Influence of Preparation Method on Structure and Photocatalytic Performance of Bi2MoO6
Abstract
1. Introduction
2. Results and Discussion
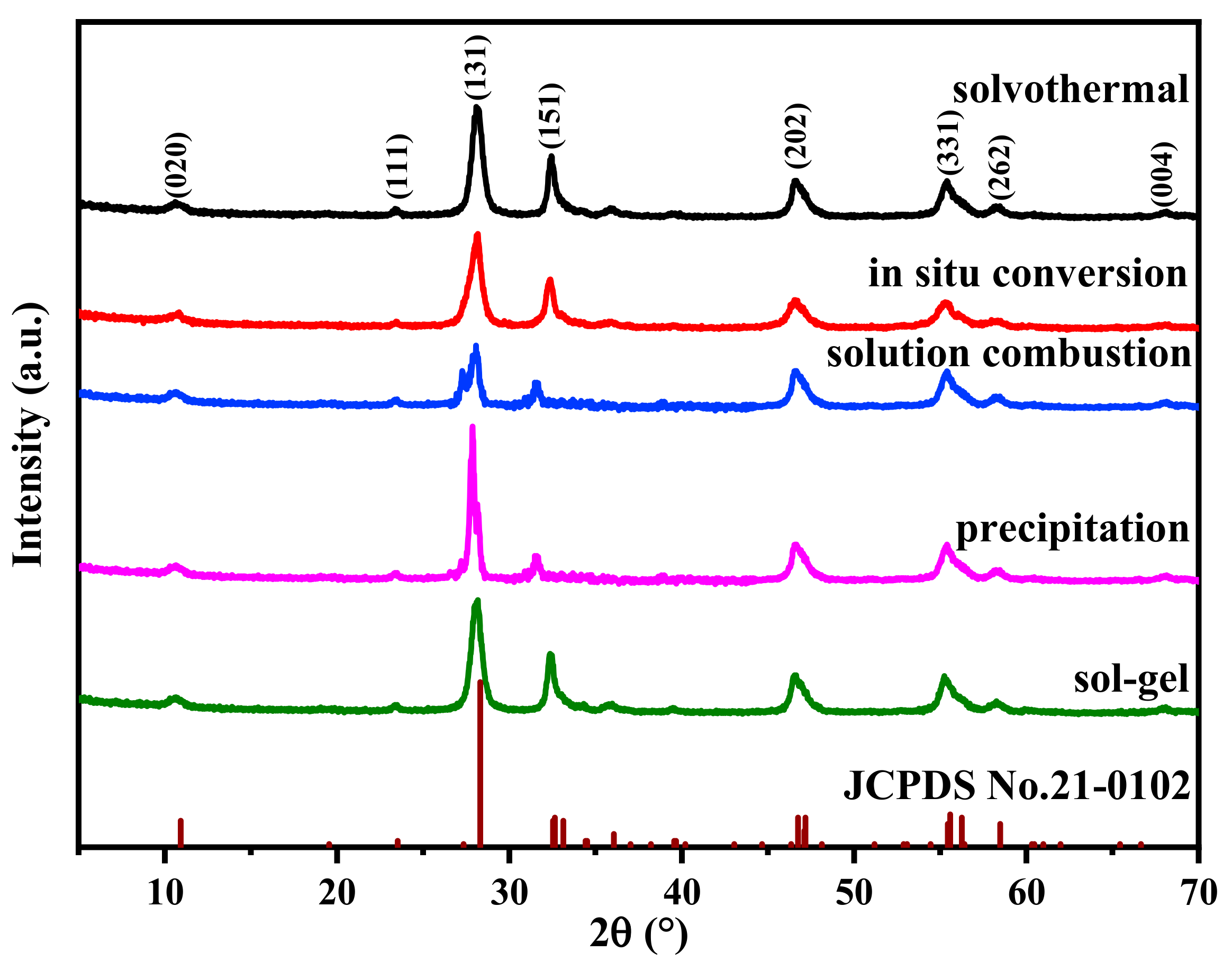
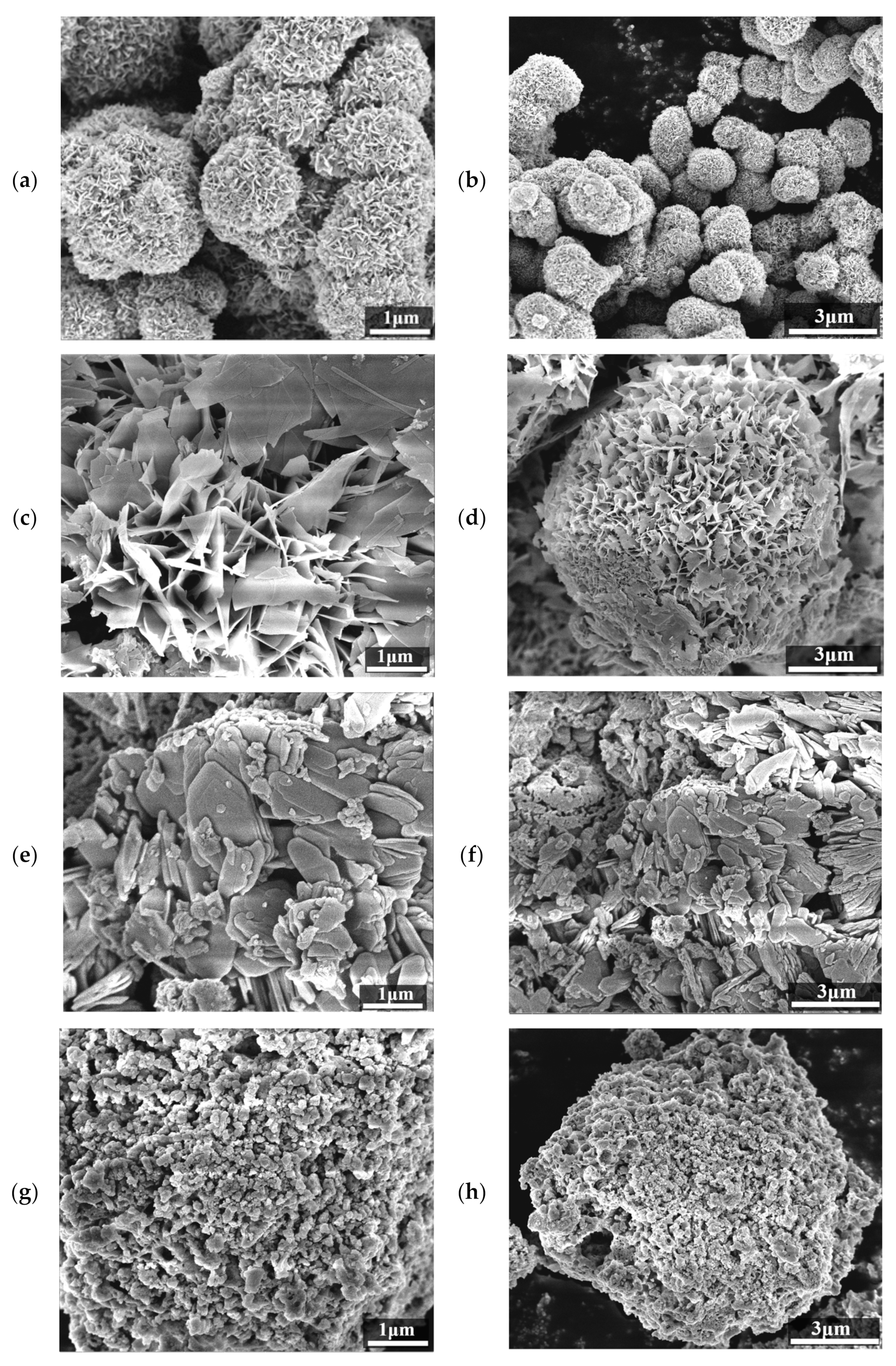
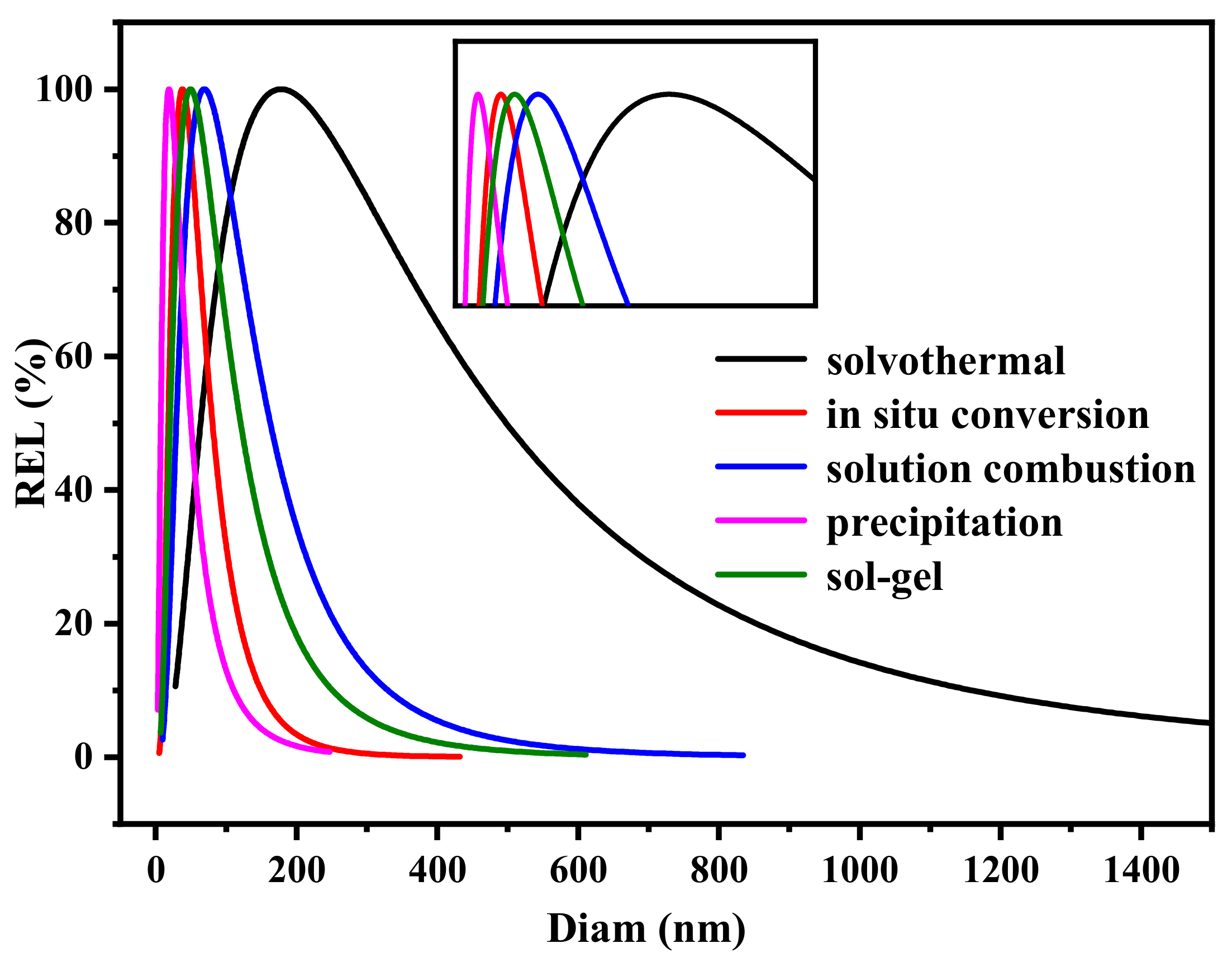
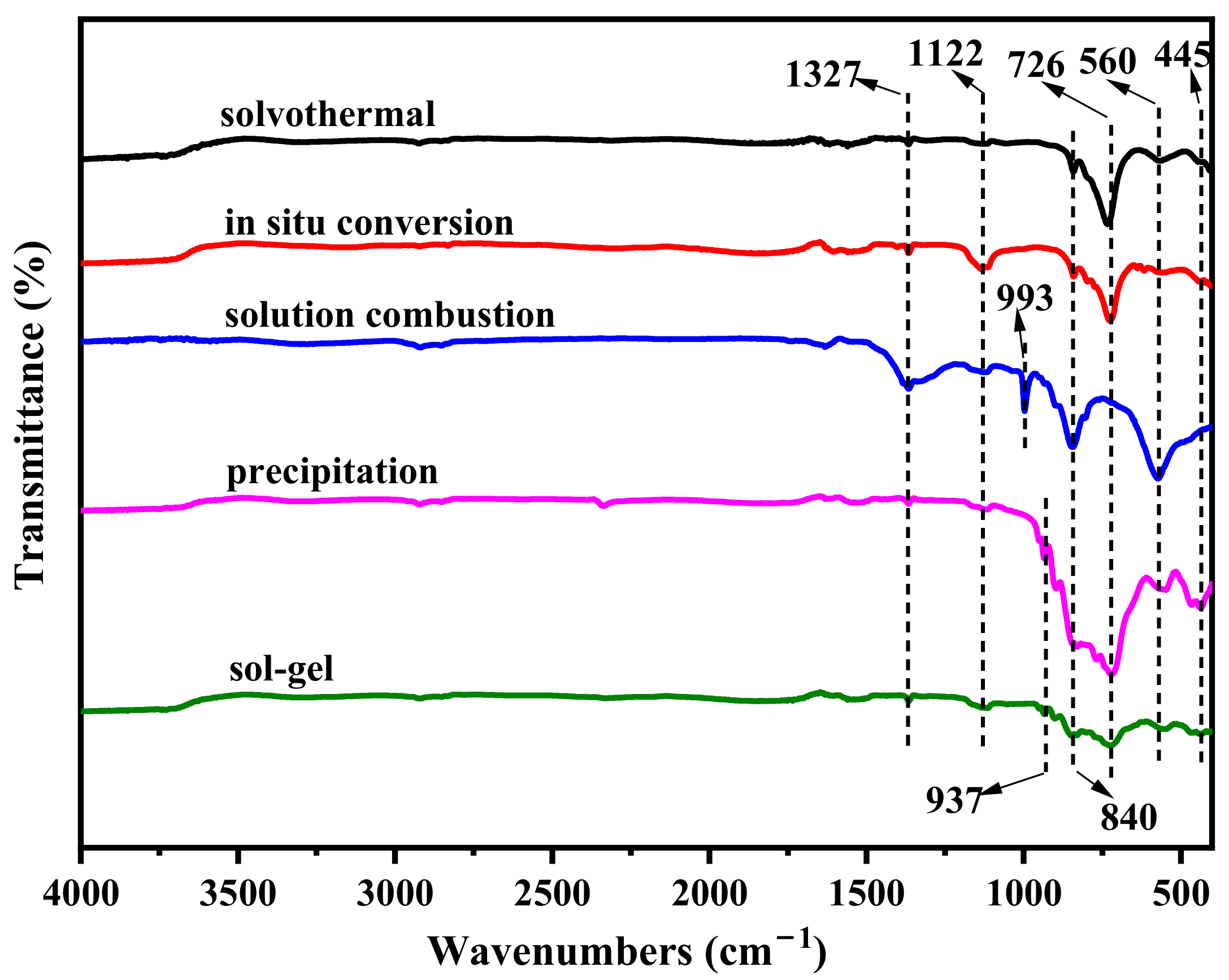
2.1. Phase and Morphology
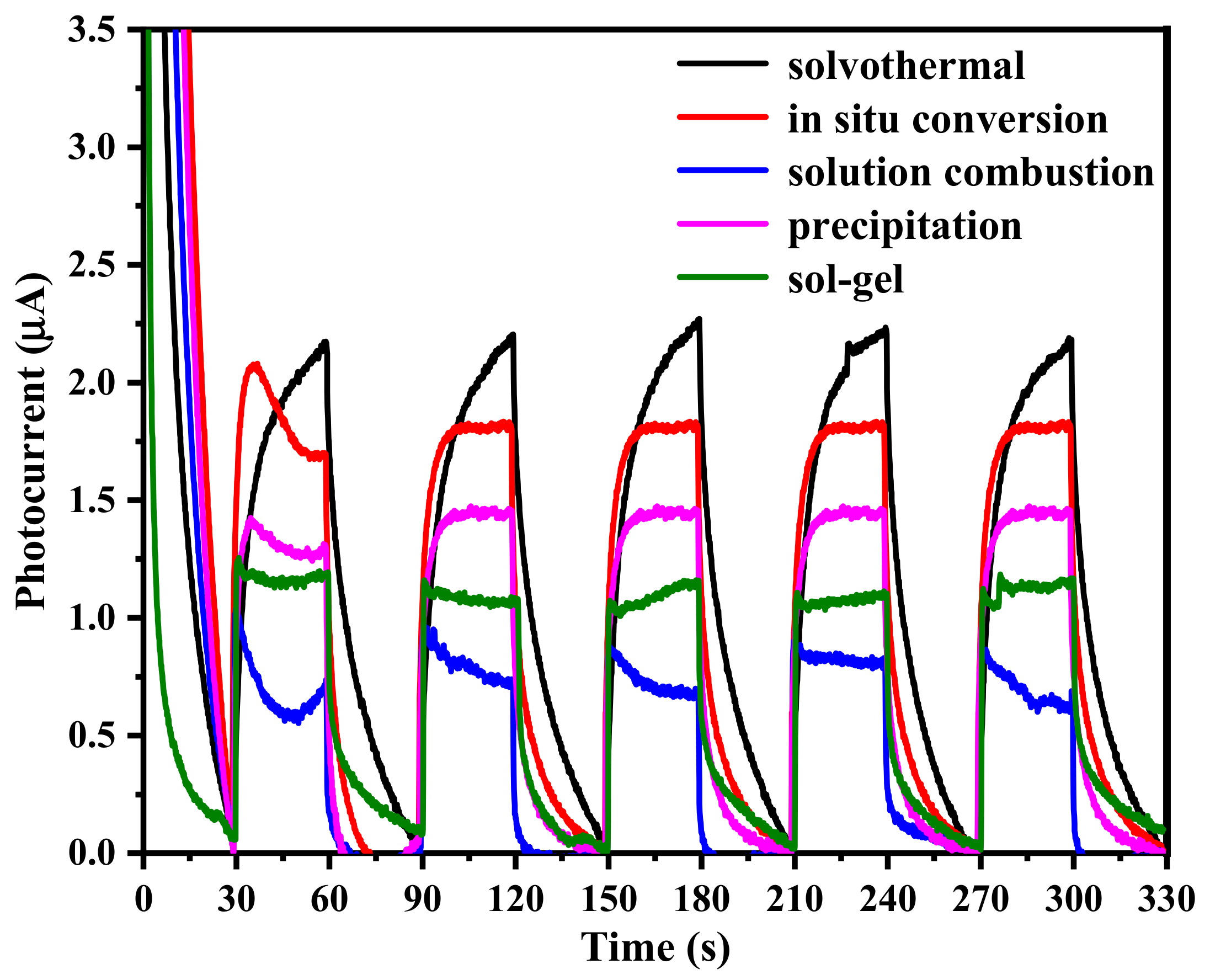
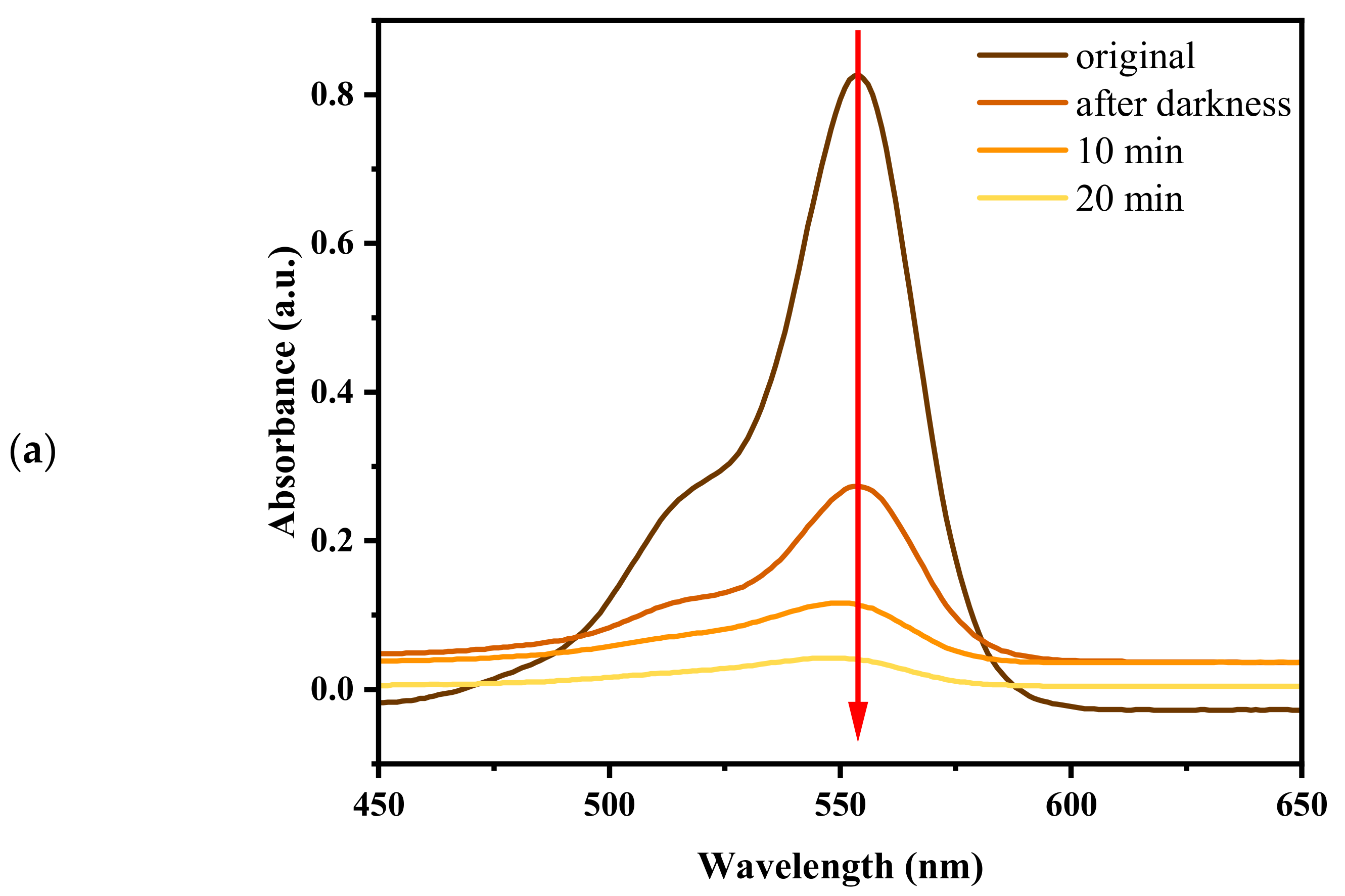
2.2. Spatial Charge Separation Capability
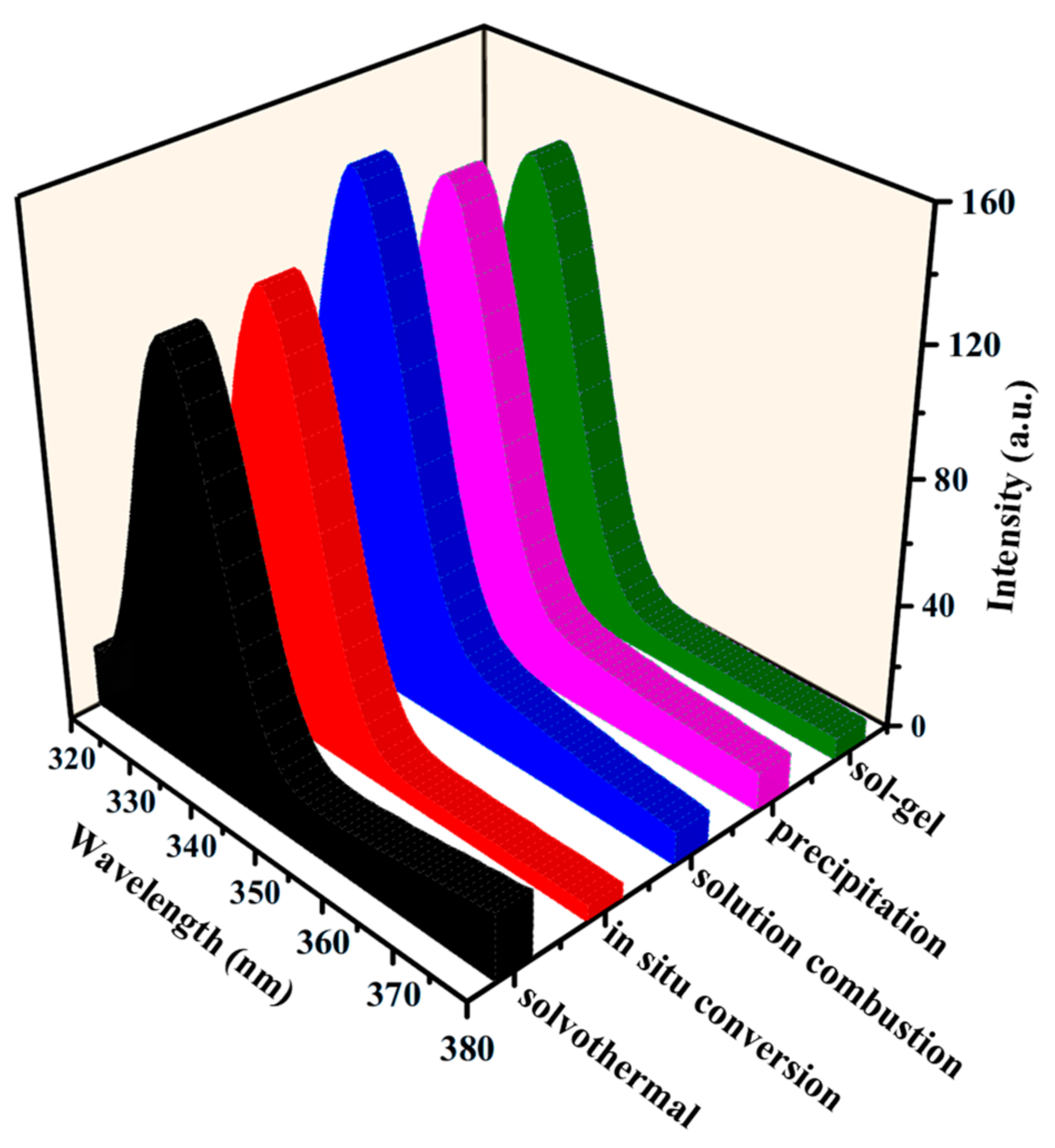
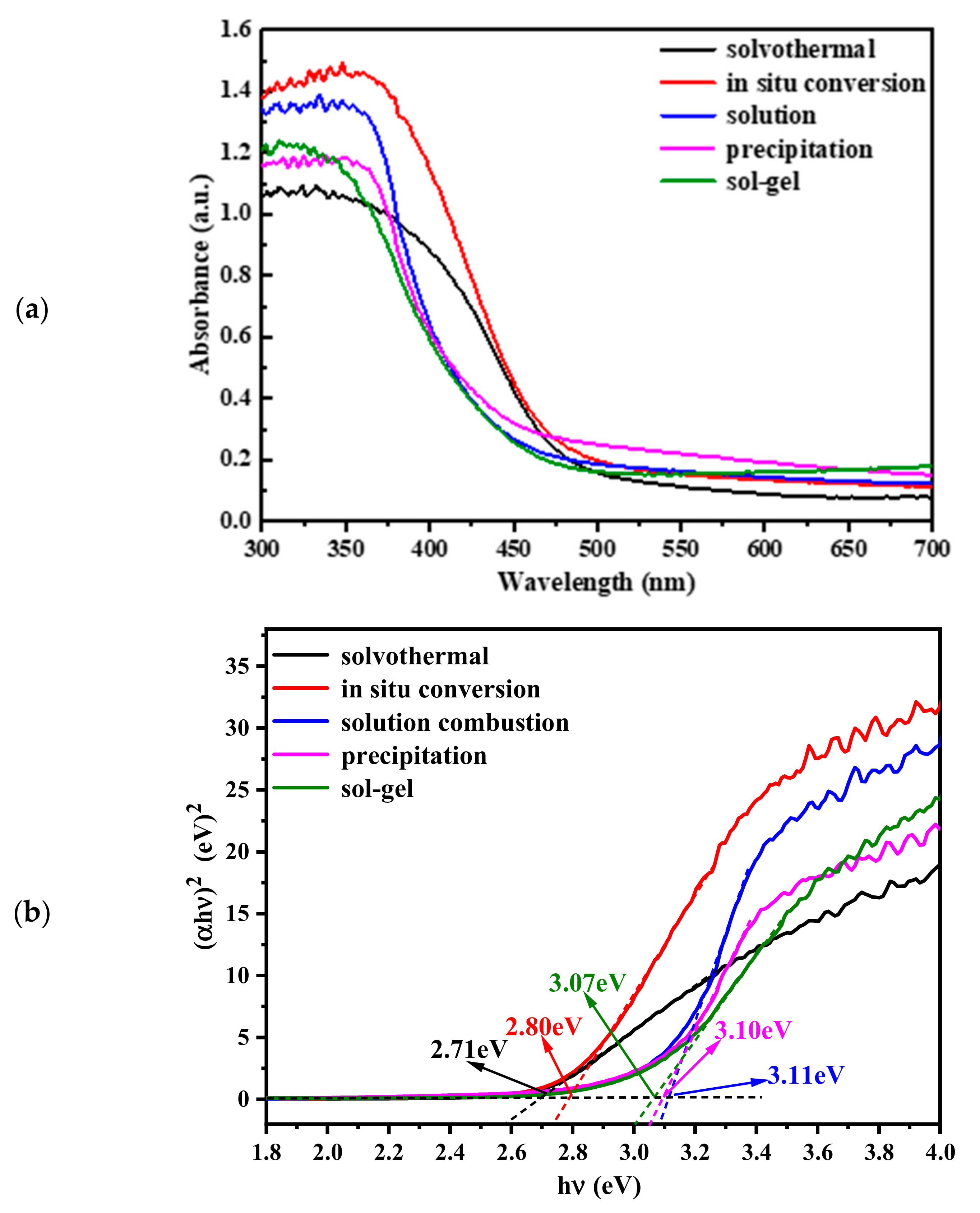
2.3. Optical Properties
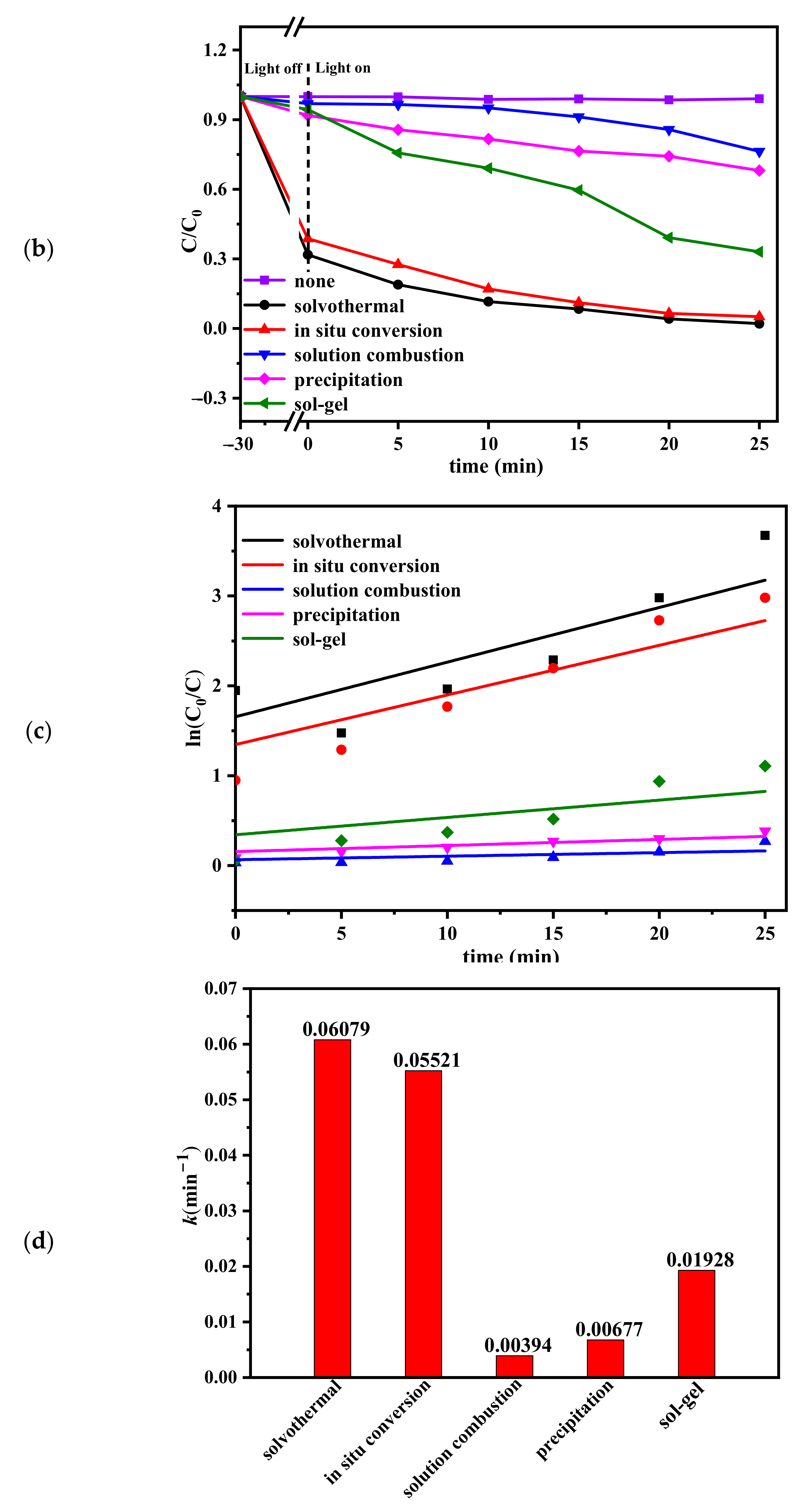
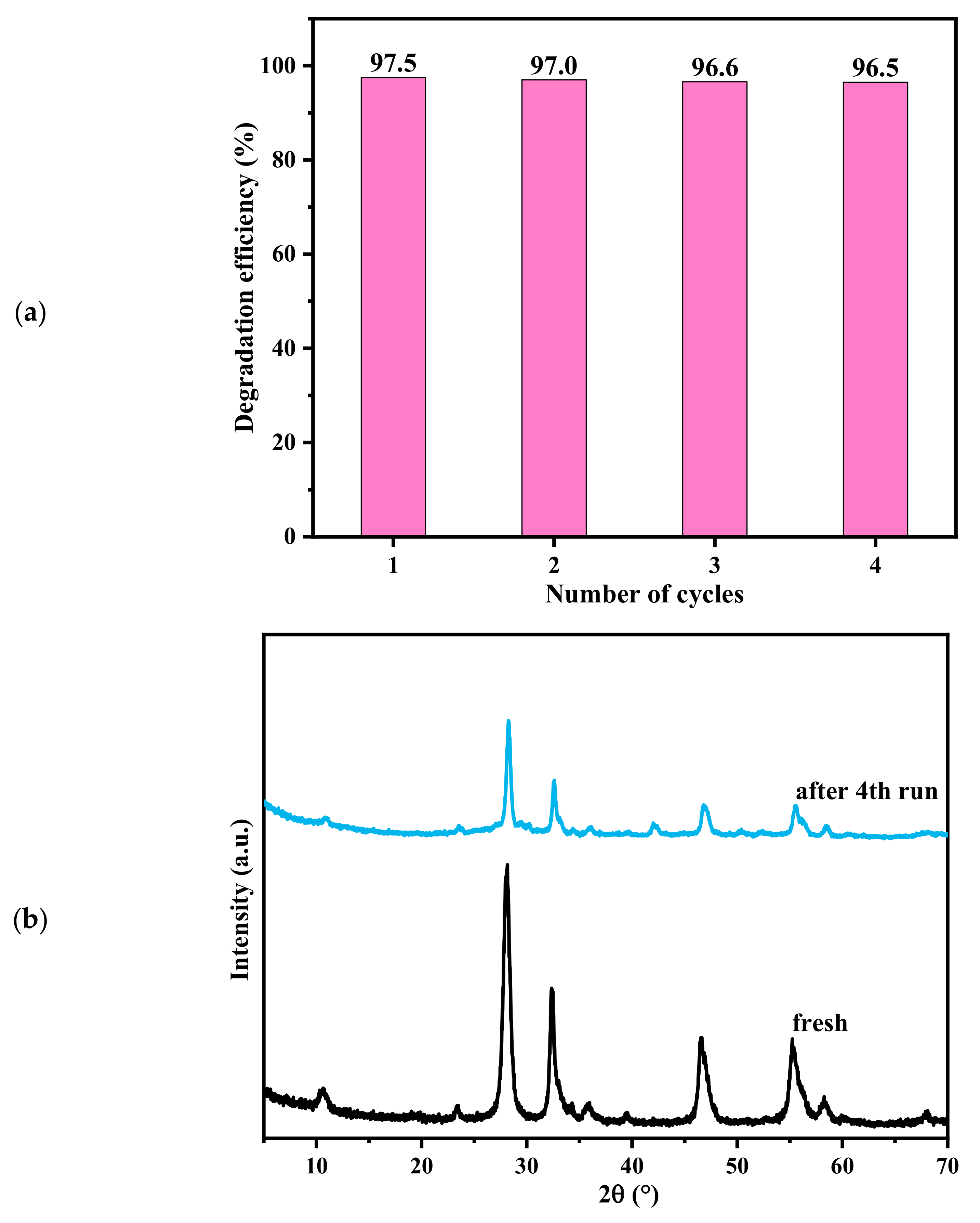
2.4. Photocatalytic Performances and Stability
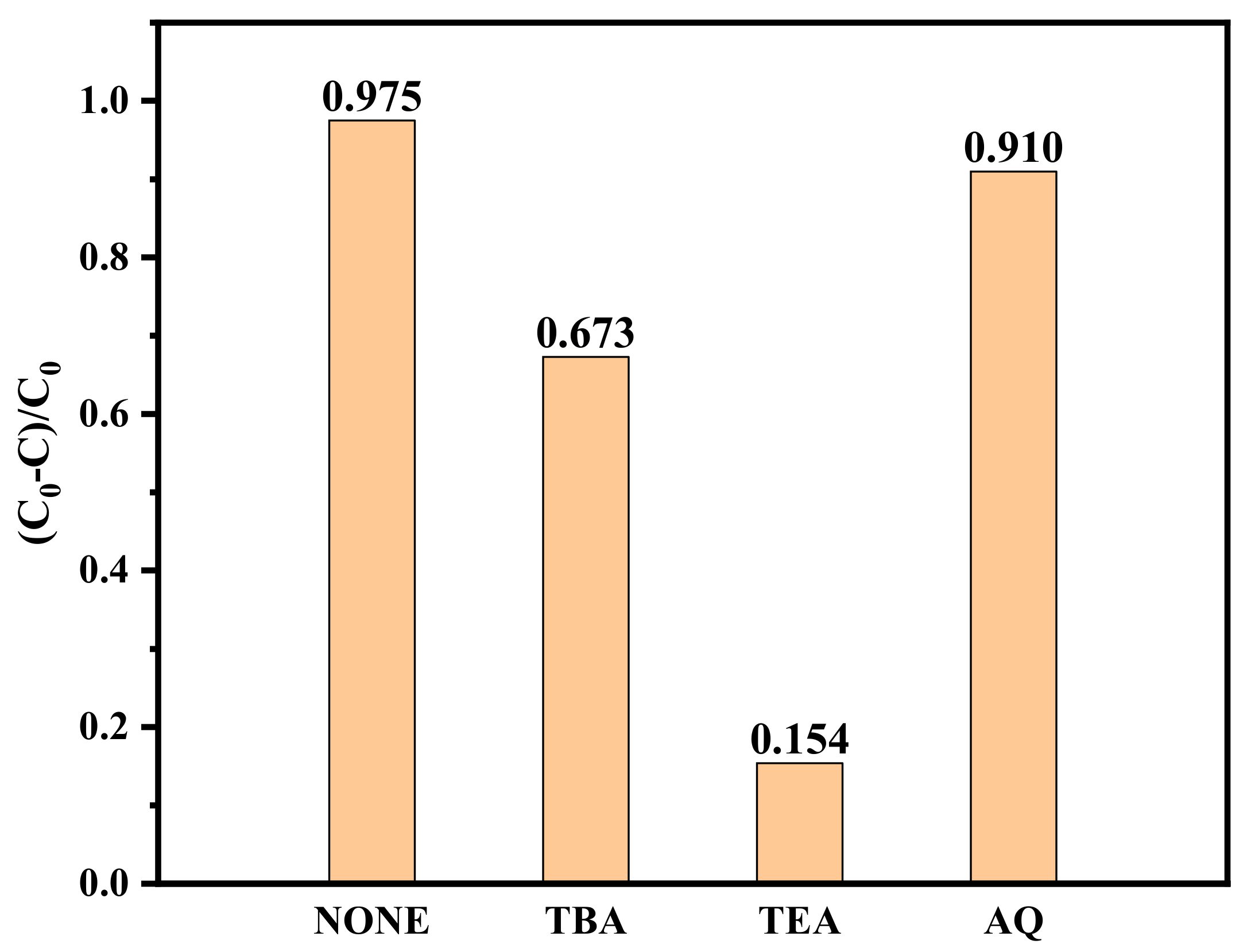
2.5. Photocatalytic Mechanism
3. Materials and Methods
3.1. Materials
3.2. Preparation of Bi2MoO6
3.3. Sample Characterization
3.4. Photoelectrochemical Measurements
3.5. Photocatalytic Performance
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Qi, S.Y.; Wu, S.Q.; Zhang, K.Y.; Guan, L.; Hu, X.; Li, H.Y. Design, preparation and mechanism of Bi2MoO6-modified cobalt-doped CdS solid solution photocatalysts. J. Alloys Compd. 2024, 1003, 175565. [Google Scholar] [CrossRef]
- Sui, G.Z.; Li, J.L.; Du, L.J.; Zhuang, Y.; Zhang, Y.L.; Zou, Y.F.; Li, B. Preparation and characterization of g-C3N4/Ag-TiO2 ternary hollow sphere nano heterojunction catalyst with high visible light photocatalytic performance. J. Alloys Compd. 2020, 823, 153851. [Google Scholar] [CrossRef]
