Crystal Structure of the N112A Mutant of the Light-Driven Sodium Pump KR2
Abstract
1. Introduction
2. Materials and Methods
2.1. Cloning, Protein Expression, and Purification
2.2. Crystallization
2.3. Acquisition and Treatment of Diffraction Data
2.4. Structure Determination and Refinement
2.5. Modeling of the O-State Structures of KR2-N112X Mutants
3. Results and Discussion
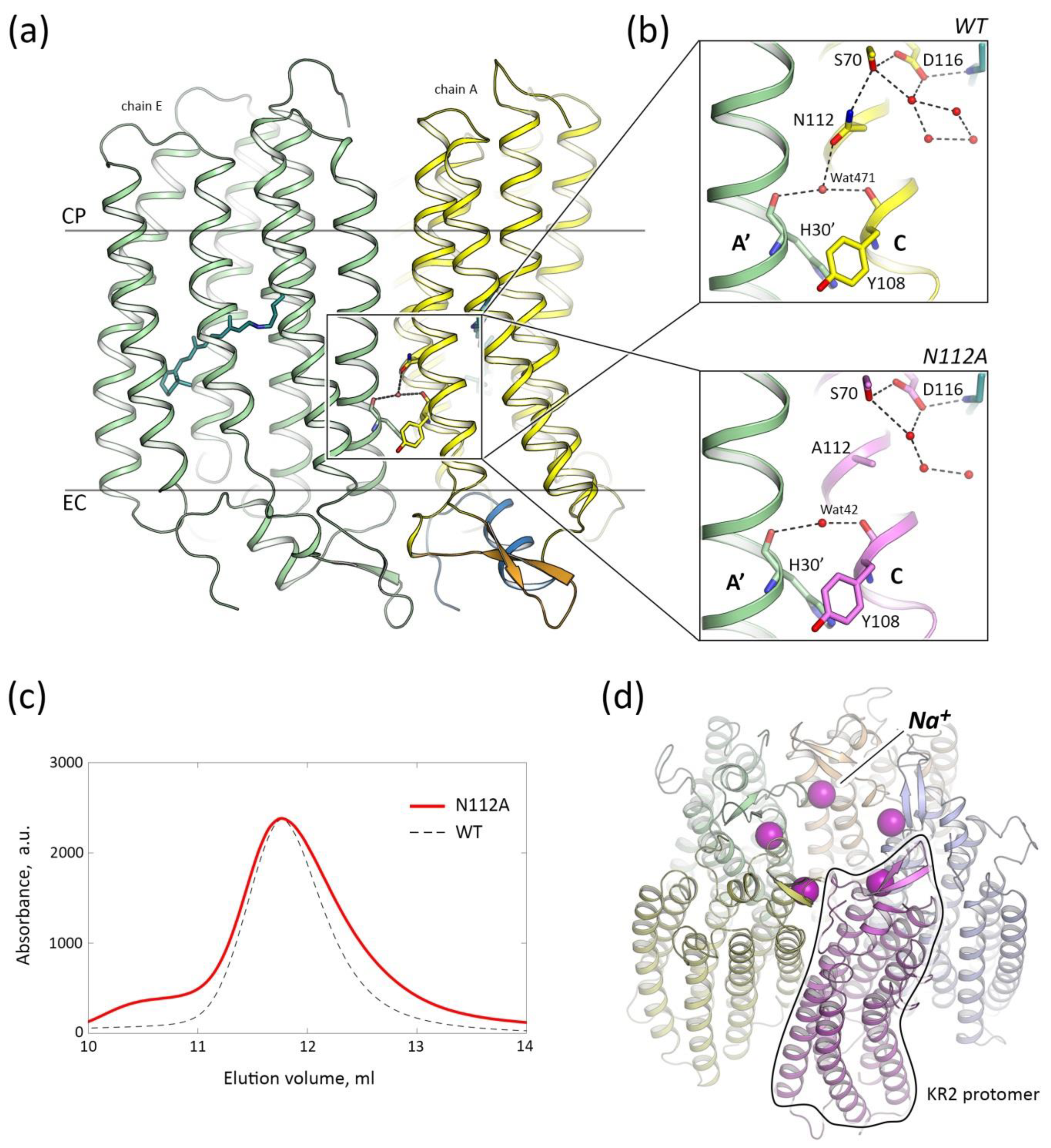
3.1. N112A Mutation Does Not Alter the Pentameric Assembly of KR2
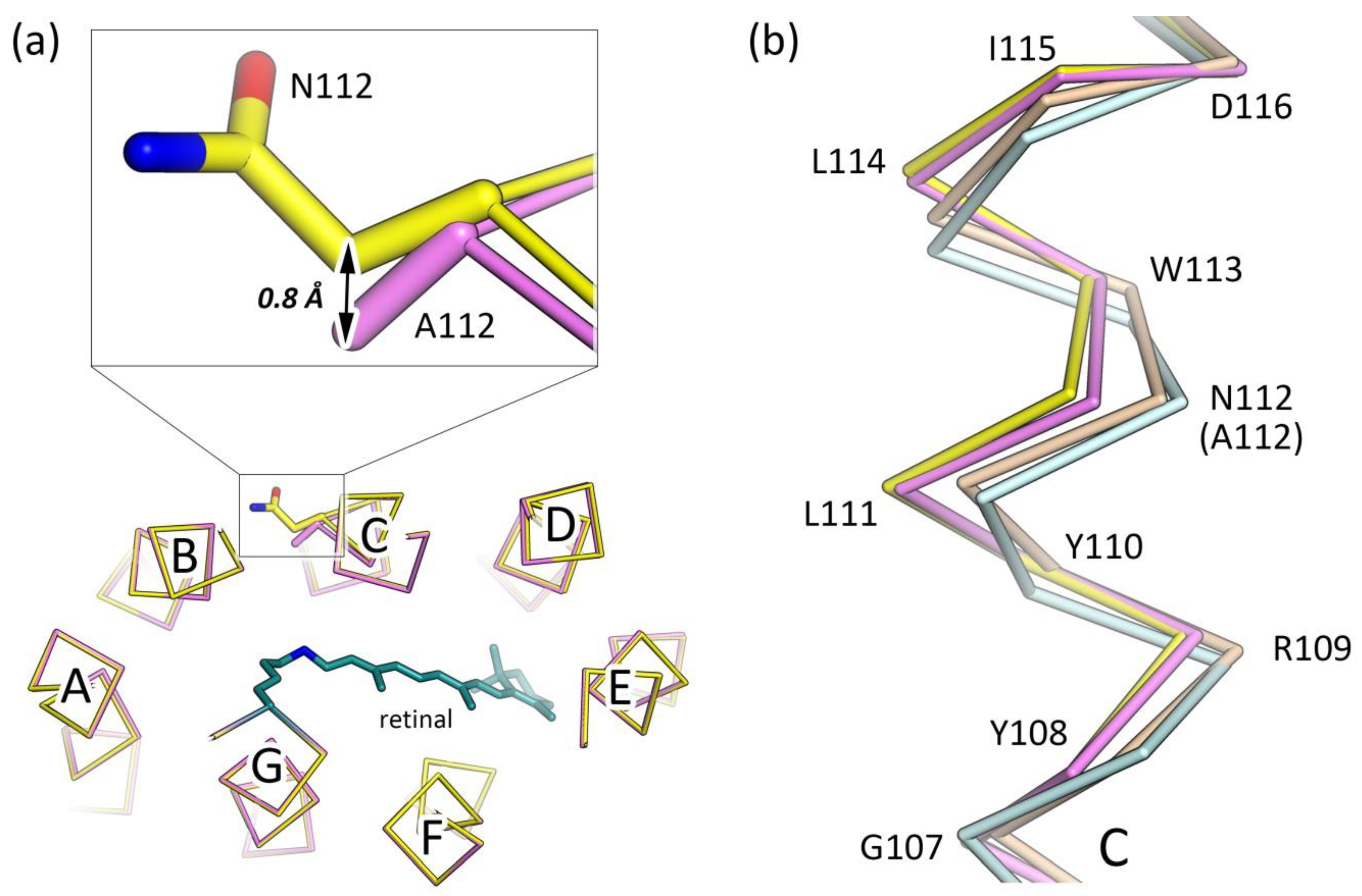
3.2. Crystal Structure of the N112A Protomer
3.3. The Organization of the RSB Region of KR2-N112A
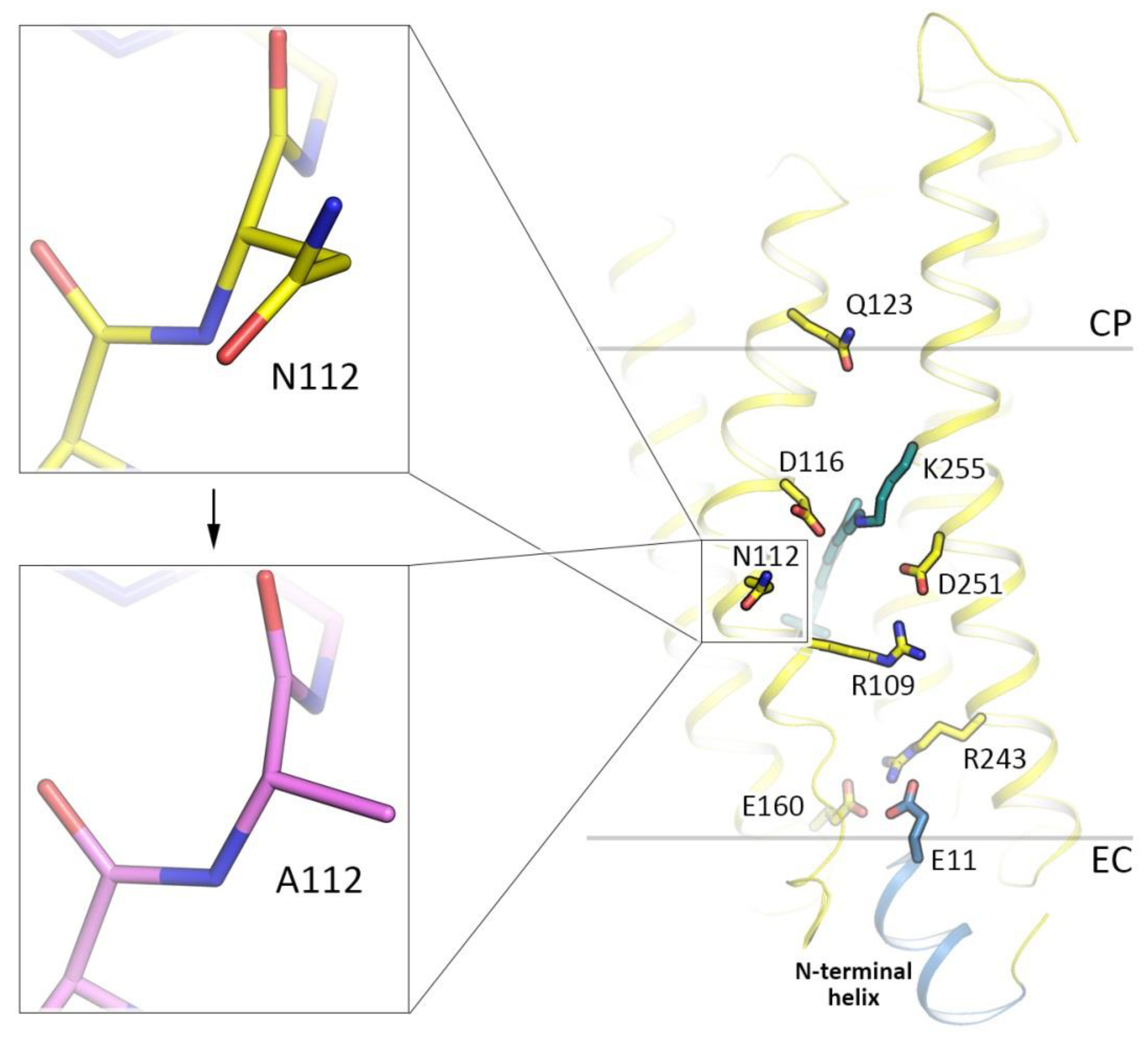
3.4. Role of N112 in the Ion Selectivity of KR2
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| WT | Wild type |
| KR2 | Krokinobacter eikastus rhodopsin 2 |
| SEC | Size exclusion chromatography |
| RSB | Retinal Schiff base |
| SBC1 | Schiff base cavity 1 |
References
- Gushchin, I.; Gordeliy, V. Microbial Rhodopsins. In Membrane Protein Complexes: Structure and Function; Springer: Singapore, 2018; pp. 19–56. [Google Scholar]
- Gushchin, I.; Shevchenko, V.; Polovinkin, V.; Kovalev, K.; Alekseev, A.; Round, E.; Borshchevskiy, V.; Balandin, T.; Popov, A.; Gensch, T.; et al. Crystal structure of a light-driven sodium pump. Nat. Struct. Mol. Biol. 2015, 22, 390–396. [Google Scholar] [CrossRef]
- Gushchin, I.; Chervakov, P.; Kuzmichev, P.; Popov, A.N.; Round, E.; Borshchevskiy, V.; Ishchenko, A.; Petrovskaya, L.; Chupin, V.; Dolgikh, D.A.; et al. Structural insights into the proton pumping by unusual proteorhodopsin from nonmarine bacteria. Proc. Natl. Acad. Sci. USA 2013, 110, 12631–12636. [Google Scholar] [CrossRef]
- Luecke, H.; Schobert, B.; Richter, H.T.; Cartailler, J.P.; Lanyi, J.K. Structure of bacteriorhodopsin at 1.55 Å resolution. J. Mol. Biol. 1999, 291, 899–911. [Google Scholar] [CrossRef]
- Kovalev, K.; Volkov, D.; Astashkin, R.; Alekseev, A.; Gushchin, I.; Haro-Moreno, J.M.; Chizhov, I.; Siletsky, S.; Mamedov, M.; Rogachev, A.; et al. High-resolution structural insights into the heliorhodopsin family. Proc. Natl. Acad. Sci. USA 2020, 117, 4131–4141. [Google Scholar] [CrossRef]
