Colossal Negative Magnetoresistance Effect in a La1.37Sr1.63Mn2O7 Single Crystal Grown by Laser-Diode-Heated Floating-Zone Technique
Abstract
1. Introduction
2. Results and Discussion
2.1. Single Crystal Growth
2.2. Structure Study
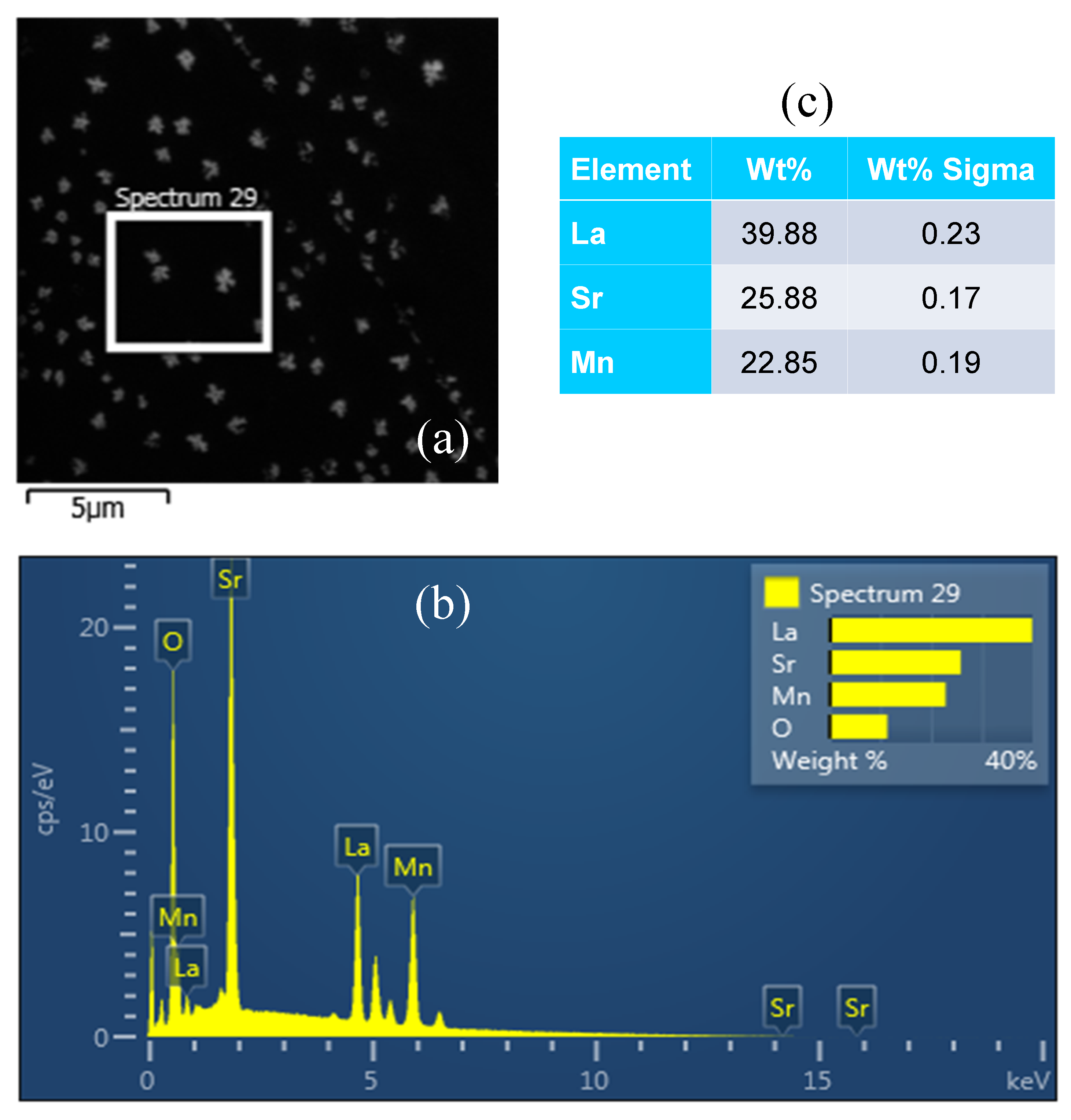
2.3. Scanning Electronic Microscopy
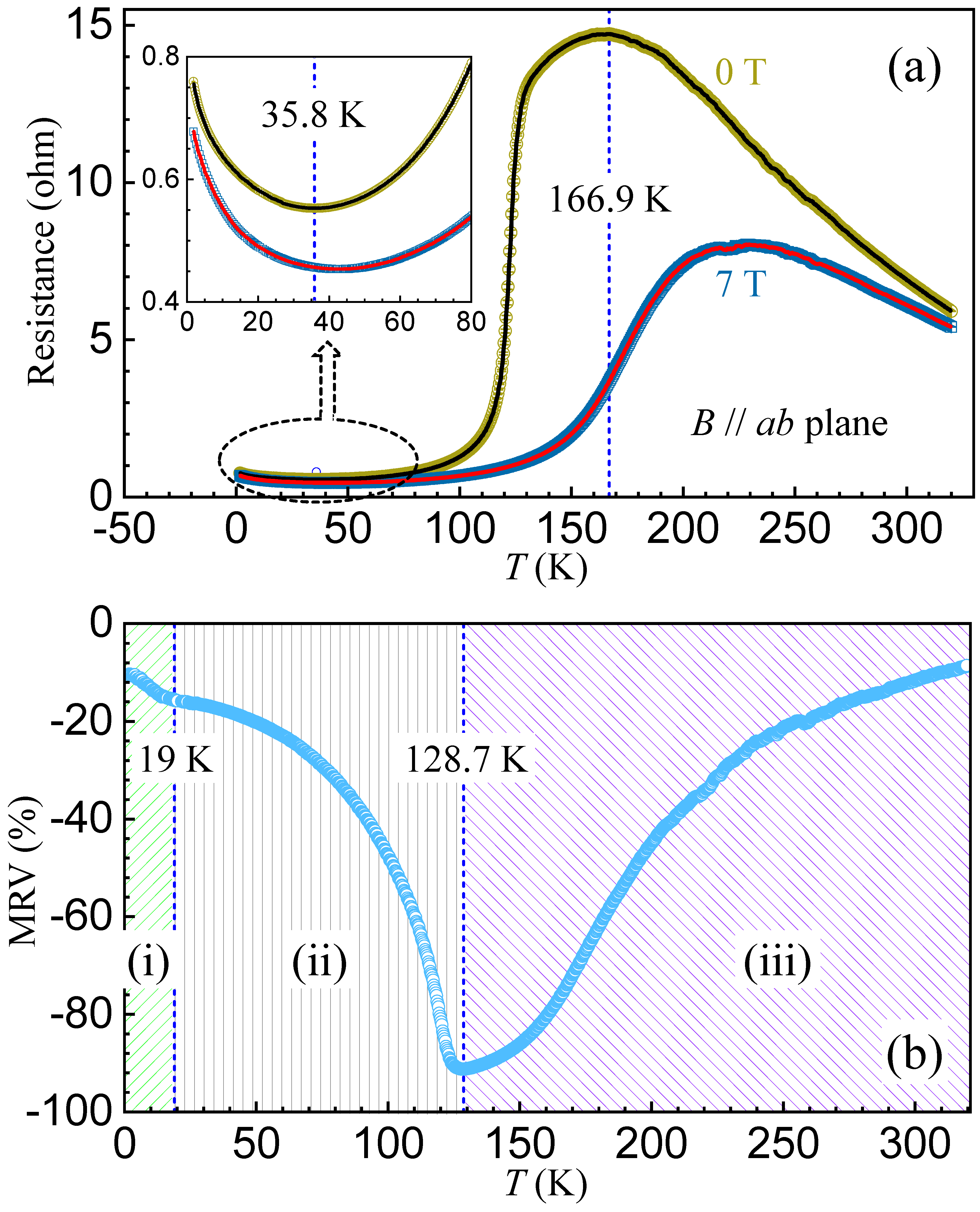
2.4. Magnetoresistance versus Temperature
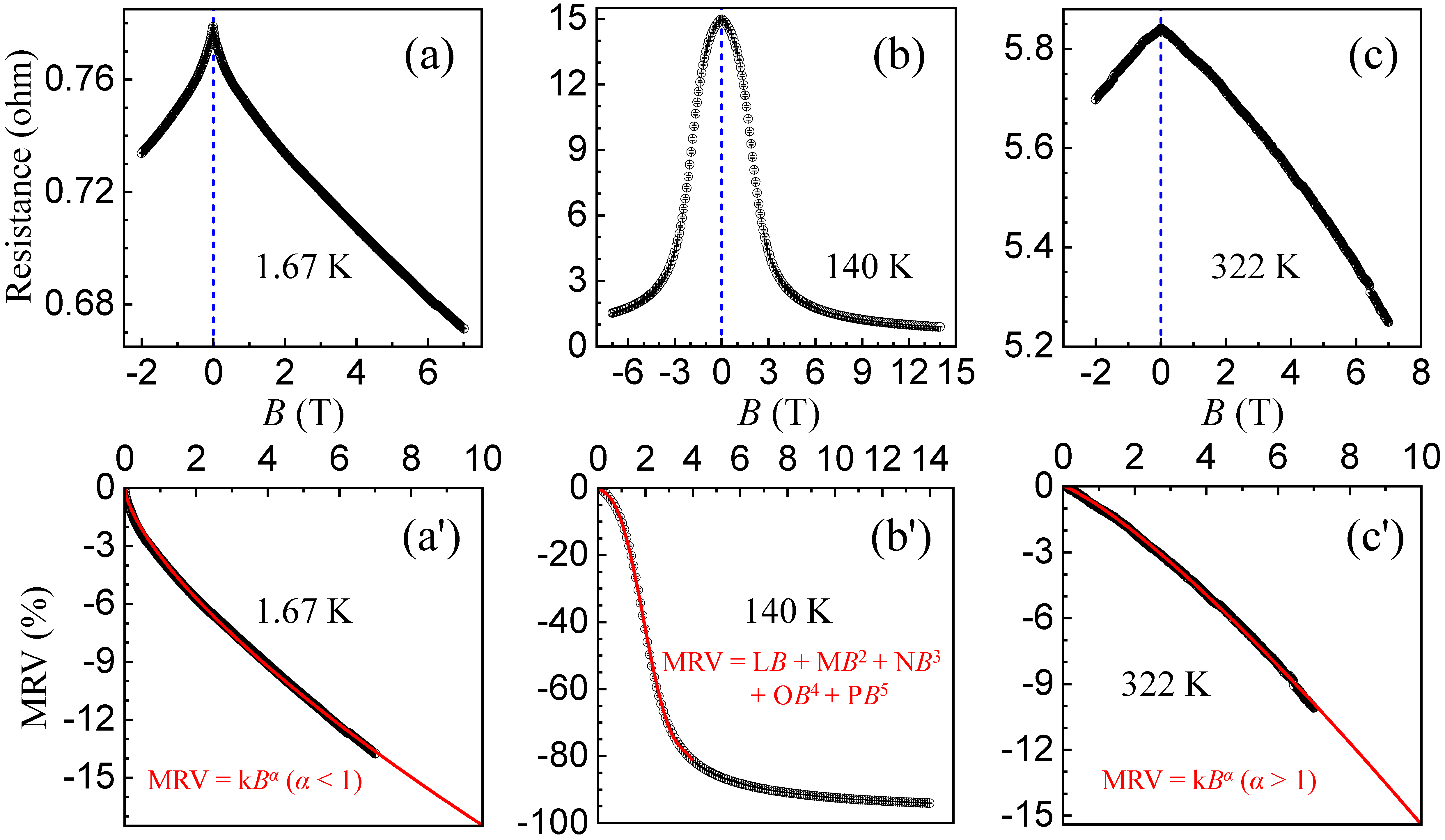
2.5. Magnetoresistance versus Applied Magnetic Field
3. Materials and Methods
3.1. Polycrystal Synthesis
3.2. Preparation of Single Crystal Growth
3.3. In-House Characterizations
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jonker, G.H.; Santen, J.H.V. Ferromagnetic compounds of manganese with perovskite structure. Physica 1950, 16, 337. [Google Scholar] [CrossRef]
- Helmot, R.V.; Wecker, J.; Holzapfel, B.; Schultz, L.; Samwer, K. Giant Negative Magnetoresistance in Perovskitelike La2/3Ba1/3MnOx Ferromagnetic Films. Phys. Rev. Lett. 1993, 71, 2331. [Google Scholar] [CrossRef]
- Chahara, K.; Ohno, T.; Kasai, M.; Kozono, Y. Magnetoresistance in magic manganese oxide with intrinsic antiferromagic spin structure. Appl. Phys. Lett. 1993, 63, 1990. [Google Scholar] [CrossRef]
