Thermostructural and Elastic Properties of PbTe and Pb0.884Cd0.116Te: A Combined Low-Temperature and High-Pressure X-ray Diffraction Study of Cd-Substitution Effects
Abstract
:1. Introduction
1.1. General Issues
1.2. Cationic Substitutions
1.3. PbX-CdX (X = Te, Se, S) Solid Solution
1.4. Knowledge on Thermostructural and Elastic Properties for PbTe and Pb1−xCdxTe
1.5. Pb-Te and PbTe-CdTe System
1.6. Aim
2. Materials and Methods
3. Results: Thermostructural and Elastic Properties of PbTe and Pb0.884Cd0.116Te
3.1. Effect of Cd Substitution on Temperature Variation of Unit Cell Size, Thermal Expansion Coefficient and Cationic and Anionic Mean Square Displacements
3.1.1. General Issues
3.1.2. Variation of Unit Cell Size of PbTe and Pb0.884Cd0.116Te with Temperature
3.1.3. Variation of Thermal Expansion Coefficient with Temperature
3.1.4. Variation of Mean Square Displacements with Temperature
- (1)
- The fitted <u2>stat(T) curves for PbTe and Pb0.884Cd0.116Te behave differently. Namely:
- (a)
- The MSDs at 0 K, <u2>(T = 0), increase significantly (by about 0.002–0.004 Å2) with x rising from 0 to 0.116. We attribute this increase to the appearance of the static disorder expressed by the nonzero <u2>stat term resulting from fitting Equation (4) (the values of <u2>stat are quoted in Table 9). This effect is graphically presented in the insets of Figure 4a,b, where the variation of fitted <u2>stat values is displayed. Appearance of marginally small negative fitted value for anionic site in PbTe (instead of zero that represents the lack of disorder) is attributed to be the effect of imperfections of fitted <u2>(T) data points. The quoted values (Table 9) show that the disorder in the anionic sublattice is considerably higher than that at the cationic site. Summarizing, an increase of the static disorder term, <u2>stat, in Equation (4), from approximately zero to a value of the order of 3 × 10−3 Å2 is observed for the mixed crystal in respect to PbTe crystal. Namely, the rise is from 0.38(4) × 10−3 Å2 to 2.03(6) × 10−3 Å2 for cations, and from −0.54(7) × 10−3 Å2 (a value marginally different from zero) to 3.4(1) × 10−3 Å2 for anions.
- (b)
- At higher temperatures, the cationic MSDs are nearly equal for the two crystals, whereas the anionic ones differ markedly in the whole temperature range.
- (c)
- The slope of the cationic <u2>(T) curve decreases with rising x, whereas the anionic one apparently increases. The property of Equation (4) is that the slope of <u2>(T) is governed at high temperatures by the Debye temperature (for high slope the Debye temperature is low and vice versa; the corresponding θD values are discussed in detail in Section 3.3 and Section 4).
- (2)
- The MSDs for the cationic and anionic sites behave differently for x = 0 than for x = 0.116.
- (3)
- Comparison of Figure 4a,b shows that the cationic and anionic MSDs of Pb0.884Cd0.116Te are of comparable values in a broad temperature range. As this effect must depend on x, we expect that for x < 0.116, the <u2> values of anions are lower than those of cations, whereas for x > 0.116 (if the structure is stabilized), the anionic ones are higher.
3.2. Effect of Substitution of Cd in the PbTe Lattice on Variation of Unit-Cell Size and of Bulk Modulus with Pressure
3.3. Effect of Cd Substitution on Values of Debye Temperature
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| APW | augmented plane-wave |
| BMEOS | Birch-Murnaghan Equation of State |
| CDM | crystal dynamics models |
| CM | calorimetry |
| DFPT | density functional perturbation theory |
| DFT | density functional theory |
| DM | dilatometry |
| DPS | double parton scattering (nuclear inelastic scattering) |
| EC | elastic constants |
| FP | full potential |
| FPBTF | first principles Boltzmann transport framework |
| GGA | generalized gradient approximation |
| GULP | computer program for the symmetry adapted simulation of solids, authored by Julian D. Gale |
| HPM | heat-pulse method |
| HS | hydrostatic conditions |
| HSEsolSOC | revised Heyd-Scuseria-Ernzerhof functional + spin-orbit coupling |
| LAPW | linearized augmented plane-wave |
| LDA | local density approximation |
| LDY | lattice dynamics calculations |
| LEDPXRD | laboratory energy-dispersive X-ray diffraction |
| LKF | Lin-Kleinman formalism |
| LSCXRD | laboratory single-crystal X-ray diffraction |
| MD | molecular dynamics |
| MSD | mean square displacement |
| n.a. | not available |
| ND | neutron diffraction |
| NNI | nearest-neighbor interaction model by Kagan and Maslow |
| NPD | neutron powder diffraction |
| NS | neutron scattering |
| PAW | projector augmented wave method |
| PBE | Perdew-Bucke-Ernzerhof exchange-correlation functional |
| PBEsol | Perdew–Bucke–Ernzerhof exchange-correlation functional revised for solids |
| PM | the paramagnetic resonance. θD estimated by the temperature-dependent hyperfine splitting constant A(T) |
| PTW | plane temperature waves method |
| QHA | quasiharmonic approximation |
| QHS | quasi-hydrostatic conditions |
| RT | room temperature |
| SCXRD | single crystal X-ray diffraction |
| SCXRDS | single crystal X-ray difraction at synchrotron |
| SO | soft-constraint based online |
| SME | slave mode expansion |
| SP | spectroscopy |
| SPXRD | synchrotron powder X-ray diffraction |
| SV | sound velocity method |
| TC | thermal conductivity |
| TEC | thermal expansion coefficient |
| THD | thermodynamic calculations |
| UPE | ultrasonic pulse-echo method |
| UIM | ultrasonic interferometry |
| UWV | ultrasonic wave velocity |
| UWVSC | ultrasonic wave velocity in single crystal |
| XRD/ND/PDF | X-ray diffraction and neutron diffraction, analyzed with pair distribution function (PDF) method |
Appendix A
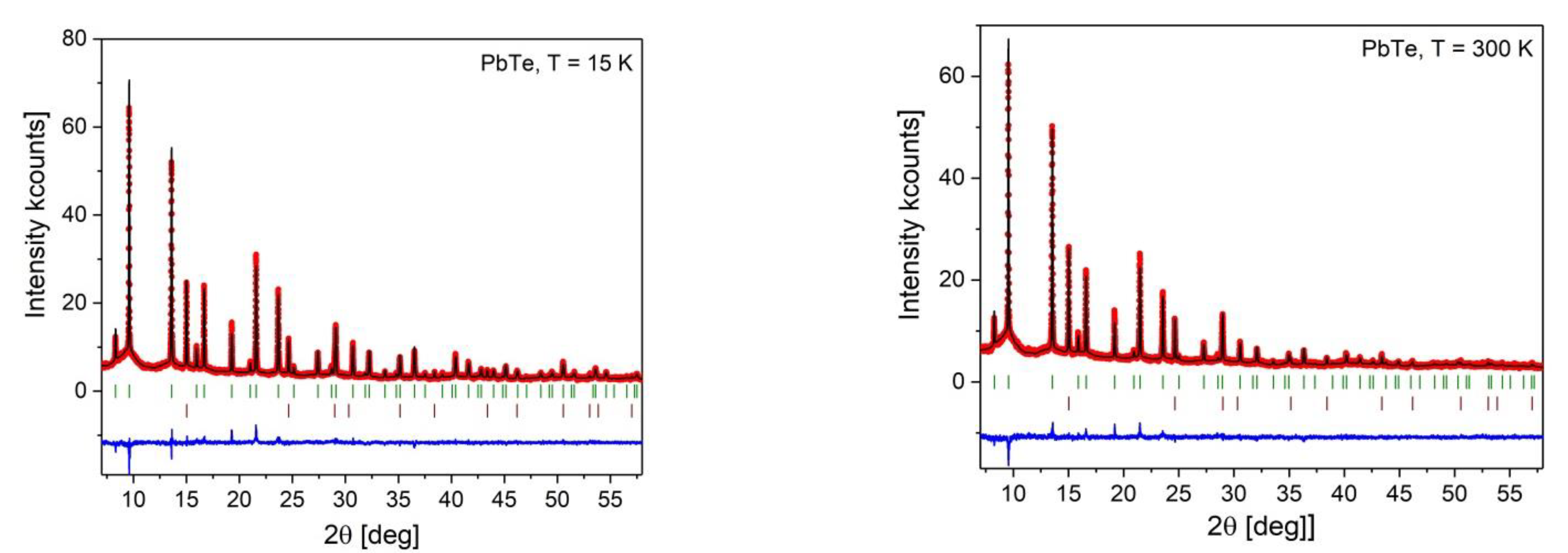
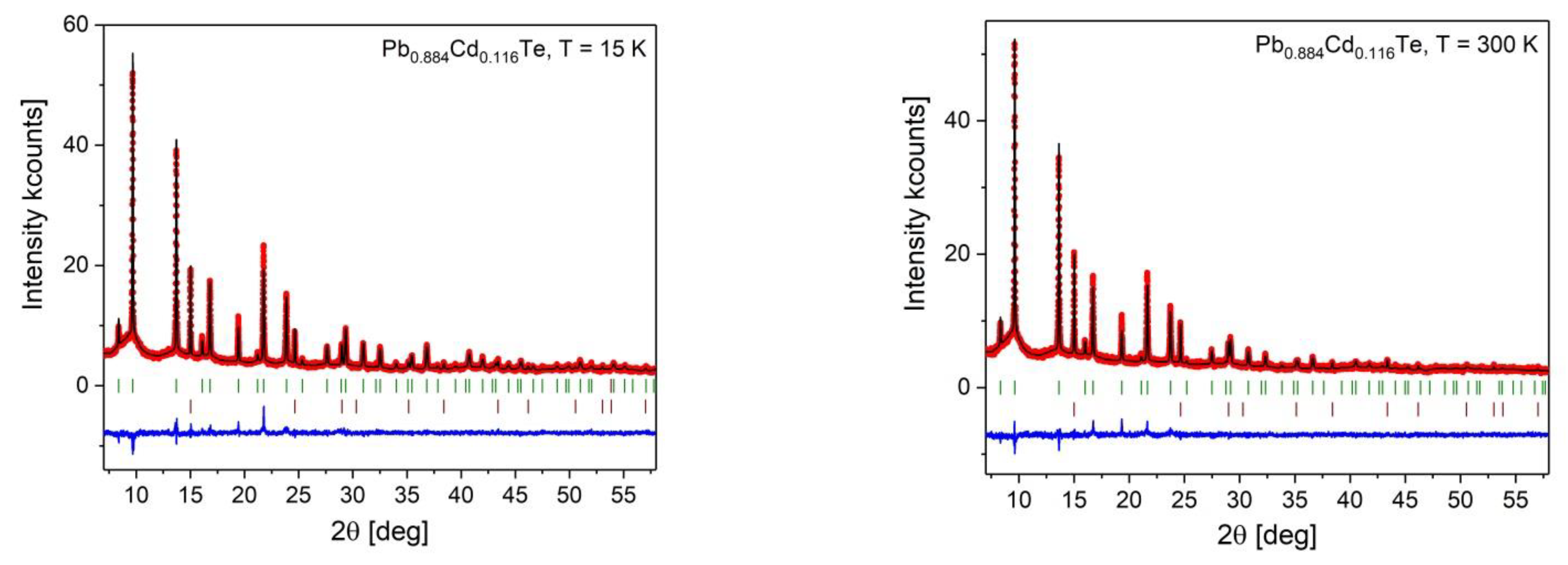
Appendix B
| T [K] | PbTe | Pb0.884Cd0.116Te | ||||
|---|---|---|---|---|---|---|
| a [Å] | <uC2> [Å2] | <uA2> [Å2] | a [Å] | <uC2> [Å2] | <uA2> [Å2] | |
| 15 | 6.4298(4) | 0.0020(1) | 0.0016(2) | 6.3775(5) | 0.0041(2) | 0.0057(3) |
| 20 | 6.4299(3) | 0.0027(1) | 0.0018(2) | 6.3773(4) | 0.0041(2) | 0.0055(3) |
| 25 | 6.4301(3) | 0.0024(1) | 0.0018(2) | 6.3776(5) | 0.0047(2) | 0.0055(3) |
| 30 | 6.4301(4) | 0.0028(1) | 0.0016(2) | 6.3777(5) | 0.0046(2) | 0.0052(3) |
| 35 | 6.4305(4) | 0.0030(1) | 0.0018(2) | 6.3783(5) | 0.0047(2) | 0.0055(3) |
| 40 | 6.4310(4) | 0.0037(1) | 0.0010(2) | 6.3787(7) | 0.0046(2) | 0.0059(3) |
| 45 | 6.4312(3) | 0.0036(1) | 0.0019(2) | 6.3792(6) | 0.0053(2) | 0.0058(3) |
| 50 | 6.4316(3) | 0.0038(1) | 0.0021(2) | 6.3793(6) | 0.0054(2) | 0.0060(3) |
| 55 | 6.4323(3) | 0.0044(1) | 0.0027(2) | 6.3800(7) | 0.0053(2) | 0.0066(3) |
| 60 | 6.4327(3) | 0.0047(1) | 0.0021(2) | 6.3805(5) | 0.0047(2) | 0.0067(3) |
| 65 | 6.4333(3) | 0.0052(1) | 0.0024(2) | 6.3810(7) | 0.0054(2) | 0.0064(3) |
| 70 | 6.4338(3) | 0.0059(1) | 0.0026(2) | 6.3817(6) | 0.0056(2) | 0.0071(3) |