- Luo, C.W.; Liao, M.C.; Peng, D.Y.; Xiong, J.; Zeng, H.Y. Fe-doped MOF(Ti)/Bi2MoO6 heterostructure for boosting oxidation/reduction activities under visible light. Colloid Surf. A 2024, 19, 118565. [Google Scholar] [CrossRef]
- Dong, S.; Feng, J.L.; Fan, M.H.; Pi, Y.Q.; Hu, L.M.; Han, X.; Liu, M.L.; Sun, J.Y.; Sun, J.H. Recent developments in heterogeneous photocatalytic water treatment using visible light-responsive photocatalysts: A review. Rsc Adv. 2015, 5, 14610–14630. [Google Scholar] [CrossRef]
- Yang, S.; Lu, Q.P.; Wang, F.G.; Zhi, Y.H.; Chen, J.Y.; Wang, Y.H.; Zhang, H.; Yin, H.Q.; Sun, P.; Cao, W.B. S-scheme SnO/TiO2 heterojunction with high hole mobility for boosting photocatalytic degradation of gaseous benzene. Chem. Eng. J. 2023, 478, 147345. [Google Scholar] [CrossRef]
- Buddiga, L.R.; Gajula, G.R.; Sailaja, B.B.V.; Ch, M.L.V.P. Visible light photocatalytic exploit of P/Zr doped TiO2 nano particles for dye degradation of rose Bengal. Appl. Surf. Sci. 2023, 18, 100492. [Google Scholar] [CrossRef]
- Lu, P.; Peng, Y.Q.; Yang, Y.; Chen, S.Y.; Shang, J.; Yang, C.; Xu, M.M.; Bai, J.W.; Zhao, Z.L.; Hu, X.L. Visible-light-driven photocatalytic for degrading tetracycline wastewater by BiOI/Bi2O3 Z-scheme heterojunction. J. Environ. Chem. Eng. 2024, 12, 114395. [Google Scholar] [CrossRef]