- Zabelskii, D.; Alekseev, A.; Kovalev, K.; Oliviera, A.-S.; Balandin, T.; Soloviov, D.; Bratanov, D.; Volkov, D.; Vaganova, S.; Astashkin, R.; et al. Viral channelrhodopsins: Calcium-dependent Na+/K+ selective light-gated channels. bioRxiv 2020. [Google Scholar] [CrossRef]
- Shihoya, W.; Inoue, K.; Singh, M.; Konno, M.; Hososhima, S.; Yamashita, K.; Ikeda, K.; Higuchi, A.; Izume, T.; Okazaki, S.; et al. Crystal structure of heliorhodopsin. Nature 2019, 574, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Needham, D.M.; Yoshizawa, S.; Hosaka, T.; Poirier, C.; Choi, C.J.; Hehenberger, E.; Irwin, N.A.T.; Wilken, S.; Yung, C.-M.; Bachy, C.; et al. A distinct lineage of giant viruses brings a rhodopsin photosystem to unicellular marine predators. Proc. Natl. Acad. Sci. USA 2019, 116, 20574–20583. [Google Scholar] [CrossRef] [PubMed]
- Oesterhelt, D.; Stoeckenius, W. Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nat. New Biol. 1971, 233, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Matsuno-Yagi, A.; Mukohata, Y. ATP synthesis linked to light-dependent proton uptake in a red mutant strain of Halobacterium lacking bacteriorhodopsin. Arch. Biochem. Biophys. 1980, 199, 297–303. [Google Scholar] [CrossRef]
- Govorunova, E.G.; Sineshchekov, O.A.; Li, H.; Spudich, J.L. Microbial Rhodopsins: Diversity, Mechanisms, and Optogenetic Applications. Annu. Rev. Biochem. 2017, 86, 845–872. [Google Scholar] [CrossRef]
- Ernst, O.P.; Lodowski, D.T.; Elstner, M.; Hegemann, P.; Brown, L.S.; Kandori, H. Microbial and Animal Rhodopsins: Structures, Functions, and Molecular Mechanisms. Chem. Rev. 2014, 114, 126–163. [Google Scholar] [CrossRef] [PubMed]
- Schobert, B.; Lanyi, J.K. Halorhodopsin is a light-driven chloride pump. J. Biol. Chem. 1982, 257, 10306–10313. [Google Scholar] [CrossRef] [PubMed]
- Bogomolni, R.A.; Spudich, J.L. The photochemical reactions of bacterial sensory rhodopsin-I. Flash photolysis study in the one microsecond to eight second time window. Biophys. J. 1987, 52, 1071–1075. [Google Scholar] [CrossRef]
