Selective Hydrogenation of Benzene to Cyclohexene over Ru-Zn Catalysts: Mechanism Investigation on NaOH as a Reaction Additive
Abstract
:1. Introduction
2. Results
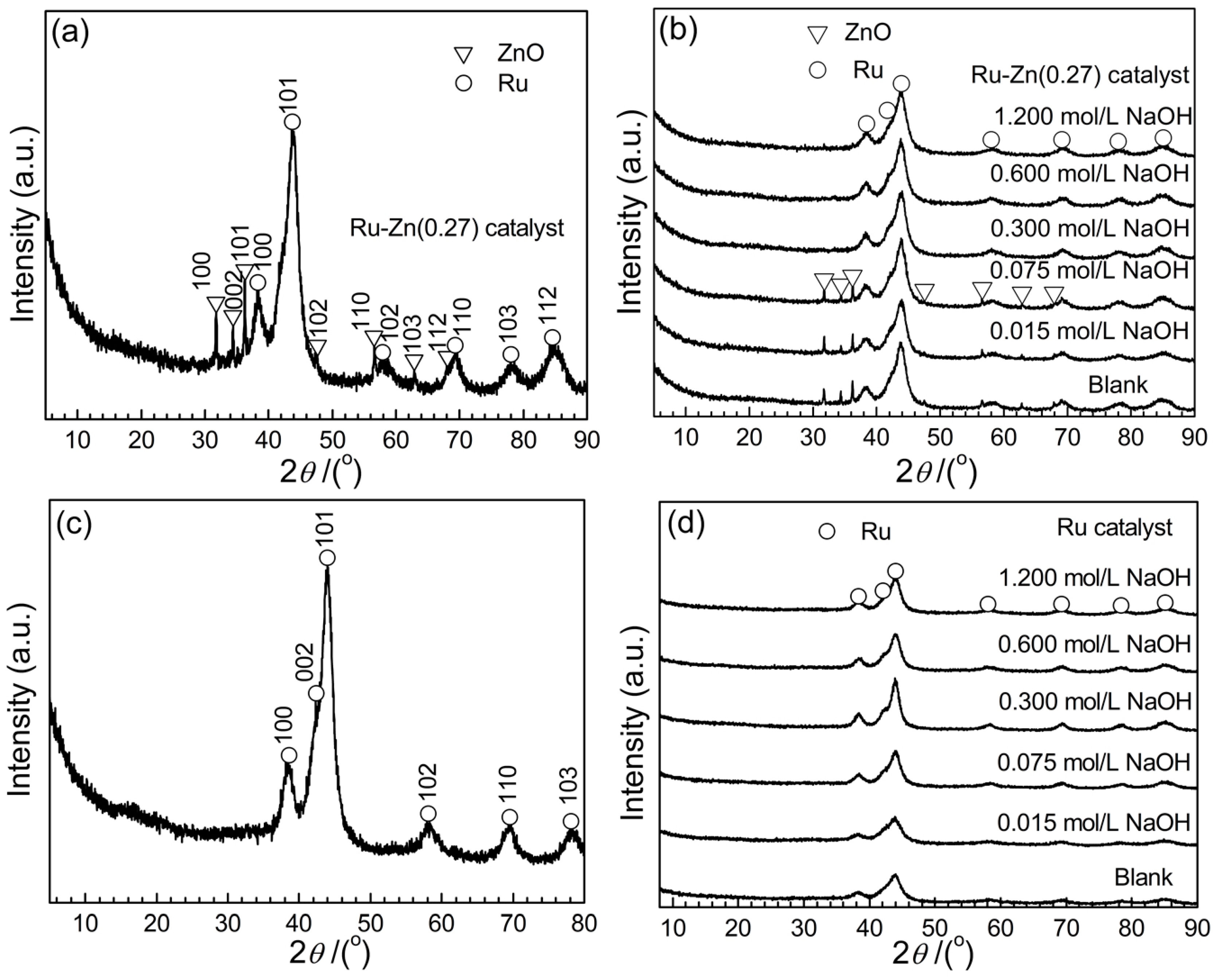
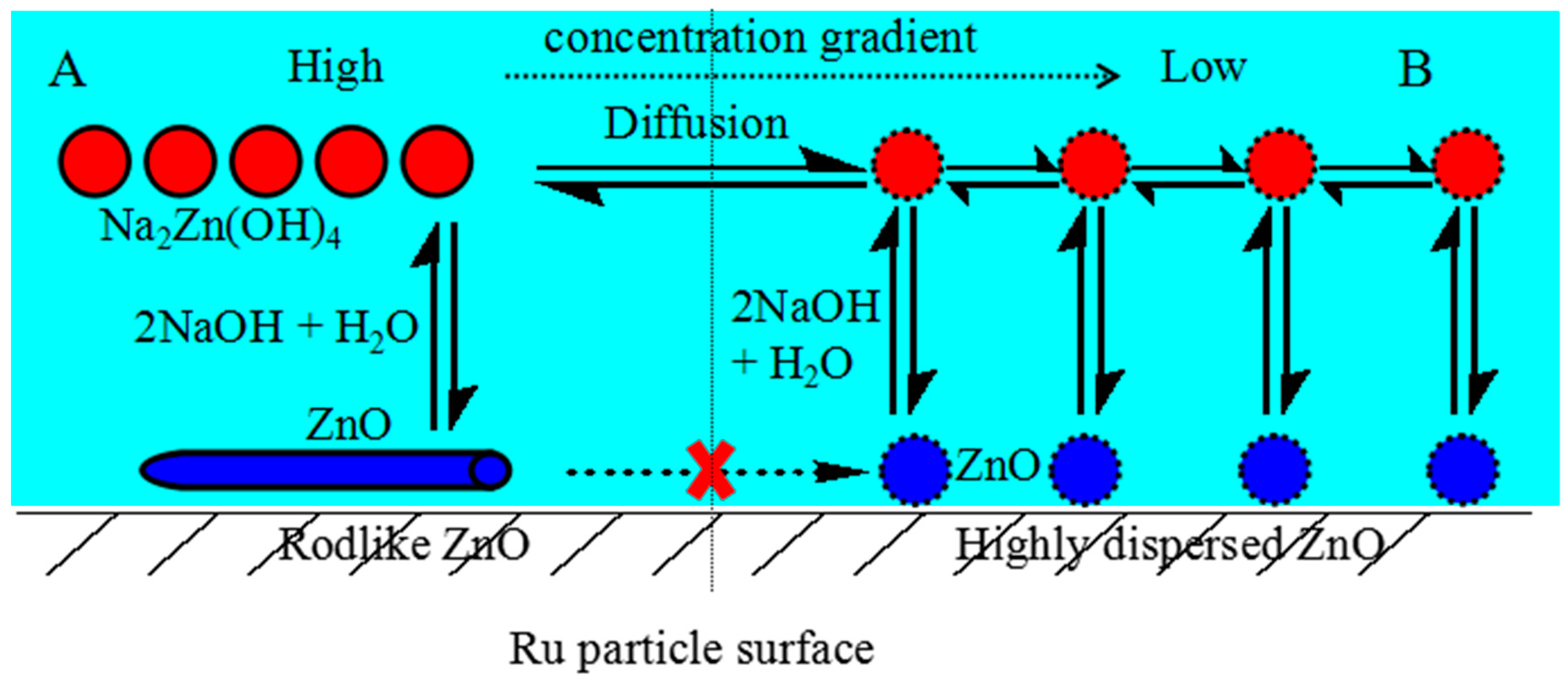
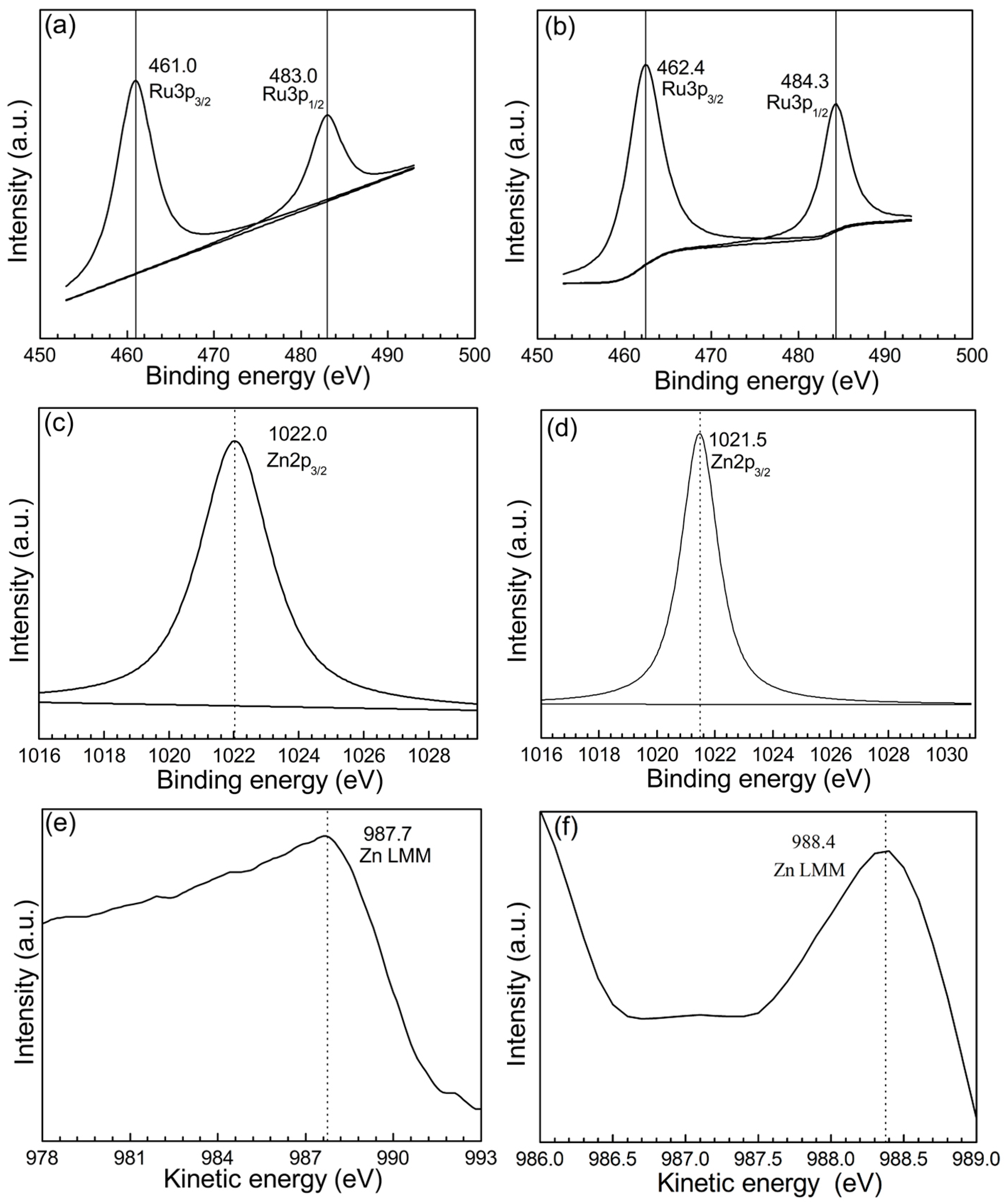
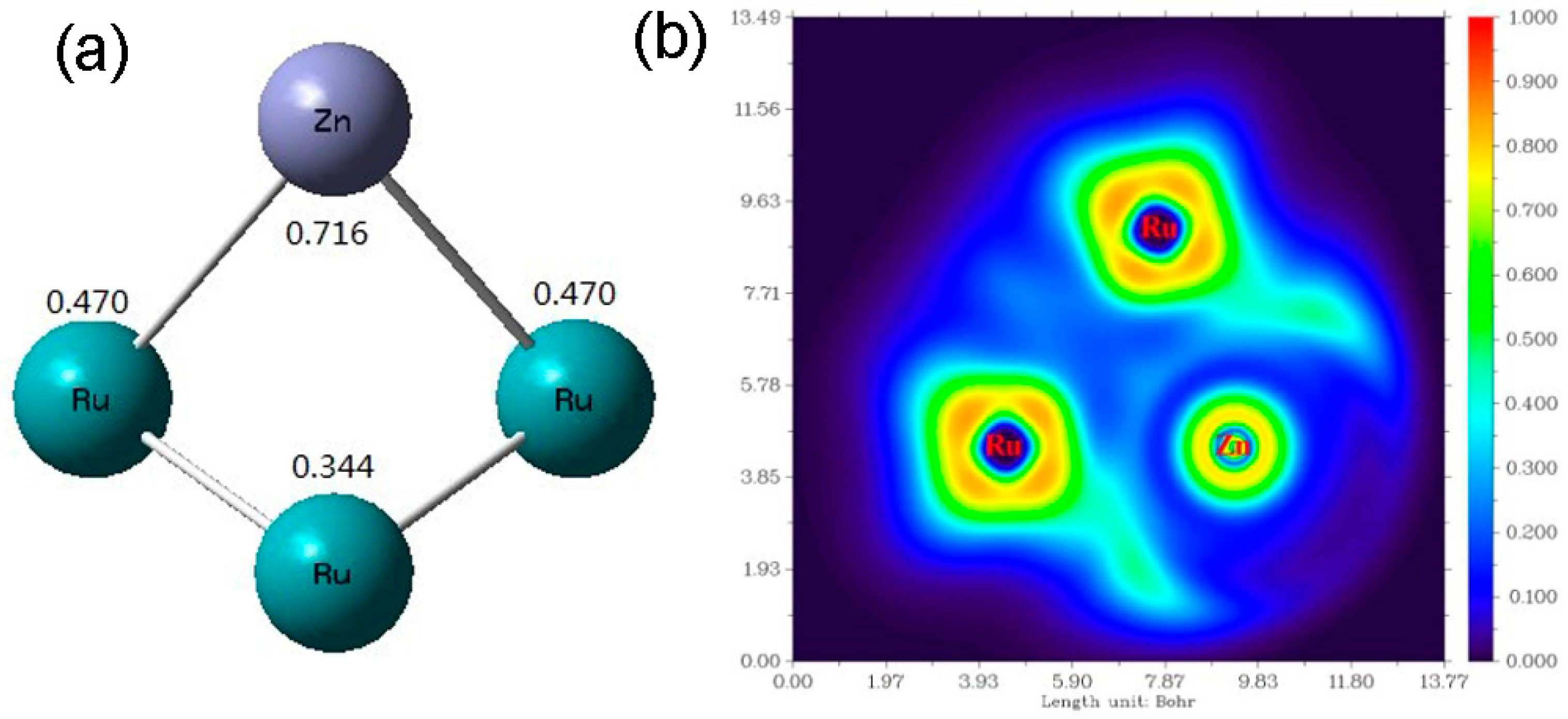
2.1. Catalyst Characterization
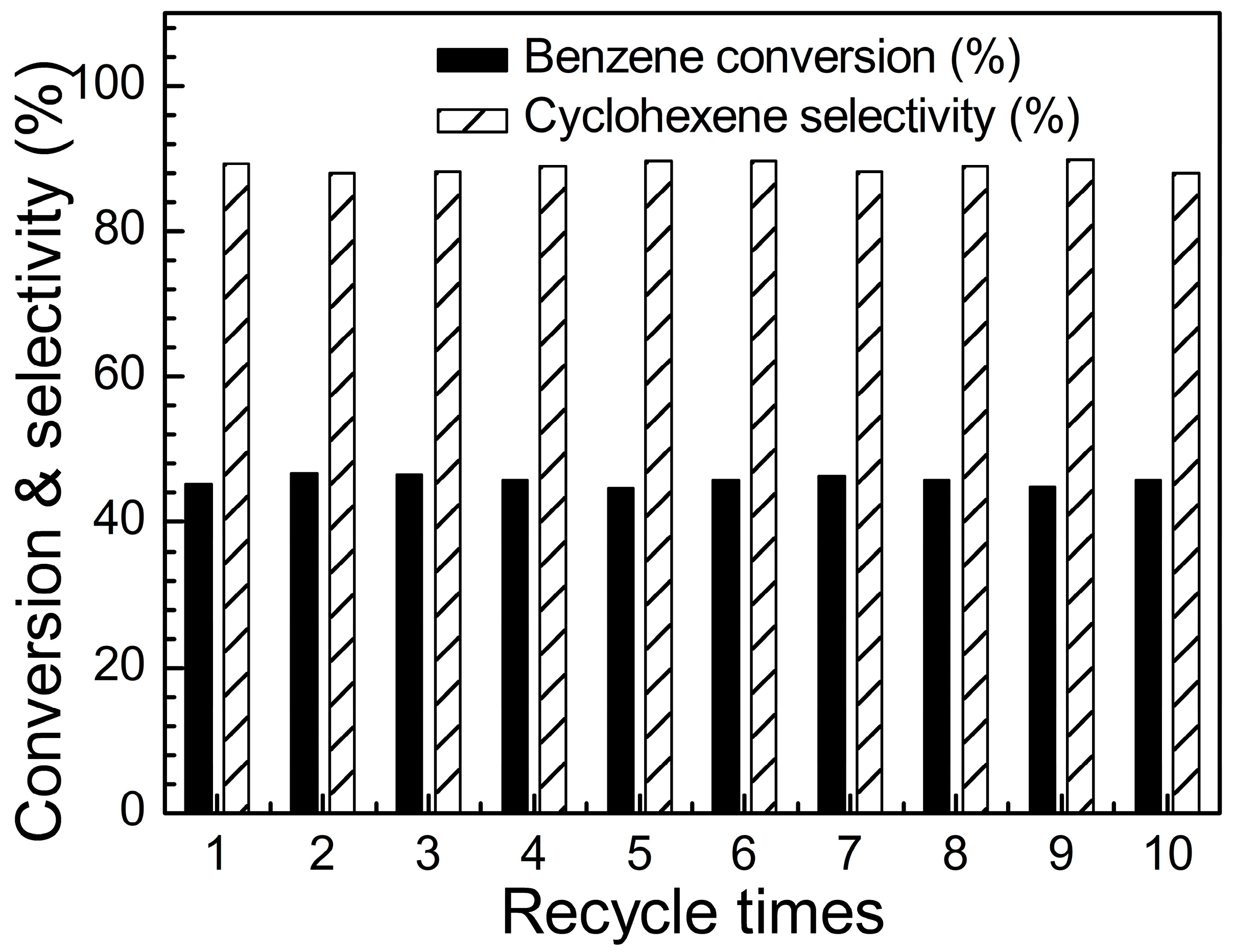
2.2. Catalytic Performance
3. Materials and Methods
3.1. Chemicals
3.2. Preparation of Catalysts
3.3. Catalytic Experimental Procedure
3.4. Catalysts Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Z.B.; Zhang, Q.; Lu, X.F.; Chen, S.J.; Liu, C.J. Ru-Zn catalysts for selective hydrogenation of benzene using coprecipitation in low alkalinity. Chin. J. Catal. 2015, 36, 400–407. [Google Scholar] [CrossRef]
- Fan, G.F.; Li, R.X.; Li, X.J.; Chen, H. Effect of organic on partial hydrogenation of benzene. Catal. Commun. 2008, 9, 1394–1397. [Google Scholar] [CrossRef]
- Yuan, P.Q.; Wang, B.Q.; Ma, Y.M.; He, H.M.; Cheng, Z.M.; Yuan, W.K. Hydrogenation of cyclohexene over Ru-Zn/Ru(0001) surface alloy: A first principles density functional study. J. Mol. Catal. A Chem. 2009, 301, 140–145. [Google Scholar] [CrossRef]
- He, H.M.; Yuan, P.Q.; Ma, Y.M.; Cheng, Z.M.; Yuan, W.K. Theoretical and experimental study on the partial hydrogenation of benzene over Ru-Zn/ZrO2 catalyst. Chin. J. Catal. 2009, 30, 312–318. [Google Scholar]
