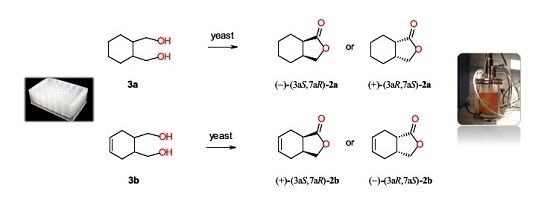
Microbial Stereoselective One-Step Conversion of Diols to Chiral Lactones in Yeast Cultures
Abstract
1. Introduction
2. Results and Discussion
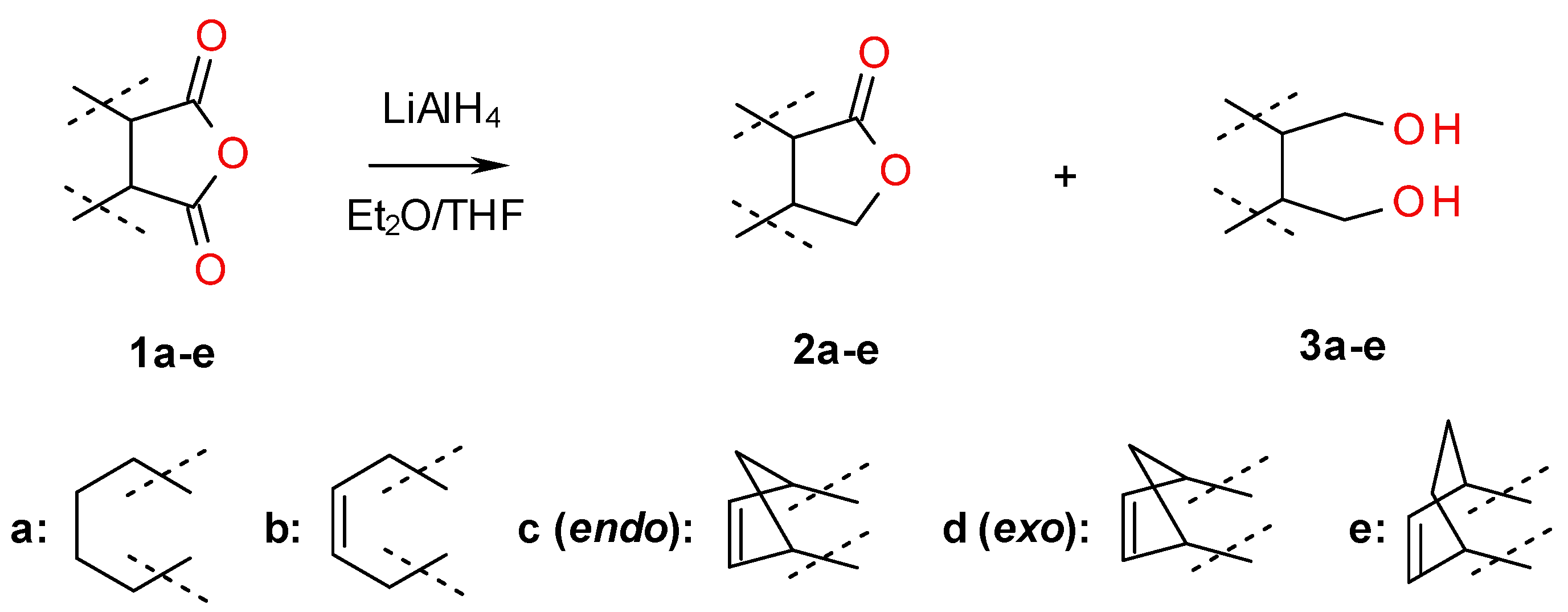
2.1. Synthesis of Diols 3a–f and Lactones 2a–f

2.2. Screening Scale Biotransformations of Monocyclic Meso Diols 3a–b

| Strain | Time (day) | Conversion of Diol 3a (%) | Lactone 2a | |
|---|---|---|---|---|
| ee (%) | Isomer | |||
| Candida pelliculosa ZP22 | 14 | 92 | 70 | (+)-(3aR,7aS) |
| Candida viswanathi AM120 | 21 | 9 | 0 | racemic |
| Saccharomyces cerevisiae AM464 | 21 | 20 | 95 | (+)-(3aR,7aS) |
| Saccharomyces pastorianus 906 | 21 | >99 | 0 | racemic |
| Yarrowia lipolytica AR71 | 21 | 60 | 68 | (+)-(3aR,7aS) |
| Yarrowia lipolytica AR72 | 21 | 44 | 58 | (+)-(3aR,7aS) |
| Rhodotorula glutinis AM242 | 14 | 20 | 50 | (–)-(3aS,7aR) |
| Rhodotorula marina 77 | 21 | 12 | 10 | (–)-(3aS,7aR) |
| Rhodotorula rubra AM82 | 21 | 28 | 6 | (–)-(3aS,7aR) |
| Rhodotorula rubra AM4 | 21 | 18 | 10 | (–)-(3aS,7aR) |
