Merging Metallic Catalysts and Sonication: A Periodic Table Overview
Abstract
:1. Introduction
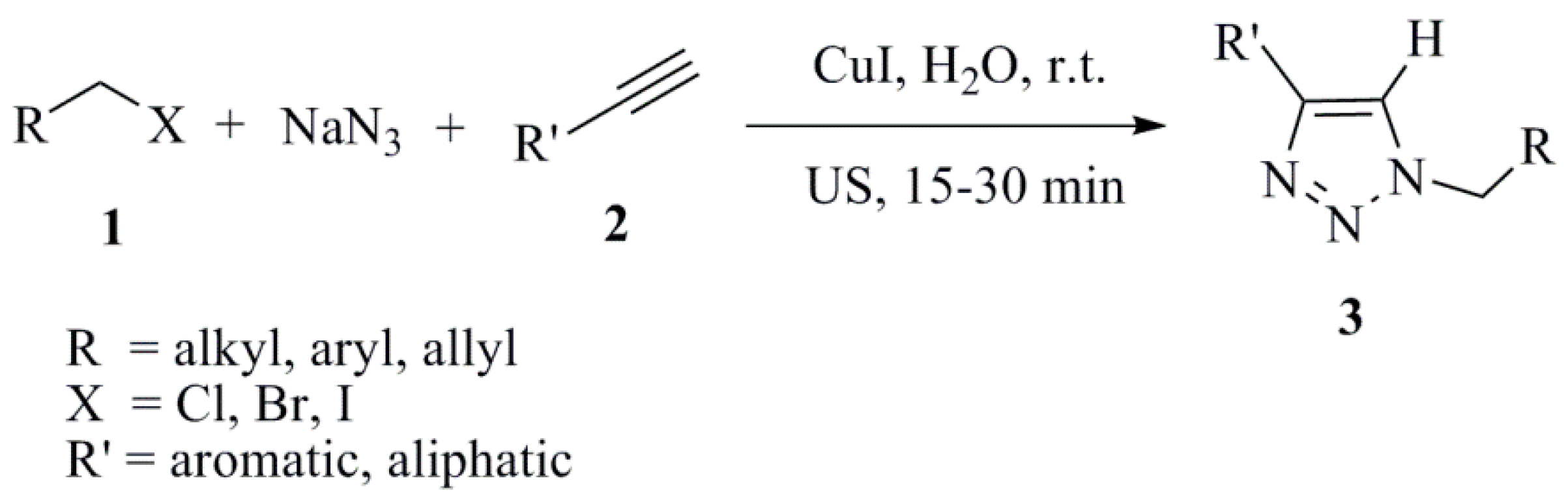
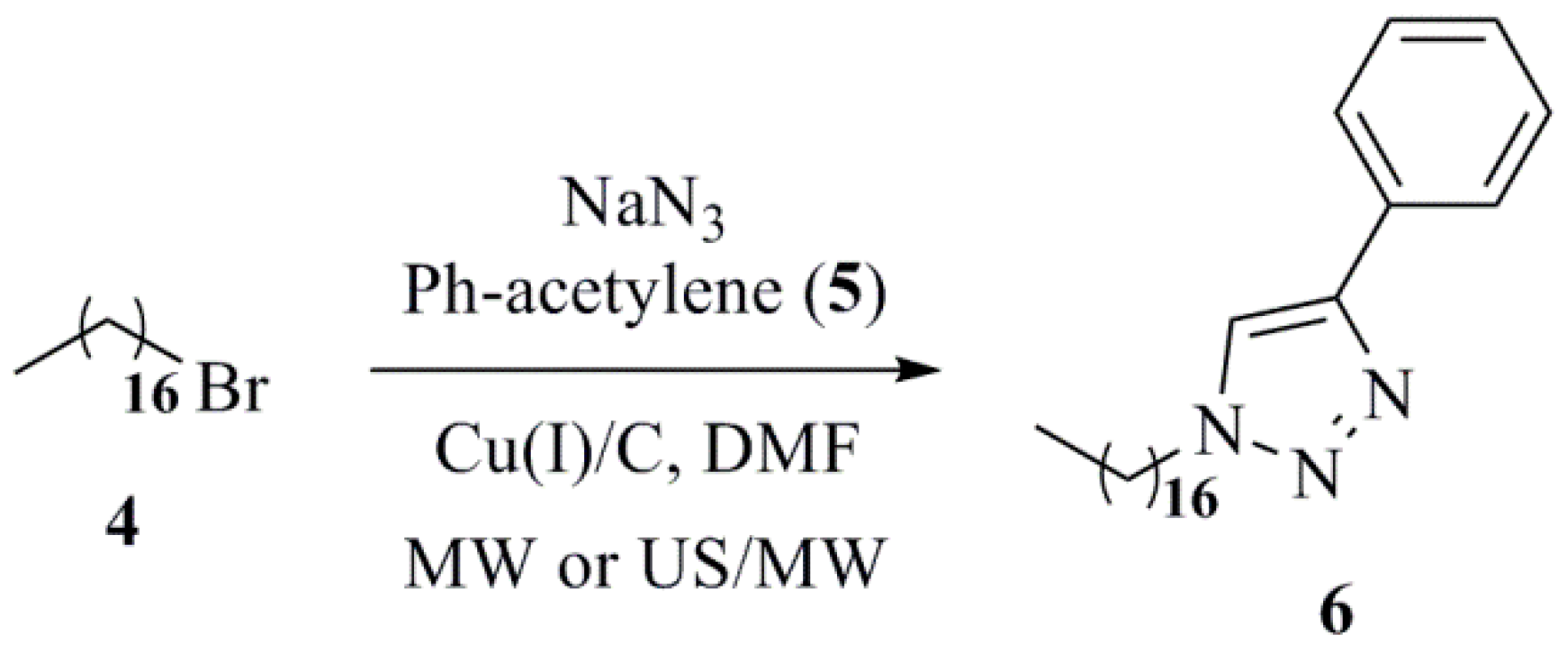
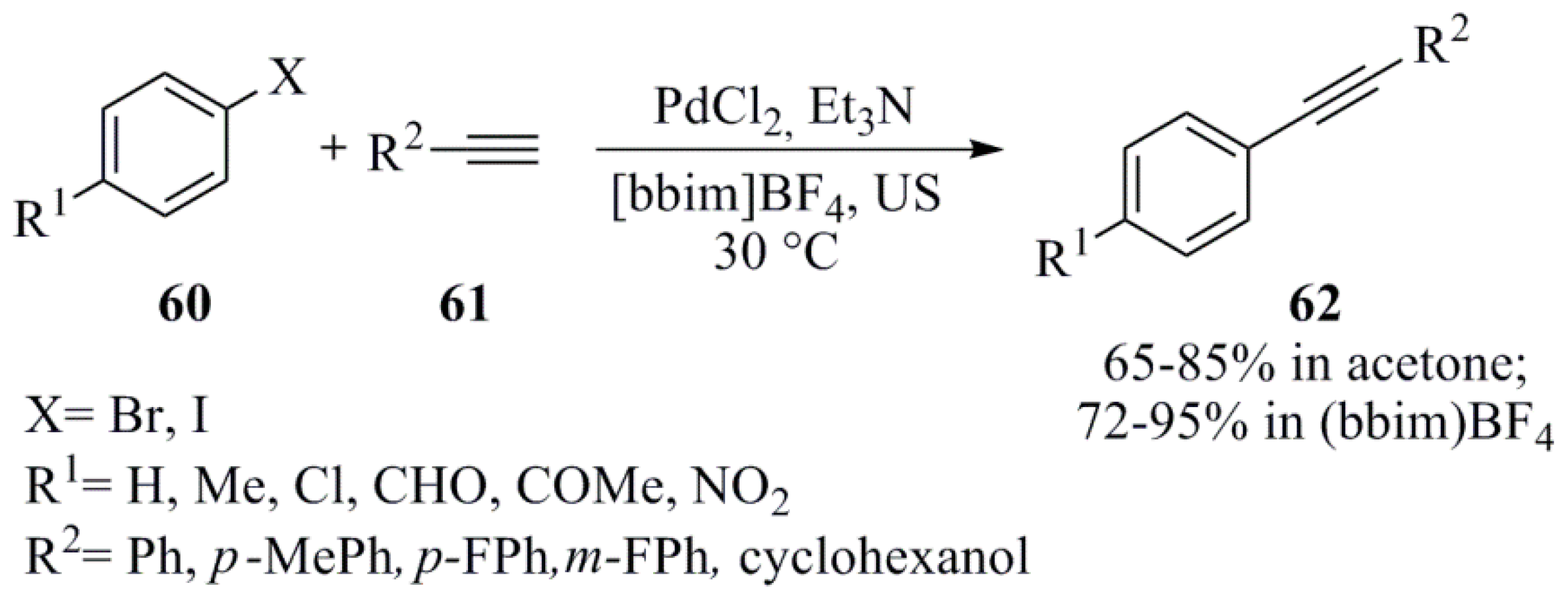
2. Transition Metals and Supported Transition Metal Reagents
3. Main Group Metal Derivatives
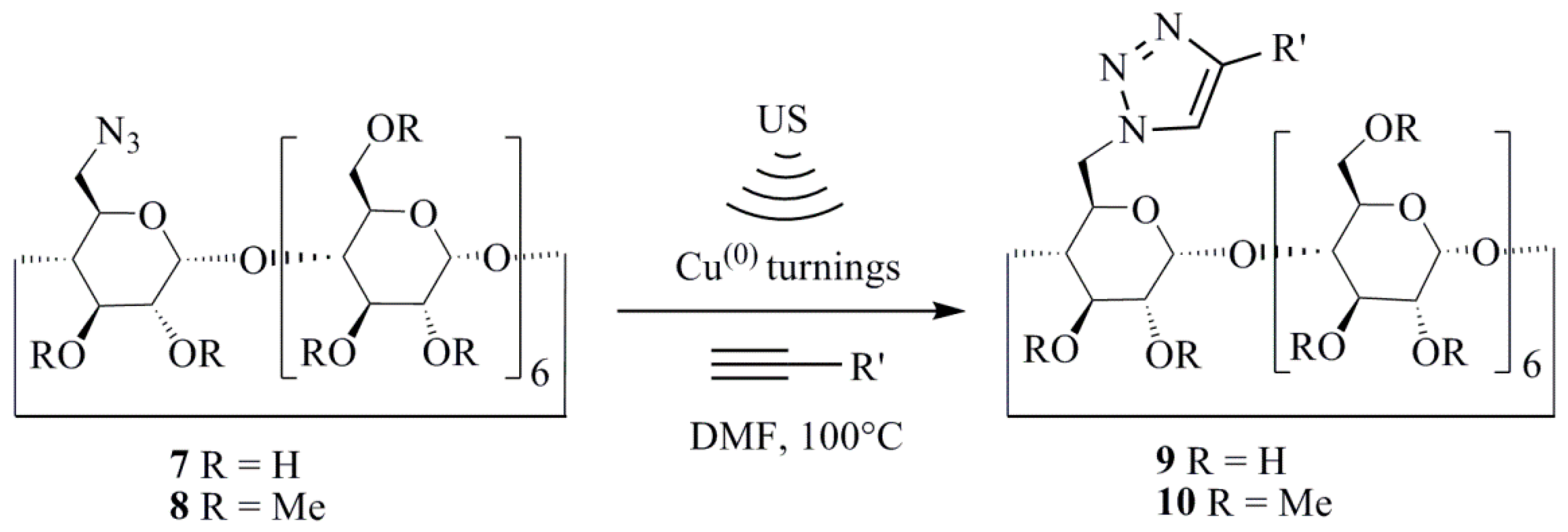
4. Nanostructured Materials from Transition Elements
4.1. First-Row Transition Metal NPs
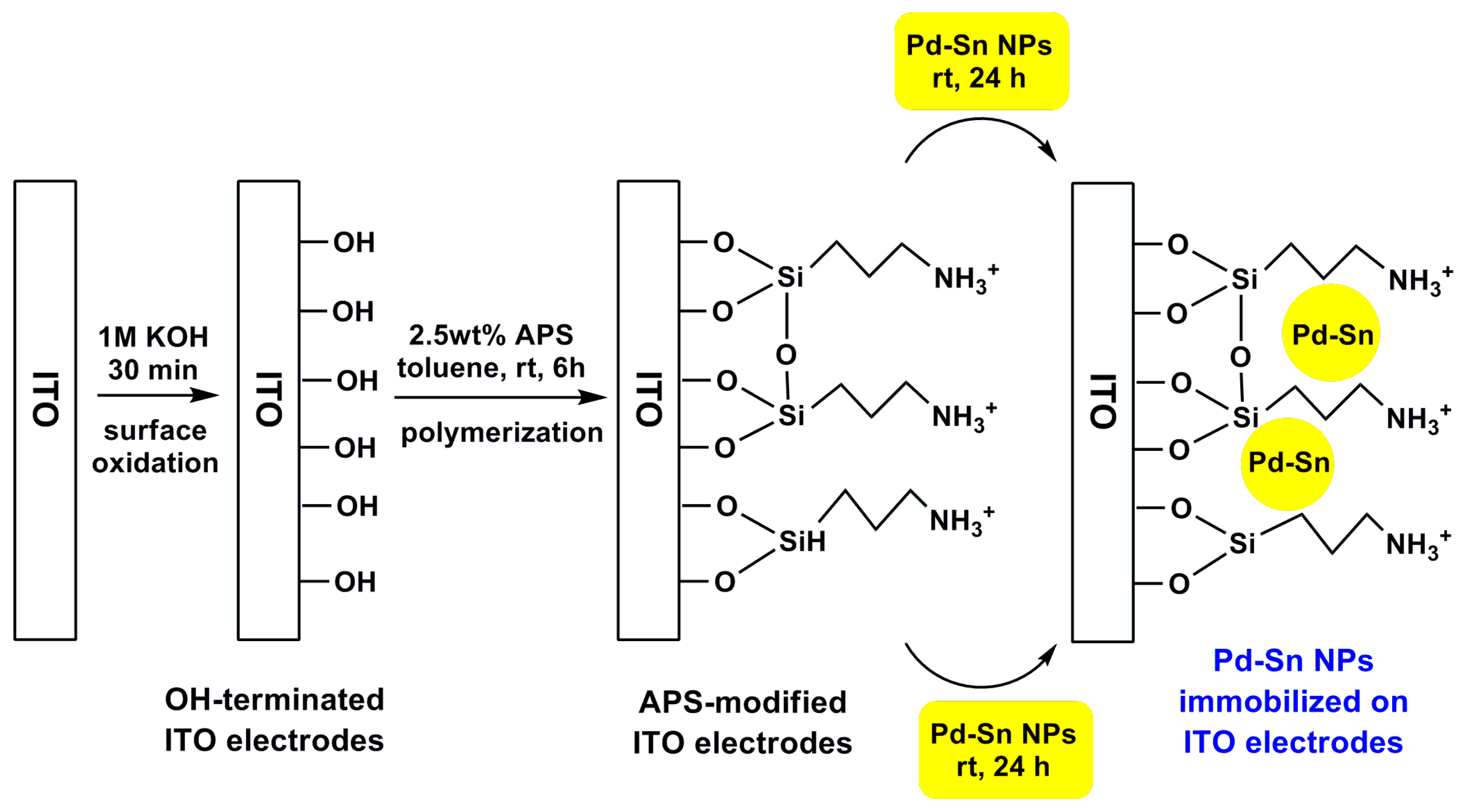
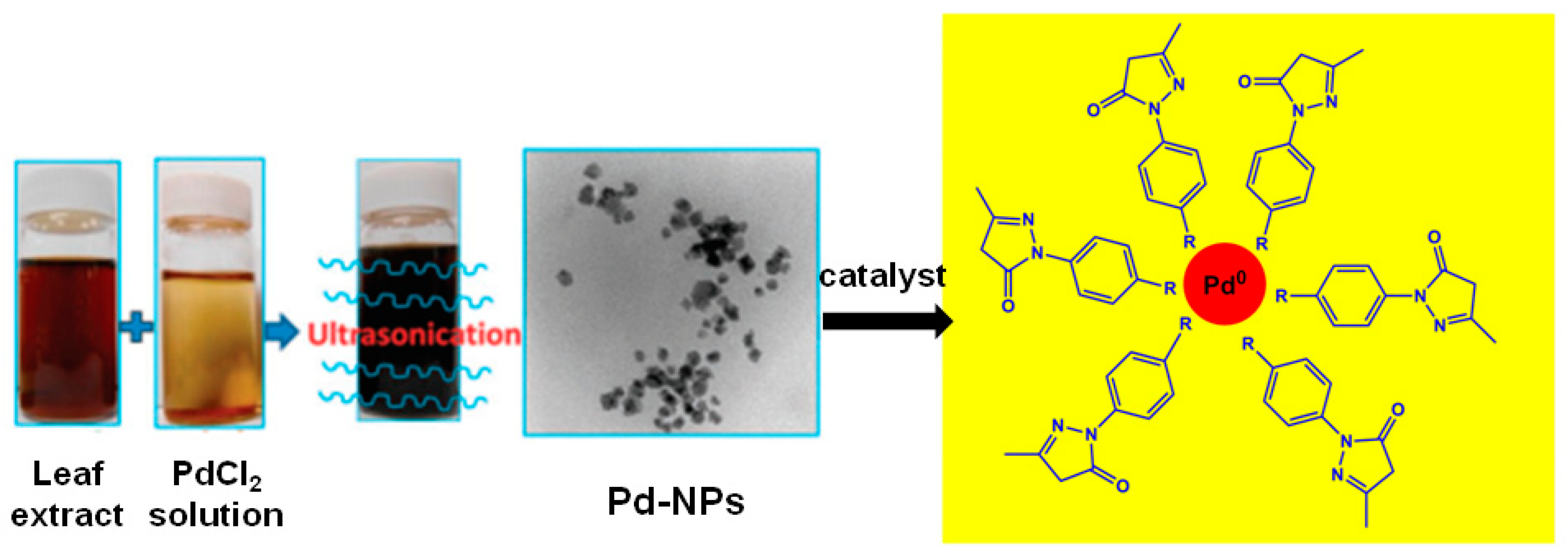
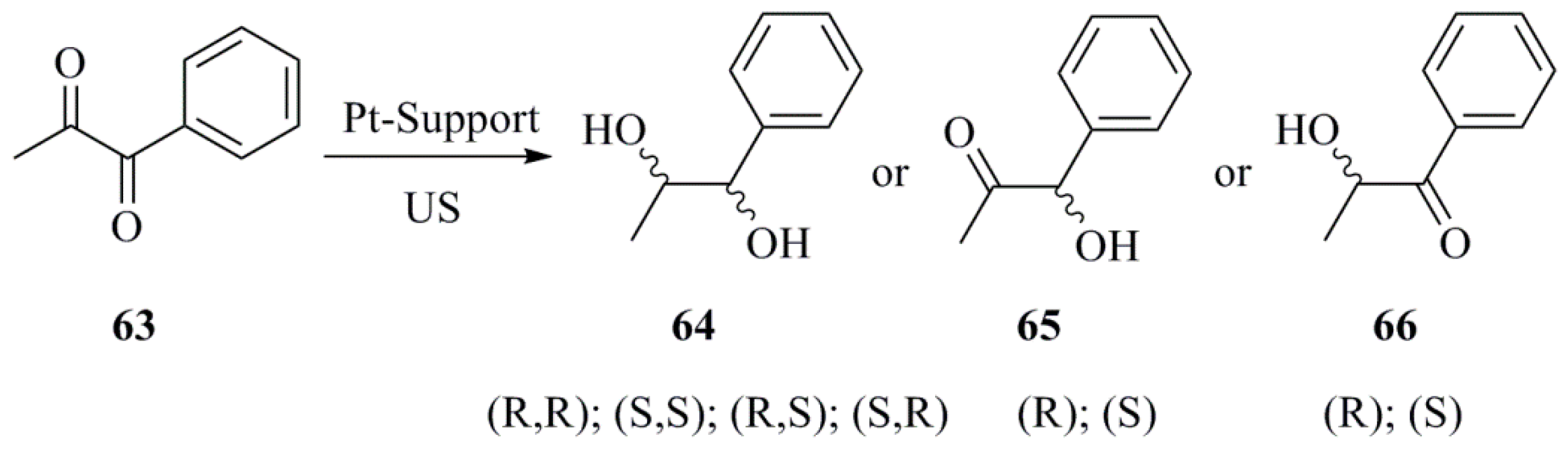
4.2. Pd and Pt NPs
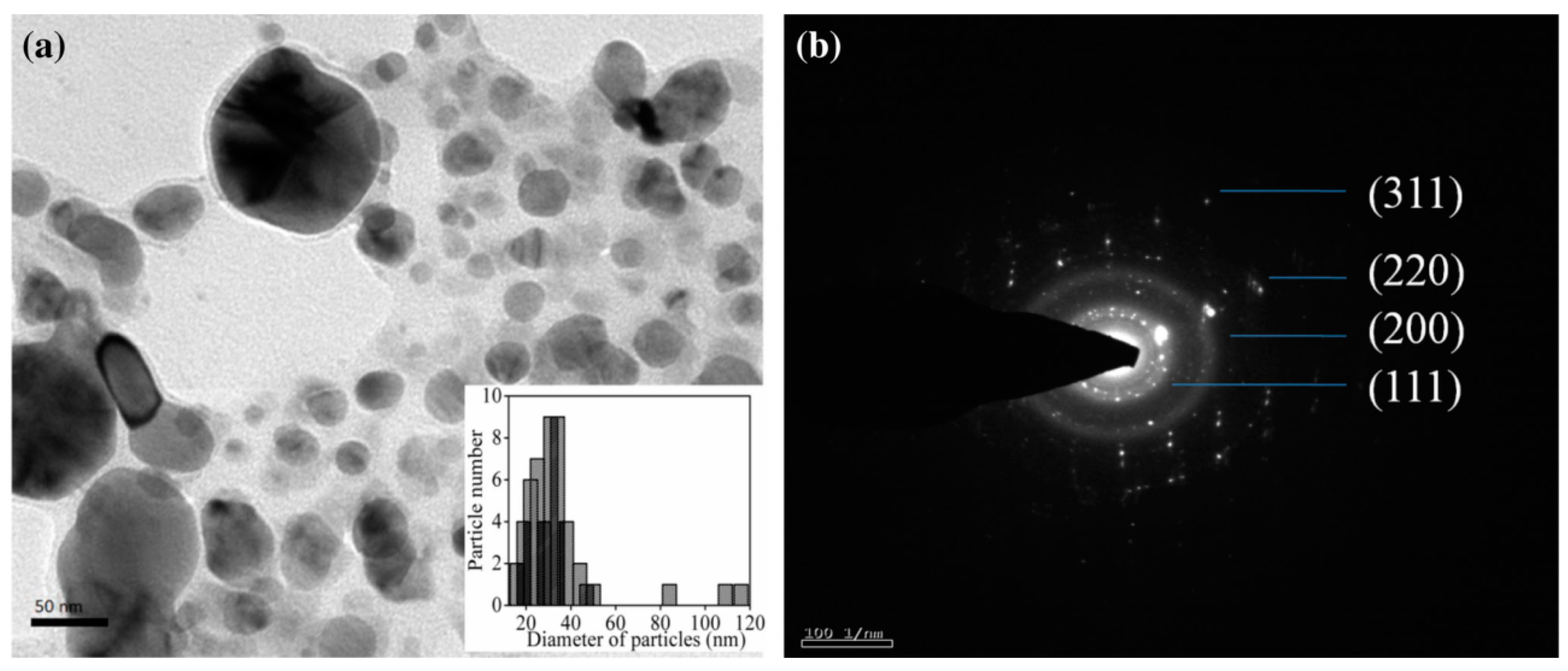
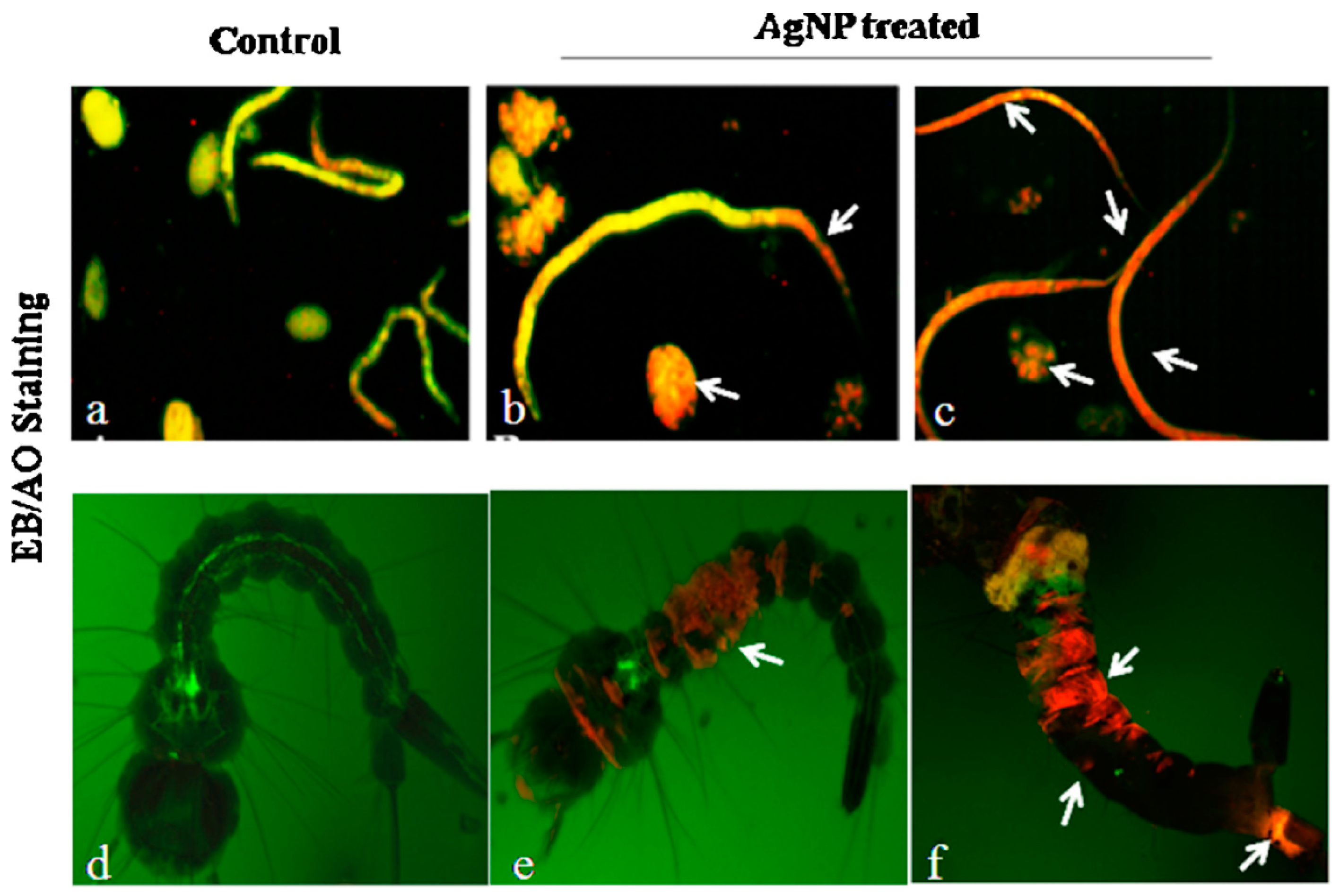
4.3. Ag and Au NPs
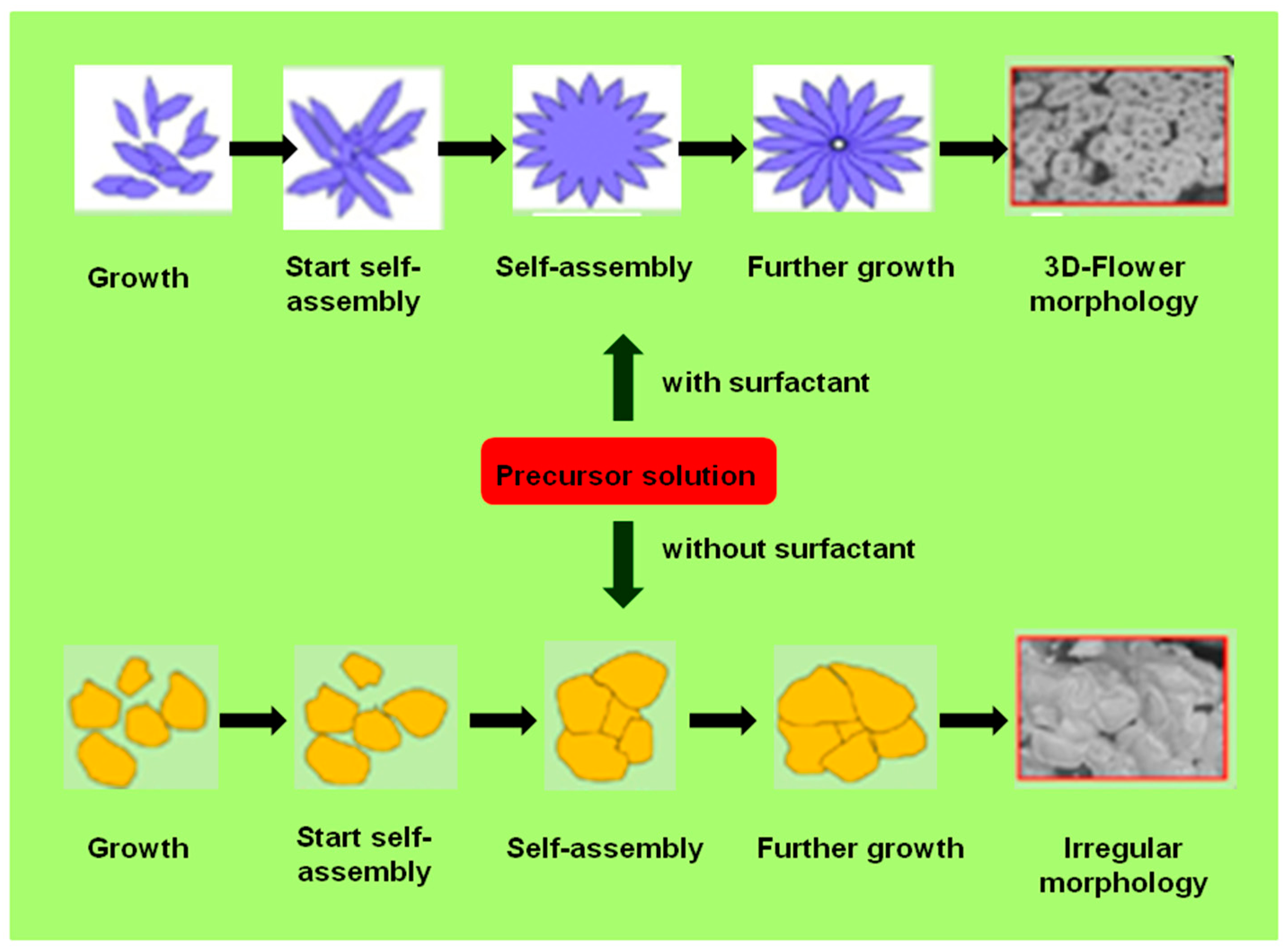
4.4. Lanthanide NPs
4.5. Main-Group NPs
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Suslick, K.S.; Doktycz, S.J. Effects of ultrasound on surfaces and solids. In Advances in Sonochemistry; Mason, T.J., Ed.; JAI Press Ltd.: London, UK, 1990; Volume 1, pp. 197–230. [Google Scholar]
- Pugin, B.; Turner, A.T. Influence of ultrasound on reactions with metals. In Advances in Sonochemistry; Mason, T.J., Ed.; JAI Press Ltd.: London, UK, 1990; Volume 1, pp. 81–118. [Google Scholar]
- Tomlinson, W.J. Effect of ultrasonically induced cavitation on corrosion. In Advances in Sonochemistry; Mason, T.J., Ed.; JAI Press Ltd.: London, UK, 1990; Volume 1, pp. 173–195. [Google Scholar]
- Luche, J.-L. Sonochemistry. From experiment to theoretical considerations. In Advances in Sonochemistry; Mason, T.J., Ed.; JAI Press Ltd.: London, UK, 1993; Volume 3, pp. 85–124. [Google Scholar]
- Cintas, P.; Luche, J.-L. Organometallic sonochemistry. In Synthetic Organic Sonochemistry; Luche, J.-L., Ed.; Plenum Press: New York, NY, USA, 1998; Chapter 5; pp. 167–234. [Google Scholar]
- Luche, J.-L. Ultrasonically promoted carbonyl addition reactions. In Advances in Sonochemistry; Mason, T.J., Ed.; JAI Press Ltd.: London, UK, 1990; Volume 1, pp. 119–171. [Google Scholar]
- Cintas, P.; Palmisano, G.; Cravotto, G. Power ultrasound in metal-assisted synthesis: From classical Barbier-like reactions to click chemistry. Ultrason. Sonochem. 2011, 18, 836–841. [Google Scholar] [CrossRef] [PubMed]