| 75 | 6.4342(5) | 0.0057(2) | 0.0029(2) | 6.3823(5) | 0.0072(2) | 0.0072(3) |
| 80 | 6.4347(5) | 0.0058(2) | 0.0025(2) | 6.3829(6) | 0.0071(2) | 0.0074(3) |
| 85 | 6.4350(3) | 0.0062(1) | 0.0034(2) | 6.3833(5) | 0.0069(2) | 0.0084(3) |
| 90 | 6.4358(4) | 0.0064(2) | 0.0034(2) | 6.3840(4) | 0.0078(2) | 0.0077(3) |
| 95 | 6.4364(5) | 0.0072(2) | 0.0035(3) | 6.3848(4) | 0.0075(2) | 0.0087(3) |
| 100 | 6.4371(4) | 0.0074(2) | 0.0026(3) | 6.3855(4) | 0.0079(2) | 0.0089(3) |
| 110 | 6.4381(3) | 0.0083(2) | 0.0034(2) | 6.3863(5) | 0.0085(2) | 0.0079(4) |
| 120 | 6.4394(3) | 0.0092(2) | 0.0043(3) | 6.3875(4) | 0.0094(2) | 0.0092(4) |
| 130 | 6.4408(4) | 0.0095(2) | 0.0044(3) | 6.3891(4) | 0.0095(2) | 0.0094(3) |
| 140 | 6.4418(4) | 0.0100(2) | 0.0060(3) | 6.3903(4) | 0.0099(2) | 0.0097(4) |
| 150 | 6.4429(3) | 0.0111(2) | 0.0057(3) | 6.3915(4) | 0.0102(2) | 0.0104(4) |
| 160 | 6.4440(3) | 0.0117(2) | 0.0059(3) | 6.3931(5) | 0.0115(2) | 0.0115(4) |
| 170 | 6.4451(3) | 0.0120(2) | 0.0069(3) | 6.3941(5) | 0.0108(2) | 0.0129(4) |
| 180 | 6.4466(3) | 0.0118(2) | 0.0064(3) | 6.3953(5) | 0.0117(2) | 0.0103(4) |
| 190 | 6.4477(3) | 0.0133(2) | 0.0073(3) | 6.3969(5) | 0.0121(2) | 0.0129(4) |
| 200 | 6.4491(4) | 0.0144(2) | 0.0072(3) | 6.3981(5) | 0.0127(2) | 0.0132(4) |
| 210 | 6.4503(3) | 0.0147(2) | 0.0070(3) | 6.3997(4) | 0.0132(2) | 0.0131(4) |
| 220 | 6.4514(4) | 0.0142(2) | 0.0097(3) | 6.4007(5) | 0.0137(3) | 0.0138(4) |
| 230 | 6.4527(3) | 0.0167(2) | 0.0085(3) | 6.4020(4) | 0.0156(3) | 0.0137(4) |
| 240 | 6.4539(3) | 0.0170(3) | 0.0088(3) | 6.4035(3) | 0.0170(3) | 0.0135(4) |
| 250 | 6.4552(3) | 0.0166(2) | 0.0096(3) | 6.4045(3) | 0.0173(3) | 0.0153(4) |
| 260 | 6.4564(3) | 0.0171(2) | 0.0094(3) | 6.4062(3) | 0.0160(3) | 0.0148(4) |
| 270 | 6.4578(3) | 0.0180(3) | 0.0109(4) | 6.4071(3) | 0.0192(3) | 0.0160(5) |
| 280 | 6.4588(3) | 0.0192(3) | 0.0111(4) | 6.4086(4) | 0.0171(3) | 0.0165(5) |
| 290 | 6.4602(3) | 0.0178(3) | 0.0123(4) | 6.4100(4) | 0.0188(3) | 0.0176(5) |
| 300 | 6.4616(3) | 0.0195(3) | 0.0124(4) | 6.4116(4) | 0.0200(3) | 0.0166(5) |
| T [K] | PbTe | Pb0.884Cd0.116Te | ||||||
|---|---|---|---|---|---|---|---|---|
| a [Å] | α [MK−1] | <uC2> [Å2] | <uA2> [Å2] | a [Å] | α [MK−1] | <uC2> [Å2] | <uA2> [Å2] | |
| 0 | 6.42972(5) | 0 | 0.0021(1) | 0.0011(1) | 6.37725(6) | 0 | 0.0036(1) | 0.0052(1) |
| 10 | 6.42973(5) | 0.70(7) | 0.0022(1) | 0.0012(1) | 6.37726(6) | 0.9(1) | 0.0037(1) | 0.0053(1) |
| 20 | 6.42986(6) | 4.2(3) | 0.0025(1) | 0.0013(1) | 6.37742(7) | 5.0(4) | 0.0040(1) | 0.0054(1) |
| 30 | 6.43026(9) | 8.5(4) | 0.0030(1) | 0.0015(1) | 6.37788(11) | 9.8(6) | 0.0043(1) | 0.0056(1) |
| 40 | 6.43091(12) | 11.7(4) | 0.0035(1) | 0.0017(1) | 6.37861(15) | 13.2(6) | 0.0048(1) | 0.0059(1) |
| 50 | 6.43173(15) | 13.9(4) | 0.0041(1) | 0.0020(1) | 6.37952(18) | 15.4(6) | 0.0052(1) | 0.0062(1) |
| 60 | 6.43266(17) | 15.3(4) | 0.0047(1) | 0.0023(1) | 6.38054(22) | 16.9(5) | 0.0057(1) | 0.0066(1) |
| 70 | 6.43367(20) | 16.2(4) | 0.0053(1) | 0.0027(1) | 6.38165(25) | 17.8(5) | 0.0062(1) | 0.0070(2) |
| 80 | 6.43474(22) | 16.9(4) | 0.0059(1) | 0.0030(1) | 6.38281(28) | 18.5(5) | 0.0068(1) | 0.0074(2) |
| 90 | 6.43584(24) | 17.4(4) | 0.0066(1) | 0.0033(1) | 6.38400(31) | 19.0(5) | 0.0072(2) | 0.0079(2) |
| 100 | 6.43697(27) | 17.8(4) | 0.0072(1) | 0.0037(1) | 6.38523(35) | 19.3(5) | 0.0078(2) | 0.0083(2) |
| 110 | 6.43812(29) | 18.0(4) | 0.0079(1) | 0.0041(1) | 6.38647(38) | 19.6(5) | 0.0084(2) | 0.0087(2) |
| 120 | 6.43929(31) | 18.3(4) | 0.0085(1) | 0.0045(1) | 6.38773(41) | 19.8(5) | 0.0089(2) | 0.0091(2) |
| 130 | 6.44047(34) | 18.5(4) | 0.0092(1) | 0.0049(1) | 6.38900(44) | 20.0(5) | 0.0095(2) | 0.0096(2) |
| 140 | 6.44167(36) | 18.6(4) | 0.0098(1) | 0.0052(2) | 6.39028(47) | 20.1(5) | 0.0100(2) | 0.0100(2) |
| 150 | 6.44287(39) | 18.7(4) | 0.0105(1) | 0.0056(2) | 6.39157(51) | 20.2(5) | 0.0106(2) | 0.0105(2) |
| 160 | 6.44408(41) | 18.8(4) | 0.0111(1) | 0.0060(2) | 6.39287(54) | 20.3(6) | 0.0111(2) | 0.0109(2) |
| 170 | 6.44529(44) | 18.9(4) | 0.0118(1) | 0.0064(2) | 6.39417(58) | 20.4(6) | 0.0117(2) | 0.0113(2) |
| 180 | 6.44652(47) | 19.0(4) | 0.0125(1) | 0.0068(2) | 6.39547(62) | 20.4(6) | 0.0122(3) | 0.0118(2) |
| 190 | 6.44774(50) | 19.1(4) | 0.0131(1) | 0.0072(2) | 6.39678(66) | 20.5(6) | 0.0128(3) | 0.0122(2) |
| 200 | 6.44898(53) | 19.1(5) | 0.0138(1) | 0.0076(2) | 6.39809(70) | 20.5(6) | 0.0133(3) | 0.0127(3) |
| 210 | 6.45021(56) | 19.2(5) | 0.0144(1) | 0.0080(2) | 6.39941(74) | 20.6(6) | 0.0139(3) | 0.0131(3) |
| 220 | 6.45145(59) | 19.3(5) | 0.0151(1) | 0.0084(2) | 6.40073(78) | 20.6(7) | 0.0144(3) | 0.0136(3) |
| 230 | 6.45270(62) | 19.3(5) | 0.0158(1) | 0.0088(2) | 6.40205(82) | 20.6(7) | 0.0150(3) | 0.0140(3) |
| 240 | 6.45394(65) | 19.3(5) | 0.0164(1) | 0.0091(2) | 6.40337(87) | 20.7(7) | 0.0156(3) | 0.0145(3) |
| 250 | 6.45519(69) | 19.4(5) | 0.0171(1) | 0.0095(2) | 6.40469(91) | 20.7(7) | 0.0161(3) | 0.0149(3) |
| 260 | 6.45644(72) | 19.4(5) | 0.0177(1) | 0.0099(2) | 6.40602(96) | 20.7(7) | 0.0167(3) | 0.0154(3) |
| 270 | 6.45770(76) | 19.5(6) | 0.0184(1) | 0.0103(2) | 6.40734(100) | 20.7(8) | 0.0172(4) | 0.0158(3) |
| 280 | 6.45896(79) | 19.5(6) | 0.0191(1) | 0.0107(2) | 6.40867(105) | 20.7(8) | 0.0178(4) | 0.0163(3) |
| 290 | 6.46022(83) | 19.5(6) | 0.0197(2) | 0.0111(2) | 6.41000(110) | 20.7(8) | 0.0183(4) | 0.0167(3) |
| 300 | 6.46148(87) | 19.6(6) | 0.0204(2) | 0.0115(2) | 6.41133(116) | 20.7(8) | 0.0189(4) | 0.0172(3) |
| PbTe | Pb0.884Cd0.116Te | ||
|---|---|---|---|
| p [GPa] | V [Å3] | p [GPa] | V [Å3] |
| 0.33(2) | 271.1(15) | 0.30(2) | 265.40(50) |
| 0.80(2) | 267.9(12) | 0.80(2) | 261.10(80) |
| 1.30(2) | 266.8(15) | 1.40(2) | 256.50(16) |
| 1.80(2) | 264.8(16) | 2.50(2) | 251.78(10) |
| 2.40(2) | 259.5(16) | 3.00(2) | 247.41(12) |
| 3.00(2) | 257.2(15) | 3.50(2) | 245.37(11) |
| 3.70(2) | 254.7(12) | 4.00(2) | 241.37(14) |
| 4.50(2) | 251.8(15) | 4.50(2) | 241.23(14) |
| p [GPa] | PbTe | Pb0.884Cd0.116Te | ||
|---|---|---|---|---|
| V [Å3] | K [GPa] | V [Å3] | K [GPa] | |
| 0 | 273.25 | 45.6(2.5) | 273.25 | 33.5(2.8) |
| 0.5 | 270.33 | 47.6(2.5) | 270.33 | 35.5(2.8) |
| 1.0 | 267.57 | 49.6(2.5) | 267.57 | 37.5(2.8) |
| 1.5 | 264.93 | 51.5(2.5) | 264.93 | 39.4(2.8) |
| 2.0 | 262.42 | 53.5(2.5) | 262.42 | 41.3(2.8) |
| 2.5 | 260.02 | 55.4(2.5) | 260.02 | 43.2(2.8) |
| 3.0 | 257.72 | 57.3(2.5) | 257.72 | 45.1(2.8) |
| 3.5 | 255.52 | 59.1(2.5) | 255.52 | 47.0(2.8) |
| 4.0 | 253.41 | 61.1(2.5) | 253.41 | 48.8(2.8) |
| 4.5 | 251.37 | 62.9(2.5) | 251.37 | 50.7(2.8) |
| 5.0 | 249.41 | 64.8(2.5) | 249.41 | 52.5(2.8) |
Appendix C
| Function | Fitted Equation | Parameters for PbTe | Parameters for Pb0.884Cd0.116Te |
|---|---|---|---|
| V(T) | Equation (1) | = 265.813(6) Å3, Q = 2.86(3) × 10−18, b = 1.3(6), θD = 135.2(3.9) K | = 259.358(7) Å3, Q = 2.63(4) × 10−18, b = 0.4(7), θD = 130.1(4.4) K |
| <u2>(T), for cationic site | Equation (4) | θD = 102.8(3) K, <u2>stat = 0.00038(4) Å2 | θD = 114.5(5) K, <u2>stat = 0.00203(6) Å2 |
