Catalytic Upgrading of Biomass-Derived Furfuryl Alcohol to Butyl Levulinate Biofuel over Common Metal Salts
Abstract
:1. Introduction
2. Results and Discussion
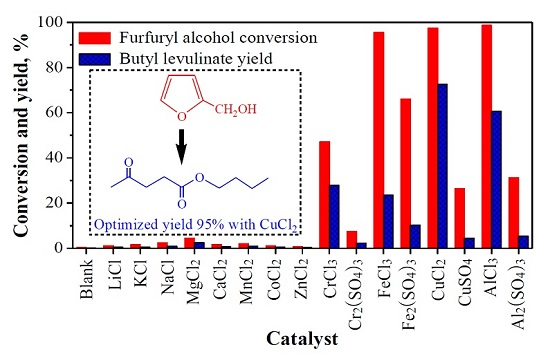
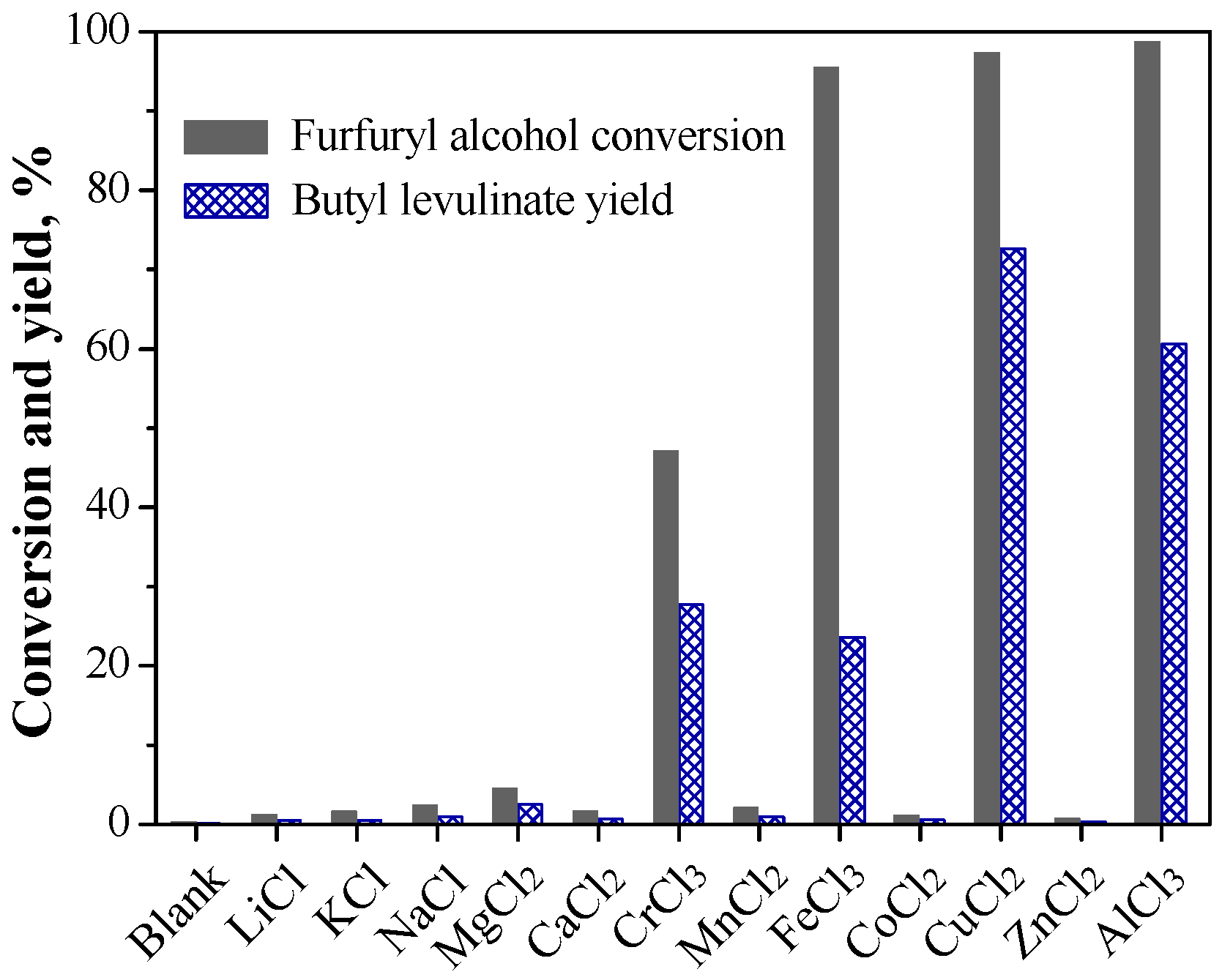
2.1. Various Metal Salts’ Catalytic Conversion of Furfuryl Alcohol
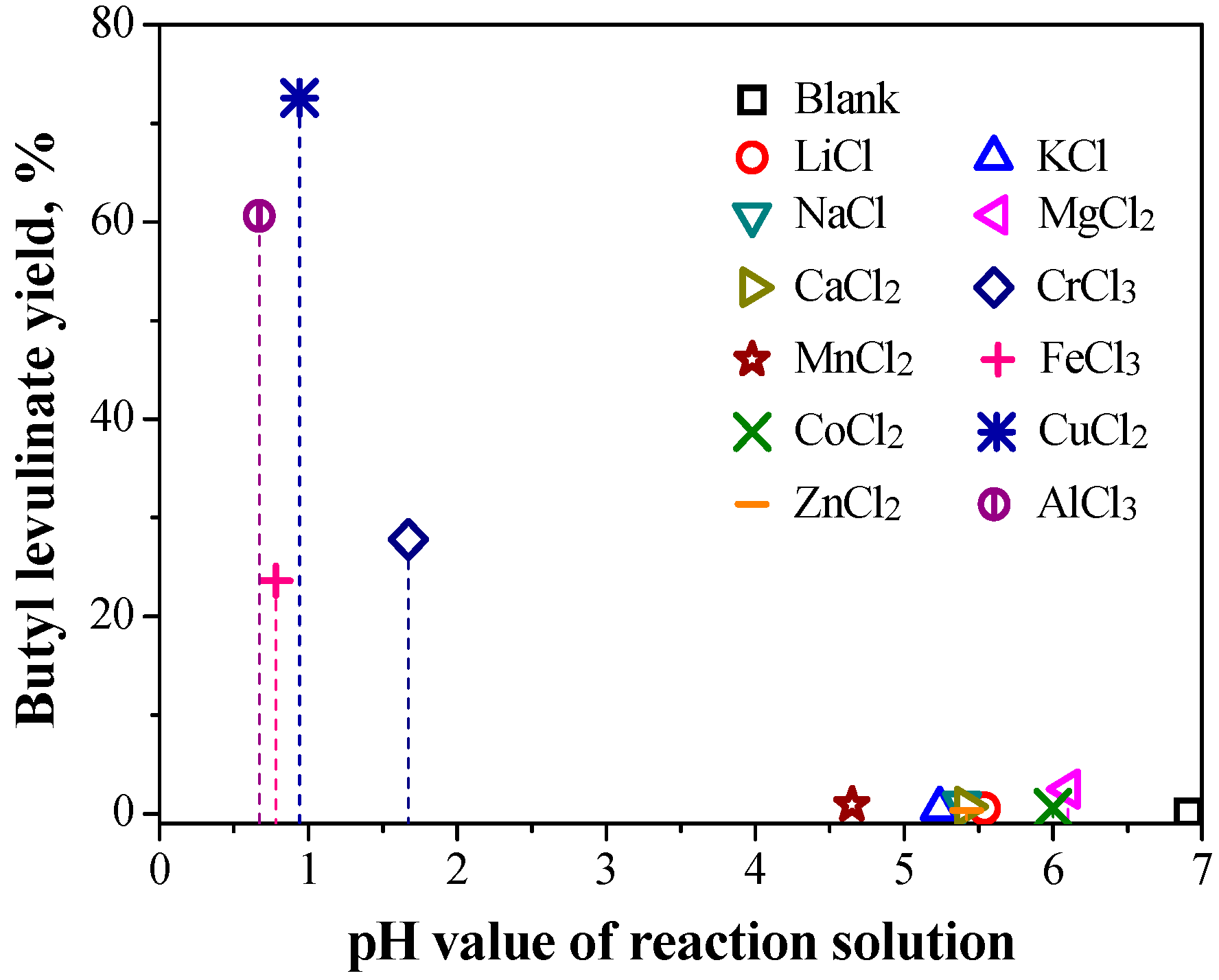
2.1.1. Catalytic Performance of Various Metal Salts
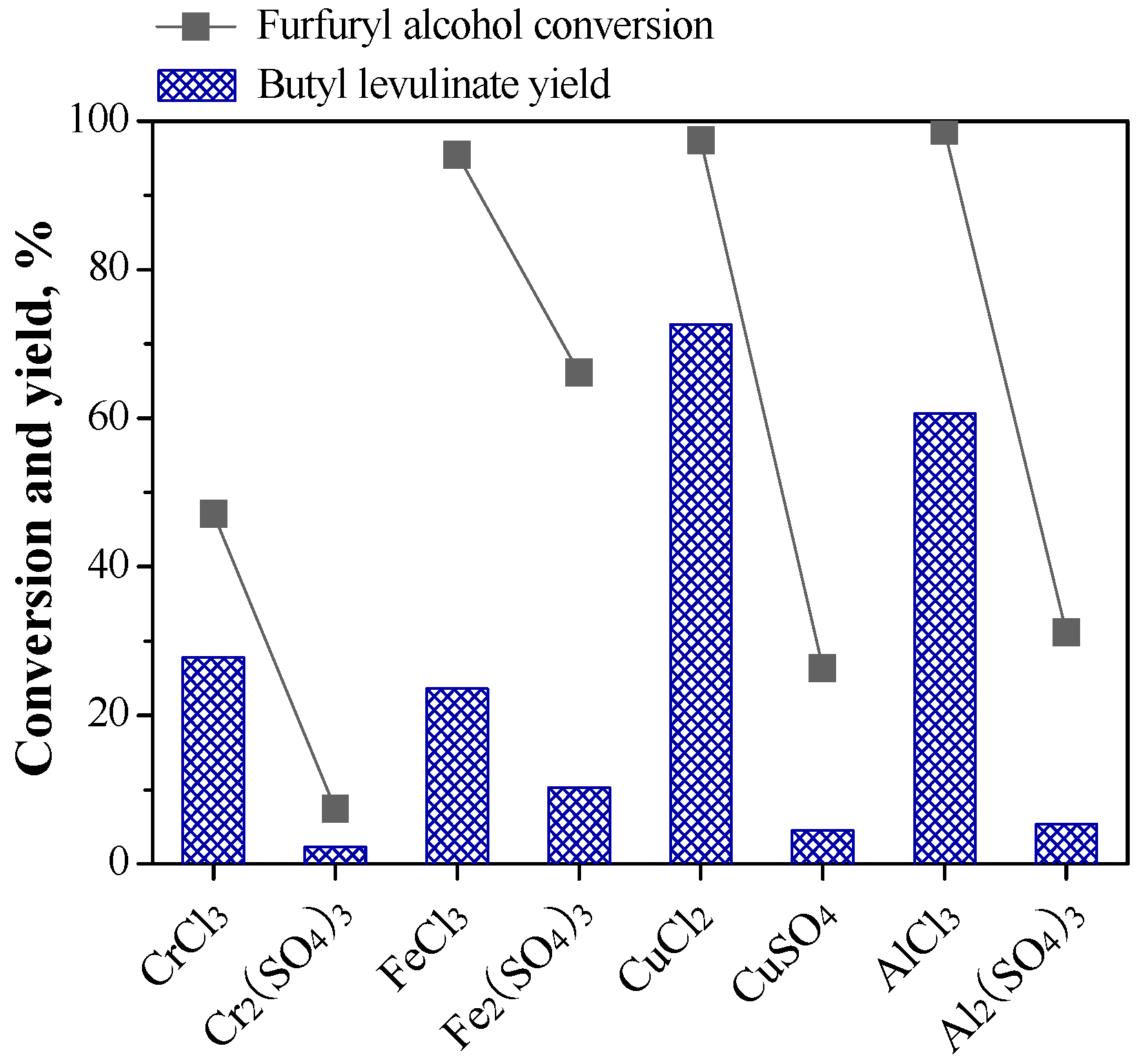
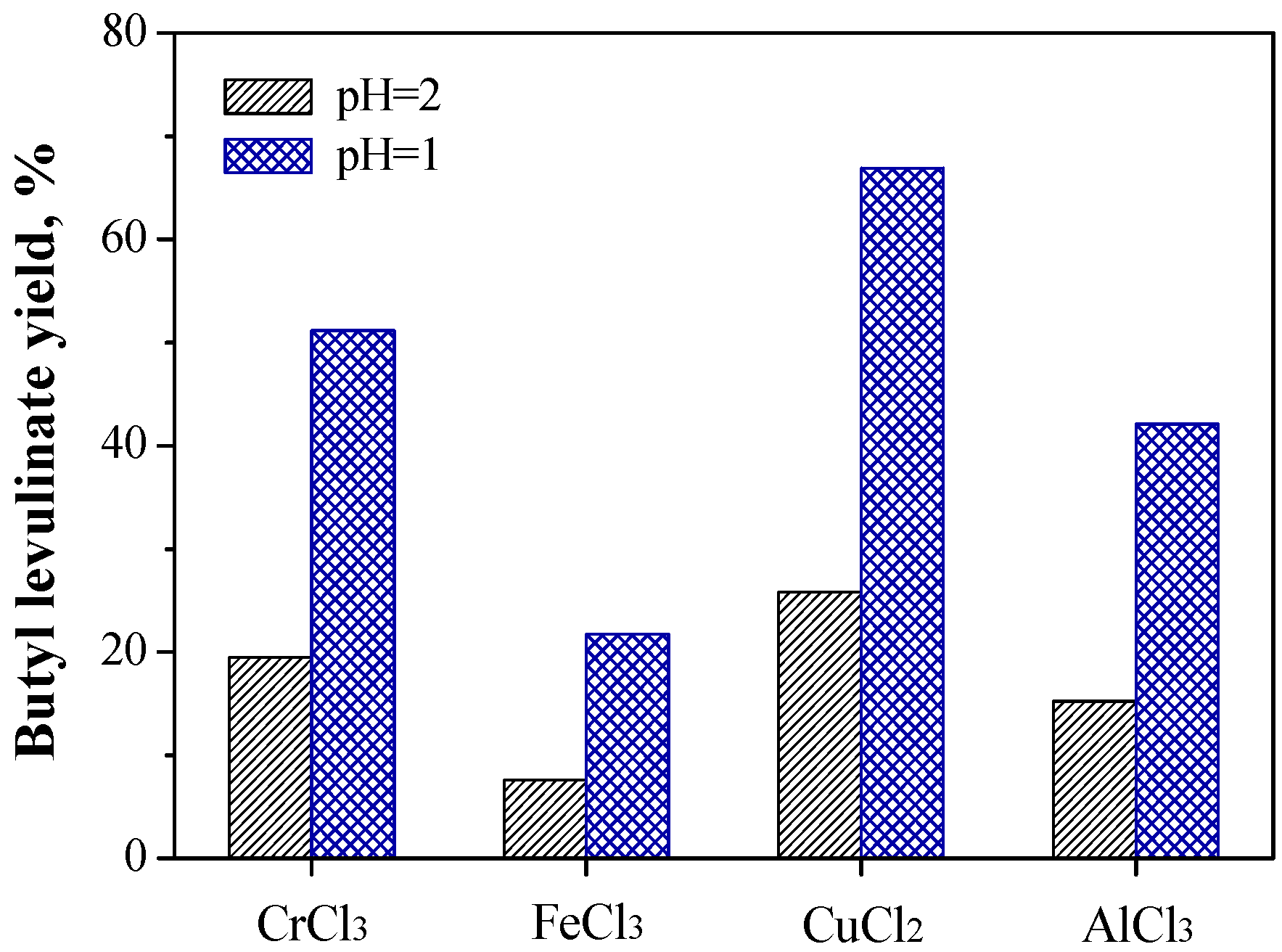
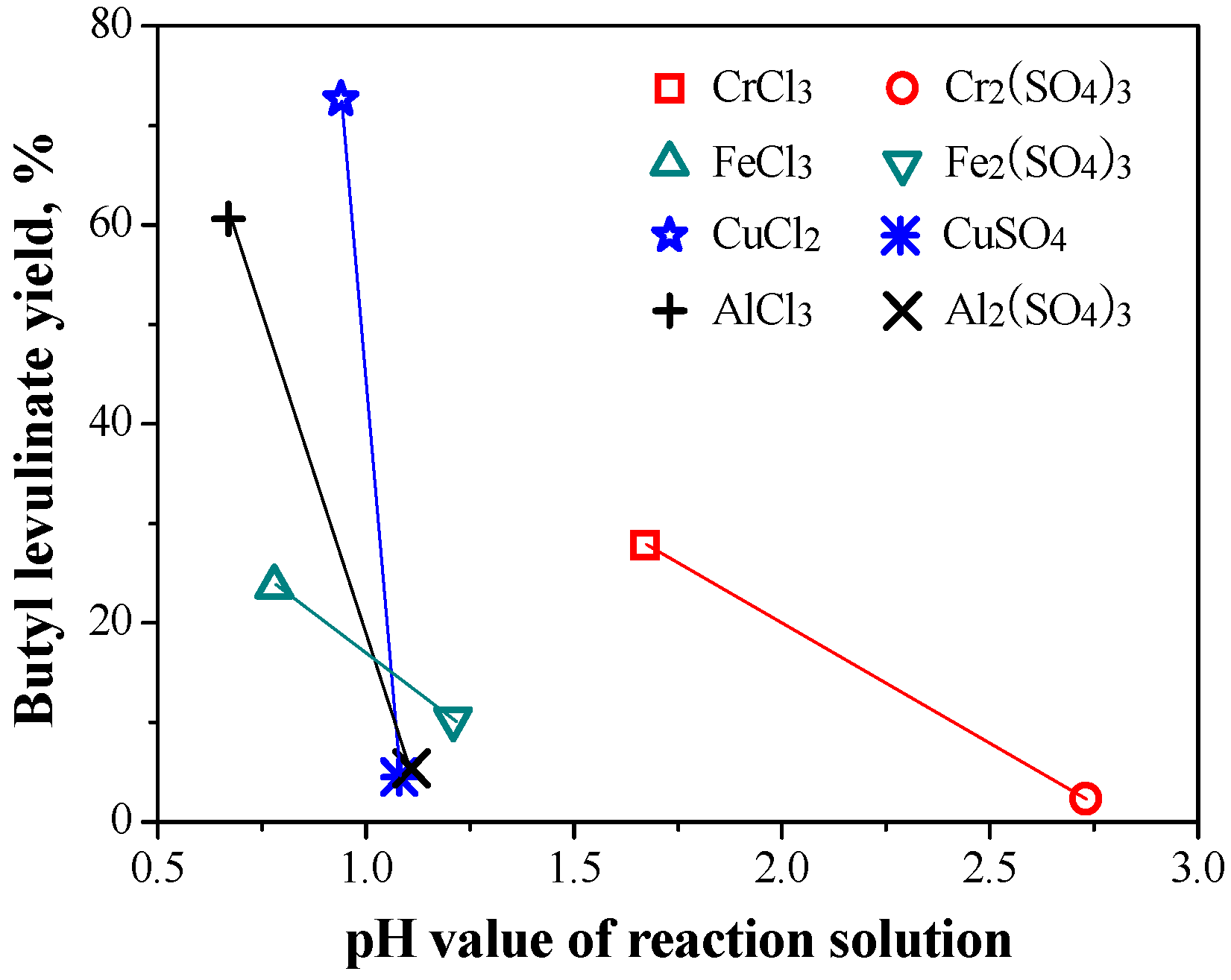
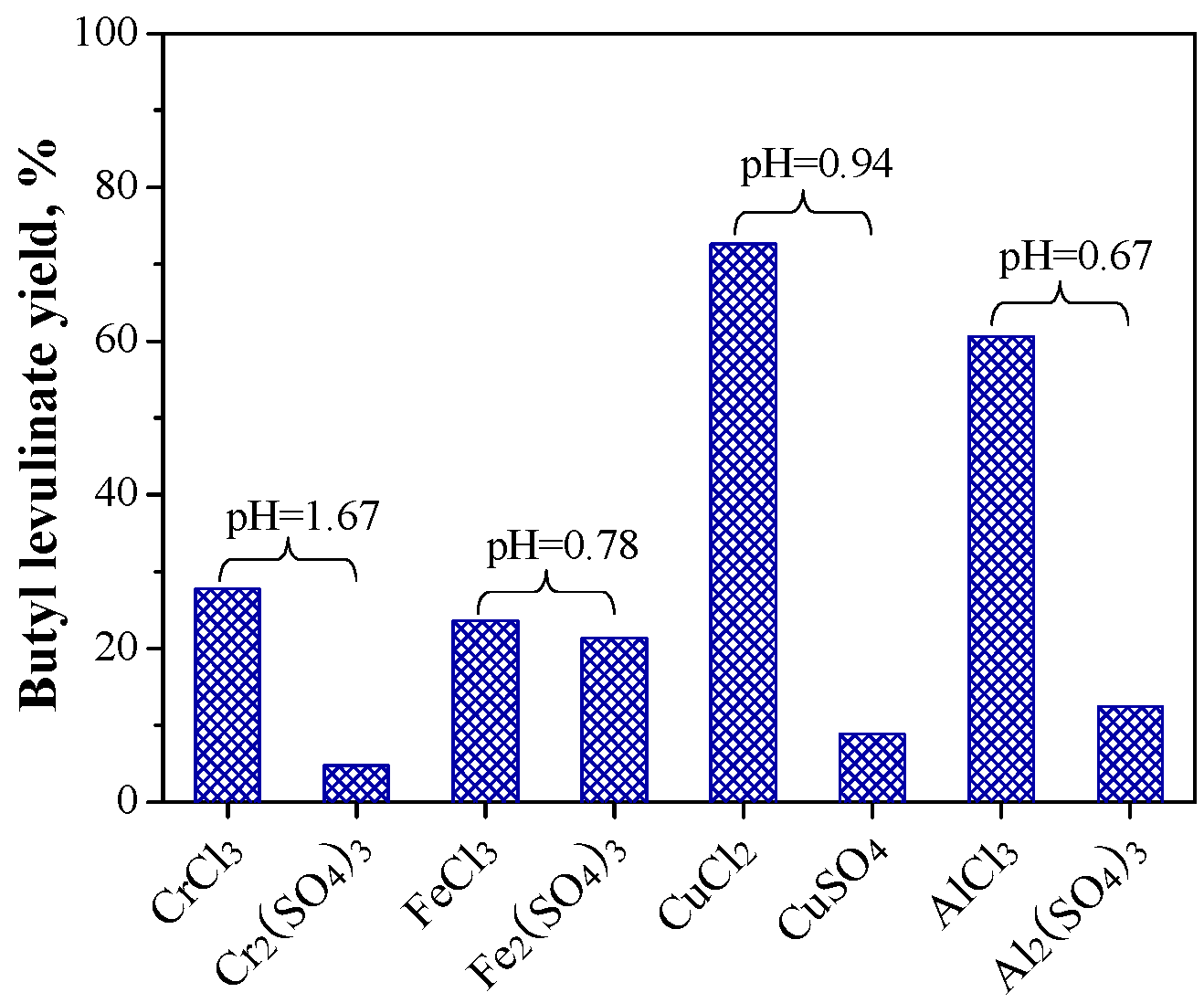
2.1.2. Relationship between Reactivity and Acidity of Reaction System
2.2. Optimization of Butyl Levulinate Production Catalyzed by CuCl2
2.2.1. Catalyst Dosage
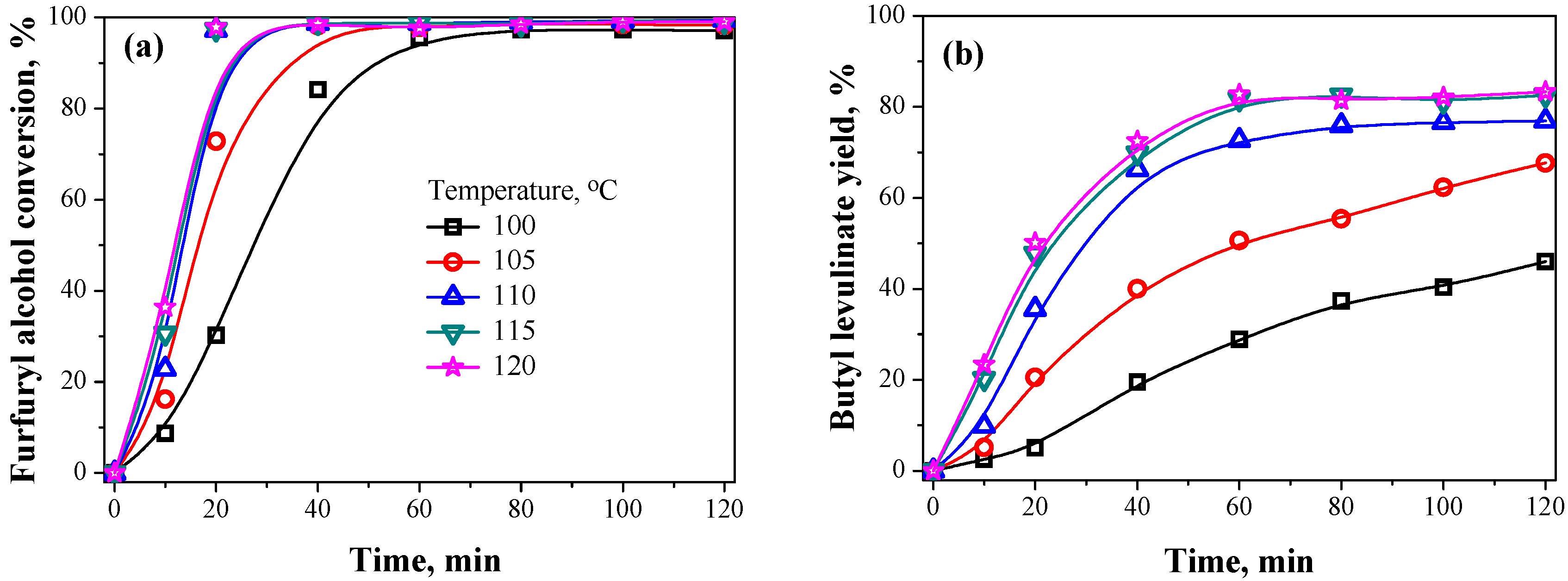
2.2.2. Reaction Temperature
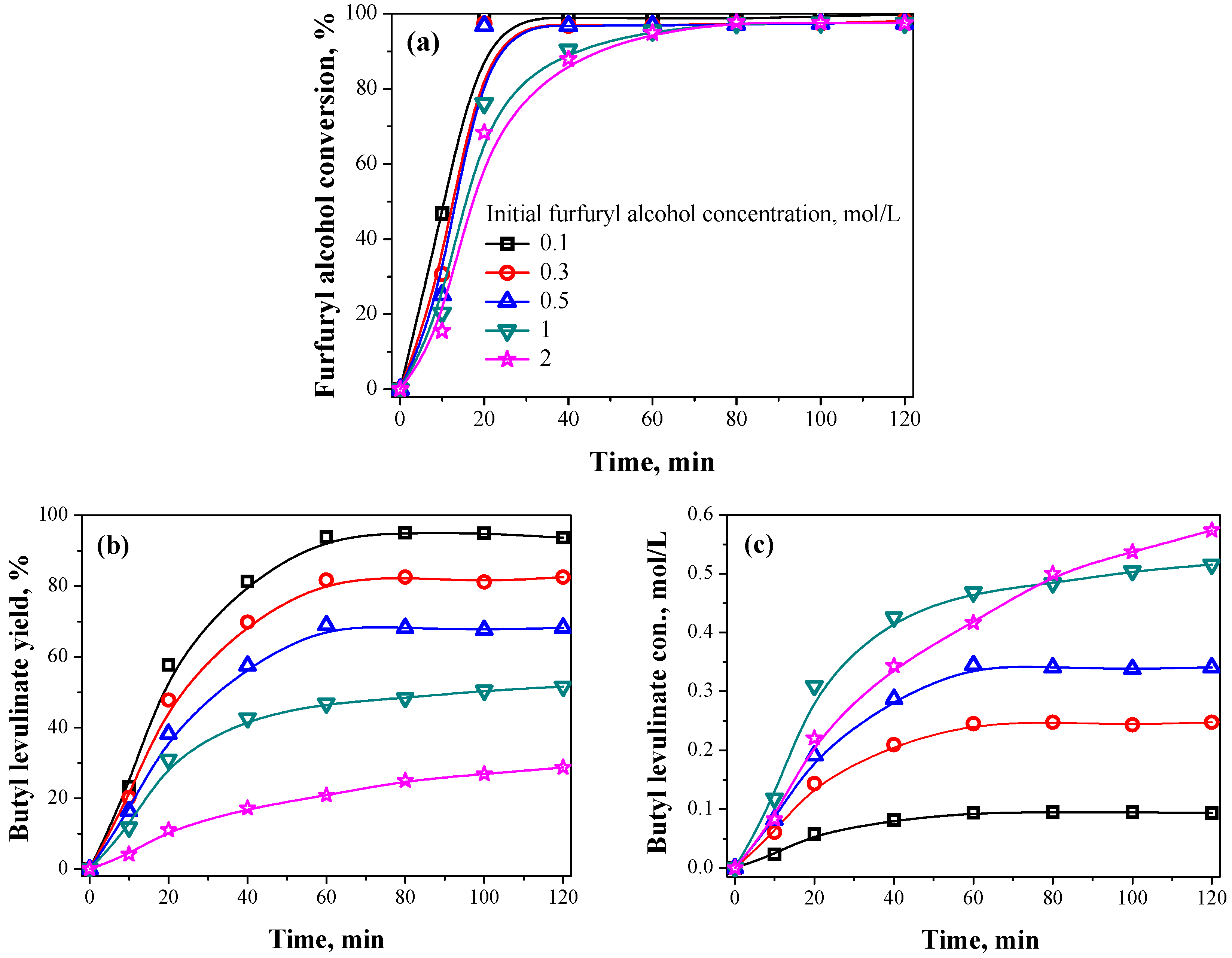
2.2.3. Initial Furfuryl Alcohol Concentration
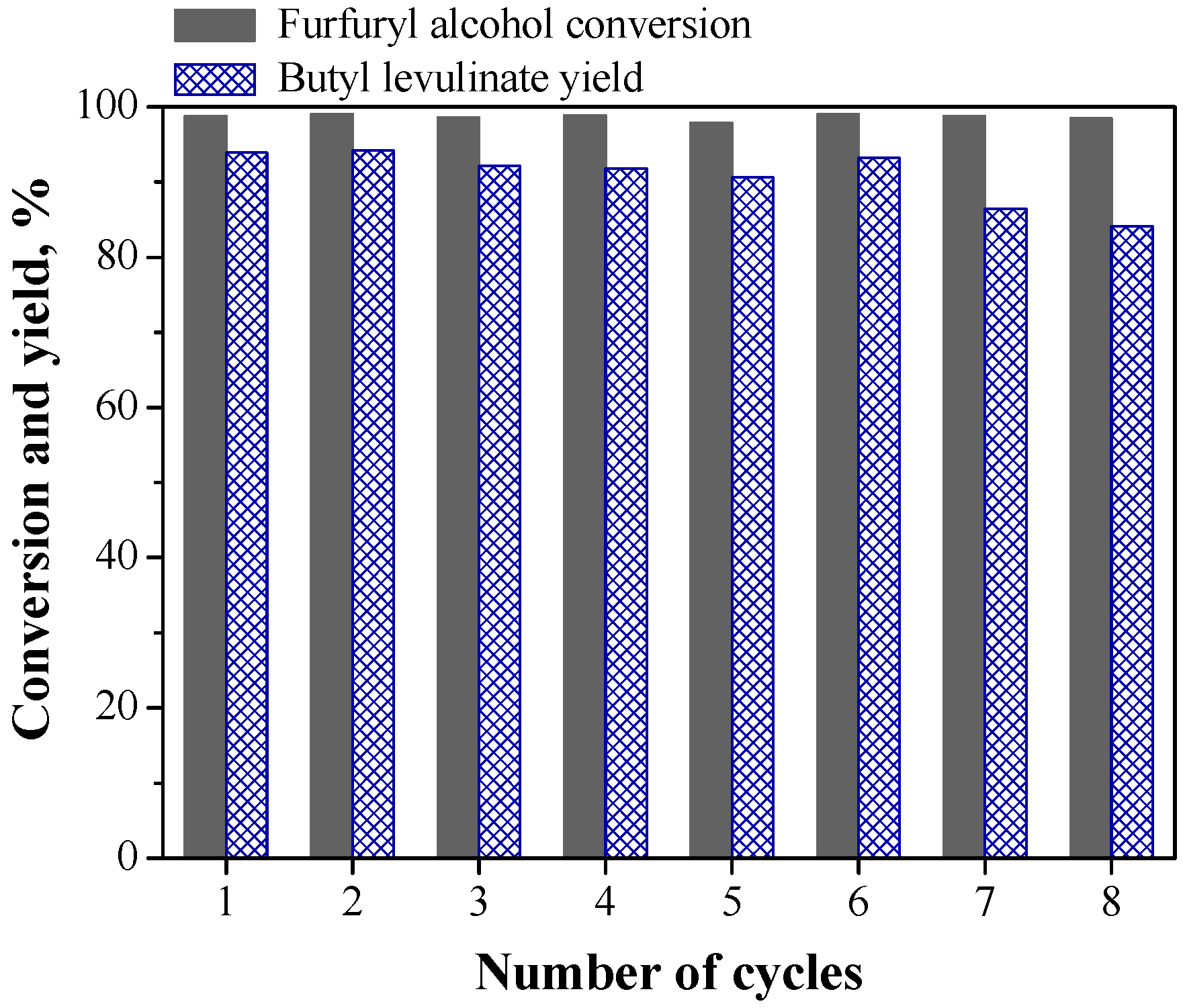
2.3. Reusability of CuCl2 Catalyst
3. Materials and Methods
3.1. Materials
3.2. General Procedure for the Synthesis of Butyl Levulinate
3.3. Products Analysis
3.4. pH Measurement of the Reaction Solution
3.5. Recycling of the Catalyst
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Naik, S.N.; Goud, V.V.; Rout, P.K.; Dalai, A.K. Production of first and second generation biofuels: A comprehensive review. Renew. Sustain. Energy Rev. 2010, 14, 578–597. [Google Scholar] [CrossRef]
