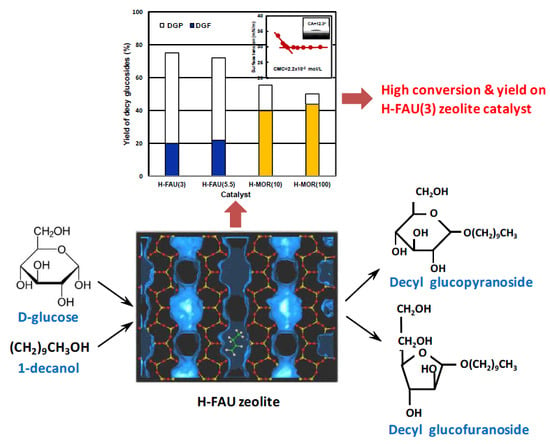
Microporous Zeolites as Catalysts for the Preparation of Decyl Glucoside from Glucose with 1-Decanol by Direct Glucosidation
Abstract
:1. Introduction
2. Results and Discussion
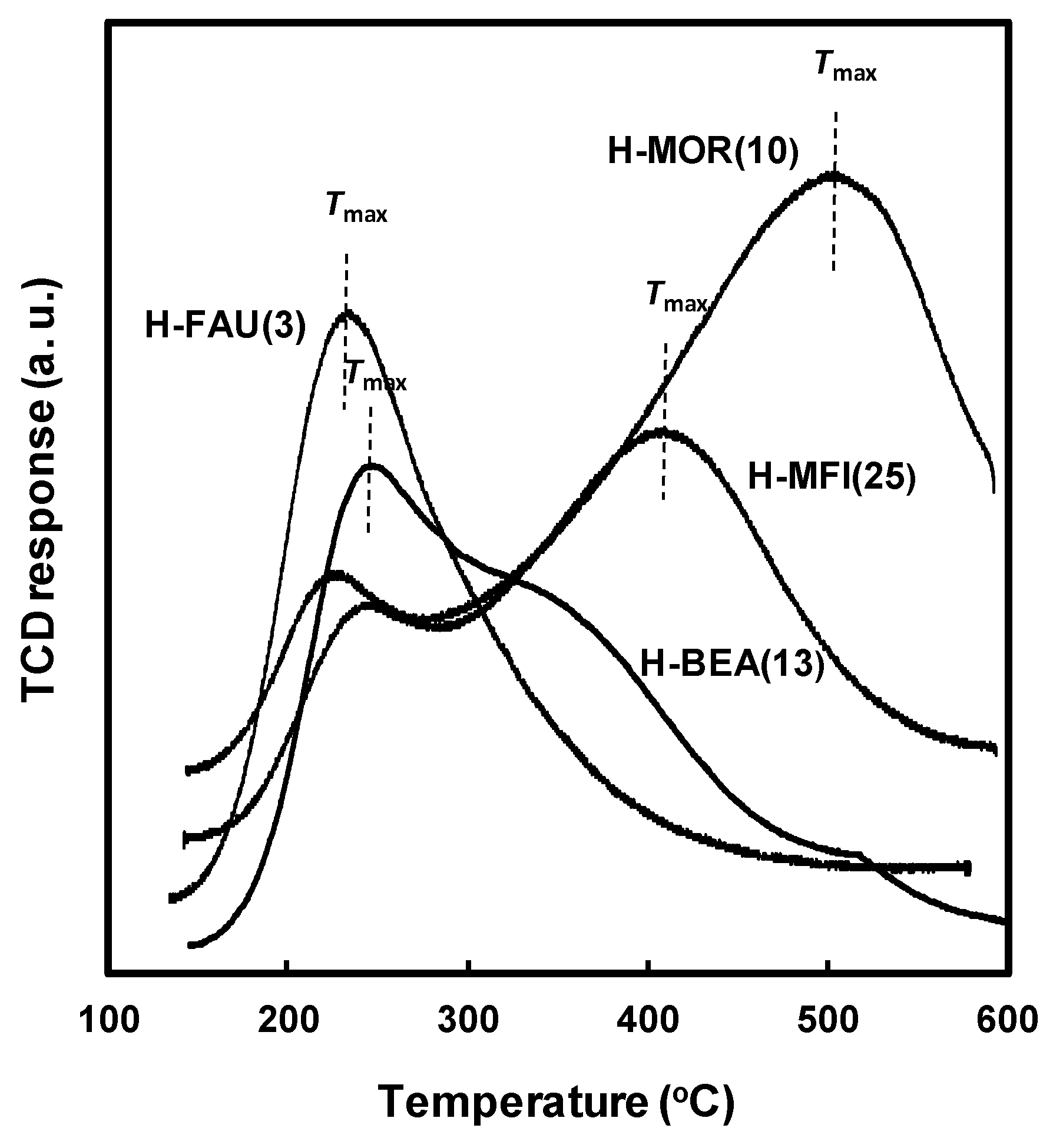
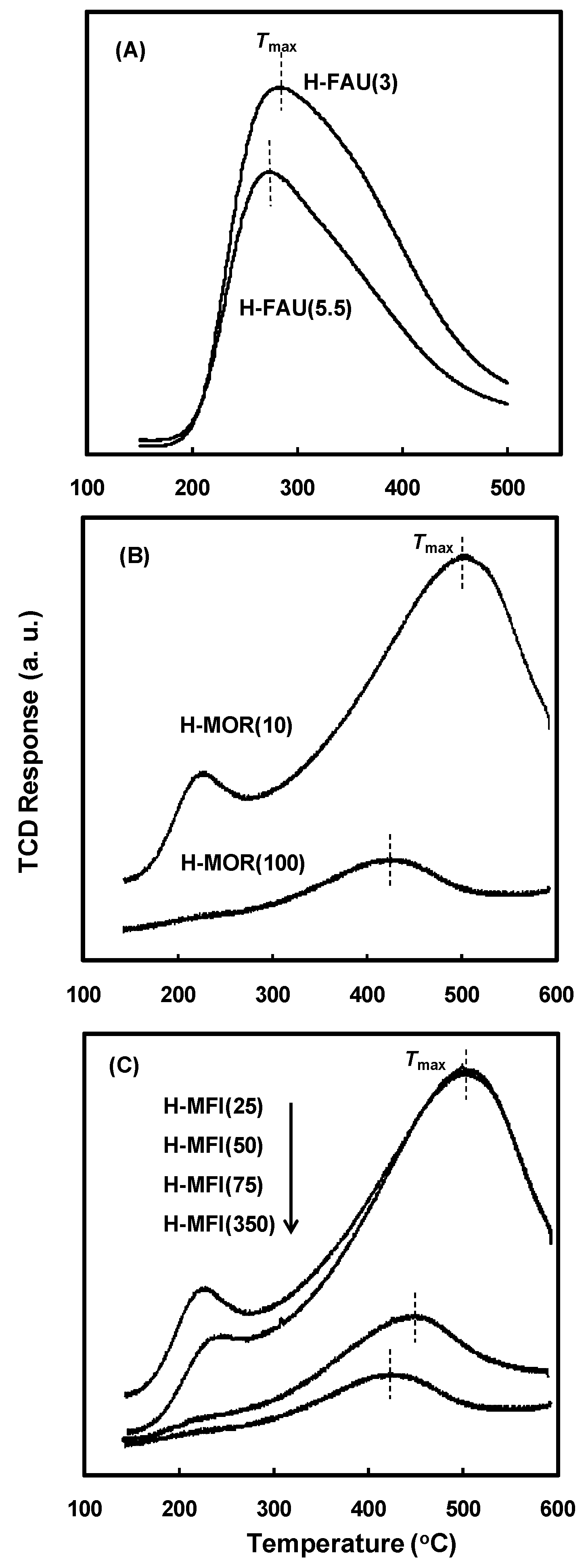
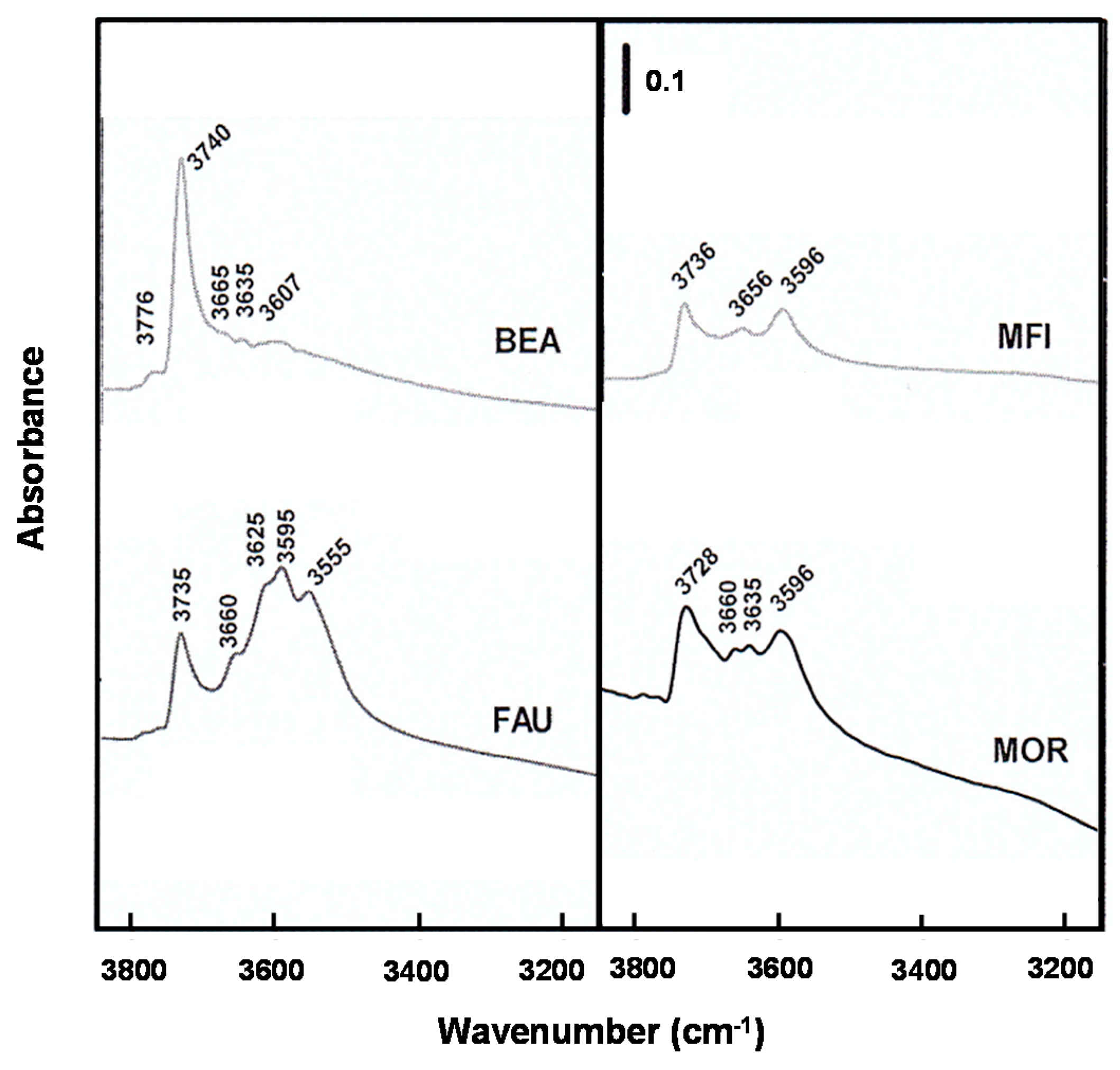
2.1. Physicochemical Properties of the Zeolite Catalysts
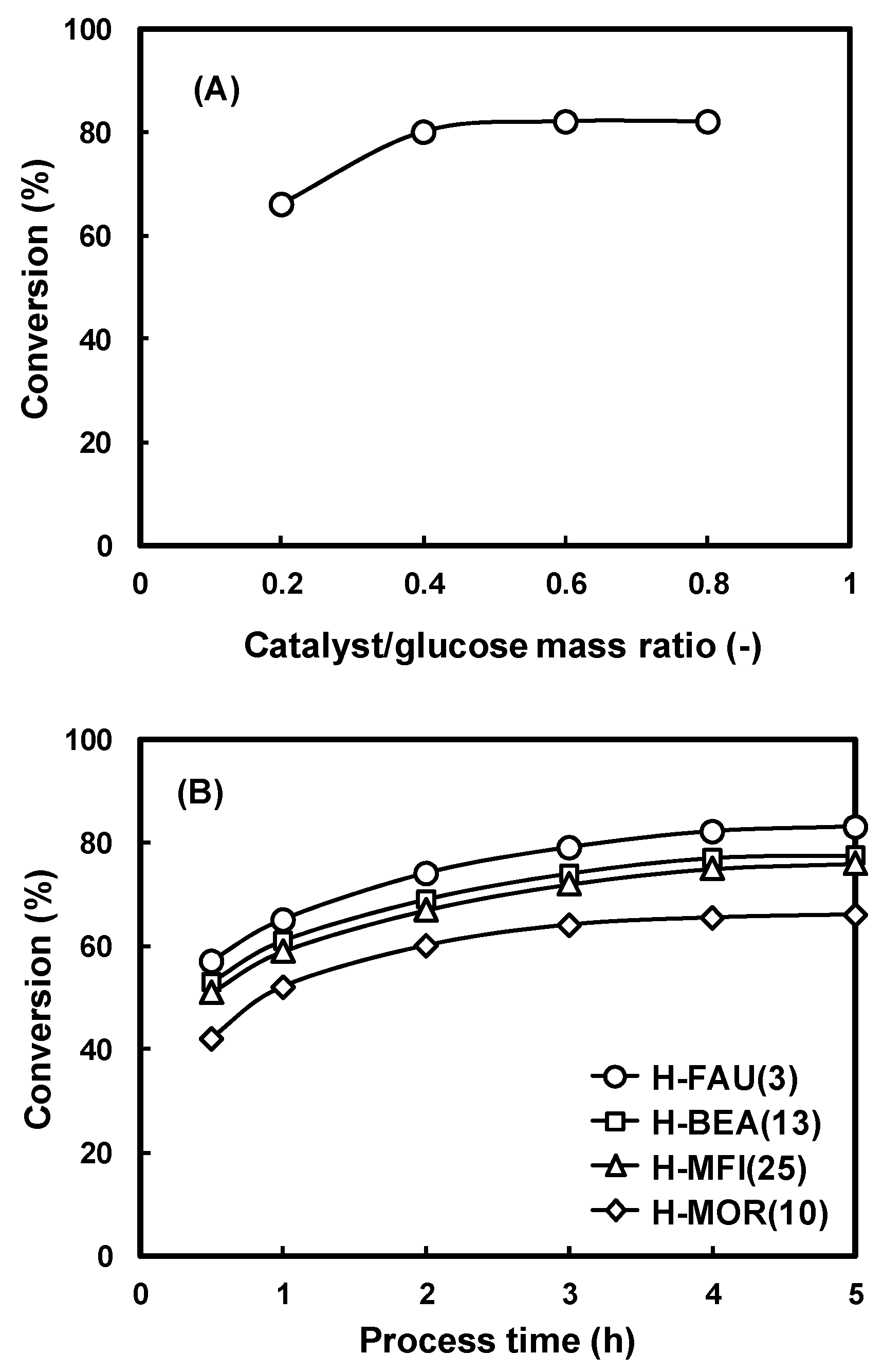
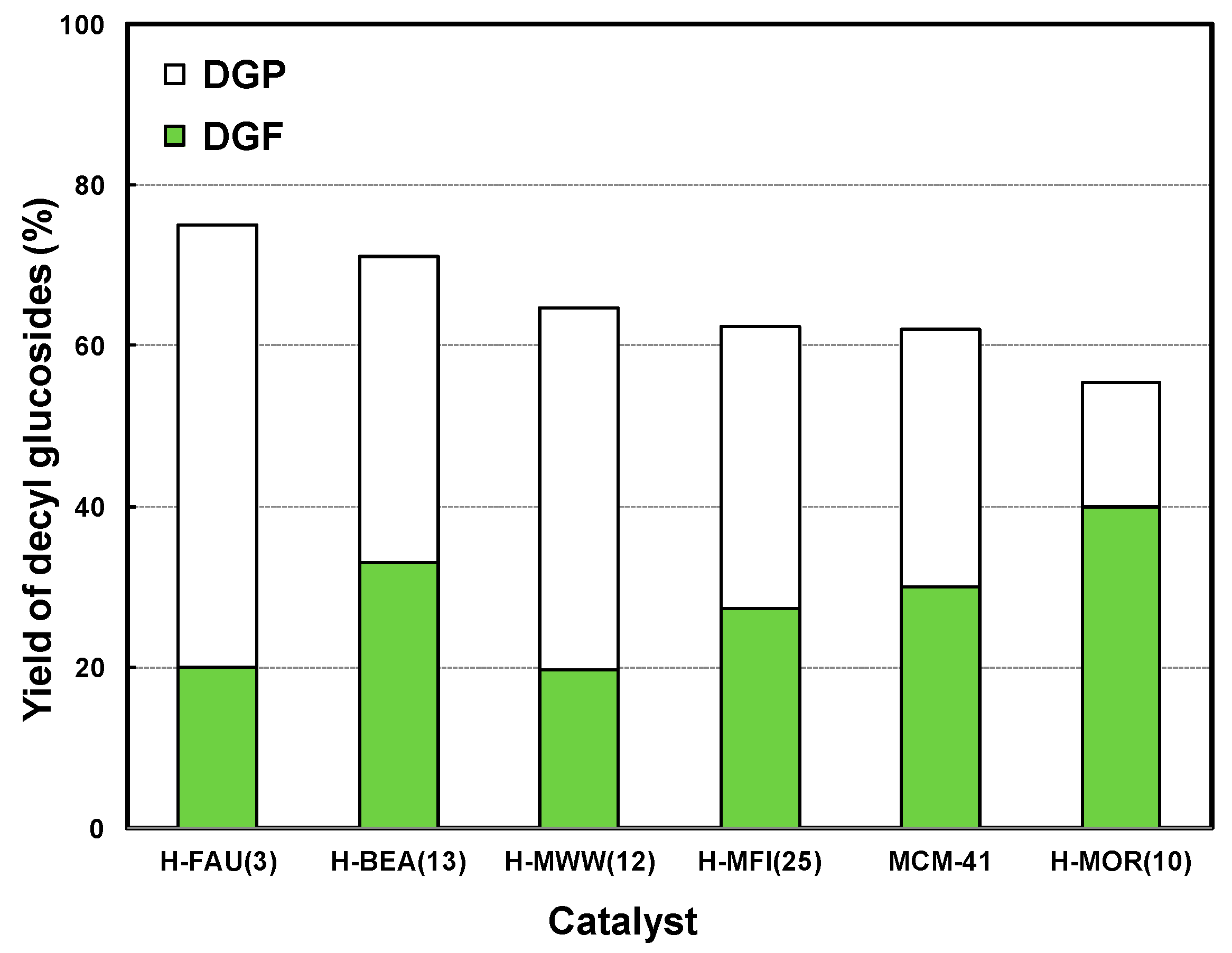
2.2. Catalytic Activities of the Zeolite Catalysts.
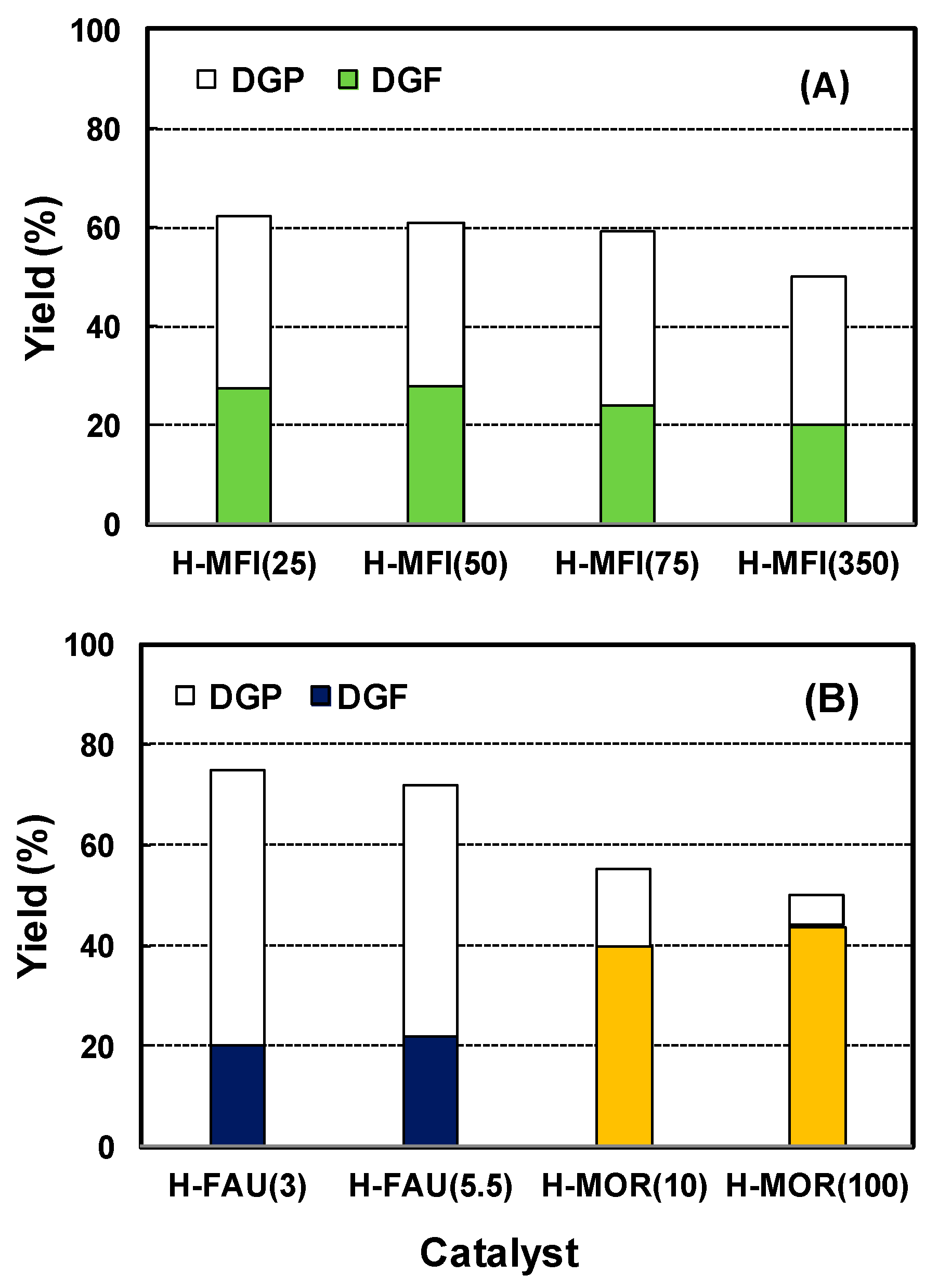
2.3. Effect of Acidic Properties of the Zeolite Catalysts
2.4. Effect of Pore Topologies of the Zeolite Catalysts
3. Materials and Methods
3.1. Catalysts
3.2. Direct Glucosidation Reaction
3.3. Characterization of the Zeolite Catalysts and Products
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Scott, M.; Jones, M.N. The biodegradation of surfactants in the environment. Biochim. Biophys. Acta 2000, 1508, 235–251. [Google Scholar] [CrossRef]
- Soares, A.; Guieysse, B.; Jefferson, B.; Cartmell, E.; Lester, J. Nonylphenol in the environment: A critical review on occurrence, fate, toxicity and treatment in wastewaters. Environ. Int. 2008, 34, 1033–1049. [Google Scholar] [CrossRef] [PubMed]