- Béjà, O.; Spudich, E.N.; Spudich, J.L.; Leclerc, M.; DeLong, E.F. Proteorhodopsin phototrophy in the ocean. Nature 2001, 411, 786–789. [Google Scholar] [CrossRef] [PubMed]
- Nagel, G.; Szellas, T.; Huhn, W.; Kateriya, S.; Adeishvili, N.; Berthold, P.; Ollig, D.; Hegemann, P.; Bamberg, E. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc. Natl. Acad. Sci. USA 2003, 100, 13940–13945. [Google Scholar] [CrossRef]
- Nagel, G. Channelrhodopsin-1: A Light-Gated Proton Channel in Green Algae. Science 2002, 296, 2395–2398. [Google Scholar] [CrossRef]
- Govorunova, E.G.; Sineshchekov, O.A.; Spudich, J.L. Structurally Distinct Cation Channelrhodopsins from Cryptophyte Algae. Biophys. J. 2016, 110, 2302–2304. [Google Scholar] [CrossRef]
- Oppermann, J.; Fischer, P.; Silapetere, A.; Liepe, B.; Rodriguez-Rozada, S.; Flores-Uribe, J.; Peter, E.; Keidel, A.; Vierock, J.; Kaufmann, J.; et al. MerMAIDs: A family of metagenomically discovered marine anion-conducting and intensely desensitizing channelrhodopsins. Nat. Commun. 2019, 10, 3315. [Google Scholar] [CrossRef]
- Govorunova, E.G.; Sineshchekov, O.A.; Janz, R.; Liu, X.; Spudich, J.L. Natural light-gated anion channels: A family of microbial rhodopsins for advanced optogenetics. Science 2015, 349, 647–650. [Google Scholar] [CrossRef]
- Inoue, K.; Ono, H.; Abe-Yoshizumi, R.; Yoshizawa, S.; Ito, H.; Kogure, K.; Kandori, H. A light-driven sodium ion pump in marine bacteria. Nat. Commun. 2013, 4, 1678. [Google Scholar] [CrossRef]
- Kim, K.; Kwon, S.-K.; Jun, S.-H.; Cha, J.S.; Kim, H.; Lee, W.; Kim, J.F.; Cho, H.-S. Crystal structure and functional characterization of a light-driven chloride pump having an NTQ motif. Nat. Commun. 2016, 7, 12677. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Ito, S.; Kato, Y.; Nomura, Y.; Shibata, M.; Uchihashi, T.; Tsunoda, S.P.; Kandori, H. A natural light-driven inward proton pump. Nat. Commun. 2016, 7, 13415. [Google Scholar] [CrossRef] [PubMed]