- Peng, Z.K.; Liu, X.; Lin, H.N.; Wang, Z.; Li, Z.J.; Li, B.J.; Liu, Z.Y.; Liu, S.C. Surface engineering on a nanocatalyst: Basic zinc salt nanoclusters improve catalytic performances of Ru nanoparticles. J. Mater. Chem. A 2016, 4, 17694–17703. [Google Scholar] [CrossRef]
- Fan, G.Y.; Jiang, W.D.; Wang, J.B.; Li, R.X.; Chen, H.; Li, X.J. Selective hydrogenation of benzene to cyclohexene over RuCoB/γ-Al2O3 without additive. Catal. Commun. 2008, 10, 98–102. [Google Scholar] [CrossRef]
- Sun, H.J.; Chen, L.X.; Huang, Z.X.; Sun, L.L.; Li, Y.Y.; Liu, S.C.; Liu, Z.Y. Effect of reduction medium and reduction temperature on the performance of Ru-Zn catalysts for selective hydrogenation of benzene to cyclohexene. Chem. Ind. Eng. Prog. 2017, 36, 2962–2970. [Google Scholar]
- Sun, H.J.; Li, Y.Y.; Li, S.H.; Zhang, Y.X.; Liu, S.C.; Liu, Z.Y.; Ren, B.Z. ZnSO4 and La2O3 as Co-Modifier of the Monoclinic Ru Catalyst for Selective Hydrogenation of Benzene to Cyclohexene. Acta Phys. Chim. Sin. 2014, 30, 1332–1340. [Google Scholar]
- Foppa, L.; Dupont, J. Benzene partial hydrogenation: Advances and perspectives. Chem. Soc. Rev. 2015, 44, 1886–1897. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.J.; Chen, L.X.; Huang, Z.X.; Liu, S.C.; Liu, Z.Y. Particle Size Effect of Ru-Zn Catalysts on Selective Hydrogenation of Benzene to Cyclohexene. Chem. J. Chin. Univ. 2015, 36, 1969–1976. [Google Scholar]
- Sun, H.J.; Chen, X.L.; Huang, Z.X.; Liu, Z.Y.; Liu, S.C. Effect of NaOH concentration on performance of Ru-Zn catalyst for selective hydrogenation of benzene to cyclohexene. CIESC J. 2016, 67, 1324–1332. [Google Scholar]