| <u2>(T), for anionic site | Equation (4) | θD = 169.2(1.1) K, <u2>stat = −0.00054(7) Å2 | θD = 158.1(1.3) K, <u2>stat = 0.0034(1) Å2 |
| V(p) | Equation (6) | V0 = 273.3(7) Å3 K0 = 45.6(2.5) GPa K′ = 4 (fixed) | V0 = 267.7(1.5) Å3 K0 = 33.5(2.8) GPa K′ = 4 (fixed) |
Appendix D
| Compound | K0 [GPa] | K′ | Method | Ref. | Year |
|---|---|---|---|---|---|
| PbTe | 45 | n.a. | LDA | ||
| 48 | n.a. | LDA | (a) | 1983 | |
| 51.7 | 4.52 | LAPW LDA | (b) | 1997 | |
| 51.44 (0 K) 40.30 (0 K) 49.82 (0 K) 39.5 (0 K) | 5.50 4.27 5.76 3.92 | FP-LAPW LDA FP-LAPW GGA FP-LAPW LDA+SO FP-LAPW GGA+SO | (c) | 2000 | |
| 41.4 51.4 | 3.352 4.080 | GGA LDA | (d) | 2002 | |
| 37.5 (0 K) | n.a. | FP-APW PBE | (e) | 2007 | |
| 40.4 (0 K) 50.3 (0 K) 30.7 | n.a. n.a. n.a. | LDA/GGA “ “ | (f) | 2009 | |
| 39.05 | 4.32 | FP-LAPW | (g) | 2011 | |
| 46.0 46.1 | 4.27 4.53 | LDA LDA+SO | (h) | 2012 | |
| 41.0 | n.a. | MD (GULP) | (i) | 2012 | |
| 39.1 | n.a. | PAW PBE | (j) | 2013 | |
| 47 (0 K) | n.a. | FP-LAPW | (k) | 2014 | |
| 38.54 (300 K) ~45.5 (0 K) | n.a. n.a. | PBEsol “ | (l) | 2014 | |
| 34.04 (0 K) | n.a. | LDA, GGA | (m) | 2014 | |
| 46.61 (0 K) | n.a. | PBEsol | (n) | 2015 | |
| 44.1(&) | n.a. | HSEsolSOC | (o) | 2016 | |
| 41.1 | n.a. | FP-LAPW | (p) | 2017 | |
| 36.19 (100 K) 37.52 (300 K) | n.a. n.a. | LDY “ | (q) | 2019 | |
| 48.242 (0 K) | 5.576 (0 K) | LDA | (r) | 2020 | |
| 43.6 | 4.6 | GGA-PBE | (s) | 2020 | |
| Pb0.969Cd0.031Te | 46.42 | n.a. | GGA | (n) | 2015 |
| Pb0.884Cd0.116Te | 45.90 | n.a. | GGA | ($) | 2021 |
References
- Faber, K.T.; Malloy, K.J. (Eds.) The Mechanical Properties of Semiconductors; Academic Press: Boston, MA, USA, 1992; Volume 37. [Google Scholar]
- Zhang, X.; Zhao, L.D. Thermoelectric materials: Energy conversion between heat and electricity. J. Mater. 2015, 1, 92–105. [Google Scholar] [CrossRef] [Green Version]
- Heremans, J.P.; Jovovic, V.; Toberer, E.S.; Saramat, A.; Kurosaki, K.; Charoenphakdee, A.; Yamanaka, S.; Snyder, G.J. Enhancement of thermoelectric efficiency in PbTe by distortion of the electronic density of states. Science 2008, 321, 554–557. [Google Scholar] [CrossRef] [Green Version]
- Jaworski, C.M.; Wiendlocha, B.; Jovovic, V.; Heremans, J.P. Combining alloy scattering of phonons and resonant electronic levels to reach a high thermoelectric figure of merit in PbTeSe and PbTeS alloys. Energy Environ. Sci. 2011, 4, 4155–4162. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, X.; Cui, T.; Niu, Y.; Wang, Y.; Wang, M.; Ma, Y.; Zou, G. Enhanced thermoelectric performance of PbTe within the orthorhombic Pnma phase. Phys. Rev. B 2007, 76, 155127. [Google Scholar] [CrossRef]
- Dynowska, E.; Szuszkiewicz, W.; Szczepanska, A.; Romanowski, P.; Dobrowolski, W.; Lathe, C.; Slynko, E.V. High-Pressure Studies of Bulk Pb1−xBaxTe Mixed Crystals. In Jahresbericht/Hamburger Synchrotronstrahlungslabor HASYLAB am Deutschen Elektronen-Synchrotron DESY= Annual Report; HASYLAB: Hamburg, Germany, 2005; pp. 361–362. [Google Scholar]
- Bukała, M.; Sankowski, P.; Buczko, R.; Kacman, P. Structural and electronic properties of Pb1−xCdxTe and Pb1−xMnxTe ternary alloys. Phys. Rev. B 2012, 86, 085205. [Google Scholar] [CrossRef] [Green Version]
- Joseph, E.; Amouyal, Y. Enhancing thermoelectric performance of PbTe-based compounds by substituting elements: A first principles study. J. Electron. Mater. 2015, 44, 1460–1468. [Google Scholar] [CrossRef]
- Joseph, E.; Amouyal, Y. Towards a predictive route for selection of doping elements for the thermoelectric compound PbTe from first-principles. J. Appl. Phys. 2015, 117, 175102. [Google Scholar] [CrossRef]
- Śpiewak, P.; Kurzydłowski, K.J. Electronic Structure and Transport Properties of Doped Lead Chalcogenides from First Principles. MRS Adv. 2016, 1, 4003–4010. [Google Scholar] [CrossRef]
- Sealy, B.J.; Crocker, A.J. A comparison of phase equilibria in some II-IV-VI compounds based on PbTe. J. Mater. Sci. 1973, 8, 1731–1736. [Google Scholar] [CrossRef]
- Crocker, A.J.; Rogers, L.M. Valence band structure of PbTe. J. Phys. Colloq. 1968, 29, C4-129–C4-132. [Google Scholar] [CrossRef]
- Pei, Y.; LaLonde, A.D.; Heinz, N.A.; Snyder, G.J. High thermoelectric figure of merit in PbTe alloys demonstrated in PbTe–CdTe. Adv. Energy Mater. 2012, 2, 670–675. [Google Scholar] [CrossRef]
- Ahn, K.; Biswas, K.; He, J.; Chung, I.; Dravid, V.; Kanatzidis, M.G. Enhanced thermoelectric properties of p-type nanostructured PbTe–MTe (M = Cd, Hg) materials. Energy Environ. Sci. 2013, 6, 1529–1537. [Google Scholar] [CrossRef]
- Sarkar, S.; Zhang, X.; Hao, S.; Hua, X.; Bailey, T.P.; Uher, C.; Wolverton, C.; Dravid, V.P.; Kanatzidis, M.G. Dual alloying strategy to achieve a high thermoelectric figure of merit and lattice hardening in p-type nanostructured PbTe. ACS Energy Lett. 2018, 3, 2593–2601. [Google Scholar] [CrossRef]
- Rosenberg, A.J.; Woolley, J.C.; Nikolic, P.; Grierson, R. Solid solutions of CdTe and InTe in PbTe and SnTe. I. Crystal chemistry. Trans. Metall. Soc. AIME 1964, 230, 342–349. [Google Scholar]
- Kulvitit, Y.; Rolland, S.; Granger, R.; Pelletier, C.M. Relation entre composition paramètre de maille et bande interdite des composés Pb1−xCdxTe. Rev. Phys. Appliquée 1980, 15, 1501–1504. [Google Scholar] [CrossRef]
- Marx, R.; Range, K.J. Homogene, abschreckbare Hochdruckphasen mit NaCl-Struktur in den systemen CdTe-SnTe und CdTe-PbTe. J. Less Common Met. 1989, 155, 49–59. [Google Scholar] [CrossRef]
- Szot, M.; Szczerbakow, A.; Dybko, K.; Kowalczyk, L.; Smajek, E.; Domukhovski, V.; Łusakowska, E.; Dziawa, P.; Mycielski, A.; Story, T.; et al. Experimental and Theoretical Analysis of PbTe–CdTe Solid Solution Grown by Physical Vapour Transport Method. Acta Phys. Polon. A 2009, 116, 959–961. [Google Scholar] [CrossRef]
- Wald, F.; Rosenberg, A.J. Solid solutions of CdSe and InSe in PbSe. J. Phys. Chem. Solids 1965, 26, 1087–1091. [Google Scholar] [CrossRef]
- Crocker, A.J. Phase equilibria in PbTe/CdTe alloys. J. Mater. Sci. 1968, 3, 534–539. [Google Scholar] [CrossRef]
- Sealy, B.J.; Crocker, A.J. Some physical properties of the systems Pb1−xMgxSe and Pb1−xCdxSe. J. Mater. Sci. 1973, 8, 1247–1252. [Google Scholar] [CrossRef]
- Qian, X.; Wu, H.; Wang, D.; Zhang, Y.; Wang, J.; Wang, G.; Wang, G.; Zheng, L.; Pennycook, S.J.; Zhao, L.D. Synergistically optimizing interdependent thermoelectric parameters of n-type PbSe through alloying CdSe. Energy Environ. Sci. 2019, 12, 1969–1978. [Google Scholar] [CrossRef]
- Bethke, P.M.; Barton, P.B., Jr. Sub-solidus relations in the system PbS-CdS. Am. Mineral. J. Earth Planet. Mater. 1971, 56, 2034–2039. [Google Scholar]
- Tan, G.L.; Liu, L.; Wu, W. Mid-IR band gap engineering of CdxPb1−xS nanocrystals by mechanochemical reaction. AIP Adv. 2014, 4, 067107. [Google Scholar] [CrossRef] [Green Version]
- ICSD Database (FIZ, Karlsruhe), Records 620316, 620315, 620322. Available online: https://icsd.products.fiz-karlsruhe.de/ (accessed on 21 August 2021).
