Large Area Free-Standing Single Crystalline Films of p-Quinquephenyl: Growth, Structure and Photoluminescence Properties
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Crystal Growth
2.3. Optical and Laser Confocal Microscopy
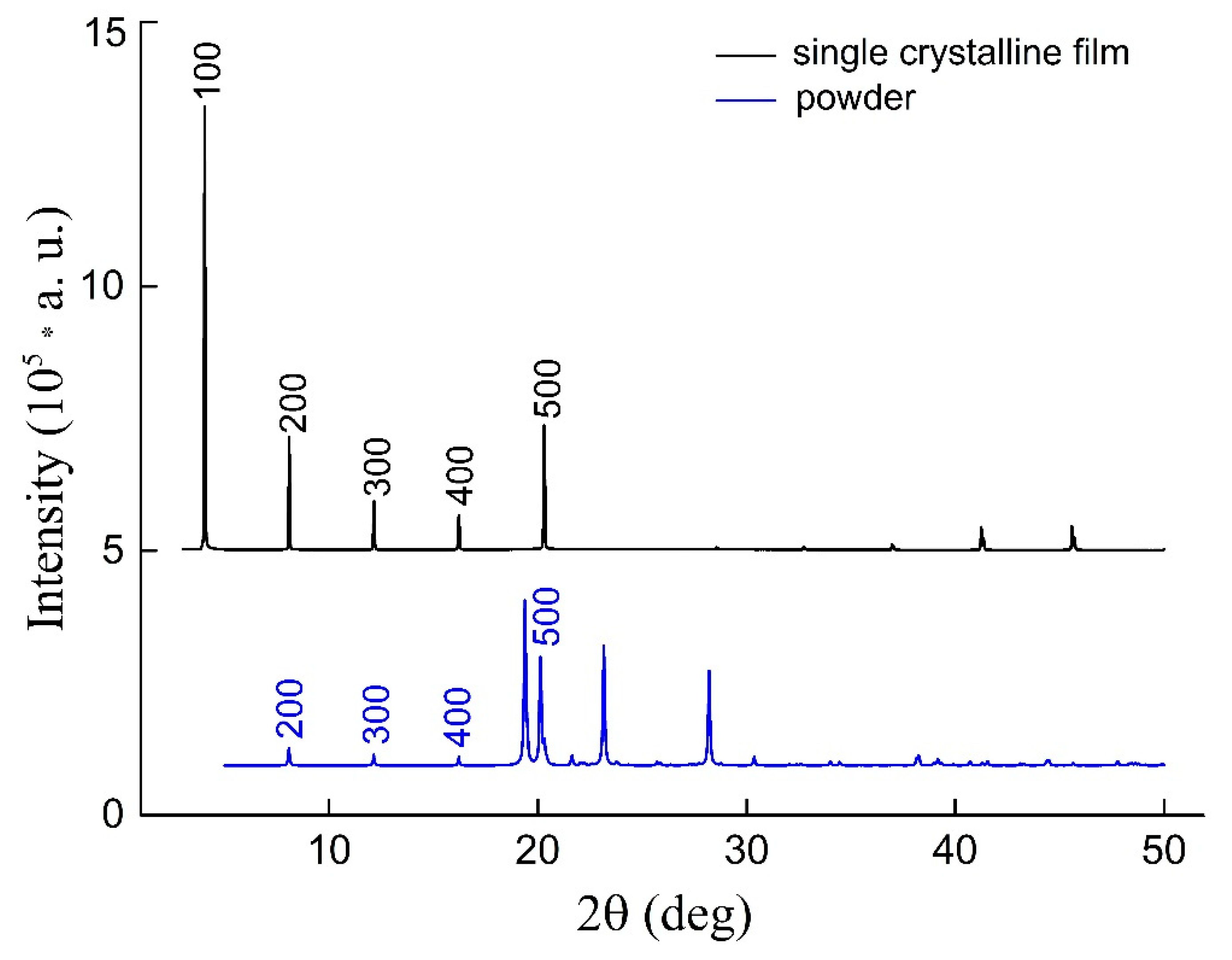
2.4. X-Ray Diffraction
2.5. Absorption and Photoluminescence Spectra
3. Results
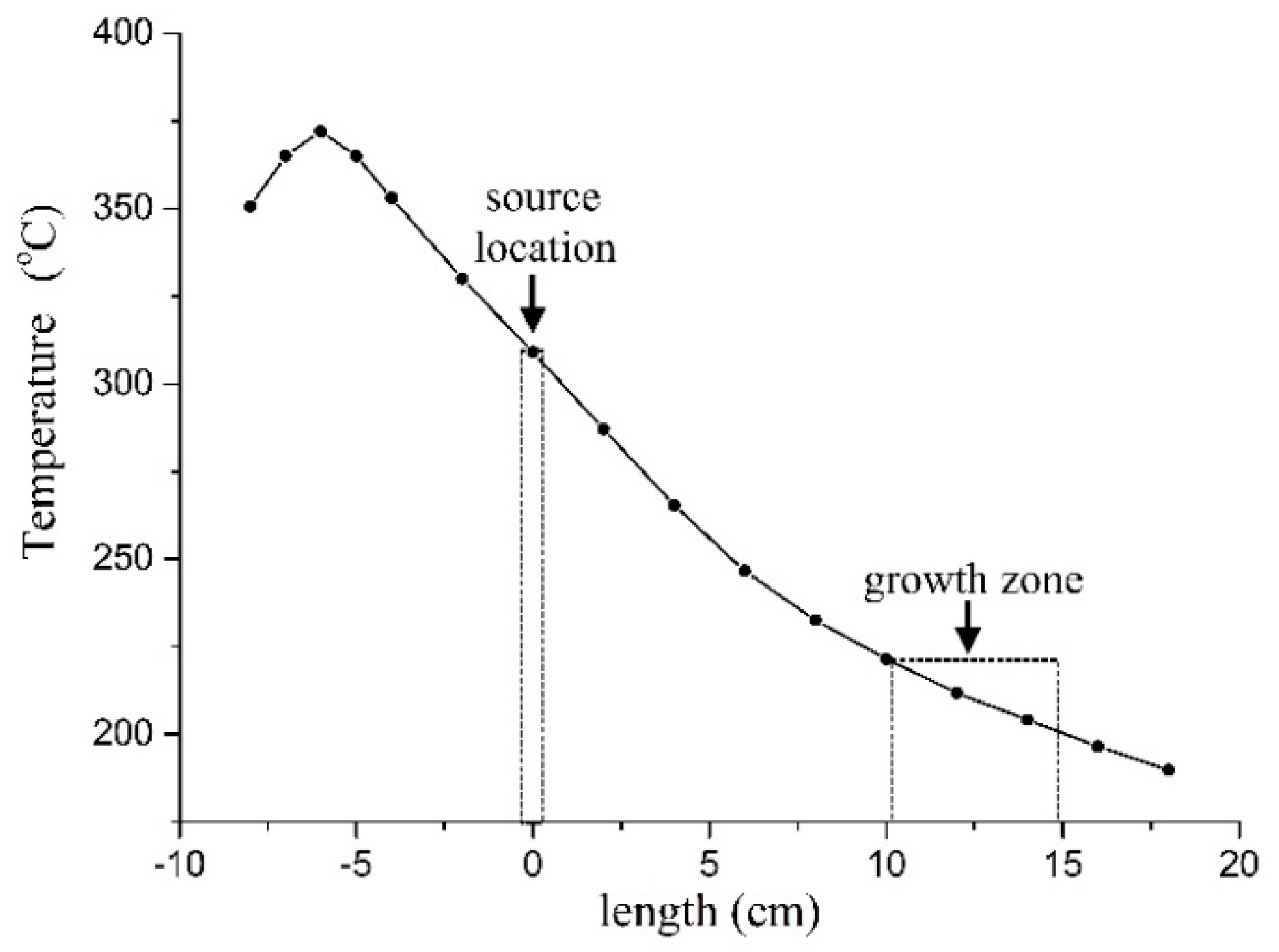
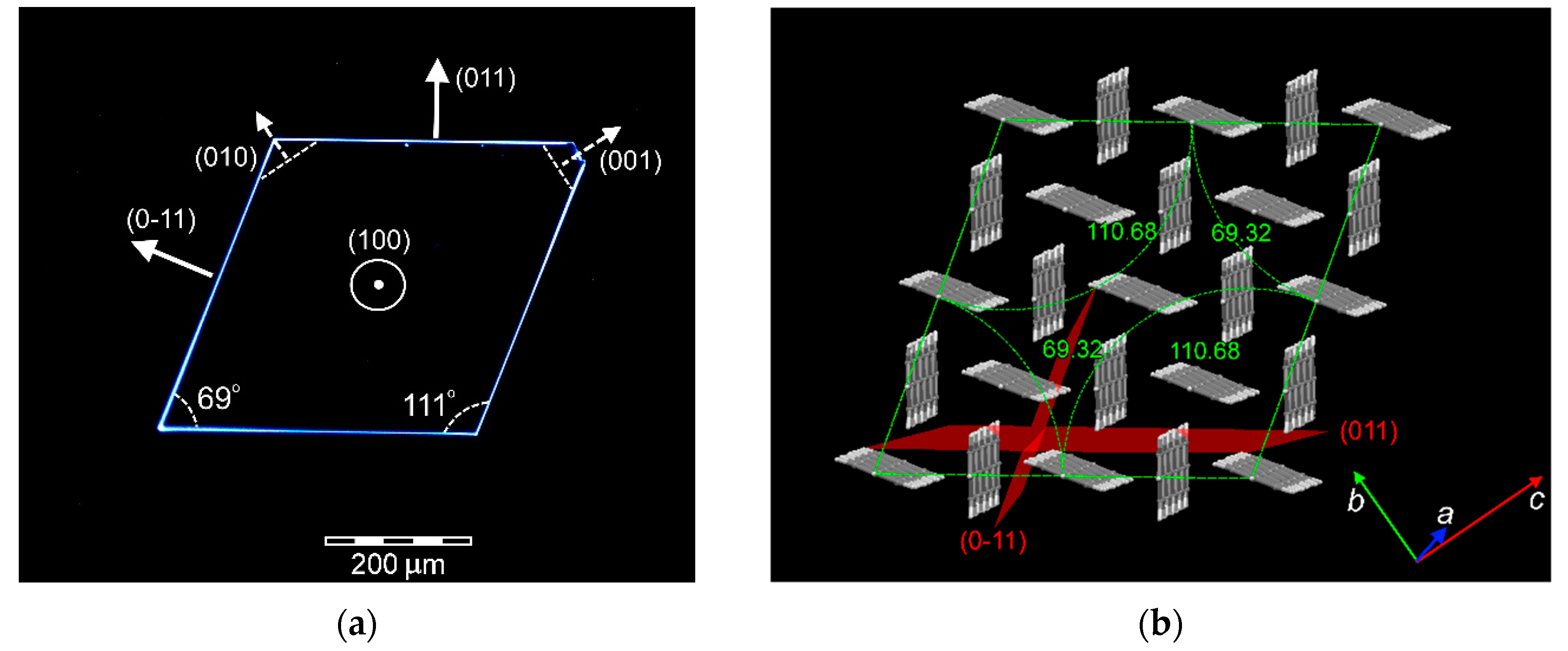
3.1. Crystal Growth and Morphology
3.1.1. Growth from Solution
3.1.2. Growth by PVT Method
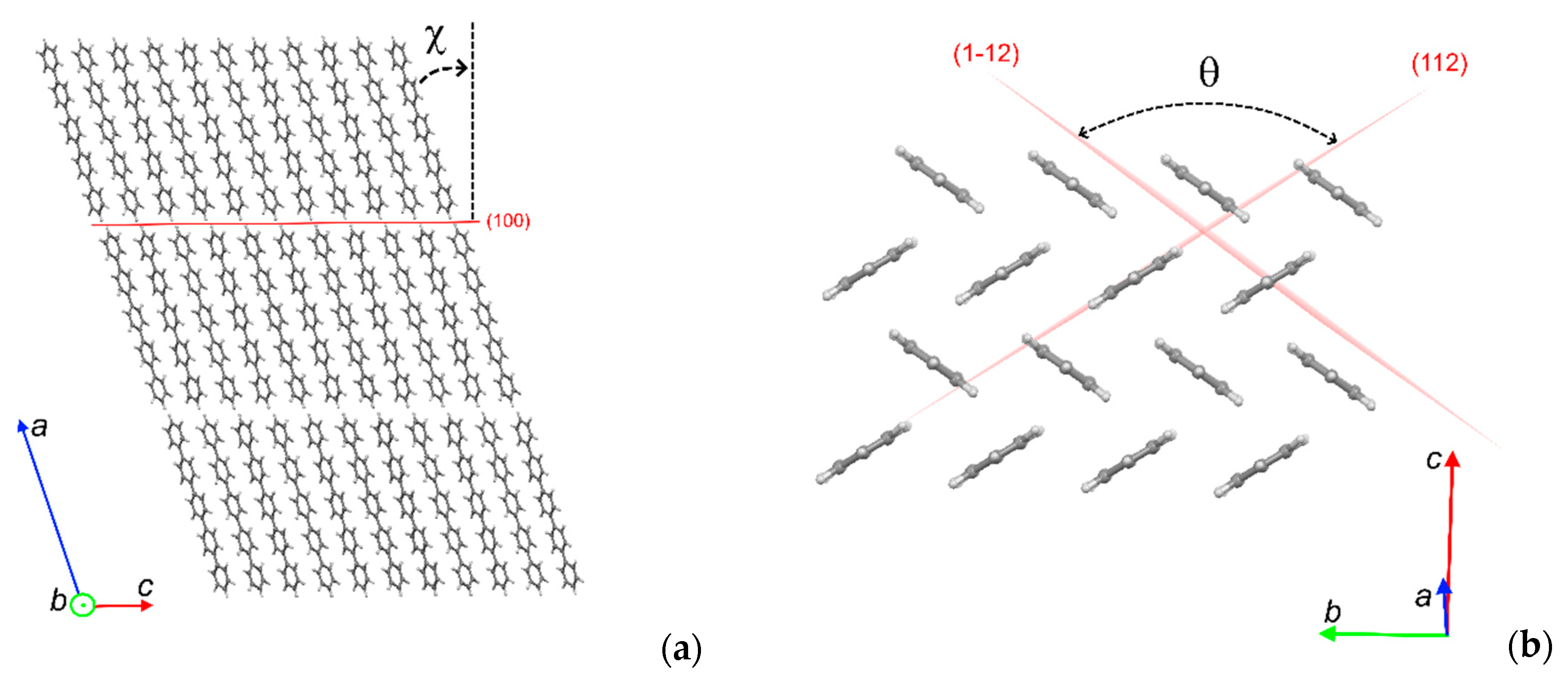
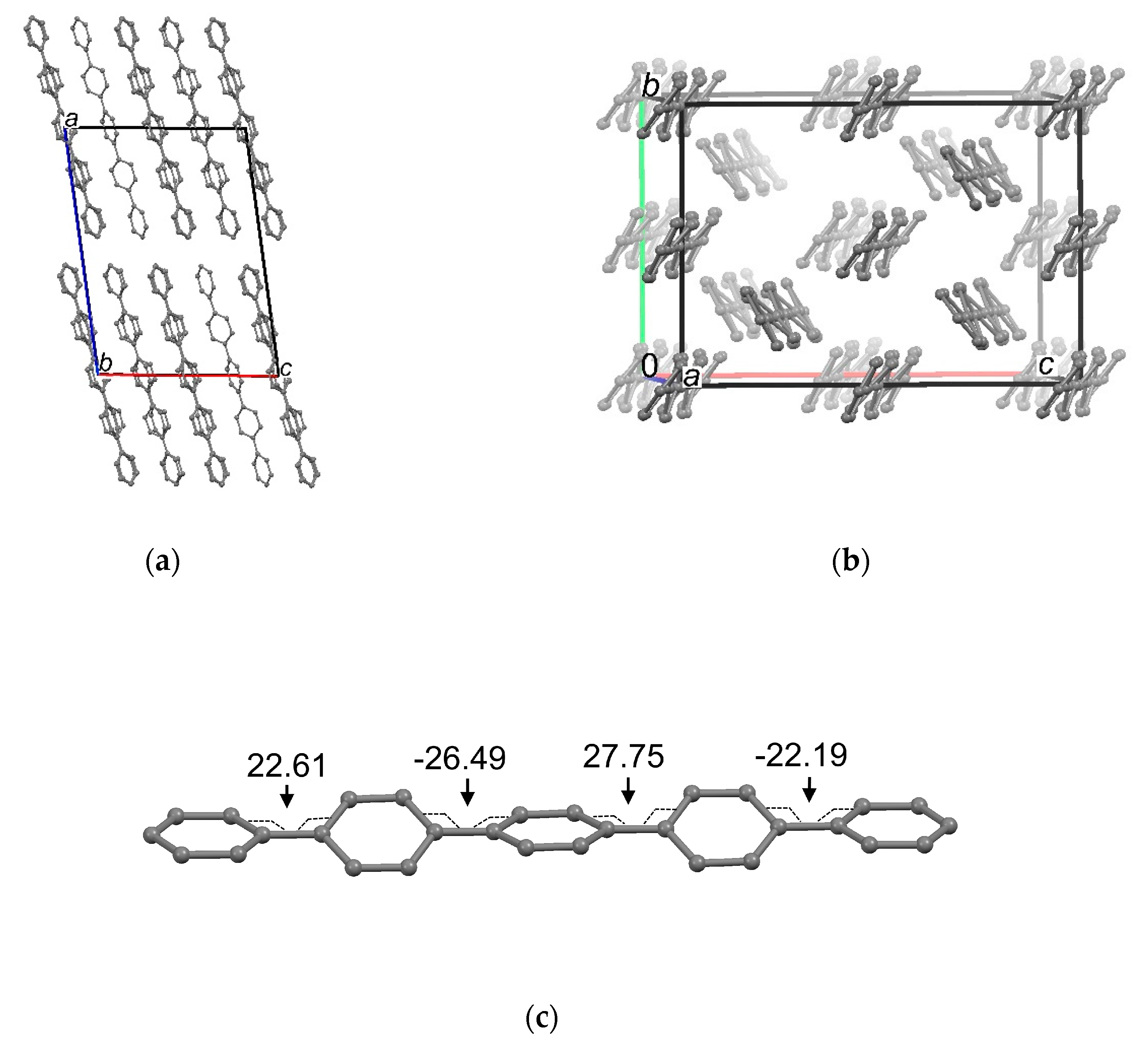
3.2. Crystal Structure
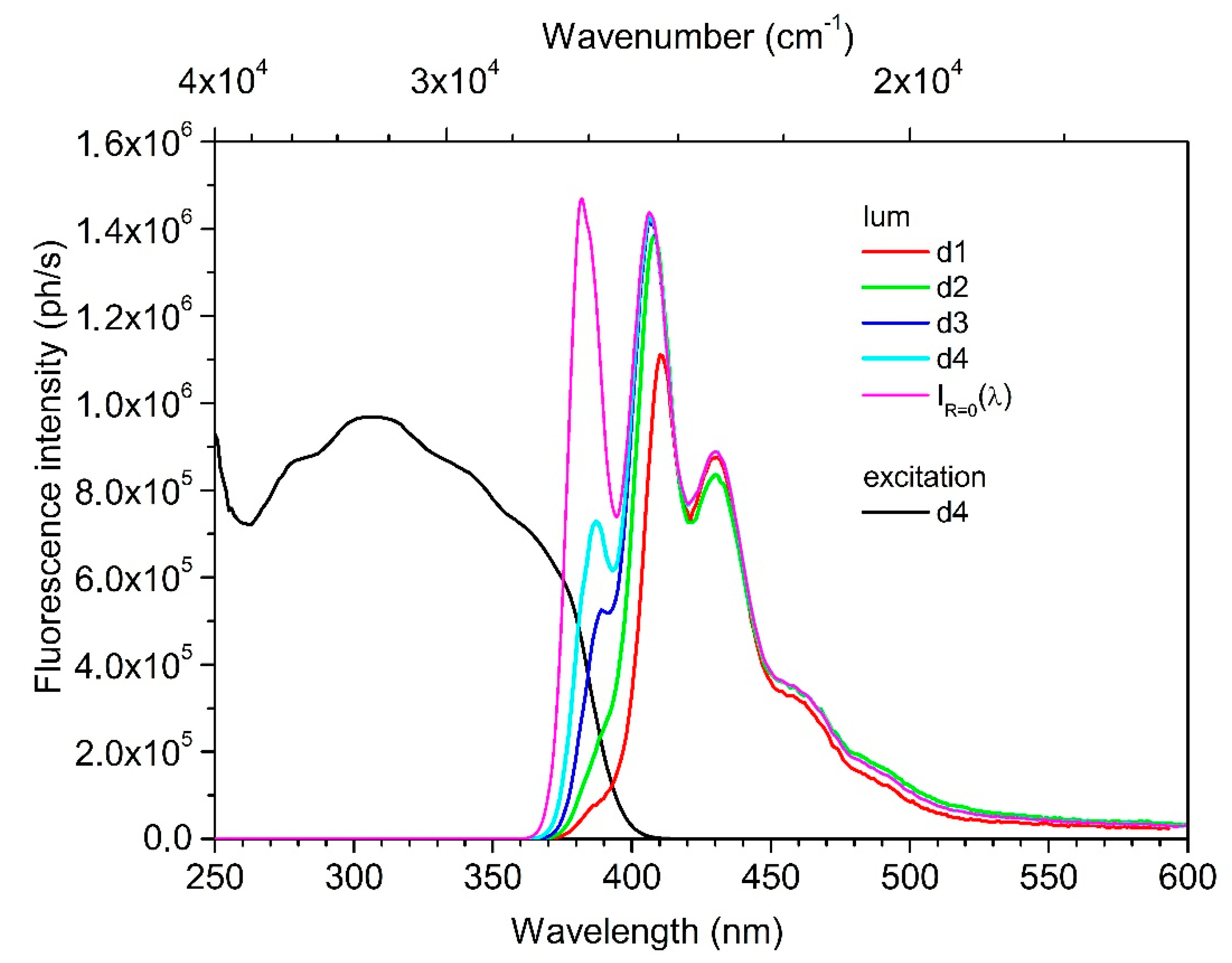
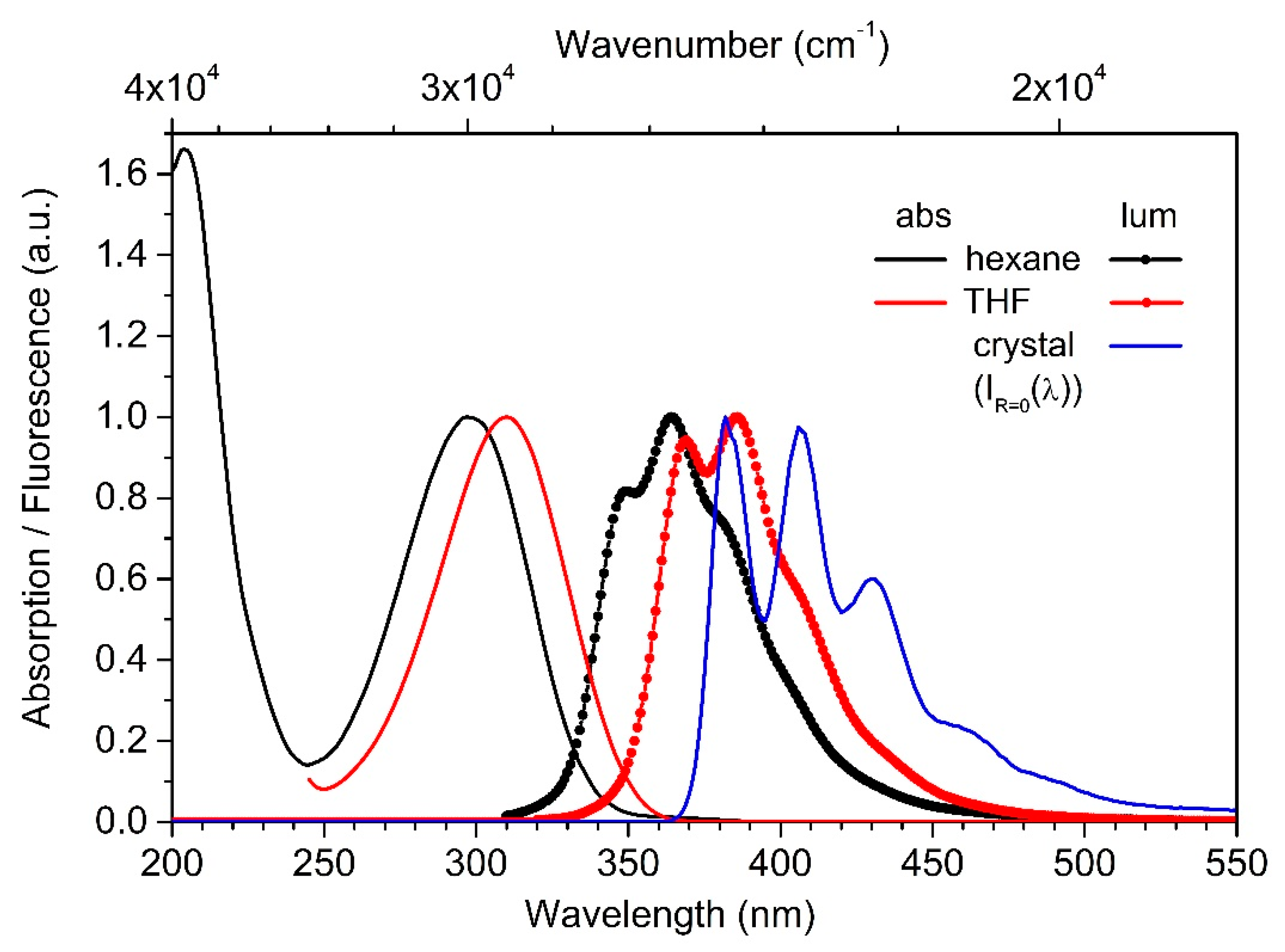
3.3. Photophysical Properties
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- De Boer, R.W.I.; Gershenson, M.E.; Morpurgo, A.F.; Podzorov, V. Organic single-crystal field-effect transistors. Phys. Stat. Sol. 2004, 201, 1302–1331. [Google Scholar] [CrossRef]
- Reese, C.; Bao, Z. Organic single-crystal field-effect transistors. Materialstoday 2007, 10, 20–27. [Google Scholar] [CrossRef]
- Hasegawa, T.; Takeya, J. Organic field-effect transistors using single crystals. Sci. Technol. Adv. Mater. 2009, 10, 024314. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Dong, H.; Hu, W. Organic single crystal field-effect transistors: Advances and perspectives. J. Mater. Chem. 2010, 20, 4994–5007. [Google Scholar] [CrossRef]