- Murata, T.; Terai, T.; Fukuda, T.; Kakeshita, T.; Kishio, K. Influnce of Grain Boundray on Magnetoresistance in Hole Doped Manganites La0.7Ca0.3MnO3 and (La0.75Y0.25)0.7Sr0.3MnO3. Trans. Mater. Res. Soc. Jpn. 2003, 44, 2589. [Google Scholar]
- Urushibara, A.; Moritomo, Y.; Arima, T.; Asamitsu, A.; Kido, G.; Tokura, Y. Insulator-Metal Transition and Giant Magnetoresistance in La1-xSrxMnO3. Phys. Rev. B 1995, 51, 14103. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, P.; Ramirez, A.P.; Bao, W.; Cheong, S.-W. Study of tProduction in pCollisions Using Total Transverse Energy. Phys. Rev. Lett. 1995, 75, 3336. [Google Scholar] [CrossRef]
- Hwang, H.Y.; Cheong, S.-W.; Radaelli, P.G.; Marezio, M.; Batlogg, B. Lattice Effects on the Magnetoresistance in Doped LaMnO3. Phys. Rev. Lett. 1995, 75, 914. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Zhou, J.-S. New forms of phase segregation. Nature (London) 1997, 386, 229. [Google Scholar] [CrossRef]
- Teresa, J.M.D.; Ibarra, M.R.; Algarabel, P.A.; Ritter, C.; Marquina, C.; Blasco, J.G.J.; Moral, A.D.; Arnold, Z. Evidence for magnetic polarons in the magnetoresistive perovskites. Nature (London) 1997, 386, 256. [Google Scholar] [CrossRef]
