Performance of Mn-Fe-Ce/GO-x for Catalytic Oxidation of Hg0 and Selective Catalytic Reduction of NOx in the Same Temperature Range
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Catalysts
2.2. The Performance of the Prepared Materials
2.3. Effect of Individual Flue Gas Components
2.3.1. The Effect of O2 on Mercury Oxidation over the Ce-Mn-Fe/GO-20% Catalyst
2.3.2. The Effect of SO2 on Mercury Oxidation over the Ce-Mn-Fe/GO-20% Catalyst
2.3.3. The Effect of NO on Mercury Oxidation over the Ce-Mn-Fe/GO-20% Catalyst
2.3.4. The Effect of Water Vapor on Mercury Oxidation over the Ce-Mn-Fe/GO-20% Catalyst
2.4. Effect of Individual Flue Gas Components on NOx Removal over the Ce-Mn-Fe/GO-20% Catalyst under NH3
2.5. Proposed Hg0 Oxidation Mechanism
2.6. Regeneration
3. Experimental
3.1. Catalyst Preparation
3.2. Material Characterizations
3.3. Hg0 Removal Test
3.4. NH3-SCR Catalytic Activity Measurement
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, D.Y.; Zhao, S.J.; Qu, Z.; Yan, N.Q. Cu-BTC as a novel material for elemental mercury removal from sintering gas. Fuel 2018, 217, 297–305. [Google Scholar] [CrossRef]
- Zhao, B.; Yi, H.H.; Tang, X.L.; Li, Q.; Liu, D.D.; Gao, F.Y. Copper modified activated coke for mercury removal from coal-fired flue gas. Chem. Eng. J. 2016, 286, 585–593. [Google Scholar] [CrossRef]
- Pavlish, J.H.; Sondreal, E.A.; Mann, M.D.; Olson, E.S.; Galbreath, K.C.; Laudal, D.L.; Benson, S.A. Status review of mercury control options for coal-fired power plants. Fuel Process. Technol. 2003, 82, 89–165. [Google Scholar] [CrossRef]
- Xu, W.; Shao, M.; Yang, Y.; Liu, R.; Wu, Y.; Zhu, T. Mercury emission from sintering process in the iron and steel industry of china. Fuel Process. Technol. 2017, 159, 340–344. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Mason, R.P.; Chan, H.M.; Jacob, D.J.; Pirrone, N. Mercury as a global pollutant: Sources, pathways, and effects. Environ. Sci. Technol. 2013, 47, 4967–4983. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, C.B.; Liu, H.M. Experiment and mechanism research on gas-phase As2O3 adsorption of Fe2O3/γ-Al2O3. Fuel 2016, 181, 1034–1040. [Google Scholar] [CrossRef]
