Synthesis, Characterization and Catalytic Polymerization of N-Methyl Imidazolium Phosphotungstic Catalyst
Abstract
:1. Introduction
2. Results and Discussion
2.1. IR Analysis
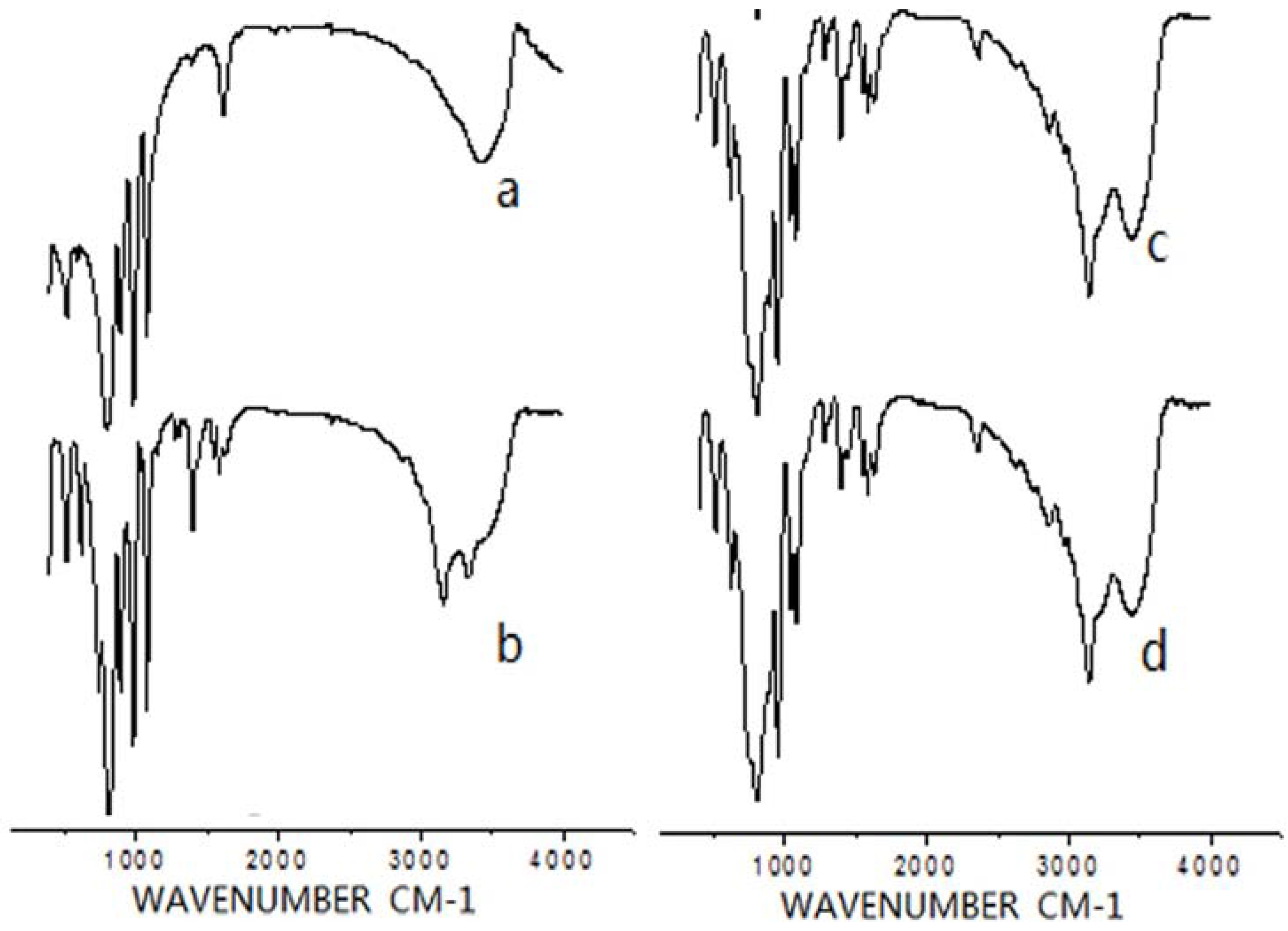
2.2. 1H NMR Analysis
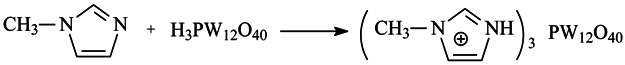
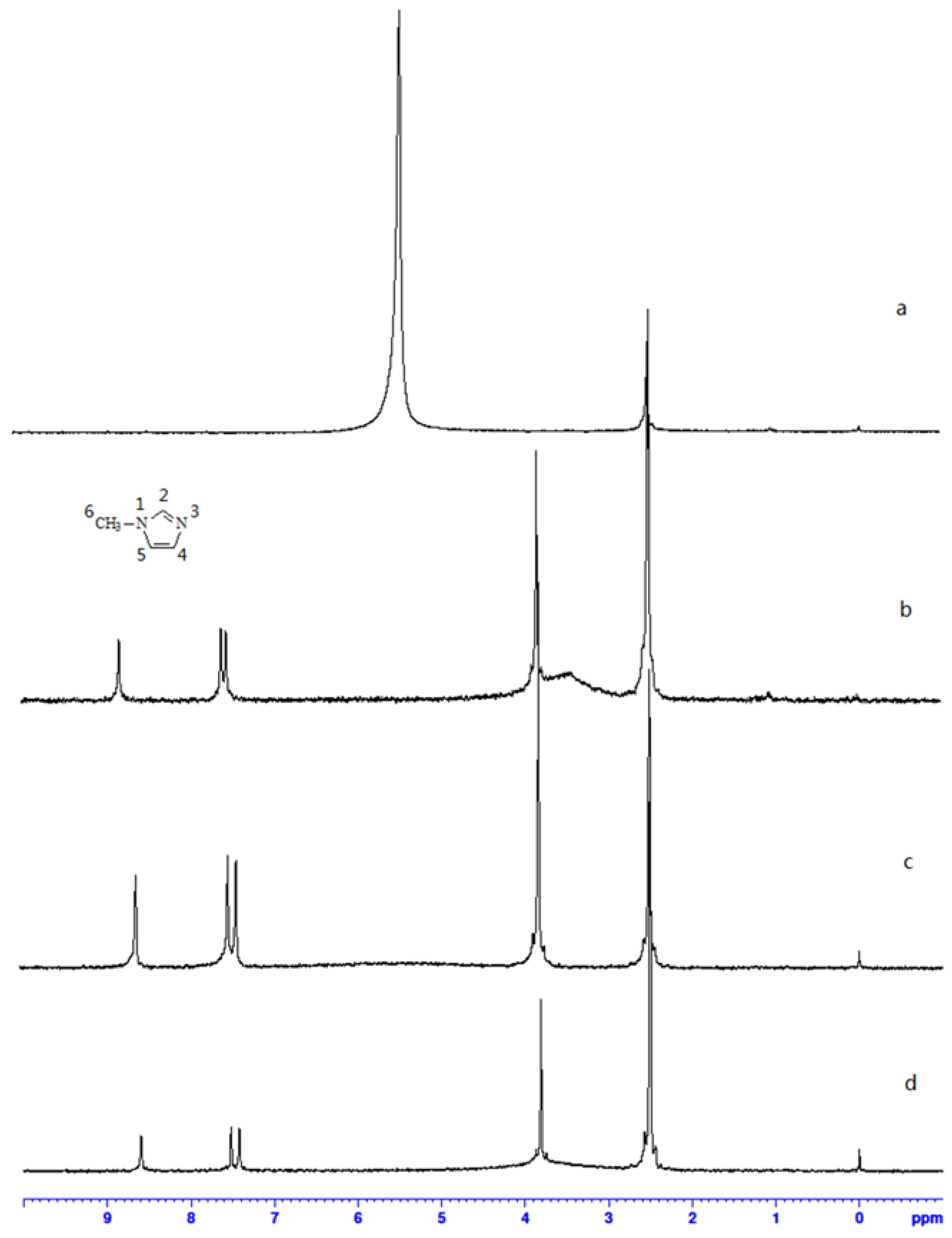
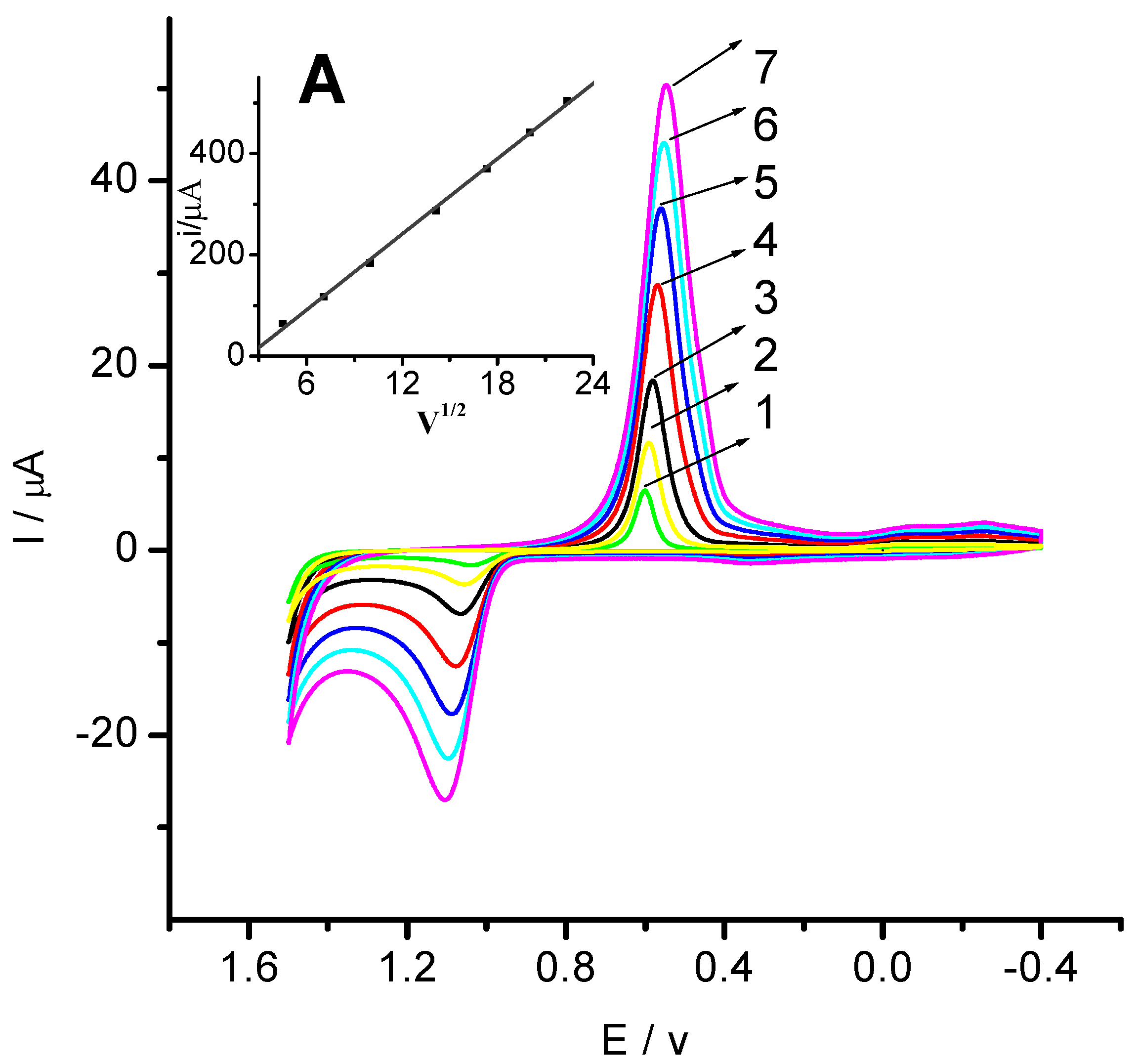
2.3. Electro-Chemical Property Analysis
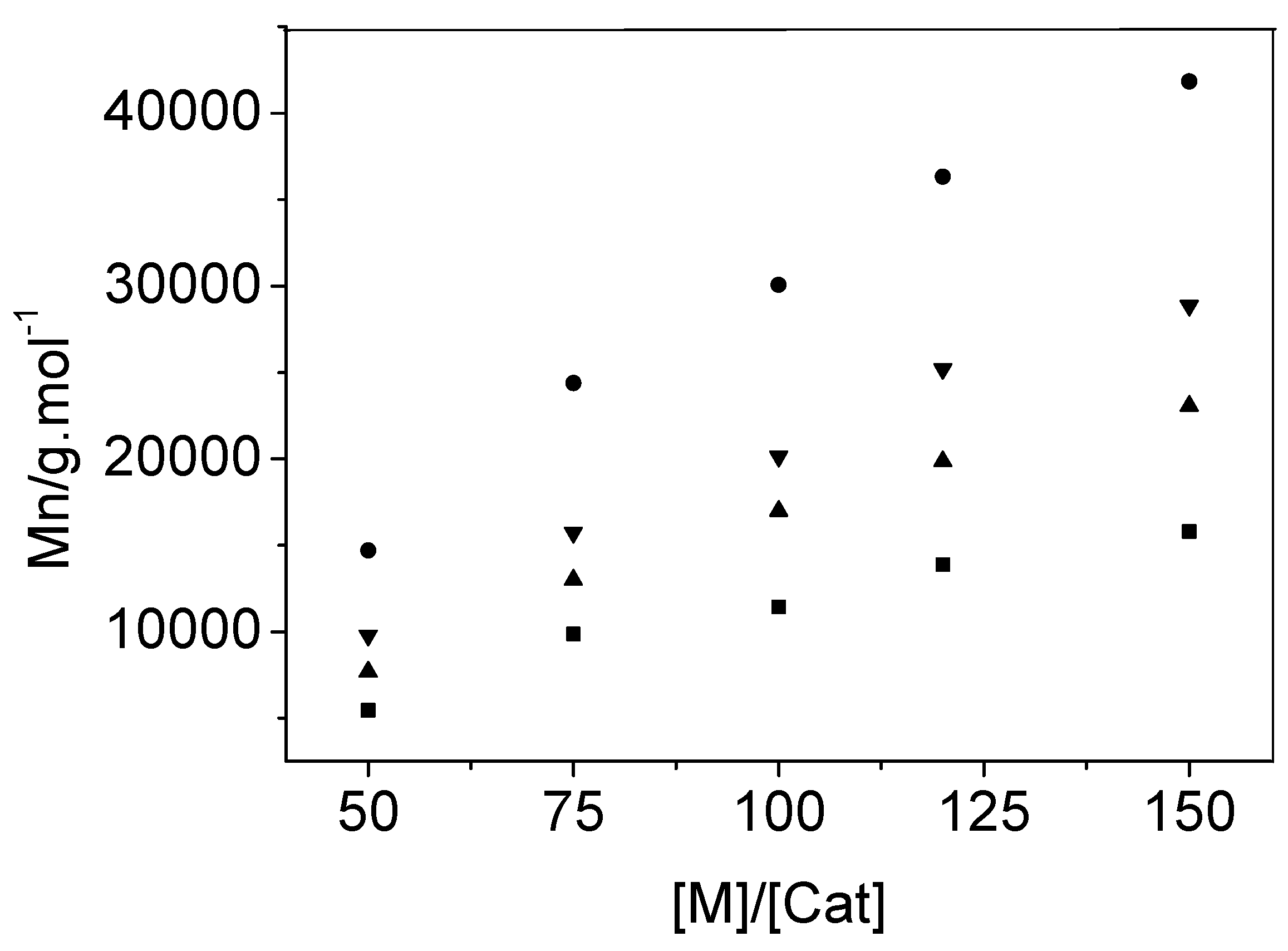
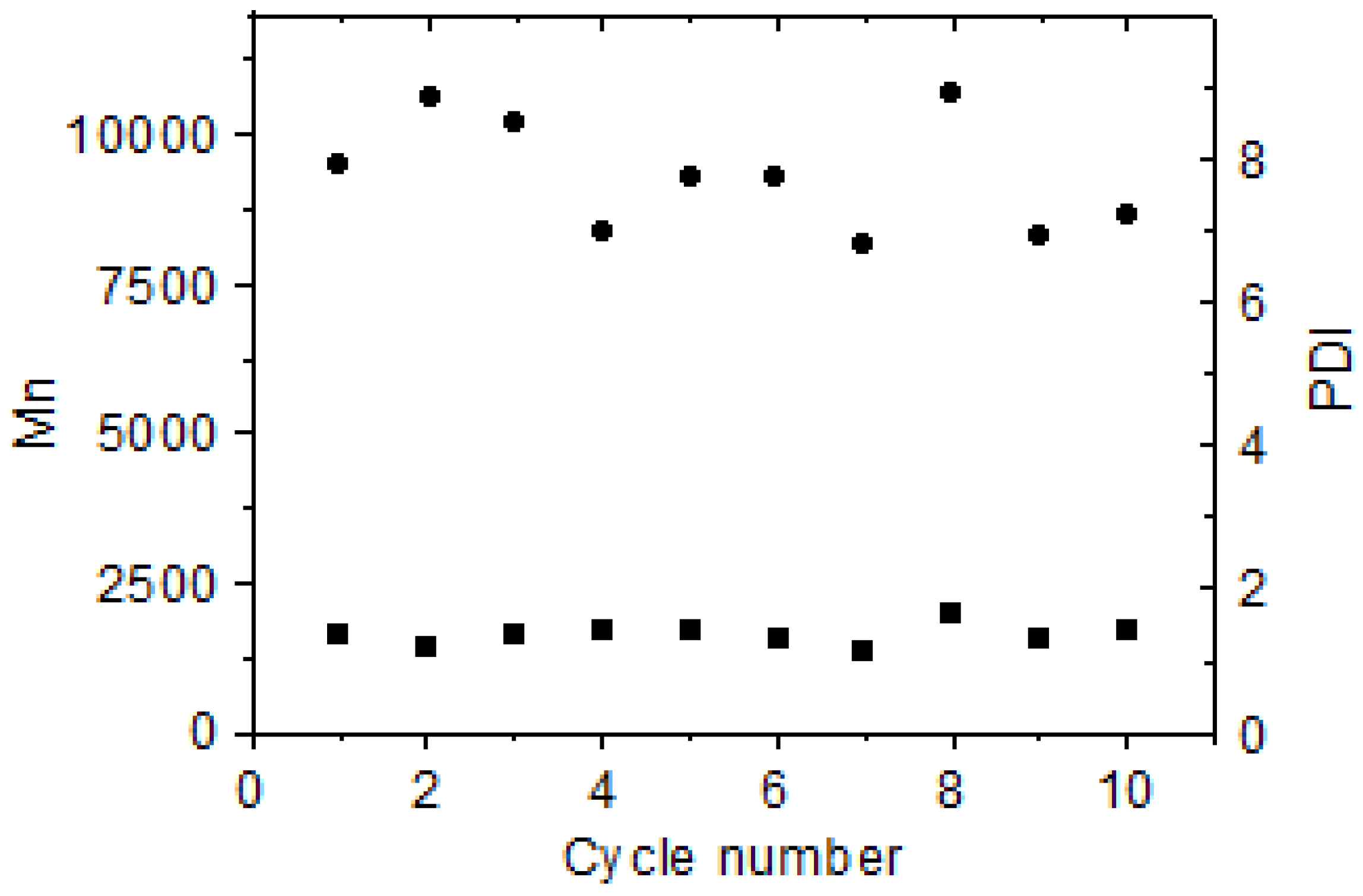
2.4. Catalytic Activity
| Reaction a | Temperature/°C | Time/min | Power/W | Conversion/% b | Mn c | PDI c |
|---|---|---|---|---|---|---|
| 1 d | 20 | 40 | 300 | 63 | 4531 | 1.26 |
| 2 d | 20 | 50 | 300 | 70 | 4917 | 1.23 |
| 3 d | 20 | 60 | 300 | 72 | 5464 | 1.21 |
| 4 d | 30 | 60 | 300 | 65 | 5006 | 1.62 |
| 5 d | 40 | 60 | 300 | 43 | 3564 | 1.67 |
| 6 d | 50 | 60 | 300 | 16 | 2833 | 2.16 |
| 7 d | 20 | 60 | 500 | 65 | 4654 | 2.23 |
| 8 d | 20 | 60 | 800 | 58 | 1303 | 3.11 |
| 9 d,* | 20 | 120 | 300 | 58 | 11437 | 1.31 |
