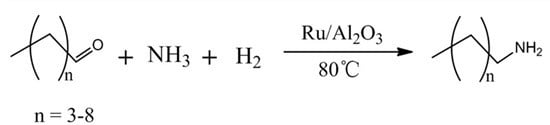
Heterogeneous Ru-Based Catalysts for One-Pot Synthesis of Primary Amines from Aldehydes and Ammonia
Abstract
:1. Introduction
2. Results and Discussion
2.1. Influence of Support on the Catalytic Performance
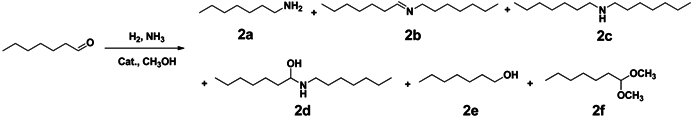 | ||||||||
|---|---|---|---|---|---|---|---|---|
| Entry | Catalysts | Con. (%) | Yield (%) b | |||||
| 2a | 2b | 2c | 2d | 2e | 2f | |||
| 1 | Ru/γ-Al2O3 | >99 | 94 | 0.1 | 0.5 | 0.8 | 0.2 | - |
| 2 | Ru/θ-Al2O3 | >99 | 92 | 1.0 | 0.8 | 0.8 | 0.5 | - |
| 3 | Ru/α-Al2O3 | >99 | 66 | 23 | 0.6 | 0.9 | 0.2 | - |
| 4 | Ru/CaO | >99 | 5 | 67 | <0.1 | 1.1 | 0.2 | - |
| 5 | Ru/MgO | >99 | 53 | 12 | 0.3 | 1.1 | 0.3 | - |
| 6 | Ru/ZrO2 | >99 | 90 | 1.0 | 2.2 | 0.9 | <0.1 | - |
| 7 | Ru/CeO2 | >99 | 70 | 0.3 | 0.2 | 1.1 | 0.1 | 15 |
| 8 | Ru/Nb2O5 | >99 | 18 | 28 | 0.3 | 1 | 0.3 | - |
| 9 | Ru/SnO2 | >99 | 0 | 17 | 0.5 | 2.9 | 20 | - |
| 10 | Ru/ZSM-5 | >99 | 9 | 57 | 0.1 | 1.6 | 7 | - |
| 11 | Ru/MCM-41 | >99 | 9 | 77 | <0.1 | 0.9 | 0.4 | - |
2.2. Effect of Various Solvents on the Reaction
| Entry | Solvent | Yield (%) b |
|---|---|---|
| 1 | methanol | 94 |
| 2 | toluene | 84 |
| 3 | THF | 84 |
| 4 | cyclohexane | 72 |
| 5 | 1,4-dioxane | 39 |
| 6 | MTBE | 34 |
| 7 | H2O | 32 |
2.3. Reaction Pathways

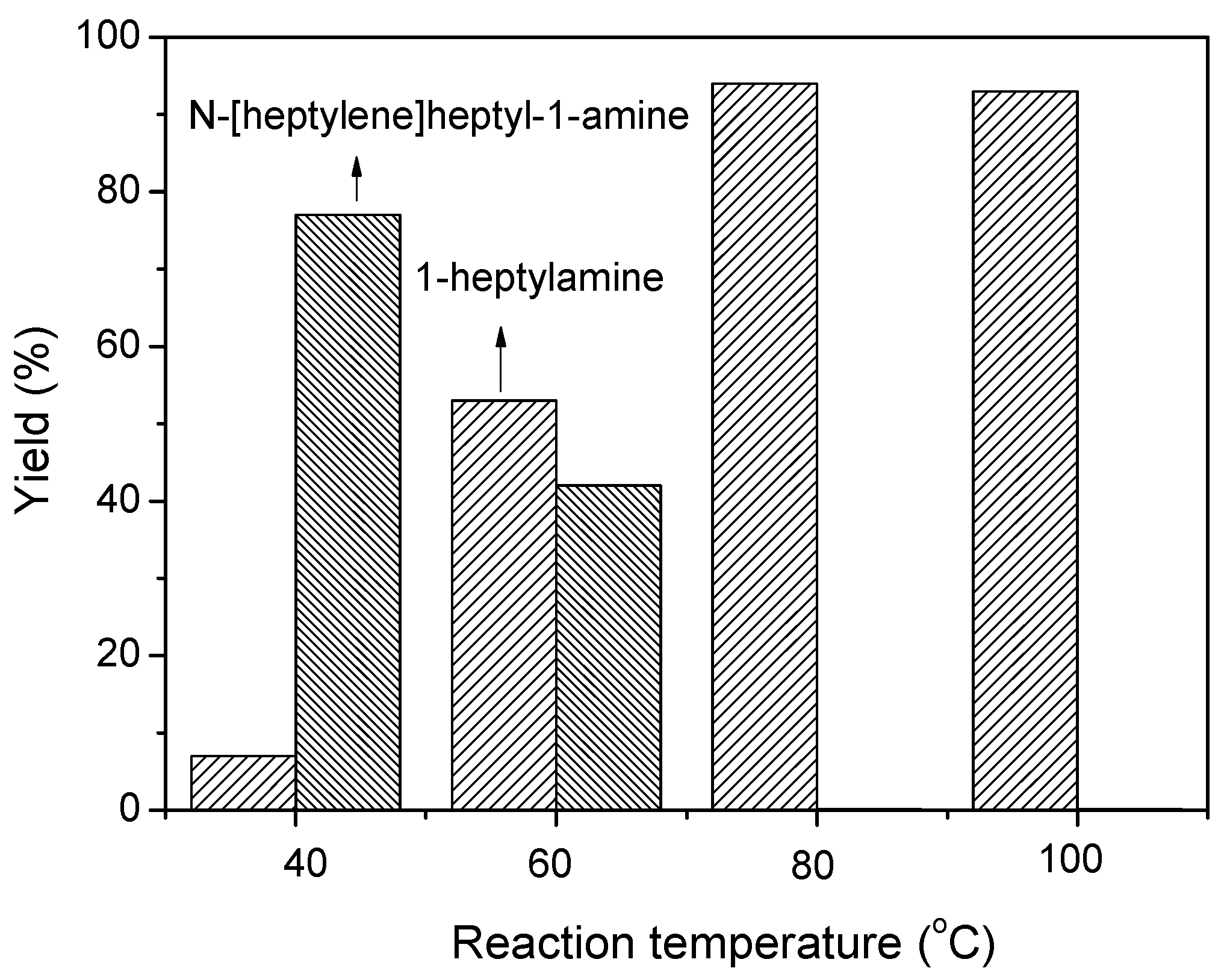
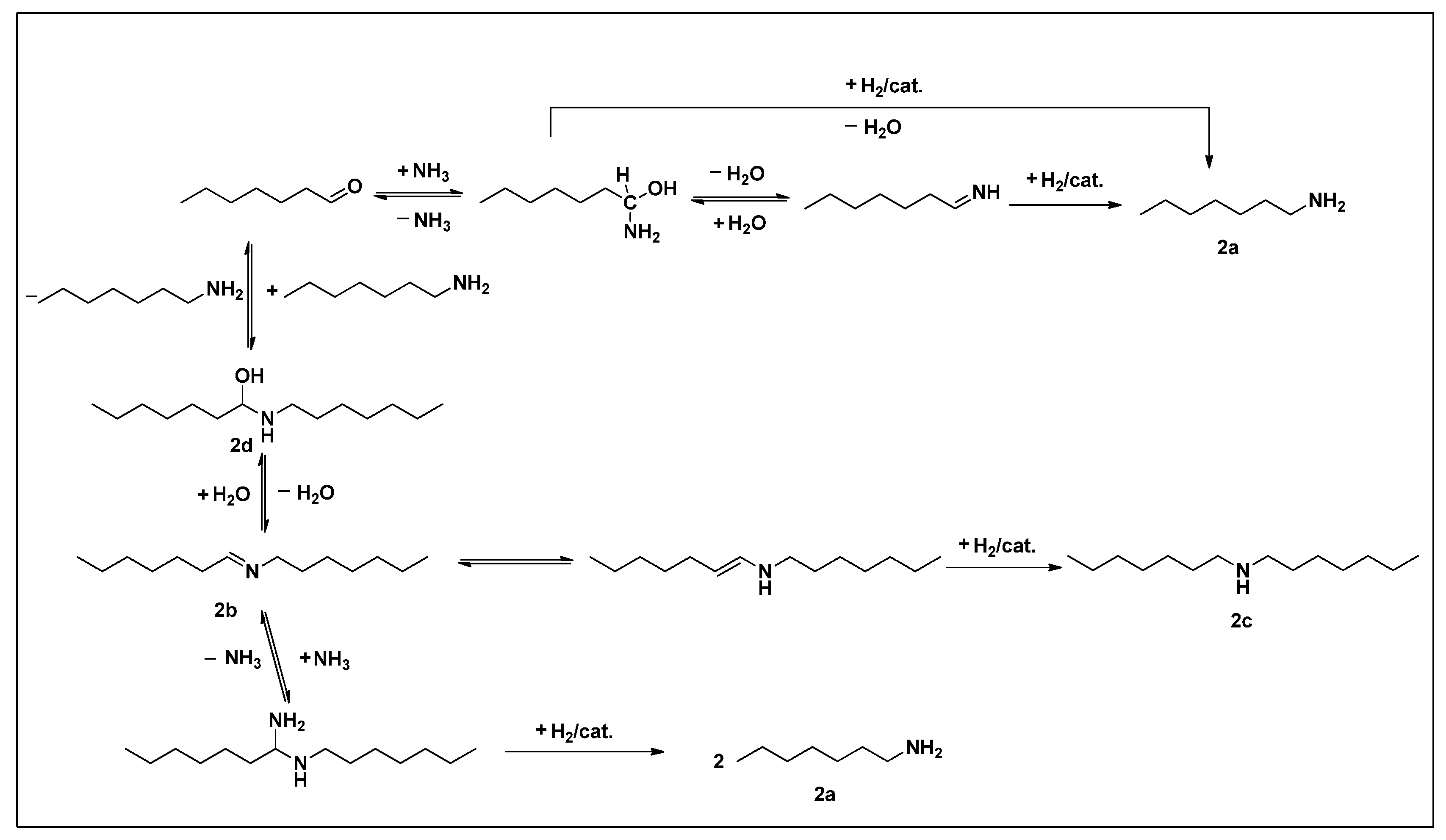
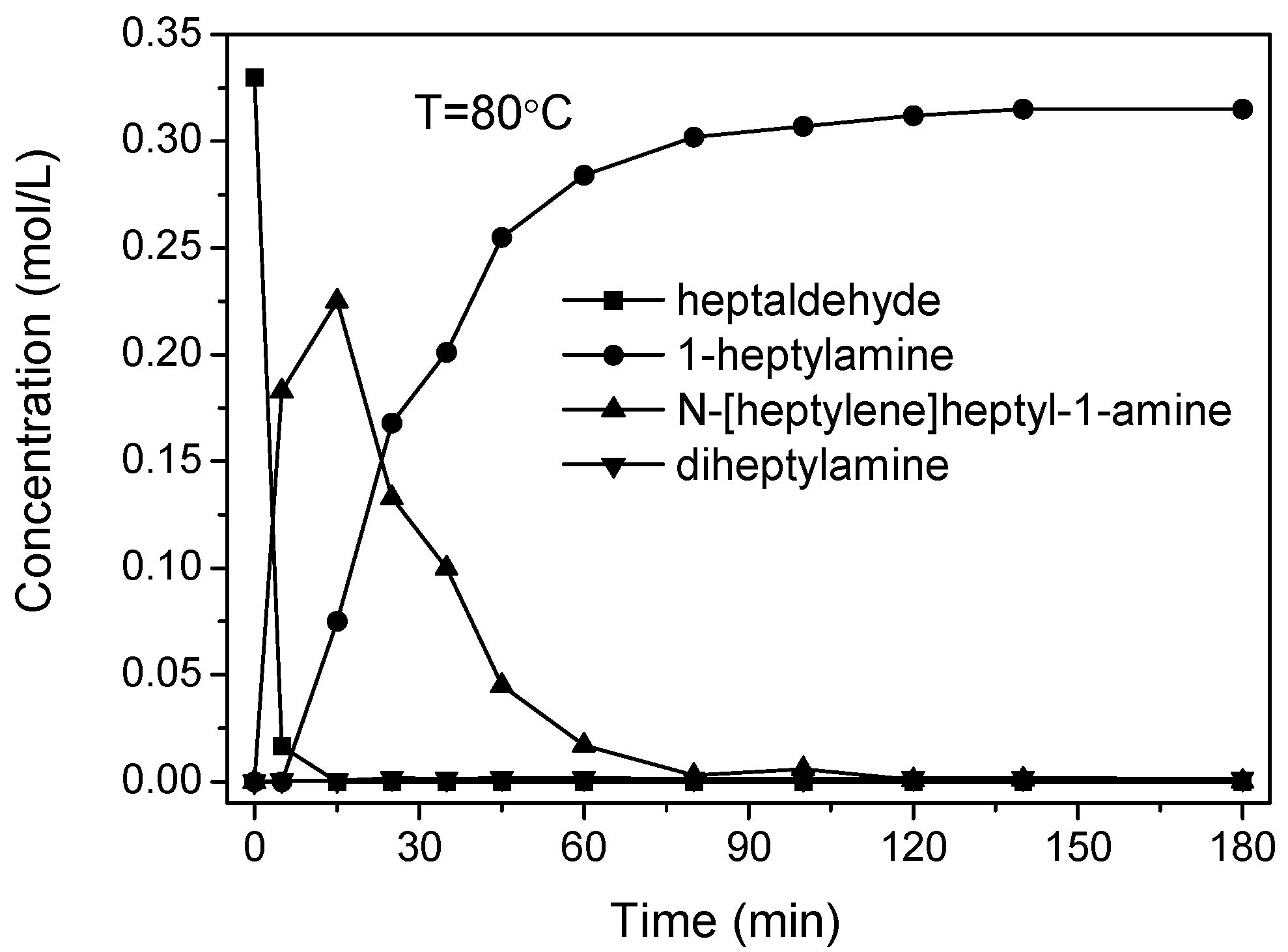
2.4. Scope of Reactions
| Entry | Substrate | Product | Yield (%) b |
|---|---|---|---|
| 1 |  |  | 94 |
| 2 |  |  | 92 |
| 3 |  | 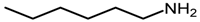 | 89 |
| 4 | 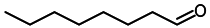 |  | 92 |
| 5 | 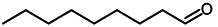 |  | 95 |
| 6 |  |  | 95 |
| 7 c | 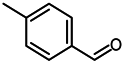 | 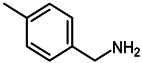 | 97 |
