Oxygen Inhomogeneity and Reversibility in Single Crystal LaNiO3−δ
Abstract
1. Introduction
2. Experimental Details
2.1. Crystal Growth
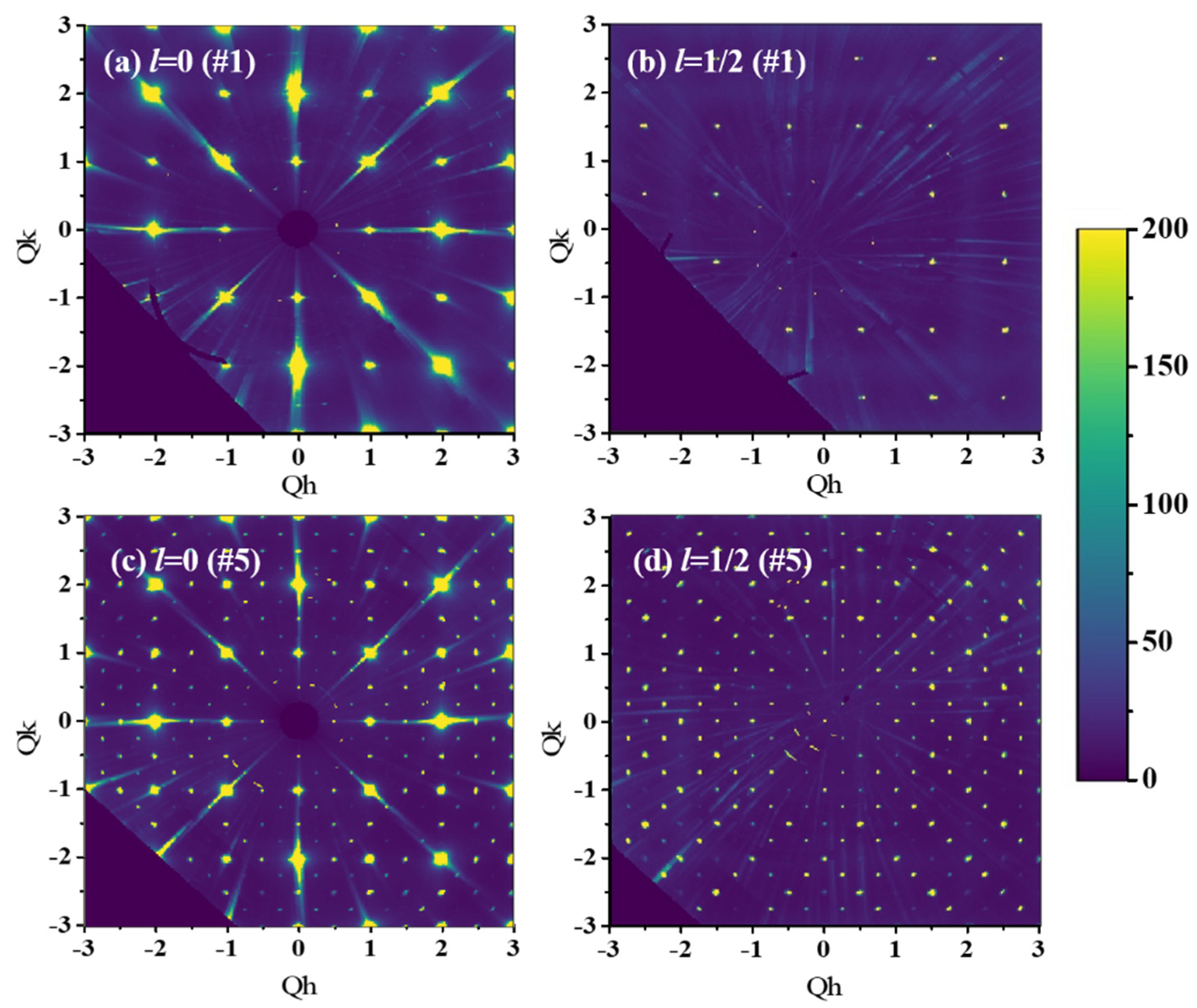
2.2. Single Crystal X-Ray Diffraction
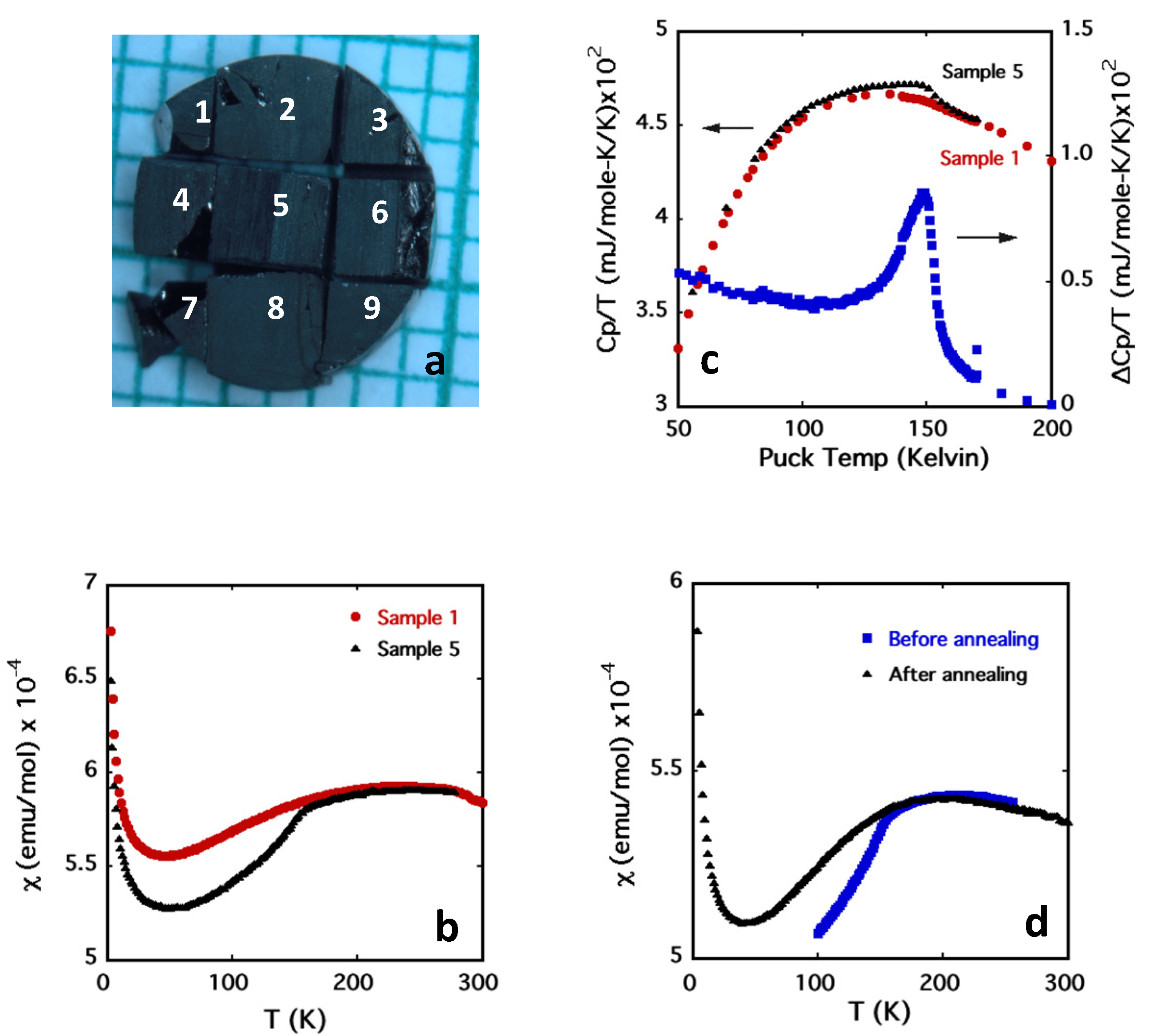
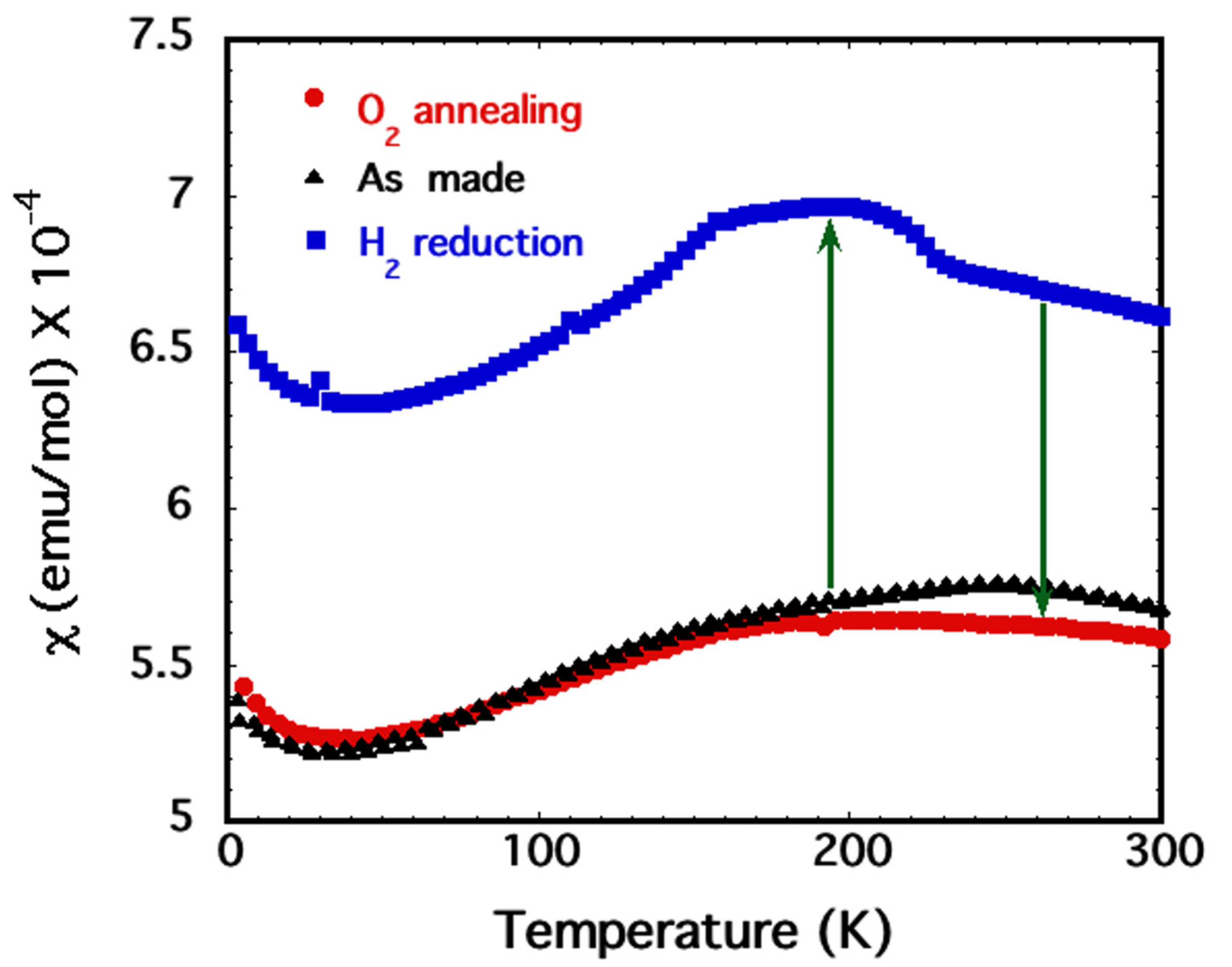
2.3. Magnetic Susceptibility
2.4. Heat Capacity
2.5. High Pressure Oxygen Annealing
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mazin, I.I.; Khomskii, D.I.; Lengsdorf, R.; Alonso, J.A.; Marshall, W.G.; Ibberson, R.M.; Abd-Elmeguid, M.M. Charge ordering as alternative to Jahn-Teller distortion. Phys. Rev. Lett. 2007, 98, 176406. [Google Scholar] [CrossRef]
- Zhou, J.S.; Marshall, L.G.; Goodenough, J.B. Mass enhancement versus Stoner enhancement in strongly correlated metallic perovskites: LaNiO3 and LaCuO3. Phys. Rev. B 2014, 89, 245138. [Google Scholar] [CrossRef]
- Disa, A.S.; Walker, F.J.; Ismail-Beigi, S.; Ahn, C.H. Research Update: Orbital polarization in LaNiO3-based heterostructures. APL Mater. 2015, 3, 062303. [Google Scholar] [CrossRef]
- Medarde, M.; Fernández-Díaz, M.T.; Lacorre, P. Long-range charge order in the low-temperature insulating phase of PrNiO3. Phys. Rev. B 2008, 78, 212101. [Google Scholar] [CrossRef]
- Allen, S.J.; Hauser, A.J.; Mikheev, E.; Zhang, J.Y.; Moreno, N.E.; Son, J.; Stemmer, S. Gaps and pseudogaps in perovskite rare earth nickelates. APL Mater. 2015, 3, 062503. [Google Scholar] [CrossRef]
- Lee, S.; Chen, R.; Balents, L. Metal-insulator transition in a two-band model for the perovskite nickelates. Phys. Rev. B 2011, 84, 165119. [Google Scholar] [CrossRef]
- Green, R.J.; Haverkort, M.W.; Sawatzky, G.A. Bond disproportionation and dynamical charge fluctuations in the perovskite rare-earth nickelates. Phys. Rev. B 2016, 94, 195127. [Google Scholar] [CrossRef]
