Ethylene Polymerization Using (Imino)vanadium(V) Dichloride Complexes Containing (Anilido)methyl-pyridine, -quinoline Ligands–Halogenated Al Alkyls Catalyst Systems
Abstract
:1. Introduction
| Run | V complex | Al cocat. | Al/V b | C4',C6' | Polyethylene (PE) | ||||
|---|---|---|---|---|---|---|---|---|---|
| (μmol) | Activity c | C4'/% d | C6'/% d | Activity e | Mw f | Mw/Mn f | |||
| 1 | 1a (0.2) g | MAO | 500 | 57800 | 96.8 | 3.2 | |||
| 2 | 1a (0.1) g | MAO | 1500 | 76500 | 97.0 | 3.0 | |||
| 3 | 1b (0.5) g | MAO | 1000 | 35700 | 92.1 | 7.9 | |||
| 4 | 1a (5.0) h | Et2AlCl | 100 | 137 | 5.92 i | ||||
| 5 | 1a (5.0) h | Me2AlCl | 200 | 704 | 6.76 i | ||||
| 6 | 2a (5.0) j | MAO | 1000 | 43 | 71.6 | 28.4 | 53 | ||
| 7 | 3a (2.0) j | MAO | 1000 | 201 | 92.4 | 7.6 | 30 | ||
| 8 | 3a (2.0) j | MAO | 1500 | 249 | 92 | 8 | 45 | ||
| 9 | 4a (0.2) j | MAO | 600 | 50300 | 95.2 | 4.8 | |||
| 10 | 4b (0.2) j | MAO | 700 | 41500 | 97.1 | 2.9 | |||
| 11 | 5a (2.0) k | MAO | 3000 | 78 | 2.98 | 2.0 | |||
| 12 | 5b (2.0) k | MAO | 3000 | 189 | 2.93 | 2.6 | |||
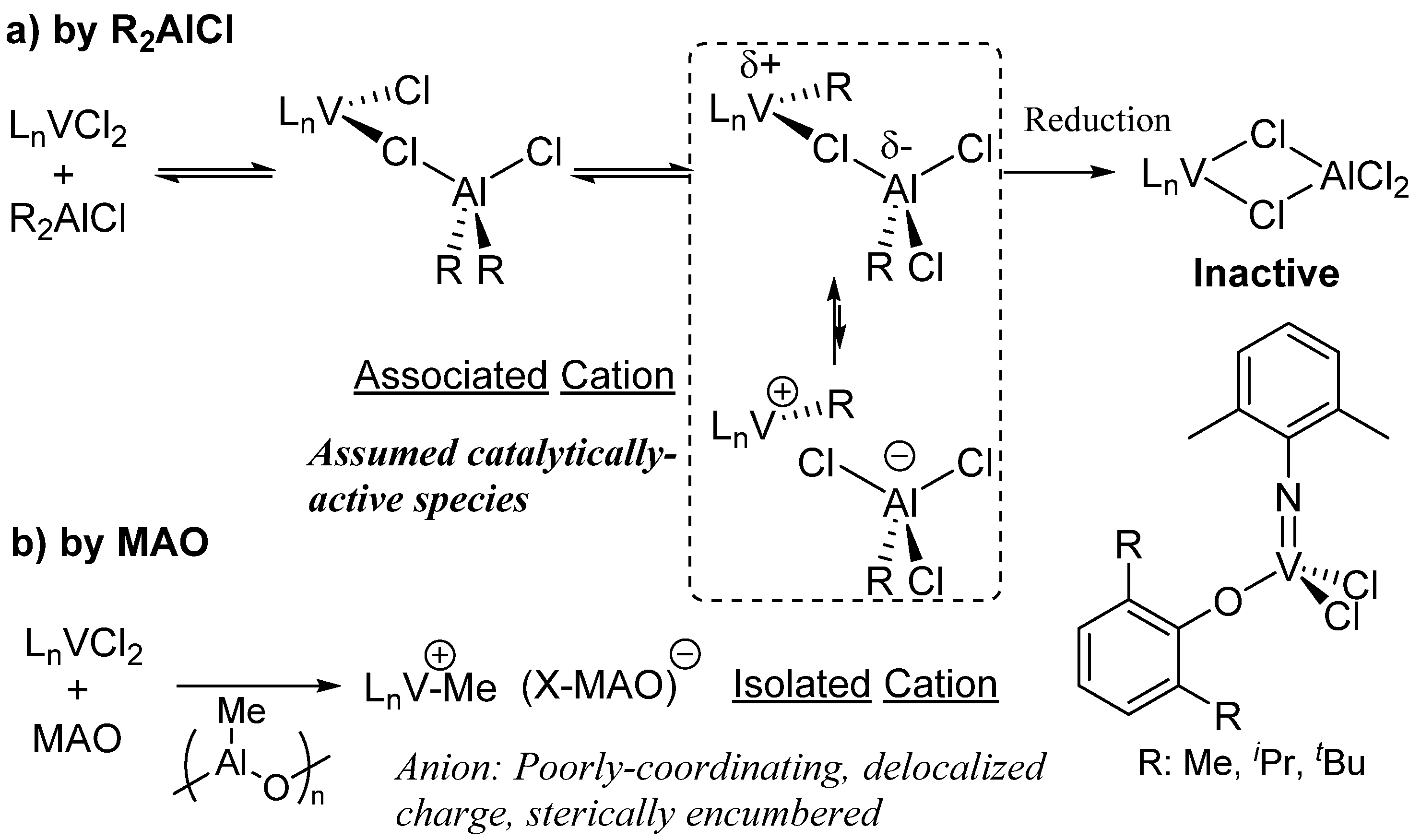
2. Results and Discussion
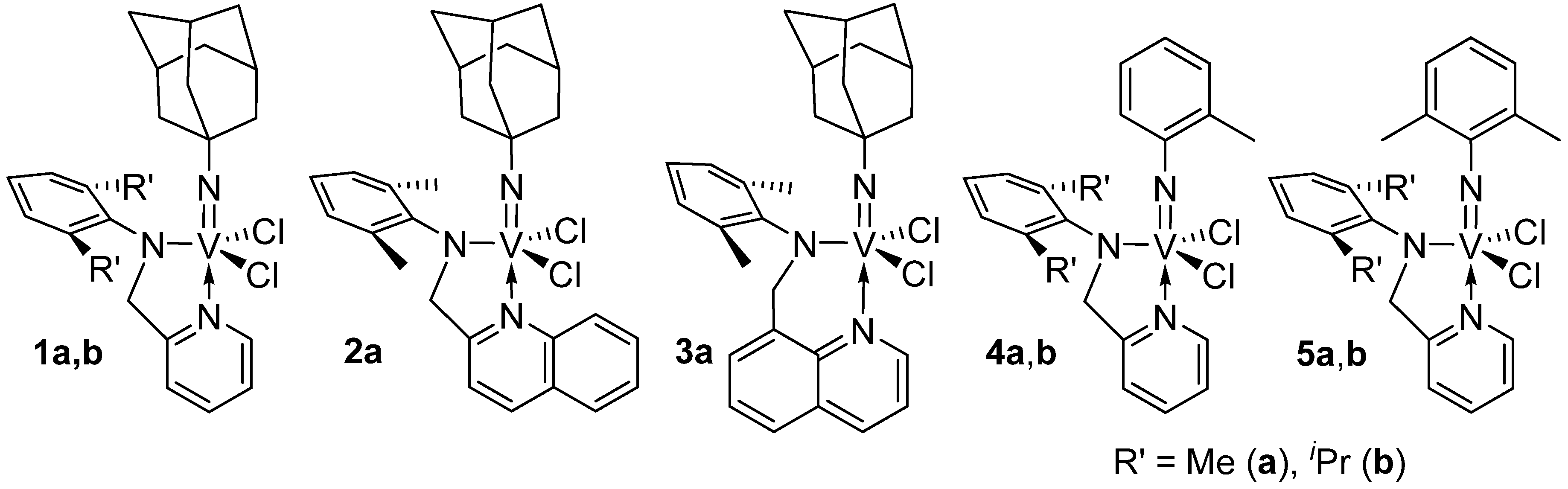
2.1. Ethylene Polymerization Using V(NAd)Cl2[2-ArNCH2(C9H6N)], V(NAd)Cl2[8-ArNCH2(C9H6N)]–Me2AlCl Catalyst Systems
| Run | Vanadium complex | Al/V b | Yield | Activity c | |
|---|---|---|---|---|---|
| L (V cat.) | /μmol | /mg | |||
| 5 d | 2-ArNCH2(C5H4N) (1a) e | 0.5 | 200 | 58.7 | 704 |
| 13 d | 2-ArNCH2(C5H4N) (1a) f | 1.0 | 500 | 116 | 696 |
| 14 | 2-ArNCH2(C9H6N) (2a) | 0.2 | 2500 | 70.2 | 2110 |
| 15 | 2-ArNCH2(C9H6N) (2a) | 0.2 | 5000 | 122 | 3670 |
| 16 | 2-ArNCH2(C9H6N) (2a) | 0.2 | 7500 | 152 | 4560 |
| 17 | 2-ArNCH2(C9H6N) (2a) | 0.2 | 10000 | 120 | 3600 |
| 18 | 8-ArNCH2(C9H6N) (3a) | 0.2 | 1000 | 79.6 | 2390 |