- Manickam, S.; Kumar Rana, R. Production of nanomaterials using ultrasonic cavitation—A simple, energy efficient and technological approach. In Ultrasound Technologies for Food and Bioprocessing; Feng, H., Barbosa-Cánovas, G.V., Weiss, J., Eds.; Springer: Heidelberg, Germany, 2011; Chapter 15; pp. 405–444. [Google Scholar]
- Gedanken, A. Doping nanoparticles into polymers and ceramics using ultrasonic irradiation. Ultrason. Sonochem. 2007, 14, 418–430. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zeiger, B.W.; Suslick, K.S. Sonochemical synthesis of nanomaterials. Chem. Soc. Rev. 2013, 42, 2555–2567. [Google Scholar] [CrossRef] [PubMed]
- Verdan, S.; Burato, G.; Comet, M.; Reinert, L.; Fuzellier, H. Structural changes of metallic surfaces induced by ultrasound. Ultrason. Sonochem. 2003, 10, 291–295. [Google Scholar] [CrossRef]
- Skorb, E.V.; Möhwald, H.; Irrgang, T.; Fery, A.; Andreeva, D.V. Ultrasound-assisted design of metal nanocomposites. Chem. Commun. 2010, 46, 7897–7899. [Google Scholar] [CrossRef] [PubMed]
- Cravotto, G.; Cintas, P. Power ultrasound in organic synthesis: Moving cavitational chemistry from academia to innovative and large-scale applications. Chem. Soc. Rev. 2006, 35, 180–196. [Google Scholar] [CrossRef] [PubMed]
- Gedanken, A. Using sonochemistry for the fabrication of nanomaterials. Ultrason. Sonochem. 2004, 11, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Gawande, M.B.; Bonifácio, V.D.B.; Luque, R.; Branco, P.S.; Varma, R.S. Solvent-free and catalysts-free chemistry: A benign pathway to sustainability. ChemSusChem 2014, 7, 24–44. [Google Scholar] [CrossRef] [PubMed]
- Thirumurugan, P.; Matosiuk, D.; Jozwiak, K. Click chemistry for drug development and diverse chemical-biology applications. Chem. Rev. 2013, 113, 4905–4979. [Google Scholar] [CrossRef] [PubMed]
- Witczak, Z.J.; Bielski, R. Click Chemistry in Glycoscience: New Developments and Strategies; Wiley-VCH: Weinheim, Germany, 2013. [Google Scholar]