- Dodds, E.C.; Lawson, W. Molecular structure in relation to oestrogenic activity. Compounds without a phenanthrene nucleus. Proc. R. Soc. Lond. B 1938, 125, 222–232. [Google Scholar] [CrossRef]
- Soto, A.M.; Justicia, H.; Wray, J.; Sonnenschein, C. p-Nonyl-phenol: An estrogenic xenobiotic released from “modified” polystyrene. Environ. Health Perspect. 1991, 92, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Chattopadhyay, S.; Gong, E.Y.; Ahn, R.S.; Lee, K. Antiandrogenic effects of bisphenol A and nonylphenol on the function of androgen receptor. Toxicol. Sci. 2003, 75, 40–46. [Google Scholar] [CrossRef] [PubMed]
- De Nijs, P.M.; Maat, L.; Kieboom, A.P.G. Two-step chemo-enzymatic synthesis of octyl 6-O-acyl-α-d-glucopyranoside surfactants from glucose. Recl. Trav. Chim. Pays-Bas 1990, 109, 429–433. [Google Scholar] [CrossRef]
- Lee, S.M.; Lee, J.Y.; Yu, H.P.; Lim, J.C. Synthesis of environment friendly nonionic surfactants from sugar base and characterization of interfacial properties for detergent application. J. Ind. Eng. Chem. 2016, 38, 157–166. [Google Scholar] [CrossRef]
- Ferlin, N.; Duchet, L.; Kovensky, J.; Grand, E. Microwave-assisted synthesis of long-chain alkyl glucopyranosides. Carbohydr. Res. 2008, 343, 2819–2821. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.; von Rybinski, W.; Stoll, G. Alkyl Polyglucosides: Technology, Properties and Applications; VCH: Weinheim, Germany, 1997. [Google Scholar]