| 19 | 8-ArNCH2(C9H6N) (3a) | 0.2 | 2000 | 73.6 | 2210 |
| 20 | 8-ArNCH2(C9H6N) (3a) | 0.2 | 5000 | 58.7 | 1760 |
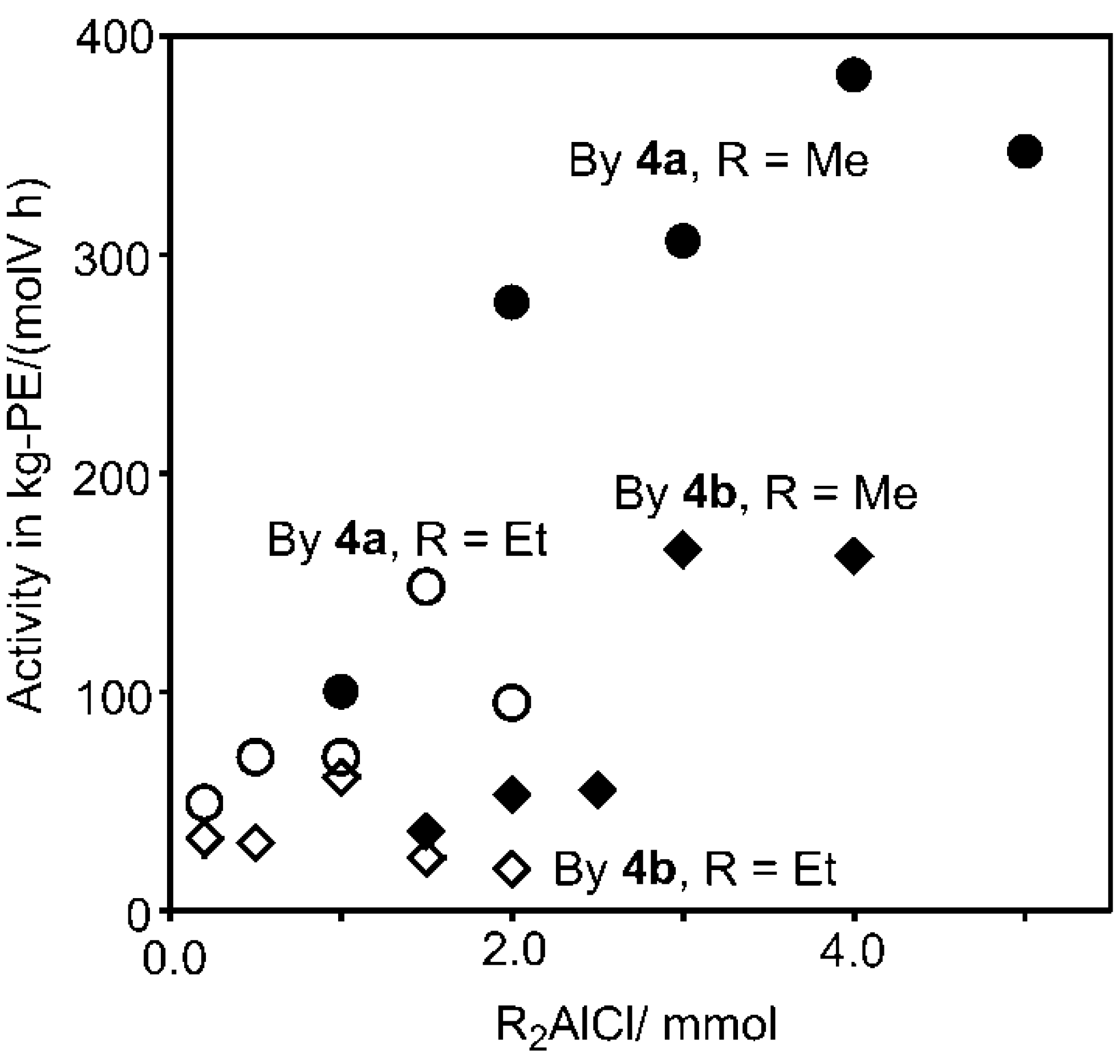
2.2. Ethylene Polymerization Using V(N-2-MeC6H4)Cl2[2-(2,6-R'2C6H3)NCH2(C5H4N)]–Halogenated Al Alkyls Catalyst Systems
| Run | Vanadium complex | Al cocat. | Al/V b | Yield | Activity c | |
|---|---|---|---|---|---|---|
| R (V cat.) | /μmol | (mmol) | /mg | |||
| 5d | 1-adamantyl (1a) e | 0.5 | Me2AlCl (0.10) | 200 | 58.7 | 704 |
| 13d | 1-adamantyl (1a) f | 1.0 | Me2AlCl (0.50) | 500 | 116 | 696 |
| 21 | 2-MeC6H4 (4a) | 1.0 | Me2AlCl (1.0) | 1000 | 16.7 | 100 |
| 22 | 2-MeC6H4 (4a) | 1.0 | Me2AlCl (2.0) | 2000 | 46.4 | 278 |
| 23 | 2-MeC6H4 (4a) | 1.0 | Me2AlCl (3.0) | 3000 | 51.0 | 306 |
| 25 | 2-MeC6H4 (4a) | 1.0 | Me2AlCl (4.0) | 4000 | 63.7 | 382 |
| 26 | 2-MeC6H4 (4a) | 1.0 | Me2AlCl (5.0) | 5000 | 57.8 | 347 |
| 27 | 2-MeC6H4 (4b) | 2.0 | Me2AlCl (1.5) | 750 | 11.9 | 36 |
| 28 | 2-MeC6H4 (4b) | 2.0 | Me2AlCl (2.0) | 1000 | 17.8 | 53 |
| 29 | 2-MeC6H4 (4b) | 2.0 | Me2AlCl (2.5) | 1250 | 18.2 | 55 |
| 30 | 2-MeC6H4 (4b) | 2.0 | Me2AlCl (3.0) | 1500 | 55.1 | 165 |
| 31 | 2-MeC6H4 (4b) | 2.0 | Me2AlCl (4.0) | 2000 | 54.1 | 162 |
| 32 | 2-MeC6H4 (4a) | 1.0 | Et2AlCl (0.20) | 200 | 8.1 | 49 |
| 33 | 2-MeC6H4 (4a) | 1.0 | Et2AlCl (0.50 | 500 | 11.7 | 70 |
| 34 | 2-MeC6H4 (4a) | 1.0 | Et2AlCl (1.0) | 1000 | 11.6 | 70 |
| 35 | 2-MeC6H4 (4a) | 1.0 | Et2AlCl (1.5) | 1500 | 24.6 | 148 |
| 36 | 2-MeC6H4 (4a) | 1.0 | Et2AlCl (2.0) | 2000 | 15.9 | 95 |
| 37 | 2-MeC6H4 (4b) | 1.0 | Et2AlCl (0.20) | 200 | 5.5 | 33 |
| 38 | 2-MeC6H4 (4b) | 1.0 | Et2AlCl (0.50) | 500 | 5.1 | 31 |
| 39 | 2-MeC6H4 (4b) | 1.0 | Et2AlCl (1.0) | 1000 | 10.1 | 61 |
| 40 | 2-MeC6H4 (4b) | 1.0 | Et2AlCl (1.5) | 1500 | 4.0 | 24 |