- Xu, M.H.; Qiao, Y.; Zhang, C.G.; Li, L.C.; Liu, J. Modeling of homogeneous mercury speciation using detailed chemical kinetics. Combust. Flame. 2003, 132, 208–218. [Google Scholar] [CrossRef]
- Zhang, S.B.; Zhao, Y.C.; Yang, J.P.; Zhang, Y.; Sun, P.; Yu, X.H.; Zhang, J.Y.; Zheng, C.G. Simultaneous NO and mercury removal over MnOx/TiO2 catalyst in different atmospheres. Fuel Process. Technol. 2017, 166, 282–290. [Google Scholar] [CrossRef]
- Yang, J.P.; Zhao, Y.C.; Chang, L.; Zhang, J.Y.; Zheng, C.G. Mercury adsorption and oxidation over cobalt oxide loaded magnetospheres catalyst from fly ash in oxyfuel combustion flue gas. Environ. Sci. Technol. 2015, 49, 8210–8218. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.N.; Duan, Y.F.; Wang, H.; Zhao, S.L.; Chen, M.M.; Liu, M.; Wei, H.Q. Effects of acidic gases on mercury adsorption by activated carbon in simulated oxy-fuel combustion flue gas. Energy Fuels 2017, 31, 9745–9751. [Google Scholar] [CrossRef]
- Pacyna, E.G.; Pacyna, J.M.; Sundseth, K.; Munthe, J.; Kindbom, K.; Wilson, S.; Steenhuisen, F.; Maxson, P. Global emission of mercury to the atmosphere from anthropogenic sources in 2005 and projections to 2020. Atmos. Environ. 2010, 44, 2487–2499. [Google Scholar] [CrossRef]
- Cai, J.H.; Jia, C.Q. Mercury removal from aqueous solution using coke-derived sulfur-impregnated activated carbons. Ind. Eng. Chem. Res. 2010, 49, 2716–2721. [Google Scholar] [CrossRef]
- Xu, W.Q.; Wang, H.R.; Zhu, T.Y.; Kuang, J.Y.; Jing, P.F. Mercury removal from coal combustion flue gas by modified fly ash. J. Environ. Sci. 2013, 25, 393–398. [Google Scholar] [CrossRef]
