Adsorption Characteristics of Gas Molecules (H2O, CO2, CO, CH4, and H2) on CaO-Based Catalysts during Biomass Thermal Conversion with in Situ CO2 Capture
Abstract
:1. Introduction
2. Results and Discussion
2.1. Comparison of Adsorption Characteristics of CO2 on Two Catalyst Surfaces at Different Temperatures
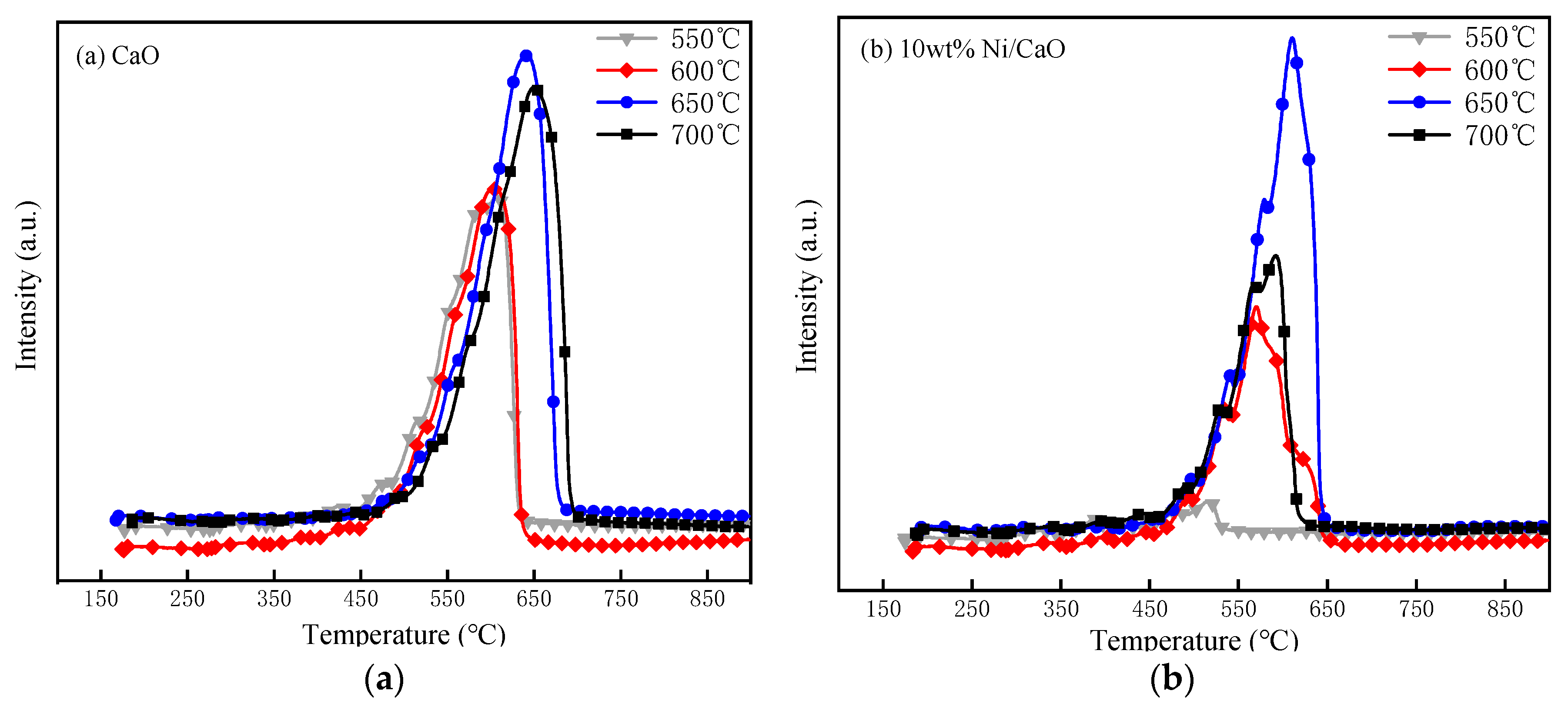
2.2. Analysis of CO2 Temperature-Programmed Desorption (CO2-TPD) Experimental Results at Different Temperatures
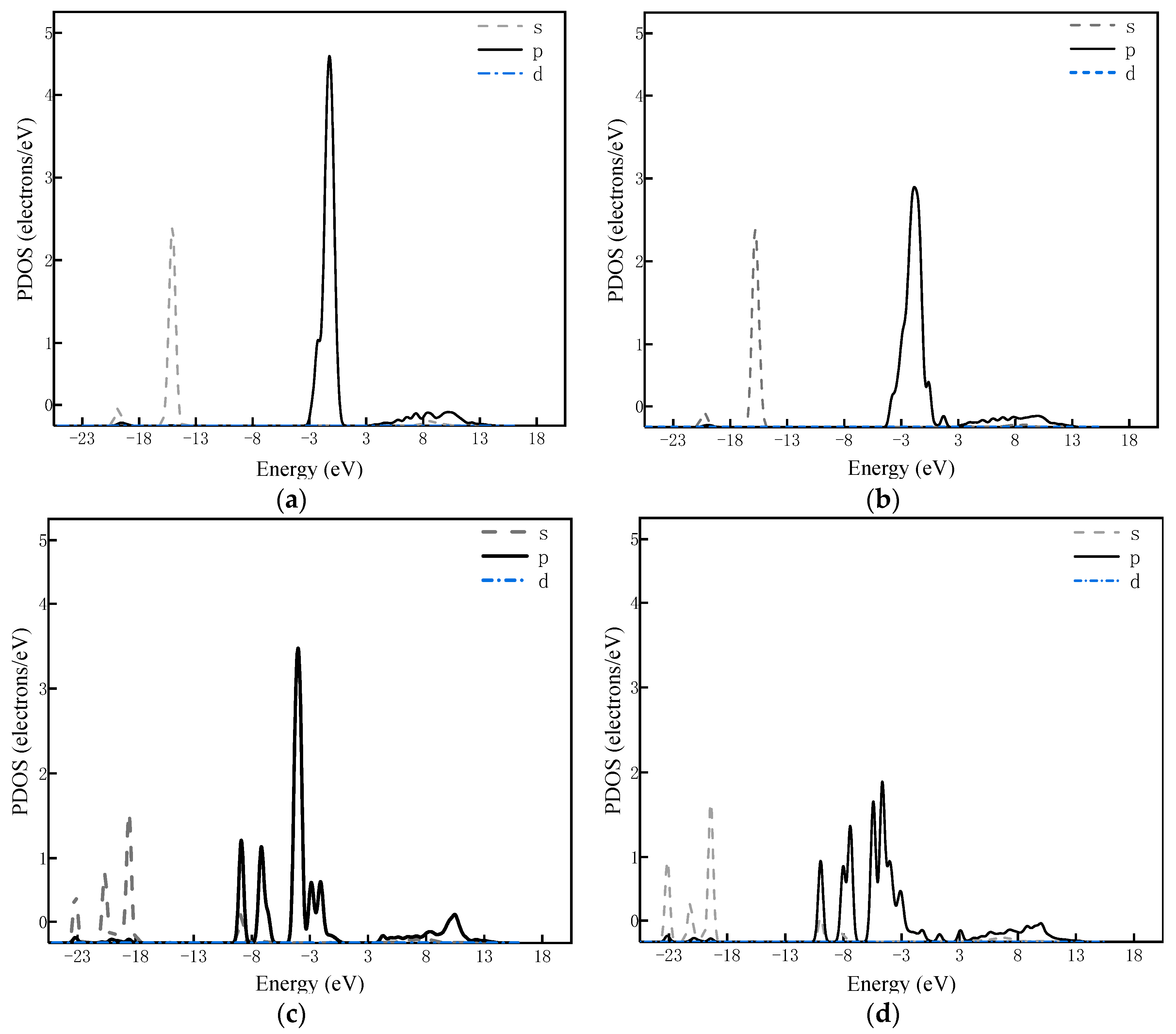
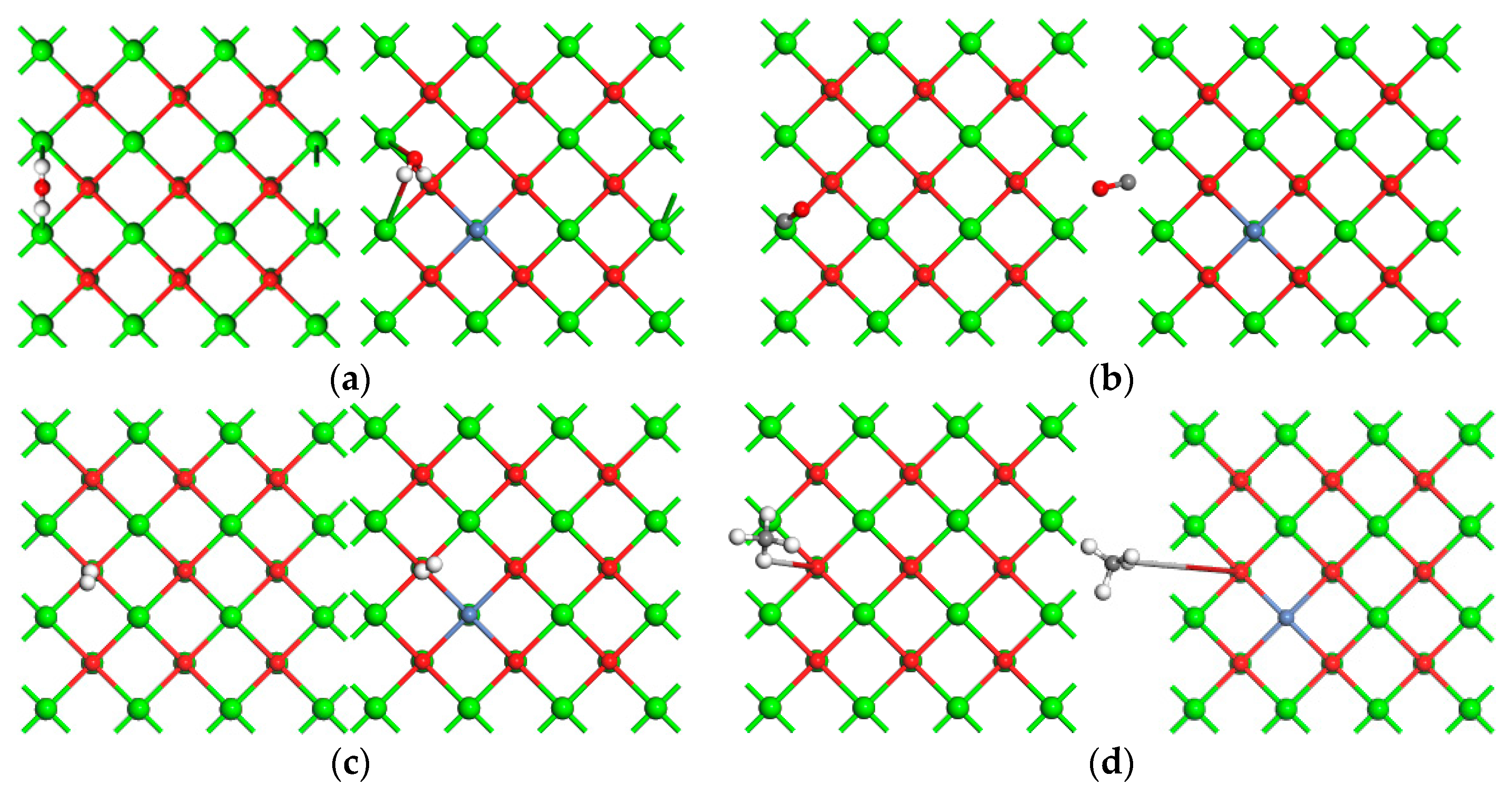
2.3. Adsorption of H2O, CO, H2, and CH4 on Catalysts’ Surfaces
3. Computational and Experimental Methods
3.1. Computational Details
3.2. Experimental Details
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhao, B.; Zhang, X.; Xu, A.; Ding, W.; Sun, L.; Chen, L.; Guan, H.; Yang, S.; Zhou, W. A study of the in-situ CO2 removal pyrolysis of Chinese herb residue for syngas production. Sci. Total Environ. 2018, 626, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Xu, A.; Zhou, W.; Zhang, X.; Zhao, B.; Chen, L.; Sun, L.; Ding, W.; Yang, S.; Guan, H.; Bai, B. Gas production by catalytic pyrolysis of herb residues using Ni/CaO catalysts. J. Anal. Appl. Pyrolysis 2018, 130, 216–223. [Google Scholar] [CrossRef]