- Srirangan, K.; Akawi, L.; Moo-Young, M.; Chou, C.P. Towards sustainable production of clean energy carriers from biomass resources. Appl. Energy 2012, 100, 172–186. [Google Scholar] [CrossRef]
- Ullah, K.; Sharma, V.K.; Dhingra, S.; Braccio, G.; Ahmad, M.; Sofia, S. Assessing the lignocellulosic biomass resources potential in developing countries: A critical review. Renew. Sustain. Energy Rev. 2015, 51, 682–698. [Google Scholar] [CrossRef]
- Deng, W.; Zhang, Q.; Wang, Y. Catalytic transformation of cellulose and its derived carbohydrates into chemicals involving C–C bond cleavage. J. Energy Chem. 2015, 24, 595–607. [Google Scholar] [CrossRef]
- Gandini, A. Furans as offspring of sugars and polysaccharides and progenitors of a family of remarkable polymers: A review of recent progress. Polym. Chem. UK 2010, 1, 245–251. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, Z.; Song, L. Efficient and selective alcoholysis of furfuryl alcohol to alkyl levulinates catalyzed by double SO3H-functionalized ionic liquids. Green Chem. 2014, 16, 1436–1443. [Google Scholar]
- Gao, X.; Peng, L.; Li, H.; Chen, K. Formation of humin and alkyl levulinate in the acid-catalyzed conversion of biomass-derived furfuryl alcohol. BioResources 2015, 10, 6548–6564. [Google Scholar] [CrossRef]