- Xu, Y.X.; Chen, T.W. Development of nanostructured based ZnO@WO3 photocatalyst and its photocatalytic and electrochemical properties: Degradation of Rhodamine B. Int. J. Electrochem. Sci. 2023, 18, 100055. [Google Scholar] [CrossRef]
- Lin, Z.X.; Xu, J.X.; Gu, H.J.; Huang, J.Y.; Lin, J.H.; Shao, J.; Wang, D.D.; Li, H.J. A review on research progress in photocatalytic degradation of organic pollutants by Bi2MoO6. J. Environ. Chem. Eng. 2023, 11, 110911. [Google Scholar] [CrossRef]
- Hajiali, M.; Farhadian, M.; Tangestaninejad, S.; Khosravi, M. Synthesis and characterization of Bi2MoO6/MIL-101(Fe) as a novel composite with enhanced photocatalytic performance: Effect of water matrix and reaction mechanism. Adv. Power Technol. 2022, 33, 103546. [Google Scholar] [CrossRef]
- Ly, M.J.; Wang, C.M.; Rong, Y.Z.; Wei, J.W.; Yang, Y.K.; Liu, Y.Y.; Wei, G.X.; Zhang, Q.; Wang, C.; Xiu, J.S. Advances in modification of Bi2MoO6 and its photocatalysis: A review. J. Alloys Compd. 2024, 982, 173759. [Google Scholar] [CrossRef]
- Li, Z.X.; Qi, W.K.; Li, L.D.; Ma, Z.Y.; Lai, W.D.; Li, L.; Jin, X.S.; Zhang, Y.C.; Zhang, W.M. Preparation of carbon nanofibers supported Bi2MoO6 nanosheets as counter electrode materials on Titanium mesh substrate for dye-sensitized solar cells. Sol. Energy 2021, 214, 502–509. [Google Scholar] [CrossRef]