- Zhao, B.; Zhang, X.; Chen, L.; Sun, L.; Si, H.; Chen, G. High quality fuel gas from biomass pyrolysis with calcium oxide. Bioresour. Technol. 2014, 156, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Charisiou, N.D.; Baklavaridis, A.; Papadakis, V.G.; Goula, M.A. Synthesis Gas Production via the Biogas Reforming Reaction Over Ni/MgO–Al2O3 and Ni/CaO–Al2O3 Catalysts. Waste Biomass Valorization 2016, 7, 725–736. [Google Scholar] [CrossRef]
- Bennici, S.; Jeguirim, M.; Limousy, L.; Haddad, K.; Vaulot, C.; Michelin, L.; Josien, L.; Zorpas, A.A. Influence of CO2 Concentration and Inorganic Species on the Gasification of Lignocellulosic Biomass Derived Chars. Waste Biomass Valorization 2019, 1–8. [Google Scholar] [CrossRef]
- Wang, M.; Qi, Y.; Ma, R.; Fu, Z.; Ge, P.; Ji, S.; Wu, J.; Qian, X. Investigation of CaO Influences on Fst Gasification Characteristics of Biomass in a Fixed-bed Reactor. Waste Biomass Valorization 2019, 1–8. [Google Scholar]
- Li, J.; Wang, H.; Zhu, Q.; Li, H. Coupling relationship of fluidization behavior, reaction and particle structure of Ni/MgO catalyst toward fluidized CO methanation. Chem. Eng. J. 2019, 357, 298–308. [Google Scholar] [CrossRef]
- Yang, H.; Wang, D.; Li, B.; Zeng, Z.; Qu, L.; Zhang, W.; Chen, H. Effects of potassium salts loading on calcium oxide on the hydrogen production from pyrolysis-gasification of biomass. Bioresour. Technol. 2017, 249, 744–750. [Google Scholar] [CrossRef]