- Uehara, M.; Mori, S.; Chen, C.H.; Cheong, S.-W. Percolative phase separation underlies colossal magnetoresistance in mixed-valent manganites. Nature (London) 1999, 399, 560. [Google Scholar] [CrossRef]
- Li, H.-F.; Xiao, Y.; Schmitz, B.; Persson, J.; Schmidt, W.; Meuffels, P.; Roth, G.; Brückel, T. Possible magnetic-polaron-switched positive and negative magnetoresistance in the GdSi single crystals. Sci. Rep. 2012, 2, 750. [Google Scholar] [CrossRef] [PubMed]
- Ruddlesden, S.N.; Popper, P. The compound Sr3Ti2O7 and its structure. Acta Cryst. 1958, 11, 54. [Google Scholar] [CrossRef]
- Seshadri, R.; Maigan, A.; Hervieu, M.; Nguyen, N.; Raveau, B. Complex magnetotransport in LaSr2Mn2O7. Solid State Commun. 1997, 101, 453. [Google Scholar] [CrossRef]
- Kubota, M.; Fujioka, H.; Ohoyama, K.; Moritomo, Y.; Yoshizawa, H.; Endoh, Y. Relation between crystal and magnetic structures of layered manganite La2-2xSr1+2xMn2O7 (0.30 < x < 0.50). J. Phys. Soc. Jpn. 2000, 69, 1606. [Google Scholar]
- Li, H.-F. Synthesis of CMR Manganites and Ordering Phenomena in Complex Transition Metal Oxides; Forschungszentrum Jülich GmbH: Jülich, Germany, 2008. [Google Scholar]