| 10 e | 20 | 60 | 300 | 75 | 7407 | 1.27 |
| 11 f | 20 | 60 | 300 | 59 | 7689 | 1.21 |
| 12 g | 20 | 60 | 300 | 68 | 8230 | 1.54 |
3. Experimental Section
3.1. Materials and Instruments
3.2. Catalyst Preparation
3.3. Electrochemical Property Determination
3.4. Typical Polymerization Process
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Ramesh Kumar, C.; Gatla, S.; Mathon, O.; Pascarelli, S.; Lingaiah, N. The role of niobia location on the acidic and catalytic functionalities of heteropoly tungstate. Appl. Catal. A 2015, 502, 297–304. [Google Scholar] [CrossRef]
- Yu, F.L.; Liu, C.Y.; Yuan, B.; Xie, C.X.; Yu, S.T. Self-assembly heteropoly acid catalyzed oxidative desulfurization of fuel with oxygen. Catal. Commun. 2015, 68, 49–52. [Google Scholar] [CrossRef]
- Alessandro, S.; Irene, G.; Ada, S.; Vincenzo, B.; Antonino, S.A. Enhancement of Oxygen Reduction and Mitigation of Ionomer Dry-Out Using Insoluble Heteropoly Acids in Intermediate Temperature Polymer-Electrolyte Membrane Fuel Cells. Energies 2015, 8, 7805–7817. [Google Scholar]