- Bziz, I.; Atmani, E.H.; Fazouan, N.; Aazi, M.; Es-Smairi, A. Ab-initio study of structural, electronic and optical properties of CdS. In Proceedings of the 2019 7th International Renewable and Sustainable Energy Conference (IRSEC), Agadir, Morocco, 27–30 November 2019; 2019; pp. 1–6. [Google Scholar] [CrossRef]
- Kiyosawa, T.; Takahashi, S.; Koguchi, N. Solid solubility range and lattice parameter of semiconducting Pb1−xCdxS1−ySey for mid-infra-red lasers. Phys. Status Solidi A 1989, 111, K21–K25. [Google Scholar] [CrossRef]
- Knight, K.S. Does Altaite Exhibit Emphanitic Behavior? a High Resolution Neutron Powder Diffraction Investigation of the Crystallographic and Thermoelastic Properties of PbTe Between 10 and 500 K. Does Altaite Exhibit Emphanitic Behaviour? Can. Mineral. 2016, 54, 1493–1503. [Google Scholar] [CrossRef]
- Liu, Y.; Ibáñez, M. Tidying up the mess. Science 2021, 371, 678–679. [Google Scholar] [CrossRef] [PubMed]
- Kastbjerg, S.; Bindzus, N.; Søndergaard, M.; Johnsen, S.; Lock, N.; Christensen, M.; Takata, M.; Spackman Brummerstedt, M.A.; Iversen, B. Direct evidence of cation disorder in thermoelectric lead chalcogenides PbTe and PbS. Adv. Funct. Mater. 2013, 23, 5477–5483. [Google Scholar] [CrossRef]
- Wang, H.; LaLonde, A.D.; Pei, Y.; Snyder, G.J. The criteria for beneficial disorder in thermoelectric solid solutions. Adv. Funct. Mater. 2013, 23, 1586–1596. [Google Scholar] [CrossRef]
- Knight, K.S. A high-resolution neutron powder diffraction investigation of galena (PbS) between 10 K and 350 K: No evidence for anomalies in the lattice parameters or atomic displacement parameters in galena or altaite (PbTe) at temperatures corresponding to the saturation of cation disorder. J. Phys. Condens. Matter 2014, 26, 385403. [Google Scholar] [CrossRef]
- Christensen, S.; Bindzus, N.; Sist, M.; Takata, M.; Iversen, B.B. Structural disorder, anisotropic micro-strain and cation vacancies in thermo-electric lead chalcogenides. Phys. Chem. Chem. Phys. 2016, 18, 15874–15883. [Google Scholar] [CrossRef]
- Szot, M.; Pfeffer, P.; Dybko, K.; Szczerbakow, A.; Kowalczyk, L.; Dziawa, P.; Minikayev, R.; Zajarniuk, T.; Piotrowski, P.; Gutowska, M.U.; et al. Two-valence band electron and heat transport in monocrystalline PbTe-CdTe solid solutions with Cd content up to 10 atomic percent. Phys. Rev. Mater. 2020, 4, 044605. [Google Scholar] [CrossRef]
- Tsang, Y.W.; Cohen, M.L. Calculation of the temperature dependence of the energy gaps in PbTe and SnTe. Phys. Rev. B 1971, 3, 1254. [Google Scholar] [CrossRef]
- Noda, Y.; Masumoto, K.; Ohba, S.; Saito, Y.; Toriumi, K.; Iwata, Y.; Shibuya, I. Temperature dependence of atomic thermal parameters of lead chalcogenides, PbS, PbSe and PbTe. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 1987, 43, 1443–1445. [Google Scholar] [CrossRef]
- Božin, E.S.; Malliakas, C.D.; Souvatzis, P.; Proffen, T.; Spaldin, N.A.; Kanatzidis, M.G.; Billinge, S.J. Entropically stabilized local dipole formation in lead chalcogenides. Science 2010, 330, 1660–1663. [Google Scholar] [CrossRef] [PubMed]
- Sangiorgio, B.; Bozin, E.S.; Malliakas, C.D.; Fechner, M.; Simonov, A.; Kanatzidis, M.G.; Billinge, S.J.L.; Spaldin, N.A.; Weber, T. Correlated local dipoles in PbTe. Phys. Rev. Mater. 2018, 2, 085402. [Google Scholar] [CrossRef]
- Holm, K.A.; Roth, N.; Zeuthen, C.M.; Iversen, B.B. Anharmonicity and correlated dynamics of PbTe and PbS studied by single crystal x-ray scattering. Phys. Rev. B 2021, 103, 224302. [Google Scholar] [CrossRef]
- Male, J.P.; Hanus, R.; Snyder, G.J.; Hermann, R.P. Thermal Evolution of Internal Strain in Doped PbTe. Chem. Mater. 2021, 33, 4765–4772. [Google Scholar] [CrossRef]
- Minikayev, E.D.; Dziawa, P.; Kamińska, E.; Szczerbakow, A.; Trots, D.; Szuszkiewicz, W. High-temperature studies of Pb1−xCdxTe solid solution: Structure stability and CdTe solubility limit, Synchrotron Radiation. Nat. Sci. 2009, 8, 8. [Google Scholar]
- Minikayev, R.; Dynowska, E.; Kaminska, E.; Szczerbakow, A.; Trots, D.; Story, T.; Szuszkiewicz, W. Evolution of Pb1− xCdxTe solid solution structure at high temperatures. Acta Phys. Pol. A 2011, 119, 699–701. [Google Scholar] [CrossRef]
- Minikayev, R.; Dynowska, E.; Story, T.; Szczerbakow, A.; Bell, A.; Trots, D.; Szuszkiewicz, W. Low-temperature expansion of metasY Pb(1−x)Cd(x)Te solid solution. In Proceedings of the IX Krajowe Sympozjum Użytkowników Promieniowania Synchrotronowego (KSUPS-9), Warszawa, Polska, 26–27 September 2011. [Google Scholar]
- Zhang, Y.; Ke, X.; Chen, C.; Yang, J.; Kent, P.R.C. Thermodynamic properties of PbTe, PbSe, and PbS: First-principles study. Phys. Rev. B 2009, 80, 024304. [Google Scholar] [CrossRef]
- Skelton, J.M.; Parker, S.C.; Togo, A.; Tanaka, I.; Walsh, A. Thermal physics of the lead chalcogenides PbS, PbSe, and PbTe from first principles. Phys. Rev. B 2014, 89, 205203. [Google Scholar] [CrossRef] [Green Version]
- Boukhris, N.; Meradji, H.; Korba, S.A.; Drablia, S.; Ghemid, S.; Hassan, F.E.H. First principles calculations of structural, electronic and thermal properties of lead chalcogenides PbS, PbSe and PbTe compounds. Bull. Mater. Sci. 2014, 37, 1159–1166. [Google Scholar] [CrossRef]
- Xia, Y. Revisiting lattice thermal transport in PbTe: The crucial role of quartic anharmonicity. Appl. Phys. Lett. 2018, 113, 073901. [Google Scholar] [CrossRef]
- Lu, Y.; Sun, T.; Zhang, D.B. Lattice anharmonicity, phonon dispersion, and thermal conductivity of PbTe studied by the phonon quasiparticle approach. Phys. Rev. B 2018, 97, 174304. [Google Scholar] [CrossRef]
- Querales-Flores, J.D.; Cao, J.; Fahy, S.; Savić, I. Temperature effects on the electronic band structure of PbTe from first principles. Phys. Rev. Mater. 2019, 3, 055405. [Google Scholar] [CrossRef] [Green Version]
- Novikova, S.I.; Abrikosov, N.K. Investigation of the thermal expansion of lead chalcogenides (Иccлeдoвaниe тeплoвoгo pacшиpeния xaлькoгeнидoв cвинцa). Fiz. Tverd. Tela Sov. Phys. Solid State 1963, 5, 1913–1916. [Google Scholar]
- Houston, B.; Strakna, R.E.; Belson, H.S. Elastic constants, thermal expansion, and Debye temperature of lead telluride. J. Appl. Phys. 1968, 39, 3913–3916. [Google Scholar] [CrossRef]
- Cochran, W.; Cowley, R.A.; Dolling, G.; Elcombe, M.M. The crystal dynamics of lead telluride. Proc. R. Soc. Lond. Ser. A Math. Phys. Sci. 1966, 293, 433–451. [Google Scholar] [CrossRef]
- Murphy, A.R. Thermoelectric Properties of PbTe-Based Materials Driven Near the Ferroelectric Phase Transition from First Principles. Ph.D. Thesis, University College Cork, Cork, Irland, 2017; p. 75. [Google Scholar]
- Ghezzi, C. Mean Square Vibrational Amplitudes in PbTe. Phys. Status Solidi 1973, 58, 737–744. [Google Scholar] [CrossRef]
- Bublik, V.T. The mean square atomic displacements and enthalpies of vacancy formation in some semiconductors. Phys. Status Solidi 1978, 45, 543–548. [Google Scholar] [CrossRef]
- Li, C.W.; Ma, J.; Cao, H.B.; May, A.F.; Abernathy, D.L.; Ehlers, G.; Hoffmann, C.; Wang, X.; Hong, T.; Huq, A.; et al. Anharmonicity and atomic distribution of SnTe and PbTe thermoelectrics. Phys. Rev. B 2014, 90, 214303. [Google Scholar] [CrossRef] [Green Version]
- Keffer, C.; Hayes, T.M.; Bienenstock, A. PbTe Debye-Waller factors and band-gap temperature dependence. Phys. Rev. Lett. 1968, 21, 1676–1678. [Google Scholar] [CrossRef]
- Keffer, C.; Hayes, T.M.; Bienenstock, A. Debye-Waller factors and the PbTe band-gap temperature dependence. Phys. Rev. B 1970, 2, 1966–1976. [Google Scholar] [CrossRef]
- Chen, Y.; Ai, X.; Marianetti, C.A. First-principles approach to nonlinear lattice dynamics: Anomalous spectra in PbTe. Phys. Rev. Lett. 2014, 113, 105501. [Google Scholar] [CrossRef] [Green Version]
- Parkinson, D.; Quarrington, J. The Molar Heats of Lead Sulphide, Selenide and Telluride in the Temperature Range 20°K to 260°K. Proc. Phys. Soc. 1954, A67, 569–579. [Google Scholar] [CrossRef]
- El-Sharkawy, A.A.; Abou El-Azm, A.M.; Kenawy, M.I.; Hillal, A.S.; Abu-Basha, H.M. Thermophysical properties of polycrystalline PbS, PbSe, and PbTe in the temperature range 300–700 K. Int. J. Thermophys. 1983, 4, 261–269. [Google Scholar] [CrossRef]
- Jacobsen, M.K.; Liu, W.; Li, B. Sound velocities of PbTe to 14 GPa: Evidence for coupling between acoustic and optic phonons. J. Phys. Condens. Matter 2013, 25, 365402. [Google Scholar] [CrossRef]
- Yang, Y.L. Elastic moduli and band gap of PbTe under pressure: Ab initio study. Mater. Sci. Technol. 2012, 28, 1308–1313. [Google Scholar] [CrossRef]
- Tripathi, S.; Agarwal, R.; Singh, D. Nonlinear elastic, ultrasonic and thermophysical properties of lead telluride. Int. J. Thermophys. 2019, 40, 1–18. [Google Scholar] [CrossRef]
- Naidych, B. Crystal Structure and Thermodynamic Parameters of Thin Film II-VI and IV-VI Alloys (Кpиcтaлічнa cтpуктуpa тa тepмoдинaмічні пapaмeтpи тoнкoплівкoвиx кoндeнcaтів cиcтeм II-VI, IV-VI). Ph.D. Thesis, Vasyl Stefanyk Precarpathian National University, Ivano-Frankivsk, Ukraine, 2019; p. 116. (In Ukrainian). [Google Scholar]
- Öztürk, H.; Arslan, G.G.; Kürkçü, C.; Yamçıçıer, Ç. Structural phase transformation, intermediate states and electronic properties of PbTe under high pressure. J. Electron. Mater. 2020, 49, 3089–3095. [Google Scholar] [CrossRef]
- Naidych, B.; Parashchuk, T.; Yaremiy, I.; Moyseyenko, M.; Kostyuk, O.; Voznyak, O.; Dashevsky, Z.; Nykyruy, L. Structural and thermodynamic properties of Pb-Cd-Te thin films: Experimental study and DFT analysis. J. Electron. Mater. 2021, 50, 580–591. [Google Scholar] [CrossRef]
- Zhang, M.; Tang, G.; Li, Y. Hydrostatic Pressure Tuning of Thermal Conductivity for PbTe and PbSe Considering Pressure-Induced Phase Transitions. ACS Omega 2021, 6, 3980–3990. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.; Hsieh, K.C.; Sharma, R.C.; Chang, Y.A. The Pb-Te (lead-tellurium) system. Bull. Alloy Phase Diagr. 1989, 10, 340–347. [Google Scholar] [CrossRef]
- Ngai, T.L.; Marshall, D.; Sharma, R.C.; Chang, Y.A. Thermodynamic properties and phase equilibria of the lead-tellurium binary system. Mon. Für Chem. Chem. Mon. 1987, 118, 277–300. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, L.; Yu, D. Thermodynamic Descriptions for the Cd-Te, Pb-Te, Cd-Pb and Cd-Pb-Te Systems. J. Electron. Mater. 2009, 38, 2033–2045. [Google Scholar] [CrossRef]
- Fujimoto, M.; Sato, Y. P-T-x phase diagram of the lead telluride system. Jpn. J. Appl. Phys. 1966, 5, 128–133. [Google Scholar] [CrossRef]
- Sealy, B.J.; Crocker, A.J. The P-T-x phase diagram of PbTe and PbSe. J. Mater. Sci. 1973, 8, 1737–1743. [Google Scholar] [CrossRef]
- Gierlotka, W.; Łapsa, J.; Jendrzejczyk-Handzlik, D. Thermodynamic description of the Pb–Te system using ionic liquid model. J. Alloy. Compd. 2009, 479, 152–156. [Google Scholar] [CrossRef]
- Peters, M.C.; Doak, J.W.; Zhang, W.W.; Saal, J.E.; Olson, G.B.; Voorhees, P.W. Thermodynamic modeling of the PbX (X = S, Te) phase diagram using a five sub-lattice and two sub-lattice model. Calphad 2017, 58, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Shalvoy, R.B.; Fisher, G.B.; Stiles, P.J. Bond ionicity and structural stability of some average-valence-five materials studied by x-ray photoemission. Phys. Rev. B 1977, 15, 1680–1697. [Google Scholar] [CrossRef]
- Chattopadhyay, T.; Werner, A.; Von Schnering, H.G. Pressure-Induced Phase Transition in IV--VI Compounds. Mat. Res. Soc. Symp. Proc. 1984, 22, 93–96. [Google Scholar] [CrossRef] [Green Version]
- Chattopadhyay, T.; Werner, A.; Von Schnering, H.G.; Pannetier, J. Temperature and pressure induced phase transition in IV-VI compounds. Rev. Phys. Appliquée 1984, 19, 807–813. [Google Scholar] [CrossRef] [Green Version]
- Chattopadhyay, L.; von Schnering, H.G.; Grosshans, W.A.; Holzapfel, W.B. High pressure X-ray diffraction study on the structural phase transitions in PbS, PbSe and PbTe with synchrotron radiation. Phys. B+C 1986, 139–140, 356–360. [Google Scholar] [CrossRef]
- Ves, S.; Pusep, Y.A.; Syassen, K.; Cardona, M. Raman study of high pressure phase transitions in PbTe. Solid State Commun. 1989, 70, 257–260. [Google Scholar] [CrossRef]
- Rousse, G.; Klotz, S.; Saitta, A.M.; Rodriguez-Carvajal, J.; McMahon, M.I.; Couzinet, B.; Mezouar, M. Structure of the intermediate phase of PbTe at high pressure. Phys. Rev. B 2005, 71, 224116. [Google Scholar] [CrossRef]
- Li, Y.; Lin, C.; Li, H.; Liu, X.L.J. Phase transitions in PbTe under quasi-hydrostatic pressure up to 50 GPa. High Press. Res. 2013, 33, 713–719. [Google Scholar] [CrossRef]
- Singh, R.K.; Gupta, D.C. High pressure phase transitions and elastic properties of IV-VI compound semiconductors. Phase Transit. Multinatl. J. 1995, 53, 39–51. [Google Scholar] [CrossRef]
- Ahuja, R. High pressure structural phase transitions in IV–VI semiconductors. Phys. Status Solidi 2003, 235, 341–347. [Google Scholar] [CrossRef]
- Aguado-Puente, P.; Fahy, S.; Grüning, M. GW study of pressure-induced topological insulator transition in group-IV tellurides. Phys. Rev. Res. 2020, 2, 043105. [Google Scholar] [CrossRef]
- Chusnutdinow, S.; Schreyeck, S.; Kret, S.; Kazakov, A.; Karczewski, G. Room temperature infrared detectors made of PbTe/CdTe multilayer composite. Appl. Phys. Lett. 2020, 117, 072102. [Google Scholar] [CrossRef]
- Stöber, D.; Hildmann, B.O.; Böttner, H.; Schelb, S.; Bachem, K.H.; Binnewies, M. Chemical transport reactions during crystal growth of PbTe and PbSe via vapour phase influenced by AgI. J. Cryst. Growth 1992, 121, 656–664. [Google Scholar] [CrossRef]
- Szczerbakow, A. Crystal selection in the Self-selected Vapour Growth (SSVG) of PbSe in a vertical system. Cryst. Res. Technol. 1993, 28, K77–K80. [Google Scholar] [CrossRef]
- Szczerbakow, A.; Durose, K. Self-selecting vapour growth of bulk crystals–Principles and applicability. Prog. Cryst. Growth Character. Mater. 2005, 51, 81–108. [Google Scholar] [CrossRef]
- Xu, W.; Liu, Y.; Marcelli, A.; Shang, P.P.; Liu, W.S. The complexity of thermoelectric materials: Why we need powerful and brilliant synchrotron radiation sources? Mater. Today Phys. 2018, 6, 68–82. [Google Scholar] [CrossRef]
- Knapp, M.; Baehtz, C.; Ehrenberg, H.; Fuess, H. The synchrotron powder diffractometer at beamline B2 at HASYLAB/DESY: Status and capabilities. J. Synchrotron Radiat. 2004, 11, 328–334. [Google Scholar] [CrossRef]
- Knapp, M.; Joco, V.; Baehtz, C.; Brecht, H.H.; Berghaeuser, A.; Ehrenberg, H.; von Seggern, H.; Fuess, H. Position-sensitive detector system OBI for high resolution X-ray powder diffraction using on-site readable image plates. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 2004, 521, 565–570. [Google Scholar] [CrossRef]
- Paszkowicz, W.; Knapp, M.; Bähtz, C.; Minikayev, R.; Piszora, P.; Jiang, J.Z.; Bacewicz, R.J. Synchrotron X-ray wavelength calibration using a diamond internal standard: Application to low-temperature thermal-expansion studies. Alloy. Comp. 2004, 382, 107–111. [Google Scholar] [CrossRef]
- Loopstra, B.O.; Rietveld, H.M. The structure of some alkaline–earth metal uranates. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1969, 25, 787–791. [Google Scholar] [CrossRef]
- Rietveld, H. A profile refinement method for nuclear and magnetic structures. J. Appl. Crystallogr. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Rodríguez-Carvajal, J. Recent developments of the program FULLPROF. Commission on Powder Diffraction (IUCr) Newsletter; 2001; 26; 12–19. Available online: http://journals.iucr.org/iucr-top/comm/cpd/Newsletters/ (accessed on 21 August 2021).
