Green Synthesis of Ni-Cu-Zn Based Nanosized Metal Oxides for Photocatalytic and Sensor Applications
Abstract
:1. Introduction
2. Experimental
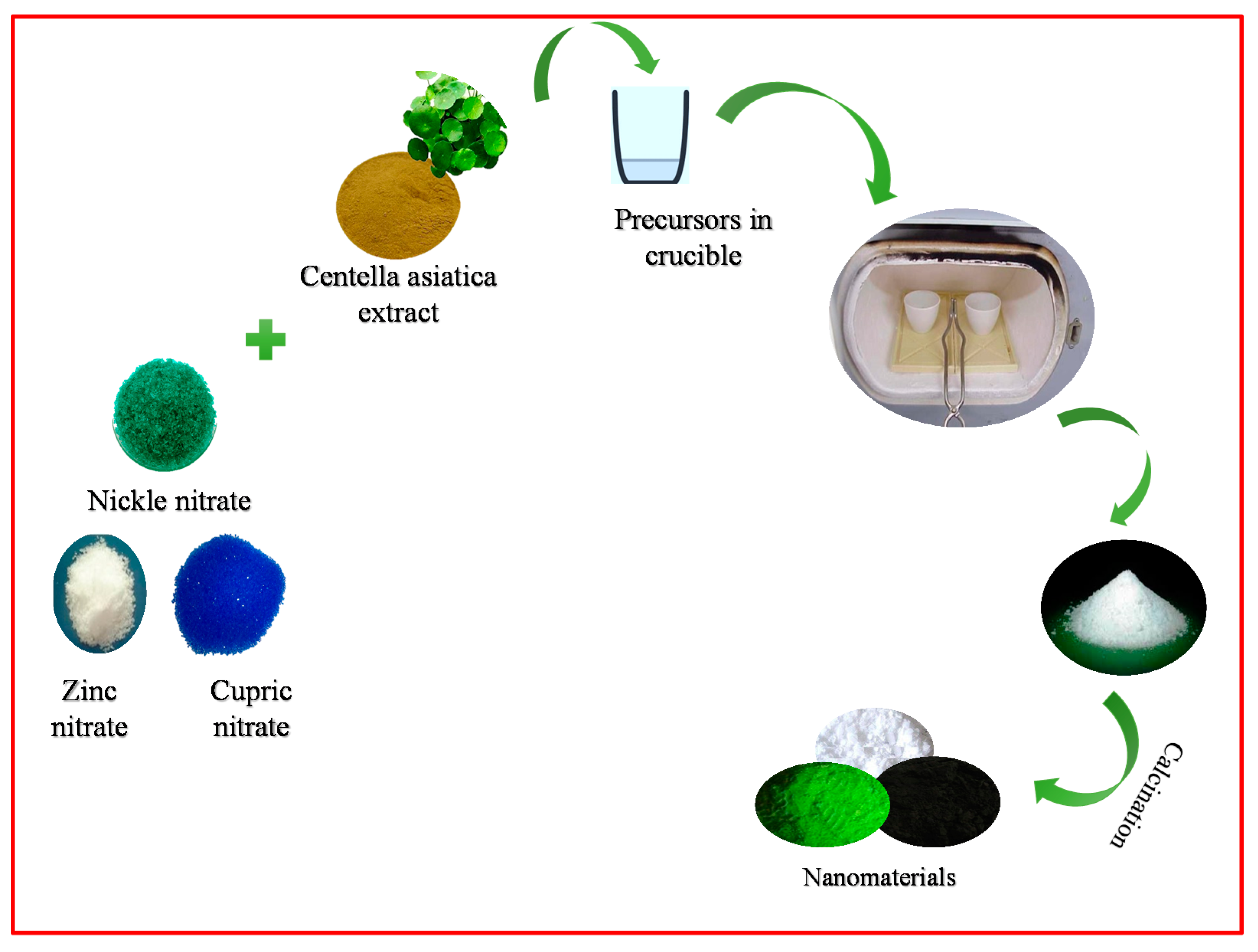
2.1. Preparation of NiO, CuO, and ZnO NPs
2.2. Characterization
3. Results and Discussion
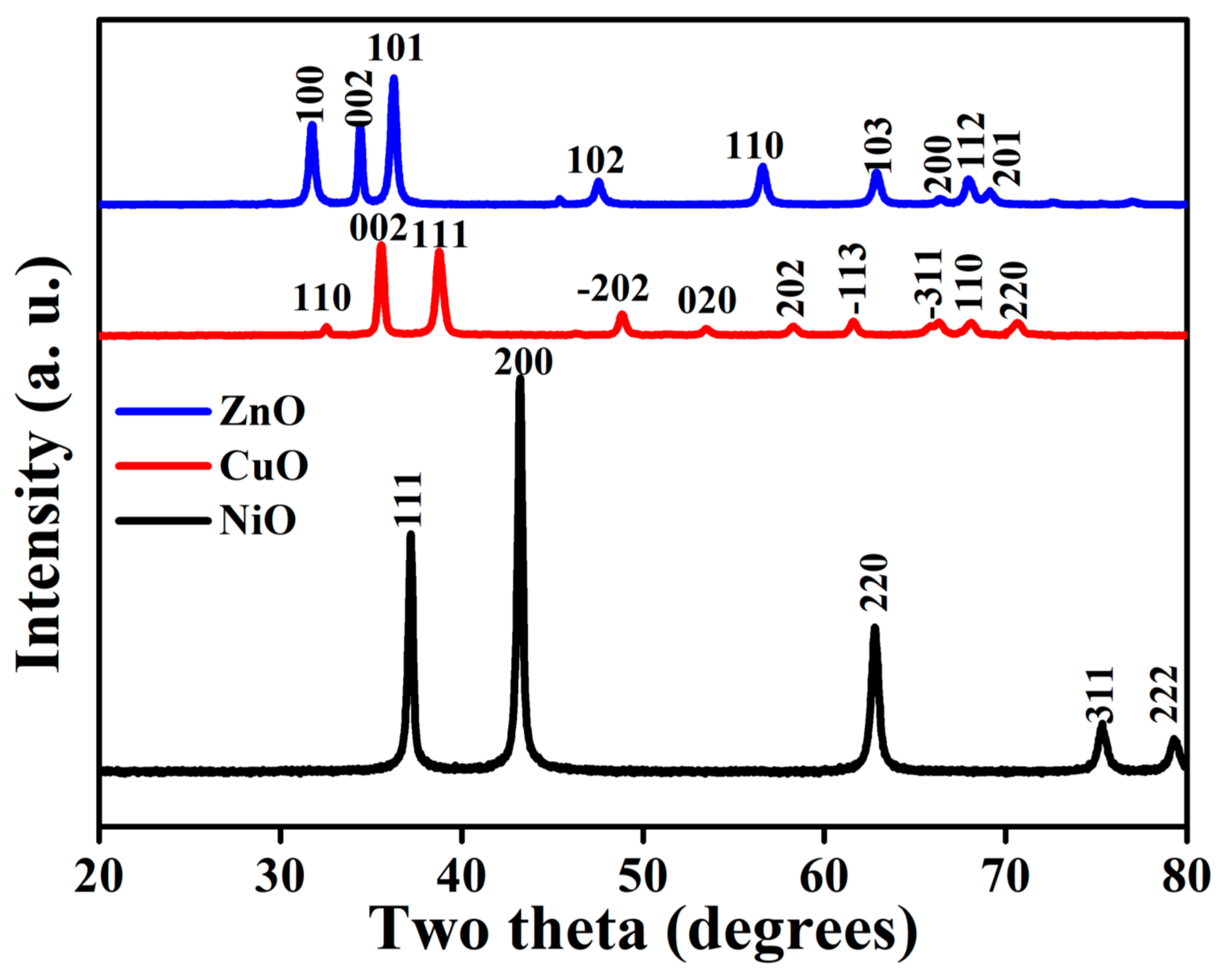
3.1. Structural Analysis
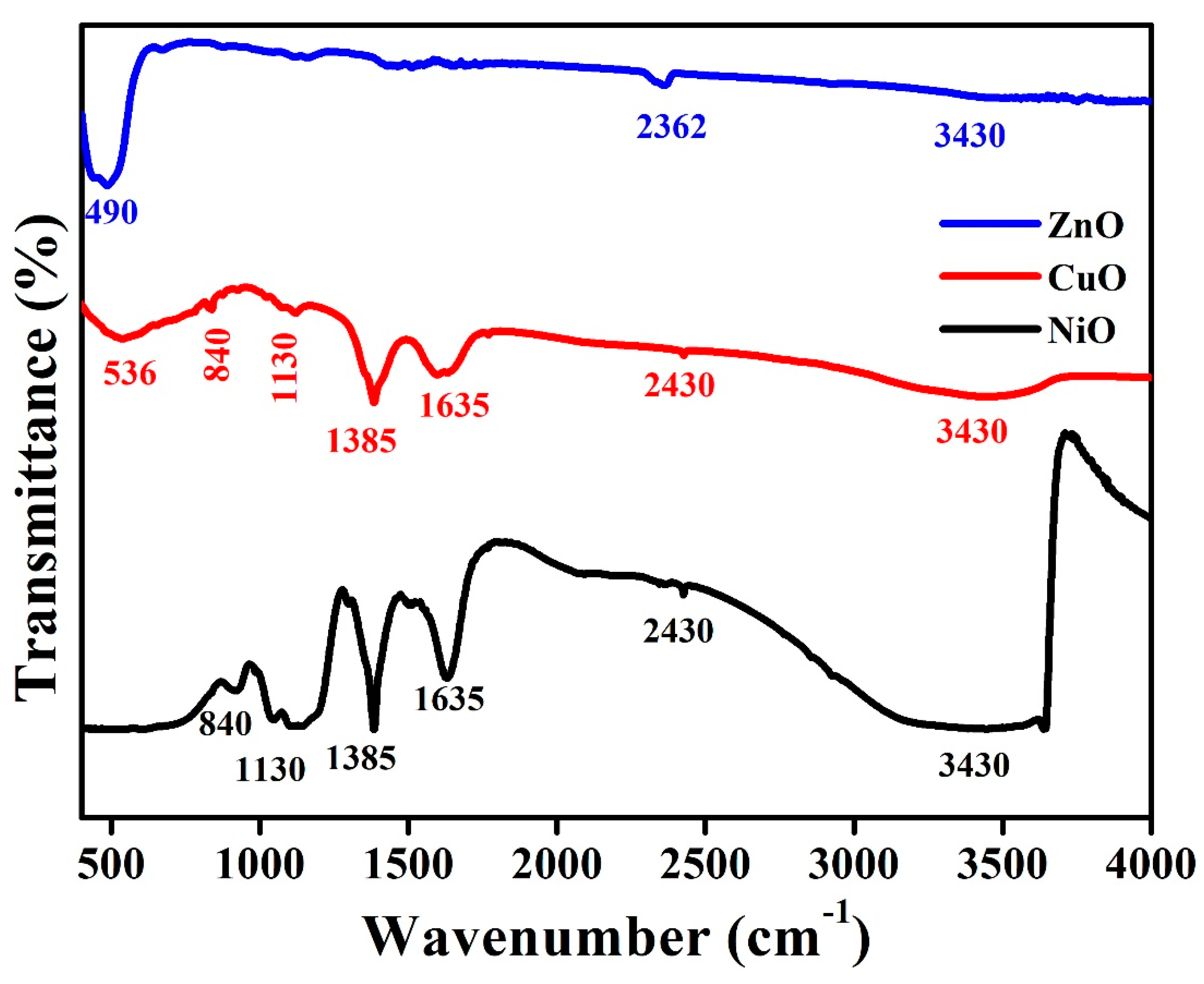
3.2. Functional Group Analysis
3.3. Morphological Analysis
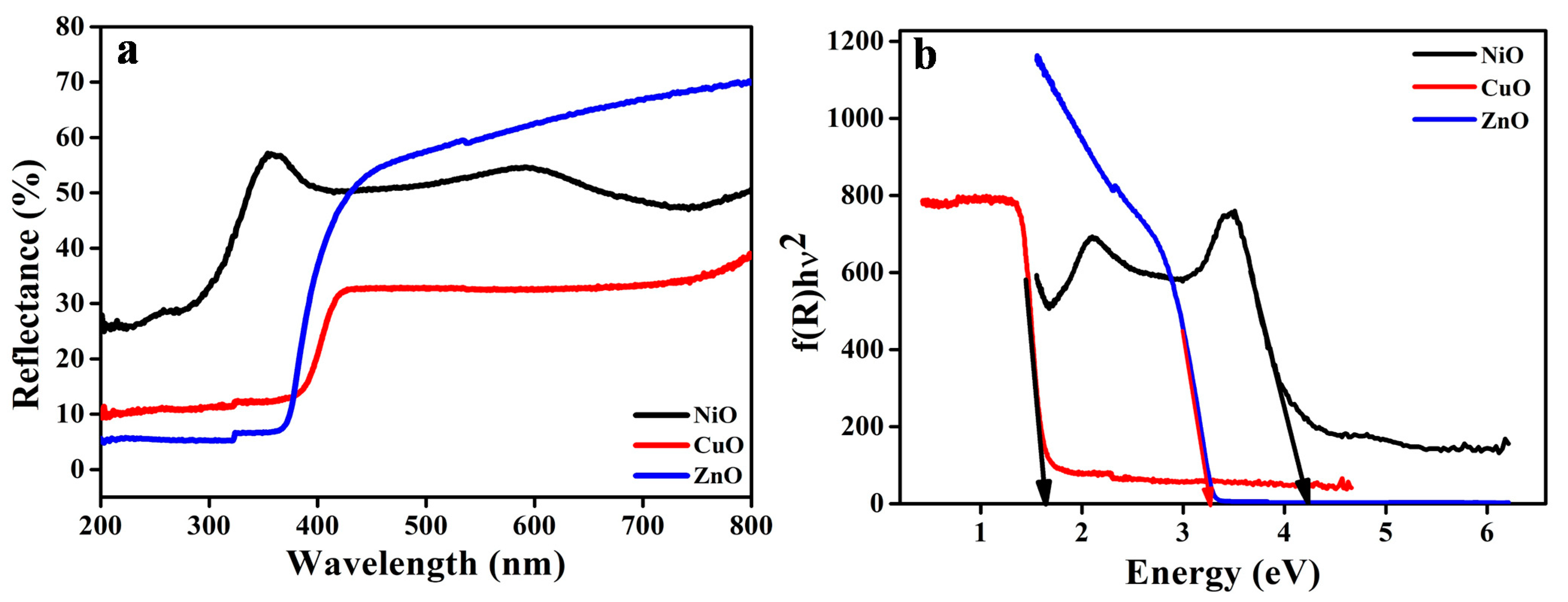
3.4. DRS Analysis
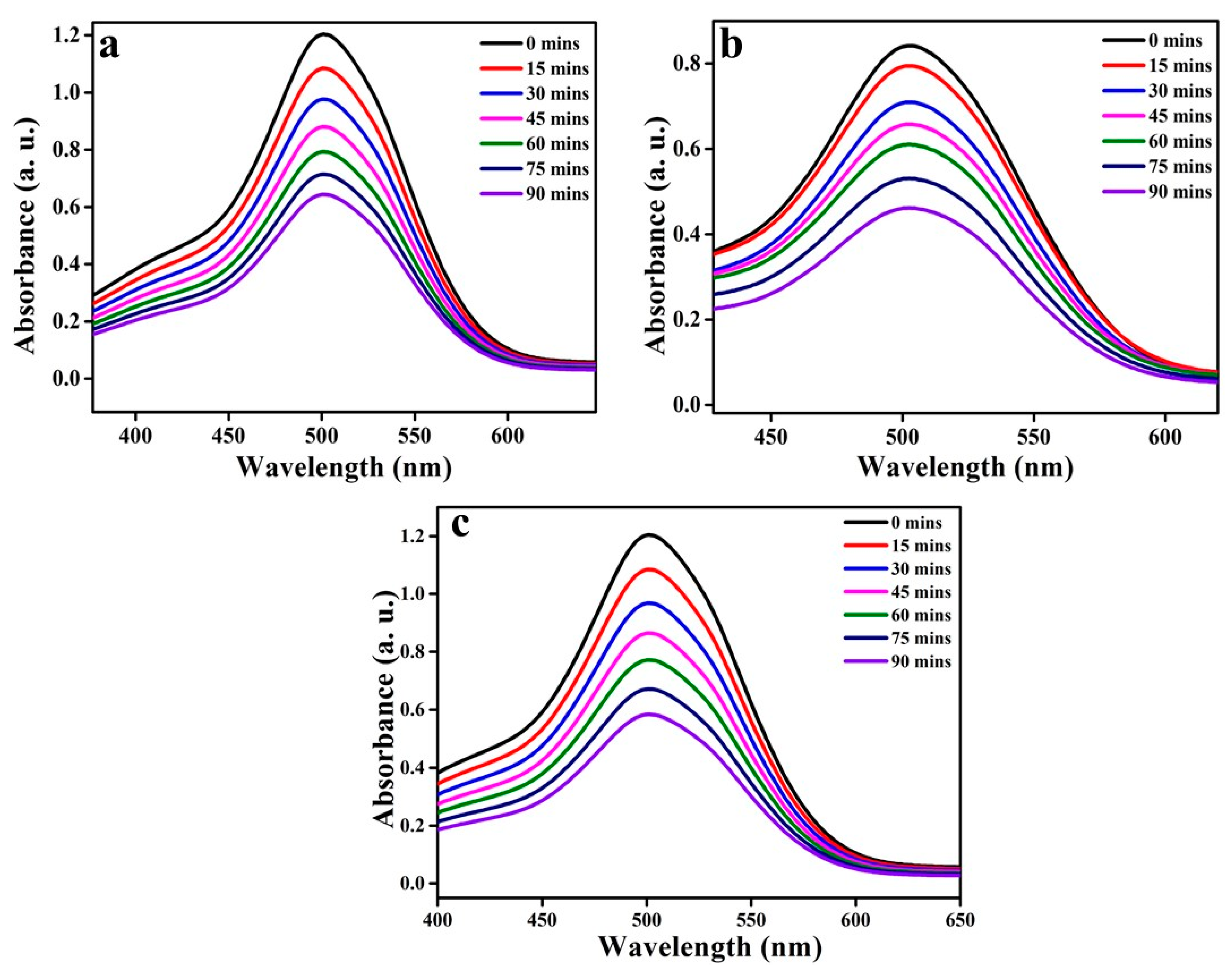
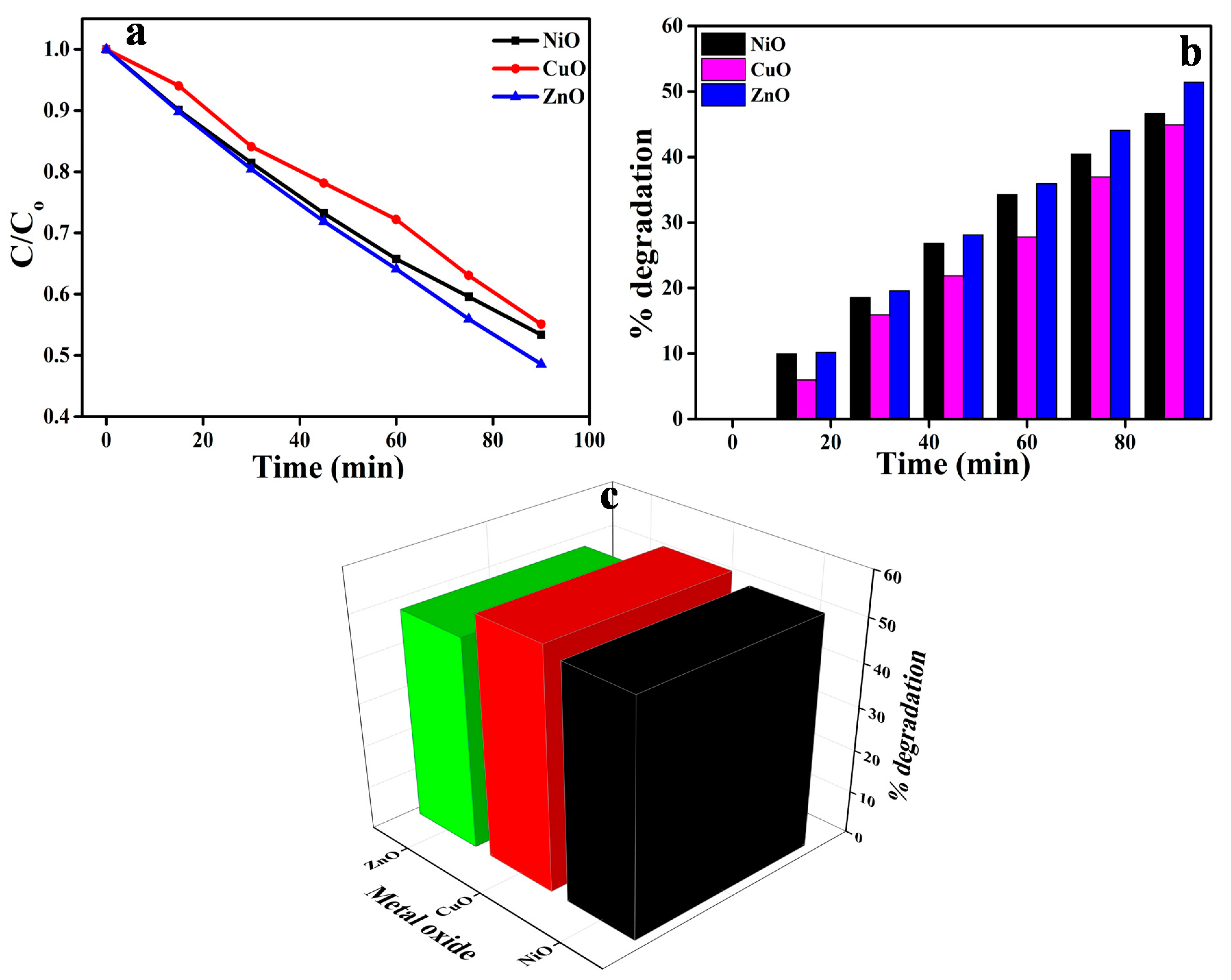
3.5. Photocatalytic Activity of NiO, CuO, and ZnO on AR88 Dye
3.6. Mechanism of Photodegradation
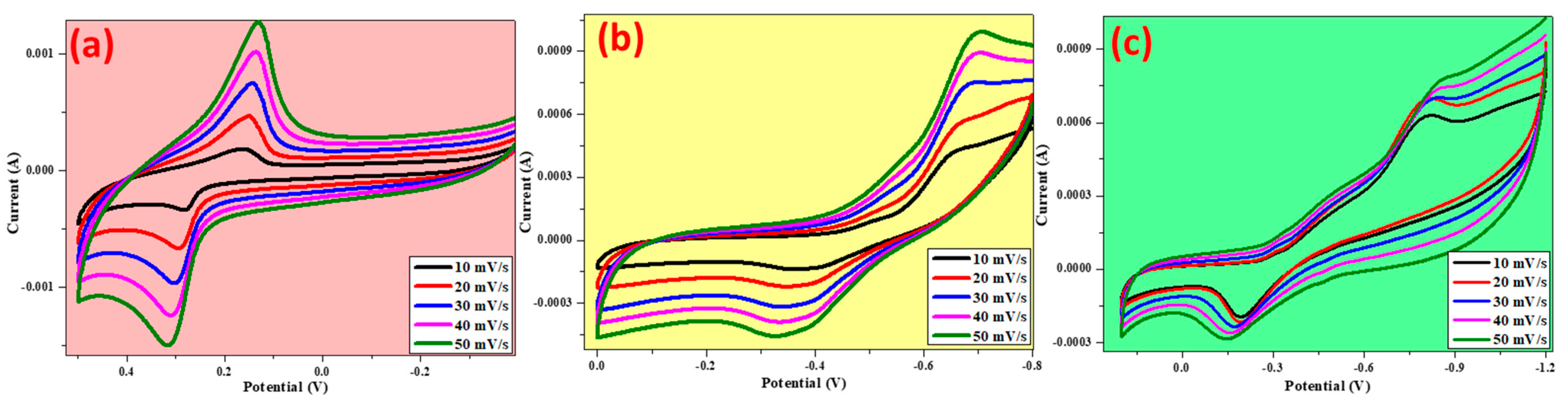
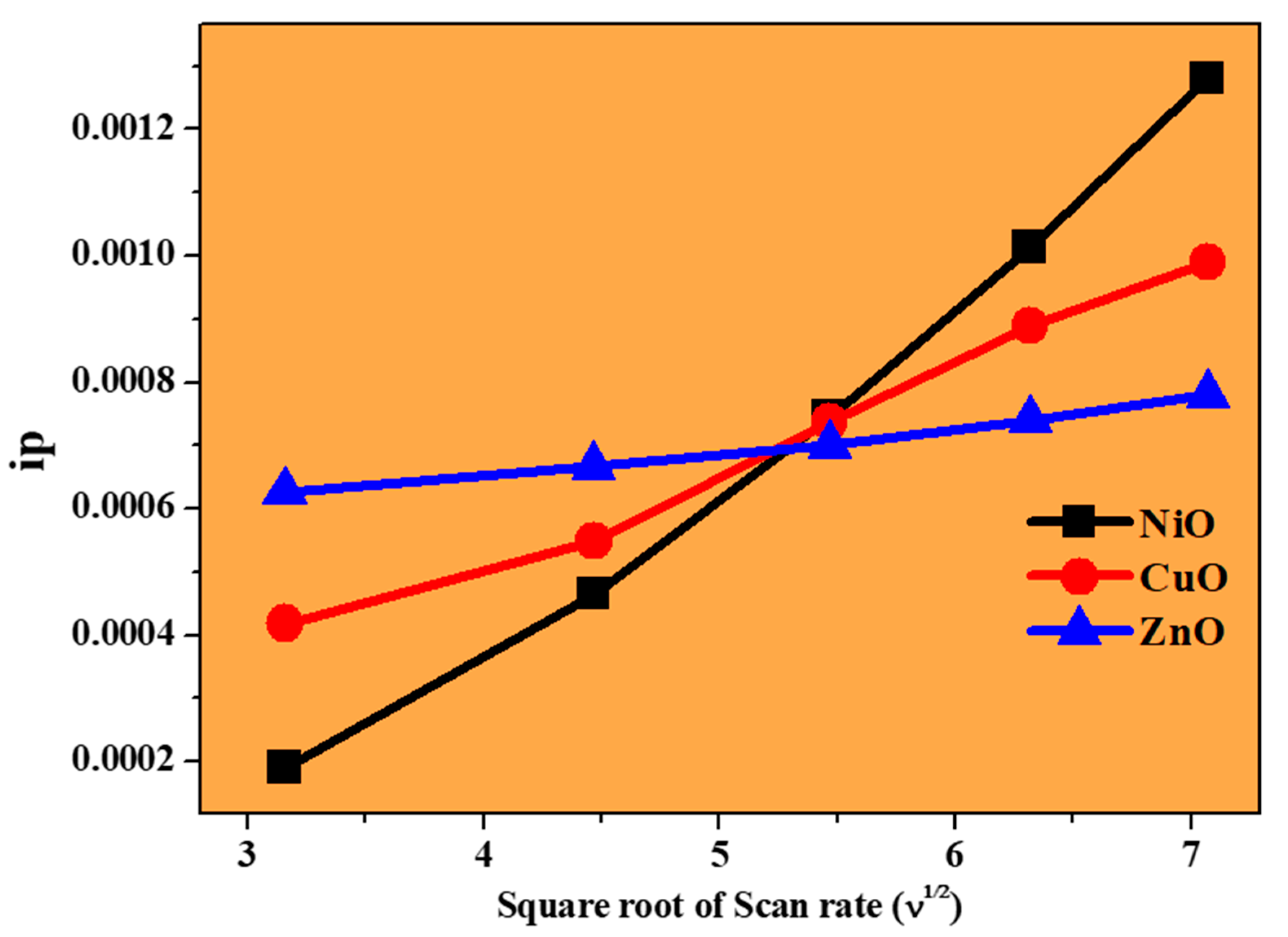
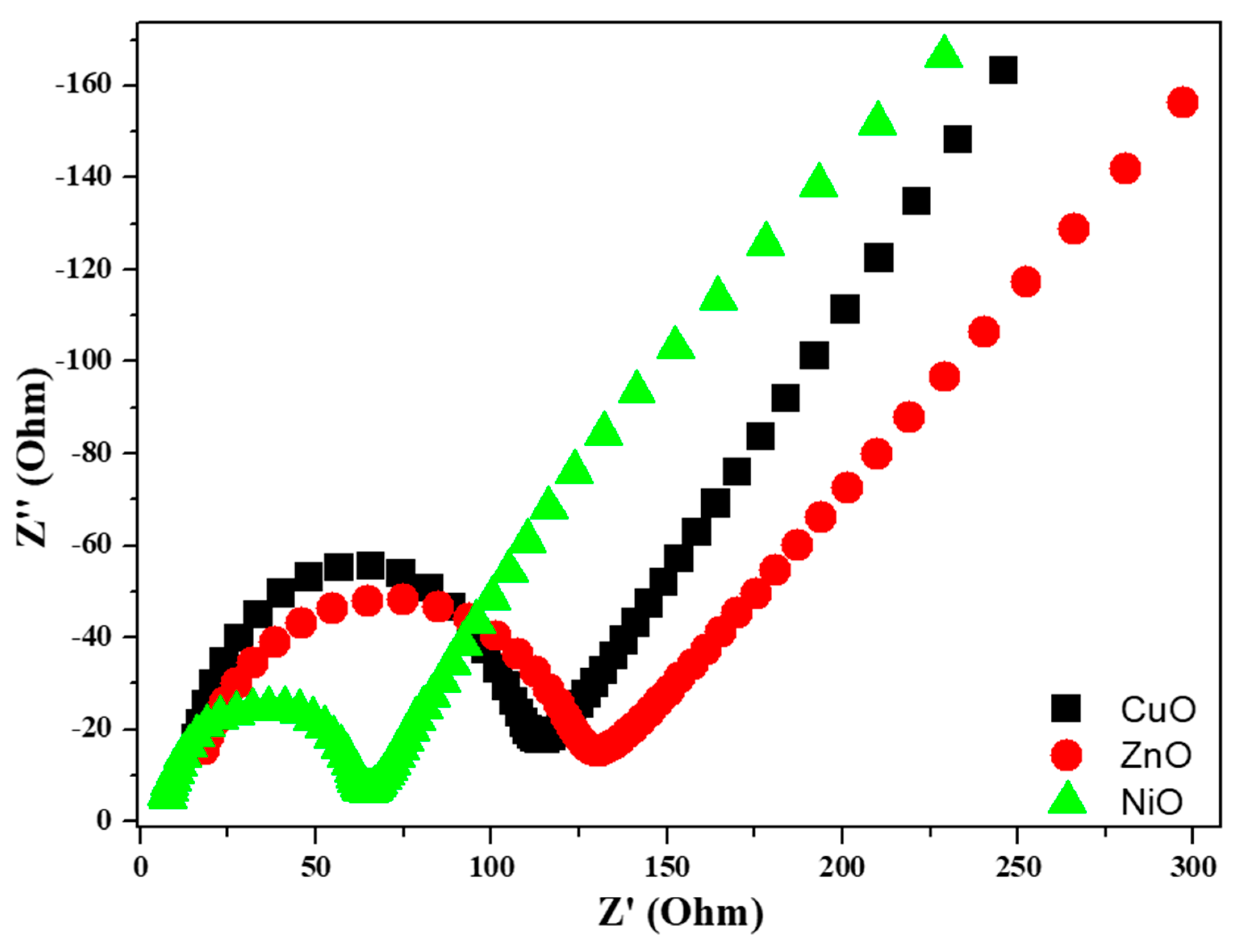
3.7. Electrochemical Studies
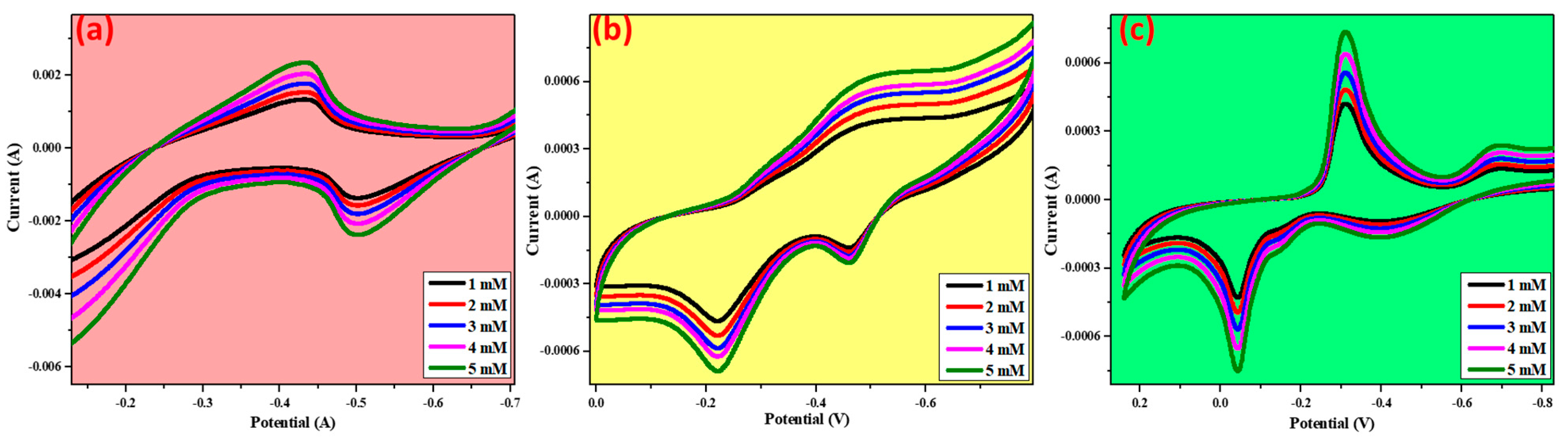
3.8. Glucose Sensing
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- GLASS; WHO. Investing in Water and Sanitation: Increasing Access, Reducing Inequalities, UN-Water Global Analysis and Assessment of Sanitation and Drinking-Water, GLAAS 2014 Report (GLASS, 2014); WHO: Geneva, Switzerland, 2014. [Google Scholar]
- UNESCO. Water and Jobs: The UN World Water Development Report (UNESCO-WWAP, 2016); UNESCO: Paris, France, 2016. [Google Scholar]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 Photocatalysis: Mechanisms and Materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.N.; Jnaneshwara, D.; Nagabhushana, H.; Prashantha, S.; Chandrasekhar, M.; Ravikumar, C.; Kumar, M.A.; Basavaraju, N.; Shekhar, T.S.; Premkumar, H. Enhanced