| Strain | Time (day) | Conversion of Diol 3b (%) | Lactone 2b | |
|---|---|---|---|---|
| ee (%) | Isomer | |||
| Candida pelliculosa ZP22 | 14 | >99 | 68 | (+)-(3aS,7aR) |
| Saccharomyces cerevisiae AM464 | 21 | >99 | 40 | (–)-(3aR,7aS) |
| Yarrowia lipolytica AR71 | 21 | >99 | 50 | (+)-(3aS,7aR) |
| Strain | Time | Lactone 2a | Lactone 2b | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH 4.5 | pH 7.2 | pH 8.5 | pH 4.5 | pH 7.2 | pH 8.5 | ||||||||
| day | (%) | ee (%) | (%) | ee (%) | (%) | ee (%) | (%) | ee (%) | (%) | ee (%) | (%) | ee (%) | |
| Candida pelliculosa ZP22 | 14 | >99 | 68 | >99 | 66 | 95 | 64 | >99 | 64 | >99 | 68 | >99 | 70 |
| Saccharomyces cerevisiae AM464 | 21 | 0 | - | 0 | - | 0 | - | >99 | 54 | >99 | 24 | >99 | 50 |
| Yarrowia lipolytica AR71 | 21 | 89 | 58 | 94 | 58 | 94 | 56 | 59 | 50 | 60 | 50 | 52 | 50 |
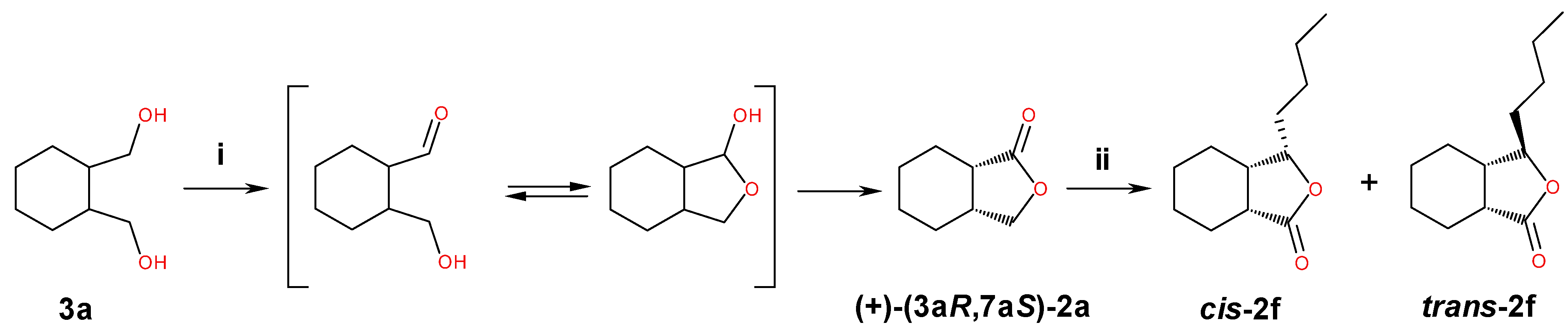
2.3. Preparative-Scale Biotransformations of Monocyclic Meso Diols 3a–b
2.4. Screening-Scale Biotransformations of Bicyclic Meso Diols 3c–e