- Zhang, M.Z.; Wang, P.; Dong, Y.; Sui, H.; Xiao, D.D. Study of elemental mercury oxidation over an SCR catalyst with calcium chloride addition. Chem. Eng. J. 2014, 253, 243–250. [Google Scholar] [CrossRef]
- Straube, S.; Hahn, T.; Koeser, H. Adsorption and oxidation of mercury in tail-end SCR-DeNOx plants-bench scale investigations and speciation experiments. Appl. Catal. B Environ. 2008, 79, 286–295. [Google Scholar] [CrossRef]
- Mei, Z.J.; Shen, Z.M.; Zhao, Q.J.; Wang, W.H.; Zhang, Y.J. Removal and recovery of gas-phase element mercury by metal oxide-loaded activated carbon. J. Hazard. Mater. 2008, 152, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Pavlish, J.H.; Hamre, L.L.; Zhuang, Y. Mercury control technologies for coal combustion and gasification systems. Fuel 2010, 89, 838–847. [Google Scholar] [CrossRef]
- Li, C.W.; Zhang, A.C.; Zhang, L.X.; Song, J.; Su, S.; Sun, Z.J.; Xiang, J. Enhanced photocatalytic activity and characterization of magnetic Ag/BiOI/ZnFe2O4 composites for Hg0 removal under fluorescent light irradiation. Appl. Surf. Sci. 2018, 433, 914–926. [Google Scholar] [CrossRef]
- Gao, Y.S.; Zhang, Z.; Wu, J.W.; Duan, L.H.; Umar, A.; Sun, L.Y.; Guo, Z.H.; Wang, Q. A critical review on the heterogeneous catalytic oxidation of elemental mercury in flue gases. Environ. Sci. Technol. 2013, 47, 10813–10823. [Google Scholar] [CrossRef] [PubMed]
- Li, H.L.; Zhu, L.; Wu, S.K.; Liu, Y.; Shih, K. Synergy of CuO and CeO2 combination for mercury oxidation under low-temperature selective catalytic reduction atmosphere. Int. J. Coal Geol. 2017, 170, 69–76. [Google Scholar] [CrossRef]
- Liu, R.; Xu, W.; Tong, L.; Zhu, T. Role of NO in Hg0 oxidation over a commercial selective catalytic reduction catalyst V2O5–WO3/TiO2. J. Environ. Sci. 2015, 38, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Yin, L.B.; Zhuo, Y.Q.; Xu, Q.S.; Zhang, L.; Chen, C.H. Performance of CuOx-neutral Al2O3 sorbents on mercury removal from simulated coal combustion flue gas. Fuel Process. Technol. 2015, 131, 403–408. [Google Scholar] [CrossRef]
