SO42−/Sn-MMT Solid Acid Catalyst for Xylose and Xylan Conversion into Furfural in the Biphasic System
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Catalyst
2.2. Furfural Production from Xylose and Xylan
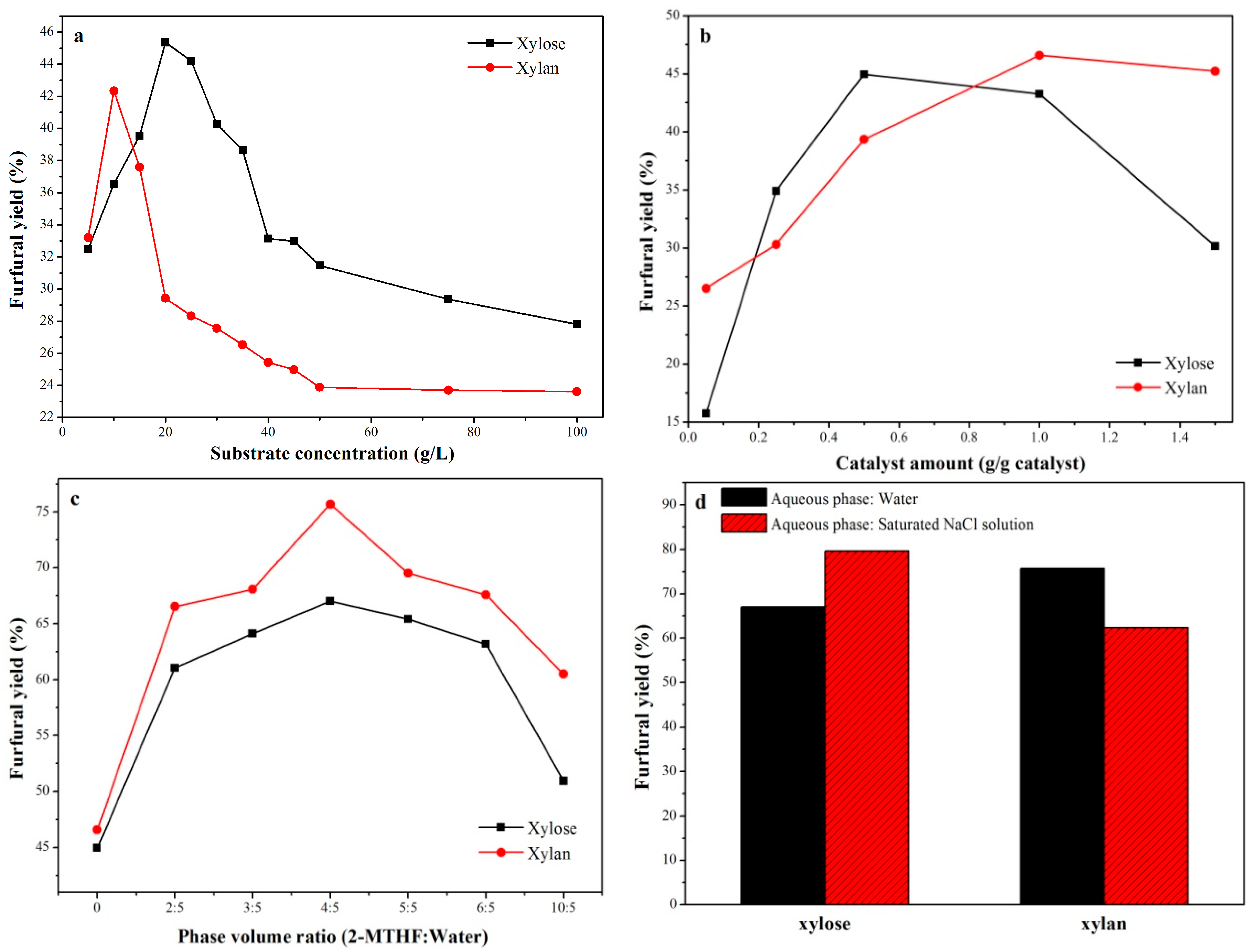
2.2.1. Effect of Substrate Concentration
2.2.2. Effect of the Catalyst Amount
2.2.3. Effect of the 2-MTHF/Water Volume Ratio
2.2.4. Effect of Saturated NaCl Solution in the Aqueous Phase
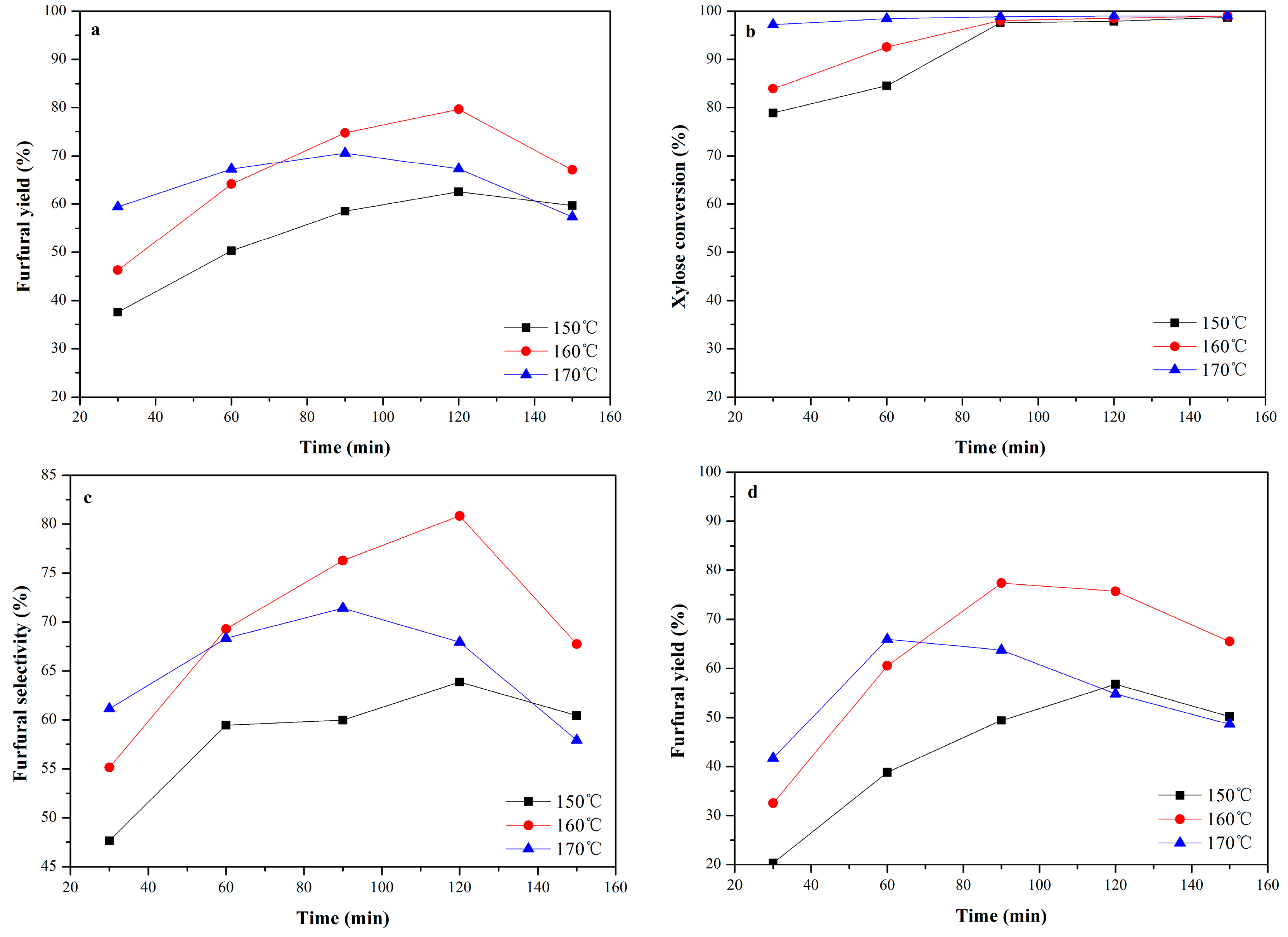
2.2.5. Effect of Reaction Temperature and Time
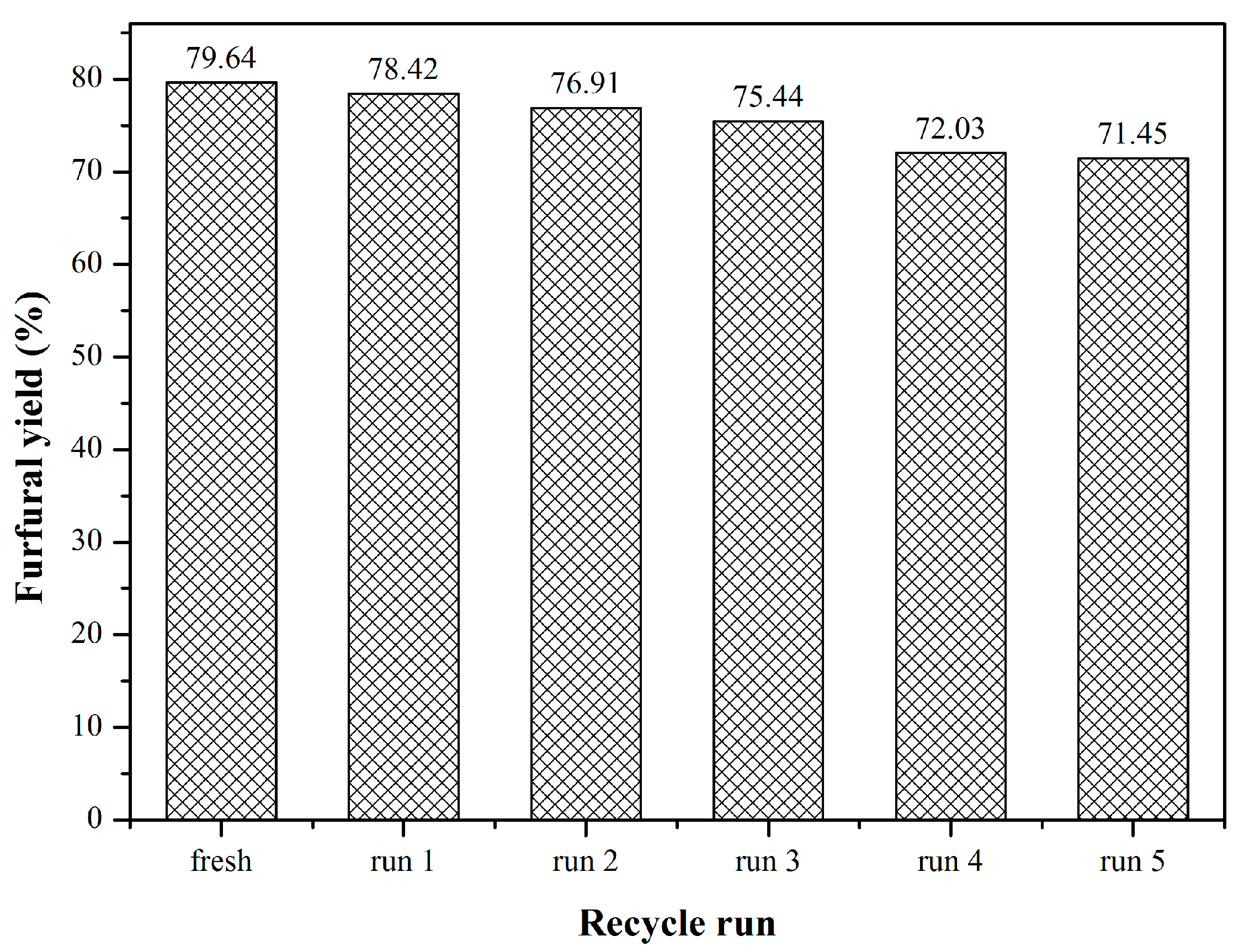
2.3. Catalyst Recyclability
3. Materials and Methods
3.1. Materials
3.2. Preparation of the Catalyst
3.3. Characterization of Catalyst
3.4. Procedure for the Transformation of Xylose and Xylan into Furfural
3.5. Product Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mardhiah, H.H.; Ong, H.C.; Masjuki, H.H.; Lim, S.; Lee, H.V. A review on latest developments and future prospects of heterogeneous catalyst in biodiesel production from non-edible oils. Renew. Sustain. Energy Rev. 2017, 67, 1225–1236. [Google Scholar] [CrossRef]
- Guldhe, A.; Singh, P.; Ansari, F.A.; Singh, B.; Bux, F. Biodiesel synthesis from microalgal lipids using tungstated zirconia as a heterogeneous acid catalyst and its comparison with homogeneous acid and enzyme catalysts. Fuel 2017, 187, 180–188. [Google Scholar] [CrossRef]
- Liu, S.; Amada, Y.; Tamura, M.; Nakagawa, Y.; Tomishige, K. One-pot selective conversion of furfural into 1,5-pentanediol over a Pd-added Ir–ReOx/SiO2 bifunctional catalyst. Green Chem. 2014, 16, 617–626. [Google Scholar] [CrossRef]
- Agirrezabal-Telleria, I.; Hemmann, F.; Jäger, C.; Arias, P.L.; Kemnitz, E. Functionalized partially hydroxylated MgF2 as catalysts for the dehydration of d-xylose to furfural. J. Catal. 2013, 305, 81–91. [Google Scholar] [CrossRef]