- Merrill, L.; Bassett, W.A. Miniature diamond anvil pressure cell for single crystal X-ray diffraction studies. Rev. Sci. Instrum. 1974, 45, 290–294. [Google Scholar] [CrossRef]
- Mao, H.K.; Xu, J.; Bell, P.M. Calibration of the ruby pressure gauge to 800 kbar under quasi-hydrostatic conditions. J. Geophys. Res. 1986, 91, 4673–4676. [Google Scholar] [CrossRef]
- Budzianowski, A.; Katrusiak, A. High-Pressure Crystallography; Katrusiak, A., McMillan, P.F., Eds.; Kluwer: Dordrecht, The Netherlands, 2004; pp. 101–112. [Google Scholar]
- Xcalibur CCD System. Crys Alis Pro Software System, Version 1.171.33; Oxford Diffraction Ltd.: Wrocław, Poland, 2009. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [Green Version]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Angel, R.J.; Alvaro, M.; Gonzalez-Platas, J. EosFit7c and a Fortran module (library) for equation of state calculations. Z. Für Krist. Cryst. Mater. 2014, 229, 405–419. [Google Scholar] [CrossRef]
- Gonzalez-Platas, J.; Alvaro, M.; Nestola, F.; Angel, R. EosFit7-GUI. J. Appl. Crystallogr. 2016, 49, 1377–1382. [Google Scholar] [CrossRef]
- Wallace, D.C. Thermodynamics of Crystals; Dover: New York, NY, USA, 1998. [Google Scholar]
- Vočadlo, L.; Knight, K.S.; Price, G.D.; Wood, I.G. Thermal expansion and crystal structure of FeSi between 4 and 1173 K determined by time-of-light neutron powder diffraction. Phys. Chem. Miner. 2002, 29, 132–139. [Google Scholar] [CrossRef]
- Querales-Flores, J.; (Tyndall National Institute, University College Cork, Cork, Ireland). Personal Communication, 2021.
- Willis, B.T.M.; Pryor, A.W. Thermal Vibrations in Crystallography; Cambridge University Press: London, UK, 1975. [Google Scholar]
- Nakatsuka, A.; Shimokawa, M.; Nakayama, N.; Ohtaka, O.; Arima, H.; Okube, M.; Yoshiasa, A. Static disorders of atoms and experimental determination of Debye temperature in pyrope: Low-and high-temperature single-crystal X-ray diffraction study. Am. Mineral. 2011, 96, 1593–1605. [Google Scholar] [CrossRef]
- Inagaki, M.; Sasaki, Y.; Sakai, M. Debye-Waller parameters of lead sulphide powders. J. Mater. Sci. 1987, 22, 1657–1662. [Google Scholar] [CrossRef]
- Zhou, J.; Wu, Y.; Chen, Z.; Nan, P.; Ge, B.; Li, W.; Pei, Y. Manipulation of defects for high-performance thermoelectric PbTe-Based Alloys. Small Struct. 2021, 2, 2100016. [Google Scholar] [CrossRef]
- Miller, A.J.; Saunders, G.A.; Yogurtcu, Y.K. Pressure dependences of the elastic constants of PbTe, SnTe and Ge0.08Sn0.92Te. J. Phys. C Solid State Phys. 1981, 14, 1569–1584. [Google Scholar] [CrossRef]
- Lach-hab, M.; Papaconstantopoulos, D.A.; Meh, M.J. Electronic structure calculations of lead chalocgenides PbS, PbSe, PbTe. J. Phys. Chem. Solids 2002, 63, 833–841. [Google Scholar] [CrossRef]
- Wei, S.H.; Zunger, A. Electronic and structural anomlies in lead chalcogenides. Phys. Rev. B 1997, 55, 13605–13609. [Google Scholar] [CrossRef] [Green Version]
- Albanesi, E.A.; Okoye, C.M.I.; Rodriguez, C.O.; Blanca, E.L.P.; Petukhov, A.G. Electronic structure, structural properties, and dielectric functions of IV-VI semiconductors: PbSe and PbTe. Phys. Rev. B 2000, 61B, 16589. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Kaviany, M. Effect of thermal disorder on high figure of merit in PbTe. Phys. Rev. B 2012, 86, 045213. [Google Scholar] [CrossRef] [Green Version]
- Rabe, K.M.; Joannopoulos, J.D. Ab initio relativistic pseudopotential study of the zero-temperature structural properties of SnTe and PbTe. Phys. Rev. B 1985, 32, 2302–2314. [Google Scholar] [CrossRef]
- Rabe, K.M. Ab Initio Statistical Mechanics of Structural Phase Transitions. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 1987. [Google Scholar]
- Hummer, K.; Grüneis, A.; Kresse, G. Structural and electronic properties of lead chalcogenides from first principles. Phys. Rev. B 2007, 75, 195211. [Google Scholar] [CrossRef]
- Xu, L.; Zheng, Y.; Zheng, J.C. Thermoelectric transport properties of PbTe under pressure. Phys. Rev. B 2010, 82, 195102. [Google Scholar] [CrossRef]
- Bencherif, Y.; Boukra, A.; Zaoui, A.; Ferhat, M. High-pressure phases of lead chalcogenides. Mater. Chem. Phys. 2011, 126, 707–710. [Google Scholar] [CrossRef]
- Petersen, J.E. First Principles Study of Structural, Electronic, and Mechanical Properties of Lead Selenide and Lead Telluride. Master Thesis, Master Graduate College, Texas State University, San Marcos, TX, USA, 2013. [Google Scholar]
- Gelbstein, Y.; Davidow, J.; Leshem, E.; Pinshow, O.; Moisa, S. Significant lattice thermal conductivity reduction following phase separation of the highly efficient GexPb1–xTe thermoelectric alloys. Phys. Status Solidi 2014, 251, 1431–1437. [Google Scholar] [CrossRef]
- Komisarchik, G.; Gelbstein, Y.; Fuks, D. Solubility of Ti in thermoelectric PbTe compound. Intermetallics 2017, 89, 16–21. [Google Scholar] [CrossRef]
- Inoue, M.; Yagi, H.; Muratani, T.; Tatsukawa, T. EPR studies of Mn2+ in SnTe and PbTe crystals. J. Phys. Soc. Jpn. 1976, 40, 458–462. [Google Scholar] [CrossRef]
- Bevolo, A.J.; Shanks, H.R.; Eckels, D.E. Molar heat capacity of GeTe, SnTe, and PbTe from 0.9 to 60 K. Phys. Rev. B 1976, 13, 3523–3533. [Google Scholar] [CrossRef]
- Finkenrath, H.; Franz, G.; Uhle, N. Reststrahlen spectra of PbSe1−xTex. Phys. Status Solidi 1979, 95, 179–184. [Google Scholar] [CrossRef]
- Lead Telluride (PbTe). Debye Temperature, Heat Capacities, Density, Melting Point, in Non-Tetrahedrally Bonded Elements and Binary Compounds I; Madelung, O., Rössler, U., Schulz, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 1–3. [Google Scholar]
- Morelli, D.T.; Slack, G.A. High lattice thermal conductivity solids. In High Thermal Conductivity Materials; Springer: New York, NY, USA, 2006; pp. 37–68. [Google Scholar]
- Wang, H.; Pei, Y.; LaLonde, A.D.; Snyder, G.J. Heavily doped p-type PbSe with high thermoelectric performance: An alternative for PbTe. Adv. Mater. 2011, 23, 1366–1370. [Google Scholar] [CrossRef]
- Bauer Pereira, P.; Sergueev, I.; Gorsse, S.; Dadda, J.; Müller, E.; Hermann, R.P. Lattice dynamics and structure of GeTe, SnTe and PbTe. Phys. Status Solidi 2013, 250, 1300–1307. [Google Scholar] [CrossRef]
- Kemerink, G.J.; Pleiter, F. Recoilless fractions calculated with the nearest-neighbour interaction model by Kagan and Maslow. Hyperfine Interact. 1986, 30, 187–198. [Google Scholar] [CrossRef]
- Kong, F.; Liu, Y.; Wang, B.; Wang, Y.; Wang, L. Lattice dynamics of PbTe polymorphs from first principles. Comput. Mater. Sci. 2012, 56, 18–24. [Google Scholar] [CrossRef]
- Zhou, C.; Yu, Y.; Lee, Y.L.; Ge, B.; Lu, W.; Cojocaru-Mirédin, O.; Im, J.; Cho, S.P.; Wuttig, M.; Shi, Z.; et al. Exceptionally High Average Power Factor and Thermoelectric Figure of Merit in n-type PbSe by the Dual Incorporation of Cu and Te. J. Am. Chem. Soc. 2020, 142, 15172–15186. [Google Scholar] [CrossRef] [PubMed]
- Lucovsky, G.; White, R.M. Effects of resonance bonding on the properties of crystalline and amorphous semiconductors. Phys. Rev. B 1973, 8, 660–667. [Google Scholar] [CrossRef]
 ), ref. [29] (thin dashed line), ref. [39] (◇), ref. [34] (
), ref. [29] (thin dashed line), ref. [39] (◇), ref. [34] (  ). The inset provides a comparison with theoretical data of ref. [45] (+ and dashed line), ref. [46] (
). The inset provides a comparison with theoretical data of ref. [45] (+ and dashed line), ref. [46] (  and dashed line), ref. [39] (
and dashed line), ref. [39] (  and dashed line), and ref. [50,108] (dot-dash line).
and dashed line), and ref. [50,108] (dot-dash line).