- Sun, H.J.; Chen, J.J.; Huang, Z.X.; Liu, Z.Y.; Liu, S.C. Selective hydrogenation of benzene to cyclohexene over the nano-sized Ru-Zn catalyst modified by Rrabic gum. Chin. J. Inorg. Chem. 2016, 32, 202–210. [Google Scholar]
- Nagahara, H.; Ono, M.; Konishi, M.; Fukuoka, Y. Partial hydrogenation of benzene to cyclohexene. Appl. Surf. Sci. 1997, 121–122, 448–451. [Google Scholar] [CrossRef]
- Wu, J.M.; Yang, Y.F.; Chen, J.L. Study on the causes of catalyst inactivation of benzene semi-hydrogenation. Chem. Ind. Eng. Prog. 2003, 22, 295–297. [Google Scholar]
- Liu, J.L.; Zhu, L.J.; Pei, Y.; Zhuang, J.H.; Li, H.; Li, H.X.; Qiao, M.H.; Fan, K.N. Ce-promoted Ru/SBA-15 catalysts prepared by a “two solvent” impregnation method for selective hydrogenation of benzene to cyclohexene. Appl. Catal. A Gen. 2009, 353, 282–287. [Google Scholar] [CrossRef]
- Zhou, G.B.; Pei, Y.; Jiang, Z.; Fan, K.N.; Qiao, M.H.; Sun, B.; Zong, B.N. Doping effects of B in ZrO2 on structural and catalytic properties of Ru/B-ZrO2 catalysts for benzene partial hydrogenation. J. Catal. 2014, 311, 393–403. [Google Scholar] [CrossRef]
- Liao, H.G.; Ouyang, D.H.; Zhang, J.; Xiao, Y.J.; Liu, P.L.; Hao, F.; You, K.Y.; Luo, H.A. Benzene hydrogenation over oxide-modified MCM-41 supported ruthenium-lanthanum catalyst: The influence of zirconia crystal form and surface hydrophilicity. Chem. Eng. J. 2014, 243, 207–216. [Google Scholar] [CrossRef]
- Ning, J.B.; Xu, J.; Liu, J.; Lu, F. Selective hydrogenation of benzene to cyclohexene over colloidal ruthenium catalyst stabilized by silica. Catal. Lett. 2006, 109, 175–180. [Google Scholar] [CrossRef]