- Goldmann, A.S.; Glassner, M.; Inglis, A.J.; Barner-Kowollik, C. Post-functionalization of polymers via orthogonal ligation chemistry. Macromol. Rap. Commun. 2013, 34, 810–849. [Google Scholar] [CrossRef] [PubMed]
- Sreedhar, B.; Surendra Reddy, P. Sonochemical synthesis of 1,4-disubstituted 1,2.3-triazoles in aqueous medium. Synth. Commun. 2007, 37, 805–812. [Google Scholar] [CrossRef]
- Cintas, P.; Martina, K.; Robaldo, B.; Garella, A.; Boffa, L.; Cravotto, G. Improved protocols for microwave-assisted Cu(I)-catalyzed Huisgen 1,3-dipolar cycloadditions. Collect. Czech. Chem. Commun. 2007, 72, 1014–1024. [Google Scholar] [CrossRef]
- Cravotto, G.; Fokin, V.V.; Garella, D.; Binello, A.; Barge, A. Huisgen 1,3-dipolar cycloaddition catalyzed by metallic copper under ultrasound. J. Comb. Chem. 2010, 12, 13–15. [Google Scholar] [CrossRef] [PubMed]
- Cintas, P.; Barge, A.; Tagliapietra, T.; Boffa, L.; Cravotto, G. Alkyne-azide click reaction catalyzed by metallic copper under ultrasound. Nat. Protoc. 2010, 5, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Klufers, P.; Piotrowski, H.; Uhlendorf, J. Homoleptic cuprates(II) with multiply deprotonated α-cyclodextrin ligands. Chem. Eur. J. 1997, 3, 601–608. [Google Scholar] [CrossRef]
- Boffa, L.; Calcio Gaudino, E.; Martina, K.; Jicsinszky, L.; Cravotto, G. A new class of cationic cyclodextrins: Synthesis and chemico-physical properties. New J. Chem. 2010, 34, 2013–2019. [Google Scholar] [CrossRef]
- Jacob, K.; Stolle, A.; Ondruschka, B.; Jandt, K.D.; Keller, T.F. Cu on porous glass: An easily recyclable catalyst for the microwave-assisted azide–alkyne cycloaddition in water. Appl. Catal. A Gen. 2013, 451, 94–100. [Google Scholar] [CrossRef]
- Tu, N.P.; Hochlowski, J.E.; Djuric, S.W. Ultrasound-assisted click chemistry in continuous flow. Mol. Divers. 2012, 16, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Sreedhar, B.; Surendra Reddy, P.; Veda Prakash, B.; Ravindra, A. Ultrasound-assisted rapid and efficient synthesis of propargylamines. Tetrahedron Lett. 2005, 46, 7019–7022. [Google Scholar] [CrossRef]