- Shevchenko, V.; Mager, T.; Kovalev, K.; Polovinkin, V.; Alekseev, A.; Juettner, J.; Chizhov, I.; Bamann, C.; Vavourakis, C.; Ghai, R.; et al. Inward H+ pump xenorhodopsin: Mechanism and alternative optogenetic approach. Sci. Adv. 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Tsunoda, S.P.; Singh, M.; Tomida, S.; Hososhima, S.; Konno, M.; Nakamura, R.; Watanabe, H.; Bulzu, P.-A.; Banciu, H.L.; et al. Schizorhodopsins: A family of rhodopsins from Asgard archaea that function as light-driven inward H+ pumps. Sci. Adv. 2020, 6, eaaz2441. [Google Scholar] [CrossRef]
- Bulzu, P.-A.; Andrei, A.-Ş.; Salcher, M.M.; Mehrshad, M.; Inoue, K.; Kandori, H.; Beja, O.; Ghai, R.; Banciu, H.L. Casting light on Asgardarchaeota metabolism in a sunlit microoxic niche. Nat. Microbiol. 2019, 4, 1129–1137. [Google Scholar] [CrossRef]
- Pushkarev, A.; Inoue, K.; Larom, S.; Flores-Uribe, J.; Singh, M.; Konno, M.; Tomida, S.; Ito, S.; Nakamura, R.; Tsunoda, S.P.; et al. A distinct abundant group of microbial rhodopsins discovered using functional metagenomics. Nature 2018, 558, 595–599. [Google Scholar] [CrossRef]
- Yutin, N.; Koonin, E.V. Proteorhodopsin genes in giant viruses. Biol. Direct 2012, 7, 34. [Google Scholar] [CrossRef]
- Bratanov, D.; Kovalev, K.; Machtens, J.-P.; Astashkin, R.; Chizhov, I.; Soloviov, D.; Volkov, D.; Polovinkin, V.; Zabelskii, D.; Mager, T.; et al. Unique structure and function of viral rhodopsins. Nat. Commun. 2019, 10, 4939. [Google Scholar] [CrossRef]
- Luck, M.; Mathes, T.; Bruun, S.; Fudim, R.; Hagedorn, R.; Tran Nguyen, T.M.; Kateriya, S.; Kennis, J.T.M.; Hildebrandt, P.; Hegemann, P. A Photochromic Histidine Kinase Rhodopsin (HKR1) That Is Bimodally Switched by Ultraviolet and Blue Light. J. Biol. Chem. 2012, 287, 40083–40090. [Google Scholar] [CrossRef]
- Yoshida, K.; Tsunoda, S.P.; Brown, L.S.; Kandori, H. A unique choanoflagellate enzyme rhodopsin exhibits light-dependent cyclic nucleotide phosphodiesterase activity. J. Biol. Chem. 2017, 292, 7531–7541. [Google Scholar] [CrossRef]
- Gómez-Consarnau, L.; Raven, J.A.; Levine, N.M.; Cutter, L.S.; Wang, D.; Seegers, B.; Arístegui, J.; Fuhrman, J.A.; Gasol, J.M.; Sañudo-Wilhelmy, S.A. Microbial rhodopsins are major contributors to the solar energy captured in the sea. Sci. Adv. 2019, 5, eaaw8855. [Google Scholar] [CrossRef]