- Muccini, M. A bright future for organic field-effect transistors. Nat. Mater. 2006, 5, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Inada, Y.; Yamao, T.; Inada, M.; Itami, T.; Hotta, S. Giant organic single-crystals of a thiophene/phenylene co-oligomer toward device applications. Synth. Met. 2011, 161, 1869–1877. [Google Scholar] [CrossRef]
- Hotta, S.; Yamao, T.; Zulkarnaen Bisri, S.Z.; Takenobu, T.; Iwasa, Y. Organic single-crystal light-emitting field-effect transistors. J. Mater. Chem. C 2014, 2, 965–980. [Google Scholar] [CrossRef]
- Zeb, M.; Tahir, M.; Muhammad, F.; Said, S.M.; Sabri, M.F.M.; Sarker, M.R.; Ali, S.H.M.d.; Wahab, F. Amplified Spontaneous Emission and Optical Gain in Organic Single Crystal Quinquethiophene. Crystals 2019, 9, 609. [Google Scholar] [CrossRef]
- Piaggi, A.; Lanzani, G.; Bongiovanni, G.; Mura, A.; Graupner, W.; Meghdadi, F.; Leising, G.; Nisoli, M. Emission properties of para-hexaphenyl polycrystalline films. Phys. Rev. B 1997, 56, 10133. [Google Scholar] [CrossRef]
- Gundlach, D.J.; Lin, Y.-Y.; Jackson, T.N. Oligophenyl-based organic thin film transistors. Appl. Phys. Lett. 1997, 71, 3853. [Google Scholar] [CrossRef]
- Huang, G.; Zhong, G.-H.; Wang, R.-S.; Hand, J.-X.; Lind, H.-Q.; Chen, X.-J. Superconductivity and phase stability of potassium-doped p-quinquephenyl. Carbon 2019, 143, 837–843. [Google Scholar] [CrossRef]