- Cargnelutti, R.; Lang, E.S.; Schumacher, R.F. Bis(2-pyridyl)diselenoethers as versatile ligands for copper-catalyzed C-S bond formation in glycerol. Tetrahedron Lett. 2015, 56, 5218–5222. [Google Scholar] [CrossRef]
- Pirola, C.; Bianchi, C.L.; Di Michele, A.; Diodati, P.; Boffito, D.; Ragaini, V. Ultrasound and microwave assisted synthesis of high loading Fe-supported Fischer-Tropsch catalysts. Ultrason. Sonochem. 2010, 17, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Li, H.; Zhu, B.; Feng, Y.; Wang, S.; Zhang, S. Selective hydrogenation of furfural to furfuryl alcohol over catalysts prepared via sonochemistry. Ultrason. Sonochem. 2007, 14, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Arena, F.; Italiano, G.; Barbera, K.; Bonura, G.; Spadaro, L.; Frusteri, F. Basic evidences for methanol-synthesis catalyst design. Catal. Today 2009, 143, 80–85. [Google Scholar] [CrossRef]
- Shih-Yuan, A.; Dai, W.-C. A simple and highly efficient preparation of arylstannane(s) via sonochemical Barbier reaction. Tetrahedron Lett. 1996, 37, 495–498. [Google Scholar] [CrossRef]
- Lee, A.S.-Y.; Dai, W.-C. A facile and highly efficient sonochemical synthesis of organostannane via Barbier reaction. Tetrahedron 1997, 53, 859–868. [Google Scholar] [CrossRef]
- David-Quillot, F.; Lunot, S.; Marsacq, D.; Duchêne, A. A novel access to organogermanium compounds. Tetrahedron Lett. 2000, 41, 4905–4907. [Google Scholar] [CrossRef]
- Lamandé-Langle, S.; Abarbri, M.; Thibonnet, J.; Duchêne, A. A novel mode of access to polyfunctional organotin compounds and their reactivity in Stille cross-coupling reaction. J. Organomet. Chem. 2009, 694, 2368–2374. [Google Scholar] [CrossRef]
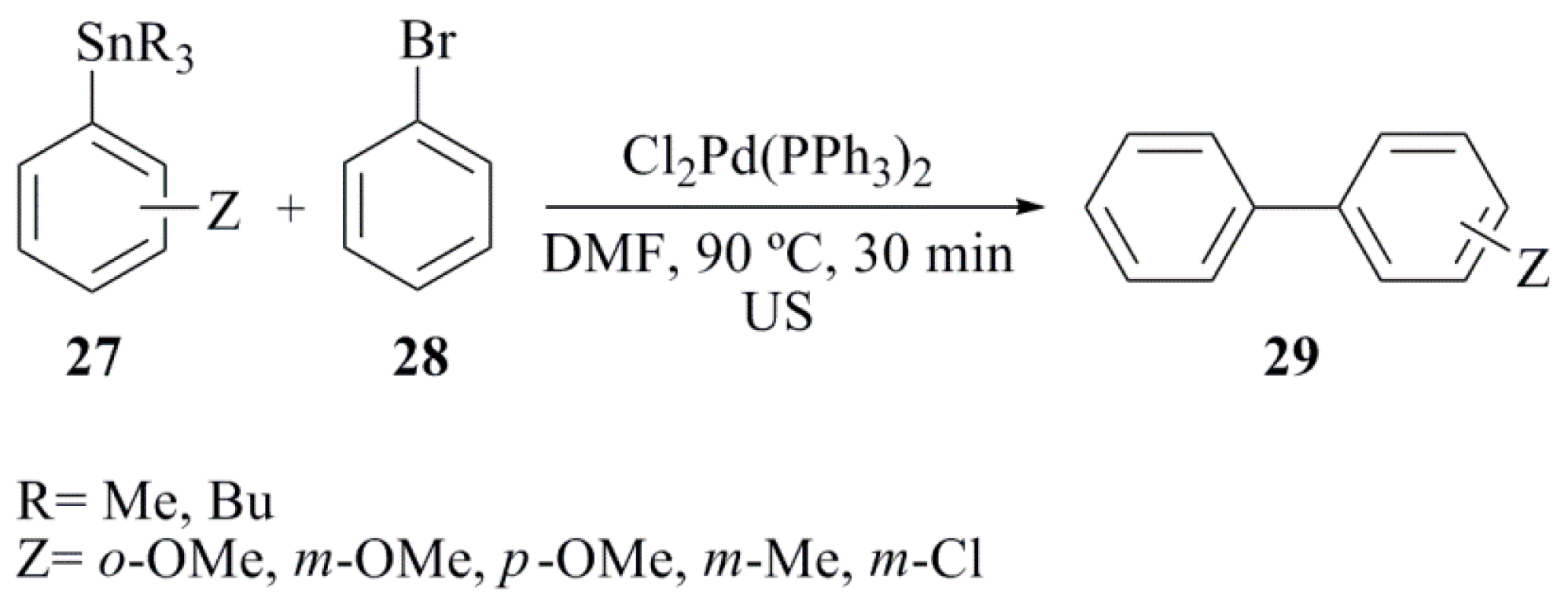
- Domini, C.E.; Silbestri, G.F.; Fernández Band, B.; Chopa, A.B. Ultrasound-assisted synthesis of unsymmetrical biaryls by Stille cross-coupling reactions. Ultrason. Sonochem. 2012, 19, 410–414. [Google Scholar] [CrossRef] [PubMed]
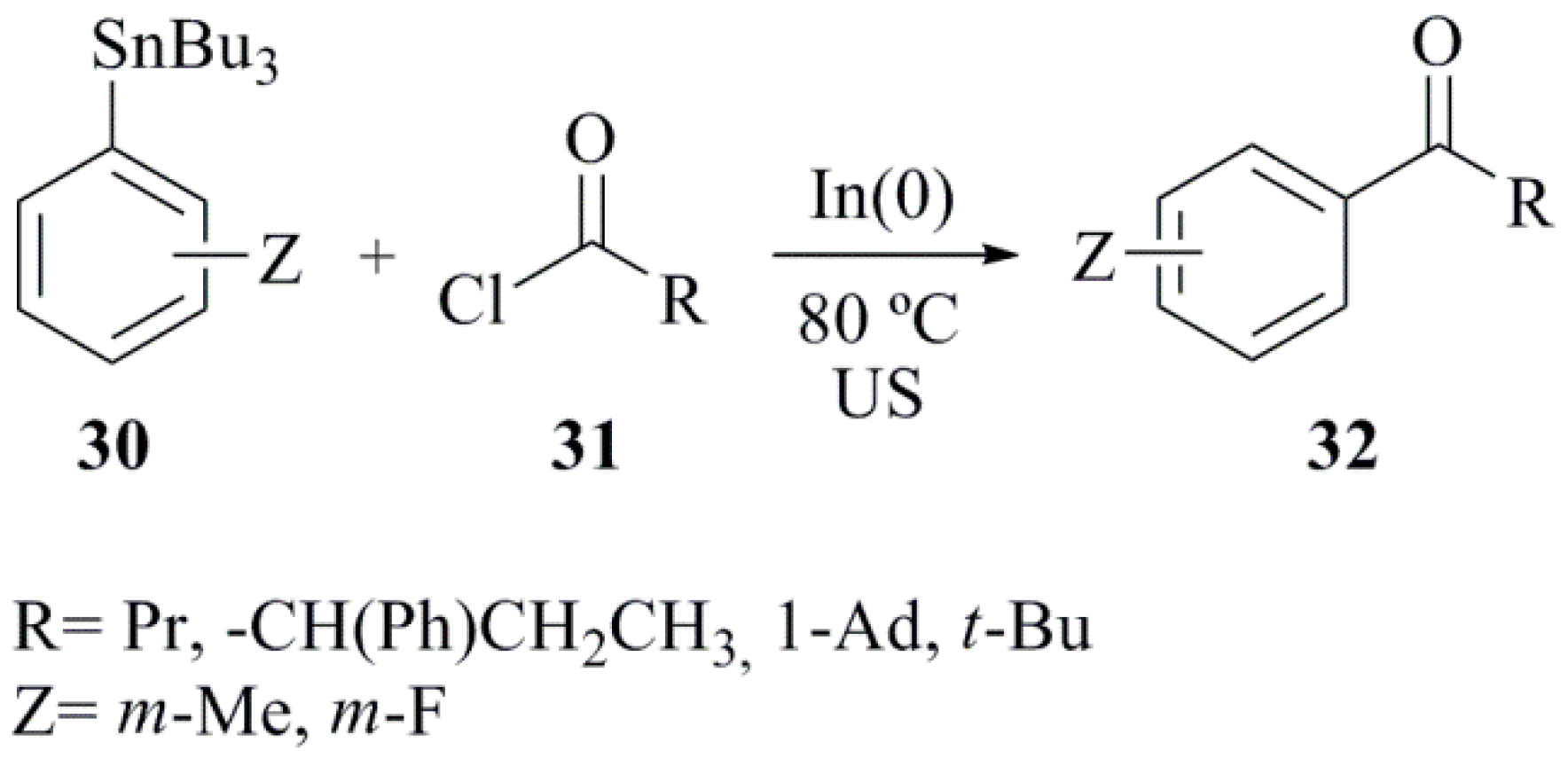