- Boyden, E.S.; Zhang, F.; Bamberg, E.; Nagel, G.; Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 2005, 8, 1263–1268. [Google Scholar] [CrossRef] [PubMed]
- Deisseroth, K. Optogenetics: 10 years of microbial opsins in neuroscience. Nat. Neurosci. 2015, 18, 1213–1225. [Google Scholar] [CrossRef] [PubMed]
- Kovalev, K.; Polovinkin, V.; Gushchin, I.; Alekseev, A.; Shevchenko, V.; Borshchevskiy, V.; Astashkin, R.; Balandin, T.; Bratanov, D.; Vaganova, S.; et al. Structure and mechanisms of sodium-pumping KR2 rhodopsin. Sci. Adv. 2019, 5, eaav2671. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Inoue, K.; Kandori, H. Kinetic Analysis of H+–Na+ Selectivity in a Light-Driven Na+ -Pumping Rhodopsin. J. Phys. Chem. Lett. 2015, 6, 5111–5115. [Google Scholar] [CrossRef]
- Kato, H.E.; Inoue, K.; Abe-Yoshizumi, R.; Kato, Y.; Ono, H.; Konno, M.; Hososhima, S.; Ishizuka, T.; Hoque, M.R.; Kunitomo, H.; et al. Structural basis for Na+ transport mechanism by a light-driven Na+ pump. Nature 2015, 521, 48–53. [Google Scholar] [CrossRef]
- Balashov, S.P.; Imasheva, E.S.; Dioumaev, A.K.; Wang, J.M.; Jung, K.-H.; Lanyi, J.K. Light-Driven Na+ Pump from Gillisia limnaea: A High-Affinity Na+ Binding Site Is Formed Transiently in the Photocycle. Biochemistry 2014, 53, 7549–7561. [Google Scholar] [CrossRef]
- Gerwert, K.; Freier, E.; Wolf, S. The role of protein-bound water molecules in microbial rhodopsins. Biochim. Biophys. Acta Bioenerg. 2014, 1837, 606–613. [Google Scholar] [CrossRef]
- Nishimura, N.; Mizuno, M.; Kandori, H.; Mizutani, Y. Distortion and a Strong Hydrogen Bond in the Retinal Chromophore Enable Sodium-Ion Transport by the Sodium-Ion Pump KR2. J. Phys. Chem. B 2019, 123, 3430–3440. [Google Scholar] [CrossRef]
- Kovalev, K.; Astashkin, R.; Gushchin, I.; Orekhov, P.; Volkov, D.; Zinovev, E.; Marin, E.; Rulev, M.; Alekseev, A.; Royant, A.; et al. Molecular mechanism of light-driven sodium pumping. Nat. Commun. 2020, 11, 2137. [Google Scholar] [CrossRef]