- Yan, X.H.; Zhang, Q.; Zhu, M.Q.; Wang, Z.B. Selective hydrogenation of benzene to cyclohexene over Ru-Zn/ZrO2 catalysts prepared by a two step impreganation method. J. Mol. Catal. A Chem. 2016, 413, 85–93. [Google Scholar] [CrossRef]
- Liu, H.Z.; Liang, S.G.; Wang, W.T.; Jiang, T.; Han, B.X. The partial hydrogenation of benzene to cyclohexene over Ru-Cu catalyst supported on ZnO. J. Mol. Catal. A Chem. 2011, 341, 35–41. [Google Scholar] [CrossRef]
- Zhang, P.; Wu, T.B.; Jiang, T.; Wang, W.T.; Liu, H.Z.; Fan, H.L.; Zhang, Z.F.; Han, B.X. Ru-Zn supported on hydroxyapatite as an effective catalyst for partial hydrogenation of benzene. Green Chem. 2013, 15, 152–159. [Google Scholar] [CrossRef]
- Wu, T.B.; Zhang, P.; Jiang, T.; Yang, D.X.; Han, B.X. Enhancing the selective hydrogenation of benzene to cyclohexene over Ru/TiO2 catalyst in the presence of a very small amount of ZnO. Sci. China Chem. 2015, 58, 93–100. [Google Scholar] [CrossRef]
- Sun, H.J.; Wang, H.X.; Jiang, H.B.; Li, S.H.; Liu, S.C.; Liu, Z.Y.; Yuan, X.M.; Yang, K.J. Eeffect of (Zn(OH)2)3(ZnSO4)(H2O)5 on the performance of Ru-Zn catalyst for benzene selective hydrogenation to cyclohexene. Appl. Catal. A 2013, 450, 160–168. [Google Scholar] [CrossRef]
- Sun, H.J.; Zhang, X.D.; Chen, Z.H.; Zhou, X.L.; Guo, W.; Liu, Z.Y.; Liu, S.C. Monolayer dispersed Ru-Zn Catalyst and its performance in the Selective Hydrogenation of Benzene to Cyclohexene. Chin. J. Catal. 2011, 32, 224–230. [Google Scholar] [CrossRef]