- Hurford, J.R.; Lee, C.K. Developments in Food Carbohydrate 2; Lee, C.K., Ed.; Applied Science Publishers Ltd.: London, UK, 1980; pp. 327–350. [Google Scholar]
- Hughes, F.A.; Lew, B.W.; Am, J. Physical and functional properties of some higher alkyl polyglucosides. Chem. Soc. 1970, 47, 162–167. [Google Scholar] [CrossRef]
- Straathof, A.J.J.; van Bekkum, H.; Kieboom, A.P.G. Efficient preparation of octyl α-d-glucopyranoside monohydrate: A recirculation procedure involving water removal by product crystallization. Starch-Stärke 1998, 40, 229–234. [Google Scholar] [CrossRef]
- Le Coz, C.J.; Meyer, M.T. Contact allergy to decyl glucoside in antiseptic after body piercing. Contact Dermat. 2003, 48, 279–280. [Google Scholar] [CrossRef]
- Andersen, K.E.; Goossens, A. Decyl glucoside contact allergy from a sunscreen product. Contact Dermat. 2006, 54, 349–350. [Google Scholar] [CrossRef] [PubMed]
- Andrade, P.; Goncalo, M.; Figueiredo, A. Allergic contact dermatitis to decyl glucoside in Tinosorb M®. Contact Dermat. 2010, 62, 119–120. [Google Scholar] [CrossRef] [PubMed]
- Blondeel, A. Contact allergy to the mild surfactant decylglucoside. Contact Dermat. 2004, 49, 304–305. [Google Scholar] [CrossRef] [PubMed]