- Chen, G.Q.; Gao, J.; Xu, L.; Fu, X.; Yin, Y.; Wu, S.; Qin, Y. Optimizing conditions for preparation of MnOx/RHA catalyst particle for the catalytic oxidation of No. Adv. Powder Technol. 2012, 23, 256–263. [Google Scholar] [CrossRef]
- He, C.; Shen, B.X.; Chen, J.H.; Cai, J. Adsorption and oxidation of elemental mercury over Ce-MnOx/Ti-PILCs. Environ. Sci. Technol. 2014, 48, 7891–7898. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.K.; Li, H.L.; Li, L.Q.; Wu, C.Y.; Zhang, J.Y.; Shih, K. Effects of flue-gas parameters on low temperature NO reduction over a Cu-promoted CeO2–TiO2 catalyst. Fuel 2015, 159, 876–882. [Google Scholar] [CrossRef]
- Zhang, A.C.; Zhang, Z.H.; Chen, J.J.; Sheng, W.; Sun, L.S.; Xiang, J. Effect of calcination temperature on the activity and structure of MnOx/TiO2 adsorbent for Hg0 removal. Fuel Process. Technol. 2015, 135, 25–33. [Google Scholar] [CrossRef]
- Liu, T.; Man, C.Y.; Guo, X.; Zheng, C.G. Experimental study on the mechanism of mercury removal with Fe2O3. Fuel 2016, 173, 209–216. [Google Scholar] [CrossRef]
- Li, H.L.; Wu, C.Y.; Li, Y.; Zhang, J.Y. Superior activity of MnOx-CeO2/TiO2 catalyst for catalytic oxidation of elemental mercury at low flue gas temperatures. Appl. Catal. B Environ. 2012, 111–112, 381–388. [Google Scholar] [CrossRef]
- Qi, G.; Yang, R.T. Characterization and FTIR studies of MnOx-CeO2 catalyst for low-temperature selective catalytic reduction of NO with NH3. J. Phy. Chem. B. 2004, 108, 15738–15747. [Google Scholar] [CrossRef]
