Broad Spectrum Microbicidal Activity of Photocatalysis by TiO2
Abstract
:1. Introduction
2. Results and Discussion
2.1. Bactericidal Activity of Photocatalysis by TiO2-Coated Glass
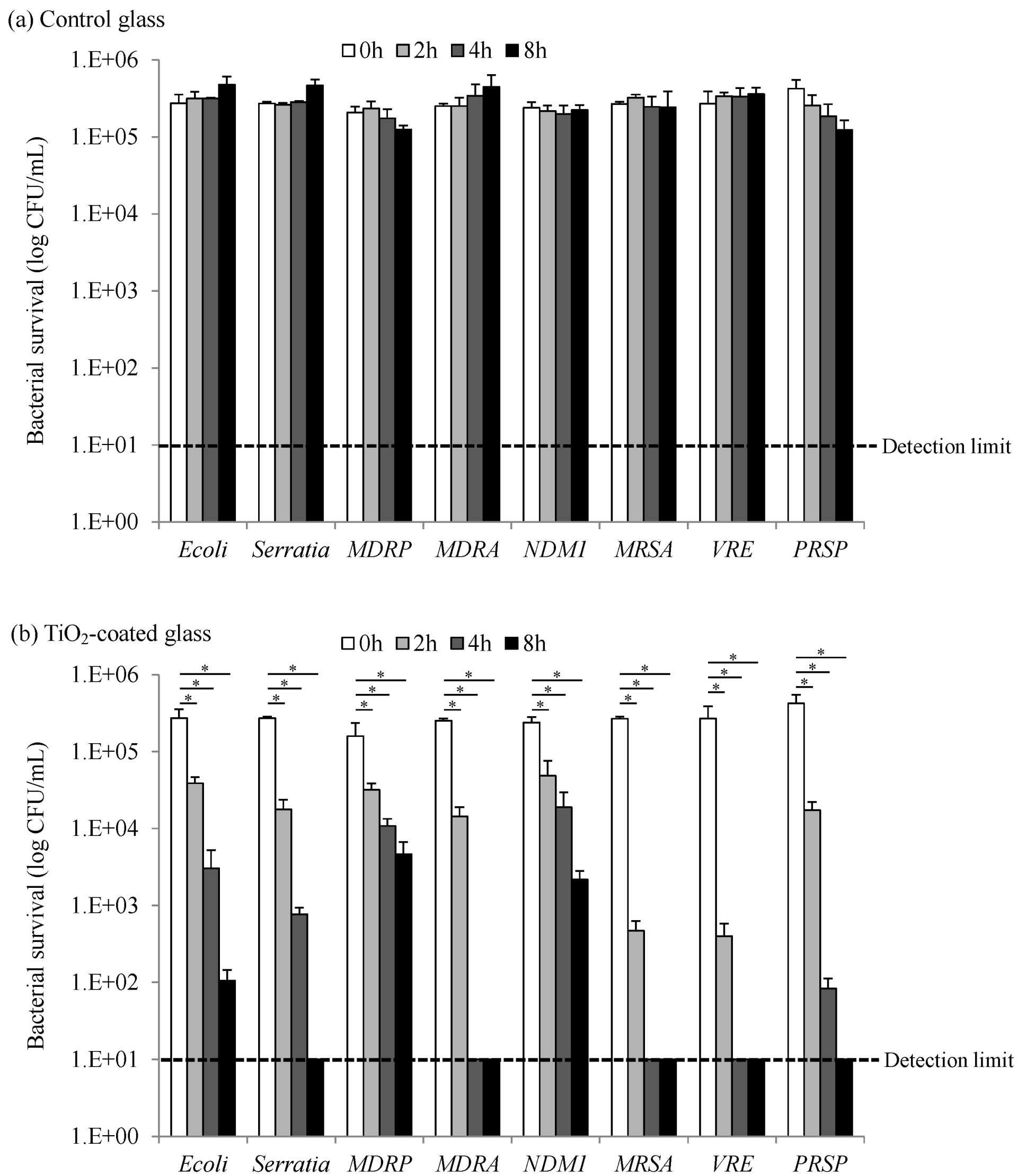
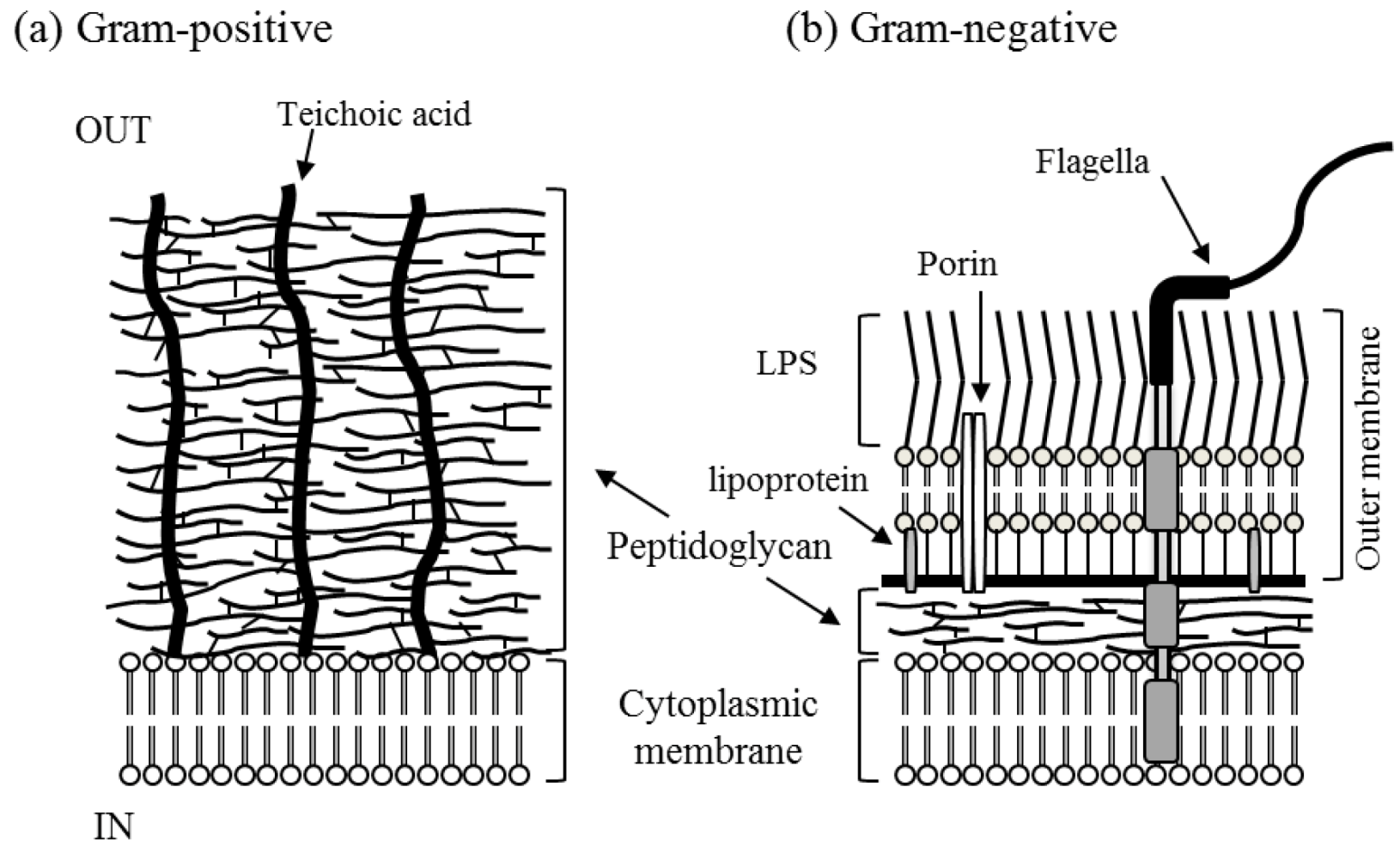
| Characteristic | Gram-positive | Gram-negative |
|---|---|---|
| Thickness of wall | thick (20–80 nm) | thin (10 nm) |
| Number of layers | 1 | 2 |
| Peptidoglycan content | >50% | 10%–20% |
| Lipid and lipoprotein content | 0%–3% | 58% |
| Lipopolysaccharide content | 0 | 13% |
2.2. Virucidal Activity of Photocatalysis by TiO2-Coated Glass
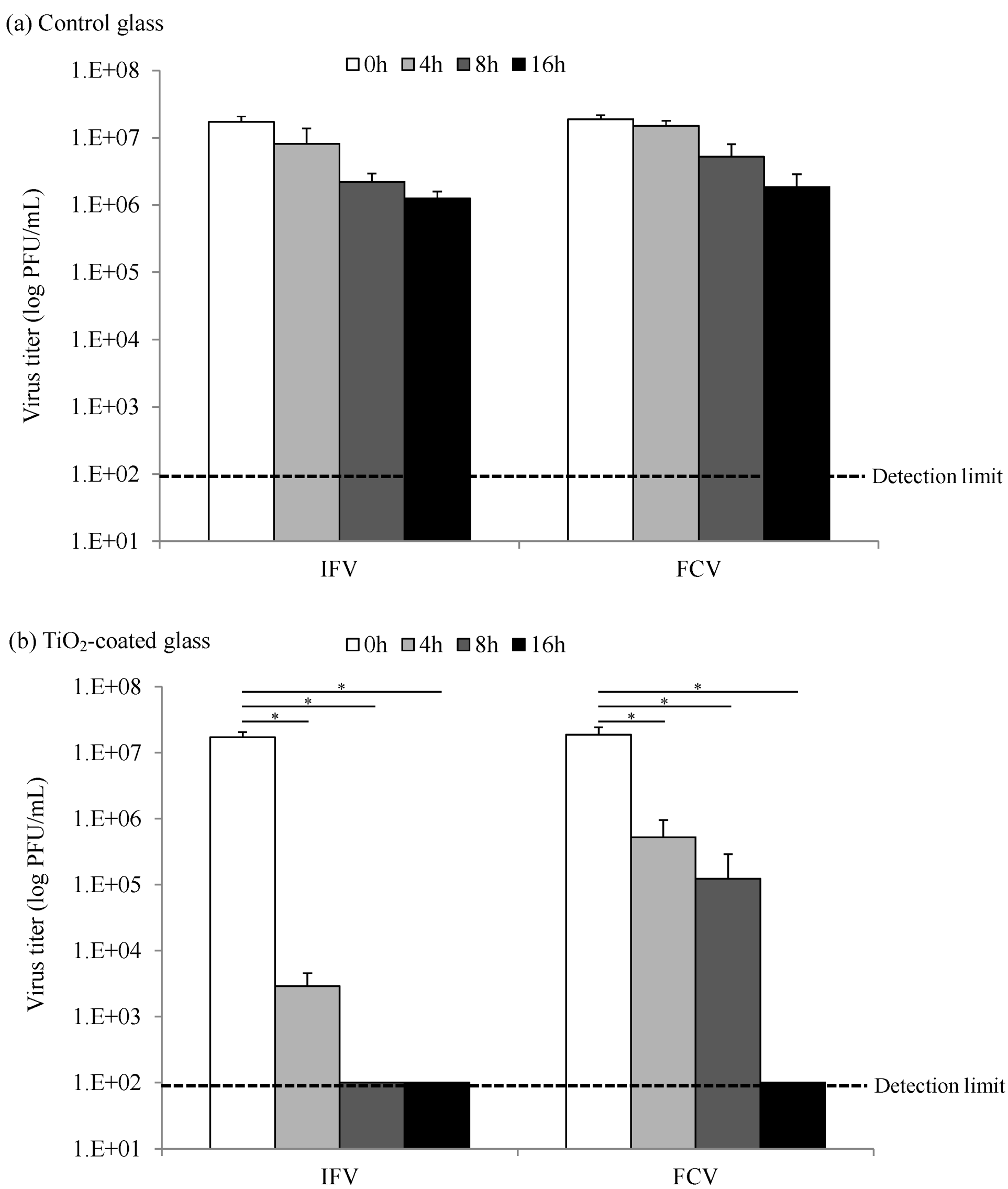
3. Experimental Section
3.1. Bacteria Strains
| Species | Structure | Types of Infections a |
|---|---|---|
| E. coli | Gram-negative rod | Diarrhea, UTI, food poisoning, sepsis |
| S. marcescens | Gram-negative rod | Nosocomial infections, Pneumonia, UTI, and wound infections |
| MDRA | Gram-negative rod | Nosocomial infections, Pneumonia, UTI, VAP |
| MDRP | Gram-negative rod | Nosocomial infections, Pneumonia, UTI, VAP, Bacteremia, cystic fibrosis |
| NDM-1 producing K. pneumoniae | Gram-negative rod | Nosocomial infections, Pneumonia, UTI, Bacteremia |