photoluminescence, electrochemical and photocatalytic activity of combustion synthesized La10Si6O27:Dy3+ nanophosphors. J. Sci. Adv. Mater. Devices 2021, 6, 49–57. [Google Scholar] [CrossRef]
- Vasudha, M.; Khan, A.A.; Bhumika, K.; Gayathri, D.; Nagaswarupa, H.; Shekhar, T.S.; Raghavendra, N.; Ravikumar, C.; Murthy, H.A.; Vinutha, K.; et al. Facile chemical synthesis of Ca3MgAl10O17 nanomaterials for photocatalytic and non-enzymatic sensor applications. Sens. Int. 2021, 2, 100082. [Google Scholar] [CrossRef]
- Ranjitha, R.; Meghana, K.N.; Kumar, V.G.D.; Bhatt, A.S.; Jayanna, B.K.; Ravikumar, C.R.; Santosh, M.S.; Madhyastha, H.; Sakai, K. Rapid photocatalytic degradation of cationic organic dyes using Li-doped Ni/NiO nanocomposites and their electrochemical performance. New J. Chem. 2021, 45, 796–809. [Google Scholar] [CrossRef]
- Avinash, B.; Ravikumar, C.; Kumar, M.A.; Santosh, M.; Pratapkumar, C.; Nagaswarupa, H.; Murthy, H.A.; Deshmukh, V.; Bhatt, A.S.; Jahagirdar, A.; et al. NiO bio-composite materials: Photocatalytic, electrochemical and supercapacitor applications. Appl. Surf. Sci. Adv. 2021, 3, 100049. [Google Scholar] [CrossRef]
- Abebe, B.; Ravikumar, C.; Zereffa, E.A.; Kumar, A.N.; Murthy, H.A. Photocatalytic and superior ascorbic acid sensor activities of PVA/Zn-Fe-Mn ternary oxide nanocomposite. Inorg. Chem. Commun. 2021, 123, 108343. [Google Scholar] [CrossRef]