- Moritomo, Y.; Asamitsu, A.; Kuwahara, H.; Tokura, Y. Giant magnetoresistance of manganese oxides with a layered perovskite structure. Nature (London) 1996, 380, 141. [Google Scholar] [CrossRef]
- Battle, P.D.; Green, M.A.; Laskey, N.S.; Millburn, J.E.; Radaelli, P.G.; Rosseinsky, M.J.; Sullivan, S.P.; Vente, J.F. Crystal and magnetic structures of the colossal magnetoresistance manganates. Sr2-xNd1+xMn2O7 (x = 0.0, 0.1). Phys. Rev. B 1996, 54, 15967. [Google Scholar] [CrossRef]
- Moritomo, Y.; Maruyama, Y.; Akimoto, T.; Nakamura, A. Layered-type antiferromagnetic state in double-layered manganites: (La1-zNdz)2-2xSr1+2xMn2O7. J. Phys. Soc. Jpn. 1998, 67, 405. [Google Scholar] [CrossRef]
- Moritomo, Y.; Ohoyama, K.; Ohashi, M. Competition of interbilayer magnetic couplings in R1.4Sr1.6Mn2O7 (R = La1-zNdz). Phys. Rev. B 1999, 59, 157. [Google Scholar] [CrossRef]
- Asano, H.; Hayakawa, J.; Matsui, M. Two-dimensional ferromagnetic ordering and magnetoresistance in the layered perovskite La2-2xCa1+2xMn2O7. Phys. Rev. B 2000, 56, 5396. [Google Scholar]
- Hirota, K.; Moritomo, Y.; Fujioka, H.; Kubota, M.; Yoshizawa, H.; Endoh, Y. Neutron-diffraction studies on the magnetic ordering process in the layered Mn perovskite La2-2xSr1+2xMn2O7 (x = 0.40, 0.45 and 0.48). J. Phys. Soc. Jpn. 1998, 67, 3380. [Google Scholar] [CrossRef]