- Wu, K.D.; He, X.X.; Ly, A.; Lahem, D.; Debliquy, M.; Zhang, C. Highly sensitive and selective gas sensors based on 2D/3D Bi2MoO6 micro-nano composites for trimethylamine biomarker detection. Appl. Surf. Sci. 2023, 629, 157443. [Google Scholar] [CrossRef]
- Murugan, R. Investigation on ionic conductivity and Raman spectra of γ-Bi2MoO6. Phys. B 2004, 352, 227–232. [Google Scholar] [CrossRef]
- Ali, H.E.; Khairy, Y. Facile low temperature synthesis and characterization of bismuth molybdate (Bi2MoO6) nanostructures: An effect surfactant concentration. Optik 2019, 178, 90–96. [Google Scholar]
- Ashrafi, M.; Farhadian, M.; Nazar, A.R.S.; Hajiali, M.; Noorbaksh, A. The tetracycline degradation in a photocatalytic fuel cell microreactor using a ZnO nanorod/Bi2MoO6/ZIF-67 photocatalyst responsive to visible light. Energy Convers. Manag. 2024, 312, 118565. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, Y.; Yu, J.; Yu, J.Q.; Yang, D.J.; Ng, T.W.; Wong, P.K.; Yu, J.C. Enhanced photocatalytic water disinfection propertiesof Bi2MoO6-RGO nanocomposites under visible light irradiation. Nanoscale 2013, 5, 6307–6310. [Google Scholar] [CrossRef]
- Yang, F.; Elnabawy, A.O.; Schimmenti, R.; Song, P.; Wang, J.W.; Peng, Z.Q.; Yao, S.; Deng, R.P.; Song, S.Y.; Lin, Y.; et al. Bismuthene for highly efficient carbon dioxide electroreduction reaction. Nat. Commun. 2020, 11, 1088. [Google Scholar] [CrossRef]