- Shi, X.; Wu, Y.; Yi, H.; Rui, G.; Li, P.; Yang, M.; Wang, G. Selective preparation of furfural from xylose over sulfonic acid functionalized mesoporous SBA-15 materials. Energies 2011, 4, 669. [Google Scholar] [CrossRef]
- Sádaba, I.; Ojeda, M.; Mariscal, R.; Granados, M.L. Silica-poly(styrenesulphonic acid) nanocomposites for the catalytic dehydration of xylose to furfural. Appl. Catal. B Environ. 2014, 150–151, 421–431. [Google Scholar] [CrossRef]
- Choudhary, V.; Pinar, A.B.; Sandler, S.I.; Vlachos, D.G.; Lobo, R.F. Xylose isomerization to xylulose and its dehydration to furfural in aqueous media. ACS Catal. 2011, 1, 1724–1728. [Google Scholar] [CrossRef]
- Takagaki, A.; Ohara, M.; Nishimura, S.; Ebitani, K. One-pot formation of furfural from xylose via isomerization and successive dehydration reactions over heterogeneous acid and base catalysts. Chem. Lett. 2010, 39, 838–840. [Google Scholar] [CrossRef]
- Wang, W.J.; Ren, J.L.; Li, H.L.; Deng, A.J.; Sun, R.C. Direct transformation of xylan-type hemicelluloses to furfural via SnCl4 catalysts in aqueous and biphasic systems. Bioresour. Technol. 2015, 183, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Izumi, Y.; Onaka, M. Liquid-phase organic reactions catalyzed by inorganic solid acids and bases. J. Mol. Catal. 1992, 74, 35–42. [Google Scholar] [CrossRef]
- Wang, J.; Masui, Y.; Onaka, M. Direct allylation of α-aryl alcohols with allyltrimethylsilane catalyzed by heterogeneous tin ion-exchanged montmorillonite. Tetrahedron Lett. 2010, 51, 3300–3303. [Google Scholar] [CrossRef]
- Li, H.L.; Ren, J.L.; Zhong, L.J.; Sun, R.C.; Liang, L. Production of furfural from xylose, water-insoluble hemicelluloses and water-soluble fraction of corncob via a tin-loaded montmorillonite solid acid catalyst. Bioresour. Technol. 2015, 176, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, X.-D.; Sun, L.; Zhang, J.; Xu, H.-P. Fatty acid methyl ester synthesis catalyzed by solid superacid catalyst SO42−/ZrO2–TiO2/La3+. Appl. Energy 2010, 87, 156–159. [Google Scholar] [CrossRef]