- Windom, B.C.; Lovestead, T.M.; Mascal, M.; Nikitin, E.B.; Bruno, T.J. Advanced distillation curve analysis on ethyl levulinate as a diesel fuel oxygenate and a hybrid biodiesel fuel. Energy Fuels 2011, 25, 1878–1890. [Google Scholar] [CrossRef]
- Joshi, H.; Moser, B.R.; Toler, J.; Smith, W.F.; Walker, T. Ethyl levulinate: A potential bio-based diluent for biodiesel which improves cold flow properties. Biomass Bioenergy 2011, 35, 3262–3266. [Google Scholar] [CrossRef]
- Alonso, D.M.; Wettstein, S.G.; Dumesic, J.A. Gamma-valerolactone, a sustainable platform molecule derived from lignocellulosic biomass. Green Chem. 2013, 15, 584–595. [Google Scholar] [CrossRef]
- Tang, X.; Zeng, X.; Li, Z.; Hu, L.; Sun, Y.; Liu, S.; Lei, T.; Lin, L. Production of γ-valerolactone from lignocellulosic biomass for sustainable fuels and chemicals supply. Renew. Sustain. Energy Rev. 2014, 40, 608–620. [Google Scholar] [CrossRef]
- Bond, J.Q.; Alonso, D.M.; Wang, D.; West, R.M.; Dumesic, J.A. Integrated catalytic conversion of γ-valerolactone to liquid alkenes for transportation fuels. Science 2010, 327, 1110–1114. [Google Scholar] [CrossRef] [PubMed]
- Démolis, A.; Essayem, N.; Rataboul, F. Synthesis and applications of alkyl levulinates. ACS Sustain. Chem. Eng. 2014, 2, 1338–1352. [Google Scholar] [CrossRef]