- Lo Fiego, M.J.; Badajoz, M.A.; Domini, C.; Chopa, A.B.; Lockhart, M.T. Indium-mediated regioselective synthesis of ketones from arylstannanes under solvent free ultrasound irradiation. Ultrason. Sonochem. 2013, 20, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Cintas, P. Synthetic organoindium chemistry: What makes indium so appealing? Synlett 1995, 1995, 1087–1096. [Google Scholar] [CrossRef]
- Shen, Z.-L.; Wang, S.-Y.; Chok, Y.-K.; Xu, Y.-H.; Loh, T.-P. Organoindium reagents: The preparation and application in organic synthesis. Chem. Rev. 2013, 113, 271–401. [Google Scholar] [CrossRef] [PubMed]
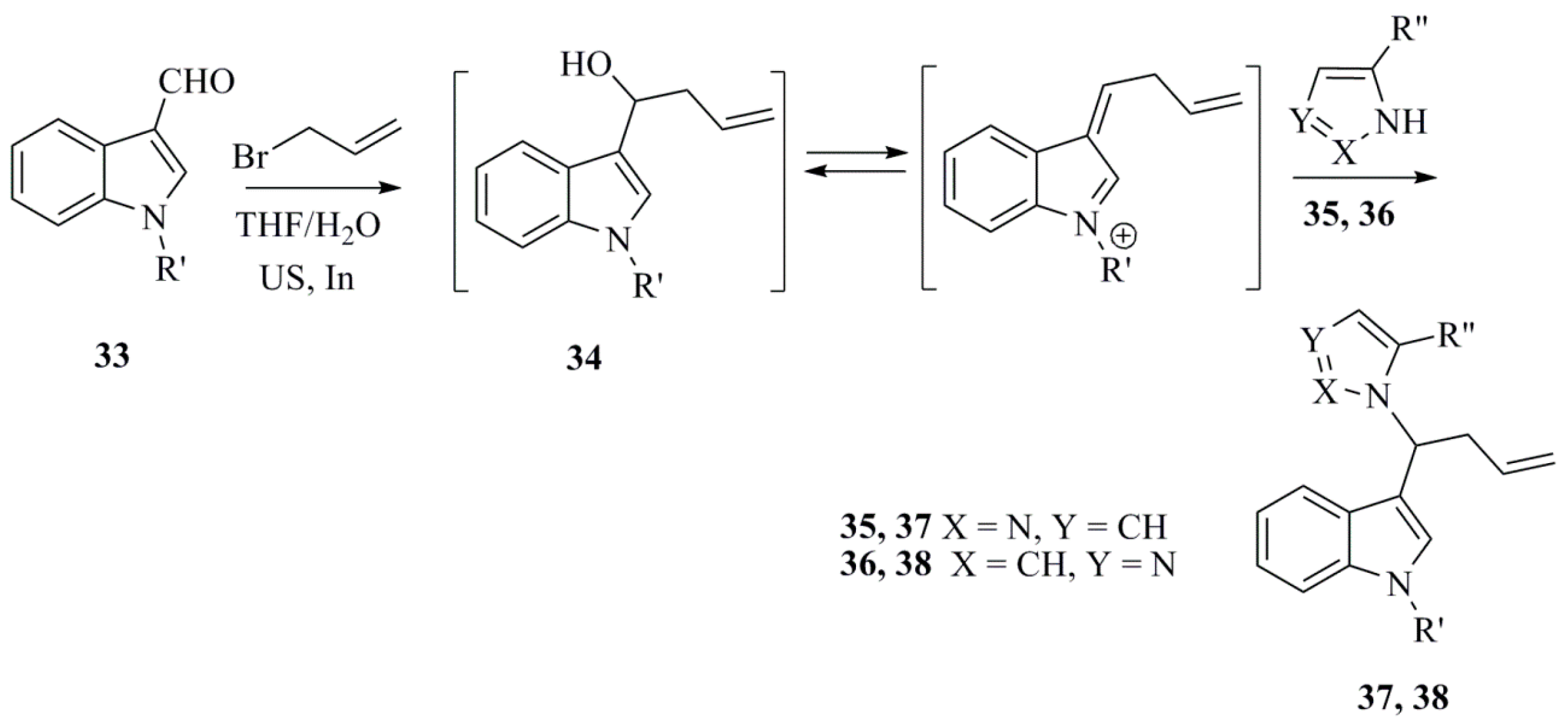
- Cravotto, G.; Giovenzana, G.B.; Maspero, A.; Pilati, T.; Penoni, A.; Palmisano, G. Allylindation of 1H-indole-3-carboxaldehyde in the presence of azoles–revisited. Tetrahedron Lett. 2006, 47, 6439–6443. [Google Scholar] [CrossRef]
- Cravotto, G.; Giovenzana, G.B.; Palmisano, G.; Penoni, A.; Pilati, T.; Sisti, M.; Stazi, F. Convolutamydine A: The first authenticated absolute configuration and enantioselective synthesis. Tetrahedron Asymmetry 2006, 17, 3070–3074. [Google Scholar] [CrossRef]
- Colombo, F.; Cravotto, G.; Palmisano, G.; Penoni, A.; Sisti, M. 3-Component indium-mediated domino allylation of 1H-indole-3-carboxaldehyde with electron-rich (het)arenes: Highly efficient access to diversely functionalized indolylbutenes. Eur. J. Org. Chem. 2008, 2008, 2801–2807. [Google Scholar] [CrossRef]
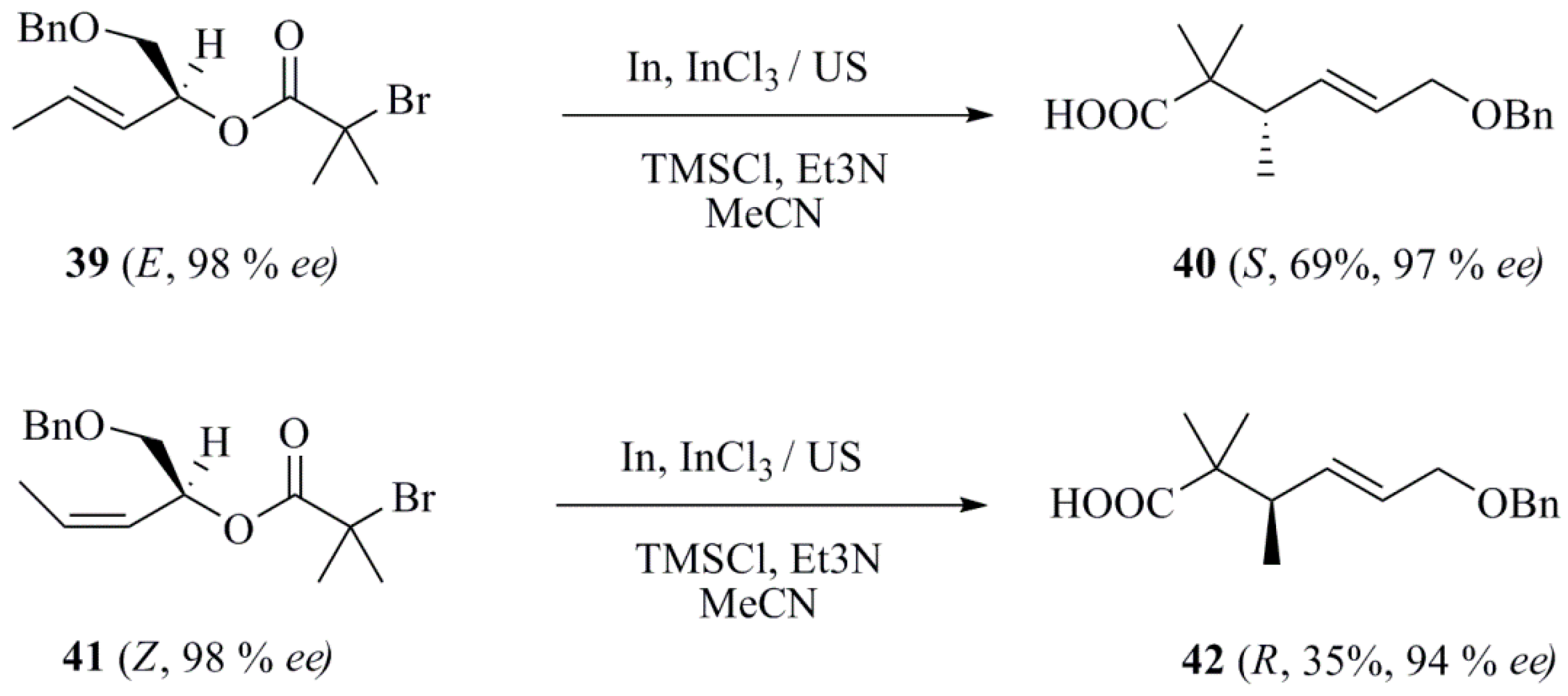
- Soengas, R.G.; Segade, Y.; Jiménez, C.; Rodríguez, J. Highly diastereoselective indium-mediated synthesis of β-lactam carbohydrates from imines. Tetrahedron 2011, 67, 2617–2622. [Google Scholar] [CrossRef]
- Soengas, R.G. A straightforward route to novel α,α-disubstituted tetrahydrofuran β-amino acids and spirodiketopiperazines from sugar lactones. Synlett 2010, 2010, 2549–2552. [Google Scholar] [CrossRef]
- Soengas, R.G.; Estévez, A.M. Indium-mediated allylation and Reformatsky reaction on glyoxylic oximes under ultrasound irradiation. Ultrason. Sonochem. 2012, 19, 916–920. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, J.; Watanabe, Y.; Koyama, N.; Nishino, Y.; Takahashi, K.; Hatakeyama, S. Tetrahedron 2011, 67, 3659–3667.