| Strain | Conversion of Diol 3d (%) | Lactone 2d | |
|---|---|---|---|
| ee (%) | Isomer | ||
| Candida viswanathi AM120 | >99 | 64 | (+)-(3aR,4S,7R,7aS) |
| Saccharomyces pastorianus 906 | >99 | 50 | (+)-(3aR,4S,7R,7aS) |
| Yarrowia lipolytica AR71 | >99 | >99 | (+)-(3aR,4S,7R,7aS) |
| Rhodotorula glutinis AM242 | >99 | 54 | (+)-(3aR,4S,7R,7aS) |
| Rhodotorula rubra AM82 | 15 | 80 | (+)-(3aR,4S,7R,7aS) |
| Rhodotorula rubra AM4 | 15 | 76 | (+)-(3aR,4S,7R,7aS) |
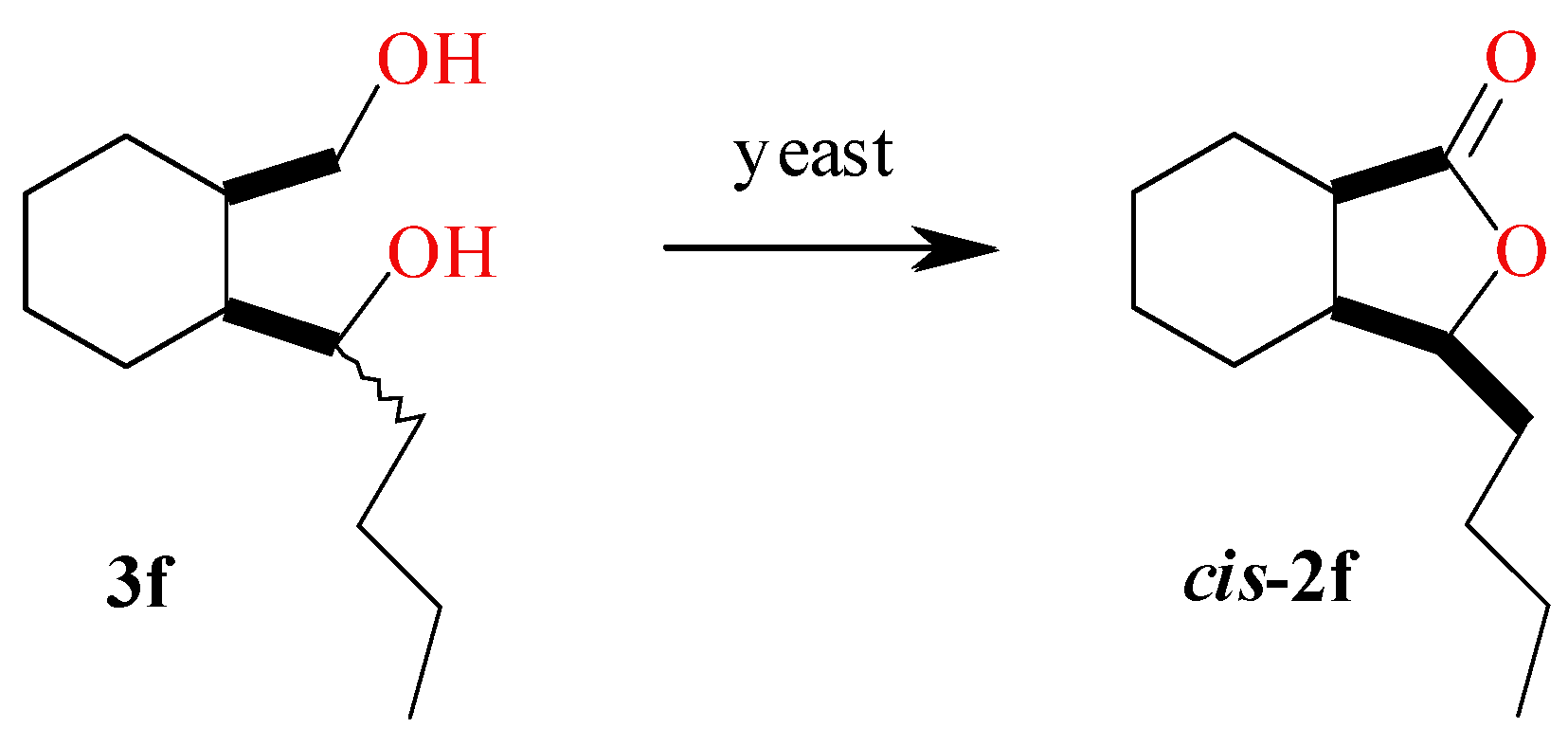
2.5. Screening Scale Biotransformations of Diol 3f
| Strain | Conversion of Diol 3f (%) | Lactone 2f ee (%) |
|---|---|---|
| Candida viswanathi AM120 | 11 | 21 |
| Candida sake AM908 | 18 | >99 |
| Candida parapsilosis AM909 | 22 | 38 |
| Yarrowia lipolytica AR71 | 4 | 62 |
| Rhodotorula marina 77 | 9 | 21 |
| Rhodotorula rubra AM82 | 11 | 98 |
| Rhodotorula rubra AM4 | 17 | >99 |
3. Experimental Section
3.1. Analysis
3.2. Chemicals
3.3. Synthesis of Meso Diols 3a–e and Lactones 2a–e
3.3.1. cis-Hexahydro-1(3H)-isobenzofuranone (±)-(2a)