- Yang, S.J.; Guo, Y.F.; Yan, N.Q.; Qu, Z.; Xie, J.K.; Yang, C.; Jia, J.P. Capture of gaseous elemental mercury from flue gas using a magnetic and sulfur poisoning resistant sorbent Mn/γ-Fe2O3 at lower temperatures. J. Hazard. Mater. 2011, 186, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.M.; Qu, Z.; Zong, C.X.; Huang, W.J.; Quan, F.Q.; Yan, N.Q. MnOx/graphene for the catalytic oxidation and adsorption of elemental mercury. Environ. Sci. Technol. 2015, 49, 6823–6830. [Google Scholar] [CrossRef] [PubMed]
- Li, J.R.; Chen, J.S.; Yu, Y.K.; He, C. Fe–Mn–Ce/ceramic powder composite catalyst for highly volatile elemental mercury removal in simulated coal-fired flue gas. J. Ind. Eng. Chem. 2015, 25, 352–358. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, V.V. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Georgakilas, V.; Otyepka, M.; Bourlinos, A.B.; Chandra, V.; Kim, N.; Kemp, K.C.; Hobza, P.; Zboril, R.; Kim, K.S. Functionalization of graphene: covalent and non-covalent approaches, derivatives and applications. Chem. Rev. 2012, 112, 6156–6214. [Google Scholar] [CrossRef] [PubMed]
- Li, H.N.; Zhu, M.Y.; Chen, W.; Xu, L.; Wang, K. Non-light-driven reduced graphene oxide anchored TiO2 nanocatalysts with enhanced catalytic oxidation performance. J. Colloid Interface Sci. 2017, 507, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.Y.; Shi, L.; He, C.X.; Gao, F.; Li, B.B.; Zhou, Q.; Hu, H.; Shao, G.H.; Wang, X.Z.; Qiu, J.S. Highly efficient synthesis of graphene/MnO2 hybrids and their application for ultrafast oxidative decomposition of methylene blue. Carbon 2014, 66, 485–492. [Google Scholar] [CrossRef]
- Qiao, S.H.; Chen, J.; Li, J.F.; Qu, Z.; Liu, P.; Yan, N.Q.; Jia, J.P. Adsorption and catalytic oxidation of gaseous elemental mercury in flue gas over MnOx/alumina. Ind. Eng. Chem. Res. 2009, 48, 3317–3322. [Google Scholar] [CrossRef]
- Wang, J.G.; Yi, H.H.; Tang, X.L.; Zhao, S.Z.; Gao, F.Y.; Yang, Z.Y. Oxygen plasma-catalytic conversion of NO over MnOx: Formation and reactivity of adsorbed oxygen. Catal. Commun. 2017, 100, 227–231. [Google Scholar] [CrossRef]