- Cooper, B.F.T.; MacDonald, C.L.B. Alternative syntheses of univalent indium salts including a direct route from indium metal. New J. Chem. 2010, 34, 1551–1555. [Google Scholar] [CrossRef]
- Downs, A.J.; Himmel, H.-J. New light on the chemistry of the group 13 metals. In The Group 13 Metals Aluminium, Gallium, Indium and Thallium: Chemical Patterns and Peculiarities, 1st ed.; Aldridge, S., Downs, A.J., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2011; p. 1. [Google Scholar]
- Araki, S.; Ito, H.; Butsugan, Y. Gallium-induced Barbier coupling of carbonyl compounds with allyl iodide. Appl. Organomet. Chem. 1988, 2, 475–478. [Google Scholar] [CrossRef]
- Goswami, D.; Chattopadhyay, A.; Sharma, A.; Chattopadhyay, S. [Bmim][Br] as a solvent and activator for the Ga-mediated Barbier allylation: Direct formation of an N-heterocyclic carbene from Ga metal. J. Org. Chem. 2012, 77, 11064–11070. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Schneider, U. Catalytic use of elemental gallium for carbon-carbon bond formation. J. Am. Chem. Soc. 2016, 138, 13119–13122. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.B.; Gedanken, A.; Kimmer, G.; Porat, Z. Ultrasonic cavitation of molten gallium: Formation of micro- and nano-spheres. Ultrason. Sonochem. 2014, 21, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.B.; Koltypin, Y.; Gedanken, A.; Porat, Z. Ultrasonic cavitation of molten gallium in water: Entrapment of organic molecules in gallium microspheres. J. Mater. Chem. A 2014, 2, 1309–1317. [Google Scholar] [CrossRef]
- Shirazi, F.S.; Akhbari, K. Sonochemical procedures; the main synthetic methods for synthesis of coinage metal ion supramolecular polymer nanostructures. Ultrason. Sonochem. 2016, 31, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, F.; Haghighi, M.; Mahboob, S. CO2-enhanced dehydrogenation of ethane over sonochemically synthesized Cr/clinoptilolite-ZrO2 nanocatalyst: Effects of ultrasound irradiation and ZrO2 loading on catalytic activity and stability. Ultrason. Sonochem. 2016, 33, 150–163. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Wang, J.; Yu, J.; Chen, J.; Chen, J. Catalytic ozonation for the degradation of acetylsalicylic acid in aqueous solution by magnetic CeO2 nanometer catalyst particles. Appl. Catal. B 2014, 144, 686–693. [Google Scholar] [CrossRef]
- Gobouri, A.A. Ultrasound enhanced photocatalytic properties of α-Fe2O3 nanoparticles for degradation of dyes used by textile industry. Res. Chem. Intermed. 2016, 42, 5099–5113. [Google Scholar] [CrossRef]
- Ziylan, A.; Ince, N.H. Catalytic ozonation of ibuprofen with ultrasound and Fe-based catalysts. Catal. Today 2014, 240, 5–8. [Google Scholar] [CrossRef]
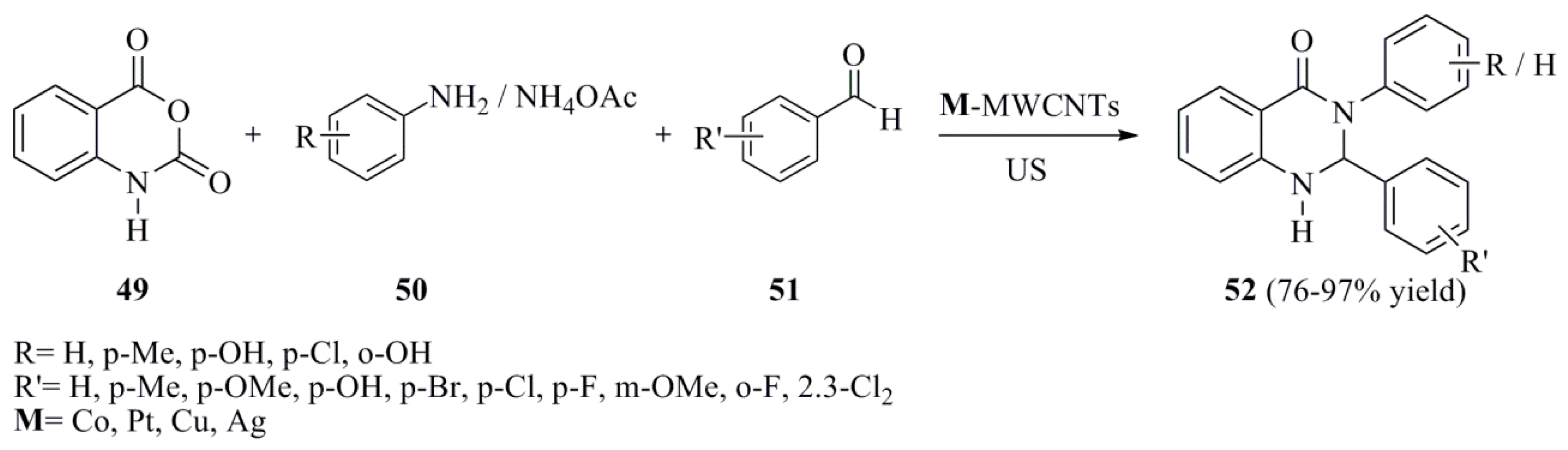
- Safari, J.; Gandomi-Ravandi, S. Application of the ultrasound in the mild synthesis of substituted 2,3-dihydroquinazolin-4(1H)-ones catalyzed by heterogeneous metal-MWCNTs nanocomposites. J. Mol. Struct. 2014, 1072, 173–178. [Google Scholar] [CrossRef]
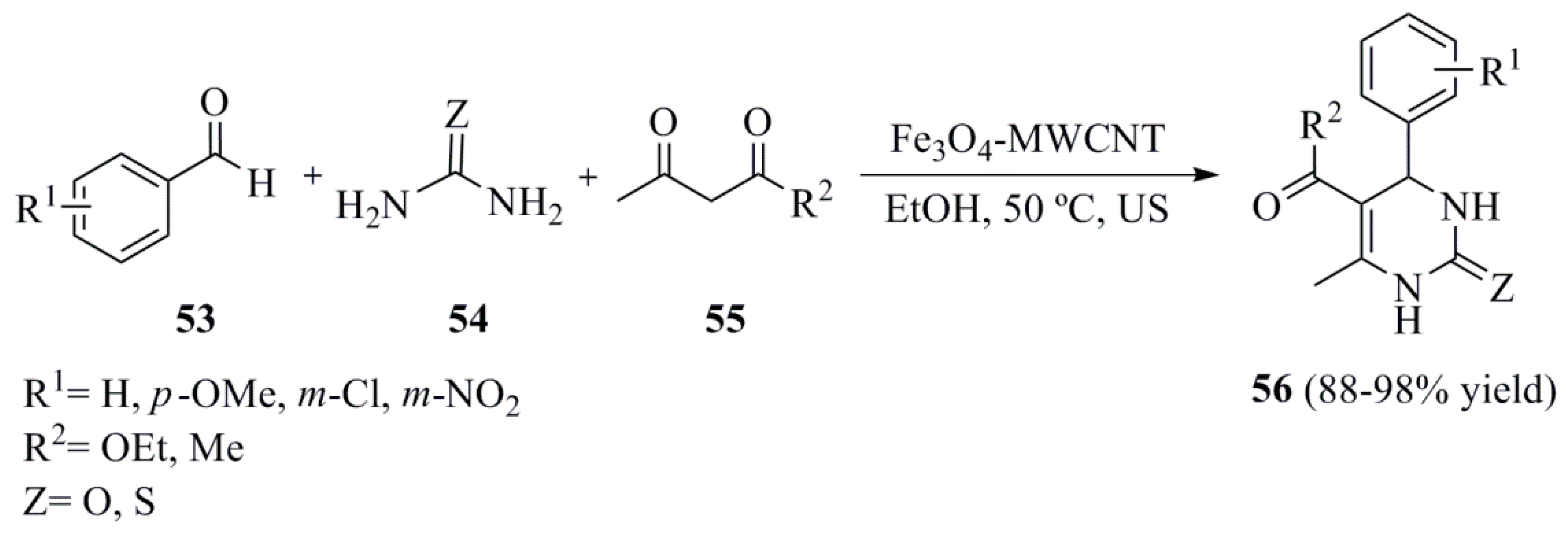
- Safari, J.; Zarnegar, Z. Biginelli reaction on Fe3O4–MWCNT nanocomposite: Excellent reactivity and facile recyclability of the catalyst combined with ultrasound irradiation. RSC Adv. 2013, 3, 17962–17967. [Google Scholar] [CrossRef]
- Jiang, B.; Zhang, C.; Wang, K.; Dou, B.; Song, Y.; Chen, H.; Xu, Y. Highly dispersed Ni/montmorillonite catalyst for glycerol steam reforming: Effect of Ni loading and calcination temperature. Appl. Therm. Eng. 2016, 109, 99–108. [Google Scholar] [CrossRef]
- Behling, R.; Chatel, G.; Valange, S. Sonochemical oxidation of vanillyl alcohol to vanillin in the presence of a cobalt oxide catalyst under mild conditions. Ultrason. Sonochem. 2017, 36, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Sharma, B.; Kim, B.-S.; Park, K.H. Double-hydrophilic block copolymer nanoreactor for the synthesis of copper nanoparticles and for application in click chemistry. J. Nanosci. Nanotechnol. 2011, 11, 6162–6166. [Google Scholar] [CrossRef] [PubMed]
- Stucchi, M.; Bianchi, C.L.; Pirola, C.; Cerrato, G.; Morandi, S.; Argirusis, C.; Sourkounie, G.; Naldonif, A.; Capucci, V. Copper NPs decorated titania: A novel synthesis by high energy US with a study of the photocatalytic activity under visible light. Ultrason. Sonochem. 2016, 31, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Astruc, D. Palladium nanoparticles as efficient green homogeneous and heterogeneous carbon-carbon coupling precatalysts: A unifying approach. Inorg. Chem. 2007, 46, 1884–1894. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zha, Z.; Gan, C.; Pan, C.; Zhou, Y.; Wang, Z.; Zhou, M.-M. Catalysis and regioselectivity of the aqueous Heck reaction by Pd(0) nanoparticles under ultrasonic irradiation. J. Org. Chem. 2006, 71, 4339–4342. [Google Scholar] [CrossRef] [PubMed]
- Gholap, A.R.; Venkatesan, K.; Pasricha, R.; Daniel, T.; Lahoti, R.J.; Srinivasan, K.V. Copper- and ligand-free Sonogashira reaction catalyzed by Pd(0) nanoparticles at ambient conditions under ultrasound irradiation. J. Org. Chem. 2005, 70, 4869–4872. [Google Scholar] [CrossRef] [PubMed]
- Saliman, M.A.; Okawa, H.; Takai, M.; Ono, Y.; Kato, T.; Sugawara, K.; Sato, M. Improved battery performance using Pd nanoparticles synthesized on the surface of LiFePO4/C by ultrasound irradiation. Jpn. J. Appl. Phys. 2016, 55, 07KE05. [Google Scholar] [CrossRef]
- Ziylan-Yavas, A.; Mizukoshi, Y.; Maeda, Y.; Ince, N.H. Supporting of pristine TiO2 with noble metals to enhance the oxidation and mineralization of paracetamol by sonolysis and sonophotolysis. Appl. Catal. B 2015, 172, 7–17. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.-E.; Momma, T.; Osaka, T. Synthesis of Pd-Sn nanoparticles by ultrasonic irradiation and their electrocatalytic activity for oxygen reduction. Electrochim. Acta 2009, 54, 3412–3418. [Google Scholar] [CrossRef]
- Brancewicz, E.; Gradzka, E.; Basa, A.; Winkler, K. Chemical synthesis and characterization of the C60-Pd polymer spherical nanoparticles. Electrochim. Acta 2014, 128, 91–101. [Google Scholar] [CrossRef]
- Tang, S.; Vongehr, S.; Zheng, Z.; Ren, H.; Meng, X. Facile and rapid synthesis of spherical porous palladium nanostructures with high catalytic activity for formic acid electro-oxidation. Nanotechnology 2012, 23, 255606. [Google Scholar] [CrossRef] [PubMed]