- Sun, H.J.; Jiang, H.B.; Li, S.H.; Wang, H.X.; Pan, Y.J.; Dong, Y.Y.; Liu, S.C.; Liu, Z.Y. Selective hydrogenation of benzene to cyclohexene over nanocomposite Ru-Mn/ZrO2 catalyst. Chin. J. Catal. 2013, 34, 684–694. [Google Scholar] [CrossRef]
- Sun, H.J.; Dong, Y.Y.; Li, S.H.; Jiang, H.B.; Zhang, Y.X.; Liu, Z.Y.; Liu, S.C. The role of La in improving the selectivity to cyclohexene of Ru catalyst for hydrogenation of benzene. J. Mol. Catal. A Chem. 2013, 368–369, 119–124. [Google Scholar] [CrossRef]
- Sun, H.J.; Pan, Y.J.; Li, S.H.; Zhang, Y.X.; Dong, Y.Y.; Liu, S.C.; Liu, Z.Y. Selective hydrogenation of benzene to cyclohexene over Ce-promoted Ru catalysts. J. Energy Chem. 2013, 22, 710–716. [Google Scholar] [CrossRef]
- Sun, H.J.; Jiang, H.B.; Dong, Y.Y.; Wang, H.X.; Pan, Y.J.; Liu, S.C.; Tang, M.S.; Liu, Z.Y. Effect of alcohols as additives on the performance of a nano-sized Ru-Zn(2.8%) catalyst for selective hydrogenation of benzene to cyclohexene. Chem. Eng. J. 2013, 218, 415–424. [Google Scholar] [CrossRef]
- Sun, H.J.; Pan, Y.J.; Wang, H.X.; Dong, Y.Y.; Liu, Z.Y.; Liu, S.C. Selective hydrogenation of benzene to cyclohexene over a Ru-Zn Catalyst with diethanolamine as an additive. Chin. J. Catal. 2012, 33, 610–620. [Google Scholar] [CrossRef]
- Zhou, G.B.; Wang, H.; Pei, Y.; Qiao, M.H.; Sun, B.; Zong, B.N. Pore size effect of Ru-Zn/ZrO2 catalyst on partial hydrogenation of benzene to cyclohexene. Acta Chim. Sin. 2017, 75, 321–328. [Google Scholar] [CrossRef]
- Kötz, R.; Lewerenz, H.J.; Stucki, S. XPS studies of oxygen evolution on Ru and RuO2 anodes. J. Electrochem. Soc. 1983, 130, 825–829. [Google Scholar] [CrossRef]