- Reddy, B.M.; Khan, A.; Yamada, Y.; Kobayashi, T.; Loridant, S.; Volta, J.-C. Structural characterization of CeO2-TiO2 and V2O5/CeO2−TiO2 catalysts by raman and XPS techniques. J. Phys. Chem. B 2003, 107, 5162–5167. [Google Scholar] [CrossRef]
- Zhou, Z.J.; Liu, X.W.; Hu, Y.C.; Liao, Z.Q.; Cheng, S.; Xu, M.H. An efficient sorbent based on CuCl2 loaded CeO2-ZrO2 for elemental mercury removal from chlorine-free flue gas. Fuel 2018, 216, 356–363. [Google Scholar] [CrossRef]
- Ding, Z.Y.; Li, L.X.; Wade, D.; Gloyna, E.F. Supercritical water oxidation of NH3 over a MnO2. Ind. Eng. Chem. Res. 1998, 37, 1707–1716. [Google Scholar] [CrossRef]
- Li, H.H.; Wang, Y.; Wang, S.K.; Wang, X.; Hu, J.J. Removal of elemental mercury in flue gas at lower temperatures over Mn-Ce based materials prepared by co-precipitation. Fuel 2017, 208, 576–586. [Google Scholar] [CrossRef]
- Wang, L.Y.; Cheng, X.X.; Wang, Z.Q.; Ma, C.Y.; Qin, Y.K. Investigation on Fe-Co binary metal oxides supported on activated semi-coke for NO reduction by Co. Appl. Catal. B Environ. 2017, 201, 636–651. [Google Scholar] [CrossRef]
- Xing, L.L.; Xu, Y.L.; Zhong, Q. Mn and Fe modified fly ash as a superior catalyst for elemental mercury capture under air conditions. Energy Fuels 2012, 26, 4903–4909. [Google Scholar] [CrossRef]
- Kang, M.; Park, E.D.; Kim, J.M.; Yie, J.E. Manganese oxide catalysts for NOx reduction with NH3 at low temperatures. Appl. Catal. A Gen. 2007, 327, 261–269. [Google Scholar] [CrossRef]
- Dupin, J.C.; Gonbeau, D.; Vinatier, P.; Levasseur, A. Systematic XPS studies of metal oxides, hydroxides and peroxides. Phys. Chem. Chem. Phys. 2000, 2, 1319–1324. [Google Scholar] [CrossRef]
- Zhao, Y.C.; Zhang, J.Y.; Tian, C.; Li, H.L.; Shao, X.Y.; Zheng, C.G. Mineralogy and chemical composition of high-calcium fly ashes and density fractions from a coal-fired power plant in china. Energy Fuels 2010, 24, 834–843. [Google Scholar] [CrossRef]