| 8 d | 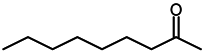 | 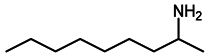 | 97 |
| 9 | 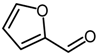 | 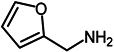 | 75 |
2.5. Recycle Study of Catalyst
3. Experimental Section
3.1. Catalyst Materials and Preparation
3.2. Characterization
3.3. General Procedure of Catalytic Reactions
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bähn, S.; Imm, S.; Neubert, L.; Zhang, M.; Neumann, H.; Beller, M. The catalytic amination of alcohols. ChemCatChem 2011, 3, 1853–1864. [Google Scholar] [CrossRef]
- Nador, F.; Moglie, Y.; Ciolino, A.; Pierini, A.; Dorn, V.; Yus, M.; Alonso, F.; Radivoy, G. Direct reductive amination of aldehydes using lithium-arene(cat.) as reducing system. A simple one-pot procedure for the synthesis of secondary amines. Tetrahedron Lett. 2012, 53, 3156–3160. [Google Scholar] [CrossRef]
- Ward, J.; Wohlgemuth, R. High-yield biocatalytic amination reactions in organic synthesis. Curr. Org. Chem. 2010, 14, 1914–1927. [Google Scholar] [CrossRef]
- Ekbote, S.; Gadge, S.; Bhanage, B. Polymer supported triphenylphosphine-palladium acetate complex PS-TPP-Pd(OAc)2 as a heterogeneous and reusable catalyst for indirect reductive amination of aldehydes. Catalysts 2014, 4, 289–298. [Google Scholar] [CrossRef]
- Willis, M.C. Palladium catalyzed coupling of ammonia and hydroxide with aryl halides: The direct synthesis of primary anilines and phenols. Angew. Chem. Int. Ed. 2007, 46, 3402–3404. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Hartwig, J.F. Palladium-catalyzed coupling of ammonia and lithium amide with aryl halides. J. Am. Chem. Soc. 2006, 128, 10028–10029. [Google Scholar] [CrossRef] [PubMed]
- Laval, S.; Dayoub, W.; Favre-Reguillon, A.; Berthod, M.; Demonchaux, P.; Mignani, G.; Lemaire, M. A mild and efficient method for the reduction of nitriles. Tetrahedron Lett. 2009, 50, 7005–7007. [Google Scholar] [CrossRef]