- Horn, H.M.; Murray, C.; Aldridge, R.D. Contact allergy to decyl glucoside. Contact Dermat. 2005, 52, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Krehic, M.; Avenel-Audran, M. Allergic contact dermatitis from decyl glucoside in an antiseptic lotion. Contact Dermat. 2009, 61, 349–350. [Google Scholar] [CrossRef] [PubMed]
- Ludot, C.; Estrine, B.; Le Bras, J.; Hoffmann, N.; Marinkovic, S.; Muzart, J. Sulfoxides and sulfones as solvents for the manufacture or alkyl polyglycosides without added catalyst. Green Chem. 2013, 15, 3027–3030. [Google Scholar] [CrossRef]
- Van Es, D.S.; Marinkovic, S.; Oduber, X.; Estrine, B. Use of furandicarboxylic acid and its decyl ester as additives in the Fischer’s glycosylation of decanol by d-glucose: Physicochemical properties of the surfactant compositions obtained. J. Surfactant Deterg. 2013, 16, 147–154. [Google Scholar] [CrossRef]
- McCurry, P.M., Jr.; Pickens, C.E. Process for Preparation of Alkylglycosides. U.S. Patent 4,950,743, 21 August 1990. [Google Scholar]
- Mazumder, N.A.; Rano, R.; Sarmah, G. A green and efficient solid acid catalyst from coal fly ash for Fischer esterification reaction. J. Ind. Eng. Chem. 2015, 32, 211–217. [Google Scholar] [CrossRef]