| MRSA | Gram-positive cocci | Nosocomial infections, skin infections, food poisoning, pneumonia, meningitis, bacteremia |
| VRE | Gram-positive cocci | Nosocomial infections, UTI, meningitis, bacteremia |
| PRSP | Gram-positive cocci | Community-acquired pneumonia, bacteremia, bacterial meningitis |
| IFV | Enveloped virus | Respiratory tract infections |
| FCV | Non-enveloped virus | Respiratory infection in cats |
3.2. Virus Strains
3.3. TiO2-Coated Glass
3.4. Photocatalytic Reaction
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Bratu, S.; Landman, D.; Haag, R.; Recco, R.; Eramo, A.; Alam, M.; Quale, J. Rapid spread of carbapenem-resistant Klebsiella pneumoniae in New York City: A new threat to our antibiotic armamentarium. Arch. Int. Med. 2005, 165, 1430–1435. [Google Scholar] [CrossRef]
- Livermore, D.M. Fourteen years in resistance. Int. J. Antimicrob. Agents 2012, 39, 283–294. [Google Scholar] [CrossRef]
- Burke, J.P. Infection control—A problem for patient safety. N. Engl. J. Med. 2003, 348, 651–656. [Google Scholar] [CrossRef]
- Lankford, M.G.; Collins, S.; Youngberg, L.; Rooney, D.M.; Warren, J.R.; Noskin, G.A. Assessment of materials commonly utilized in health care: Implications for bacterial survival and transmission. Am. J. Infect. Control 2006, 34, 258–263. [Google Scholar] [CrossRef]