- Khan, Z.; Yusup, S.; Ahmad, M.M. Performance Study of Ni Catalyst with Quicklime (CaO) as CO2 Adsorbent in Palm Kernel Shell Steam Gasification for Hydrogen Production. Adv. Mater. Res. 2014, 917, 283–291. [Google Scholar] [CrossRef]
- Jiang, S.; Lu, Y.; Wang, S.; Zhao, Y.; Ma, X. Insight into the reaction mechanism of CO2 activation for CH4 reforming over NiO-MgO: A combination of DRIFTS and DFT study. Appl. Surf. Sci. 2017, 416, 59–68. [Google Scholar] [CrossRef]
- Naeem, M.A.; Armutlulu, A.; Imtiaz, Q.; Müller, C.R. CaO-Based CO2 Sorbents Effectively Stabilized by Metal Oxides. Chemphyschem 2017, 18, 3280–3285. [Google Scholar] [CrossRef] [PubMed]
- Charisiou, N.D.; Papageridis, K.N.; Tzounis, L.; Sebastian, V.; Hinder, S.J.; Baker, M.A.; AlKetbi, M.; Polychronopoulou, K.; Goula, M.A. Ni supported on CaO-MgO-Al2O3 as a highly selective and stable catalyst for H2 production via the glycerol steam reforming reaction. Int. J. Hydrog. Energy 2018, 8, 256–273. [Google Scholar] [CrossRef]
- Tao, J.; Zhao, L.; Dong, C.; Lu, Q.; Du, X.; Dahlquist, E. Catalytic Steam Reforming of Toluene as a Model Compound of Biomass Gasification Tar Using Ni-CeO2/SBA-15 Catalysts. Energies 2013, 6, 3284–3296. [Google Scholar] [CrossRef]