- Lam, E.; Majid, E.; Leung, A.C.; Chong, J.H.; Mahmoud, K.A.; Luong, J.H. Synthesis of furfural from xylose by heterogeneous and reusable nafion catalysts. Chemsuschem 2011, 4, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Mellmer, M.A.; Sener, C.; Gallo, J.M.; Luterbacher, J.S.; Alonso, D.M.; Dumesic, J.A. Solvent effects in acid-catalyzed biomass conversion reactions. Angew. Chem. 2014, 53, 11872–11875. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhuang, J.; Lin, L.; Liu, S.; Zhang, Z. Conversion of d-xylose into furfural with mesoporous molecular sieve MCM-41 as catalyst and butanol as the extraction phase. Biomass Bioenergy 2012, 39, 73–77. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, H.; Wang, P.; Dong, H.; Peng, X. Conversion of xylan, d-xylose and lignocellulosic biomass into furfural using AlCl3 as catalyst in ionic liquid. Bioresour. Technol. 2013, 130, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Li, W.; Xu, Z.; Liu, Q.; Ma, Q.; Jameel, H.; Chang, H.M.; Ma, L. Catalytic conversion of xylose and corn stalk into furfural over carbon solid acid catalyst in gamma-valerolactone. Bioresour. Technol. 2016, 209, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Xin, K.; Song, Y.; Dai, F.; Yu, Y.; Li, Q. Liquid–liquid equilibria for the extraction of furfural from aqueous solution using different solvents. Fluid Phase Equilibr. 2016, 425, 393–401. [Google Scholar] [CrossRef]
- Wang, J.; Masui, Y.; Onaka, M. Synthesis of α-amino nitriles from carbonyl compounds, amines, and trimethylsilyl cyanide: Comparison between catalyst-free conditions and the presence of tin ion-exchanged montmorillonite. Eur. J. Org. Chem. 2010, 2010, 1763–1771. [Google Scholar] [CrossRef]