- Boettner, F.E. Alkylpolyalkoxyalkyl Glucosides and Process of Preparation Therefor. U.S. Patent 3,219,656, 23 November 1965. [Google Scholar]
- Corma, A.; Iborra, S.; Miquel, S.; Primo, J. Preparation of long-chain alkyl glucoside surfactants by one-step direct Fischer glucosidation, and by transacetalation of butyl glucosides on beta zeolite catalysts. J. Catal. 1998, 180, 218–224. [Google Scholar] [CrossRef]
- Roboson, H.; Lillerud, K.P. Verified Syntheses of Zeolitic Materials, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2001; pp. 225–227. [Google Scholar]
- Cerqueira, H.S.; Ayrault, P.; Darka, J.; Magnoux, P.; Guisnet, M. Influence of coke on the acid properties of a USHY zeolite. Micropor. Mesoporous Mater. 2000, 38, 197–205. [Google Scholar] [CrossRef]
- Kadata, N.; Igi, H.; Kim, J.-H.; Niwa, M. Determination of the acidic properties of zeolite by theoretical analysis of temperature-programmed desorption of ammonia based on adsorption equilibrium. J. Phys. Chem. B 1997, 101, 5969–5977. [Google Scholar] [CrossRef]
- Niwa, M.; Kadata, N.; Sawa, M.; Murakami, Y. Temperature-programmed desorption of ammonia with readsorption based on the derived theoretical equation. J. Phys. Chem. B 1995, 99, 8812–8816. [Google Scholar] [CrossRef]
