Fivefold Enhanced Photoelectrochemical Properties of ZnO Nanowire Arrays Modified with C3N4 Quantum Dots
Abstract
:1. Introduction
2. Results and Discussion
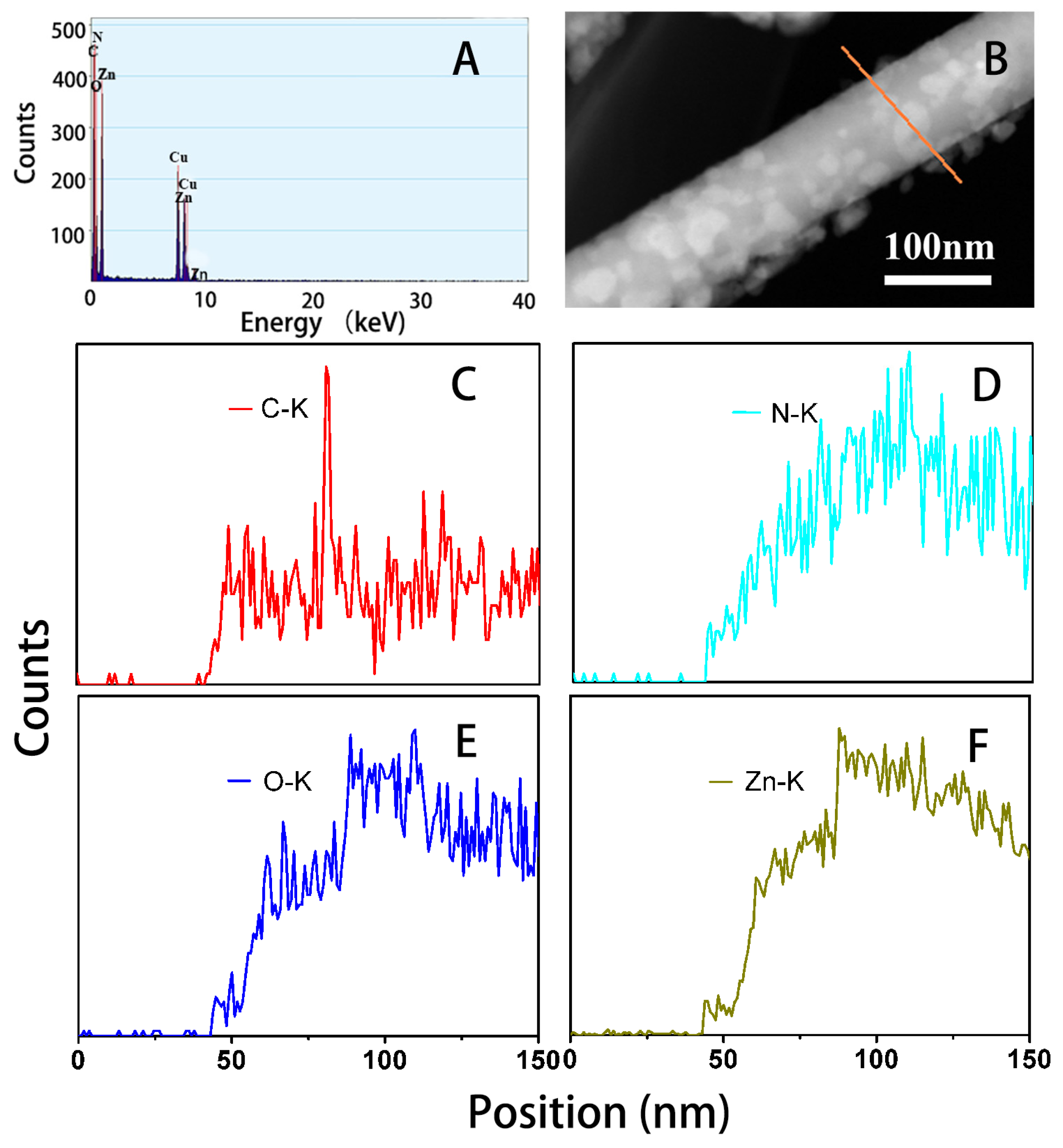
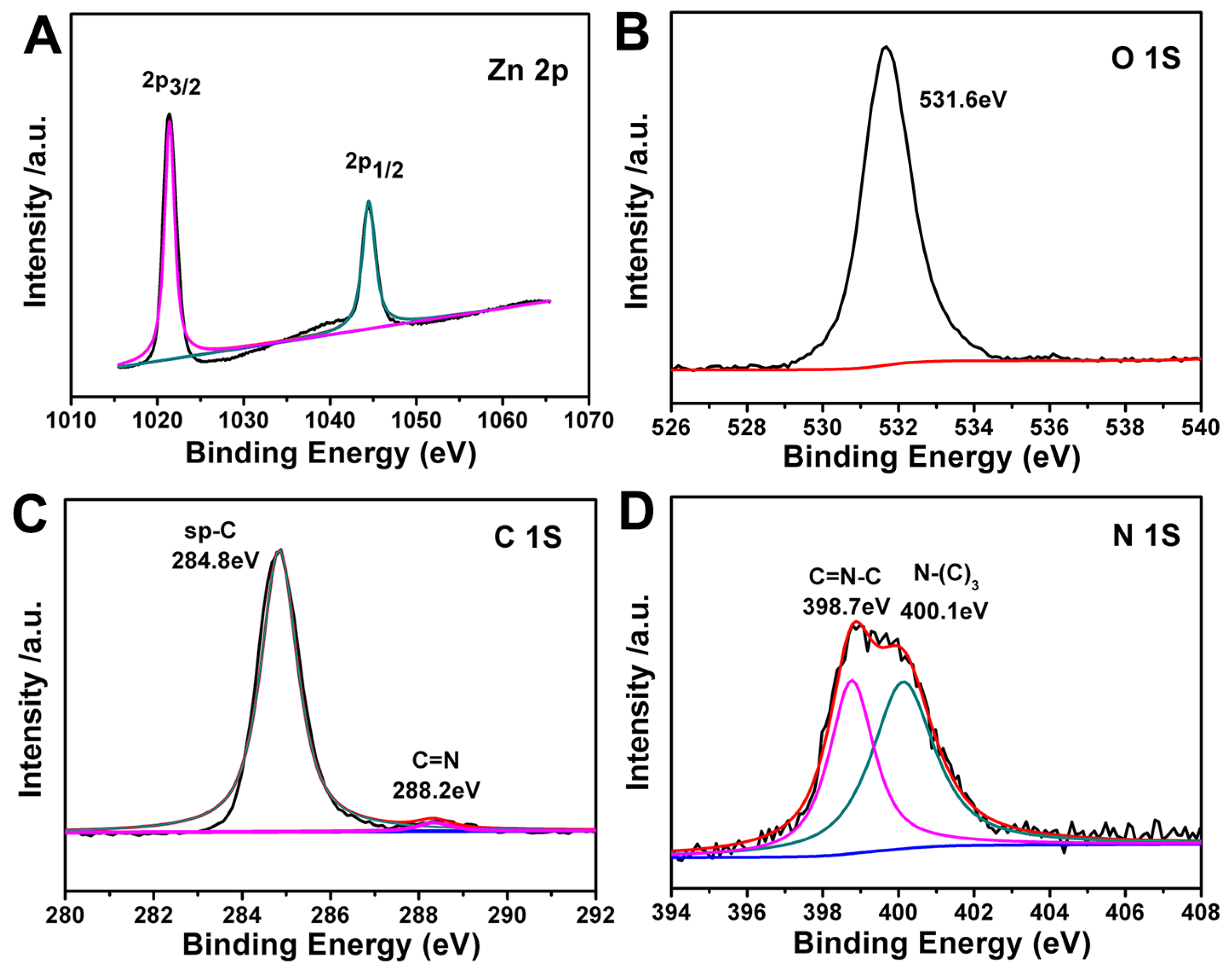
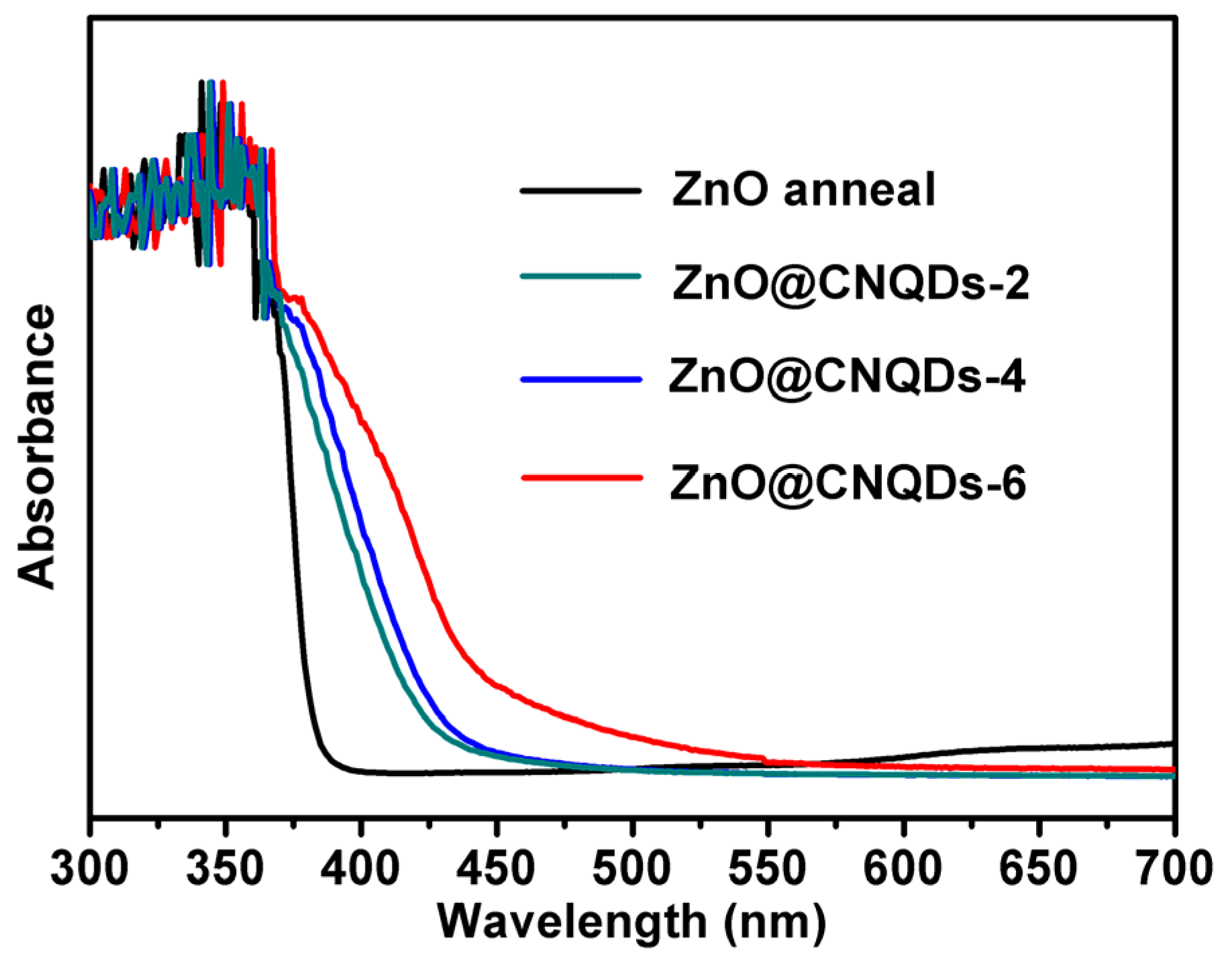
2.1. Morphology and Structure
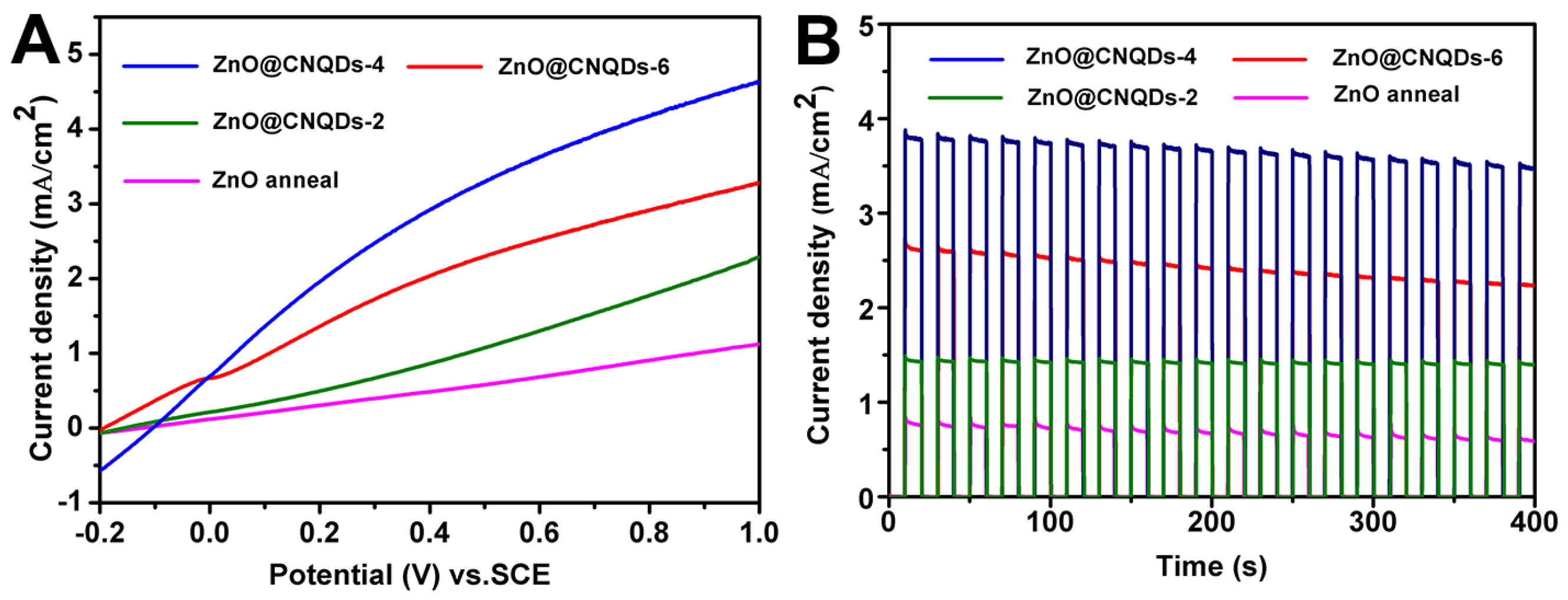
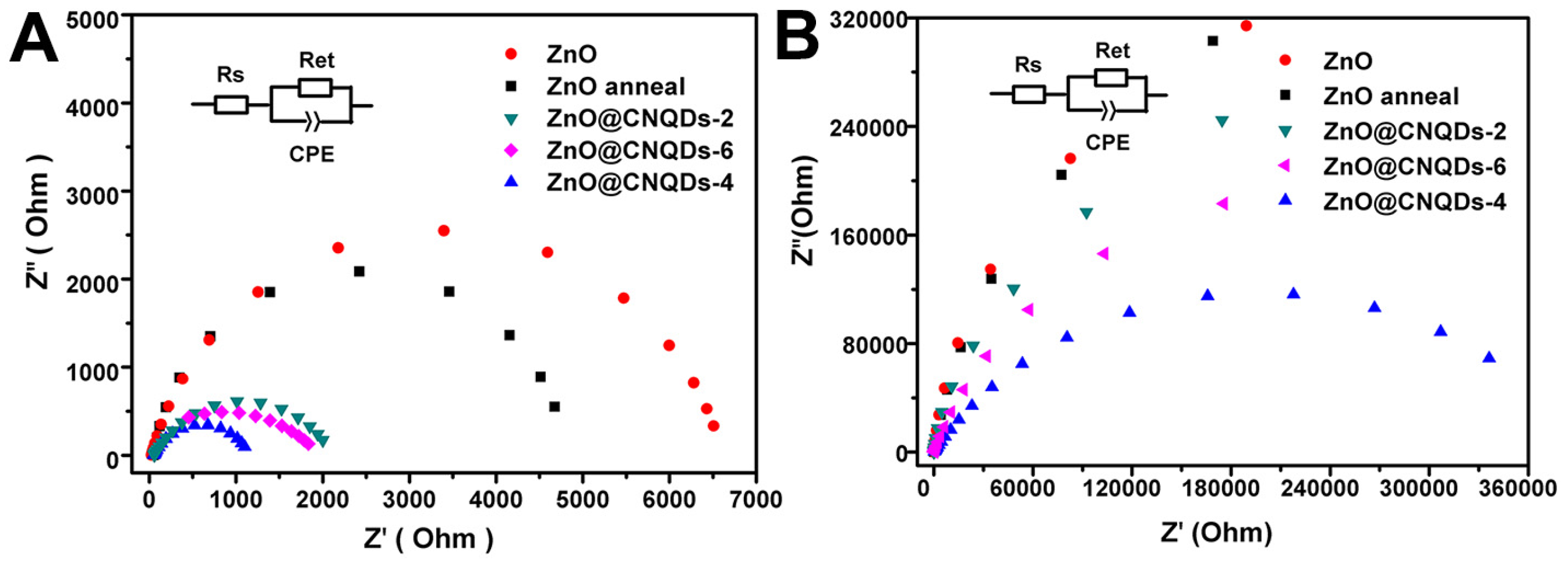
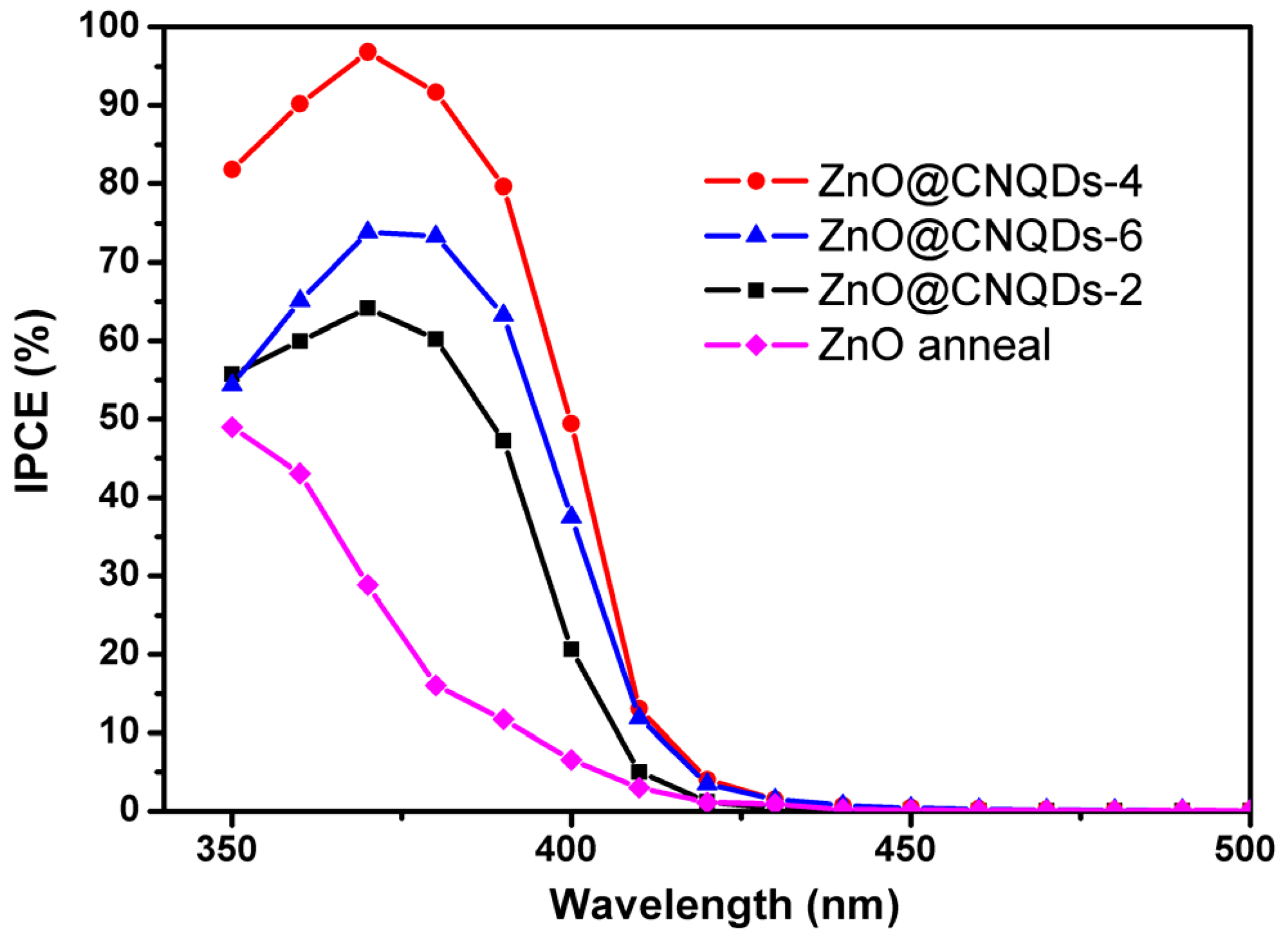
2.2. PEC Performance
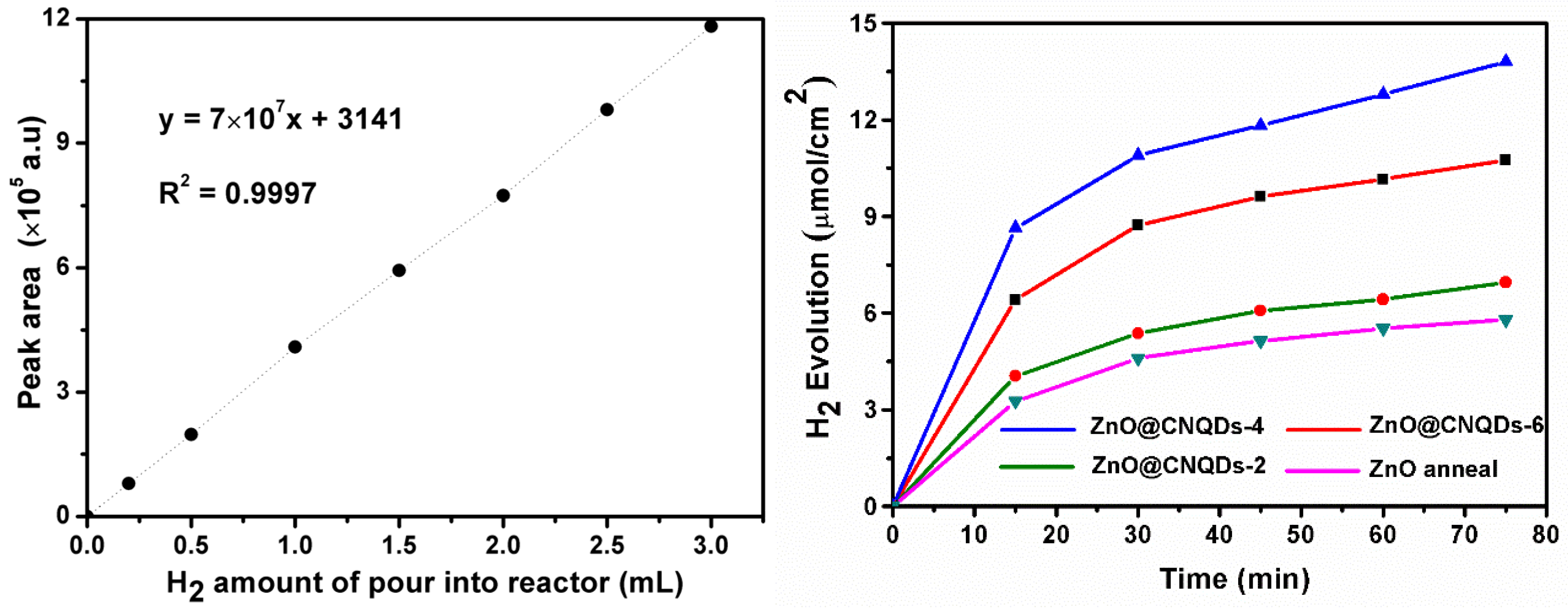
2.3. Photocatalytic Hydrogen Production
2.4. Mechanism of the Photoelectrochemistry Hydrogen Evolution
3. Materials and Methods
3.1. Catalyst Preparation
3.1.1. g-C3N4 Quantum Dots (CNQDs) Preparation
3.1.2. ZnO Nanowire Arrays Preparation
3.1.3. ZnO@CNQD Composites Electrode Preparation
3.2. Characterization
3.3. PEC Measurements and Photoelectric Hydrogen Production
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Harvey, B.G.; Merriman, W.W.; Koontz, T.A. High-Density Renewable Diesel and Jet Fuels Prepared from Multi cyclic Sesqui terpanes and a 1-Hexene-Derived Synthetic Paraffinic Kerosene. Energy Fuels 2015, 29, 2431–2436. [Google Scholar] [CrossRef]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N. S. Solar Water Splitting Cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- Bak, T.; Nowotny, J.; Rekas, M.; Sorrell, C.C. Photo-electrochemical hydrogen generation from water using solar energy. Materials-related aspects. Int. J. Hydrogen Energy 2002, 27, 991–1022. [Google Scholar] [CrossRef]