- Estifaee, P.; Haghighi, M.; Mohammadi, N.; Rahmani, F. CO oxidation over sonochemically synthesized Pd–Cu/Al2O3 nanocatalyst used in hydrogen purification: Effect of Pd loading and ultrasound irradiation time. Ultrason. Sonochem. 2014, 21, 1155–1165. [Google Scholar] [CrossRef] [PubMed]
- Colmenares, J.C.; Magdziarz, A.; Łomot, D.; Chernyayeva, O.; Lisovytskiy, D. A new photocatalytic tool in VOCs abatement: Effective synergetic combination of sonication and light for the synthesis of monometallic palladium-containing TiO2. Appl. Catal. B 2014, 147, 624–632. [Google Scholar] [CrossRef]
- Colmenares, J.C.; Magdziarz, A. Method of Depositing Metal Nanoparticles on the Surface of Semiconductor Materials and Surface Obtained by this Process. Patent No. P.401693, 20 November 2012. [Google Scholar]
- Colmenares, J.C.; Lisowski, P.; Łomot, D.; Chernyayeva, O.; Lisovytskiy, D. Sonophotodeposition of bimetallic photocatalysts Pd–Au/TiO2: Application to selective oxidation of methanol to methyl formate. ChemSusChem 2015, 8, 1676–1685. [Google Scholar] [CrossRef] [PubMed]
- Basavegowda, N.; Mishra, K.; Lee, Y.R. Ultrasonic-assisted green synthesis of palladium nanoparticles and their nanocatalytic application in multicomponent reaction. New J. Chem. 2015, 39, 972–977. [Google Scholar] [CrossRef]
- Colmenares, J.C.; Aramendía, M.A.; Marinas, A.; Marinas, J.M.; Urbano, F.J. Synthesis, characterization and photocatalytic activity of different metal-doped titania systems. Appl. Catal. A Gen. 2006, 306, 120–127. [Google Scholar] [CrossRef]
- Aramendía, M.A.; Colmenares, J.C.; Marinas, A.; Marinas, J.M.; Moreno, J.M.; Navío, J.A.; Urbano, F.J. Effect of the redox treatment of Pt/TiO2 system on its photocatalytic behaviour in the gas phase selective photooxidation of propan-2-ol. Catal. Today 2007, 128, 235–244. [Google Scholar] [CrossRef]
- Colmenares, J.C.; Aramendia, M.A.; Marinas, A.; Marinas, J.M.; Urbano, F.J. Titania nano-photocatalysts synthesized by ultrasound and microwave methodologies: Application in depuration of water from 3-chloropyridine. J. Mol. Catal. A Chem. 2010, 331, 58–63. [Google Scholar] [CrossRef]
- López-Tenllado, F.J.; Marinas, A.; Urbano, F.J.; Colmenares, J.C.; Hidalgo, M.C.; Marinas, J.M.; Moreno, J.M. Selective photooxidation of alcohols as test reaction for photocatalytic activity. Appl. Catal. B 2012, 128, 150–158. [Google Scholar] [CrossRef]
- Vinodgopal, K.; Neppolian, B.; Salleh, N.; Lightcap, I.V.; Grieser, F.; Ashokkumar, M.; Ding, T.T.; Kamat, P.V. Dual-frequency ultrasound for designing two dimensional catalyst surface: Reduced graphene oxide-Pt composite. Coll. Surf. A 2012, 409, 81–87. [Google Scholar] [CrossRef]
- Cravotto, G.; Cintas, P. Sonication-assisted fabrication and post-synthetic modifications of graphene-like materials. Chem. Eur. J. 2010, 16, 5246–5259. [Google Scholar] [CrossRef] [PubMed]
- Ciesielski, A.; Samori, P. Graphene via sonication assisted liquid-phase exfoliation. Chem. Soc. Rev. 2014, 43, 381–398. [Google Scholar] [CrossRef] [PubMed]
- Toukoniitty, B.; Toukoniitty, E.; Mäki-Arvela, P.; Mikkola, J.-P.; Salmi, T.; Murzin, D.Y.; Kooyman, P.J. Effect of ultrasound in enantioselective hydrogenation of 1-phenyl-1,2-propanedione: Comparison of catalyst activation, solvents and supports. Ultrason. Sonochem. 2006, 13, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Perkas, N.; Rotter, H.; Vradman, L.; Landau, M.V.; Gedanken, A. Sonochemically prepared Pt/CeO2 and its application as a catalyst in ethyl acetate combustion. Langmuir 2006, 22, 7072–7077. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, V.; Alagar, M. Ethylene glycol oxidation on Pt and Pt-Ru nanoparticle decorated polythiophene/multiwalled carbon nanotube composites for fuel cell applications. Nanotechnology 2008, 19, 045504. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.-H.; Kim, J.; Lee, Y.-H.; Kim, I.Y.; Park, M.-H.; Yang, C.-W.; Hwang, S.-J.; Kwon, Y.-U. One-pot synthesis of core-shell-like Pt3Co nanoparticle electrocatalyst with Pt-enriched surface for oxygen reduction reaction in fuel cells. Energy Environ. Sci. 2011, 4, 4947–4953. [Google Scholar] [CrossRef]
- Jiang, R.; Rong, C.; Chu, D. Surface coverage of Pt atoms on PtCo nanoparticles and catalytic kinetics for oxygen reduction. Electrochim. Acta 2011, 56, 2532–2540. [Google Scholar] [CrossRef]
- Nagao, D.; Shimazaki, Y.; Saeki, S.; Kobayashi, Y.; Konno, M. Effect of ultrasonic irradiation on carbon-supported Pt-Ru nanoparticles prepared at high metal concentration. Coll. Surf. A 2007, 302, 623–627. [Google Scholar] [CrossRef]
- Ruiz-Camacho, B.; Álvarez, O.M.; Rodríguez-Santoyo, H.H.; López-Peréz, P.A.; Fuentes-Ramírez, R. Mono and bi-metallic electrocatalysts of Pt and Ag for oxygen reduction reaction synthesized by sonication. Electrochem. Commun. 2015, 61, 5–9. [Google Scholar] [CrossRef]
- Cinelli, M.; Coles, S.R.; Nadagouda, M.N.; Blaszczyński, J.; Slowiński, R.; Varma, R.S.; Kirwan, K. A green chemistry-based classification model for the synthesis of silver nanoparticles. Green Chem. 2015, 17, 2825–2839. [Google Scholar] [CrossRef]
- Chernousova, S.; Epple, M. Silver as antibacterial agent: Ion, nanoparticle, and metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653. [Google Scholar] [CrossRef] [PubMed]
- Manjamadha, V.P.; Muthukumar, K. Ultrasound assisted green synthesis of silver nanoparticles using weed plant. Bioprocess Biosyst. Eng. 2016, 39, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Li, J.; Wang, C.; Li, S.; Lai, Y.; Chen, H.; Lin, C. Ultrasound aided photochemical synthesis of Ag loaded TiO2 nanotube arrays to enhance photocatalytic activity. J. Hazard. Mater. 2009, 171, 1045–1050. [Google Scholar] [CrossRef] [PubMed]
- Jhuang, Y.Y.; Cheng, W.T. Fabrication and characterization of silver/titanium dioxide composite nanoparticles in ethylene glycol with alkaline solution through sonochemical process. Ultrason. Sonochem. 2016, 28, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Pech-Pech, I.E.; Gervasio, D.F.; Godínez-Garcia, A.; Solorza-Feria, O.; Pérez-Robles, J.F. Nanoparticles of Ag with a Pt and Pd rich surface supported on carbon as a new catalyst for the oxygen electroreduction reaction (ORR) in acid electrolytes: Part 1. J. Power Sources 2015, 276, 365–373. [Google Scholar] [CrossRef]