- Neely, A.N.; Maley, M.P. Survival of enterococci and staphylococci on hospital fabrics and plastic. J. Clin. Microbiol. 2000, 38, 724–726. [Google Scholar]
- Kramer, A.; Schwebke, I.; Kampf, G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006, 6, 130. [Google Scholar] [CrossRef]
- Weber, D.J.; Rutala, W.A. Use of germicides in the home and the healthcare setting: Is there a relationship between germicide use and antibiotic resistance? Infect. Control Hosp. Epidemiol. 2006, 27, 1107–1119. [Google Scholar] [CrossRef]
- Ramirez, J.A.; Anzueto, A.R. Changing needs of community-acquired pneumonia. J. Antimicrob. Chemother. 2011, 66, iii3–iii9. [Google Scholar] [CrossRef]
- Nordmann, P.; Naas, T.; Poirel, L. Global spread of carbapenemase-producing enterobacteriaceae. Emerg. Infect. Dis. 2011, 17, 1791–1798. [Google Scholar] [CrossRef]
- Fraser, C.; Donnelly, C.A.; Cauchemez, S.; Hanage, W.P.; van Kerkhove, M.D.; Hollingsworth, T.D.; Griffin, J.; Baggaley, R.F.; Jenkins, H.E.; Lyons, E.J.; et al. Pandemic potential of a strain of influenza A (H1N1): Early findings. Science 2009, 324, 1557–1561. [Google Scholar] [CrossRef]