- Gao, G.; Shi, J.W.; Liu, C.; Gao, C.; Fan, Z.Y.; Niu, C.M. Mn/CeO2 catalysts for SCR of NOx with NH3: Comparative study on the effect of supports on low-temperature catalytic activity. Appl. Surf. Sci. 2017, 411, 338–346. [Google Scholar] [CrossRef]
- Ndifor, E.N.; Garcia, T.; Solsona, B.; Taylor, S.H. Influence of preparation conditions of nano-crystalline ceria catalysts on the total oxidation of naphthalene, a model polycyclic aromatic hydrocarbon. Appl. Catal. B: Environ. 2007, 76, 248–256. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Ma, C.Y.; Cheng, X.X.; Wang, Z.Q. Performance of Fe-Ba/ZSM-5 catalysts in NO + O2 adsorption and NO + CO reduction. Int. J. Hydrogen Energy 2017, 42, 7077–7088. [Google Scholar] [CrossRef]
- Zhang, A.C.; Xing, W.B.; Zhang, Z.H.; Meng, F.M.; Liu, Z.C.; Xiang, J.; Sun, L.S. Promotional effect of SO2 on CeO2-TiO2 material for elemental mercury removal at low temperature. Atmos. Pollut. Res. 2016, 7, 895–902. [Google Scholar] [CrossRef]
- Busca, G.; Lietti, L.; Ramis, G.; Berti, F. Chemical and mechanistic aspects of the selective catalytic reduction of NOx by ammonia over oxide catalysts: A review. Appl. Catal. B Environ. 1998, 18, 1–36. [Google Scholar] [CrossRef]
- Zheng, Y.J.; Jensen, A.D.; Windelin, C.; Jensen, F. Review of technologies for mercury removal from flue gas from cement production processes. Prog. Energy Combust. Sci. 2012, 38, 599–629. [Google Scholar] [CrossRef]
- Liu, Z.M.; Liu, H.Y.; Zeng, H.; Xu, Q. A novel Ce-Sb binary oxide catalyst for the selective catalytic reduction of NOx with NH3. Catal. Sci. Technol. 2016, 6, 8063–8071. [Google Scholar] [CrossRef]
- Miser, D.E.; Shin, E.J.; Hajaligol, M.R.; Rasouli, F. HRTEM characterization of phase changes and the occurrence of maghemite during catalysis by an iron oxide. Appl. Catal. A Gen. 2004, 258, 7–16. [Google Scholar] [CrossRef]
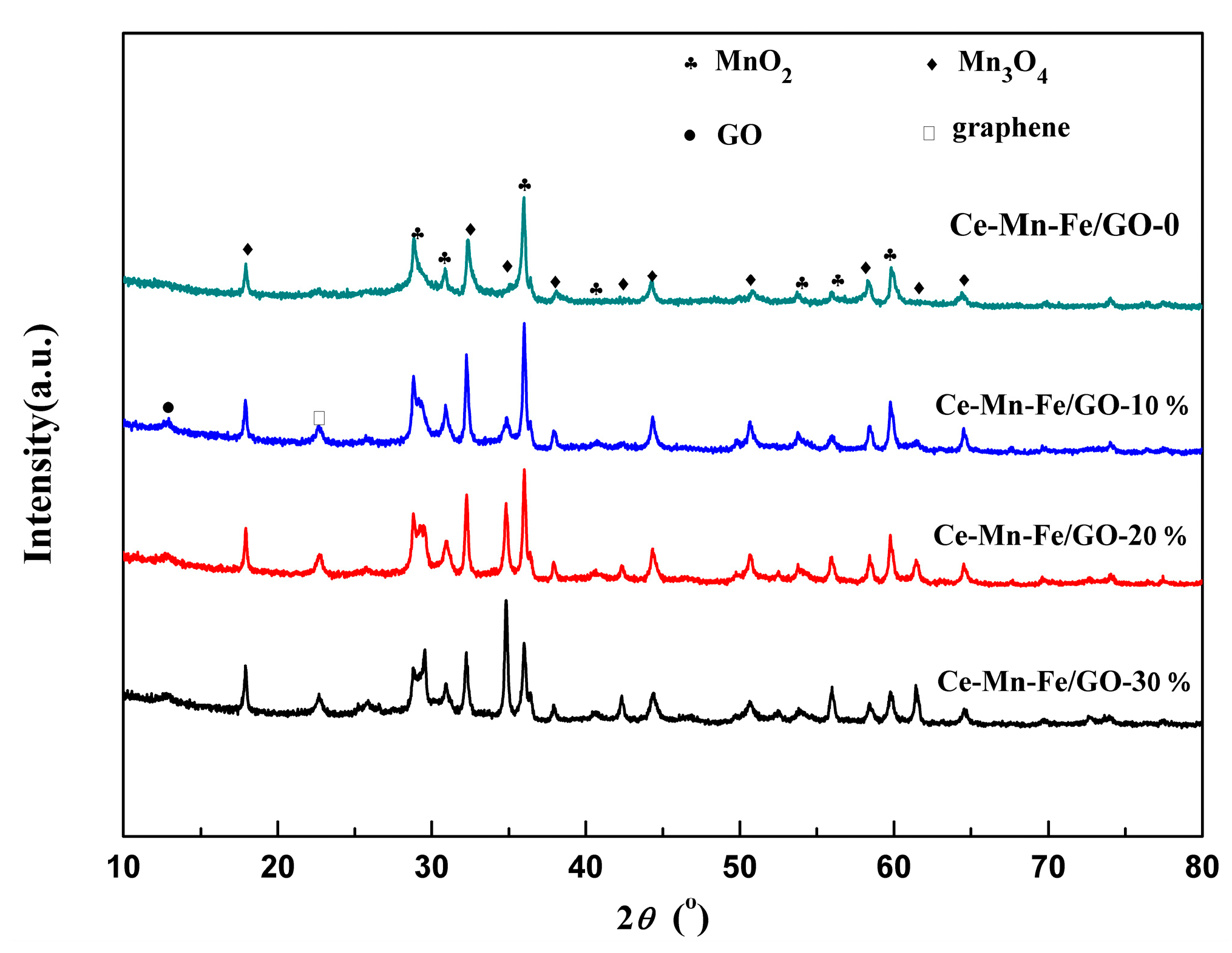