- Ahmad, E.; Alam, M.I.; Pant, K.K.; Haider, M.A. Catalytic and mechanistic insights into the production of ethyl levulinate from biorenewable feedstocks. Green Chem. 2016. [Google Scholar] [CrossRef]
- Ren, D.; Fu, J.; Li, L.; Liu, Y.; Jin, F.; Huo, Z. Efficient conversion of biomass-derived furfuryl alcohol to levulinate esters over commercial α-Fe2O3. RSC Adv. 2016, 6, 22174–22178. [Google Scholar] [CrossRef]
- Lange, J.P.; van de Graaf, W.D.; Haan, R.J. Conversion of furfuryl alcohol into ethyl levulinate using solid acid catalysts. ChemSusChem 2009, 2, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Dong, K.; Zhao, Z.K. Efficient conversion of furfuryl alcohol into alkyl levulinates catalyzed by an organic–inorganic hybrid solid acid catalyst. ChemSusChem 2011, 4, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Neves, P.; Lima, S.; Pillinger, M.; Rocha, S.M.; Rocha, J.; Valente, A.A. Conversion of furfuryl alcohol to ethyl levulinate using porous aluminosilicate acid catalysts. Catal. Today 2013, 218, 76–84. [Google Scholar] [CrossRef]
- Demma Carà, P.; Ciriminna, R.; Shiju, N.R.; Rothenberg, G.; Pagliaro, M. Enhanced heterogeneous catalytic conversion of furfuryl alcohol into butyl levulinate. ChemSusChem 2014, 7, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Neves, P.; Russo, P.A.; Fernandes, A.; Antunes, M.M.; Farinha, J.; Pillinger, M.; Ribeiro, M.F.; Castanheiro, J.E.; Valente, A.A. Mesoporous zirconia-based mixed oxides as versatile acid catalysts for producing bio-additives from furfuryl alcohol and glycerol. Appl. Catal. A 2014, 487, 148–157. [Google Scholar] [CrossRef]
- Russo, P.A.; Antunes, M.M.; Neves, P.; Wiper, P.V.; Fazio, E.; Neri, F.; Barreca, F.; Mafra, L.; Pillinger, M.; Pinna, N.; et al. Solid acids with SO3H groups and tunable surface properties: Versatile catalysts for biomass conversion. J. Mater. Chem. A 2014, 2, 11813–11824. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, C.; Xue, Y.; Wu, J.; Wang, J.; Fan, W. Graphene oxide: An efficient acid catalyst for alcoholysis and esterification reactions. ChemCatChem 2014, 6, 3080–3083. [Google Scholar] [CrossRef]
- Lu, B.; An, S.; Song, D.; Su, F.; Yang, X.; Guo, Y. Design of organosulfonic acid functionalized organosilica hollow nanospheres for efficient conversion of furfural alcohol to ethyl levulinate. Green Chem. 2015, 17, 1767–1778. [Google Scholar] [CrossRef]
- Nandiwale, K.Y.; Pande, A.M.; Bokade, V.V. One step synthesis of ethyl levulinate biofuel by ethanolysis of renewable furfuryl alcohol over hierarchical zeolite catalyst. RSC Adv. 2015, 5, 79224–79231. [Google Scholar] [CrossRef]