- Amulya, M.S.; Nagaswarupa, H.P.; Kumar, M.A.; Ravikumar, C.R.; Kusuma, K.B. Enhanced photocatalytic and elec-trochemical properties of Cu doped NiMnFe2O4 nanoparticles synthesized via probe sonication method. Appl. Surf. Sci. 2020, 2, 100038. [Google Scholar] [CrossRef]
- Waqas, A.M.; Khatoon, U.; Qurashi, A. Synthesis and characterization of Cu-SnO2 nanoparticles deposited on glass using ultrasonic spray pyrolysis and their H2S sensing properties. Curr. Nanosci. 2012, 8, 919–924. [Google Scholar] [CrossRef]
- Kumar, V.D.; Balaji, K.; Viswanatha, R.; Ambika, G.; Roopa, R.; Basavaraja, B.; Chennabasappa, M.; Kumar, C.R.; Chen, Z.; Bui, X.-T.; et al. Visible light photodegradation of 2,4-dichlorophenol using nanostructured NaBiS2: Kinetics, cytotoxicity, antimicrobial and electrochemical studies of the photocatalyst. Chemosphere 2021, 287, 132174. [Google Scholar] [CrossRef]
- Pratapkumar, C. Structural, photo catalytic and electrochemical studies on facile combustion synthesized low-cost nano chromium (III) doped polycrystalline magnesium aluminate spinels. J. Sci. Adv. Mater. Devices 2021, 6, 462–471. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Solanki, P.R.; Kaushik, A.; Agrawal, V.V.; Malhotra, B.D. Nanostructured metal oxide-based biosensors. NPG Asia Mater. 2011, 3, 17–24. [Google Scholar] [CrossRef]
- Guo, T.; Yao, M.-S.; Lin, Y.-H.; Nan, C.-W. A comprehensive review on synthesis methods for transition-metal oxide nanostructures. CrystEngComm 2015, 17, 3551–3585. [Google Scholar] [CrossRef]