- Perring, T.G.; Aeppli, G.; Kimura, T.; Tokura, Y.; Adams, M.A. Ordered stack of spin valves in a layered magnetoresistive perovskite. Phys. Rev. B 1998, 58, 14693. [Google Scholar] [CrossRef]
- Argyriou, D.N.; Mitchell, J.F.; Radaelli, P.G.; Bordallo, H.N.; Cox, D.E.; Medarde, M.; Jorgensen, J.D. Lattice effects and magnetic structure in the layered colossal magnetoresistance manganite La2-2xSr1+2xMn2O7, x = 0.3. Phys. Rev. B 1999, 59, 8695. [Google Scholar] [CrossRef]
- Kimura, T.; Tokura, Y. Layered magnetic manganites. Annu. Rev. Mater. Sci. 2000, 30, 451. [Google Scholar] [CrossRef]
- Qiu, X.; Billinge, S.J.L.; Kmety, C.R.; Mitchell, J.F. Evidence for nano-scale inhomogeneities in bilayer manganites in the Mn4+ rich region: 0.54 ≤ x ≤ 0.80. J. Phys. Chem. Solids 2004, 65, 1423. [Google Scholar] [CrossRef][Green Version]
- Zheng, H.; Li, Q.A.; Gray, K.E.; Mitchell, J.F. Charge and orbital ordered phases of La2-2xSr1+2xMn2O7-δ. Phys. Rev. B 2008, 78, 155103. [Google Scholar] [CrossRef]
- Sonomura, H.; Terai, T.; Kakeshita, T.; Osakabe, T.; Kakurai, K. Neutron diffraction study on magnetic structures in a La1.37Sr1.63Mn2O7 single crystal under hydrostatic pressures of up to 0.8 GPa. Phys. Rev. B 2013, 87, 184419. [Google Scholar] [CrossRef]