- Hutson, A.M.; Atmar, R.L.; Estes, M.K. Norovirus disease: Changing epidemiology and host susceptibility factors. Trends Microbiol. 2004, 12, 279–287. [Google Scholar] [CrossRef]
- Boone, S.A.; Gerba, C.P. Significance of fomites in the spread of respiratory and enteric viral disease. Appl. Environ. Microbiol. 2007, 73, 1687–1696. [Google Scholar] [CrossRef]
- Sakaguchi, H.; Wada, K.; Kajioka, J.; Watanabe, M.; Nakano, R.; Hirose, T.; Ohta, H.; Aizawa, Y. Maintenance of influenza virus infectivity on the surfaces of personal protective equipment and clothing used in healthcare settings. Environ. Health Prev. Med. 2010, 15, 344–349. [Google Scholar] [CrossRef]
- Goldmann, D.A. Transmission of viral respiratory infections in the home. Pediatr. Infect. Dis. J. 2000, 19, S97–S102. [Google Scholar] [CrossRef]
- Wu, H.M.; Fornek, M.; Schwab, K.J.; Chapin, A.R.; Gibson, K.; Schwab, E.; Spencer, C.; Henning, K. A norovirus outbreak at a long-term-care facility: The role of environmental surface contamination. Infect. Control Hosp. Epidemiol. 2005, 26, 802–810. [Google Scholar]
- Infection Control in Health Care Facilities. Available online: http://www.cdc.gov/flu/professionals/infectioncontrol/ (accessed on 15 October 2012).