- Marques, J.P.; Gener, I.; Ayraut, P.; Bordado, J.C.; Lopes, J.M.; Ribeiro, F.R.; Guisnet, M. Dealumination of HBEA zeolite by steaming and acid leaching: Distribution of the various aluminic species and identification of the hydroxyl groups. R. C. Chim. 2005, 8, 399–410. [Google Scholar] [CrossRef]
- Cerqueira, H.S.; Ayrault, P.; Datka, J.; Magnoux, P.; Guisnet, M. m-Xylene transformation over a USHY zeolite at 523 and 723 K: Influence of coked deposits on activity, acidity and porosity. J. Catal. 2000, 196, 149–157. [Google Scholar] [CrossRef]
- Kotrel, S.; Rosynek, M.P.; Lunsford, J.H. Quantification of acid sites in H-ZSM-5, H-β, and H-Y Zeolites. J. Catal. 1999, 182, 278–281. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Katada, N.; Niwa, M. Acidity of β zeolite with different Si/Al2 ratio as measured by temperature programmed desorption of ammonia. Micropor. Mesoporous Mater. 2000, 40, 271–281. [Google Scholar] [CrossRef]
- Lonyi, F.; Valyon, J. On the interpretation of the NH3-TPD patterns of H-ZSM-5 and H-mordenite. Micropor. Mesoporous Mater. 2001, 47, 293–301. [Google Scholar] [CrossRef]
- Bennet, A.J.; Sinnott, M.L.; Wijesundera, W.S. 18O and secondary 2H kinetic isotope effects confirm the existence of two pathways for acid-catalyzed hydrolyses of α-arabinofuranosides. J. Chem. Soc. Perkin Trans. 1985, 2, 1233–1236. [Google Scholar] [CrossRef]