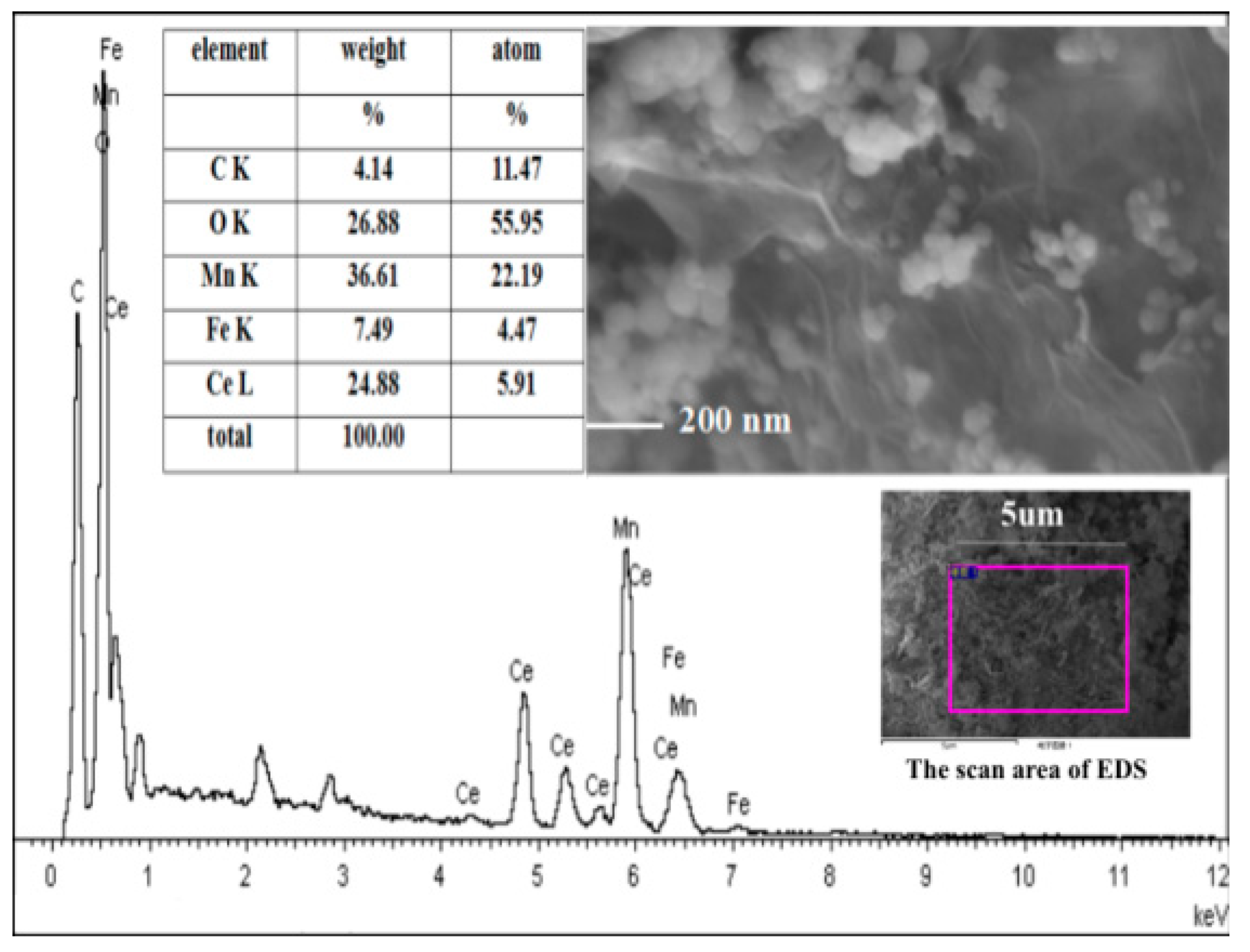
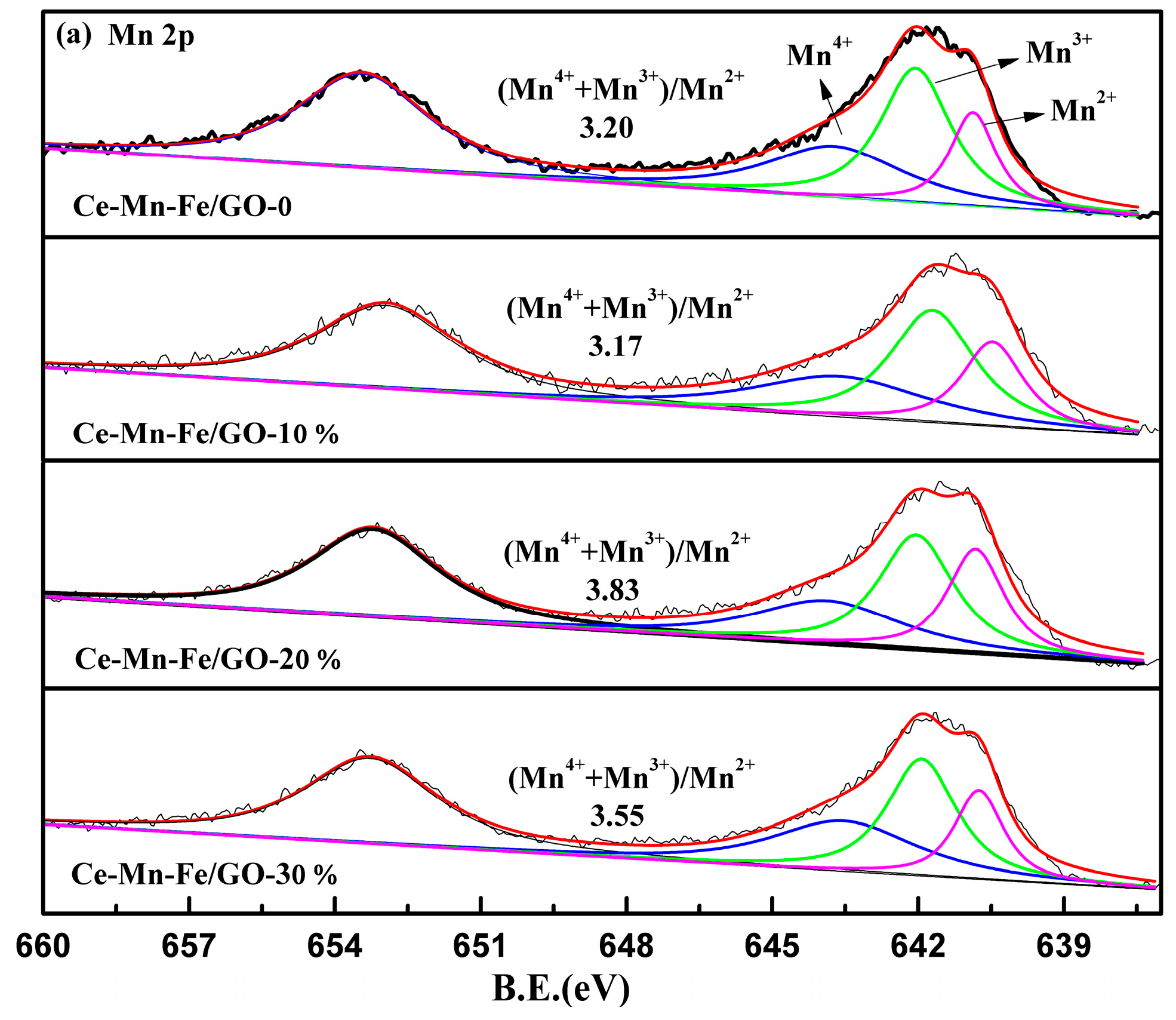
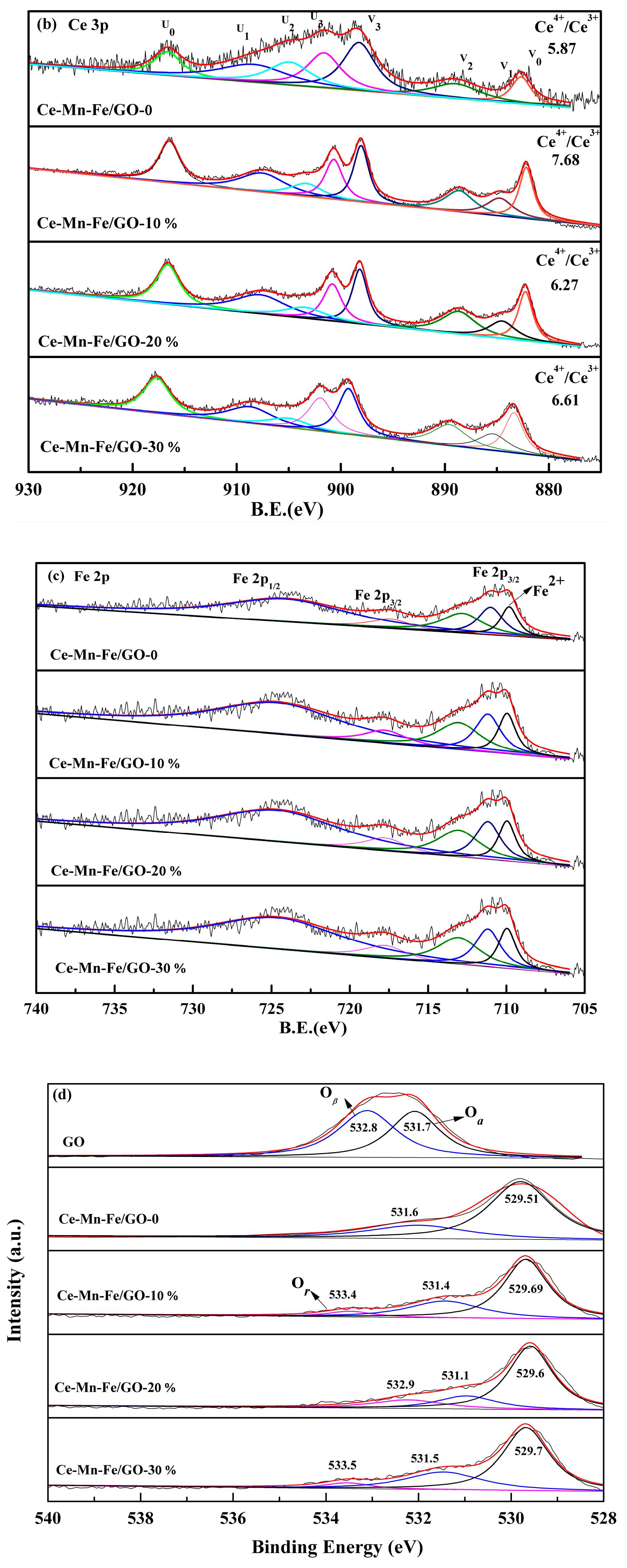
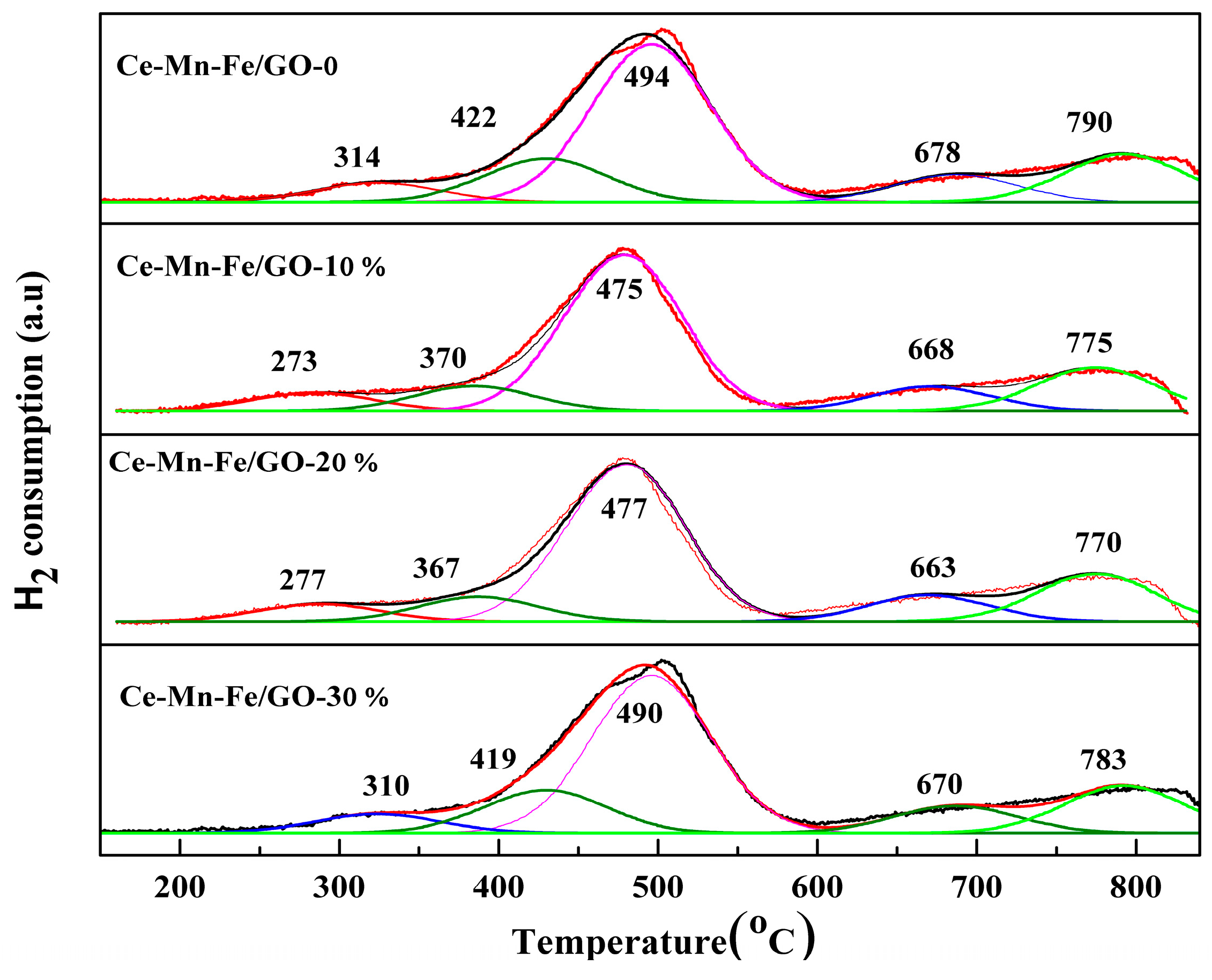
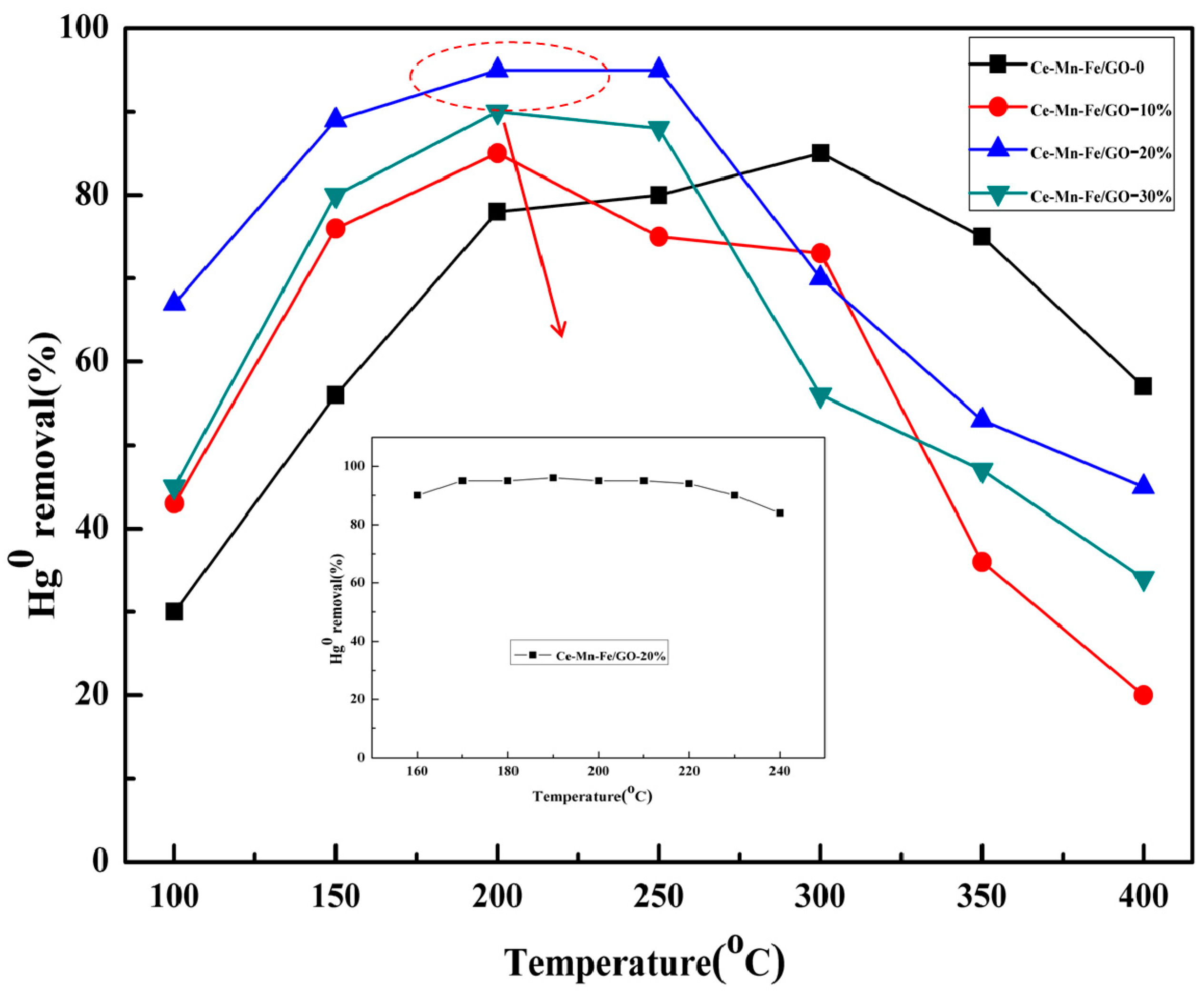
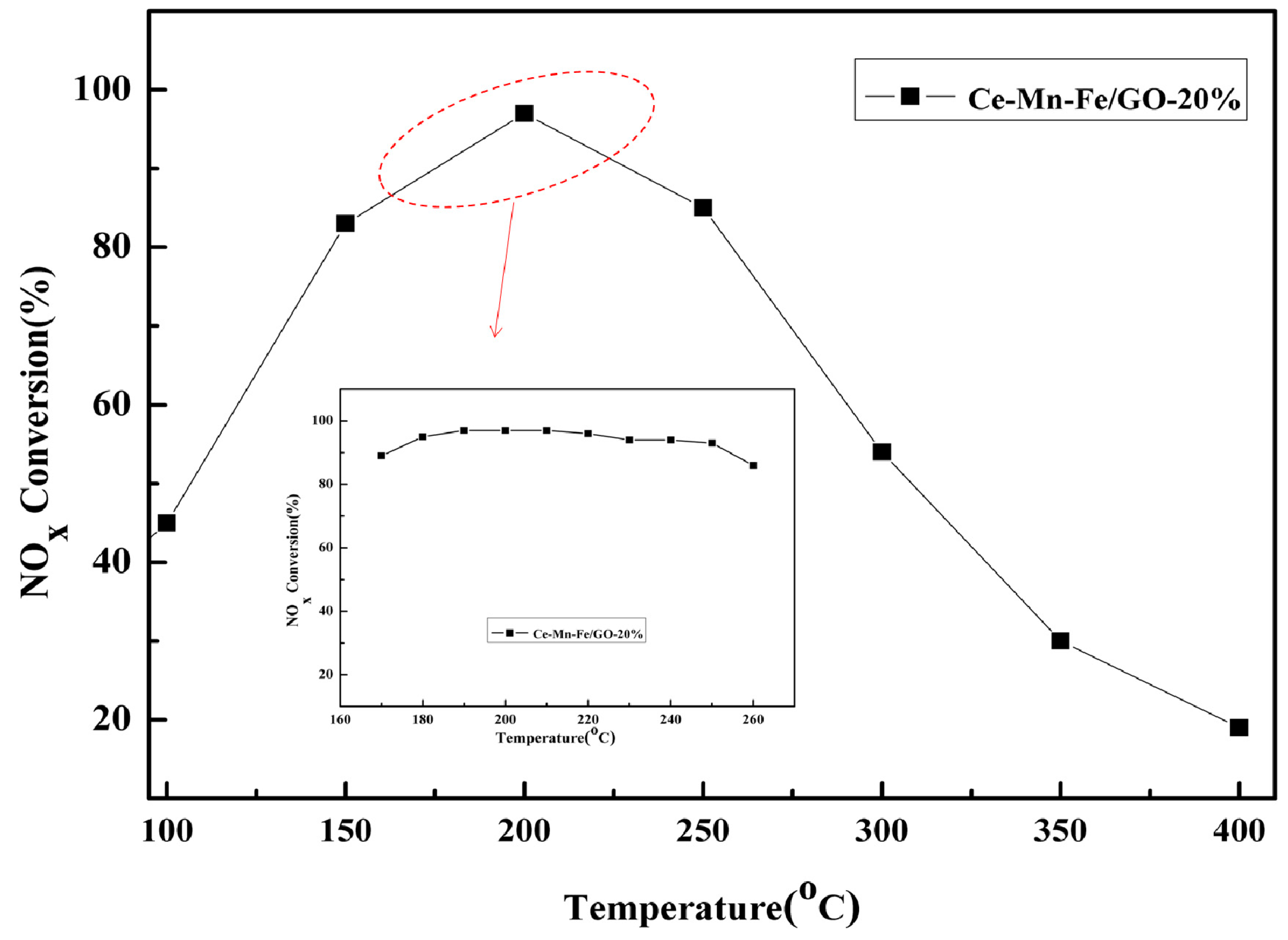
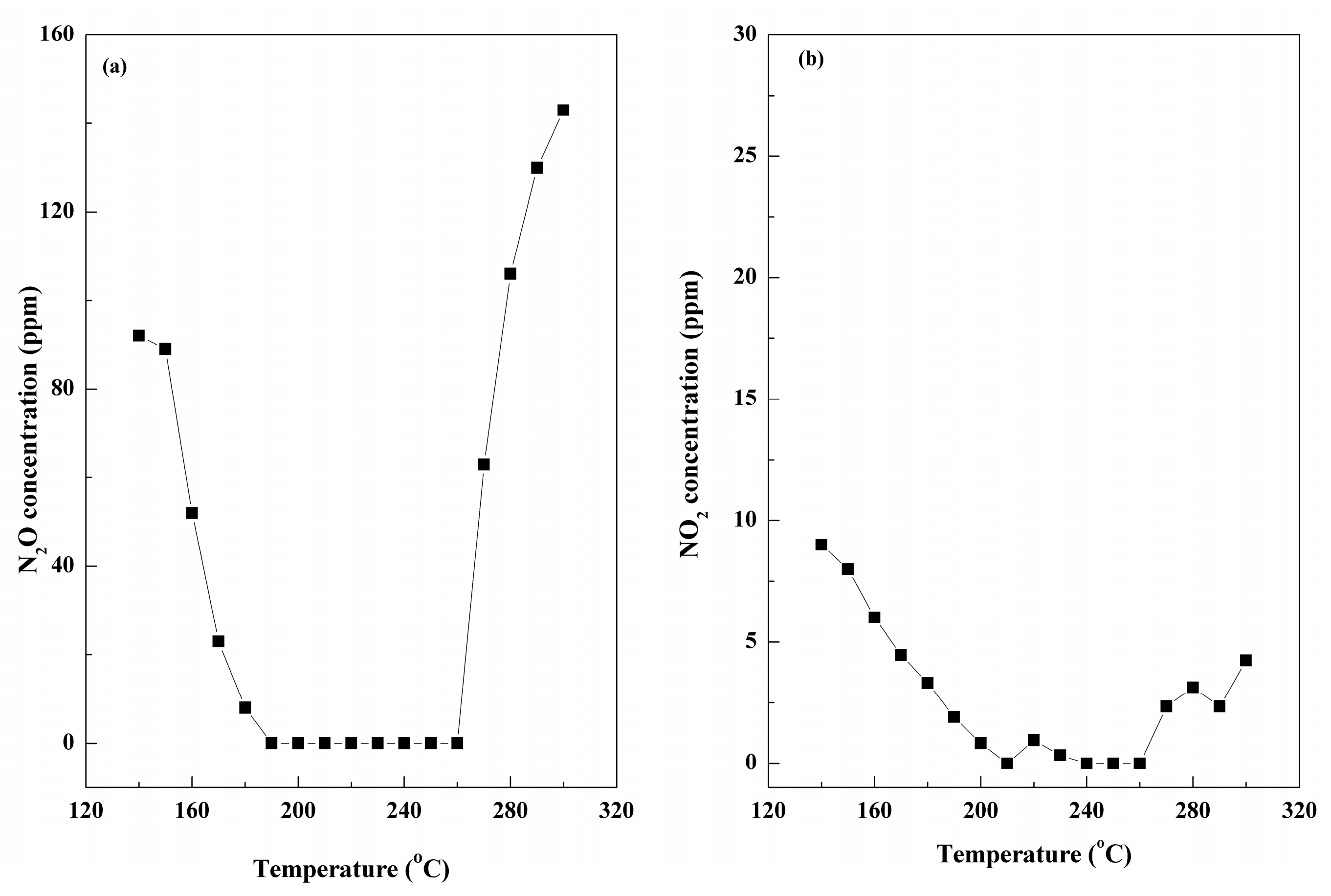

| Samples | BET Surface (m2/g) | Pore Volume (cm3/g) | Average Pore Diameter (nm) |
|---|---|---|---|
| Ce-Mn-Fe/GO-0 | 2.37 | 0.0089 | 2.321 |
| Ce-Mn-Fe/GO-10% | 54.39 | 0.0363 | 3.418 |
| Ce-Mn-Fe/GO-20% | 109.23 | 0.0463 | 3.426 |
| Ce-Mn-Fe/GO-30% | 79.95 | 0.0395 | 3.415 |
| Sample | Surface Atomic Concentrations (%) | ||
|---|---|---|---|
| Oα | Oβ | Or | |
| GO | 0 | 45.36 | 54.64 |
| Ce-Mn-Fe/GO0 | 60.24 | 30.76 | 0 |
| Ce-Mn-Fe/GO0.1 | 61.65 | 31.73 | 6.62 |
| Ce-Mn-Fe/GO0.2 | 59.89 | 32.26 | 7.85 |
| Ce-Mn-Fe/GO0.3 | 66.89 | 18.40 | 14.70 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
An, D.; Zhang, X.; Cheng, X.; Dong, Y. Performance of Mn-Fe-Ce/GO-x for Catalytic Oxidation of Hg0 and Selective Catalytic Reduction of NOx in the Same Temperature Range. Catalysts 2018, 8, 399. https://doi.org/10.3390/catal8090399
An D, Zhang X, Cheng X, Dong Y. Performance of Mn-Fe-Ce/GO-x for Catalytic Oxidation of Hg0 and Selective Catalytic Reduction of NOx in the Same Temperature Range. Catalysts. 2018; 8(9):399. https://doi.org/10.3390/catal8090399
Chicago/Turabian StyleAn, Donghai, Xiaoyang Zhang, Xingxing Cheng, and Yong Dong. 2018. "Performance of Mn-Fe-Ce/GO-x for Catalytic Oxidation of Hg0 and Selective Catalytic Reduction of NOx in the Same Temperature Range" Catalysts 8, no. 9: 399. https://doi.org/10.3390/catal8090399
APA StyleAn, D., Zhang, X., Cheng, X., & Dong, Y. (2018). Performance of Mn-Fe-Ce/GO-x for Catalytic Oxidation of Hg0 and Selective Catalytic Reduction of NOx in the Same Temperature Range. Catalysts, 8(9), 399. https://doi.org/10.3390/catal8090399