- Hisatomi, T.; Kubota, J.; Domen, K. Recent advances in semiconductors for photocatalytic and photoelectrochemical water splitting. Chem. Soc. Rev. 2014, 43, 7520–7535. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.D.; Wong, L.H.; Barber, J.; Loo, S.C. Recent advances in hybrid photocatalysts for solar fuel production. Energy Environ. Sci. 2012, 5, 5902–5918. [Google Scholar] [CrossRef]
- Chen, X.; Shen, S.; Guo, L.; Mao, S.S. Semiconductor-based photocatalytic hydrogen generation. Chem. Rev. 2010, 110, 6503–6570. [Google Scholar] [CrossRef] [PubMed]
- Pinna, N.; Neri, G.; Antoniett, M.; Niederberger, M. Nonaqueous synthesis of nanocrystalline semiconducting metal oxides for gas sensing. Angew. Chem. Int. Ed. 2004, 116, 4445–4449. [Google Scholar] [CrossRef]
- Shankar, K.; Mor, G.K.; Prakasam, H.E.; Yoriya, S.; Paulose, M.; Varghese, O.K.; Grimes, C. Highly-ordered TiO2 nanotube arrays up to 220 µm in length: use in water photoelectrolysis and dye-sensitized solar cells. Nanotechnology 2007, 18, 065707. [Google Scholar] [CrossRef]
- Zheng, J.Y.; Son, S.I.; Van, K.T.; Kang, Y.S. Preparation of α-Fe2O3 films by electrodeposition and photodeposition of Co-Pi on them to enhance their photoelectrochemical properties. RSC Adv. 2015, 5, 36307–36314. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, F.; Mou, Z.; Liu, X.; Sun, J.; Lei, W. A facile one-step synthesis of ZnO quantum dots modified poly (triazine imide) nanosheets for enhanced hydrogen evolution under visible light. Chem. Commun. 2016, 52, 13020–13023. [Google Scholar] [CrossRef] [PubMed]
- Pawar, A.U.; Kim, C.W.; Kang, M.J.; Kang, Y.S. Facile Fabrication of WO3 Nanoplates Thin Films with Dominant Crystal Facet of (002) for Water Splitting. Nano Energy 2016, 20, 156–167. [Google Scholar] [CrossRef]
- Zhang, J.Z. Metal oxide nanomaterials for solar hydrogen generation from photoelectrochemical water splitting. MRS Bull 2011, 36, 48–55. [Google Scholar] [CrossRef]
- Wang, T.; Jin, B.; Jiao, Z.; Lu, G.; Ye, J.; Bi, Y. Photo-directed growth of Au nanowires on ZnO arrays for enhancing photoelectrochemical performances. J. Mater. Chem. A 2014, 2, 15553–15559. [Google Scholar] [CrossRef]
- Mclaren, A.; Valdes-Solis, T.; Li, G.; Tsang, S.C. Shape and size effects of ZnO nanocrystals on photocatalytic activity. J. Am. Chem. Soc. 2009, 131, 12540–12541. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Jang, J.; Kim, J.; Lee, J.; Choi, W.; Lee, K. Three-Dimensional Type II ZnO/ZnSe Heterostructures and Their Visible Light Photocatalytic Activities. Langmuir 2011, 27, 10243–10250. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Jin, B.; Jiao, Z.; Lu, G.; Ye, J.; Bi, Y. Electric field-directed growth and photoelectrochemical properties of cross-linked Au-ZnO hetero-nanowire arrays. Chem. Commun. 2015, 51, 2103–2106. [Google Scholar] [CrossRef] [PubMed]