- Devan, R.S.; Patil, R.A.; Lin, J.-H.; Ma, Y.-R. One-Dimensional Metal-Oxide Nanostructures: Recent Developments in Synthesis, Characterization, and Applications. Adv. Funct. Mater. 2012, 22, 3326–3370. [Google Scholar] [CrossRef]
- Navyashree, G.R.; Nagabhushana, H.; Sunitha, D.V.; Yeshodamma, S. Synthesis of TiO2 nanoparticles by solution com-bustion technique using different plant extracts. Int. J. Eng. Res. 2016, 50, 790–991. [Google Scholar]
- Raj, N.B.; PavithraGowda, N.; Pooja, O.; Purushotham, B.; Kumar, M.A.; Sukrutha, S.; Ravikumar, C.; Nagaswarupa, H.; Murthy, H.A.; Boppana, S.B. Harnessing ZnO nanoparticles for antimicrobial and photocatalytic activities. J. Photochem. Photobiol. 2021, 6, 100021. [Google Scholar] [CrossRef]
- Saravanakkumar, D. Synthesis of NiO doped ZnO/MWCNT nanocomposite and its characterization for photocatalytic & antimicrobial applications. J. Appl. Phys. 2018, 10, 73–83. [Google Scholar]
- Ethiraj, A.S.; Kang, D.J. Synthesis and characterization of CuO nanowires by a simple wet chemical method. Nanoscale Res. Lett. 2012, 7, 70. [Google Scholar] [CrossRef] [Green Version]
- Nayak, R.; Ali, F.A.; Mishra, D.K.; Ray, D.; Aswal, V.; Sahoo, S.K.; Nanda, B. Fabrication of CuO nanoparticle: An efficient catalyst utilized for sensing and degradation of phenol. J. Mater. Res. Technol. 2020, 9, 11045–11059. [Google Scholar] [CrossRef]