- Park, H.; Millis, A.J.; Marianetti, C.A. Site-selective Mott transition in rare-earth-element nickelates. Phys. Rev. Lett. 2012, 109, 156402. [Google Scholar] [CrossRef]
- Johnston, S.; Mukherjee, A.; Elfimov, I.; Berciu, M.; Sawatzky, G.A. Charge disproportionation without charge transfer in the rare-earth-element nickelates as a possible mechanism for the metal-insulator transition. Phys. Rev. Lett. 2014, 112, 106404. [Google Scholar] [CrossRef]
- Bisogni, V.; Catalano, S.; Green, R.J.; Gibert, M.; Scherwitzl, R.; Huang, Y.; Sawatzky, G. Ground-state oxygen holes and the metal–insulator transition in the negative charge-transfer rare-earth nickelates. Nat. Commun. 2016, 7, 1–8. [Google Scholar] [CrossRef]
- Mizokawa, T.; Khomskii, D.; Sawatzky, G. Spin and charge ordering in self-doped Mott insulators. Phys. Rev. B 2000, 61, 11263. [Google Scholar] [CrossRef]
- Varignon, J.; Grisolia, M.N.; Iniguez, J.; Barthélémy, A.; Bibes, M. Complete phase diagram of rare-earth nickelates from first-principles. NPJ Quantum Mater. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Shamblin, J.; Heres, M.; Zhou, H.; Sangoro, J.; Lang, M.; Neuefeind, J.; Johnston, S. Experimental evidence for bipolaron condensation as a mechanism for the metal-insulator transition in rare-earth nickelates. Nat. Commun. 2018, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Louca, D.; Yano, S.; Marshall, L.G.; Zhou, J.; Goodenough, J.B. Insulating pockets in metallic LaNiO3. Adv. Electron. Mater. 2016, 2, 1500261. [Google Scholar] [CrossRef]
- Torrance, J.B.; Lacorre, P.; Nazzal, A.I.; Ansaldo, E.J.; Niedermayer, C. Systematic study of insulator-metal transitions in perovskites RNiO3 (R = Pr, Nd, Sm, Eu) due to closing of charge-transfer gap. Phy. Rev. B 1992, 45, 8209. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. Sect. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Raccah, P.M. Complex vs band formation in perovskite oxides. J. Appl. Phys. 1965, 36, 1031–1032. [Google Scholar] [CrossRef]