- Peng, Y.H.; Zhang, Y.; Tian, F.H.; Zhang, J.Q.; Yu, J.Q. Structure Tuning of Bi2MoO6 and Their Enhanced Visible Light Photocatalytic Performances. Crit. Rev. Solid State 2017, 42, 347–372. [Google Scholar] [CrossRef]
- Wang, M.; Han, J.; Guo, P.Y.; Sun, M.Z.; Zhang, Y.; Tong, Z.; You, M.Y.; Lv, C.M. Hydrothermal synthesis of B-doped Bi2MoO6 and its high photocatalytic performance for the degradation of Rhodamine B. J. Phys. Chem. Solids 2018, 113, 86–93. [Google Scholar] [CrossRef]
- Cruz, A.M.L.; Alfaro, S.O. Synthesis and characterization of γ-Bi2MoO6 prepared by co-precipitation: Photoassisted degradation of organic dyes under vis-irradiation. J. Mol. Catal. A-Chem. 2010, 320, 85–91. [Google Scholar] [CrossRef]
- Li, J.; Nie, X.; Meng, L.J.; Zhang, X.J.; Bai, L.M.; Chai, D.F.; Zhang, W.Z.; Zhang, Z.F.; Dong, G.H. Microwave-assisted hydrothermal synthesis of Ag/Bi2MoO6/ZnO heterojunction with nano Ag as electronic accelerator pump for high-efficienty photocatalytic degradation of levofloxacin. Appl. Surf. Sci. 2024, 678, 161143. [Google Scholar] [CrossRef]
- Wang, Y.J.; Wang, Q.Y.; Zhang, H.; Wu, Y.; Jia, Y.; Jin, R.C.; Gao, S.M. CTAB-assisted solvothermal construction of hierarchical Bi2MoO6/Bi5O7 Br with improved photocatalytic performances. Sep. Purif. Technol. 2020, 242, 11. [Google Scholar] [CrossRef]