3.3.2. cis-3a,4,7,7a-Tetrahydro-1(3H)-isobenzofuranone (±)-(2b)
3.3.3. cis-endo-3a,4,7,7a-Tetrahydro-4,7-methanoisobenzofuran-1(3H)-one (±)-(2c)
3.3.4. cis-exo-3a,4,7,7a-Tetrahydro-4,7-methanoisobenzofuran-1(3H)-one (±)-(2d)
3.3.5. cis-endo-3a,4,7,7a-Tetrahydro-4,7-ethanoisobenzofuran-1(3H)-one (±)-(2e)
3.4. Synthesis of Diol 3f and Lactone 2f
3.4.1. 1-(2-(Hydroxymethyl)cyclohexyl)pentan-1-ol (±)-(3f)
3.4.2. trans-3-Butylhexahydro-1(3H)-isobenzofuranone (±)-(2f)
3.5. Growth Conditions
- A:
- 40 g glucose, 15 g (NH4)3PO4, 7 g KH2PO4, 0.8 g MgSO4·7H2O, 0.1 g NaCl, 6 × 10−3 g ZnSO4·7H2O, 5 × 10−3 g CuSO4·5H2O, 1 × 10−3 g MnSO4·4H2O;
- C:
- 30 g saccharose, 3 g NaNO3, 1 g KH2PO4, 0.5 g MgSO4·7H2O, 0.5 g KCl, 0.01 g FeSO4;
- E:
- 10 g starch, 4 g yeast extract, 0.1 g K2HPO4, 0.05 g MgSO4·7H2O;
- G:
- 10 g glucose, 0.5 g asparagine, 0.5 g K2HPO4;
- M:
- 40 g glucose, 2 g asparagine, 0.5 g thiamine, 0.5 g KH2PO4, 0.25 g MgSO4·7H2O;
- P:
- 30 g glucose, 10 g peptone;
- S:
- 10 g glucose, 2.5 × 10−3 g genistein, 2.5 g K2HPO4, 2.5 g NaNO3.