- Wang, J.; Ren, J.; Liu, X.; Xi, J.; Xia, Q.; Zu, Y.; Lu, G.; Wang, Y. Direct conversion of carbohydrates to 5-hydroxymethylfurfural using Sn-Mont catalyst. Green Chem. 2012, 14, 2506. [Google Scholar] [CrossRef]
- Shinde, S.; Rode, C. Selective self-etherification of 5-(hydroxymethyl)furfural over Sn-Mont catalyst. Catal. Commun. 2017, 88, 77–80. [Google Scholar] [CrossRef]
- Liu, D.; Yuan, P.; Liu, H.; Cai, J.; Tan, D.; He, H.; Zhu, J.; Chen, T. Quantitative characterization of the solid acidity of montmorillonite using combined FTIR and TPD based on the NH3 adsorption system. Appl. Clay Sci. 2013, 80–81, 407–412. [Google Scholar] [CrossRef]
- Liu, D.; Yuan, P.; Liu, H.; Cai, J.; Qin, Z.; Tan, D.; Zhou, Q.; He, H.; Zhu, J. Influence of heating on the solid acidity of montmorillonite: A combined study by DRIFT and Hammett indicators. Appl. Clay Sci. 2011, 52, 358–363. [Google Scholar] [CrossRef]
- Reddy, C.R.; Bhat, Y.S.; Nagendrappa, G.; Jai Prakash, B.S. Brønsted and Lewis acidity of modified montmorillonite clay catalysts determined by FT-IR spectroscopy. Catal. Today 2009, 141, 157–160. [Google Scholar] [CrossRef]
- Azzouz, A.; Nistor, D.; Miron, D.; Ursu, A.V.; Sajin, T.; Monette, F.; Niquette, P.; Hausler, R. Assessment of acid–base strength distribution of ion-exchanged montmorillonites through NH3 and CO2-TPD measurements. Thermochim. Acta 2006, 449, 27–34. [Google Scholar] [CrossRef]
- Chen, W.-H.; Ko, H.-H.; Sakthivel, A.; Huang, S.-J.; Liu, S.-H.; Lo, A.-Y.; Tsai, T.-C.; Liu, S.-B. A solid-state NMR, FT-IR and TPD study on acid properties of sulfated and metal-promoted zirconia: Influence of promoter and sulfation treatment. Catal. Today 2006, 116, 111–120. [Google Scholar] [CrossRef]
