H3PW12O40/SBA-15 for the Solventless Synthesis of 3-Substituted Indoles
Abstract
:1. Introduction
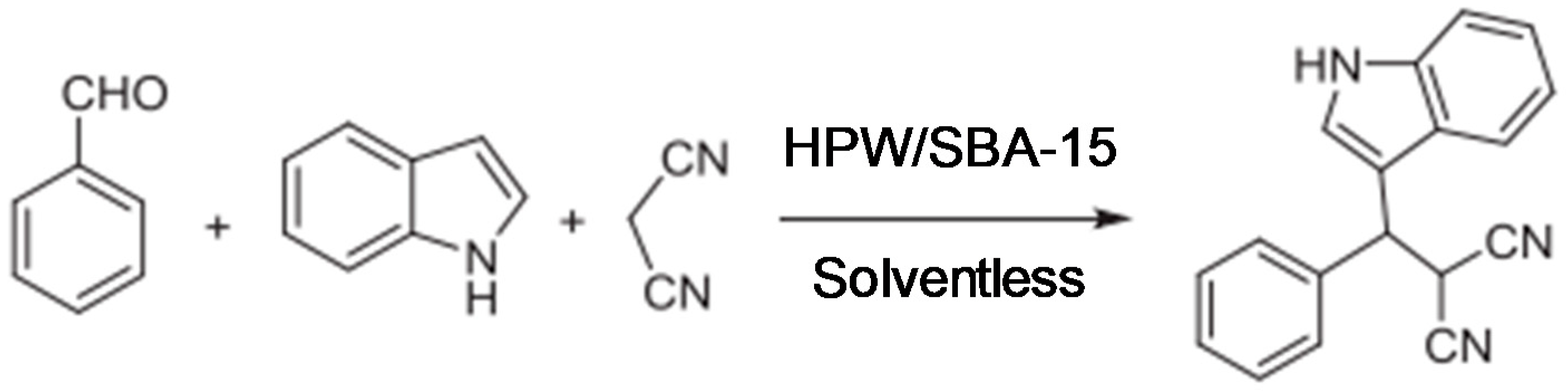
2. Results and Discussion
2.1. Physicochemical Properties of HPW/SBA-15
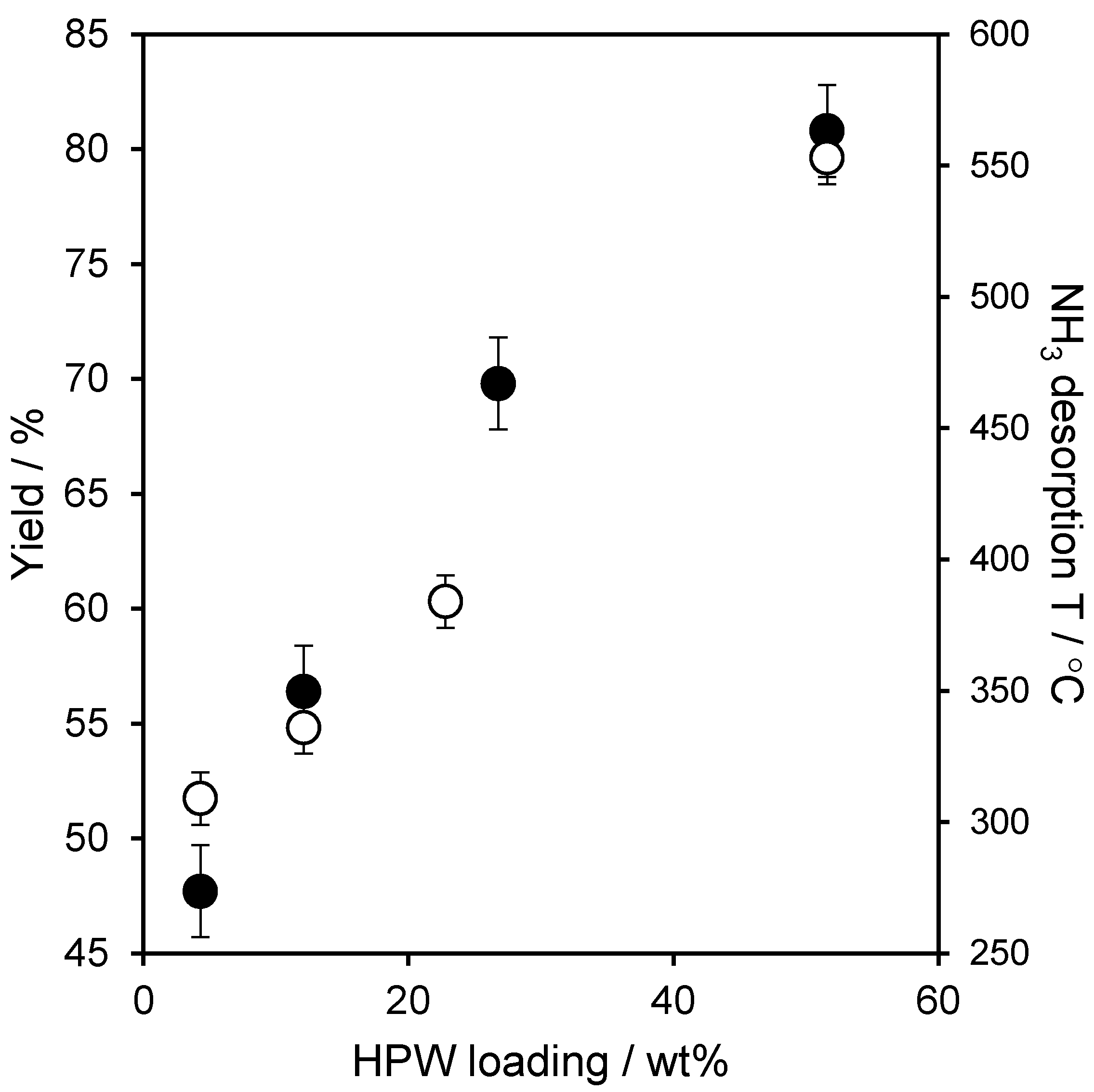
2.2. Catalytic Performance
3. Experimental
3.1. Catalyst Synthesis
3.2. Catalyst Characterization and Synthesis of 2-[(1H-Indol-3-yl)(Aryl)methyl]Malononitriles
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Taguchi, A.; Schüth, F. Ordered mesoporous materials in catalysis. Micropor. Mesopor. Mater. 2005, 77, 1–45. [Google Scholar] [CrossRef]
- Corma, A. Inorganic solid acids and their use in acid-catalyzed hydrocarbon reactions. Chem. Rev. 1995, 95, 559–614. [Google Scholar] [CrossRef]
- Rinaldi, R.; Schüth, F. Design of solid catalysts for the conversion of biomass. Energy Environ. Sci. 2009, 2, 610–626. [Google Scholar] [CrossRef]
- Dacquin, J.-P.; Pirez, C.; Lee, A.F.; Wilson, K. Pore-expanded SBA-15 sulfonic acid silicas for biodiesel synthesis. Chem. Commun. 2012, 48, 212–214. [Google Scholar] [CrossRef] [PubMed]
- Tayebee, R.; Mohammadzadeh Kakhki, R.; Audebert, P.; Amini, M.M.; Salehi, M.; Mahdizadeh Ghohe, N.; Karimipour, G.R. A robust UV–visible light-driven SBA-15-PS/phthalhydrazide nanohybrid material with enhanced photocatalytic activity in the photodegradation of methyl orange. Appl. Organomet. Chem. 2018, 32, e4391. [Google Scholar] [CrossRef]
- Morales, G.; Paniagua, M.; Iglesias, J.; Hernández, B.; Wilson, K.; Lee, A.F.; Brown, D.R.; Granollers, M.; Osatiashtiani, A.; Melero, J.A. Conformal sulfated zirconia monolayer catalysts for the one-pot synthesis of ethyl levulinate from glucose. Chem. Commun. 2014, 50, 11742–11745. [Google Scholar] [CrossRef] [PubMed]