- Hosoi, Y.; Koch, N.; Sakurai, Y.; Ishii, H.; Kampen, T.U.; Salvan, G.; Zahn, D.R.T.; Leising, G.; Ouchi, Y.; Seki, K. Structural study of thin films of neutral and potassium-doped oligophenylenes on Cu. Surf. Sci. 2005, 589, 19–31. [Google Scholar] [CrossRef]
- Kintzel, E.J., Jr.; Smilgies, D.-M.; Skofronick, J.G.; Safron, S.A.; Van Winkl, D.H. Ultrathin film growth of p-phenylene oligomers on alkali halide substrates. J. Cryst. Growth 2006, 289, 345–350. [Google Scholar] [CrossRef]
- Resel, R.; Koini, M.; Novak, J.; Berkebile, S.; Koller, G.; Ramsey, M. Epitaxial Order Driven by Surface Corrugation: Quinquephenyl Crystals on a Cu (110)-(2 × 1) O Surface. Crystals 2019, 9, 373. [Google Scholar] [CrossRef]
- Birks, B. The Theory and Practice of Scintillation Counting, 1st ed.; Pergamon Press Ltd.: Oxford, UK, 1964; p. 684. [Google Scholar] [CrossRef]
- Higashino, T.; Akiyama, Y.; Kojima, H.; Kawamoto, T.; Mori, T. Organic Semiconductors and Conductors with tert-Butyl Substituents. Crystals 2012, 2, 1222–1238. [Google Scholar] [CrossRef]
- Postnikov, V.A.; Odarchenko, Y.; Iovlev, A.V.; Bruevich, V.V.; Pereverzev, A.Y.; Kudryashova, L.G.; Sobornov, V.V.; Vidal, L.; Chernyshov, D.; Luponosov, Y.N.; et al. Molecularly Smooth Single-Crystalline Films of Thiophene–Phenylene Co-Oligomers Grown at the Gas–Liquid Interface. Cryst. Growth Des. 2014, 14, 1726. [Google Scholar] [CrossRef]