- Guptasarma, P.; Williamsen, M.S.; Ray, S.K. Floating zone growth of bulk single crystals of complex oxides. Mater. Res. Soc. Symp. Proc. 2005, 848. [Google Scholar] [CrossRef]
- Ito, T.; Ushiyama, T.; Yanagisawa, Y.; Tomioka, Y.; Shindo, I.; Yanase, A. Laser-diode-heated floating zone (LDFZ) method appropriate to crystal growth of incongruently melting materials. J. Cryst. Growth 2013, 363, 264. [Google Scholar] [CrossRef]
- Wu, S.; Zhu, Y.H.; Gao, H.S.; Xiao, Y.G.; Xia, J.C.; Zhou, P.F.; Ouyang, D.F.; Li, Z.; Chen, Z.Q.; Tang, Z.K.; et al. Super-necking crystal growth and structural and magnetic properties of SrTb2O4 single crystals. ACS Omega 2020. accepted. [Google Scholar]
- Ouladdiaf, B.; Archer, J.; McIntyre, G.J.; Hewat, A.W.; Brau, D.; York, S. OrientExpress: A new system for Laue neutron diffraction. Phys. B. Condens. Matter 2006, 385–386, 1052. [Google Scholar] [CrossRef]
- Rodríguez-Carvajal, J. Recent advances in magnetic-structure determination by neutron powder diffraction. Phys. B. Condens. Matter 1993, 192, 55. [Google Scholar] [CrossRef]