- Pech-Pech, I.E.; Gervasio, D.F.; Pérez-Robles, J.F. Nanoparticles of Ag with a Pt and Pd rich surface supported on carbon as a new catalyst for the oxygen electroreduction reaction (ORR) in acid electrolytes: Part 2. J. Power Sources 2015, 276, 374–381. [Google Scholar] [CrossRef]
- Vu, T.A.; Dao, C.D.; Hoang, T.T.T.; Dang, P.T.; Tran, H.T.K.; Nguyen, K.T.; Le, G.H.; Nguyen, T.V.; Lee, G.D. Synthesis of novel silver vanadates with high photocatalytic and antibacterial activities. Mater. Lett. 2014, 123, 176–180. [Google Scholar] [CrossRef]
- Lei, Z.; Zhang, L.; Wei, X. One-step synthesis of silver nanoparticles by sonication or heating using amphiphilic block copolymer as templates. J. Coll. Interface Sci. 2008, 324, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.K.; Roy, P.; Saini, P.; Mondal, M.K.; Chowdhury, P.; Babu, S.P.S. Carbohydrate polymer inspired silver nanoparticles for filaricidal and mosquitocidal activities: A comprehensive view. Carbohydr. Polym. 2016, 137, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Kuo, T.-C.; Hsu, T.-C.; Liu, Y.-C.; Yang, K.-H. Size-controllable synthesis of surface-enhanced Raman scattering-active gold nanoparticles coated on TiO2. Analyst 2012, 137, 3847–3853. [Google Scholar] [CrossRef] [PubMed]
- Wen, B.; Ma, J.; Chen, C.; Ma, W.; Zhu, H.; Zhao, J. Supported noble metal nanoparticles as photo/sono-catalysts for synthesis of chemicals and degradation of pollutants. Sci. China Chem. 2011, 54, 887–897. [Google Scholar] [CrossRef]
- George, P.P.; Gedanken, A.; Perkas, N.; Zhong, Z. Selective oxidation of CO in the presence of air over gold-based catalysts Au/TiO2/C (sonochemistry) and Au/TiO2/C (microwave). Ultrason. Sonochem. 2008, 15, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Mizukoshi, Y.; Makise, Y.; Shuto, T.; Hu, J.; Tominaga, A.; Shironita, S.; Tanabe, S. Immobilization of noble metal nanoparticles on the surface of TiO2 by the sonochemical method: Photocatalytic production of hydrogen from an aqueous solution of ethanol. Ultrason. Sonochem. 2007, 14, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Kan, C.; Zhu, J.; Wang, C. Ag nanoparticle-filled polymer shell formed around Au nanoparticle core via ultrasound-assisted spherulite growth. J. Cryst. Growth 2009, 311, 1565–1570. [Google Scholar] [CrossRef]
- Park, J.-E.; Atobe, M.; Fuchigami, T. Sonochemical synthesis of conducting polymer-metal nanoparticles nanocomposite. Electrochim. Acta 2005, 51, 849–854. [Google Scholar] [CrossRef]
- Fang, Y.; Li, X.; Hu, Y.; Li, F.; Lin, X.; Tian, M.; Anc, X.; Fud, Y.; Jina, J.; Ma, J. Ultrasonication-assisted ultrafast preparation of multiwalled carbon nanotubes/Au/Co3O4 tubular hybrids as superior anode materials for oxygen evolution reaction. J. Power Sources 2015, 300, 285–293. [Google Scholar] [CrossRef]
- Yusof, N.S.M.; Ashokkumar, M. Sonochemical synthesis of gold nanoparticles by using high intensity focused ultrasound. ChemPhysChem 2015, 16, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Vilela, S.M.F.; Ananias, D.; Fernandes, J.A.; Silva, P.; Gomes, A.C.; Silva, N.J.O.; Rodrigues, M.O.; Tomé, J.P.C.; Valente, A.A.; Ribeiro-Claro, P.; et al. Multifunctional micro- and nanosized metal-organic frameworks assembled from bisphosphonates and lanthanides. J. Mater. Chem. C 2014, 2, 3311–3327. [Google Scholar] [CrossRef]
- Amith Yadav, H.J.; Eraiah, B.; Nagabhushana, H.; Darshan, G.P.; Daruka Prasad, B.; Sharma, S.C.; Premkumar, H.B.; Anantharaju, K.S.; Vijayakumar, G.R. Facile ultrasound route to prepare micro/nano superstructures for multifunctional applications. ACS Sust. Chem. Eng. 2017, 5, 2061–2074. [Google Scholar] [CrossRef]
- Gottapu, S.; Padhi, S.K.; Krishna, M.G.; Muralidharan, K. Poly(vinylpyrrolidone) stabilized aluminum nanoparticles obtained by the reaction of SiCl4 with LiAlH4. New J. Chem. 2015, 39, 5203–5207. [Google Scholar] [CrossRef]
- Kumar, V.B.; Mastai, Y.; Porat, Z.; Gedanken, A. Chiral imprinting in molten gallium. New J. Chem. 2015, 39, 2690–2696. [Google Scholar] [CrossRef]
- Nissan, I.; Kumar, V.B.; Porat, Z.; Makovec, D.; Shefi, O.; Gedanken, A. Sonochemically-fabricated Ga@C-dots@Ga nanoparticle-aided neural growth. J. Mater. Chem. B 2017, 5, 1371–1379. [Google Scholar] [CrossRef]
- Safari, J.; Gandomi-Ravandi, S.; Akbari, Z. Sonochemical synthesis of 1,2,4,5-tetrasubstituted imidazoles using nanocrystalline MgAl2O4 as an effective catalyst. J. Adv. Res. 2013, 4, 509–514. [Google Scholar] [CrossRef] [PubMed]

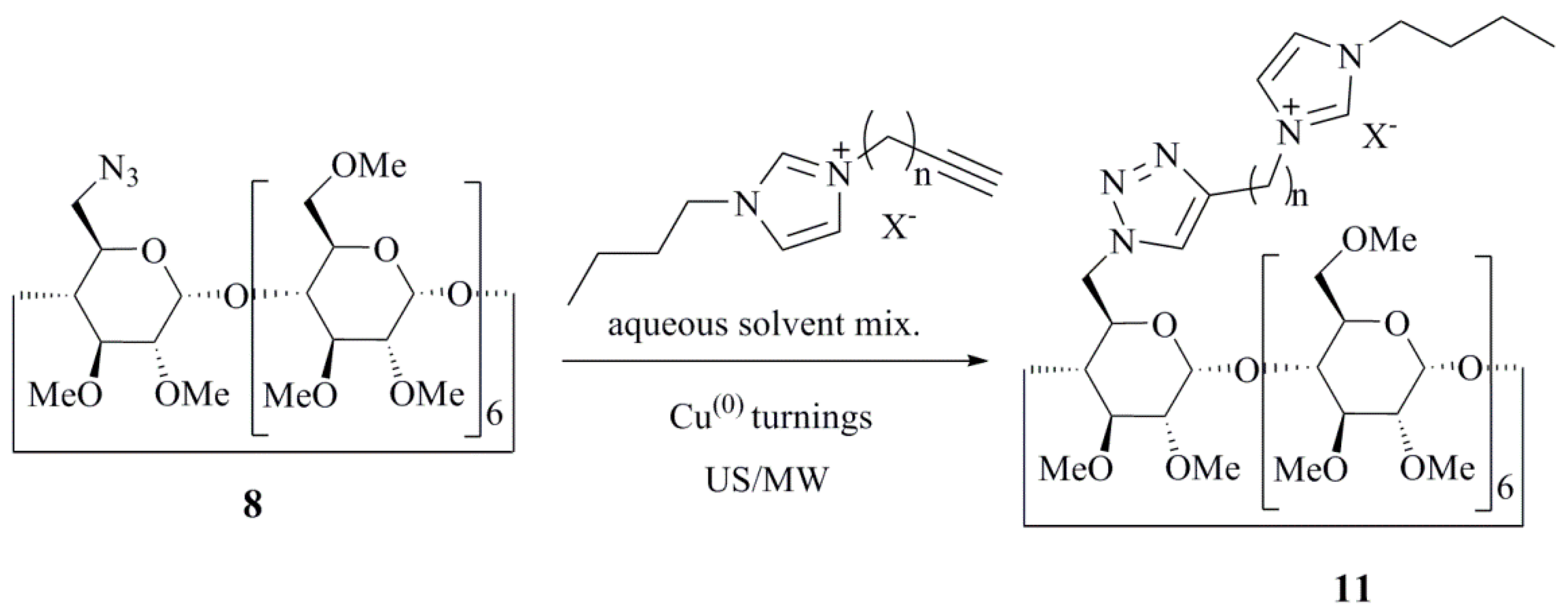
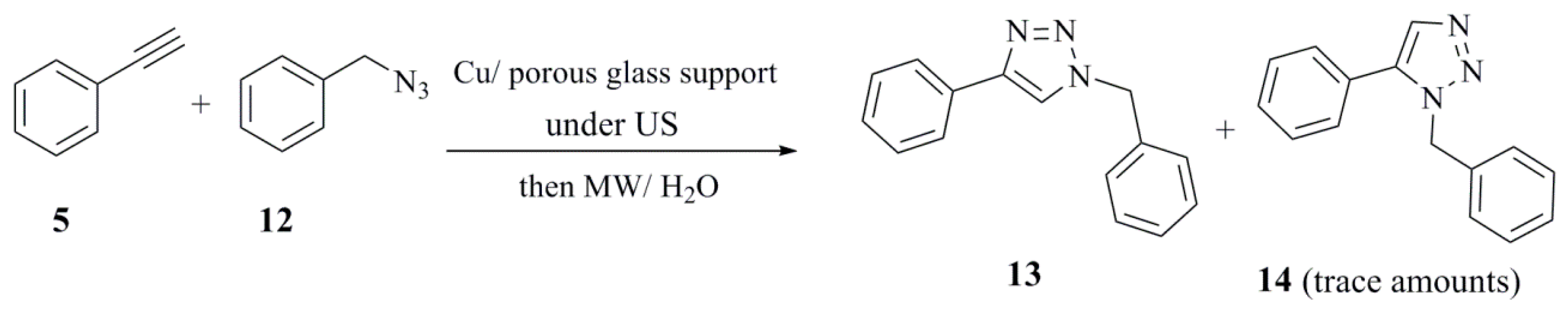
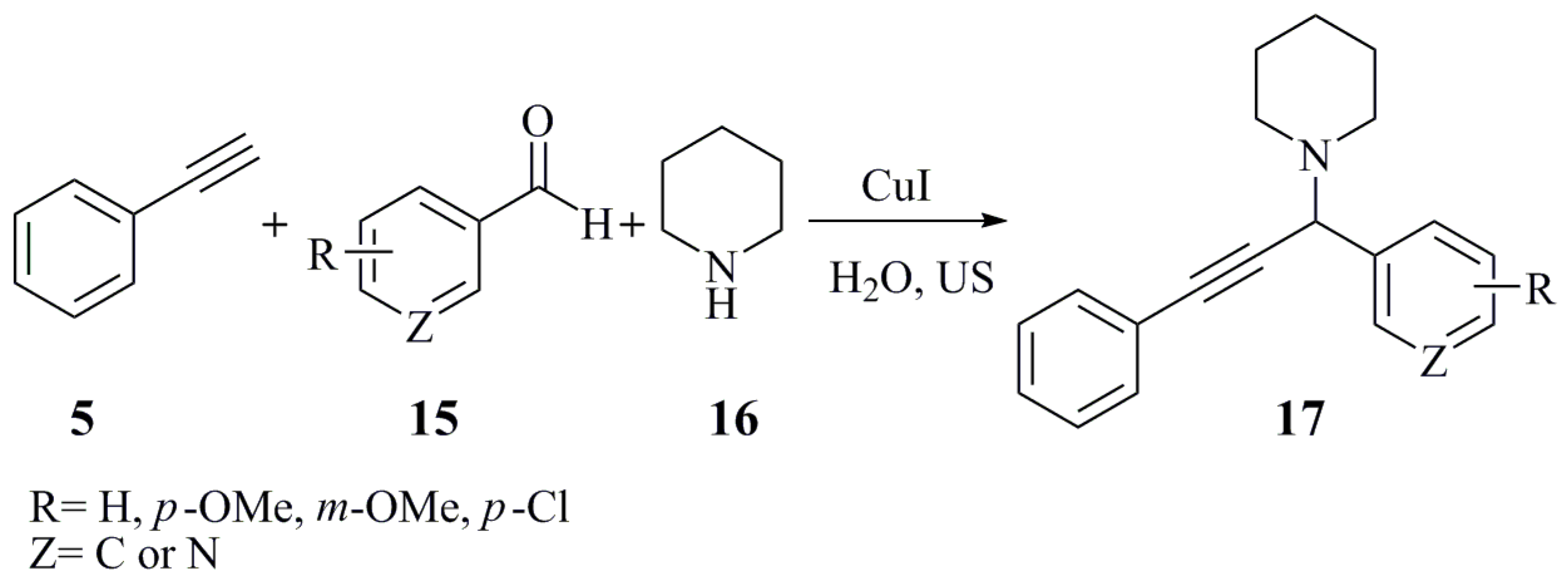

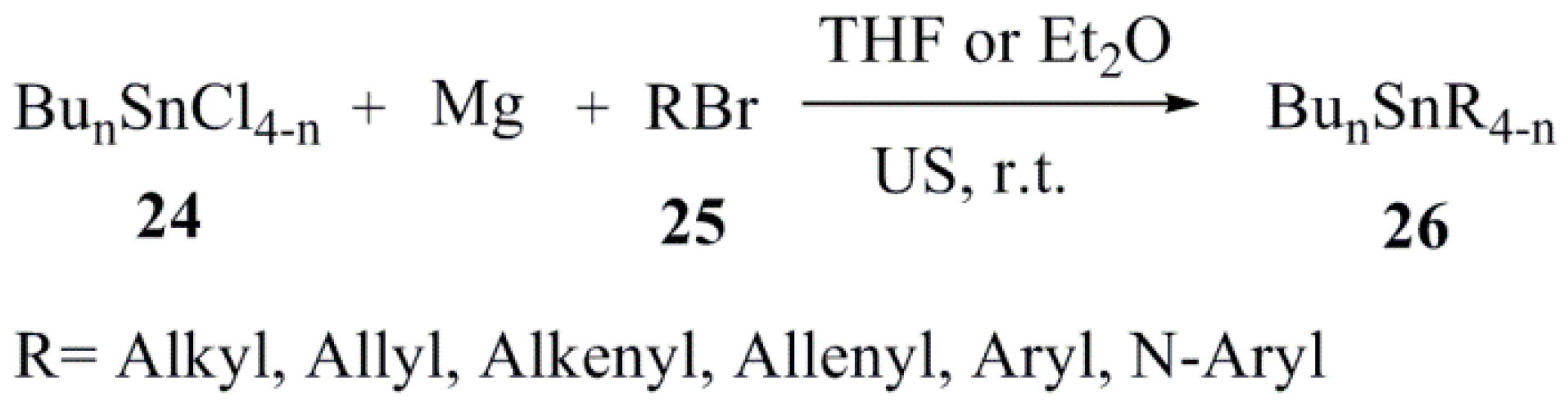


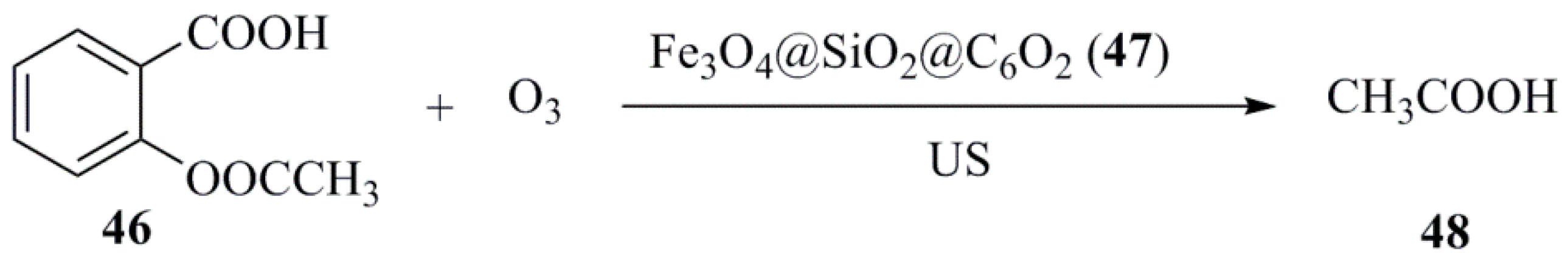

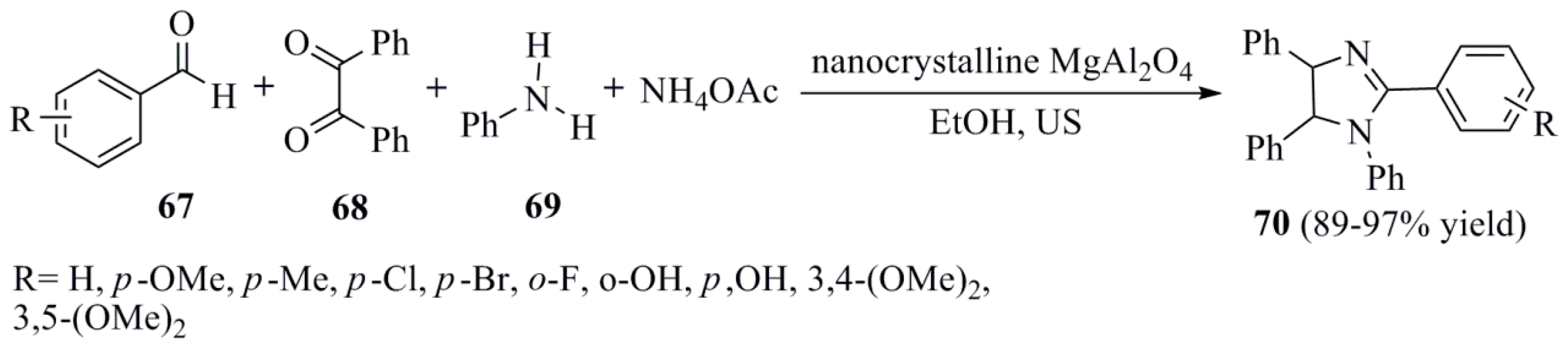
| Catalyst (10 mol %) | Conditions (at 85 °C) | Reaction Time | Yield (%) |
|---|---|---|---|
| Cu(II)/C, l-ascorbic acid (a) | oil bath | 2 h | 79 |
| Cu(II)/C, l-ascorbic acid | MW | 10 min | 83 |
| recycled Cu(II)/C (b) | MW | 30 min | 84 |
| Cu(II)/C | oil bath | 3 h | 30 |
| Cu(II)/C | MW | 45 min | 81 |
| Cu(I)/C | oil bath | 2 h | 78 |
| Cu(I)/C | MW | 10 min | 89 |
| Cu(I)/C | MW/US | 10 min | 93 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domini, C.E.; Álvarez, M.B.; Silbestri, G.F.; Cravotto, G.; Cintas, P. Merging Metallic Catalysts and Sonication: A Periodic Table Overview. Catalysts 2017, 7, 121. https://doi.org/10.3390/catal7040121
Domini CE, Álvarez MB, Silbestri GF, Cravotto G, Cintas P. Merging Metallic Catalysts and Sonication: A Periodic Table Overview. Catalysts. 2017; 7(4):121. https://doi.org/10.3390/catal7040121
Chicago/Turabian StyleDomini, Claudia E., Mónica B. Álvarez, Gustavo F. Silbestri, Giancarlo Cravotto, and Pedro Cintas. 2017. "Merging Metallic Catalysts and Sonication: A Periodic Table Overview" Catalysts 7, no. 4: 121. https://doi.org/10.3390/catal7040121
APA StyleDomini, C. E., Álvarez, M. B., Silbestri, G. F., Cravotto, G., & Cintas, P. (2017). Merging Metallic Catalysts and Sonication: A Periodic Table Overview. Catalysts, 7(4), 121. https://doi.org/10.3390/catal7040121