- Postnikov, V.A.; Sorokina, N.I.; Alekseeva, O.A.; Kulishov, A.A.; Sokolnikov, R.I.; Lyasnikova, M.S.; Grebenev, V.V.; Borshchev, O.V.; Skorotecky, M.S.; Surin, N.M.; et al. Growth from Solutions, Structure, and Photoluminescence of Single-Crystal Plates of p-Terphenyl and Its Trimethylsilyl Derivative. Crystallogr. Rep. 2018, 63, 819. [Google Scholar] [CrossRef]
- Postnikov, V.A.; Lyasnikova, M.S.; Kulishov, A.A.; Grebenev, V.V.; Borshchev, O.V. Solubility and Crystal Growth of p-Quaterphenyl and Its Derivative with Trimethylsilyl Terminal Substituents. Rus. J. Phys. Chem. A 2019, 93, 1741–1746. [Google Scholar] [CrossRef]
- Postnikov, V.A.; Sorokina, N.I.; Kulishov, A.A.; Lyasnikova, M.S.; Grebenev, V.V.; Voloshin, A.E.; Borshchev, O.V.; Skorotetcky, M.S.; Surin, N.M.; Svidchenko, E.A.; et al. Highly luminescent crystals of a novel linear π-conjugated thiophene–phenylene co-oligomer with a benzothiadiazole fragment. Acta Cryst. B 2019, B75, 1076–1085. [Google Scholar] [CrossRef]
- Laudise, R.A.; Kloc, C.; Simpkins, P.G.; Siegrist, T. Physical vapor growth of organic semiconductors. J. Cryst. Growth 1998, 187, 449. [Google Scholar] [CrossRef]
- Jo, S.; Takenaga, M. Morphologies of Pentacene Crystals Obtained by Physical Vapor Growth Technique. Jpn. J. Appl. Phys. 2010, 49, 078002. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, Y.; Xie, Z.; Wang, H.; Wanga, B.; Ma, Y. The thermodynamic characteristics of organic crystal growth by physical vapor transport: Towards high-quality and color-tunable crystal preparation. CrystEngComm 2014, 16, 4539. [Google Scholar] [CrossRef]