- Wakamatsu, T.; Inaki, H.; Ogawa, A.; Watanabe, M.; Ban, Y.; Wakamatsu, T.; Inaki, H.; Ogawa, A.; Watanabe, M.; Ban, Y. An efficient reduction of nitriles and amides to the corresposnding amines with tetra-n-butylammonium borohydride in dichloromethane. Heterocycles 1980. [Google Scholar] [CrossRef]
- Müller, T.E.; Beller, M. Metal-initiated amination of alkenes and alkynes. Chem. Rev. 1998, 98, 675–704. [Google Scholar] [CrossRef] [PubMed]
- Yin, B.L.; Hu, T.S.; Wu, Y.L. Hydroamination of olefin in a special conjugated spiroketal enol ether system, diastereoselective synthesis of amino-containing tonghaosu analogs. Tetrahedron Lett. 2004, 45, 2017–2021. [Google Scholar] [CrossRef]
- Klein, H.; Jackstell, R.; Kant, M.; Martin, A.; Beller, M. First catalytic hydroaminomethylation in supercritical ammonia. Chem. Eng. Technol. 2007, 30, 721–725. [Google Scholar] [CrossRef]
- Shimizu, K.I.; Kon, K.; Onodera, W.; Yamazaki, H.; Kondo, J.N. Heterogeneous Ni catalyst for direct synthesis of primary amines from alcohols and ammonia. ACS Catal. 2012, 3, 112–117. [Google Scholar] [CrossRef]
- Gunanathan, C.; Milstein, D. Selective synthesis of primary amines directly from alcohols and ammonia. Angew. Chem. Int. Ed. 2008, 47, 8661–8664. [Google Scholar] [CrossRef] [PubMed]
- Imm, S.; Bahn, S.; Neubert, L.; Neumann, H.; Beller, M. An efficient and general synthesis of primary amines by ruthenium-catalyzed amination of secondary alcohols with ammonia. Angew. Chem. Int. Ed. 2010, 49, 8126–8129. [Google Scholar] [CrossRef] [PubMed]
- Baiker, A.; Kijenski, J. Catalytic synthesis of higher aliphatic amines from the corresponding alcohols. Catal. Rev. 1985, 27, 653–697. [Google Scholar] [CrossRef]
- Hayes, K.S. Industrial processes for manufacturing amines. Appl. Catal. A 2001, 221, 187–195. [Google Scholar] [CrossRef]