3.6. Microorganisms
3.7. Biotransformations of Diols 3a–f
3.7.1. Screening-Scale Biotransformations in Microtiter Plates
3.7.2. Preparative-Scale Biotransformation in a Bioreactor
3.7.3. Preparative Oxidation of Meso Diols 3a–b Catalyzed by Candida pelliculosa ZP22
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Breuer, M.; Ditrich, K.; Habicher, T.; Hauer, B.; Keßeler, M.; Stürmer, R.; Zelinski, T. Industrial methods for the production of optically active intermediates. Angew. Chem. Int. Ed. 2004, 43, 788–824. [Google Scholar] [CrossRef] [PubMed]
- Muñoz Solano, D.; Hoyos, P.; Hernáiz, M.J.; Alcántara, A.R.; Sánchez-Montero, J.M. Industrial biotransformations in the synthesis of building blocks leading to enantiopure drugs. Bioresour. Technol. 2012, 115, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Nestl, B.M.; Nebel, B.A.; Hauer, B. Recent progress in industrial biocatalysis. Curr. Opin. Chem. Biol. 2011, 15, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Brenna, E.; Fuganti, C.; Gatti, F.G.; Serra, S. Biocatalytic methods for the synthesis of enantioenriched odor active compounds. Chem. Rev. 2011, 111, 4036–4072. [Google Scholar] [CrossRef] [PubMed]
- Muschiol, J.; Peters, C.; Oberleitner, N.; Mihovilovic, M.D.; Bornscheuer, U.T.; Rudroff, F. Cascade catalysis—Strategies and challenges en route to preparative synthetic biology. Chem. Commun. 2015, 51, 5798–5811. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Gallego, F.; Schmidt-Dannert, C. Multi-enzymatic synthesis. Curr. Opin. Chem. Biol. 2010, 14, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Deasy, R.E.; O’Riordan, N.; Maguire, A.R. Baker’s yeast mediated reduction of 2-acetyl-3-methyl sulfolane. Catalysts 2014, 4, 186–195. [Google Scholar] [CrossRef]
- Nakamura, K.; Yamanaka, R.; Matsuda, T.; Harada, T. Recent developments in asymmetric reduction of ketones with biocatalysis. Tetrahedron Asymmetr. 2003, 14, 2659–2681. [Google Scholar] [CrossRef]
- Brenna, E.; Dei Negri, C.; Fuganti, C.; Serra, S. Baker’s yeast-mediated approach to (−)-cis- and (+)-trans-aerangis lactones. Tetrahedron Asymmetr. 2001, 12, 1871–1879. [Google Scholar] [CrossRef]
- Sohoni, S.; Bapat, P.; Lantz, A. Robust, small-scale cultivation platform for Streptomyces coelicolor. Microb. Cell Fact. 2012, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Zang, R.; Zhang, X.; Yang, S.-T. A 24-microwell plate with improved mixing and scalable performance for high throughput cell cultures. Process Biochem. 2012, 47, 612–618. [Google Scholar] [CrossRef]
- Chen, A.; Chitta, R.; Chang, D.; Amanullah, A. Twenty-four well plate miniature bioreactor system as a scale-down model for cell culture process development. Biotechnol. Bioeng. 2009, 102, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.; Baganz, F. Miniature bioreactors: Current practices and future opportunities. Microb. Cell Fact. 2006. [Google Scholar] [CrossRef] [PubMed]
- Forchin, M.C.; Crotti, M.; Gatti, F.G.; Parmeggiani, F.; Brenna, E.; Monti, D. A rapid and high-throughput assay for the estimation of conversions of ene-reductase-catalysed reactions. ChemBioChem 2015, 16, 1571–1573. [Google Scholar] [CrossRef] [PubMed]
- Duetz, W. Microtiter plates as mini-bioreactors: Miniaturization of fermentation methods. Trends Microbiol. 2007, 15, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Duetz, W.; Witholt, B. Oxygen transfer by orbital shaking of square vessels and deepwell microtiter plates of various dimensions. Biochem. Eng. J. 2004, 17, 181–185. [Google Scholar] [CrossRef]
- Duetz, W.A.; Rüedi, L.; Hermann, R.; O’Connor, K.; Büchs, J.; Witholt, B. Methods for intense aeration, growth, storage, and replication of bacterial strains in microtiter plates. Appl. Environ. Microb. 2000, 66, 2641–2646. [Google Scholar] [CrossRef]