- Liu, Z.; Tian, J.; Yu, C.L.; Fan, Q.Z.; Liu, X.Q. Solvothermal fabrication of Bi2MoO6 nanocrystals with tunable oxygen vacancies and excellent photocatalytic oxidation performance in quinoline production and antibiotics degradation. Chin. J. Catal. 2022, 43, 472–484. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, X.F.; Duan, F.; Chen, Q.M. Preparation and Photocatalytic Properties of Bi2MoO6 Hollow Microspheres. Chin. J. Inorg. Chem. 2015, 31, 2152–2158. [Google Scholar]
- Saha, D.; Madras, G. Solution combustion synthesis of γ(L)-Bi2MoO6 and photocatalytic activity under solar radiation. Mater. Res. Bull. 2011, 46, 1252–1256. [Google Scholar] [CrossRef]
- Hipólito, E.L.; Cruz, A.M.L.; Cuéllar, E.L. Synthesis, characterization, and photocatalytic properties of γ-Bi2MoO6 prepared by co-precipitation assisted with ultrasound irradiation. J. Taiwan Inst. Chem. E 2014, 45, 2749–2754. [Google Scholar] [CrossRef]
- Umapathy, V.; Manikandan, A.; Antony, S.A.; Ramu, P.; Neeraja, P. Structure, morphology and opto-magnetic properties of Bi2MoO6 nano-photocatalyst synthesized by sol-gel method. Trans. Nonferrous Met. Soc. China 2015, 25, 3271–3278. [Google Scholar] [CrossRef]
- Zhao, Z.W.; Zhang, W.D.; Sun, Y.Y.; Yu, J.Y.; Zhang, Y.X.; Wang, H.; Dong, F.; Wu, Z.B. Bi Cocatalyst/Bi2MoO6 microspheres nanohybrid with SPR-promoted visible-light photocatalysis. J. Phys. Chem. C 2016, 120, 11889–11898. [Google Scholar] [CrossRef]