- Nakano, R.; Nakano, A.; Abe, M.; Inoue, M.; Okamoto, R. Regional outbreak of CTX-M-2 beta-lactamase-producing Proteus mirabilis in Japan. J. Med. Microbiol. 2012, 61, 1727–1735. [Google Scholar] [CrossRef]
- Guidelines for Environmental Infection Control in Health-Care Facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC); U.S. Department of Health and Human Services Centres for Disease Control and Prevention (CDC): Atlanta, GA, USA, 2003; pp. 1–42.
- Hashimoto, K.; Irie, H.; Fujishima, A. TiO2 photocatalysis: A historical overview and future prospects. Jpn. Soc. Appl. Phys. 2005, 44, 8269–8285. [Google Scholar] [CrossRef]
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: Design and applications. J. Photochem. Photobiol. C 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Nakata, K.; Ochiai, T.; Murakami, T.; Fujishima, A. Photoenergy conversion with TiO2 photocatalysis: New materials and recent applications. Electrochim. Acta 2012, 84, 103–111. [Google Scholar]
- Linsebigler, A.L.; Lu, G.; Yates, J.T. Photocatalysis on TiO2 surfaces: Principles, mechanisms, and selected results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Daimon, T.; Hirakawa, T.; Kitazawa, M.; Suetake, J.; Nosaka, Y. Formation of singlet molecular oxygen associated with the formation of superoxide radicals in aqueous suspensions of TiO2 photocatalysts. Appl. Catal. A 2008, 340, 169–175. [Google Scholar] [CrossRef]
- Ishibashi, K.I.; Fujishima, A.; Watanabe, T.; Hashimoto, K. Generation and deactivation processes of superoxide formed on TiO2 film illuminated by very weak UV light in air or water. J. Phys. Chem. B 2000, 104, 4934–4938. [Google Scholar] [CrossRef]
- Wei, C.; Lin, W.Y.; Zainal, Z.; Williams, N.E.; Zhu, K.; Kruzic, A.P.; Smith, R.L.; Rajeshwar, K. Bactericidal activity of TiO2 photocatalyst in aqueous media: Toward a solar-assisted water disinfection system. Environ. Sci. Technol. 1994, 28, 934–938. [Google Scholar] [CrossRef]
- Ryu, H.; Gerrity, D.; Crittenden, J.C.; Abbaszadegan, M. Photocatalytic inactivation of cryptosporidium parvum with TiO2 and low-pressure ultraviolet irradiation. Water Res. 2008, 42, 1523–1530. [Google Scholar] [CrossRef]
- Ochiai, T.; Fujishima, A. Photoelectrochemical properties of TiO2 photocatalyst and its applications for environmental purification. J. Photochem. Photobiol. C 2012, 13, 247–262. [Google Scholar] [CrossRef]
- Sunada, K.; Watanabe, T.; Hashimoto, K. Bactericidal activity of copper-deposited TiO2 thin film under weak UV light illumination. Environ. Sci. Technol. 2003, 37, 4785–4789. [Google Scholar]
- Mitoraj, D.; Janczyk, A.; Strus, M.; Kisch, H.; Stochel, G.; Heczko, P.B.; Macyk, W. Visible light inactivation of bacteria and fungi by modified titanium dioxide. Photochem. Photobiol. Sci. 2007, 6, 642–628. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Nakano, R.; Ishiguro, H.; Yao, Y.; Kajioka, J.; Fujishima, A.; Sunada, K.; Minoshima, M.; Hashimoto, K.; Kubota, Y. Photocatalytic inactivation of influenza virus by titanium dioxide thin film. Photochem. Photobiol. Sci. 2012, 11, 1293–1298. [Google Scholar] [CrossRef]