- Li, H.-F.; Su, Y.; Persson, J.; Meuffels, P.; Walter, J.M.; Skowronek, R.; Brückel, T. Neutron-diffraction study of structural transition and magnetic order in orthorhombic and rhombohedral La7/8Sr1/8Mn1-γO3+δ. J. Phys. Condens. Matter 2007, 19, 176226. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.H.; Wu, S.; Jin, S.J.; Huq, A.; Persson, J.; Gao, H.S.; Ouyang, D.F.; He, Z.B.; Yao, D.-X.; Tang, Z.K.; et al. High-temperature magnetism and crystallography of a YCrO3 single crystal. Phys. Rev. B 2020, 101, 014114. [Google Scholar] [CrossRef]
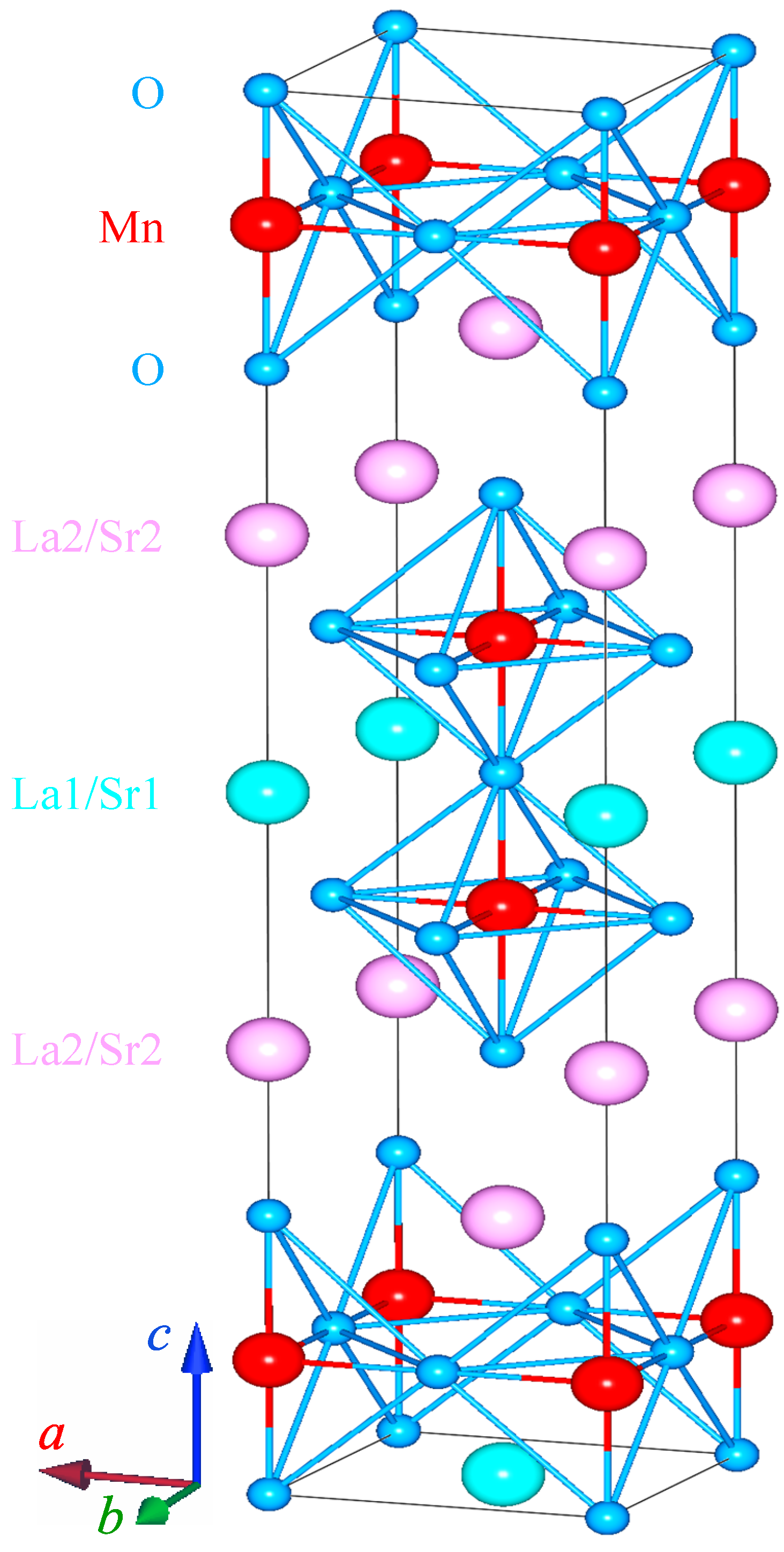

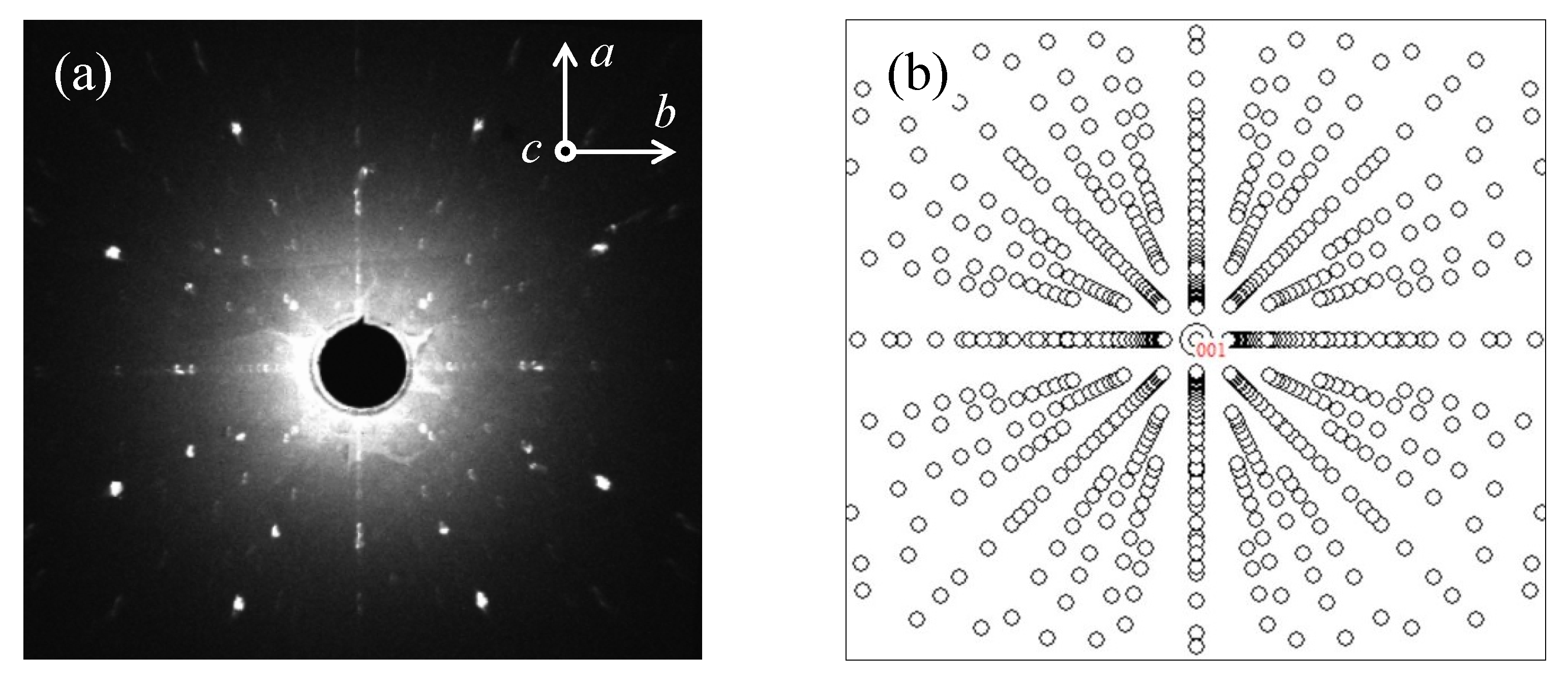
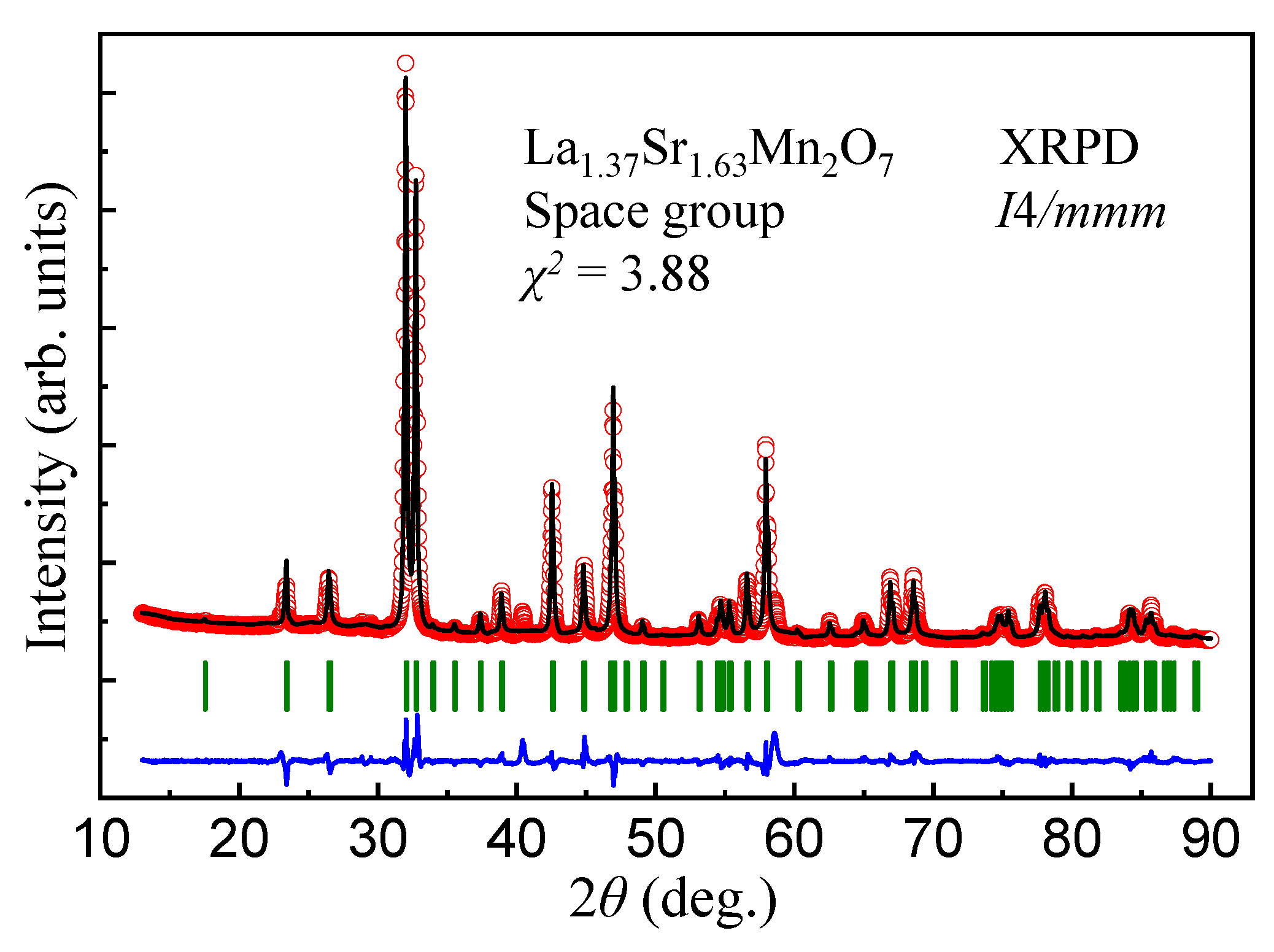
| XRPD Study of a Pulverized LaSrMnO Single Crystal (Tetragonal, Space Group I4/mmm (No. 139), ) | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a ( = b) (Å) 3.8671(1) | c (Å) 20.2108(8) | V (Å) 302.25(2) | ( = () 90 | ||||||||||||||
| Atom | Site | x | y | z | ITP | ||||||||||||
| La1/Sr1 | 2b | 0.00 | 0.00 | 0.50 | 2.41(4) | ||||||||||||
| La2/Sr2 | 4e | 0.00 | 0.00 | 0.3174(1) | 2.41(4) | ||||||||||||
| Mn | 4e | 0.00 | 0.00 | 0.0965(2) | 1.88(8) | ||||||||||||
| O1 | 2a | 0.00 | 0.00 | 0.00 | 2.2(1) | ||||||||||||
| O2 | 4e | 0.00 | 0.00 | 0.1971(7) | 2.2(1) | ||||||||||||