- Werkmeister, S.; Junge, K.; Beller, M. Copper-catalyzed reductive amination of aromatic and aliphatic ketones with anilines using environmental-friendly molecular hydrogen. Green Chem. 2012, 14, 2371–2374. [Google Scholar] [CrossRef]
- Kim, J.; Kim, H.J.; Chang, S. Synthetic uses of ammonia in transition-metal catalysis. Eur. J. Org. Chem. 2013, 2013, 3201–3213. [Google Scholar] [CrossRef]
- Mignonac, G. Nouvelle méthodegénérale de préparation des amines à partir des aldéhydes ou des cétones. Comptes Rendus 1921, 172, 223–225. [Google Scholar]
- Riermeier, T.; Haack, K.; Dingerdissen, U.; Boerner, A.; Tararov, V.; Kadyrov, R. Method for Producing Amines by Homogeneously Catalyzed Reductive Amination of Carbonyl Compounds. U.S. Patent 6,884,887 B1, 26 April 2005. [Google Scholar]
- Gross, T.; Seayad, A.M.; Ahmad, M.; Beller, M. Synthesis of primary amines: First homogeneously catalyzed reductive amination with ammonia. Org. Lett. 2002, 4, 2055–2058. [Google Scholar] [CrossRef] [PubMed]
- Ogo, S.; Uehara, K.; Abura, T.; Fukuzumi, S. pH-Dependent chemoselective synthesis of α-amino acids. Reductive amination of α-keto acids with ammonia catalyzed by acid-stable iridium hydride complexes in water. J. Am. Chem. Soc. 2004, 12, 3020–3021. [Google Scholar] [CrossRef] [PubMed]
- Talwar, D.; Salguero, N.P.; Robertson, C.M.; Xiao, J. Primary amines by transfer hydrogenative reductive amination of ketones by using cyclometalated IrIII catalysts. Chemistry 2014, 20, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Menche, D.; Arikan, F.; Li, J.; Rudolph, S. Directed reductive amination of β-hydroxy-ketones: Convergent assembly of the ritonavir/lopinavir core. Org. Lett. 2007, 9, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Pei, D.; Zhang, Y.; Wang, C.; Sun, J. A facile one-pot process for the formation of hindered tertiary amines. Molecules 2012, 17, 5151–5163. [Google Scholar] [CrossRef] [PubMed]