- Sreedhar, K.; Honig, J.M.; Darwin, M.; McElfresh, M.; Shand, P.M.; Xu, J.; Spalek, J. Electronic properties of the metallic perovskite LaNiO3: Correlated behavior of 3d electrons. Phys. Rev. B 1992, 46, 6382. [Google Scholar] [CrossRef]
- Vasanthacharya, N.Y.; Ganguly, P.; Goodenough, J.B.; Rao, C.N.R. Valence states and magnetic properties of LaNi1−xMnxO3 (for 0 ⩽ x ⩽ 0.2 and x = 0.5). J. Phy. C Solid State Phys. 1984, 17, 2745. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, H.; Ren, Y.; Mitchell, J.F. High-pressure floating-zone growth of perovskite nickelate LaNiO3 single crystals. Cryst. Growth Des. 2017, 17, 2730–2735. [Google Scholar] [CrossRef]
- Meng, N.; Ren, X.; Santagiuliana, G.; Ventura, L.; Zhang, H.; Wu, J.; Bilotti, E. Ultrahigh β-phase content poly (vinylidene fluoride) with relaxor-like ferroelectricity for high energy density capacitors. Nat. Commun. 2019, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dey, K.; Hergett, W.; Telang, P.; Abdel-Hafiez, M.M.; Klingeler, R. Magnetic properties of high-pressure optical floating-zone grown LaNiO3 single crystals. J. Cryst. Growth 2019, 524, 125157. [Google Scholar] [CrossRef]
- Zheng, H.; Zhang, J.; Wang, B.; Phelan, D.; Krogstad, M.J.; Ren, Y.; Mitchell, J.F. High pO2 Floating Zone Crystal Growth of the Perovskite Nickelate PrNiO3. Crystals 2019, 9, 324. [Google Scholar] [CrossRef]
- Wang, B.X.; Rosenkranz, S.; Rui, X.; Zhang, J.; Ye, F.; Zheng, H.; Phelan, D. Antiferromagnetic defect structure in LaNiO3−δ single crystals. Phys. Rev. Mater. 2018, 2, 064404. [Google Scholar] [CrossRef]