- Ishiguro, H.; Nakano, R.; Yao, Y.; Kajioka, J.; Fujishima, A.; Sunada, K.; Minoshima, M.; Hashimoto, K.; Kubota, Y. Photocatalytic inactivation of bacteriophages by TiO2-coated glass plates under low-intensity, long-wavelength UV irradiation. Photochem. Photobiol. Sci 2011, 10, 1825–1829. [Google Scholar] [CrossRef]
- Ishiguro, H.; Yao, Y.; Nakano, R.; Hara, M.; Sunada, K.; Hashimoto, K.; Kajioka, J.; Fujishima, A.; Kubota, Y. Photocatalytic activity of Cu2+/TiO2-coated cordierite foam inactivates bacteriophages and Legionella pneumophila. Appl. Catal. B 2013, 129, 56–61. [Google Scholar] [CrossRef]
- Gerrity, D.; Ryu, H.; Crittenden, J.; Abbaszadegan, M. Photocatalytic inactivation of viruses using titanium dioxide nanoparticles and low-pressure UV light. J. Environ. Sci. Health. A 2008, 43, 1261–1270. [Google Scholar] [CrossRef]
- Kavitha, T.; Gopalan, A.I.; Lee, K.P.; Park, S.Y. Glucose sensing, photocatalytic and antibacterial properties of graphene-ZnO nanoparticle hybrids. Carbon 2012, 50, 2994–3000. [Google Scholar] [CrossRef]
- Synnott, D.W.; Seery, M.K.; Hinder, S.J.; Michlits, G.; Pillai, S.C. Anti-bacterial activity of indoor-light activated photocatalysts. Appl. Catal. B 2013, 130–131, 106–111. [Google Scholar]
- Nixdorff, K.; Gmeiner, J.; Martin, H.H. Interaction of lipopolysaccharide with detergents and its possible role in the detergent resistance of the outer membrane of Gram-negative bacteria. Biochim. Biophys. Acta 1978, 510, 87–98. [Google Scholar] [CrossRef]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar]
- Blake, D.M.; Maness, P.C.; Huang, Z.; Wolfrum, E.J.; Huang, J.; Jacoby, W.A. Application of the photocatalytic chemistry of titanium dioxide to disinfection and the killing of cancer cells. Sep. Purif. Rev. 1999, 28, 1–50. [Google Scholar] [CrossRef]
- Maness, P.C.; Smolinski, S.; Blake, D.M.; Huang, Z.; Wolfrum, E.J.; Jacoby, W.A. Bactericidal activity of photocatalytic TiO2 reaction: Toward an understanding of its killing mechanism. Appl. Environ. Microbiol. 1999, 65, 4094–4098. [Google Scholar]
- Sunada, K.; Kikuchi, Y.; Hashimoto, K.; Fujishima, A. Bactericidal and detoxification effects of TiO2 thin film photocatalysts. Environ. Sci. Technol. 1998, 32, 726–728. [Google Scholar] [CrossRef]
- Macnab, R.M. How bacteria assemble flagella. Annu. Rev. Microbiol. 2003, 57, 77–100. [Google Scholar] [CrossRef]
- Ibáñez, J.A.; Litter, M.I.; Pizarro, R.A. Photocatalytic bactericidal effect of TiO2 on Enterobacter cloacae. J. Photochem. Photobiol. A 2003, 157, 81–85. [Google Scholar] [CrossRef]
- Horowitz, B.; Wiebe, M.E.; Lippin, A.; Stryker, M.H. Inactivation of viruses in labile blood derivatives. I. Disruption of lipid-enveloped viruses by tri(n-butyl)phosphate detergent combinations. Transfusion 1985, 25, 516–522. [Google Scholar]
- Beekes, M.; Mielke, M.; Pauli, G.; Baier, M.; Kurth, R. Aspects of risk assessment and risk management of nosocomial transmission of classical and variant Creutzfeldt-Jakob disease with special attention to German regulations. Contrib. Microbiol. 2004, 11, 117–135. [Google Scholar] [CrossRef]
- Rutala, W.A.; Weber, D.J.; Healthcare Infection Control Practices Advisory Committee (HICPAC). Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008; Centres for Disease Control and Prevention (CDC): Atlanta, GA, USA, 2008. [Google Scholar]