- Lopez-Romero, J.M.; Rico, R.; Martınez-Mallorquın, R.; Hierrezuelo, J.; Guillen, E.; Cai, C.; Otero, J.C.; Lopez-Tocon, I. Synthesis of penta-p-phenylenes with oligo(ethylene glycol) side chains. Tetrahedron Lett. 2007, 48, 6075. [Google Scholar] [CrossRef]
- CrysAlis Pro Software System, Version 1.171.39.46; Rigaku Corporation: Oxford, UK, 2018.
- Petricek, V.; Dusek, M.; Palatinus, L. Crystallographic Computing System JANA2006, General features. Z. Kristallogr. 2014, 229, 345. [Google Scholar] [CrossRef]
- Palatinus, L. Ab initio determination of incommensurately modulated structures by charge flipping in superspace. Acta Cryst. 2004, A60, 604. [Google Scholar] [CrossRef]
- Shumilkina, E.A.; Borshchev, O.V.; Ponomarenko, S.A.; Surin, N.M.; Pleshkova, A.P.; Muzafarov, A.M. Synthesis and optical properties of linear and branched bithienylsilanes. Mendeleev Commun. 2007, 17, 34–36. [Google Scholar] [CrossRef]
- Berlman, I.B. Handbook of Fluorescence Spectra of Aromatic Molecules; Academic Press: New York, NY, USA, 1971. [Google Scholar]
- Lally, C.H.; Davies, G.J.; Jones, W.G.; Smith, N.J.T. UV quantum efficiencies of organic fluors. Nucl. Instrum. Methods Phys. Res. 1996, 117, 421. [Google Scholar] [CrossRef]
- Ezhumalai, Y.; Wang, T.-H.; Hsu, H.-F. Regioselective Synthesis of Tetraphenyl-1,3-butadienes with Aggregation-Induced Emission. Org. Lett. 2015, 17, 536. [Google Scholar] [CrossRef]