- Moriga, T.; Usaka, O.; Nakabayashi, I.; Hirashima, Y.; Kohno, T.; Kikkawa, S.; Kanamaru, F. Reduction of the perovskite-type LnNiO3 (Ln = Pr, Nd) to Ln3Ni3O7 with monovalent nickel ions. Solid State Ionics 1994, 74, 211. [Google Scholar] [CrossRef]
- Moriga, T.; Usaka, O.; Imamura, T.; Nakabayashi, I.; Matsubara, I.; Kinouchi, T.; Kanamaru, F. Synthesis, Crystal Structure, and Properties of Oxygen-Deficient Lanthanum Nickelate LaNiO3-x (0 ≤ x ≤ 0.5). Bull. Chem. Soc. Jpn. 1994, 67, 687–693. [Google Scholar] [CrossRef]
- Zhang, J.; Phelan, D.; Botana, A.S.; Chen, Y.S.; Zheng, H.; Krogstad, M.; Rosenkranz, S. Intertwined density waves in a metallic nickelate. arXiv 2020, arXiv:2004.07897. [Google Scholar]
- Abbate, M.; Zampieri, G.; Prado, F.; Caneiro, A.; Gonzalez-Calbet, J.M.; Vallet-Regi, M. Electronic structure and metal-insulator transition in LaNiO3−δ. Phys. Rev. B 2002, 65, 155101. [Google Scholar] [CrossRef]
- Alonso, J.A.; Martínez-Lope, M.J. Preparation and crystal structure of the deficient perovskite LaNiO2.5, solved from neutron powder diffraction data. J. Chem. Soc. Dalton Trans. 1995, 17, 2819–2824. [Google Scholar] [CrossRef]
- Alonso, J.A.; Martínez-Lope, M.J.; García-Muñoz, J.L.; Fernández-Díaz, M.T. A structural and magnetic study of the defect perovskite from high-resolution neutron diffraction data. J. Phys. Condens. Matter. 1997, 9, 6417. [Google Scholar] [CrossRef]
- Crespin, M.; Levitz, P.; Gatineau, L. Reduced Forms of LaNi03 Perovskite Part 1.-Evidence for New Phases: La2Ni2O5 and LaNiO2. J. Chem. Soc. Faraday Trans. 2 1983, 79, 1181–1194. [Google Scholar] [CrossRef]
- Gonzalez-Calbet, J.M.; Sayagues, M.J.; Vallet-Regi, M. An electron diffraction study of new phases in the LaNiO3−x system. Solid State Ionics. 1989, 32, 721726. [Google Scholar] [CrossRef]