| 41 | 2-MeC6H4 (4b) | 1.0 | Et2AlCl (2.0) | 2000 | 3.2 | 19 |
| 42 | 2,6-Me2C6H3 (5a) g | 1.0 | Et2AlCl (0.10) | 100 | 140 | 840 |
| 43 | 2,6-Me2C6H3 (5b) g | 0.2 | Et2AlCl (0.04) | 200 | 200 | 6000 |
3. Experimental Section
3.1. General Procedure
3.2. Ethylene Polymerization
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Hagen, H.; Boersma, J.; van Koten, G. Homogeneous vanadium-based catalysts for the Ziegler–Natta polymerization of α-olefins. Chem. Soc. Rev. 2002, 31, 357–364. [Google Scholar] [CrossRef]
- Gambarotta, S. Vanadium-based Ziegler/Natta: Challenges, promises, problems. Coord. Chem. Rev. 2003, 237, 229–243. [Google Scholar] [CrossRef]
- Nomura, K.; Zhang, W. (Imido)vanadium(V)-alkyl, -alkylidene complexes exhibiting unique reactivity towards olefins and alcohols. Chem. Sci. 2010, 1, 161–173. [Google Scholar] [CrossRef]
- Redshaw, C. Vanadium procatalysts bearing chelating aryloxides: structure–activity trends in ethylene polymerisation. Dalton Trans. 2010, 39, 5595–5604. [Google Scholar] [CrossRef]
- Nomura, K.; Zhang, S. Design of vanadium complex catalysts for precise olefin polymerization. Chem. Rev. 2011, 111, 2342–2362. [Google Scholar] [CrossRef]
- Nomura, K.; Sagara, A.; Imanishi, Y. Olefin polymerization and ring-opening metathesis polymerization of norbornene by (arylimido)(aryloxo)vanadium(V) complexes of the type VX2(NAr)(OAr¢). Remarkable effect of aluminum cocatalyst for the coordination and insertion and ring-opening metathesis. Macromolecules 2002, 35, 1583–1590. [Google Scholar]
- Wang, W.; Nomura, K. Remarkable effects of aluminum cocatalyst and comonomer in ethylene copolymerizations catalyzed by (arylimido)(aryloxo)vanadium complexes: efficient synthesis of high molecular weight ethylene/norbornene copolymer. Macromolecules 2005, 38, 5905–5913. [Google Scholar] [CrossRef]