- Postnikov, V.A.; Sorokina, N.I.; Alekseeva, O.A.; Grebenev, V.V.; Lyasnikova, M.S.; Borshchev, O.V.; Surin, N.M.; Svidchenko, E.A.; Ponomarenko, S.A.; Voloshin, A.E. Growth from Solution, Structure, and Optical Properties of Single-Crystal para-Quaterphenyl Films. Crystallogr. Rep. 2018, 63, 139–148. [Google Scholar] [CrossRef]
- Baker, K.N.; Knachel, H.C.; Fratini, A.V.; Adams, W.W. Structural transformations in crystalline oligomers of polyparaphenylene. Mat. Res. Soc. Symp. Proc. 1988, 134, 497–503. [Google Scholar] [CrossRef]
- Baker, K.N.; Fratini, A.V.; Resch, T.; Knachel, H.C. Crystal structures, phase transitions and energy calculations of poly(p-phenylene) oligomers. Polymer 1993, 34, 1571. [Google Scholar] [CrossRef]
- Postnikov, V.A.; Lyasnikova, M.S.; Kulishov, A.A.; Sorokina, N.I.; Voloshin, A.E.; Skorotetcky, M.S.; Borshchev, O.V.; Ponomarenko, S.A. Growth Anisotropy and Crystal Structure of Linear Conjugated Oligomers. Phys. Solid State 2019, 61, 2321. [Google Scholar] [CrossRef]
- Taniguchi, M.; Lindsey, J.S. Database of absorption and fluorescence spectra of >300 common compounds for use in PhotochemCAD. Photochem. Photobiol. 2018, 94, 290. [Google Scholar] [CrossRef] [PubMed]
- Seoul, C.; Song, W.-J.; Kang, G.-W.; Lee, C. Effect of molar mass on electroluminescence op poly(p-phenylene). Synth. Met. 2002, 130, 9. [Google Scholar] [CrossRef]
- Postnikov, V.A. Formation and growth of biphenyl and naphthalene large single crystal sheets on liquid-air interface. Kondens. Sredy I Mezhfaznye Granitsy Condens. Matter Interphases 2013, 15, 160. [Google Scholar]