- Hengne, A.M.; Kamble, S.B.; Rode, C.V. Single pot conversion of furfuryl alcohol to levulinic esters and γ-valerolactone in the presence of sulfonic acid functionalized ILs and metal catalysts. Green Chem. 2013, 15, 2540–2547. [Google Scholar] [CrossRef]
- Zhao, H.; Holladay, J.E.; Brown, H.; Zhang, Z.C. Metal chlorides in ionic liquid solvents convert sugars to 5-hydroxymethylfurfural. Science 2007, 316, 1597–1600. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Lin, L.; Zhang, J.; Zhuang, J.; Zhang, B.; Gong, Y. Catalytic conversion of cellulose to levulinic acid by metal chlorides. Molecules 2010, 15, 5258–5272. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zou, H.; Nan, J.; Wu, L.; Yang, X.; Su, Y.; Lu, T.; Xu, J. Conversion of carbohydrate biomass to methyl levulinate with Al2(SO4)3 as a simple, cheap and efficient catalyst. Catal. Commun. 2014, 50, 13–16. [Google Scholar] [CrossRef]
- Huang, Y.B.; Yang, T.; Zhou, M.C.; Pan, H.; Fu, Y. Microwave-assisted alcoholysis of furfural alcohol into alkyl levulinates catalyzed by metal salts. Green Chem. 2016, 18, 1516–1523. [Google Scholar] [CrossRef]
- Kean, J.R.; Graham, A.E. Indium (III) triflate promoted synthesis of alkyl levulinates from furyl alcohols and furyl aldehydes. Catal. Commun. 2015, 59, 175–179. [Google Scholar] [CrossRef]