- Tayebee, R.; Amini, M.M.; Pouyamanesh, S.; Aliakbari, A. A new inorganic–organic hybrid material Al-SBA-15-TPI/H6P2W18O62 catalyzed one-pot, three-component synthesis of 2H-indazolo [2,1-b] phthalazine-triones. Dalton Trans. 2015, 44, 5888–5897. [Google Scholar] [CrossRef] [PubMed]
- Julião, D.; Mirante, F.; Ribeiro, S.O.; Gomes, A.C.; Valença, R.; Ribeiro, J.C.; Balula, S.S. Deep oxidative desulfurization of diesel fuels using homogeneous and SBA-15-supported peroxophosphotungstate catalysts. Fuel 2019, 241, 616–624. [Google Scholar] [CrossRef]
- Lucas, N.; Gurrala, L.; Athawale, A. Heteropolyacids supported on mesoporous AlSBA-15 as efficient catalysts for esterification of levulinic acid. J. Por. Mater. 2019. [Google Scholar] [CrossRef]
- Eicher, T.; Hauptmann, S.; Speicher, A. The Chemistry of Heterocycles: Structures, Reactions, Synthesis, and Applications; Wiley-VCH: Weinheim, Germany, 2013; pp. 125–147. [Google Scholar]
- Ziarani, G.M.; Moradi, R.; Ahmadi, T.; Lashgari, N. Recent advances in the application of indoles in multicomponent reactions. RSC Adv. 2018, 8, 12069–12103. [Google Scholar] [CrossRef]
- Anselmo, D.; Adán, E.C.E.; Belmonte, M.M.; Kleij, A.W. Zn-Mediated synthesis of 3-substituted indoles using a three-component reaction approach. Eur. J. Inorg. Chem. 2012, 4694. [Google Scholar] [CrossRef]
- Chen, W.; Cai, Y.; Fu, X.; Liu, X.; Lin, L.; Feng, X. Enantioselective one-pot synthesis of 2-amino-4-(indol-3-yl)-4H-chromenes. Org. Lett. 2011, 13, 4910. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Ke, F.; Zhou, L.; Li, Z.; Xiang, H.; Wu, D.; Zhou, X. Synthesis of 3-indole derivatives by copper sulfonato Salen catalyzed three-component reactions in water. Chem. Commun., 2011, 47, 3912. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Allam, B.K.; Raghuvanshi, D.S.; Singh, K.N. An efficient tetrabutylammonium fluoride (TBAF)-catalyzed three-component synthesis of 3-substituted indole derivatives under solvent-free conditions. Adv. Synth. Catal. 2013, 355, 1840. [Google Scholar] [CrossRef]
- Frattini, L.; Isaacs, M.A.; Parlett, C.M.; Wilson, K.; Kyriakou, G.; Lee, A.F. Support enhanced α-pinene isomerization over HPW/SBA-15. Appl. Catal. B Environ. 2017, 200, 10–18. [Google Scholar] [CrossRef]
- Bardin, B.B.; Bordawekar, S.V.; Neurock, M.; Davis, R.J. Acidity of Keggin-Type Heteropolycompounds Evaluated by Catalytic Probe Reactions, Sorption Microcalorimetry, and Density Functional Quantum Chemical Calculations. J. Phys. Chem. B 1998, 102, 10817–10825. [Google Scholar] [CrossRef]
- Wang, L.; Huang, M.; Zhu, X.; Wan, Y. Polyethylene glycol (PEG-200)-promoted sustainable one-pot three-component synthesis of 3-indole derivatives in water. Appl. Catal. A Gen. 2013, 454, 160–163. [Google Scholar] [CrossRef]
- He, Y.-H.; Cao, J.-F.; Li, R.; Xiang, Y.; Yang, D.-C.; Guan, Z. L-Proline-catalyzed multicomponent synthesis of 3-indole derivatives. Tetrahedron 2015, 71, 9299–9306. [Google Scholar] [CrossRef]
- Rajesh, U.C.; Kholiya, R.; Thakur, A.; Rawat, D.S. [TBA][Gly] ionic liquid promoted multi-component synthesis of 3-substituted indoles and indolyl-4H-chromenes. Tetrahedron Lett. 2015, 56, 1790–1793. [Google Scholar] [CrossRef]
- Ghohe, N.M.; Tayebee, R.; Amini, M.M.; Osatiashtiani, A.; Isaacs, M.A.; Lee, A.F. H5PW10V2O40@VOx/SBA-15-NH2 catalyst for the solventless synthesis of 3-substituted indoles. Tetrahedron 2017, 73, 5862–5871. [Google Scholar] [CrossRef]