- Kumar, M.R.A.; Abebe, B.; Nagaswarupa, H.P.; Murthy, H.C.A.; Ravikumar, C.R.; Sabir, F.K. Enhanced photocatalytic and electrochemical performance of TiO2-Fe2O3 nanocomposite: Its applications in dye decolorization and as supercapacitors. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Kusuma, K.; Manju, M.; Ravikumar, C.; Nagaswarupa, H.; Amulya, M.S.; Anilkumar, M.; Avinash, B.; Gurushantha, K.; Ravikantha, N. Photocatalytic and electrochemical sensor for direct detection of paracetamol comprising γ-aluminium oxide nanoparticles synthesized via sonochemical route. Sens. Int. 2020, 1, 100039. [Google Scholar] [CrossRef]
- Ravikumar, C.; Kumar, M.A.; Nagaswarupa, H.; Prashantha, S.; Bhatt, A.S.; Santosh, M.; Kuznetsov, D. CuO embedded β-Ni(OH) 2 nanocomposite as advanced electrode materials for supercapacitors. J. Alloy. Compd. 2018, 736, 332–339. [Google Scholar] [CrossRef]
- Kumar, C.R. Synthesis and characterization of β-Ni(OH)2 embedded with MgO and ZnO nanoparticles as nanohybrids for energy storage devices. Mater. Res. Express 2017, 4, 065503. [Google Scholar] [CrossRef]
- He, Y.; Chen, W.; Li, X.; Zhang, Z.; Fu, J.; Zhao, C.; Xie, E. Freestanding Three-Dimensional Graphene/MnO2 Composite Networks as Ultralight and Flexible Supercapacitor Electrodes. ACS Nano 2013, 7, 174–182. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods, Fundamentals and Applications, 2nd ed.; Wiley: New York, NY, USA, 2001. [Google Scholar]
- Viswanathan, V.V.; Salkind, A.J.; Kelley, J.J.; Ockerman, J.B. Effect of state of charge on impedance spectrum of sealed cells Part II: Lead acid batteries. J. Appl. Electrochem. 1995, 25, 729–739. [Google Scholar] [CrossRef]