- Zhou, Y.; Niu, S.; Li, J. Activity of the carbon-based heterogeneous acid catalyst derived from bamboo in esterification of oleic acid with ethanol. Energy Convers. Manag. 2016, 114, 188–196. [Google Scholar] [CrossRef]
- Li, H.L.; Deng, A.J.; Ren, J.L.; Liu, C.Y.; Wang, W.J.; Peng, F.; Sun, R.C. A modified biphasic system for the dehydration of D-xylose into furfural using SO42−/TiO2-ZrO2/La3+ as a solid catalyst. Catal. Today 2014, 234, 251–256. [Google Scholar] [CrossRef]
- Lou, W.Y.; Zong, M.H.; Duan, Z.Q. Efficient production of biodiesel from high free fatty acid-containing waste oils using various carbohydrate-derived solid acid catalysts. Bioresour. Technol. 2008, 99, 8752–8758. [Google Scholar] [CrossRef] [PubMed]
- Chheda, J.N.; Dumesic, J.A. An overview of dehydration, aldol-condensation and hydrogenation processes for production of liquid alkanes from biomass-derived carbohydrates. Catal. Today 2007, 123, 59–70. [Google Scholar] [CrossRef]
- Deng, A.; Lin, Q.; Yan, Y.; Li, H.; Ren, J.; Liu, C.; Sun, R. A feasible process for furfural production from the pre-hydrolysis liquor of corncob via biochar catalysts in a new biphasic system. Bioresour. Technol. 2016, 216, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Roman-Leshkov, Y.; Barrett, C.J.; Liu, Z.Y.; Dumesic, J.A. Production of dimethylfuran for liquid fuels from biomass-derived carbohydrates. Nature 2007, 447, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Guenic, S.L.; Delbecq, F.; Ceballos, C.; Len, C. Microwave-assisted dehydration of d-xylose into furfural by diluted inexpensive inorganic salts solution in a biphasic system. J. Mol. Catal. A Chem. 2015, 410, 1–7. [Google Scholar] [CrossRef]
- Dias, A.S.; Lima, S.; Carriazo, D.; Rives, V.; Pillinger, M.; Valente, A.A. Exfoliated titanate, niobate and titanoniobate nanosheets as solid acid catalysts for the liquid-phase dehydration of d-xylose into furfural. J. Catal. 2006, 244, 230–237. [Google Scholar] [CrossRef]