- Farris, D.D. Process for preparation of alkyl glycosides. European Patent 0096917, 28 December 1983. [Google Scholar]
- Thiem, J.; Böocker, T. Alkyl glycoside surfactants-synthesis and properties. Spec. Publ. R. Soc. Chem. 1992, 107, 123–147. [Google Scholar]
- Weitkemp, J.; Hunger, M. Acid and base catalysis on zeolites. In Studies in Surface Science and Catalysis; Cejka, J., van Bekkum, H., Corma, A., Schuth, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2007; Chapter 22. [Google Scholar]
- Yue, C.; Xie, W.; Liu, Y.; Wu, H.; Li, X.; Wu, P. Hydrothermal synthesis of MWW-type analogues using linear-type quaternary alkylammonium hydroxides as structure-directing agents. Micropor. Mesoporous Mater. 2011, 142, 347–353. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Williams, R.T. Physisorption hysteresis loops and the characterization of nanoporous materials. Adsorpt. Sci. Technol. 2004, 22, 773–782. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]

| Zeolite | Si/Al Molar Ratio (-) | Pore Topology (MR) | Pore Size (Å) | Pore Volume (cm3/g) | BET Surface Area (m2/g) | Cage Volume a (Å3) |
|---|---|---|---|---|---|---|
| MFI | 25 | 10 | 5.1 × 5.5[100] | 0.12 | 413 | - |
| 5.3 × 5.6[010] | ||||||
| MOR | 10 | 12 | 6.5 × 7.0[001] | 0.13 | 419 | - |
| 8 | 2.6 × 5.7[001] | |||||