- Ashwini, R.; Dileepkumar, V.; Balaji, K.; Viswanatha, R.; Ravikumar, C.; Srivastava, C.; Santosh, M.S. Ternary alkali metal chalcogenide engineered reduced graphene oxide (rGO) as a new class of composite (NaFeS2-rGO) and its electrochemical performance. Sens. Int. 2021, 2, 100125. [Google Scholar] [CrossRef]
- Wang, X.; Hu, C.; Liu, H.; Du, G.; He, X.; Xi, Y. Synthesis of CuO nanostructures and their application for nonenzymatic glucose sensing. Sens. Actuators B Chem. 2010, 144, 220–225. [Google Scholar] [CrossRef]
- Ibupoto, Z.H.; Khun, K.; Lu, J.; Willander, M. The synthesis of CuO nanoleaves, structural characterization, and their glucose sensing application. Appl. Phys. Lett. 2013, 102, 103701. [Google Scholar] [CrossRef]
- Li, R.; Liu, X.; Wang, H.; Wu, Y.; Chan, K.; Lu, Z. Sandwich nanoporous framework decorated with vertical CuO nanowire arrays for electrochemical glucose sensing. Electrochim. Acta 2019, 299, 470–478. [Google Scholar] [CrossRef]
- Mishra, S.; Yogi, P.; Sagdeo, P.R.; Kumar, R. Mesoporous Nickel Oxide (NiO) Nanopetals for Ultrasensitive Glucose Sensing. Nanoscale Res. Lett. 2018, 13, 16. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Xie, Y.; Wei, C.; Lu, X.; Li, X.; Song, Y. Hierarchical NiO Superstructures/Foam Ni Electrode Derived from Ni Metal-Organic Framework Flakes on Foam Ni for Glucose Sensing. Electrochim. Acta 2015, 174, 846–852. [Google Scholar] [CrossRef]
- Kavitha, T.; Gopalan, A.I.; Lee, K.-P.; Park, S.-Y. Glucose sensing, photocatalytic and antibacterial properties of graphene–ZnO nanoparticle hybrids. Carbon 2012, 50, 2994–3000. [Google Scholar] [CrossRef]