- Xing, R.; Qi, W.; Huber, G.W. Production of furfural and carboxylic acids from waste aqueous hemicellulose solutions from the pulp and paper and cellulosic ethanol industries. Energy Environ. Sci. 2011, 4, 2193–2205. [Google Scholar] [CrossRef]
- Zhang, T.; Kumar, R.; Wyman, C.E. Enhanced yields of furfural and other products by simultaneous solvent extraction during thermochemical treatment of cellulosic biomass. RSC Adv. 2013, 3, 9809. [Google Scholar] [CrossRef]
- Binder, J.B.; Blank, J.J.; Cefali, A.V.; Raines, R.T. Synthesis of furfural from xylose and xylan. ChemSusChem 2010, 3, 1268–1272. [Google Scholar] [CrossRef] [PubMed]
- Lessard, J.; Morin, J.-F.; Wehrung, J.-F.; Magnin, D.; Chornet, E. High yield conversion of residual pentoses into furfural via zeolite catalysis and catalytic hydrogenation of furfural to 2-methylfuran. Top. Catal. 2010, 53, 1231–1234. [Google Scholar] [CrossRef]
- Agirrezabal-Telleria, I.; Requies, J.; Güemez, M.B.; Arias, P.L. Dehydration of d-xylose to furfural using selective and hydrothermally stable arenesulfonic SBA-15 catalysts. Appl. Catal. B Environ. 2014, 145, 34–42. [Google Scholar] [CrossRef]
- Bhaumik, P.; Dhepe, P.L. Exceptionally high yields of furfural from assorted raw biomass over solid acids. RSC Adv. 2014, 4, 26215. [Google Scholar] [CrossRef]
- Dias, A.S.; Pillinger, M.; Valente, A.A. Dehydration of xylose into furfural over micro-mesoporous sulfonic acid catalysts. J. Catal. 2005, 229, 414–423. [Google Scholar] [CrossRef]
- Dias, A.S.; Lima, S.; Pillinger, M.; Valente, A.A. Modified versions of sulfated zirconia as catalysts for the conversion of xylose to furfural. Catal. Lett. 2007, 114, 151–160. [Google Scholar] [CrossRef]