| O3 | 8g | 0.00 | 0.50 | 0.0958(5) | 2.2(1) | ||||||||||||
| = 5.73, = 9.20, = 4.67, and = 3.88 | |||||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.; Zhu, Y.; Xia, J.; Zhou, P.; Ni, H.; Li, H.-F. Colossal Negative Magnetoresistance Effect in a La1.37Sr1.63Mn2O7 Single Crystal Grown by Laser-Diode-Heated Floating-Zone Technique. Crystals 2020, 10, 547. https://doi.org/10.3390/cryst10060547
Wu S, Zhu Y, Xia J, Zhou P, Ni H, Li H-F. Colossal Negative Magnetoresistance Effect in a La1.37Sr1.63Mn2O7 Single Crystal Grown by Laser-Diode-Heated Floating-Zone Technique. Crystals. 2020; 10(6):547. https://doi.org/10.3390/cryst10060547
Chicago/Turabian StyleWu, Si, Yinghao Zhu, Junchao Xia, Pengfei Zhou, Haiyong Ni, and Hai-Feng Li. 2020. "Colossal Negative Magnetoresistance Effect in a La1.37Sr1.63Mn2O7 Single Crystal Grown by Laser-Diode-Heated Floating-Zone Technique" Crystals 10, no. 6: 547. https://doi.org/10.3390/cryst10060547
APA StyleWu, S., Zhu, Y., Xia, J., Zhou, P., Ni, H., & Li, H.-F. (2020). Colossal Negative Magnetoresistance Effect in a La1.37Sr1.63Mn2O7 Single Crystal Grown by Laser-Diode-Heated Floating-Zone Technique. Crystals, 10(6), 547. https://doi.org/10.3390/cryst10060547