- Kim, J.Y.; Jo, S.-Y.; Sun, G.-J.; Katoch, A.; Choi, S.-W.; Kim, S.S. Tailoring the surface area of ZnO nanorods for improved performance in glucose sensors. Sens. Actuators B Chem. 2014, 192, 216–220. [Google Scholar] [CrossRef]
- Ahmad, R.; Tripathy, N.; Khan, M.Y.; Bhat, K.S.; Ahn, M.-S.; Khang, G.; Hahn, Y.-B. Hierarchically assembled ZnO nanosheets microspheres for enhanced glucose sensing performances. Ceram. Int. 2016, 42, 13464–13469. [Google Scholar] [CrossRef]
- Dhananjaya, N.; Ambujakshi, N.; Raveesha, H.; Pratibha, S.; Ravikumar, C. Electrochemical Sensor and luminescence applications of Chonemorpha fragrans leaf extract mediated ZnO/Ag nanostructures. Appl. Surf. Sci. Adv. 2021, 4, 100075. [Google Scholar] [CrossRef]
- Avinash, B.; Ravikumar, C.; Kumar, M.A.; Nagaswarupa, H.; Santosh, M.; Bhatt, A.S.; Kuznetsov, D. Nano CuO: Electrochemical sensor for the determination of paracetamol and d-glucose. J. Phys. Chem. Solids 2019, 134, 193–200. [Google Scholar] [CrossRef]
- Girish, K.; Prashantha, S.; Nagabhushana, H.; Ravikumar, C.; Nagaswarupa, H.; Naik, R.; Premakumar, H.; Umesh, B. Multi-functional Zn2TiO4: Sm3+ nanopowders: Excellent performance as an electrochemical sensor and an UV photocatalyst. J. Sci. Adv. Mater. Devices 2018, 3, 151–160. [Google Scholar] [CrossRef]