- Wang, W.; Nomura, K. Notable effects of aluminum alkyls and solvents for highly efficient ethylene (co)polymerizations catalyzed by (arylimido)-(aryloxo)vanadium complexes. Adv. Synth. Catal. 2006, 348, 743–750. [Google Scholar] [CrossRef]
- Onishi, Y.; Katao, S.; Fujiki, M.; Nomura, K. Synthesis and structural analysis of (arylimido)vanadium(V) complexes containing phenoxyimine ligands: new, efficient catalyst precursors for ethylene polymerization. Organometallics 2008, 27, 2590–2596. [Google Scholar] [CrossRef]
- Zhang, S.; Katao, S.; Sun, W.-H.; Nomura, K. Synthesis of (arylimido)vanadium(V) complexes containing (2-anilidomethyl)pyridine ligands and their use as the catalyst precursors for olefin polymerization. Organometallics 2009, 28, 5925–5933. [Google Scholar] [CrossRef]
- Macchioni, A. Ion pairing in transition-metal organometallic chemistry. Chem. Rev. 2005, 105, 2039–2073. [Google Scholar] [CrossRef]
- Li, H.; Marks, T.J. Nuclearity and cooperativity effects in binuclear catalysts and cocatalysts for olefin polymerization. Proc. Natl. Acad. Sci. USA 2006, 103, 15295–15302. [Google Scholar] [CrossRef]
- Bochmann, M. The chemistry of catalyst activation: the case of group 4 polymerization catalysts. Organometallics 2010, 29, 4711–4740. [Google Scholar] [CrossRef]
- Christman, D.L. Preparation of polyethylene in solution. J. Polym. Sci. Part A-1 1972, 10, 471–487. [Google Scholar] [CrossRef]
- Zhang, S.; Nomura, K. Highly efficient dimerization of ethylene by (imido)vanadium complexes containing (2-anilidomethyl)pyridine ligands: notable ligand effect toward activity and selectivity. J. Am. Chem. Soc. 2010, 132, 4960–4965. [Google Scholar] [CrossRef]
