3.1. NaCl–KCl–H2O System
Images of the NaCl–KCl–H
2O system seeded with NaCl are displayed in
Figure 4. The seed presents abundant fluid inclusions, noticeable as dark spots in the crystals [
6]; it underwent slight dissolution at the beginning of the experiment, evidenced by a small decrease in size in the first minutes. After 10 min, the NaCl seed crystal started to grow, developing smooth surfaces, well-formed edges, and cubic habit. Around 20 min after the evaporation started, several individual crystals appeared in the droplet. This nucleation event is probably heterogeneous, triggered by factors such as nano-impurity particles in solution, phase boundaries [
31] and local supersaturation spots. These new crystals developed hopper habits, indicated by the smooth square-ring on the border and fluid inclusions distribution, suggested by the dark spots in their center. The emergence of such crystals is unwanted, as they consume supersaturation and interfere with the epitaxial phenomena around the seeded crystal. The control of the supersaturation, although desirable, is unfeasible in ternary solutions of highly soluble salts undergoing simultaneous crystallization. Simultaneous crystallization can only happen in the eutonic point, an invariant combination of the concentration of both salts, temperature, and pressure, with no degree of freedom. Highly soluble inorganic salts have very narrow metastable zones, leaving no room to adjust supersaturation levels. Hence, the slightest amount of water evaporation induces crystallization almost immediately and maintains the solution at the eutonic concentration [
5]. For this reason, instead of controlling supersaturation, we turned our efforts to maintain the same evaporation rates in all experiments. By keeping constant evaporation rates and both compounds close to saturation levels, the residual supersaturation of each salt can be approximated as the driving force for its respective crystallization.
After circa 35 min, crystals can be seen on the surface of the crystals nucleated in solution (blue circles in
Figure 4). It is unclear, however, whether these crystals can be evidence of epitaxial growth. As these crystals arise stochastically in the solution, it is also possible that nuclei from different salts formed both spatially and temporally close to one another, such that the polycrystalline sets circled in blue are simply agglomerates. Nuclei of different composition will have different growth rates, hence there will be a delay regarding the detection of those crystals, which can mislead our interpretation into characterizing them as epitaxy generated. Unfortunately, for the system NaCl–KCl–H
2O (
Figure 4) it was not possible to identify the crystals composition directly through optical microscopy. A closer look at those crystals using SEM was also not possible, as recognition and matching between optical microscopy and SEM images were not feasible.
NaCl seed surface grew free of foreign crystals until around 30 min of experiment, when a single crystal appeared at its surface (red circle in
Figure 4). Both crystals continued to grow until the end of the experiment.
NaCl seed developed flat and smooth surfaces, usually associated with slow growth. This is surprising as hopper’s growth, commonly associated to NaCl growth [
32], was expected. The hopper habit is favored at high supersaturations [
33,
34], which is not consistent with the formation of smooth surfaces. To better understand this unexpected behavior, the growth of single NaCl crystal was observed in a NaCl-H
2O droplet solution saturated under the same conditions used in the ternary system experiments, in which the hopper habit can be promptly seen after a few minutes of evaporation (
Figure S1). Thus, at eutonic concentrations of the ternary system NaCl–KCl–H
2O during evaporation NaCl growth resulted in smoother crystal surface.
Crystal growth is commonly divided into (i) diffusion of the growth units from the bulk solution to the crystal surface and (ii) integration of these units to the crystalline surface, and is influenced by factors such as temperature, solvent, supersaturation and by the ions and molecules presents in the surface layer. For relatively high soluble compounds, such as NaCl, the diffusion is usually the limiting step, as integration of the units to the crystal surface can be considered instantaneous. However, smooth growth is known to be surface integration-controlled [
1]. Considering this scenario, a possible explanation for the development of smooth surfaces on the NaCl seed may be that the presence of the K
+ ions in the solution causes them to accumulate on the stagnant liquid layer around the growing crystal, and thus at the surface of the growing crystal. The presence of foreign ions might slow the incorporation of NaCl growth units to the NaCl-substrate crystal, forcing slower and more ordered growth, leading to smoother surfaces. It is important to highlight that, even though K
+ ions are present at the growing surface of NaCl crystals, the incorporation of these ions on the NaCl crystal lattice, also known as isomorphic substitution, is not expected under the temperature and pressure conditions in this work [
35,
36].