- Miriyala, B.; Bhattacharyya, S.; Williamson, J.S. Chemoselective reductive alkylation of ammonia with carbonyl compounds: Synthesis of primary and symmetrical secondary amines. Tetrahedron 2004, 60, 1463–1471. [Google Scholar] [CrossRef]
- Menche, D.; Hassfeld, J.; Li, J.; Menche, G.; Ritter, A.; Rudolph, S. Hydrogen bond catalyzed direct reductive amination of ketones. Org. Lett. 2006, 8, 741–744. [Google Scholar] [CrossRef] [PubMed]
- Clinton, F. Sodium cyanoborohydride—A highly selective reducing agent for organic functional groups. Synthesis 1975, 3, 135–146. [Google Scholar]
- Hiroi, R.; Miyoshi, N.; Wada, M. Solvent-free one-pot reduction of imines generated in situ from aldehydes and aniline by tributyltin hydride on silica gel. Chem. Lett. 2002, 31, 274–275. [Google Scholar] [CrossRef]
- Ranu, B.C.; Majee, A.; Sarkar, A. One-pot reductive amination of conjugated aldehydes and ketones with silica gel and zinc borohydride. J. Org. Chem. 1998, 63, 370–373. [Google Scholar] [CrossRef]
- Dovell, F.S.; Greenfield, H. Base-metal sulfides as reductive alkylation catalysts. J. Org. Chem. 1964, 29, 1265–1267. [Google Scholar] [CrossRef]
- Winans, C.F. Method of Preparaing Secondary Amones. U.S. Patent 2,217,630, 8 October 1940. [Google Scholar]
- Bódis, J.; Lefferts, L.; Müller, T.E.; Pestman, R.; Lercher, J.A. Activity and selectivity control in reductive amination of butyraldehyde over noble metal catalysts. Catal. Lett. 2005, 104, 23–28. [Google Scholar] [CrossRef]
- Merger, F.; Otterbach, A.; Witzel, T.; Renz, H. 2,2-Dialkylpentane 1,5-Diisocyanates, 2,2-Dialkylpentane 1,5-Diurethanes and 2,2-Dialkylpentane 1,5-Dicarbamoyl Chlorides, and Their Preparation and Use. U.S. Patent 5,554,787 A1, 10 September 1996. [Google Scholar]