- Ai, G.; Li, H.; Liu, S.; Mo, R.; Zhong, J. Solar Water Splitting by TiO2/CdS/Co–Pi Nanowire Array Photoanode Enhanced with Co–Pi as Hole Transfer Relay and CdS as Light Absorber. Adv. Funct. Mater. 2015, 25, 5706–5713. [Google Scholar] [CrossRef]
- Guo, C.X.; Xie, J.; Yang, H.; Li, C.M. Au@CdS Core–Shell Nanoparticles-Modified ZnO Nanowires Photoanode for Efficient Photoelectrochemical Water Splitting. Adv. Sci. 2015, 2, 2084–2088. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Jiao, Z.; Bi, Y. Efficient Charge Separation between Bi2MoO6 Nanosheets and ZnO Nanowires for Enhanced Photoelectrochemical Properties. J. Mater. Chem. A 2015, 3, 19702–19705. [Google Scholar] [CrossRef]
- Hao, X.Q.; Jin, Z.L.; Min, S.X.; Lu, G.X. Modulating photogenerated electron transfer with selectively exposed Co–Mo facets on a novel amorphous g-C3N4/CoxMo1−xS2 photocatalyst. RSC Adv. 2016, 6, 23709–23717. [Google Scholar] [CrossRef]
- Hao, X.Q.; Jin, Z.L.; Lu, G.X. Enhanced Surface Electron Transfer with the Aid of Methyl Viologen on the Co3O4-g-C3N4 Photocatalyst. Chem. Lett. 2016, 45, 116–118. [Google Scholar] [CrossRef]
- Li, C.; Wang, Z.; Sui, X.; Zhang, L.; Gu, D. Graphitic-C3N4 quantum dots modified carbon nanotubes as novel support material for low Pt loading fuel cell catalyst. RSC Adv. 2016, 6, 32290–32297. [Google Scholar] [CrossRef]