Optical microscopy images obtained in situ during crystallization of the ternary system NaCl–KCl–H
2O seeded with KCl are shown in
Figure 5. During the first 12 min of crystallization, a slight dissolution of the seed crystal took place. Thereafter crystal growth healed the edges of the seed crystal. At 18 min, the surface started to develop roughness which became visibly new crystals at 22 min. After 26 min, crystals start growing on the surfaces of the KCl seed. These nucleation events are possibly epitaxial, visible in the photomicrographs and confirmed through EDS analysis. Between 35 and 42 min of crystallization, the crystals that were formed in opposite sides of the seed crystal surface (marked with a red circle in
Figure 5) reached one another at the center of that surface and seemed to partially engulf the KCl seed.
SEM-EDS analysis of the NaCl seed grown in the ternary system is shown in
Figure 6A. Its outer edges are well formed, and facetted epitaxial KCl crystals developed on the substrate. Some KCl crystals are aligned with the substrate crystal, some are not. The epitaxy generated KCl deposits have not partially engulfed the NaCl substrate surface, as have NaCl deposits upon the KCl substrate (
Figure 6C). Besides, epitaxial nucleation and growth of KCl on NaCl is minor compared to epitaxy of NaCl on KCl seed. This may occur because the surface of the NaCl seed grew smooth, containing less kinks and steps that promote nucleation. Alternatively, it could be due to differences between the solubilities of NaCl and KCl. The KCl and NaCl crystals that were formed in solution away from the seed were also examined by SEM-EDS (
Figure 6B), occurring both as single crystals and mixed polycrystals. Dust-like particles on the surface of the crystals arise due to drying of the reminiscent solution layer on top of the crystal, as reported elsewhere [
5]. As KCl formation on the seed surface was not substantial, it is likely that upon evaporation the KCl supersaturation rose, so the initial burst of primary crystals in solution was probably from KCl. Yet, NaCl single particles, which are not expected, are also visible in
Figure 6B, most likely due to local supersaturation and primary heterogeneous nucleation.
The KCl-seed crystal was also analyzed using SEM-EDS (
Figure 6C). Most crystals on the surface of the KCl-seed are NaCl. The KCl core of the seed could not be detected with EDS, as the excrescences grown on top of it significantly hinder a clear signal from the engulfed crystal. There are at least two sets of crystals in clear epitaxial relation: the grown KCl seed, mostly covered by NaCl crystals, and the set on the left, diagonally oriented regarding the seed but with parallel edges of NaCl and KCl. Besides, KCl crystals developed on the surface of the epitaxially grown NaCl crystal, evidencing epitaxy of both compounds: NaCl on KCl and KCl on NaCl. These epitaxially grown KCl and NaCl crystals engulfed the KCl seed. Most often, the epitaxially grown crystals followed the crystallographic orientation of the substrate.
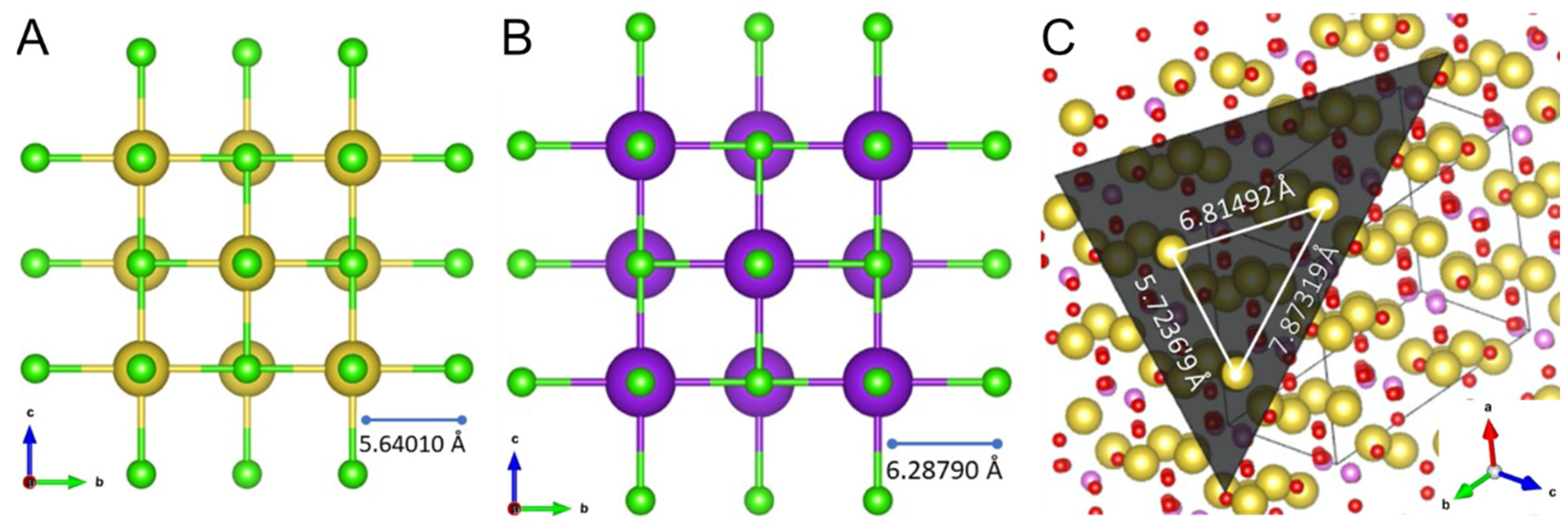
In a rough approximation, one can use the lattice constants to evaluate such mismatch (unit cell parameters are linear—1D—and epitaxial match or mismatch is 2D, thus, this comparison should not be straightforward). Both NaCl and KCl belong to the cubic crystal system, displaying only one lattice constant, respectively, a = 5.64010 Å and 6.28790 Å [
37], as shown in
Figure 7. Hence, the mismatch < 15% is compatible with epitaxial growth in this system. Even so, formation of a thin stressed growth layer of KCl over NaCl is also visible, consistent with the Stranski–Krastanov mechanism [
12,
27,