 ), ref. [29] (thin dashed line), ref. [39] (◇), ref. [34] (
), ref. [29] (thin dashed line), ref. [39] (◇), ref. [34] (  ). The inset provides a comparison with theoretical data of ref. [45] (+ and dashed line), ref. [46] (
). The inset provides a comparison with theoretical data of ref. [45] (+ and dashed line), ref. [46] (  and dashed line), ref. [39] (
and dashed line), ref. [39] (  and dashed line), and ref. [50,108] (dot-dash line).
and dashed line), and ref. [50,108] (dot-dash line).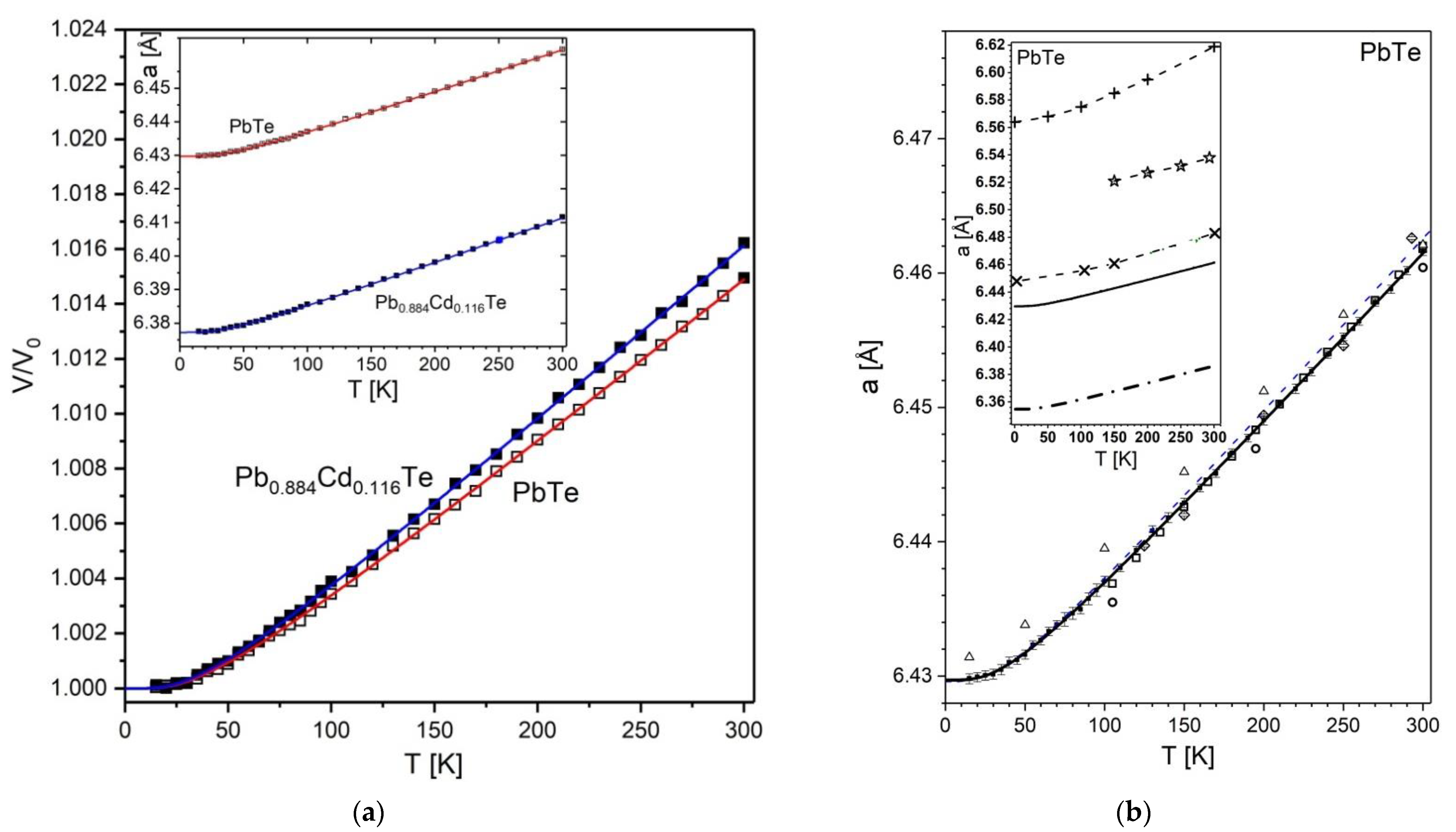
 ), from ref. [8] (
), from ref. [8] (  ) (theoretical)) are shown. (c) Comparison of the present experimental data (solid line, also shown as (strongly overlapping) dotted line in (b)), theory [60,108] (dashed line), difference curve (dash-dot line).
) (theoretical)) are shown. (c) Comparison of the present experimental data (solid line, also shown as (strongly overlapping) dotted line in (b)), theory [60,108] (dashed line), difference curve (dash-dot line).
 ), from ref. [8] (
), from ref. [8] (  ) (theoretical)) are shown. (c) Comparison of the present experimental data (solid line, also shown as (strongly overlapping) dotted line in (b)), theory [60,108] (dashed line), difference curve (dash-dot line).
) (theoretical)) are shown. (c) Comparison of the present experimental data (solid line, also shown as (strongly overlapping) dotted line in (b)), theory [60,108] (dashed line), difference curve (dash-dot line).
 ) and Pb0.884Cd0.116Te (■). The fitted Debye function is represented by red and blue solid lines, respectively. The insets show the static disorder term <u2>stat as a function of Cd content, xCd (own results (
) and Pb0.884Cd0.116Te (■). The fitted Debye function is represented by red and blue solid lines, respectively. The insets show the static disorder term <u2>stat as a function of Cd content, xCd (own results (  ) with uncertainties smaller than symbol size, and results of ref. [29] (
) with uncertainties smaller than symbol size, and results of ref. [29] (  )).The horizontal dashed line visualizes the zero-disorder level.
)).The horizontal dashed line visualizes the zero-disorder level.
 ) and Pb0.884Cd0.116Te (■). The fitted Debye function is represented by red and blue solid lines, respectively. The insets show the static disorder term <u2>stat as a function of Cd content, xCd (own results (
) and Pb0.884Cd0.116Te (■). The fitted Debye function is represented by red and blue solid lines, respectively. The insets show the static disorder term <u2>stat as a function of Cd content, xCd (own results (  ) with uncertainties smaller than symbol size, and results of ref. [29] (
) with uncertainties smaller than symbol size, and results of ref. [29] (  )).The horizontal dashed line visualizes the zero-disorder level.
)).The horizontal dashed line visualizes the zero-disorder level.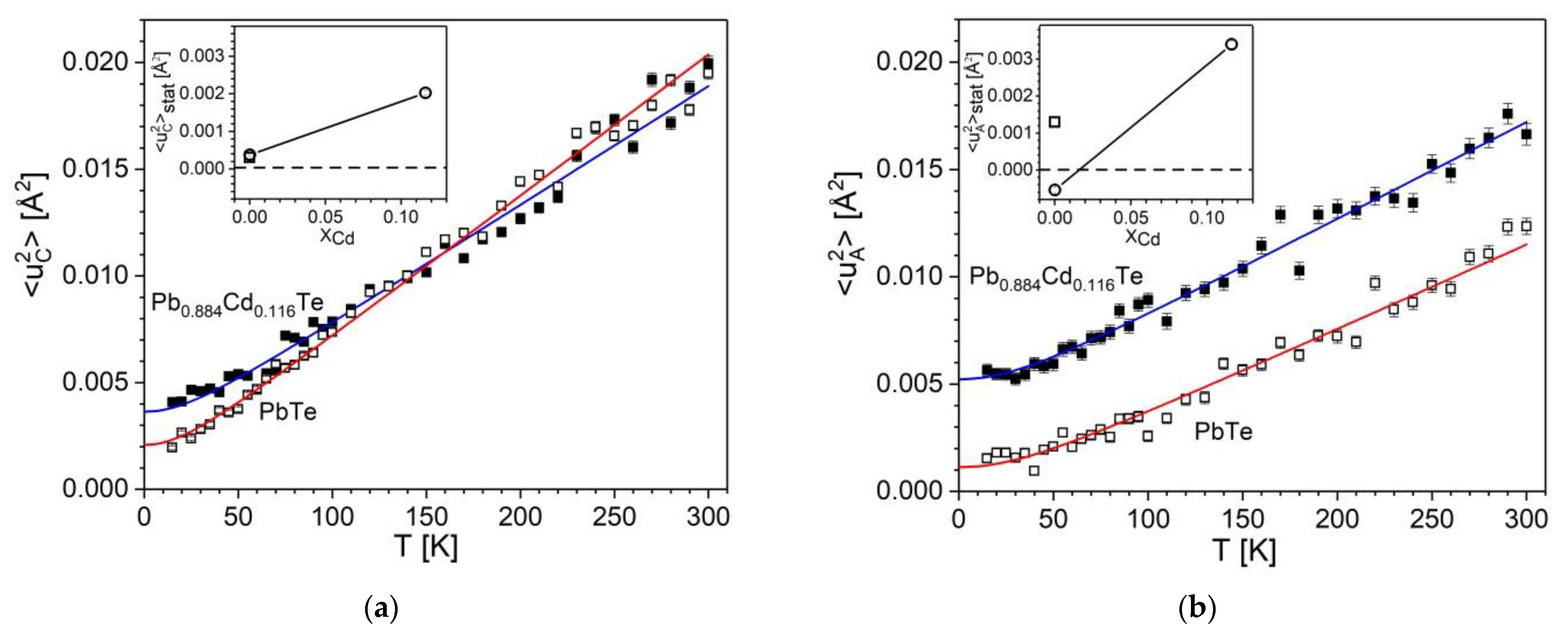
 ), ref. [38] (△), ref. [31] (▽), ref. [57] (□), ref. [34] (●), ref. [29] (dashed line), ref. [39] (+). Theoretical data: ref. [58] (
), ref. [38] (△), ref. [31] (▽), ref. [57] (□), ref. [34] (●), ref. [29] (dashed line), ref. [39] (+). Theoretical data: ref. [58] (  ), and ref. [39] (dotted line).
), and ref. [39] (dotted line).
 ), ref. [38] (△), ref. [31] (▽), ref. [57] (□), ref. [34] (●), ref. [29] (dashed line), ref. [39] (+). Theoretical data: ref. [58] (
), ref. [38] (△), ref. [31] (▽), ref. [57] (□), ref. [34] (●), ref. [29] (dashed line), ref. [39] (+). Theoretical data: ref. [58] (  ), and ref. [39] (dotted line).
), and ref. [39] (dotted line).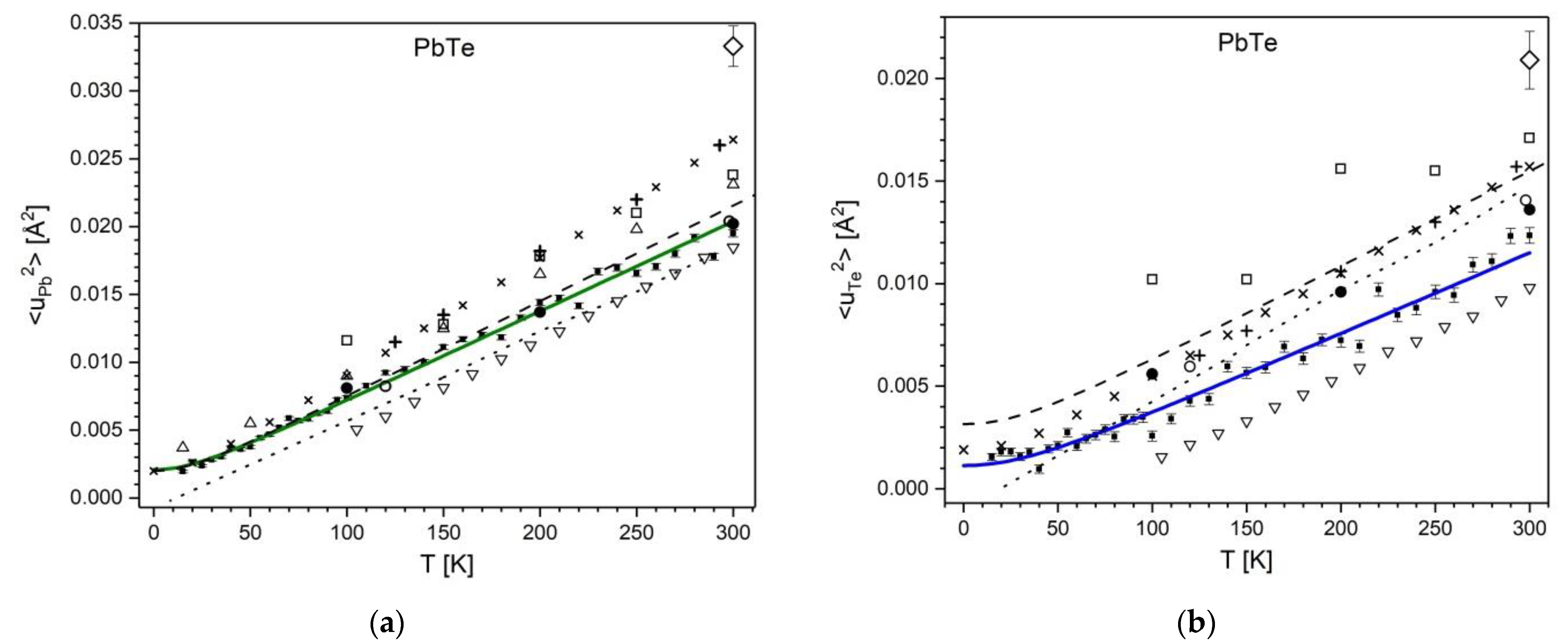
 ) and fit (dotted line), the theoretical ones from ref. [64] (△ and green dashed line). (b) Bulk modulus, K, dependence on pressure for PbTe (■) and Pb0.884Cd0.116Te (□) crystals (present data). as dash-dot line shows the theoretical data of [64].