- Li, G.; Lian, Z.; Wang, W.; Zhang, D.; Li, H. Nanotube-confinement induced size-controllable g-C3N4 quantum dots modified single-crystalline TiO2 nanotube arrays for stable synergetic photoelectrocatalysis. Nano Energy 2016, 19, 446–454. [Google Scholar] [CrossRef]
- Pan, D.; Xi, C.; Li, Z.; Wang, L.; Chen, Z.; Lu, B.; Wu, M. Electrophoretic fabrication of highly robust, efficient, and benign hetero junction photoelectrocatalysts based on graphene-quantum-dot sensitized TiO2 nanotube arrays. J. Mater. Chem. A 2013, 1, 3551–3555. [Google Scholar] [CrossRef]
- Hao, X.Q.; Jin, Z.L.; Xu, J.; Min, S.X.; Lu, G.X. Functionalization of TiO2 with graphene quantum dots for efficient photocatalytic hydrogen evolution. Superlattices Microstruct. 2016, 94, 237–244. [Google Scholar] [CrossRef]
- Hao, X.Q.; Yang, H.; Jin, Z.L.; Xu, J.; Min, S.X.; Lu, G.X. Quantum confinement effect of graphene-like C3N4 nanosheets for efficient photocatalytic hydrogen production from water splitting. Acta Phys. Chim. Sin. 2016, 32, 2581–2592. [Google Scholar]
- Zhuo, S.; Shao, M.; Lee, S. Comment on “Upconversion and downconversion fluorescent graphene quantum dots: ultrasonic preparation and photocatalysis”. ACS Nano 2012, 6, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Li, X.; Yang, Z.; Xu, S.; Zhang, W.; Su, Y.; Hu, N.; Lu, W.; Feng, J.; Zhang, Y. Morphology control and photocatalysis enhancement by in-situ hybridization of Cu2O with nitrogen-doped carbon quantum dots. Langmuir 2016, 32, 9418–9427. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Zhu, L.; Chen, G. Ultrasmall graphitic carbon nitride quantum dots decorated self-organized TiO2, nanotube arrays with highly efficient photoelectrochemical activity. Appl. Catal. B Environ. 2016, 186, 127–135. [Google Scholar] [CrossRef]
- Niu, P.; Zhang, L.; Liu, G.; Cheng, H. Graphene-Like Carbon Nitride Nanosheets for Improved Photocatalytic Activities. Adv. Funct. Mater. 2012, 22, 4763–4770. [Google Scholar] [CrossRef]
- Wang, W.; Yu, J.C.; Xia, D.; Wong, P.K.; Li, Y. Graphene and g-C3N4 nanosheets cowrapped elemental α-sulfur as a novel metal-free heterojunction photocatalyst for bacterial inactivation under visible-light. Environ. Sci. Technol. 2013, 47, 8724–8732. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Yan, X.; Kang, Z.; Liang, Q.; Liao, X.; Zhang, Y. Band alignment engineering for improved performance and stability of ZnFe2O4, modified CdS/ZnO nanostructured photoanode for PEC water splitting. Nano Energy. 2016, 24, 25–31. [Google Scholar] [CrossRef]
- Vijayan, T.A.; Chandramohan, R.; Valanarasu, S.; Thirumalai, J.; Venkateswaran, S.; Mahalingam, T.; Srikumar, S.R. Optimization of growth conditions of ZnO nano thin films by chemical double dip technique. Sci. Technol. Adv. Mater. 2008, 9, 035007. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.C.; Li, Z.S.; Zou, Z.G. Photodegradation performance of g-C3N4 fabricated by directly heating melamine. Langmuir 2009, 25, 10397–10410. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Ma, D.; Zheng, H.; Li, X.; Cheng, M.; Bao, X. Fe-doped and -mediated graphitic carbon nitride nanosheets for enhanced photocatalytic performance under natural sunlight. Microporous Mesoporous Mater. 2008, 110, 216–222. [Google Scholar] [CrossRef]
- Thomas, A.; Fischer, A.; Goettmann, F.; Antonietti, M.; Müller, J.; Schlöglb, R.; Carlsson, J.M. ChemInform Abstract: Graphitic Carbon Nitride Materials: Variation of Structure and Morphology and Their Use as Metal-Free Catalysts. J. Mater. Chem. 2008, 18, 4893–4908. [Google Scholar] [CrossRef]
- Choi, Y.; Baek, M.; Zhang, Z.; Dao, V.D.; Choi, H.S.; Yong, K. A two-storey structured photoanode of a 3D Cu2ZnSnS4/CdS/ZnO@ steel composite nanostructure for efficient photoelectrochemical hydrogen generation. Nanoscale 2015, 7, 15291–15299. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Zhang, H.; Ren, W.; Dong, W.; Sun, Y. Three dimensional urchin-like ordered hollow TiO2/ZnO nanorods structure as efficient photoelectrochemical anode. Nano Energy 2013, 2, 779–786. [Google Scholar] [CrossRef]
- Wang, G.; Yang, X.; Qian, F.; Zhang, J.Z.; Li, Y. Double-Sided CdS and CdSe Quantum Dot Co-Sensitized ZnO Nanowire Arrays for Photoelectrochemical Hydrogen Generation. Nano Lett. 2010, 10, 1088–1092. [Google Scholar] [CrossRef] [PubMed]