38]. Moreover, 2D lattice matching estimations showed that no characteristic match could be observed between any of the pairs of crystals used in this study. Most of the calculated non-dimensional potential energy values are approximately 1, indicating incommensurism [
21]. Detailed lattice matching calculations can be found on the
Supplementary Information.
In our previous work, we have also observed epitaxial growth in the NaCl–KCl pair [
4,
5] during 0.5 L scale batchwise seeded evaporative crystallization from boiling solutions at approximately 110 °C. However, in those experiments, epitaxial growth of KCl on NaCl seeds was more abundant than vice-versa, in disagreement with the droplet crystallization experiments. In the present study, epitaxial NaCl crystals have covered the KCl substrate surface almost completely, unlike KCl deposits upon a NaCl substrate. Besides, these excrescences were seen in much higher proportions when compared to the analogue experiment seeded with NaCl. This inversion on the ‘epitaxy preference’ follows an inversion on the solubility of the salts due to the change in temperature. The salt with the higher solubility is observed to show preferential epitaxial growth in relation to the less soluble salt in both cases (25 °C—NaCl 5.24 mol·kg
−1, KCl 1.95 mol·kg
−1; 110 °C—NaCl 4.64 mol·kg
−1, KCl 4.85 mol·kg
−1). This is probably the case, as due to higher residual supersaturation, in the presence of a foreign surface, heterogeneous nucleation on the substrate seed is more likely to occur.
It is also possible that the surface roughness of the parent crystal also plays a role in controlling epitaxial growth. A rough surface contains more kinks and steps, which are known to ease heterogeneous nucleation. Here, under simultaneous NaCl and KCl crystallization, the NaCl crystal surface is smooth, whereas the KCl surface is rough. Hence, this explains why NaCl is a poorer substrate for epitaxial growth than KCl and why seeding with KCl lead to the large extent of particle engulfment by the NaCl excrescences. Mithen and Sear [
40] also reported crystallization on crystalline surfaces to be largely influenced by the surface features, e.g., geometry and the intermolecular interactions between surface and nucleus. This also explains the higher number of crystals nucleating earlier around the seed-crystal when NaCl was seeded, in comparison to KCl-seeded experiments. As less supersaturation is getting consumed on growth/epitaxial growth on the seed, heterogeneous nucleation is induced elsewhere.
3.2. NaCl–Na2SO4–H2O System
In the NaCl–Na
2SO
4–H
2O droplet under evaporation, the NaCl seed crystal presented a slight dissolution at the first minutes of the experiment, but after 14 min of evaporation, it started to grow (
Figure 8). The edges, rounded by dissolution, healed and the typical hopper habit started to develop. The corners overgrowth of the NaCl seed crystal is easily visible at 26 min. After 40 min of evaporation, tiny crystals appeared all over the droplet (glass-droplet interface), growing to sizes up to 50 microns. Interestingly, none of the newly formed crystals were formed in the NaCl seed surface. Photomicrographs of the droplet after 61 and 63 min were taken with crossed polarizers and a gypsum plate to highlight the differences between the two compounds. Crystalline sodium sulphate may crystallize as the anhydrous salt (thenardite, orthorhombic crystal system) or the decahydrate form (Na
2SO
4·10H
2O, mirabilite, monoclinic). Both forms are birefringent and produce interference colors under crossed polarizers. Crystalline NaCl, on the other hand, belongs to the cubic system and is isotropic to light and does not produce interference colors under crossed polarizers.