- Lima, S.; Pillinger, M.; Valente, A.A. Dehydration of d-xylose into furfural catalysed by solid acids derived from the layered zeolite Nu-6(1). Catal. Commun. 2008, 9, 2144–2148. [Google Scholar] [CrossRef]
- Li, H.; Chen, X.; Ren, J.; Deng, H.; Peng, F.; Sun, R. Functional relationship of furfural yields and the hemicellulose-derived sugars in the hydrolysates from corncob by microwave-assisted hydrothermal pretreatment. Biotechnol. Biofuels 2015, 8, 127. [Google Scholar] [CrossRef] [PubMed]

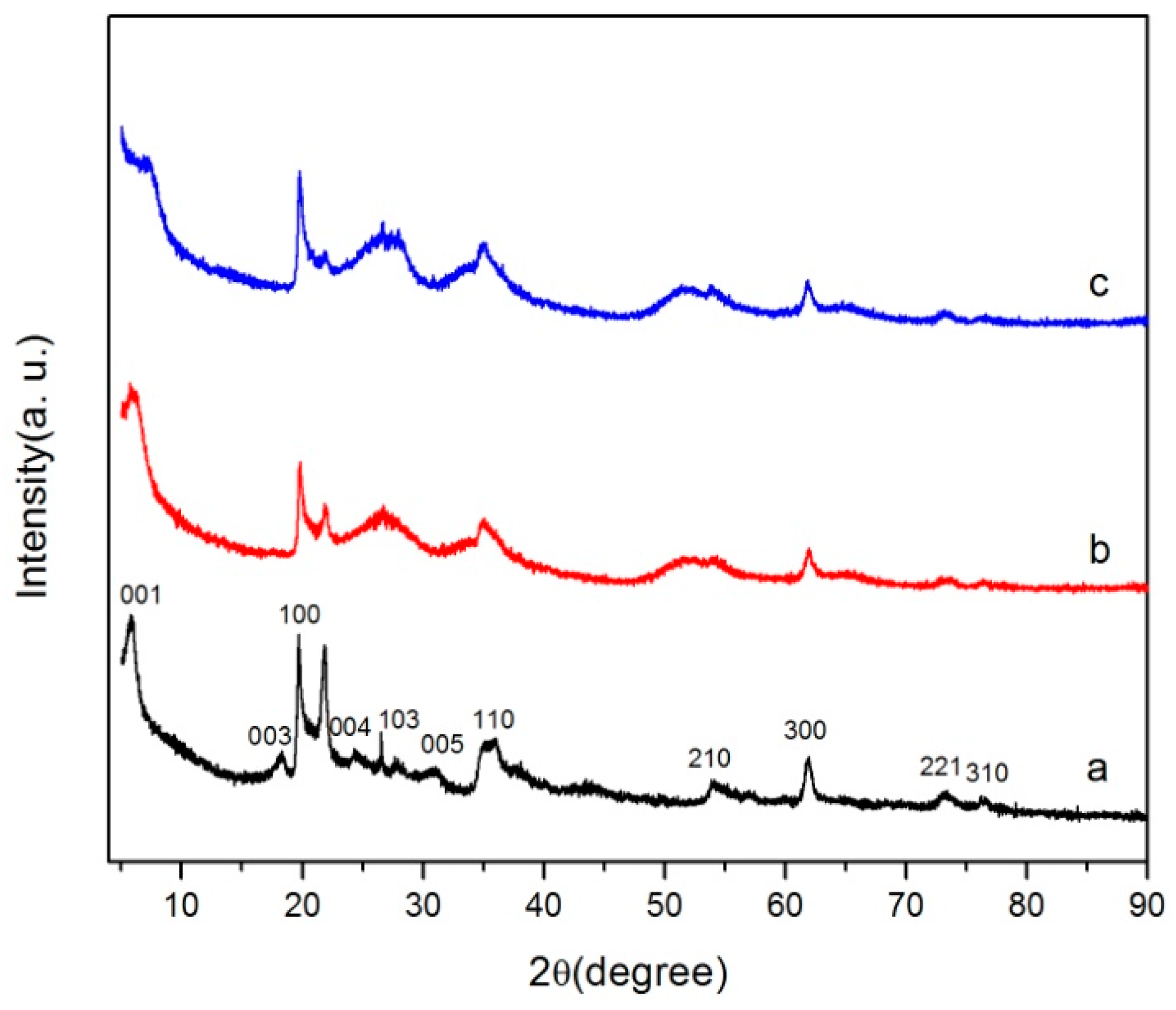
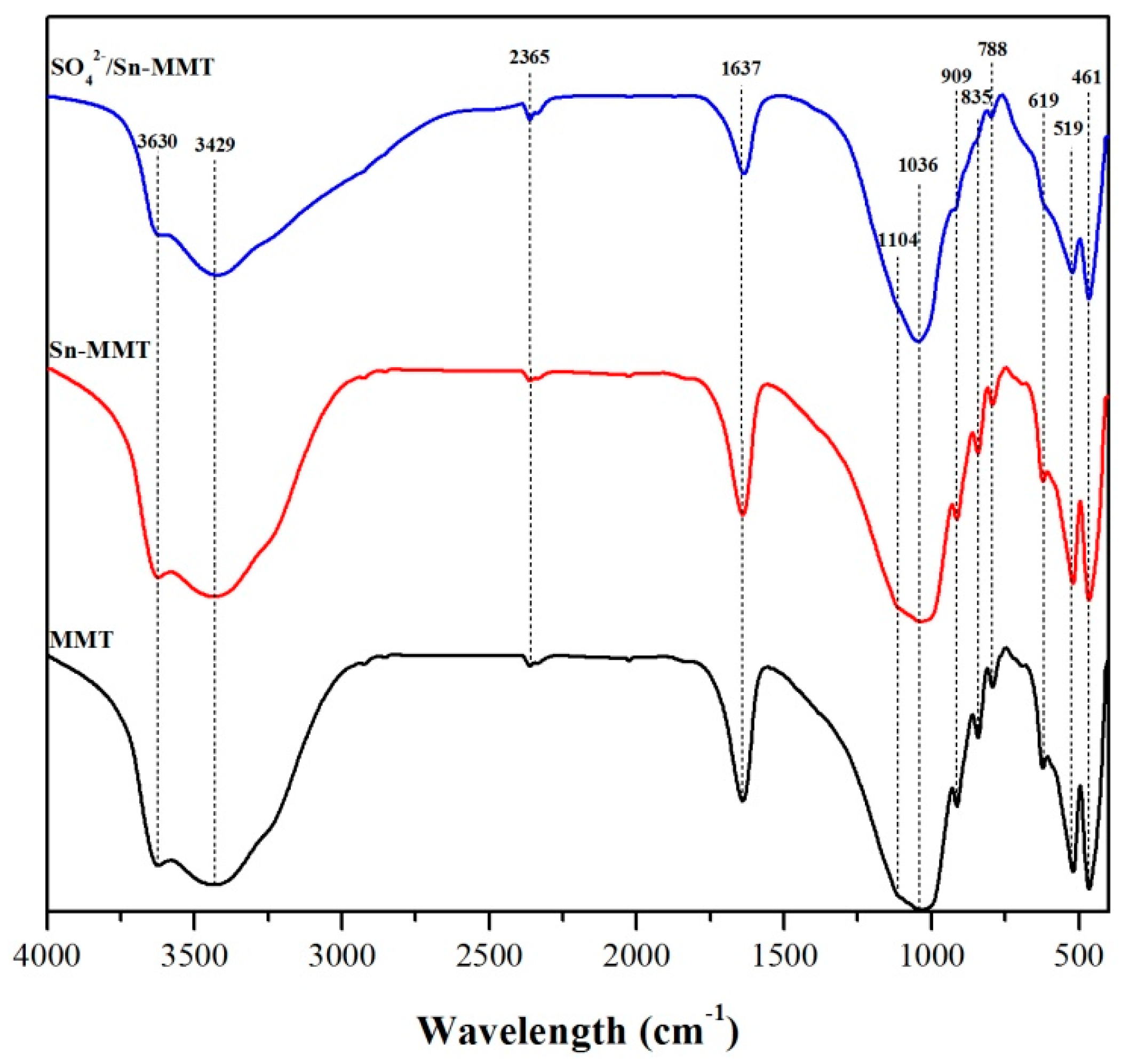
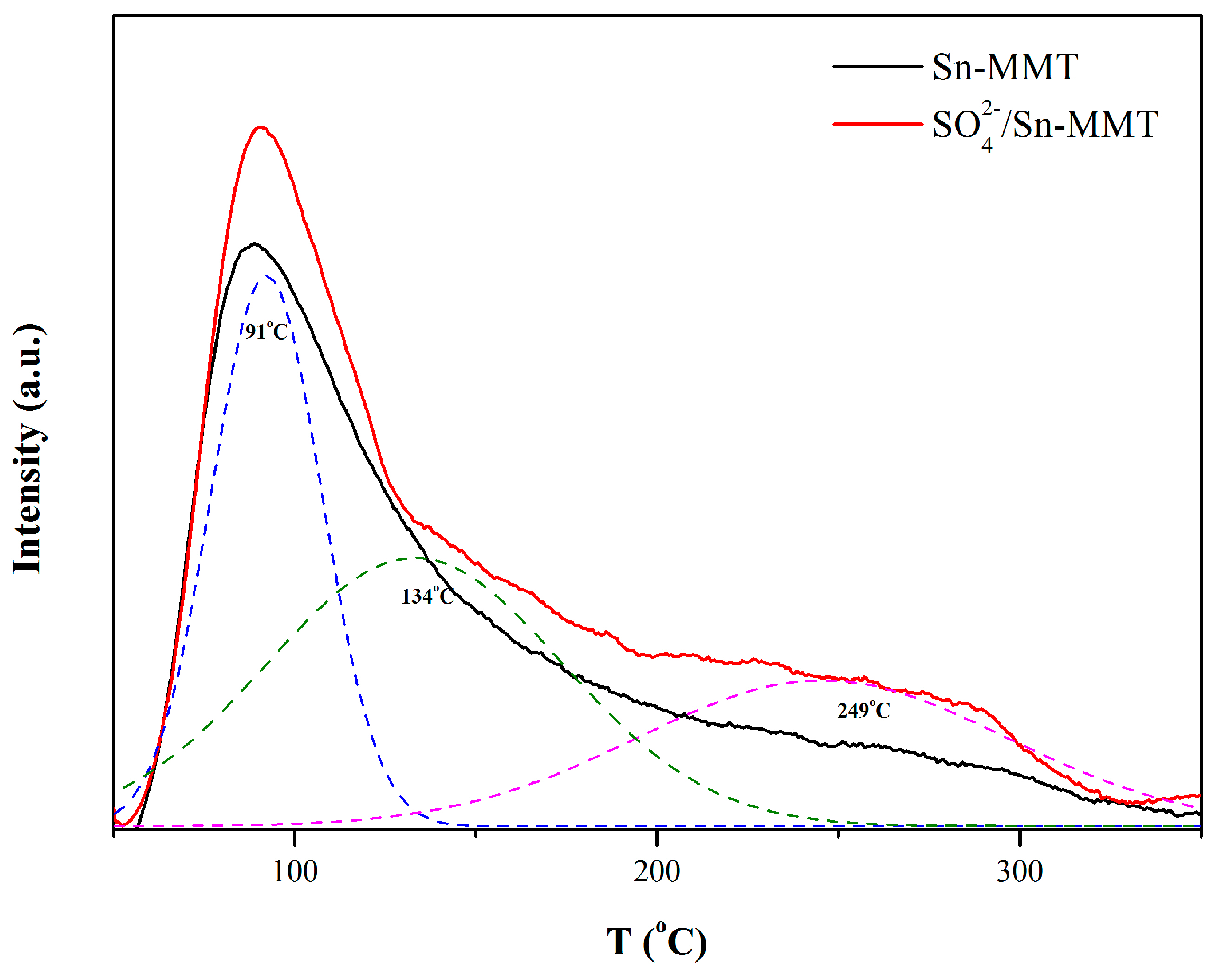
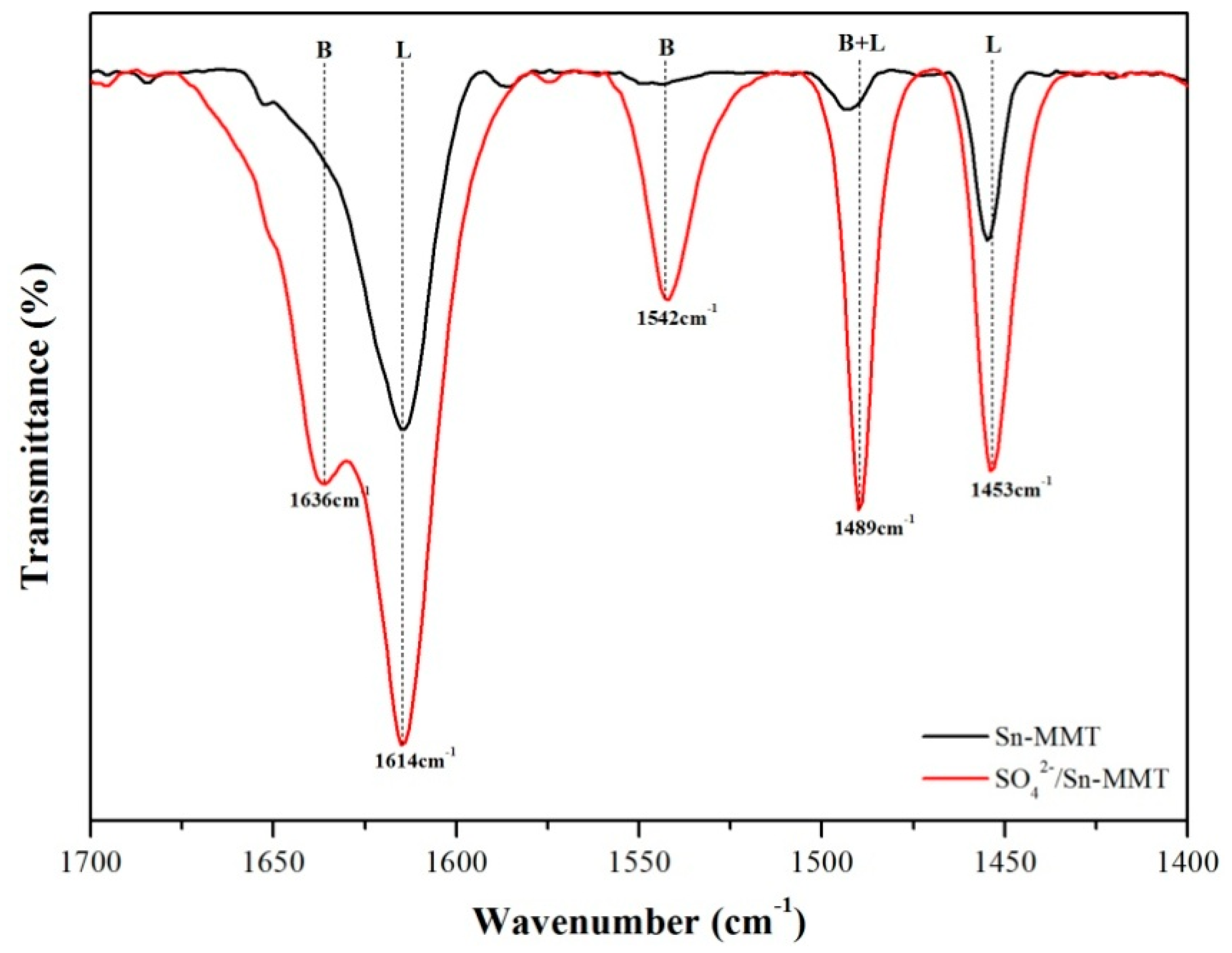
| Position (cm−1) | Assignment |
|---|---|
| 3630 | Structural Al–OH stretching |
| 3429 | H–OH stretching of adsorbed water |
| 2365 | O=C=O stretching |
| 1637 | H–OH bending of adsorbed water |
| 1104 | Si–O stretching |
| 1036 | Si–O–Si stretching |
| 909 | Al–OH bending |
| 835 | Al–Mg–OH deformation |
| 788 | Si–O stretching of quartz |
| 619 | Coupled Al–O and Si–O out-of-plane |
| 519 | Al–O–Si deformation |
| 461 | Si–O–Si deformation |
| Samples | Elemental Content (wt %) | BET Surface Area (m2 g−1) | Micropore Volume (cm3 g−1) | Pore Diameter (nm) | |||
|---|---|---|---|---|---|---|---|
| Ca | Na | Sn | S | ||||
| MMT | 3.44 | 0.54 | - | - | 42.38 | 0.18 | 9.15 |
| Sn-MMT | 0.94 | 0.16 | 15.32 | - | 178.94 | 0.27 | 12.37 |
| SO42−/Sn-MMT | 0.65 | 0.12 | 11.20 | 2.79 | 153.52 | 0.10 | 8.61 |
| No. | Substrate | Catalyst | Solvent | Temperature | Time | Yield | Refs. |
|---|---|---|---|---|---|---|---|
| 1 | Xylose | CrCl2, LiBr | N,N-dimethylacetamide | 100 °C | 4 h | 56% | [38] |
| 2 | Xylose | MCM-41-SO3H | toluene/water | 140 °C | 24 h | 75.5% | [42] |
| 3 | Xylose | PSZ-MCM-41 | toluene/water | 160 °C | 4 h | 42.8% | [43] |