- Li, B.; Yang, H.; Wei, L.; Shao, J.; Wang, X.; Chen, H. Absorption-enhanced steam gasification of biomass for hydrogen production: Effects of calcium-based absorbents and NiO-based catalysts on corn stalk pyrolysis-gasification. Int. J. Hydrog. Energy 2017, 42, 5840–5848. [Google Scholar] [CrossRef]
- Radfarnia, H.R.; Iliuta, M.C. Development of Al-stabilized CaO–nickel hybrid sorbent–catalyst for sorption-enhanced steam methane reforming. Chem. Eng. Sci. 2014, 109, 212–219. [Google Scholar] [CrossRef]
- Wei, L.; Xu, S.; Liu, J.; Liu, C.; Liu, S. Hydrogen Production in Steam Gasification of Biomass with CaO as a CO2 Absorbent. Energy Fuels 2008, 22, 1997–2004. [Google Scholar] [CrossRef]
- Vanderbilt, D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B Condens. Matter 1990, 41, 7892. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, Y.; Li, Y.; Huang, S.; Qi, J.; Liu, R. A DFT Study on the Adsorption of CO2 Molecules on CaO(001) Surface at Different Coverages. Chin. J. Struct. Chem. 2019, 38, 17–24. [Google Scholar]
- Sun, X.; Li, B.; Metiu, H. Methane Dissociation on Li-, Na-, K-, and Cu-Doped Flat and Stepped CaO(001). J. Phys. Chem. C 2013, 117, 7114–7122. [Google Scholar] [CrossRef]
- Perdew, J.P.; Chevary, J.A.; Vosko, S.H.; Jackson, K.A.; Pederson, M.R.; Singh, D.J.; Fiolhais, C. Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B Condens. Matter 1993, 46, 6671–6687. [Google Scholar] [CrossRef]