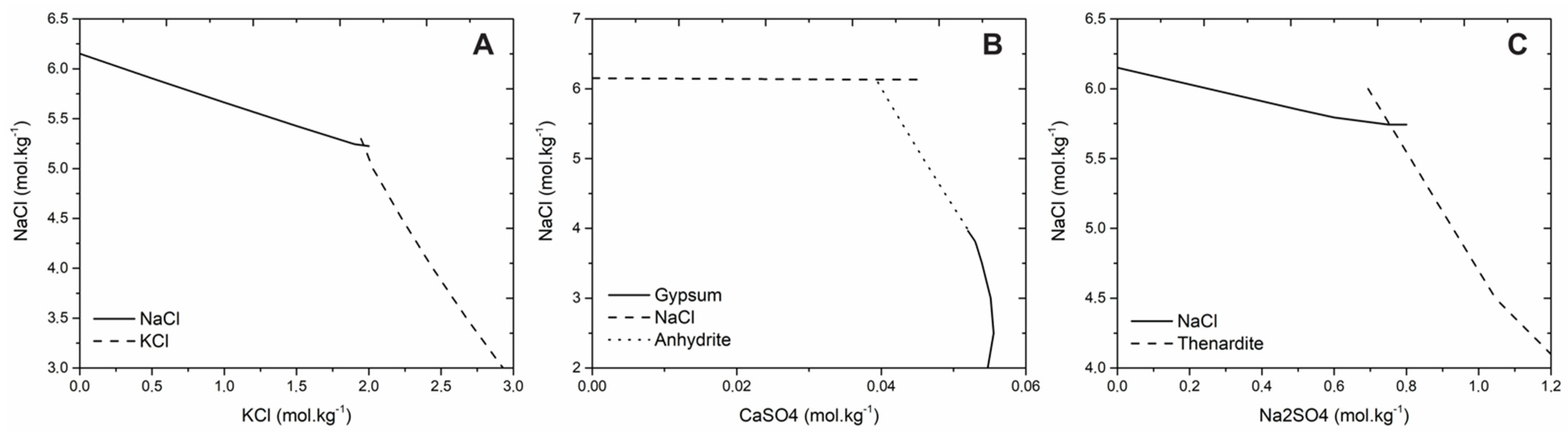
According to the phase diagram (
Figure 1), under the concentration and temperature used in the experiments, the anhydrous salt Na
2SO
4 (thenardite) is the stable form. The bipyramidal habit observed in the experiments is also characteristic of thenardite. It is noteworthy that, in a Na
2SO
4-H
2O solution, the formation of thenardite is not expected below 32.4 °C, unless relative humidity is under 71% [
41].
In situ photomicrographs of a single Na
2SO
4 seed crystal in a droplet of the NaCl–Na
2SO
4–H
2O system are presented in
Figure 9. The Na
2SO
4 seed crystal was abraded during the first 10 min, and, after that, crystal growth took place and crystal edges were reconstructed, so what appears to be a top view of a typical bipyramidal habit developed. After approximately 35 min of evaporation, two NaCl crystals nucleated on adjacent faces of the pre-existent Na
2SO
4 crystal and grew crystallographically aligned with the parent crystal whilst tiny Na
2SO
4 crystals appeared all over the droplet (highlighted by the gypsum plate at t = 58 min). Two minutes later, a new NaCl crystal appeared on the surface of a primarily nucleated Na
2SO
4 crystal, in the vicinity of the seed. Growth of NaCl crystals continued until they reached the same size of the Na
2SO
4 seed crystal, while the newly formed Na
2SO
4 crystals showed a much less growth. This is expected as Na
2SO
4 solubility at the eutonic condition is almost ten times lower than the solubility of NaCl (
Figure 1), thus the net yield of Na
2SO
4 is also almost ten times lower. Moreover, the growth of the sulphate seed was probably also hindered by the deposited NaCl crystals on its surface, which might have led to the increase of the Na
2SO
4 supersaturation and to the observed Na
2SO
4 nucleation event. It is likely that, given enough time, the epitaxial NaCl crystals would completely engulf the seeded Na
2SO
4 crystal. NaCl crystals displayed hopper habit, visible in the two ‘epitaxial’ NaCl crystals and the other crystal at 38 min (
Figure 9). Furthermore, underdeveloped crystalline faces can be observed on the NaCl deposits on the seed facing the NaCl deposit on the newly nucleated sodium sulphate, most likely due to a depletion of available ions for crystal growth.