- Wehner, P.S.; Mercer, P.N.; Apai, G. Interaction of H2 and CO with Rh4(CO)12 supported on ZnO. J. Catal. 1983, 84, 244–247. [Google Scholar] [CrossRef]
- Gaarenstroom, S.W.; Winograd, N. Initial and final state effects in the ESCA spectra of cadmium and silver oxides. J. Chem. Phys. 1977, 67, 3500–3506. [Google Scholar] [CrossRef]
- Folkesson, B. ESCA Studies on the Charge Distribution in Some Dinitrogen Complexes of Rhenium, Iridium, Ruthenium, and Osmium. Acta Chem. Scand. 1973, 27, 287–302. [Google Scholar] [CrossRef]
- Nefedov, V.I.; Firsov, M.N.; Shaplygin, I.S. Electronic structures of MRhO2, MRh2O4, RhMO4 and Rh2MO6 on the basis of X-ray spectroscopy and ESCA data. J. Electron Spectrosc. Relat. Phenom. 1982, 26, 65–78. [Google Scholar] [CrossRef]
- Schӧn, G. Auger and direct electron spectra in X-ray photoelectron studies of zinc, zinc oxide, gallium and gallium oxide. J. Electron Spectrosc. Relat. Phenom. 1973, 2, 75–86. [Google Scholar] [CrossRef]
- Sun, H.J.; Guo, W.; Zhou, X.L.; Chen, Z.H.; Liu, Z.Y.; Liu, S.C. Process in Ru-based Amorphous Alloy Catalysts for Benzene Selective Hydrogenation to Cyclohexene. Chin. J. Catal. 2011, 32, 1–16. [Google Scholar] [CrossRef]
- Mazzieri, V.A.; L’Argentire, P.C.; Coloma-Pascual, F.; Fgoli, N.S. Effect of Chlorine on the Properties of Ru/Al2O3. Ind. Eng. Chem. Res. 2003, 42, 2269–2272. [Google Scholar] [CrossRef]