- Nigam, S.; Rutan, S. Principles and Applications of Solvatochromism. Appl. Spectrosc. 2001, 55, 362A. [Google Scholar] [CrossRef]
- Beale, R.N.; Roe, E.M.F. The Spectrophotometric Determination of Steric Hindrance in Some Stilbene Derivatives. J. Am. Chem. Soc. 1952, 74, 2302. [Google Scholar] [CrossRef]
- Murrell, J.N. The Theory of the Electronic Spectra of Organic Molecules; John Wiley and Sons, Inc.: New York, NY, USA, 1963; p. 328. [Google Scholar]



| Formula | C30H22 | |
| Temperature (K) | 293 | 85 |
| Crystal system | monoclinic | triclinic |
| Space group | P21/c | P−1 |
| a (Å) | 22.059(1) | 22.042(1) |
| b (Å) | 5.579(1) | 11.018(1) |
| c (Å) | 8.070(1) | 15.970(1) |
| α (°) | 90.0 | 90.049(6) |
| β (°) | 97.989(7) | 98.203(5) |
| γ (°) | 90.0 | 90.425(5) |
| Vo (Å3) | 983.55(6) | 3838.6(1) |
| Z | 2 | 8 |
| Dcalc (g/cm3) | 1.292 | 1.324 |
| No. of measured, independent and observed [I > 3σ(I)] reflections | 20,654, 5233, 1648 | 84,275, 37,255, 5057 |
| No. of refined parameters | 180 | 1081 |
| R/wR | 0.062/0.052 | 0.057/0.068 |
| S | 2.15 | 1.85 |
| Δρmin/Δρmax (e·Å−3) | −0.47/0.33 | −0.45/0.54 |
| E0−0 eV/cm−1 | ΔE0−0 eV/cm−1 | PLQY | |
|---|---|---|---|
| crystal | 3.31/26,700 | - | 0.95 |
| DMF *) | 3.54/28,500 | 0.23/1800 | 0.89 |
| THF | 3.56/28,700 | 0.25/2000 | 0.93 |