- Zhang, M.Y.; Shao, C.L.; Mu, J.B.; Huang, X.M.; Zhang, Z.Y.; Guo, Z.C.; Zhang, P.; Liu, Y.C. Hierarchical heterostructures of Bi2MoO6 on carbon nanofibers: Controllable solvothermal fabrication and enhanced visible photocatalytic properties. J. Mater. Chem. 2012, 22, 577–584. [Google Scholar] [CrossRef]
- Li, J.L.; Liu, X.J.; Piao, X.Q.; Sun, Z.; Pan, L.K. Novel carbon sphere@Bi2MoO6 core-shell structure for efficient visible light photocatalysis. RSC Adv. 2015, 5, 16592–16597. [Google Scholar] [CrossRef]
- Li, Z.Q.; Chen, X.T.; Xue, Z.L. Bi2MoO6 microstructures: Controllable synthesis, growth mechanism, and visible-light-driven photocatalytic activities. CrystEngComm 2013, 15, 498–508. [Google Scholar] [CrossRef]
- Khazaee, Z.; Khavar, A.H.C.; Mahjou, A.R.; Motaeea, A.; Srivastavac, V.; Sillanpääc, M. Template-confined growth of X-Bi2MoO6 (X: F, Cl, Br, I) nanoplates with open surfaces for photocatalytic oxidation; experimental and DFT insights of the halogen doping. Sol. Energy 2020, 196, 567–581. [Google Scholar] [CrossRef]
- Hao, Z.Q.; Xu, L.L.; Wei, B.; Fan, L.L.; Liu, Y.; Zhang, M.y.; Gao, H. Nanosize α-Bi2O3 decorated Bi2MoO6 via an alkali etching process for enhanced photocatalytic performance. RSC Adv. 2015, 5, 12346–12353. [Google Scholar] [CrossRef]
- Miao, Y.C.; Pan, G.F.; Huo, Y.N.; Li, H.X. Aerosol-spraying preparation of Bi2MoO6: A visible photocatalyst inhollow microspheres with a porous outer shell and enhanced activity. Dyes Pigment. 2013, 99, 382–389. [Google Scholar] [CrossRef]