- Struijk, J.; Moene, R.; Kamp, T.V.D.; Scholten, J.J.F. Partial liquid phase hydrogenation of benzene to cyclohexene over ruthenium catalysts in the presence of an aqueous salt solution II. Influence of various salts on the performance of the catalyst. Appl. Catal. A Gen. 1992, 89, 77–102. [Google Scholar] [CrossRef]
- Liu, J.L.; Zhu, Y.; Liu, J.; Pei, Y.; Li, Z.H.; Li, H.; Li, H.X.; Qiao, M.H.; Fan, K.N. Discrimination of the roles of CdSO4 and ZnSO4 in liquid phase hydrogenation of benzene to cyclohexene. J. Catal. 2009, 268, 100–105. [Google Scholar] [CrossRef]
- Sun, H.J.; Qin, H.A.; Huang, Z.X.; Su, M.F.; Li, Y.Y.; Liu, S.C.; Liu, Z.Y. Effect of reaction modifier ZnSO4 and pretreatment on performance of Ru-Zn catalyst for selective hydrogenation of benzene to cyclohexene. Chin. J. Inorg. Chem. 2017, 33, 73–80. [Google Scholar]



| Sample | n(Zn)/n(Ru) (mol/mol) 1 | n(Na)/n(Ru) (mol/mol) 1 | Ru Particle Size (nm) 2 |
|---|---|---|---|
| Ru-Zn (0.27) | 0.27 | n.d. 3 | 4.2 |
| Ru-Zn (0.27) + Blank | 0.27 | n.d. | 4.2 |
| Ru-Zn (0.27) + 0.075 mol L−1 NaOH | 0.26 | n.d. | 4.3 |
| Ru-Zn (0.27) + 0.150 mol L−1 NaOH | 0.25 | n.d. | 4.2 |
| Ru-Zn (0.27) + 0.300 mol L−1 NaOH | 0.23 | n.d. | 4.1 |
| Ru-Zn (0.27) + 0.600 mol L−1 NaOH | 0.16 | n.d. | 4.3 |
| Ru-Zn (0.27) + 1.200 mol L−1 NaOH | 0.14 | n.d. | 4.1 |
| Ru | n.d. | n.d. | 4.2 |
| Ru + Blank | n.d. | n.d. | 4.2 |
| Ru + 0.075 mol L−1 NaOH | n.d. | n.d. | 4.1 |
| Ru + 0.150 mol L−1 NaOH | n.d. | n.d. | 4.2 |
| Ru + 0.300 mol L−1 NaOH | n.d. | n.d. | 4.1 |
| Ru + 0.600 mol L−1 NaOH | n.d. | n.d. | 4.3 |
| Ru + 1.200 mol L−1 NaOH | n.d. | n.d. | 4.2 |