- Zhao, L.; Wu, Y.; Han, J.; Lu, Q.; Yang, Y.; Zhang, L. Mechanism of Mercury Adsorption and Oxidation by Oxygen over the CeO2(111) Surface: A DFT Study. Materials 2018, 11, 485. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Gui, Y.; Kang, J.; Wang, W.; Tang, C. A DFT Study on the Adsorption of H2S and SO2 on Ni Doped MoS2 Monolayer. Nanomaterials 2018, 8, 646. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Zhuo, Y.; Lou, Y.; Zhu, Z.; Li, L. SeO2 adsorption on CaO surface: DFT study on the adsorption of a single SeO2 molecule. Appl. Surf. Sci. 2017, 413, 366–371. [Google Scholar] [CrossRef]
- Pan, Y.X.; Liu, C.J.; Ge, Q. Effect of surface hydroxyls on selective CO2 hydrogenation over Ni4/γ-Al2O3: A density functional theory study. J. Catal. 2010, 272, 227–234. [Google Scholar] [CrossRef]
- Besson, R.; Vargas, M.R.; Favergeon, L. CO2 adsorption on calcium oxide: An atomic-scale simulation study. Surf. Sci. 2012, 606, 490–495. [Google Scholar] [CrossRef]
- Yang, C. DFT Study of the Effect of Temperature on ZnO Adsorbed on α-AI2O3(0001) Surface. Chin. J. Chem. Phys. 2006, 19, 137–142. [Google Scholar] [CrossRef]
- Liang, X.Q.; Qiu, L.F.; Huang, P.; Yang, C. A DFT Study of TiO2 Adsorption on GaN(0001) Line Defect Surface. Adv. Mater. Res. 2013, 807, 2836–2841. [Google Scholar] [CrossRef]