| 4 | Xylose | Dealumin. HNu-6 (2) | toluene/water | 170 °C | 4 h | 47% | [44] |
| 5 | Xylose | H-mordenite 13 | toluene/water | 260 °C | 0.05 h | 98% | [39] |
| 6 | Xylose | Arenesulfonic SBA-15 | toluene/water | 160 °C | 20 h | 86% | [40] |
| 7 | Hydrolysates from corncob | Tin-loaded montmorillonite | SBP/NaCl-DMSO | 190 °C | 10 min | 57.80% | [45] |
| 8 | Isolated hemicellulose | Silicoaluminophosphate | toluene/water | 170 °C | 10 h | 82% | [41] |
| 9 | Bagasse | Silicoaluminophosphate | toluene/water | 170 °C | 8 h | 93% | [41] |
| 10 | Maple wood | Sulfuric acid | methyl isobutyl ketone/water | 170 °C | 50 min | 85.3% | [37] |
| 11 | Xylose | SO42−/Sn-MMT | 2-MTHF/water-NaCl | 160 °C | 2 h | 79.64% | In this work |
| 12 | Xylan | SO42−/Sn-MMT | 2-MTHF/water | 160 °C | 1.5 h | 77.35% | In this work |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, Q.; Li, H.; Wang, X.; Jian, L.; Ren, J.; Liu, C.; Sun, R. SO42−/Sn-MMT Solid Acid Catalyst for Xylose and Xylan Conversion into Furfural in the Biphasic System. Catalysts 2017, 7, 118. https://doi.org/10.3390/catal7040118
Lin Q, Li H, Wang X, Jian L, Ren J, Liu C, Sun R. SO42−/Sn-MMT Solid Acid Catalyst for Xylose and Xylan Conversion into Furfural in the Biphasic System. Catalysts. 2017; 7(4):118. https://doi.org/10.3390/catal7040118
Chicago/Turabian StyleLin, Qixuan, Huiling Li, Xiaohui Wang, Longfei Jian, Junli Ren, Chuanfu Liu, and Runcang Sun. 2017. "SO42−/Sn-MMT Solid Acid Catalyst for Xylose and Xylan Conversion into Furfural in the Biphasic System" Catalysts 7, no. 4: 118. https://doi.org/10.3390/catal7040118
APA StyleLin, Q., Li, H., Wang, X., Jian, L., Ren, J., Liu, C., & Sun, R. (2017). SO42−/Sn-MMT Solid Acid Catalyst for Xylose and Xylan Conversion into Furfural in the Biphasic System. Catalysts, 7(4), 118. https://doi.org/10.3390/catal7040118