- Peng, L.; Gao, X.; Chen, K. Catalytic upgrading of renewable furfuryl alcohol to alkyl levulinates using AlCl3 as a facile, efficient, and reusable catalyst. Fuel 2015, 60, 123–131. [Google Scholar] [CrossRef]
- Stames, W.H.; Frantz, S.; Chung, H.T. Aluminum chloride [alias its reaction product(s) with ethanol] for the stabilization of poly (vinyl chloride)? Polym. Degrad. Stab. 1997, 56, 103–108. [Google Scholar]
- Choudhary, V.; Mushrif, S.H.; Ho, C.; Anderko, A.; Nikolakis, V.; Marinkovic, N.S.; Frenkel, A.I.; Sandler, S.I.; Vlachos, D.G. Insights into the interplay of Lewis and Brønsted acid catalysts in glucose and fructose conversion to 5-(hydroxymethyl)furfural and levulinic acid in aqueous media. J. Am. Chem. Soc. 2013, 135, 3997–4006. [Google Scholar] [CrossRef] [PubMed]
- Corma, A.; Garcia, H. Lewis acids: From conventional homogeneous to green homogeneous and heterogeneous catalysis. Chem. Rev. 2003, 103, 4307–4366. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, T.; Ohnishi, R.; Tanabe, K. Recent survey of catalysis by solid metal sulfates. Catal. Rev. 1974, 8, 29–63. [Google Scholar] [CrossRef]
- Maldonado, G.M.G.; Assary, R.S.; Dumesic, J.A.; Curtiss, L.A. Acid-catalyzed conversion of furfuryl alcohol to ethyl levulinate in liquid ethanol. Energy Environ. Sci. 2012, 5, 8990–8997. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhang, Z.; Huang, K.; Fang, Z. Efficient conversion of carbohydrates into 5-ethoxymethylfurfural in ethanol catalyzed by AlCl3. Fuel 2013, 113, 625–631. [Google Scholar] [CrossRef]