- Tamura, M.; Shimizu, K.I.; Satsuma, A. Comprehensive Ir study on acid/base properties of metal oxides. Appl. Catal. A 2012, 433, 135–145. [Google Scholar] [CrossRef]
- Busca, G. The surface acidity of solid oxides and its characterization by Ir spectroscopic methods. An attempt at systematization. Phys. Chem. Chem. Phys. 1999, 1, 723–736. [Google Scholar] [CrossRef]
- Martin, D.; Duprez, D. Evaluation of the acid-base surface properties of several oxides and supported metal catalysts by means of model reactions. J. Mol. Catal. A Chem. 1997, 118, 113–128. [Google Scholar] [CrossRef]
- Hoffmann, S.; Nicoletti, M.; List, B. Catalytic asymmetric reductive amination of aldehydes via dynamic kinetic resolution. J. Am. Chem. Soc. 2006, 128, 13074–13075. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, B.; Guo, X.; Zhang, B.; Chen, X.; Guan, J.; Qi, Y.; Han, S.; Mu, X. Heterogeneous Ru-Based Catalysts for One-Pot Synthesis of Primary Amines from Aldehydes and Ammonia. Catalysts 2015, 5, 2258-2270. https://doi.org/10.3390/catal5042258
Dong B, Guo X, Zhang B, Chen X, Guan J, Qi Y, Han S, Mu X. Heterogeneous Ru-Based Catalysts for One-Pot Synthesis of Primary Amines from Aldehydes and Ammonia. Catalysts. 2015; 5(4):2258-2270. https://doi.org/10.3390/catal5042258
Chicago/Turabian StyleDong, Bo, Xingcui Guo, Bo Zhang, Xiufang Chen, Jing Guan, Yunfei Qi, Sheng Han, and Xindong Mu. 2015. "Heterogeneous Ru-Based Catalysts for One-Pot Synthesis of Primary Amines from Aldehydes and Ammonia" Catalysts 5, no. 4: 2258-2270. https://doi.org/10.3390/catal5042258
APA StyleDong, B., Guo, X., Zhang, B., Chen, X., Guan, J., Qi, Y., Han, S., & Mu, X. (2015). Heterogeneous Ru-Based Catalysts for One-Pot Synthesis of Primary Amines from Aldehydes and Ammonia. Catalysts, 5(4), 2258-2270. https://doi.org/10.3390/catal5042258