- Igarashi, A.; Zhang, S.; Nomura, K. Ethylene dimerization/polymerization catalyzed by (adamantylimido)vanadium(V) complexes containing (2-anilidomethyl)pyridine ligands: factors affecting the ethylene reactivity. Organometallics 2012, 31, 3575–3581. [Google Scholar] [CrossRef]
- Nomura, K.; Igarashi, A.; Katao, S.; Zhang, W.; Sun, W.-H. Synthesis and structural analysis of (imido)vanadium(V) complexes containing chelate (anilido)methyl-imine ligands: Ligand effect in ethylene dimerization. Inorg. Chem. 2013, in press. [Google Scholar]
- Redshaw, C.; Rowan, M.A.; Homden, D.M.; Dale, S.H.; Elsegood, M.R.J.; Matsui, S.; Matsuura, S. Vanadyl C and N-capped tris(phenolate) complexes: influence of procatalyst geometry on catalytic activity. Chem. Commun. 2006, 3329–3331. [Google Scholar]
- Wu, J.-Q.; Mu, J.-S.; Zhang, S.-W.; Li, Y.-S. Vanadium(V) complexes containing tetradentate amine trihydroxy ligands as catalysts for copolymerization of cyclic olefins. J. Polym. Sci. Part A 2010, 48, 1122–1132. [Google Scholar] [CrossRef]
- Redshaw, C.; Warford, L.; Dale, S.H.; Elsegood, M.R.J. Vanadyl complexes bearing bi- and triphenolate chelate ligands: highly active ethylene polymerisation procatalysts. Chem. Commun. 2004, 1954–1955. [Google Scholar]
- Arbaoui, A.; Redshaw, C.; Homden, D.M.; Wright, J.A.; Elsegood, M.R.J. Vanadium-based imido-alkoxide pro-catalysts bearing bisphenolate ligand for ethylene and e-caprolactone polymerisation. Dalton Trans. 2009, 8911–8922. [Google Scholar]
- Redshaw, C.; Rowan, M.A.; Warford, L.; Homden, D.M.; Arbaoui, A.; Elsegood, M.R.J.; Dale, S.H.; Yamato, T.; Casas, C.P.; Matsui, S.; et al. Oxo- and imidovanadium complexes incorporatingmethylene- and diachtungtrenungmethyleneoxa-bridged calix[3]- and achtungtrenung-[4]arenes: synthesis, structures and ethylene polymerisation catalysis. Chem.-Eur. J. 2007, 13, 1090–1107. [Google Scholar]