- Li, H.H.; Liu, C.Y.; Li, K.W.; Wang, H. Preparation, characterization and photocatalytic properties of nanoplate Bi2MoO6 catalysts. J. Mater. Sci. 2008, 43, 7026–7034. [Google Scholar] [CrossRef]

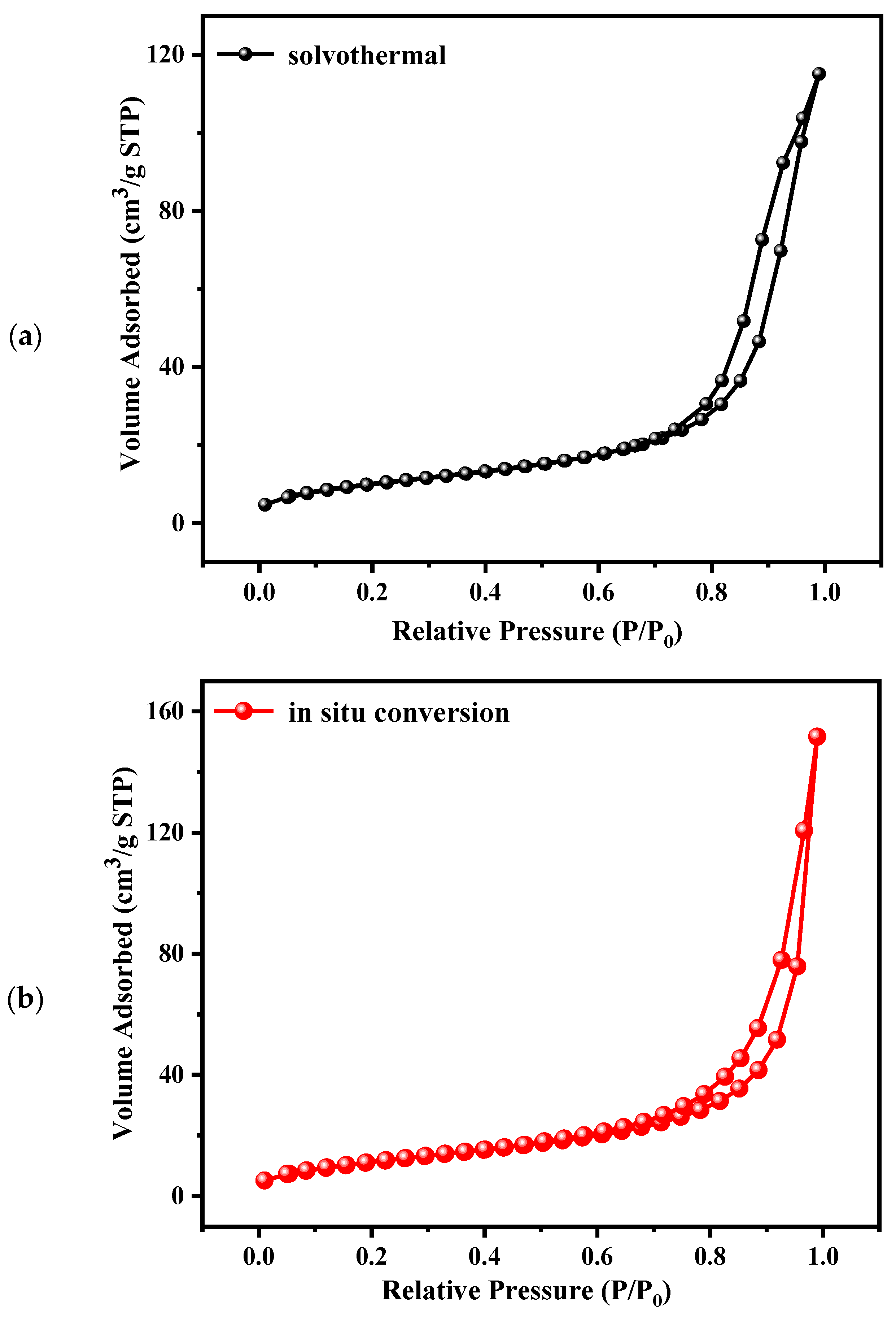
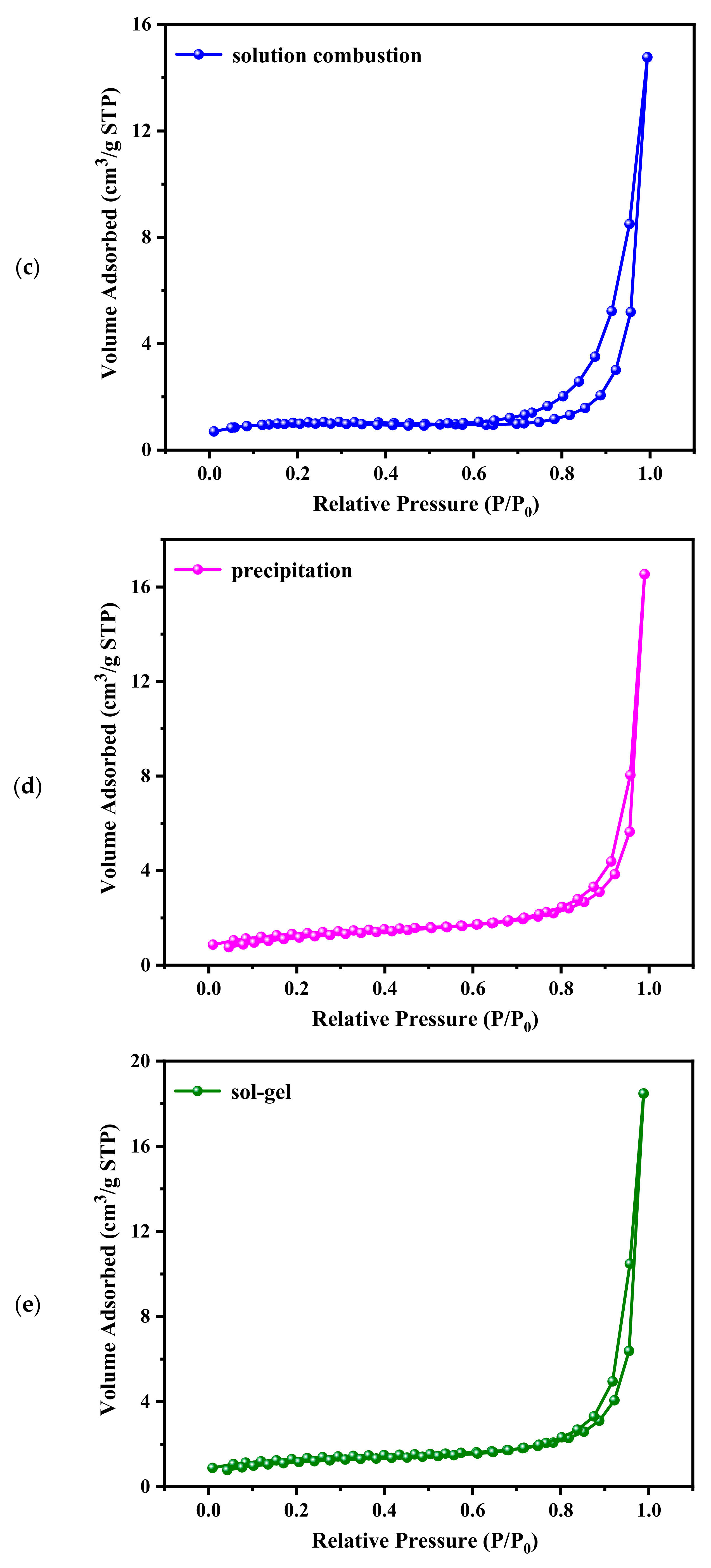
| Samples | BET Surface Area (m2/g) | Average Pore Size (nm) |
| solvothermal | 38.5 | 10.0 |
| in situ conversion | 43.0 | 7.1 |
| solution combustion | 3.3 | 9.8 |
| precipitation | 4.5 | 5.3 |
| sol-gel | 4.4 | 6.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Ge, J.; Liu, W.; Zhang, H.; Li, R. Influence of Preparation Method on Structure and Photocatalytic Performance of Bi2MoO6. Catalysts 2025, 15, 198. https://doi.org/10.3390/catal15030198
Wang Q, Ge J, Liu W, Zhang H, Li R. Influence of Preparation Method on Structure and Photocatalytic Performance of Bi2MoO6. Catalysts. 2025; 15(3):198. https://doi.org/10.3390/catal15030198
Chicago/Turabian StyleWang, Qiuqin, Jinlong Ge, Wei Liu, Hanyu Zhang, and Ruochen Li. 2025. "Influence of Preparation Method on Structure and Photocatalytic Performance of Bi2MoO6" Catalysts 15, no. 3: 198. https://doi.org/10.3390/catal15030198
APA StyleWang, Q., Ge, J., Liu, W., Zhang, H., & Li, R. (2025). Influence of Preparation Method on Structure and Photocatalytic Performance of Bi2MoO6. Catalysts, 15(3), 198. https://doi.org/10.3390/catal15030198