- Tominaga, K.I.; Mori, A.; Fukushima, Y.; Shimada, S.; Sato, K. Mixed-acid systems for the catalytic synthesis of methyl levulinate from cellulose. Green Chem. 2011, 13, 810–812. [Google Scholar] [CrossRef]
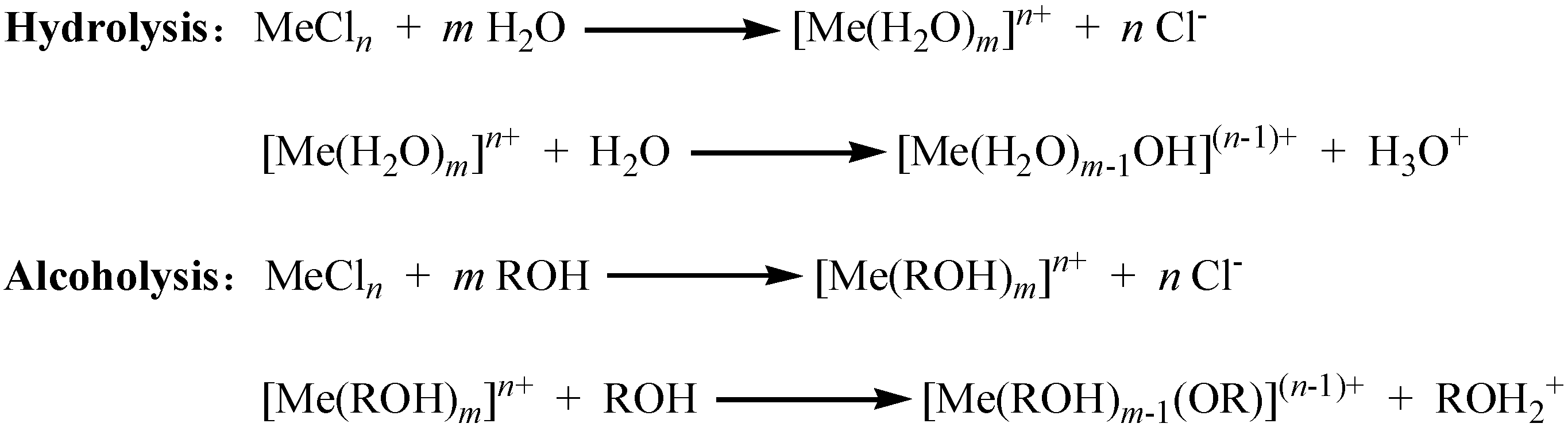

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, L.; Tao, R.; Wu, Y. Catalytic Upgrading of Biomass-Derived Furfuryl Alcohol to Butyl Levulinate Biofuel over Common Metal Salts. Catalysts 2016, 6, 143. https://doi.org/10.3390/catal6090143
Peng L, Tao R, Wu Y. Catalytic Upgrading of Biomass-Derived Furfuryl Alcohol to Butyl Levulinate Biofuel over Common Metal Salts. Catalysts. 2016; 6(9):143. https://doi.org/10.3390/catal6090143
Chicago/Turabian StylePeng, Lincai, Ruili Tao, and Yu Wu. 2016. "Catalytic Upgrading of Biomass-Derived Furfuryl Alcohol to Butyl Levulinate Biofuel over Common Metal Salts" Catalysts 6, no. 9: 143. https://doi.org/10.3390/catal6090143
APA StylePeng, L., Tao, R., & Wu, Y. (2016). Catalytic Upgrading of Biomass-Derived Furfuryl Alcohol to Butyl Levulinate Biofuel over Common Metal Salts. Catalysts, 6(9), 143. https://doi.org/10.3390/catal6090143