- Li, X.; An, M.; Zhu, Y.; Ma, H. First-Principles Study Adsorption Properties Of CO2 Molecule on CaO(100) Surfaces. In Proceedings of the 2nd International Workshop on Materials Engineering and Computer Sciences, Jinan, China, 10–11 October 2015; Volume 33, pp. 267–270. [Google Scholar]
- Wang, W.; Fan, L.; Wang, G.; Li, Y. CO2 and SO2 sorption on the alkali metals doped CaO(100) surface: A DFT-D study. Appl. Surf. Sci. 2017, 425, 972–977. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, X.; Lei, C.; Sun, L.; Xie, X.; Zhao, B. Production of syngas from pyrolysis of biomass using Fe/CaO catalysts: Effect of operating conditions on the process. J. Anal. Appl. Pyrolysis 2017, 125, 1–8. [Google Scholar] [CrossRef]
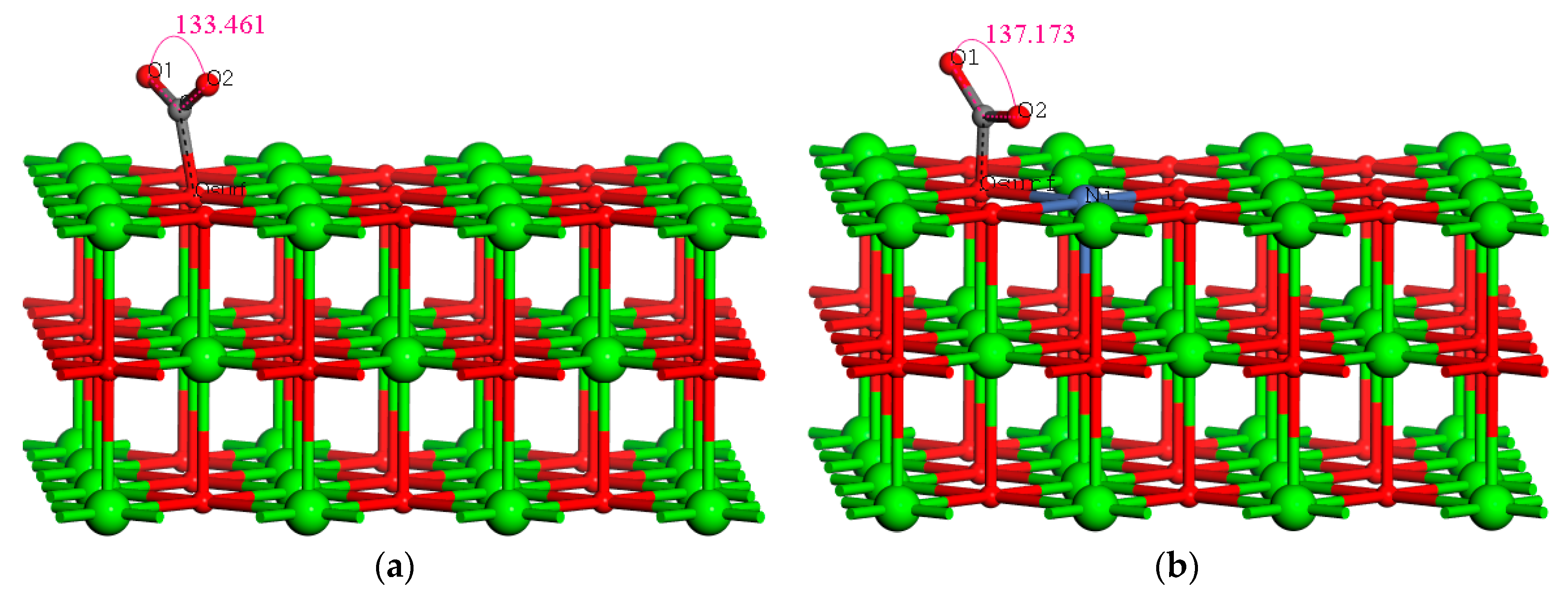
| Temperature (°C) | Bond Length (Å) C–Osurf | Bond Angle (°) O1–C–O2 | Eads(eV) | |
|---|---|---|---|---|
| CaO | 550 | 1.529 | 121.924 | 1.39 |
| 600 | 1.517 | 127.350 | 1.41 | |
| 650 | 1.487 | 133.461 | 1.45 | |
| 700 | 1.493 | 138.341 | 1.43 | |
| 10 wt % Ni/CaO | 550 | 1.392 | 132.604 | 1.69 |
| 600 | 1.373 | 136.249 | 1.74 | |
| 650 | 1.343 | 137.173 | 1.81 | |
| 700 | 1.357 | 127.228 | 1.77 |
| Orbital Population | CO2 | CO2 on CaO (100) | CO2 on 10 wt % Ni/CaO (100) |
|---|---|---|---|
| C–O1 | 1.15 | 0.97 | 0.92 |
| C–O2 | 1.15 | 0.97 | 0.84 |
| Eads (eV) | CO2 | H2O | CO | H2 | CH4 |
|---|---|---|---|---|---|
| CaO | 1.45 | 2.05 | 0.361 | 0.136 | 0.199 |