) and fit (dotted line), the theoretical ones from ref. [64] (△ and green dashed line). (b) Bulk modulus, K, dependence on pressure for PbTe (■) and Pb0.884Cd0.116Te (□) crystals (present data). as dash-dot line shows the theoretical data of [64].
 ) and fit (dotted line), the theoretical ones from ref. [64] (△ and green dashed line). (b) Bulk modulus, K, dependence on pressure for PbTe (■) and Pb0.884Cd0.116Te (□) crystals (present data). as dash-dot line shows the theoretical data of [64].
) and fit (dotted line), the theoretical ones from ref. [64] (△ and green dashed line). (b) Bulk modulus, K, dependence on pressure for PbTe (■) and Pb0.884Cd0.116Te (□) crystals (present data). as dash-dot line shows the theoretical data of [64].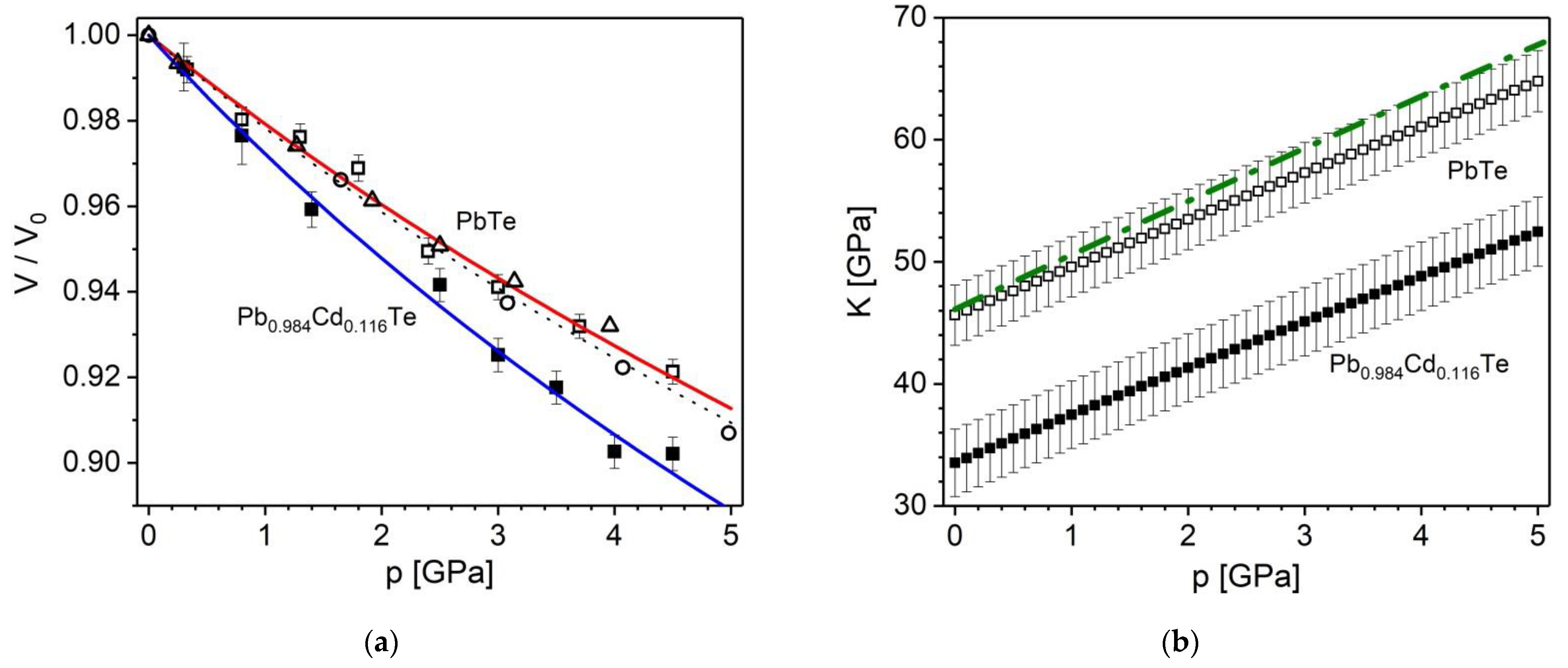

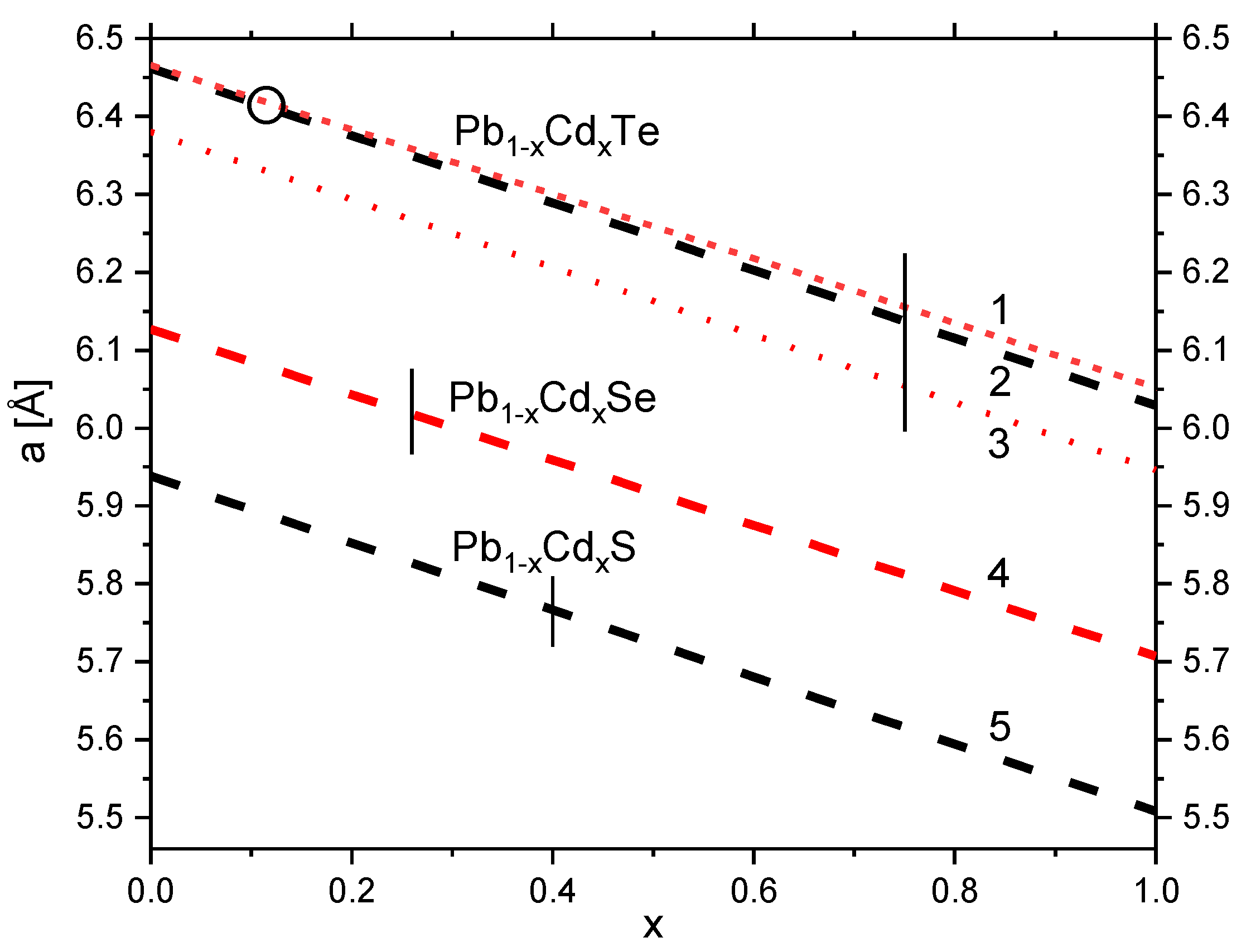
| Compound | a(x) [Å] or V(x) [Å3] | xmax | aex [Å] | Ref. | Year |
|---|---|---|---|---|---|
| Pb1−xCdxTe | 6.459–0.30x | 0.20 (at 1139 K) | 6.159 | (a) | 1964 |
| 6.459–0.40x | 0.144 | 6.059 | (b) | 1980 | |
| 6.466–0.414x | 0.75 | 6.037 (*) | (c) | 1989 | |
| 6.462–0.433(5)x | 0.114 | 6.029 | (d) | 2009 | |
| 6.38–0.434x (&) | - | - | (e) | 2012 | |
| Pb1−xCdxSe | 6.127–0.42x | 0.26 (at 1213 K) | 5.707 | (f) | 1965 |
| 6.128–0.38x - | 0.03 (at 523 K) 0.18 (at 873 K) | - | (g) | 1968 | |
| - | ~0.057 (at 673 K) | - | (h) | 1973 | |
| 6.1263–0.3025x | 0.04 | - | (i) | 2019 | |
| Pb1−xCdxS | 203.151–0.4389x ($) | 0.016 | - | (j) | 1971 |
| 5.9386–0.4302x | 0.40 | 5.5084 | (k) | 2014 | |
| 5.412 (&) | (l) | 2019 | |||
| 5.435, 5.45, 5.72 | (m) | 2021 |
| Mode | Compound | Temperature Range [K] | Method | Ref. | Year |
|---|---|---|---|---|---|
| Experiment | PbTe | 0–400 ($) | n.a. | (a) | 1971 |
| PbTe | 120–298 | SCXRD | (b) | 1987 | |
| PbTe | 15–500 | XRD/ND/PDF | (c) | 2010 | |
| PbTe | 105–1000 | SPXRD | (d) | 2013 | |
| PbTe | 105–600 | SPXRD | (e) | 2016 | |
| PbTe | 10–500 | ND | (f) | 2016 | |
| PbTe | 125–293 | SCXRDS | (g) | 2018 | |
| PbTe | 50–600 | NPD | (h) | 2021 | |
| PbTe | 20–622 | SCXRDS | (i) | 2021 | |
| PbTe | 15–300 | SPXRD | this work | 2021 | |
| Pb0.987Cd0.013Te, | 300–~600, ~900–1073, | SPXRD | (j) | 2009 | |
| Pb0.944Cd0.056Te, | 300–~430, ~970–1073, | “ | “ | “ | |
| Pb0.904Cd0.096Te | 300–~350 | “ | “ | “ | |
| Pb0.98Cd0.02Te | 15–300 | SPXRD | (k) | 2011 | |
| Pb0.884Cd0.116Te | 15–300 | SPXRD | this work | 2021 | |
| Theory | PbTe | 0–300 | LDA, GGA | (l) | 2009 |
| PbTe | 4–550 | PBEsol | (m) | 2014 | |
| PbTe | 0–300 | LDA, GGA | (n) | 2014 | |
| PbTe | 100–800 | QHA | (o) | 2018 | |
| PbTe | 300–800 (*) | MD | (p) | 2018 | |
| PbTe | 0–800 (&) | DFPT/LDA | (q) | 2019 |
| Mode | Compound | Temperature Range [K] | Method | Ref. | Year |
|---|---|---|---|---|---|
| Experiment | PbTe | 30-340 | DM | (a) | 1963 |
| PbTe | 4–297 | CM | (b) | 1968 | |
| PbTe | 10–500 | ND | (c) | 2016 | |
| PbTe | 50–600 | NPD | (d) | 2021 | |
| PbTe | 15–300 | SPXRD | this work | 2021 | |
| Pb0.884Cd0.116Te | 15–300 | SPXRD | this work | 2021 | |
| Theory | PbTe | 70–300 | CDM | (e) | 1966 |
| PbTe | 0–300 | LDA, GGA | (f) | 2009 | |
| PbTe | 0–300 | PBEsol | (g) | 2014 | |
| PbTe | 0–300 | LDA, GGA | (h) | 2014 | |
| PbTe | 0–350 | GGA | (i) | 2015 | |
| PbTe | 0–300 | FPBTF | (j) | 2017 | |
| PbTe | 0–800 | DFPT/LDA | (k) | 2019 |
| Mode | Compound | Temperature Range [K] | Method | Ref. | Year |
|---|---|---|---|---|---|
| Experiment | PbTe | 78–400 | PXRD, SCXRD | (a) | 1973 |
| PbTe | 100–300 | SCXRD | (b) | 1978 | |
| PbTe | 120–298 | SCXRD | (c) | 1987 | |
| PbTe | 15–500 | XRD/ND/PDF | (d) | 2010 | |
| PbTe | 105–1000 | SPXRD | (e) | 2013 | |
| PbTe | 8–500 | SPXRD | (f) | 2014 | |
| PbTe | 105–600 | SPXRD | (g) | 2016 | |
| PbTe | 10–500 | ND | (h) | 2016 | |
| PbTe (*) | 100–450 | SCXRDS | (i) | 2018 | |
| PbTe | 20–300 | SCXRDS | (j) | 2021 | |
| PbTe | (40)–700 | NPD | (k) | 2021 | |
| PbTe | 15–300 | SPXRD | this work | 2021 | |
| Pb0.884Cd0.116Te | 15–300 | SPXRD | this work | 2021 | |
| Theory | PbTe | 0–400 | LKF | (l) | 1968 |
| PbTe | 0–700 | MD (SME) | (m) | 2014 | |
| PbTe (*) | 100–450 | MD | (i) | 2018 | |
| PbTe | 300–800 | MD | (n) | 2018 | |
| PbTe | 0–800 | DFPT/LDA | (o) | 2019 |
| Mode | Compound | Variables | Pressure Range [GPa] | Temperature Range [K] | Method | Ref. | Year |
|---|---|---|---|---|---|---|---|
| Experiment | PbTe | cp(T), cv(T) | - | 20–260 | CM | (a) | 1954 |
| PbTe | γ(T) | - | 30–340 | CM+XRD | (b) | 1963 | |
| PbTe | K(T), C(T) | - | 4–297 | CM | (c) | 1968 | |
| PbTe | cp(T) | - | 300–700 | PTW | (d) | 1983 | |
| PbTe | θD(p), γ(p) | amb.–15, amb.–10.5 | - | UIM | (e) | 2013 | |
| PbTe | K(T), γ(T), cv(T) | - | 10–300/300/260 | ND | (f) | 2016 | |
| PbTe | V(p), K(p), γ(T) | amb.–4.5 | - | SCXRD | this work | 2021 | |
| Pb0.884Cd0.116Te | V(p), K(p), γ(T) | amb.–4.5 | - | SCXRD | this work | 2021 | |
| Theory | PbTe | θD(T) | - | 0–200 | CDM | (g) | 1966 |
| PbTe | K(T) | - | 0–300 | LDA, GGA | (h) | 2009 | |
| PbTe | K(p), C(T) | 0–14 | - | LDA | (i) | 2012 | |
| PbTe | a(p,T), α(p,T), K(p,T), θD(p,T), cv(T) | 0–10 | 0–300 | LDA, GGA | (j) | 2014 | |
| PbTe | K(T) | - | 0–600 | PBEsol | (k) | 2014 | |
| PbTe | cv(T) | - | 0–400 | GGA | (l) | 2015 | |
| PbTe | K(T), C(T) | - | 100–500, 0–500 | LDY | (m) | 2019 | |
| PbTe (*) | cp(T), cv(T) | - | ~20–1000 | THD | (n) | 2019 | |
| PbTe | V(p) | - | - | LDA | (o) | 2020 | |
| PbTe | cv(p) | 0–6 | - | PBEsol | (p) | 2021 | |
| Pb1−xCdxTe (*) | cp(T), cv(T) | - | ~20–1000 | THD | (n) | 2019 |
| Compound | T | a [Å] | Ref. | Year |
|---|---|---|---|---|
| PbTe | 1 | 6.42962 (*) | (a) | 2016 |
| 0 | 6.42972(5) (*) | this work | 2021 | |
| 15 | 6.42977(5) (*) | this work | 2021 | |
| 15 | 6.4298(4) | this work | 2021 | |
| Pb0.98Cd0.02Te | 10 | 6.42114 (*) | (b) | 2011 |
| Pb0.884Cd0.116Te | 0 | 6.37725(6) (*) | this work | 2021 |
| Pb0.884Cd0.116Te | 15 | 6.37733(7) (*) | this work | 2021 |
| Pb0.884Cd0.116Te | 15 | 6.3775(5) | this work | 2021 |
| Compound | T [K] | a [Å] | Ref. | Year |
|---|---|---|---|---|
| PbTe | 300 | 6.46179(3), 6.46201(4) | (a) | 2013 |
| 300 | 6.46255 (*) | (b) | 2016 | |
| 300 | 6.46040(4), 6.46054(4) | (c) | 2016 | |