- Shima, E.H. JIS R 1702, Fine Ceramics (Advanced Ceramics, Advanced Technical Ceramics)—Test Method for Antibacterial Activity of Photocatalytic Products under Photoirradiation and Efficacy; Japanese Standards Association: Tokyo, Japan, 2006. [Google Scholar]
- Kalbfuss, B.; Genzel, Y.; Wolff, M.; Zimmermann, A.; Morenweiser, R.; Reichl, U. Harvesting and concentration of human influenza A virus produced in serum-free mammalian cell culture for the production of vaccines. Biotechnol. Bioeng. 2007, 97, 73–85. [Google Scholar]
- Kreutz, L.C.; Seal, B.S.; Mengeling, W.L. Early interaction of feline calicivirus with cells in culture. Arch. Virol. 1994, 136, 19–34. [Google Scholar] [CrossRef]
- Van Regenmortel, M.H.V.; Fauquet, C.M.; Bishop, D.H.L. Virus Taxonomy. Classification and Nomenclature of Viruses; Academic Press: New York, NY, USA, 2000; pp. 835–849. [Google Scholar]
- Yachida, S.; Aoyama, S.; Takahashi, N.; Iritani, Y.; Katagiri, K. Plastic multiwell plates to assay avian infectious bronchitis virus in organ cultures of chicken embryo trachea. J. Clin. Microbiol. 1978, 8, 380–387. [Google Scholar]
- Tobita, K.; Sugiura, A.; Enomote, C.; Furuyama, M. Plaque assay and primary isolation of influenza A viruses in an established line of canine kidney cells (MDCK) in the presence of trypsin. Med. Microbiol. Immunol. 1975, 162, 9–14. [Google Scholar] [CrossRef]
- Fine Ceramics (Advanced Ceramics, Advanced Technical Ceramics)—Test Method for Antibacterial Activity of Semiconducting Photocatalytic Materials; Japan Industrial Standards Committee: Tokyo, Japan, 2009.
- Ochiai, T.; Nanba, H.; Nakagawa, T.; Masuko, K.; Nakata, K.; Murakami, T.; Nakano, R.; Hara, M.; Koide, Y.; Suzuki, T.; et al. Development of an O3-assisted photocatalytic water-purification unit by using a TiO2 modified titanium mesh filter. Catal. Sci. Technol. 2012, 2, 76. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nakano, R.; Hara, M.; Ishiguro, H.; Yao, Y.; Ochiai, T.; Nakata, K.; Murakami, T.; Kajioka, J.; Sunada, K.; Hashimoto, K.; et al. Broad Spectrum Microbicidal Activity of Photocatalysis by TiO2. Catalysts 2013, 3, 310-323. https://doi.org/10.3390/catal3010310
Nakano R, Hara M, Ishiguro H, Yao Y, Ochiai T, Nakata K, Murakami T, Kajioka J, Sunada K, Hashimoto K, et al. Broad Spectrum Microbicidal Activity of Photocatalysis by TiO2. Catalysts. 2013; 3(1):310-323. https://doi.org/10.3390/catal3010310
Chicago/Turabian StyleNakano, Ryuichi, Masayuki Hara, Hitoshi Ishiguro, Yanyan Yao, Tsuyoshi Ochiai, Kazuya Nakata, Taketoshi Murakami, Jitsuo Kajioka, Kayano Sunada, Kazuhito Hashimoto, and et al. 2013. "Broad Spectrum Microbicidal Activity of Photocatalysis by TiO2" Catalysts 3, no. 1: 310-323. https://doi.org/10.3390/catal3010310
APA StyleNakano, R., Hara, M., Ishiguro, H., Yao, Y., Ochiai, T., Nakata, K., Murakami, T., Kajioka, J., Sunada, K., Hashimoto, K., Fujishima, A., & Kubota, Y. (2013). Broad Spectrum Microbicidal Activity of Photocatalysis by TiO2. Catalysts, 3(1), 310-323. https://doi.org/10.3390/catal3010310