| Sample | SBET (m2 g−1) 1 | Vp (cm3 g−1) 1 | Dp (nm) 1 |
|---|---|---|---|
| Ru-Zn (0.27) | 52 | 0.17 | 5.5 |
| Ru-Zn (0.27) + Blank | 52 | 0.16 | 5.5 |
| Ru-Zn (0.27) + 0.075 mol L−1 NaOH | 49 | 0.14 | 5.8 |
| Ru-Zn (0.27) + 0.150 mol L−1 NaOH | 48 | 0.12 | 6.1 |
| Ru-Zn (0.27) + 0.300 mol L−1 NaOH | 48 | 0.12 | 6.2 |
| Ru-Zn (0.27) + 0.600 mol L−1 NaOH | 46 | 0.10 | 6.9 |
| Ru-Zn (0.27) + 1.200 mol L−1 NaOH | 45 | 0.10 | 7.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, H.; Chen, Z.; Li, C.; Chen, L.; Peng, Z.; Liu, Z.; Liu, S. Selective Hydrogenation of Benzene to Cyclohexene over Ru-Zn Catalysts: Mechanism Investigation on NaOH as a Reaction Additive. Catalysts 2018, 8, 104. https://doi.org/10.3390/catal8030104
Sun H, Chen Z, Li C, Chen L, Peng Z, Liu Z, Liu S. Selective Hydrogenation of Benzene to Cyclohexene over Ru-Zn Catalysts: Mechanism Investigation on NaOH as a Reaction Additive. Catalysts. 2018; 8(3):104. https://doi.org/10.3390/catal8030104
Chicago/Turabian StyleSun, Haijie, Zhihao Chen, Chenggang Li, Lingxia Chen, Zhikun Peng, Zhongyi Liu, and Shouchang Liu. 2018. "Selective Hydrogenation of Benzene to Cyclohexene over Ru-Zn Catalysts: Mechanism Investigation on NaOH as a Reaction Additive" Catalysts 8, no. 3: 104. https://doi.org/10.3390/catal8030104
APA StyleSun, H., Chen, Z., Li, C., Chen, L., Peng, Z., Liu, Z., & Liu, S. (2018). Selective Hydrogenation of Benzene to Cyclohexene over Ru-Zn Catalysts: Mechanism Investigation on NaOH as a Reaction Additive. Catalysts, 8(3), 104. https://doi.org/10.3390/catal8030104