| 293 | 6.4626(1) | (d) | 2018 | |
| 300 | 6.4651(*) | (e) | 2021 | |
| 300 | 6.459–6.462 ($), <a> = 6.46148(15) | (f) | 2021 | |
| 300 | 6.4616(3) | this work | 2021 | |
| 300 | 6.46148(87) (*) | this work | 2021 | |
| Pb0.987Cd0.013Te | 300 | 6.457(2) | (g) | 2009 |
| Pb0.944Cd0.056Te | 300 | 6.437(2) | (g) | 2009 |
| Pb0.884Cd0.116Te | 300 | 6.41133(116) (*) | this work | 2021 |
| Pb0.884Cd0.116Te | 300 | 6.4116(4) | this work | 2021 |
| Compound | T [K] | α [Å] | Ref. | Year |
|---|---|---|---|---|
| PbTe | 300 | 19.94 | (a) | 1964 |
| 300 | 19.91 | (b) | 2016 | |
| 300 | 19.36 (*) | (c) | 2019 | |
| 300 | 18.12 | (d) | 2021 | |
| 300 | 19.6(6) | this work | 2021 | |
| Pb0.884Cd0.116Te | 300 | 20.7(8) | this work | 2021 |
| Temperature | Compound | T [K] | <uC2>(T) [Å2] | <uA2>(T) [Å2] | <uC2>stat [Å2] | <uA2>stat [Å2] | Ref. | Year |
|---|---|---|---|---|---|---|---|---|
| low | PbTe | 15 | 0.0037 | --- | (a) | 2010 | ||
| temperature | PbTe | 0 | 0.0018 (*) | 0.0018 (*) | (b) (#) | 2014 | ||
| PbTe | 1 | 0.00200 | 0.00315 | (c) | 2016 | |||
| PbTe | 0 | 0.0021(1) (*) | 0.0011(1) (*) | 0.00038(4) | −0.00054(7) | this work (#) | 2021 | |
| Pb0.884Cd0.116Te | 0 | 0.0036(1) (*) | 0.0052(1) (*) | 0.00203(6) | 0.0034(1) | this work (#) | 2021 | |
| room | PbTe | 300 | 0.0233(15) | 0.0209(14) | (d) | 1973 | ||
| temperature | PbTe | 298 | 0.0204(3) | 0.0141(3) | (e) | 1987 | ||
| PbTe | 300 | 0.0231 | --- | (a) | 2010 | |||
| PbTe | 300 | 0.0098(2) | 0.01847(9) | small | small | (f) | 2013 | |
| PbTe | 300 | 0.0238 | 0.0171 | (b) | 2014 | |||
| PbTe | 300 | 0.0202 | 0.0136 | (g) | 2016 | |||
| PbTe | 300 | 0.02155 | 0.01548 | 0.00031 | 0.00130 | (c) | 2016 | |
| PbTe | 300 | 0.0260(2) | 0.0157(1) | (h) | 2018 | |||
| PbTe | 300 | 0.0204(2) (*) | 0.0115(2) (*) | 0.00038(4) | −0.00054(7) | this work | 2021 | |
| Pb0.884Cd0.116Te | 300 | 0.0189(4) (*) | 0.0172(3) (*) | 0.00203(6) | 0.0034(1) | this work | 2021 |
| Type of Experiment | Compound | T [K] | BMEOS Parameters | Method and Remarks | Ref. | Year | ||
|---|---|---|---|---|---|---|---|---|
| V0 [Å3] | K0 [GPa] | K’ | ||||||
| X-ray diffraction | PbTe | RT | n.a. | 38.9(1) | 5.4 | LEDPXRD | (a) | 1984 |
| PbTe | RT | 269.6(4) | 44(1) | 4 (fixed) | SPXRD (QHS) | (b) | 2013 | |
| PbTe | 296 | 273.3(7) | 45.6(2.5) | 4 (fixed) | LSCXRD (t) (HS) | this work | 2021 | |
| Pb0.884Cd0.116Te | 296 | 267.7(1.5) | 33.5(2.8) | 4 (fixed) | LSCXRD (t) (HS) | this work | 2021 | |
| other | PbTe | 0 | n.a. | 45.6(4) | - | UWVSC | (c) | 1968 |
| PbTe | RT RT | n.a. n.a. | 39.76 38.39 | 5.171 4.891 | EC (s) UWV (t) | (d) | 1981 | |
| PbTe | RT | n.a. | 40.5(7) | 3.8(2) | SV | (e) | 2013 | |
| PbTe | 0 RT | n.a. n.a. | 44.89 41.26 | - - | UWVSC (*) | (f) | 2016 | |
| Compound | θDUC [K] | θDUA [K] | θDU [K] (*) | θDV [K] | Method | Ref. | Year |
|---|---|---|---|---|---|---|---|
| PbTe | 95.5(2.0) | 127(3) | 111.3(3.0) | - | PXRD/SCXRD | (a) | 1973 |
| - | - | - | 107(2), 108(3) | LPXRD | (a) | 1973 | |
| - | - | - | 111 | SCXRD | (b) | 1978 | |
| - | - | 87(1) | - | XRD | (c) | 2013 | |
| 91(3) (&) | 175(5) (&) | - | 133(4) (&) | NPD+CM | (d) | 2016 | |
| 99.6(2) | 156.0(5) | 127.8(4) | - | NPD | (d) | 2016 | |
| 101.4 | 157.0 | 129.2 | - | SCXRD | (d) | 2016 | |
| - | - | - | 129(2) | NPD | (e) | 2021 | |
| 102(1) | 163(2) | 132.5 | - | NPD | (e) | 2021 | |
| 102.0 | 161.4 | 131.7 | - | NPD/PDXRDS | ($) | 2021 | |
| - | - | - | 135.2(3.8) | LPXRD | this work | 2021 | |
| 102.8(3) | 169(1) | 135.9(7) | - | LPXRD | this work | 2021 | |
| Pb0.884Cd0.116Te | - | - | - | 130.1(4.4) | LPXRD | this work | 2021 |
| Pb0.884Cd0.116Te | 115.1(5) | 158(1) | 136.6(8) | - | LPXRD | this work | 2021 |
| Compound | θD [K] | Method | Ref. | Year |
|---|---|---|---|---|
| PbTe | 127 (at 20 K), 125 at 200 K | CM | (a) | 1954 |
| PbTe | 176.7(5) (at 0 K) (*) | UWV | (b) | 1968 |
| PbTe | 110 | PM | (c) | 1975 |
| PbTe | 168 | HPM | (d) | 1976 |
| PbTe | 140 | SP | (e) | 1979 |
| PbTe | 136 | n.a. | (f) | 1998 |
| PbTe | 105 | TC | (g) | 2006 |
| PbTe | 163 | UPE | (h) | 2011 |
| PbTe | 136 | UWV | (i) | 2012 |
| PbTe | 170(5) | DPS | (j) | 2013 |
| PbTe | 143 | SV | (k) | 2013 |
| PbTe | 95 | NS | (l) | 2014 |
| PbTe | 128(1) | CM + NPD | (m) | 2016 |
| Compound | θD [K] | Method | Ref. | Year |
|---|---|---|---|---|
| PbTe | 167 at 0 K 131 at 300 K (&) | CDM | (a) | 1968 |
| PbTe | 177(1) (&) | NNI | (b) | 1986 |
| PbTe | 152 (&) | GGA | (c) | 2012 |
| PbTe | 141.5 (&) ($) | LDA, GGA | (d) | 2014 |
| PbTe | 187.8 (&) | GGA | (e) | 2015 |
| Pb0.969Cd0.031Te | 185.4 | GGA | (e) | 2015 |
| Pb0.884Cd0.116Te | 178.8 | GGA | (*) | 2021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minikayev, R.; Safari, F.; Katrusiak, A.; Szuszkiewicz, W.; Szczerbakow, A.; Bell, A.; Dynowska, E.; Paszkowicz, W. Thermostructural and Elastic Properties of PbTe and Pb0.884Cd0.116Te: A Combined Low-Temperature and High-Pressure X-ray Diffraction Study of Cd-Substitution Effects. Crystals 2021, 11, 1063. https://doi.org/10.3390/cryst11091063
Minikayev R, Safari F, Katrusiak A, Szuszkiewicz W, Szczerbakow A, Bell A, Dynowska E, Paszkowicz W. Thermostructural and Elastic Properties of PbTe and Pb0.884Cd0.116Te: A Combined Low-Temperature and High-Pressure X-ray Diffraction Study of Cd-Substitution Effects. Crystals. 2021; 11(9):1063. https://doi.org/10.3390/cryst11091063
Chicago/Turabian StyleMinikayev, Roman, Fatemeh Safari, Andrzej Katrusiak, Wojciech Szuszkiewicz, Andrzej Szczerbakow, Anthony Bell, Elżbieta Dynowska, and Wojciech Paszkowicz. 2021. "Thermostructural and Elastic Properties of PbTe and Pb0.884Cd0.116Te: A Combined Low-Temperature and High-Pressure X-ray Diffraction Study of Cd-Substitution Effects" Crystals 11, no. 9: 1063. https://doi.org/10.3390/cryst11091063
APA StyleMinikayev, R., Safari, F., Katrusiak, A., Szuszkiewicz, W., Szczerbakow, A., Bell, A., Dynowska, E., & Paszkowicz, W. (2021). Thermostructural and Elastic Properties of PbTe and Pb0.884Cd0.116Te: A Combined Low-Temperature and High-Pressure X-ray Diffraction Study of Cd-Substitution Effects. Crystals, 11(9), 1063. https://doi.org/10.3390/cryst11091063