- Wu, X.F.; Wu, W.; Qian, X.Y.; Wu, Q.Y.; Yan, W.F. Proton-conducting materials based on heteropoly acid and matrixes. J. Non-Cryst. Solids 2015, 426, 88–91. [Google Scholar] [CrossRef]
- Hernández-Cortez, J.G.; Manríquez, M.; Lartundo-Rojas, L.; López-Salinas, E. Study of acid-base properties of supported heteropoly acids in the reactions of secondary alcohols dehydration. Catal. Today 2014, 220–222, 32–38. [Google Scholar]
- Klein, M.; Pulidindi, I.N.; Perkas, N.; Gedanken, A. Heteropoly acid catalyzed hydrolysis of glycogen to glucose. Biomass Bioenergy 2015, 76, 61–68. [Google Scholar] [CrossRef]
- Ladera, R.M.; Fierro, J.L.G.; Ojeda, M.; Rojas, S. TiO2-supported heteropoly acids for low-temperature synthesis of dimethyl ether from methanol. J. Catal. 2014, 312, 195–203. [Google Scholar] [CrossRef]
- Santos, J.S.; Dias, J.A.; Dias, S.C.L.; de Macedo, J.L.; Garcia, F.A.C.; Almeida, L.S.; de Carvalho, E.N.C.B. Acidic characterization and activity of (NH4)xCs2.5–xH0.5PW12O40 catalysts in the esterification reaction of oleic acid with ethanol. Appl. Catal. A 2012, 443–444, 33–39. [Google Scholar] [CrossRef]