- Inoue, K.; Konno, M.; Abe-Yoshizumi, R.; Kandori, H. The Role of the NDQ Motif in Sodium-Pumping Rhodopsins. Angew. Chem. Int. Ed. 2015, 54, 11536–11539. [Google Scholar] [CrossRef]
- Tomida, S.; Ito, S.; Inoue, K.; Kandori, H. Hydrogen-bonding network at the cytoplasmic region of a light-driven sodium pump rhodopsin KR2. Biochim. Biophys. Acta Bioenerg. 2018, 1859, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Abe-Yoshizumi, R.; Inoue, K.; Kato, H.E.; Nureki, O.; Kandori, H. Role of Asn112 in a Light-Driven Sodium Ion-Pumping Rhodopsin. Biochemistry 2016, 55, 5790–5797. [Google Scholar] [CrossRef]
- Tomida, S.; Ito, S.; Mato, T.; Furutani, Y.; Inoue, K.; Kandori, H. Infrared spectroscopic analysis on structural changes around the protonated Schiff base upon retinal isomerization in light-driven sodium pump KR2. Biochim. Biophys. Acta Bioenerg. 2020, 1861, 148190. [Google Scholar] [CrossRef]
- Studier, F.W. Protein production by auto-induction in high-density shaking cultures. Protein Expr. Purif. 2005, 41, 207–234. [Google Scholar] [CrossRef]
- Landau, E.M.; Rosenbusch, J.P. Lipidic cubic phases: A novel concept for the crystallization of membrane proteins. Proc. Natl. Acad. Sci. USA 1996, 93, 14532–14535. [Google Scholar] [CrossRef] [PubMed]
- Gordeliy, V.I.; Labahn, J.; Moukhametzianov, R.; Efremov, R.; Granzin, J.; Schlesinger, R.; Büldt, G.; Savopol, T.; Scheidig, A.J.; Klare, J.P.; et al. Molecular basis of transmembrane signalling by sensory rhodopsin II-transducer complex. Nature 2002, 419, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Kabsch, W. XDS. Acta Crystallogr. Sect. D Biol. Crystallogr. 2010, 66, 125–132. [Google Scholar] [CrossRef]
- Tickle, I.J.; Flensburg, C.; Keller, P.; Paciorek, W.; Sharff, A.; Vonrhein, C.; Bricogne, G. STARANISO; Global Phasing Ltd.: Cambridge, UK, 2018. [Google Scholar]
- Vagin, A.; Teplyakov, A. Molecular replacement with MOLREP. Acta Crystallogr. Sect. D Biol. Crystallogr. 2010, 66, 22–25. [Google Scholar] [CrossRef]