- Manjunatha, A.; Pavithra, N.; Shivanna, M.; Nagaraju, G.; Ravikumar, C. Synthesis of Citrus Limon mediated SnO2-WO3 nanocomposite: Applications to photocatalytic activity and electrochemical sensor. J. Environ. Chem. Eng. 2020, 8, 104500. [Google Scholar] [CrossRef]



| Metal Oxides | Average Crystallite Size (nm) |
|---|---|
| NiO | 30–50 |
| CuO | 30–60 |
| ZnO | 25–40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, M.W.; Aamir, M.; Farhan, M.; Albuhulayqah, M.; Ahmad, M.M.; Ravikumar, C.R.; Dileep Kumar, V.G.; Ananda Murthy, H.C. Green Synthesis of Ni-Cu-Zn Based Nanosized Metal Oxides for Photocatalytic and Sensor Applications. Crystals 2021, 11, 1467. https://doi.org/10.3390/cryst11121467
Alam MW, Aamir M, Farhan M, Albuhulayqah M, Ahmad MM, Ravikumar CR, Dileep Kumar VG, Ananda Murthy HC. Green Synthesis of Ni-Cu-Zn Based Nanosized Metal Oxides for Photocatalytic and Sensor Applications. Crystals. 2021; 11(12):1467. https://doi.org/10.3390/cryst11121467
Chicago/Turabian StyleAlam, Mir Waqas, Muhammad Aamir, Mohd Farhan, Maryam Albuhulayqah, Mohamad M. Ahmad, C.R. Ravikumar, V.G. Dileep Kumar, and H.C. Ananda Murthy. 2021. "Green Synthesis of Ni-Cu-Zn Based Nanosized Metal Oxides for Photocatalytic and Sensor Applications" Crystals 11, no. 12: 1467. https://doi.org/10.3390/cryst11121467
APA StyleAlam, M. W., Aamir, M., Farhan, M., Albuhulayqah, M., Ahmad, M. M., Ravikumar, C. R., Dileep Kumar, V. G., & Ananda Murthy, H. C. (2021). Green Synthesis of Ni-Cu-Zn Based Nanosized Metal Oxides for Photocatalytic and Sensor Applications. Crystals, 11(12), 1467. https://doi.org/10.3390/cryst11121467