- Jing, F.L.; Benjamin, K.; Elisabeth, B.R.; Franck, D.; Sébastien, P. Structural Evolution under Reaction Conditions of Supported (NH4)3HPMo11VO40 Catalysts for the Selective Oxidation of Isobutane. Catalysts 2015, 5, 460–477. [Google Scholar] [CrossRef]
- Li, Y.Y.; Huang, T.P.; Wu, Q.Y.; Xu, L. Synthesis and conductive performance of quaternary molybdotungstovanadophosphoric heteropoly acid with Keggin structure. Mater. Lett. 2015, 157, 109–111. [Google Scholar] [CrossRef]
- Zhou, L.L.; Wang, L.; Zhang, S.J.; Yan, R.Y.; Diao, Y.Y. Effect of vanadyl species in Keggin-type heteropoly catalysts in selective oxidation of methacrolein to methacrylic acid. J. Catal. 2015, 329, 431–440. [Google Scholar] [CrossRef]
- Rafiee, E.; Eavani, S. Controlled immobilization of Keggin-type heteropoly acids on the surface of silica encapsulated γ-Fe2O3 nanoparticles and investigation of catalytic activity in the oxidative esterification of arylaldehydes with methanol. J. Mol. Catal. A 2013, 373, 30–37. [Google Scholar] [CrossRef]
- Alharbi, W.; Brown, E.; Kozhevnikova, E.F.; Kozhevnikov, I.V. Dehydration of ethanol over heteropoly acid catalysts in the gas phase. J. Catal. 2014, 319, 174–181. [Google Scholar] [CrossRef]
- Augusto, L.P.M.; Maíra, S.C.; Kelly, A.S.R.; Elena, V.G. Heteropoly acid catalyzed cyclization of nerolidol and farnesol: Synthesis of α-bisabolol. Appl. Catal. A 2015, 502, 271–275. [Google Scholar]
- Chen, D.; Xue, Z.; Su, Z. A new catalyst of 12-molybdophosphoric acid for cationic polymerization of styrene: Activity and mechanism studies. J. Mol. Catal. A 2003, 203, 307–312. [Google Scholar] [CrossRef]
- Chen, D.; Xue, Z.; Su, Z. Dual role study of 12-molybdophosphoric acid on styrene polymerization. J. Mol. Catal. A 2004, 208, 91–95. [Google Scholar] [CrossRef]