| hexan | 3.73/30,100 | 0.42/3400 | 0.63 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Postnikov, V.A.; Sorokina, N.I.; Lyasnikova, M.S.; Kulishov, A.A.; Voloshin, A.E.; Borshchev, O.V.; Surin, N.M.; Svidchenko, E.A.; Ponomarenko, S.A. Large Area Free-Standing Single Crystalline Films of p-Quinquephenyl: Growth, Structure and Photoluminescence Properties. Crystals 2020, 10, 363. https://doi.org/10.3390/cryst10050363
Postnikov VA, Sorokina NI, Lyasnikova MS, Kulishov AA, Voloshin AE, Borshchev OV, Surin NM, Svidchenko EA, Ponomarenko SA. Large Area Free-Standing Single Crystalline Films of p-Quinquephenyl: Growth, Structure and Photoluminescence Properties. Crystals. 2020; 10(5):363. https://doi.org/10.3390/cryst10050363
Chicago/Turabian StylePostnikov, Valery A., Nataliya I. Sorokina, Maria S. Lyasnikova, Artem A. Kulishov, Alexey E. Voloshin, Oleg V. Borshchev, Nikolay M. Surin, Evgeniya A. Svidchenko, and Sergey A. Ponomarenko. 2020. "Large Area Free-Standing Single Crystalline Films of p-Quinquephenyl: Growth, Structure and Photoluminescence Properties" Crystals 10, no. 5: 363. https://doi.org/10.3390/cryst10050363
APA StylePostnikov, V. A., Sorokina, N. I., Lyasnikova, M. S., Kulishov, A. A., Voloshin, A. E., Borshchev, O. V., Surin, N. M., Svidchenko, E. A., & Ponomarenko, S. A. (2020). Large Area Free-Standing Single Crystalline Films of p-Quinquephenyl: Growth, Structure and Photoluminescence Properties. Crystals, 10(5), 363. https://doi.org/10.3390/cryst10050363