- Emsley, P.; Cowtan, K. Coot: Model-building tools for molecular graphics. Acta Crystallogr. Sect. D Biol. Crystallogr. 2004, 60, 2126–2132. [Google Scholar] [CrossRef]
- Murshudov, G.N.; Skubák, P.; Lebedev, A.A.; Pannu, N.S.; Steiner, R.A.; Nicholls, R.A.; Winn, M.D.; Long, F.; Vagin, A.A. REFMAC5 for the refinement of macromolecular crystal structures. Acta Crystallogr. D Biol. Crystallogr. 2011, 67, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Winn, M.D.; Ballard, C.C.; Cowtan, K.D.; Dodson, E.J.; Emsley, P.; Evans, P.R.; Keegan, R.M.; Krissinel, E.B.; Leslie, A.G.W.; McCoy, A.; et al. Overview of the CCP 4 suite and current developments. Acta Crystallogr. Sect. D Biol. Crystallogr. 2011, 67, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Shibata, M.; Inoue, K.; Ikeda, K.; Konno, M.; Singh, M.; Kataoka, C.; Abe-Yoshizumi, R.; Kandori, H.; Uchihashi, T. Oligomeric states of microbial rhodopsins determined by high-speed atomic force microscopy and circular dichroic spectroscopy. Sci. Rep. 2018, 8, 8262. [Google Scholar] [CrossRef] [PubMed]
- Gushchin, I.; Shevchenko, V.; Polovinkin, V.; Borshchevskiy, V.; Buslaev, P.; Bamberg, E.; Gordeliy, V. Structure of the light-driven sodium pump KR2 and its implications for optogenetics. FEBS J. 2016, 283, 1232–1238. [Google Scholar] [CrossRef]
- Ho, B.K.; Gruswitz, F. HOLLOW: Generating Accurate Representations of Channel and Interior Surfaces in Molecular Structures. BMC Struct. Biol. 2008, 8, 49. [Google Scholar] [CrossRef]
- Harding, M.M.; Nowicki, M.W.; Walkinshaw, M.D. Metals in protein structures: A review of their principal features. Crystallogr. Rev. 2010, 16, 247–302. [Google Scholar] [CrossRef]
- Lomize, M.A.; Pogozheva, I.D.; Joo, H.; Mosberg, H.I.; Lomize, A.L. OPM database and PPM web server: Resources for positioning of proteins in membranes. Nucleic Acids Res. 2012, 40, D370–D376. [Google Scholar] [CrossRef]

| Data Collection | |
|---|---|
| Mutant | N112A |
| Protein Data Bank ID | 6YT4 |
| Space group | C2221 |
| Cell dimensions | - |
| a, b, c (Å) | 129.84, 239.70, 134.58 |
| α, β, γ (°) | 90, 90, 90 |
| Wavelength (Å) | 0.9762 |