| 10 wt % Ni/CaO | 1.81 | 2.23 | 0.461 | 0.162 | 0.183 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, B.; Wang, J.; Zhu, D.; Song, G.; Yang, H.; Chen, L.; Sun, L.; Yang, S.; Guan, H.; Xie, X. Adsorption Characteristics of Gas Molecules (H2O, CO2, CO, CH4, and H2) on CaO-Based Catalysts during Biomass Thermal Conversion with in Situ CO2 Capture. Catalysts 2019, 9, 757. https://doi.org/10.3390/catal9090757
Zhao B, Wang J, Zhu D, Song G, Yang H, Chen L, Sun L, Yang S, Guan H, Xie X. Adsorption Characteristics of Gas Molecules (H2O, CO2, CO, CH4, and H2) on CaO-Based Catalysts during Biomass Thermal Conversion with in Situ CO2 Capture. Catalysts. 2019; 9(9):757. https://doi.org/10.3390/catal9090757
Chicago/Turabian StyleZhao, Baofeng, Jingwei Wang, Di Zhu, Ge Song, Huajian Yang, Lei Chen, Laizhi Sun, Shuangxia Yang, Haibin Guan, and Xinping Xie. 2019. "Adsorption Characteristics of Gas Molecules (H2O, CO2, CO, CH4, and H2) on CaO-Based Catalysts during Biomass Thermal Conversion with in Situ CO2 Capture" Catalysts 9, no. 9: 757. https://doi.org/10.3390/catal9090757
APA StyleZhao, B., Wang, J., Zhu, D., Song, G., Yang, H., Chen, L., Sun, L., Yang, S., Guan, H., & Xie, X. (2019). Adsorption Characteristics of Gas Molecules (H2O, CO2, CO, CH4, and H2) on CaO-Based Catalysts during Biomass Thermal Conversion with in Situ CO2 Capture. Catalysts, 9(9), 757. https://doi.org/10.3390/catal9090757