- Homden, D.; Redshaw, C.; Wright, J.A.; Hughes, D.L.; Elsegood, M.R.J. Early transition metal complexes bearing a c-capped tris(phenolate) ligand incorporating a pendant imine arm: synthesis, structure, and ethylene polymerization behavior. Inorg. Chem. 2008, 47, 5799–5814. [Google Scholar] [CrossRef]
- Clowes, L.; Redshaw, C.; Hughes, D.L. Vanadium-based pro-catalysts bearing depleted 1,3-calix[4]arenes for ethylene or ε-caprolactone polymerization. Inorg. Chem. 2011, 50, 7838–7845. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Igarashi, A.; Zhang, W.; Sun, W.-H.; Nomura, K. Ethylene Polymerization Using (Imino)vanadium(V) Dichloride Complexes Containing (Anilido)methyl-pyridine, -quinoline Ligands–Halogenated Al Alkyls Catalyst Systems. Catalysts 2013, 3, 148-156. https://doi.org/10.3390/catal3010148
Igarashi A, Zhang W, Sun W-H, Nomura K. Ethylene Polymerization Using (Imino)vanadium(V) Dichloride Complexes Containing (Anilido)methyl-pyridine, -quinoline Ligands–Halogenated Al Alkyls Catalyst Systems. Catalysts. 2013; 3(1):148-156. https://doi.org/10.3390/catal3010148
Chicago/Turabian StyleIgarashi, Atsushi, Wenjuan Zhang, Wen-Hua Sun, and Kotohiro Nomura. 2013. "Ethylene Polymerization Using (Imino)vanadium(V) Dichloride Complexes Containing (Anilido)methyl-pyridine, -quinoline Ligands–Halogenated Al Alkyls Catalyst Systems" Catalysts 3, no. 1: 148-156. https://doi.org/10.3390/catal3010148
APA StyleIgarashi, A., Zhang, W., Sun, W.-H., & Nomura, K. (2013). Ethylene Polymerization Using (Imino)vanadium(V) Dichloride Complexes Containing (Anilido)methyl-pyridine, -quinoline Ligands–Halogenated Al Alkyls Catalyst Systems. Catalysts, 3(1), 148-156. https://doi.org/10.3390/catal3010148