SEM images of the experiment with NaCl seed crystals are shown in
Figure 10A. Seed surface is smooth, except for the few bipyramidal sodium sulphate crystals on its surface (
Figure 10B). These deposited crystals were not as numerous as the ones seen nucleating in solution, yet they were found in matching sizes (up to 100 µm). A hopper cavity formed during NaCl growth (
Figure 10C) is visible due to a breakage on the layer of crystalline material that formed over that cavity. This closed hopper cavity emphasizes the possibility of generation of fluid inclusions under these conditions. Fluid inclusions are not desired characteristics on products, as they lower purity when entailing a compound other than the crystal composition.
SEM image on
Figure 10D shows the Na
2SO
4 grown seed. Flat surfaces observed here are consistent with the fringes seen in polarized light, indicating a uniform variation in Na
2SO
4 crystal thickness.
Figure 10E, F focuses on the epitaxial growth of NaCl crystals on (111) plane at the Na
2SO
4 substrate. Hopper habit is clearly visible in all NaCl crystals. However, it is uncertain whether these epitaxial formations are oriented or non-oriented epitaxial crystals.
Epitaxial deposits without crystallographic orientation with respect to the seed crystal, were seen in both the NaCl–KCl–H
2O and NaCl–Na
2SO
4–H
2O systems. This is compatible with the 3D island growth mechanism proposed by Stranski–Krastanov [
12,
38], which is expected for a mismatch between the crystal lattice constants of 15% or less [
42,
43]. NaCl is seen to grown on the (111) face of Na
2SO
4 (thenardite), which belongs to the orthorhombic crystal system. Hence, the lattice mismatch can be evaluated considering the interatomic distances between the atoms on this face (
Figure 7C), as there seems to be higher coherence between NaCl (100) face and Na
2SO
4 (111) face. However, when comparing lattice parameter pairs, only one of the thenardite constants (a = 5.7236 Å) differ (slightly) from the NaCl’s constant. In their study on epitaxy-nucleated crystals over crystalline surfaces, Mithen and Sear [
24] report that oriented epitaxy relies on the coherence of the lattice parameters, i.e., crystal lattices of the nucleus and the substrate closely match. Yet, epitaxial formations were still seen in this study—although not oriented—despite poor matching between most of the lattice parameters. On the other hand, this observation agrees with Pashley’s [
18] reports for epitaxial thin film surfaces that the low misfit value, although significant under certain conditions, is not an essential criterion for epitaxial growth. Sarma et al. [
44] studied the crystallization of several pairs of organic compounds over different inorganic crystals with well-defined crystallographic planes and found little correlation between lattice matching, epitaxy and preferred nucleation orientation. They concluded that when it comes to epitaxy, multiple mechanisms take place and that intermolecular forces as well as growth anisotropy also play a role.
Similar to NaCl–KCl–H2O system, here the epitaxial growth of NaCl upon Na2SO4 is observed in large proportions than the inverse. Additionally, in the NaCl–Na2SO4–H2O system, the salt with the higher solubility showed preference for epitaxial growth in relation to the less soluble salt (25 °C—NaCl 5.74 mol·kg−1, Na2SO4 0.74 mol·kg−1). This result corroborates with previous analysis of the NaCl–KCl–H2O system regarding the higher residual supersaturation for the unseeded salt with higher solubility. It is likely that this eases the overcoming of the energy barrier for nucleation of the epitaxial crystal on the surface of the parent crystal. In the conditions studied here, some of the non-seed crystals yielded primarily nucleated single crystals in solution, instead of developing epitaxial relations with the seed. This suggests that the energy barrier for nucleation upon heterogeneous impurities in solution is at least equal to, if not lower, than the analogous barrier for nucleation upon the surface of the seed.