| Catalyst | Bulk W Loading a (wt %) | Particle Size b (nm) [16] | Acid Loading c (mmol·g−1) |
|---|---|---|---|
| 4.3 wt % HPW/SBA-15 | 3.2 | 10.5 | 0.21 |
| 12.1 wt % HPW/SBA-15 | 9.0 | 10.6 | 0.24 |
| 26.8 wt % HPW/SBA-15 | 19.9 | 10.7 | 0.29 |
| 51.6 wt % HPW/SBA-15 | 38.2 | 10.7 | 0.30 |
| Catalyst | Product Yield (%) |
|---|---|
| - | 9 |
| H3PW12O40 | 44(17) a |
| 4.3 wt % HPW/SBA-15 | 47 |
| 12.1 wt % HPW/SBA-15 | 56 |
| 25.2 wt % HPW/SBA-15 | 67 |
| 51.6 wt % HPW/SBA-15 | 81 |
| Entry | Aldehyde | Product | Yield (%) | m.p. (oC) |
|---|---|---|---|---|
| 1 | 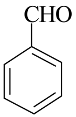 |  | 81 | 201–203 |
| 2 | 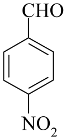 |  | 89 | 203–204 |
| 3 | 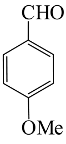 |  | 76 | 208–210 |
| 4 | 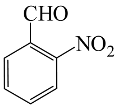 | 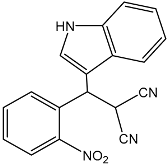 | 92 | 203–205 |
| 5 |  | 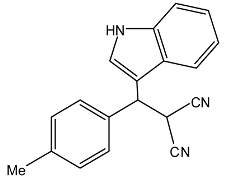 | 67 | 209–211 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tayebee, R.; Lee, A.F.; Frattini, L.; Rostami, S. H3PW12O40/SBA-15 for the Solventless Synthesis of 3-Substituted Indoles. Catalysts 2019, 9, 409. https://doi.org/10.3390/catal9050409
Tayebee R, Lee AF, Frattini L, Rostami S. H3PW12O40/SBA-15 for the Solventless Synthesis of 3-Substituted Indoles. Catalysts. 2019; 9(5):409. https://doi.org/10.3390/catal9050409
Chicago/Turabian StyleTayebee, Reza, Adam F. Lee, Lucia Frattini, and Shahrebano Rostami. 2019. "H3PW12O40/SBA-15 for the Solventless Synthesis of 3-Substituted Indoles" Catalysts 9, no. 5: 409. https://doi.org/10.3390/catal9050409
APA StyleTayebee, R., Lee, A. F., Frattini, L., & Rostami, S. (2019). H3PW12O40/SBA-15 for the Solventless Synthesis of 3-Substituted Indoles. Catalysts, 9(5), 409. https://doi.org/10.3390/catal9050409