- Rao, P.M.; Wolfson, A.; Kababya, S.; Vega, S.; Landau, M.V. Immobilization of molecular H3PW12O40 heteropolyacid catalyst in alumina-grafted silica-gel and mesostructured SBA-15 silica matrices. J. Catal. 2005, 232, 210–225. [Google Scholar]
- Yang, L.; Qi, Y.; Yuan, X.; Shen, J.; Kim, J. Direct synthesis, characterization and catalytic application of SBA-15 containing heteropolyacid H3PW12O40. J. Mol. Catal. A 2005, 229, 199–205. [Google Scholar] [CrossRef]
- Dimitratos, N.; Pina, C.D.; Falletta, E.; Bianchi, C.L.; Santo, V.D.; Rossi, M. Effect of Au in Cs2.5H1.5PVMo11O40 and Cs2.5H1.5PVMo11O40/Au/TiO2 catalysts in the gas phase oxidation of propylene. Catal. Today 2007, 122, 307–316. [Google Scholar] [CrossRef]
- Audic, N.; Clavier, H.; Mauduit, M.; Guillemin, J.C. An Ionic Liquid-Supported Ruthenium Carbene Complex: A Robust and Recyclable Catalyst for Ring-Closing Olefin Metathesis in Ionic Liquids. J. Am. Chem. Soc. 2003, 125, 9248–9249. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, J.A.; Haddleton, D.M.; Stefan, A.F.; Seddon, K.R. Copper(II) mediated living radical polymerization in an ionic liquid. Chem. Commun. 2000, 14, 1237–1238. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, D.; Deng, Z.; Liu, X.; Wang, R. Synthesis, Characterization and Catalytic Polymerization of N-Methyl Imidazolium Phosphotungstic Catalyst. Catalysts 2015, 5, 1862-1871. https://doi.org/10.3390/catal5041862
Chen D, Deng Z, Liu X, Wang R. Synthesis, Characterization and Catalytic Polymerization of N-Methyl Imidazolium Phosphotungstic Catalyst. Catalysts. 2015; 5(4):1862-1871. https://doi.org/10.3390/catal5041862
Chicago/Turabian StyleChen, Dianyu, Zhaoyi Deng, Xiaoqin Liu, and Rong Wang. 2015. "Synthesis, Characterization and Catalytic Polymerization of N-Methyl Imidazolium Phosphotungstic Catalyst" Catalysts 5, no. 4: 1862-1871. https://doi.org/10.3390/catal5041862
APA StyleChen, D., Deng, Z., Liu, X., & Wang, R. (2015). Synthesis, Characterization and Catalytic Polymerization of N-Methyl Imidazolium Phosphotungstic Catalyst. Catalysts, 5(4), 1862-1871. https://doi.org/10.3390/catal5041862