- Levitz, P.; Crespin, M.; Gatineau, L. Reduced forms of LaNiO3 perovskite. Part 2.—X-ray structure of LaNiO2 and extended X-ray absorption fine structure study: Local environment of monovalent nickel. J. Chem. Soc. Faraday Trans. 1983, 79, 1195. [Google Scholar] [CrossRef]
- Middey, S.; Rivero, P.; Meyers, D.; Kareev, M.; Liu, X.; Cao, Y.; Chakhalian, J. Polarity compensation in ultra-thin films of complex oxides: The case of a perovskite nickelate. Sci. Rep. 2014, 4, 1–7. [Google Scholar] [CrossRef]
- Moriga, T.; Kikkawa, S.; Takahashi, M.; Kanamaru, F.; Nakabayashi, I. XAFS study on reduction process of Pauli-paramagnetic LaNiO3 to antiferromagnetic La2Ni2O5. Jpn. J. Appl. Phys. 1993, 32, 764. [Google Scholar] [CrossRef]
- Moriga, T.; Usaka, O.; Nakabayashi, I.; Kinouchi, T.; Kikkawa, S.; Kanamaru, F. Characterization of oxygen- deficient phases appearing in reduction of the perovskite-type LaNiO3 to La2Ni2O5. Solid State Ionics. 1995, 79, 252. [Google Scholar] [CrossRef]
- Sanchez, R.D.; Causa, M.T.; Caneiro, A.; Butera, A.; Vallet-Regi, M.; Sayagues, M.J.; Rivas, J. Metal-insulator transition in oxygen-deficient LaNiO3−x perovskites. Phys. Rev. B 1996, 54, 16574. [Google Scholar] [CrossRef]
- Rao, C.N.R.; Gopalakrishnan, J.; Vidyasagar, K.; Ganguli, A.K.; Ramanan, A.; Ganapathi, L. Novel metal oxides prepared by ingenious synthetic routes. J. Mater. Res. 1986, 1, 280. [Google Scholar] [CrossRef]
- Vidyasagar, K.; Reller, A.; Gopalakrishnan, J.; Rao, C.N.R. Oxygen Vacancy Ordering in Superlattices of the Two Novel Oxides, La2Ni2O5 and La2Co2O5, prepared by Low Temperature Reduction of the Parent Perovskites. J. Chem. Soc. Chem. Commun. 1985, 1, 7–8. [Google Scholar] [CrossRef]