| Resolution (Å) | 47.85–2.40 (2.455–2.400) * |
| Rmerge (%) | 21.6 (273.4) * |
| Rpim (%) | 3.5 (43.6) * |
| <I/σI> | 19.6 (1.8) * |
| CC1/2 (%) | 94.3 (82.1) * |
| Completeness, spherical (%) | 94.0 (72.1) * |
| Completeness, ellipsoidal (%) | 97.5 (99.2) * |
| Multiplicity | 40.7 (40.2) * |
| Unique reflections | 77,168 (3858) * |
| Refinement | |
| Resolution (Å) | 20.00–2.40 |
| No. reflections | 73,219 |
| Rwork/Rfree (%) | 18.6/21.7 |
| No. atoms | - |
| Protein | 10,791 |
| Retinal | 100 |
| Water | 286 |
| Lipids | 510 |
| Sodium | 5 |
| Average B factors (Å2) | - |
| Protein | 44 |
| Retinal | 42 |
| Water | 44 |
| Lipids | 73 |
| Sodium | 33 |
| R.m.s. deviations | - |
| Protein bond lengths (Å) | 0.0022 |
| Protein bond angles (°) | 1.0395 |
| Ramachandran analysis | - |
| Favored (%) | 96.3 |
| Outliers (%) | 0.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maliar, N.; Kovalev, K.; Baeken, C.; Balandin, T.; Astashkin, R.; Rulev, M.; Alekseev, A.; Ilyinsky, N.; Rogachev, A.; Chupin, V.; et al. Crystal Structure of the N112A Mutant of the Light-Driven Sodium Pump KR2. Crystals 2020, 10, 496. https://doi.org/10.3390/cryst10060496
Maliar N, Kovalev K, Baeken C, Balandin T, Astashkin R, Rulev M, Alekseev A, Ilyinsky N, Rogachev A, Chupin V, et al. Crystal Structure of the N112A Mutant of the Light-Driven Sodium Pump KR2. Crystals. 2020; 10(6):496. https://doi.org/10.3390/cryst10060496
Chicago/Turabian StyleMaliar, Nina, Kirill Kovalev, Christian Baeken, Taras Balandin, Roman Astashkin, Maksim Rulev, Alexey Alekseev, Nikolay Ilyinsky, Andrey Rogachev, Vladimir Chupin, and et al. 2020. "Crystal Structure of the N112A Mutant of the Light-Driven Sodium Pump KR2" Crystals 10, no. 6: 496. https://doi.org/10.3390/cryst10060496
APA StyleMaliar, N., Kovalev, K., Baeken, C., Balandin, T., Astashkin, R., Rulev, M., Alekseev, A., Ilyinsky, N., Rogachev, A., Chupin, V., Dolgikh, D., Kirpichnikov, M., & Gordeliy, V. (2020). Crystal Structure of the N112A Mutant of the Light-Driven Sodium Pump KR2. Crystals, 10(6), 496. https://doi.org/10.3390/cryst10060496