- Boratyński, F.; Kiełbowicz, G.; Wawrzeńczyk, C. Lactones 34 [1]. Application of alcohol dehydrogenase from horse liver (HLADH) in enantioselective synthesis of δ- and ɛ-lactones. J. Mol. Catal. B 2010, 65, 30–36. [Google Scholar] [CrossRef]
- Boratyński, F.; Pannek, J.; Walczak, P.; Janik-Polanowicz, A.; Huszcza, E.; Szczepańska, E.; Martinez-Rojas, E.; Olejniczak, T. Microbial alcohol dehydrogenase screening for enantiopure lactone synthesis: Down-stream process from microtiter plate to bench bioreactor. Process Biochem. 2014, 49, 1637–1646. [Google Scholar] [CrossRef]
- Boratyński, F.; Smuga, M.; Wawrzeńczyk, C. Lactones 42. Stereoselective enzymatic/microbial synthesis of optically active isomers of whisky lactone. Food Chem. 2013, 141, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Horn, M.; Martinez-Rojas, E.; Görisch, H.; Tressl, R.; Garbe, L.A. Oxidation of 1,4-alkanediols into g-lactones via g-lactols using Rhodococcus erythropolis as biocatalyst. J. Mol. Catal. B 2007, 49, 24–27. [Google Scholar] [CrossRef]
- Romano, A.; Gandolfi, R.; Nitti, P.; Rollini, M.; Molinari, F. Acetic acid bacteria as enantioselective biocatalysts. J. Mol. Catal. B 2002, 17, 235–240. [Google Scholar] [CrossRef]
- Jakovac, I.J.; Goodbrand, H.B.; Lok, K.P.; Jones, J.B. Enzymes in organic synthesis. 24. Preparations of enantiomerically pure chiral lactones via stereospecific horse liver alcohol dehydrogenase catalyzed oxidations of monocyclic meso diols. J. Am. Chem. Soc. 1982, 104, 4659–4665. [Google Scholar] [CrossRef]
- Lok, K.P.; Jakovac, I.J.; Jones, J.B. Enzymes in organic synthesis. 34. Preparations of enantiomerically pure exo- and endo-bridged bicyclic [2.2.1] and [2.2.2] chiral lactones via stereospecific horse liver alcohol dehydrogenase catalyzed oxidations of meso diols. J. Am. Chem. Soc. 1985, 107, 2521–2526. [Google Scholar] [CrossRef]
- Bridges, A.J.; Raman, P.S.; Ng, G.S.Y.; Jones, J.B. Enzymes in organic synthesis. 31. Preparations of enantiomerically pure bicyclic [3.2.1] and [3.3.1] chiral lactones via stereospecific horse liver alcohol dehydrogenase catalyzed oxidations of meso diols. J. Am. Chem. Soc. 1984, 106, 1461–1467. [Google Scholar] [CrossRef]
- Jones, J.B.; Francis, C.J. Enzymes in organic synthesis. 32. Stereospecyfic horse liver alcohol dehydrogenase—Catalyzed oxidations of exo- and endo-oxabicyclic meso diols. Can. J. Chem. 1984, 62, 2578–2582. [Google Scholar] [CrossRef]
- Olejniczak, T.; Boratyński, F.; Białońska, A. Fungistatic activity of bicycle [4.3.0]-g-lactones. J. Agric. Food Chem. 2011, 59, 6071–6081. [Google Scholar] [CrossRef] [PubMed]
- Soni, P.; Banerjee, U.C. Biotransformations for the production of the chiral drug (S)-duloxetine catalyzed by a novel isolate of Candida tropicalis. Appl. Microbiol. Biotechnol. 2005, 67, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, A.; Yamamoto, H.; Kawada, N.; Kobayashi, Y. Industrial production of (R)-1,3-butanediol by new biocatalysts. J. Mol. Catal. B 2001, 11, 513–521. [Google Scholar] [CrossRef]
- Dehli, J.R.; Gotor, V. Dynamic kinetic resolution of 2-oxocycloalkanecarbonitriles: Chemoenzymatic syntheses of optically active cyclic β- and γ-amino alcohols. J. Org. Chem. 2002, 67, 6816–6819. [Google Scholar] [CrossRef] [PubMed]
- Stuermer, R.; Hauer, B.; Hall, M.; Faber, K. Asymmetric bioreduction of activated C=C bonds using enoate reductases from the old yellow enzyme family. Curr. Opin. Chem. Biol. 2007, 11, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Kawai, Y.; Inaba, Y.; Tokitoh, N. Asymmetric reduction of nitroalkenes with baker’s yeast. Tetrahedron Asymmetr. 2001, 12, 309–318. [Google Scholar] [CrossRef]