- Cha, H.G.; Kang, M.J.; Hwang, I.C.; Kim, H.; Yoon, K.B.; Kang, Y.S. Manual assembly of nanocrystals for enhanced photoelectrochemical efficiency of hematite film. Chem. Commun. 2015, 51, 6407–6410. [Google Scholar] [CrossRef] [PubMed]
- Van, T.K.; Pham, L.Q.; Kim, D.Y.; Zheng, J.Y.; Kim, D.; Pawar, A.U.; Kang, Y.S. Facile Fabrication of WO3 Nanoplates Thin Films with Dominant Crystal Facet of (002) for Water Splitting. Chemsuschem 2014, 7, 3505–3512. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.; Jin, Z.; Hu, H.; Lu, G.; Bi, Y. Fivefold Enhanced Photoelectrochemical Properties of ZnO Nanowire Arrays Modified with C3N4 Quantum Dots. Catalysts 2017, 7, 99. https://doi.org/10.3390/catal7040099
Yang H, Jin Z, Hu H, Lu G, Bi Y. Fivefold Enhanced Photoelectrochemical Properties of ZnO Nanowire Arrays Modified with C3N4 Quantum Dots. Catalysts. 2017; 7(4):99. https://doi.org/10.3390/catal7040099
Chicago/Turabian StyleYang, Hao, Zhiliang Jin, Hongyan Hu, Gongxuan Lu, and Yingpu Bi. 2017. "Fivefold Enhanced Photoelectrochemical Properties of ZnO Nanowire Arrays Modified with C3N4 Quantum Dots" Catalysts 7, no. 4: 99. https://doi.org/10.3390/catal7040099
APA StyleYang, H., Jin, Z., Hu, H., Lu, G., & Bi, Y. (2017). Fivefold Enhanced Photoelectrochemical Properties of ZnO Nanowire Arrays Modified with C3N4 Quantum Dots. Catalysts, 7(4), 99. https://doi.org/10.3390/catal7040099