| FAU | 3 | 12 | 7.4 × 7.4[111] | 0.24 | 589 | 1150 |
| BEA | 13 | 12 | 6.6 × 6.7[100] | 0.19 | 577 | - |
| 5.6 × 5.6[001] | ||||||
| MWW | 12 | 10 | 5.6 × 5.6[001] | 0.15 | 420 | - |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, K.-H.; Park, H.; Jeon, K.-J.; Park, Y.-K.; Jung, S.-C. Microporous Zeolites as Catalysts for the Preparation of Decyl Glucoside from Glucose with 1-Decanol by Direct Glucosidation. Catalysts 2016, 6, 216. https://doi.org/10.3390/catal6120216
Chung K-H, Park H, Jeon K-J, Park Y-K, Jung S-C. Microporous Zeolites as Catalysts for the Preparation of Decyl Glucoside from Glucose with 1-Decanol by Direct Glucosidation. Catalysts. 2016; 6(12):216. https://doi.org/10.3390/catal6120216
Chicago/Turabian StyleChung, Kyong-Hwan, Hyunwoong Park, Ki-Joon Jeon, Young-Kwon Park, and Sang-Chul Jung. 2016. "Microporous Zeolites as Catalysts for the Preparation of Decyl Glucoside from Glucose with 1-Decanol by Direct Glucosidation" Catalysts 6, no. 12: 216. https://doi.org/10.3390/catal6120216
APA StyleChung, K.-H., Park, H., Jeon, K.-J., Park, Y.-K., & Jung, S.-C. (2016). Microporous Zeolites as Catalysts for the Preparation of Decyl Glucoside from Glucose with 1-Decanol by Direct Glucosidation. Catalysts, 6(12), 216. https://doi.org/10.3390/catal6120216