- Serra, S.; Fuganti, C.; Gatti, F.G. A chemoenzymatic, preparative synthesis of the isomeric forms of p-menth-1-en-9-ol: Application to the synthesis of the isomeric forms of the cooling agent 1-hydroxy-2,9-cineole. Eur. J. Org. Chem. 2008, 1031–1037. [Google Scholar] [CrossRef]
- Fronza, G.; Fuganti, C.; Serra, S. Stereochemical course of baker's yeast mediated reduction of the tri- and tetrasubstituted double bonds of substituted cinnamaldehydes. Eur. J. Org. Chem. 2009, 2009, 6160–6171. [Google Scholar] [CrossRef]
- Sortino, M.A.; Filho, V.C.; Zacchino, S.A. Highly enantioselective reduction of the C–C double bond of N-phenyl-2-methyl- and N-phenyl-2,3-dimethyl-maleimides by fungal strains. Tetrahedron Asymmetr. 2009, 20, 1106–1108. [Google Scholar] [CrossRef]
- Csuk, R.; Glaenzer, B.I. Baker’s yeast mediated transformations in organic chemistry. Chem. Rev. 1991, 91, 49–97. [Google Scholar] [CrossRef]
- Glänzer, B.I.; Faber, K.; Griengl, H. Microbial resolution of o-acetylpantoyl lactone. Enzyme Microb. Technol. 1988, 10, 689–690. [Google Scholar] [CrossRef]
- Patel, R.N.; Hou, C.T.; Laskin, A.I.; Derelanko, P.; Felix, A. Oxidation of secondary alcohols to methyl ketones by yeasts. Appl. Environ. Microb. 1979, 38, 219–223. [Google Scholar]
- Nestl, B.; Voss, C.; Bodlenner, A.; Ellmer-Schaumberger, U.; Kroutil, W.; Faber, K. Biocatalytic racemization of sec-alcohols and α-hydroxyketones using lyophilized microbial cells. Appl. Microb. Biotechnol. 2007, 76, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Momin, R.A.; Nair, M.G. Mosquitocidal, nematicidal, and antifungal compounds from Apium graveolens L. seeds. J. Agric. Food Chem. 2001, 49, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Beck, J.J.; Chou, S.-C. The structural diversity of phthalides from the Apiaceae. J. Nat. Prod. 2007, 70, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Bartschat, D.; Beck, T.; Mosandl, A. Stereoisomeric flavor compounds. 79. Simultaneous enantioselective analysis of 3-butylphthalide and 3-butylhexahydro-phthalide stereoisomers in celery, celeriac, and fennel. J. Agric. Food Chem. 1997, 45, 4554–4557. [Google Scholar] [CrossRef]
- Walczak, P.; Pannek, J.; Boratyński, F.; Janik-Polanowicz, A.; Olejniczak, T. Synthesis and fungistatic activity of bicyclic lactones and lactams against Botrytis cinerea, Penicillium citrinum and Aspergillus glaucus. J. Agric. Food Chem. 2014, 62, 8571–8578. [Google Scholar] [CrossRef] [PubMed]
- Zaleska, I.; Piegza, M. Asymilacja nietypowych źródeł węgla przez mikroorganizmy o specyficznych uzdolnieniach do życia w wysoko stresogennych środowiskach. Acta Sci. Pol. Biotechnol. 2008, 7, 27–43. [Google Scholar]
- Le Guillou, R.; Fache, F.; Piva, O. Reductive alkylation of anhydrides and lactones: Direct access to monosubstituted lactones. C. R. Chim. 2002, 5, 571–575. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boratyński, F.; Szczepańska, E.; Pannek, J.; Olejniczak, T. Microbial Stereoselective One-Step Conversion of Diols to Chiral Lactones in Yeast Cultures. Catalysts 2015, 5, 2068-2084. https://doi.org/10.3390/catal5042068
Boratyński F, Szczepańska E, Pannek J, Olejniczak T. Microbial Stereoselective One-Step Conversion of Diols to Chiral Lactones in Yeast Cultures. Catalysts. 2015; 5(4):2068-2084. https://doi.org/10.3390/catal5042068
Chicago/Turabian StyleBoratyński, Filip, Ewa Szczepańska, Jakub Pannek, and Teresa Olejniczak. 2015. "Microbial Stereoselective One-Step Conversion of Diols to Chiral Lactones in Yeast Cultures" Catalysts 5, no. 4: 2068-2084. https://doi.org/10.3390/catal5042068
APA StyleBoratyński, F., Szczepańska, E., Pannek, J., & Olejniczak, T. (2015). Microbial Stereoselective One-Step Conversion of Diols to Chiral Lactones in Yeast Cultures. Catalysts, 5(4), 2068-2084. https://doi.org/10.3390/catal5042068