- Phelan, D.; Long, X.; Xie, Y.; Ye, Z.G.; Glazer, A.M.; Yokota, H.; Gehring, P.M. Single crystal study of competing rhombohedral and monoclinic order in lead zirconate titanate. Phys. Rev. Lett. 2010, 105, 207601. [Google Scholar] [CrossRef]
- Phelan, D. Constraints on the possible long-range orbital ordering in LaCoO3. J. Magn. Magn. Mater. 2014, 350, 183. [Google Scholar] [CrossRef]
- Shirane, G.; Shapiro, S.M.; Tranquada, J.M. Neutron Scattering with a Triple-Axis Spectrometer: Basic Techniques; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Ge, W.; Devreugd, C.P.; Phelan, D.; Zhang, Q.; Ahart, M.; Li, J.; Gehring, P.M. Lead-free and lead-based ABO3 perovskite relaxors with mixed-valence A-site and B-site disorder: Comparative neutron scattering structural study of (Na1/2Bi1/2) TiO3 and Pb(Mg1/3Nb2/3)O3. Phys. Rev. B 2013, 88, 174115. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, H.; Wang, B.-X.; Phelan, D.; Zhang, J.; Ren, Y.; Krogstad, M.J.; Rosenkranz, S.; Osborn, R.; Mitchell, J.F. Oxygen Inhomogeneity and Reversibility in Single Crystal LaNiO3−δ. Crystals 2020, 10, 557. https://doi.org/10.3390/cryst10070557
Zheng H, Wang B-X, Phelan D, Zhang J, Ren Y, Krogstad MJ, Rosenkranz S, Osborn R, Mitchell JF. Oxygen Inhomogeneity and Reversibility in Single Crystal LaNiO3−δ. Crystals. 2020; 10(7):557. https://doi.org/10.3390/cryst10070557
Chicago/Turabian StyleZheng, Hong, Bi-Xia Wang, D. Phelan, Junjie Zhang, Yang Ren, M. J. Krogstad, S. Rosenkranz, R. Osborn, and J. F. Mitchell. 2020. "Oxygen Inhomogeneity and Reversibility in Single Crystal LaNiO3−δ" Crystals 10, no. 7: 557. https://doi.org/10.3390/cryst10070557
APA StyleZheng, H., Wang, B.-X., Phelan, D., Zhang, J., Ren, Y., Krogstad, M. J., Rosenkranz, S., Osborn, R., & Mitchell, J. F. (2020). Oxygen Inhomogeneity and Reversibility in Single Crystal LaNiO3−δ. Crystals, 10(7), 557. https://doi.org/10.3390/cryst10070557