Furthermore, on NaCl–KCl–H2O system, surface roughness could be somewhat associated with epitaxial growth. Here, the NaCl–Na2SO4–H2O system partially follows this ‘rule’, as NaCl grows upon the Na2SO4 surfaces up to sizes comparable to the seeded crystal, whereas just tiny sparse Na2SO4 appear on the flat NaCl substrate surface along with several other idiomorphic bipyramidal Na2SO4 crystals. Na2SO4 seed surfaces are not visibly rough but given its initial abraded state, a higher roughness, when compared to the NaCl seed, is expected.
In the NaCl–Na
2SO
4–H
2O system, the NaCl hopper habit also prevails. In evaporative crystallization in a droplet, crystal growth removes ions from the solution, which are only partially replenished in the growth layer of the crystal by natural convection, so the ion availability decreases [
32,
45]. As the consumption of SO
4−2 ions is much lower than the other ions—due to the lower driving force, i.e., supersaturation, for sodium sulphate formation—the accumulation of SO
4−2 ions around the growing layer may sterically hamper NaCl growth by hindering growth units to reach the surface. Consequently, the growth process becomes partially limited by diffusion. As diffusion is more effective in the vicinity of the edges and corners of the crystal, these regions grow faster than the centers of the faces, giving rise to the hopper habit. For the NaCl–Na
2SO
4–H
2O and NaCl–KCl–H
2O systems, both crystallizing salts require a common ion (Na
+ and Cl
−, respectively). That not only causes competition for the common ion but also promotes a sharper decrease of the available ions.
3.3. NaCl–CaSO4–H2O System
Images of the eutonic solution of the NaCl–CaSO
4–H
2O system seeded with a single NaCl crystal are presented in
Figure 11. In the first 10 min of evaporation, the seed crystal did grow slightly, and its edges were healed, i.e., rounded edges became flat. From 10 min on, NaCl growing seed is seen to develop hopper habit. Up to 60 min, overgrown edges are seen to increase and reach each other, forming a cavity in the center of the face of the cubic crystal, which remained open.
Even though during evaporation the solution became supersaturated with respect to anhydrous CaSO
4, which crystallized, calcium sulphate crystals are not visible through optical microscopy. SEM-EDS image displays the NaCl seed (
Figure 12A); its face central cavities can be clearly observed, as well as a step-like-structure, comprising the inner (hollow) walls of the cavity. Two of the edges are seen broken because, due to hopper growth, edges are thin and fragile. Calcium sulphate in solution was reported to interfere with the growth of NaCl by Zago et al. [
4,
7]. In their work, evaporation was performed at 110 °C and hopper growth was also observed, as well as more prominent fluid inclusions in the product. CaSO
4 was also related to increased roughness on NaCl crystal surfaces.
Figure 12B reveals shapeless deposits of CaSO
4 on the surface of the NaCl parent crystal. Tiny CaSO
4 crystals seen are xenomorphic or anhedral, as their shape is determined by the surrounding crystals [
46]. No isolated CaSO
4 single crystals were seen. Some elongated/needle-like crystals, which can be CaSO
4, are seen partially buried on the NaCl surface, likely due to the higher NaCl growth rate compared to CaSO
4 that ends up in particle engulfment. This is not completely unexpected, as the solubility of NaCl is approximately 150 times higher than CaSO
4 solubility. Thus, the much larger amount of crystallized NaCl can easily grow a layer over tiny calcium sulphate crystals, burying them on the crystalline structure. Moreover, despite the thermodynamic predictions of CaSO
4 crystallization as anhydrite, its low kinetics [
47] plus concentration fluctuations and local supersaturation conditions—due to natural convection only—may lead to the formation of other CaSO
4 phases, such as gypsum. The formation of amorphous phases and hemihydrate (CaSO
4·0.5H
2O) as intermediates in the precipitation of gypsum has been reported elsewhere [
48,
49].
Overall, previous studies reported in the literature mention only lattice matching and recently intermolecular interactions and surface geometry as criteria for epitaxial growth [
12,
24,
40,
44]. However, in all previous studies, a single compound was nucleating and growing on top of an inert crystalline substrate. For this reason, relative solubilities between the crystallizing compounds were not a parameter. Here, it is observed that a higher proportion of epitaxial growth on the surface of the seeded crystal occurs when the most soluble compound is not seeded, e.g., NaCl on KCl, when KCl was seeded and NaCl on Na
2SO
4 when Na
2SO
4 was seeded. The solubility ratios for NaCl regarding KCl, Na
2SO
4 and CaSO
4 are, respectively, 2.7:1, 7.8:1 and 152:1. Although CaSO
4 was not seeded here, evidence of its seeds serving as nuclei for the formation of NaCl seeds were found in previous studies [
7].