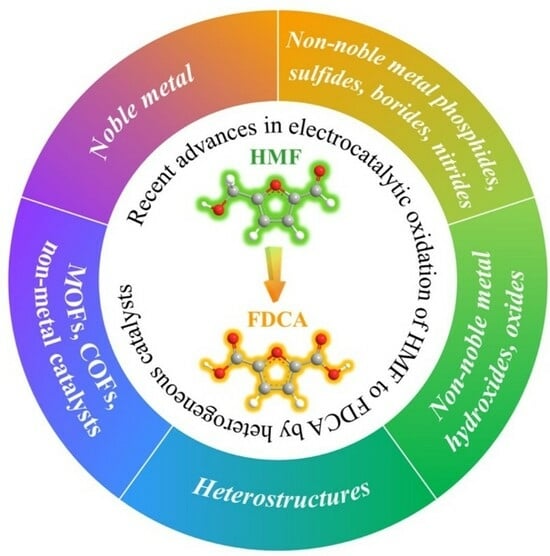
Recent Advances in Electrocatalytic Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid by Heterogeneous Catalysts
Abstract
1. Introduction
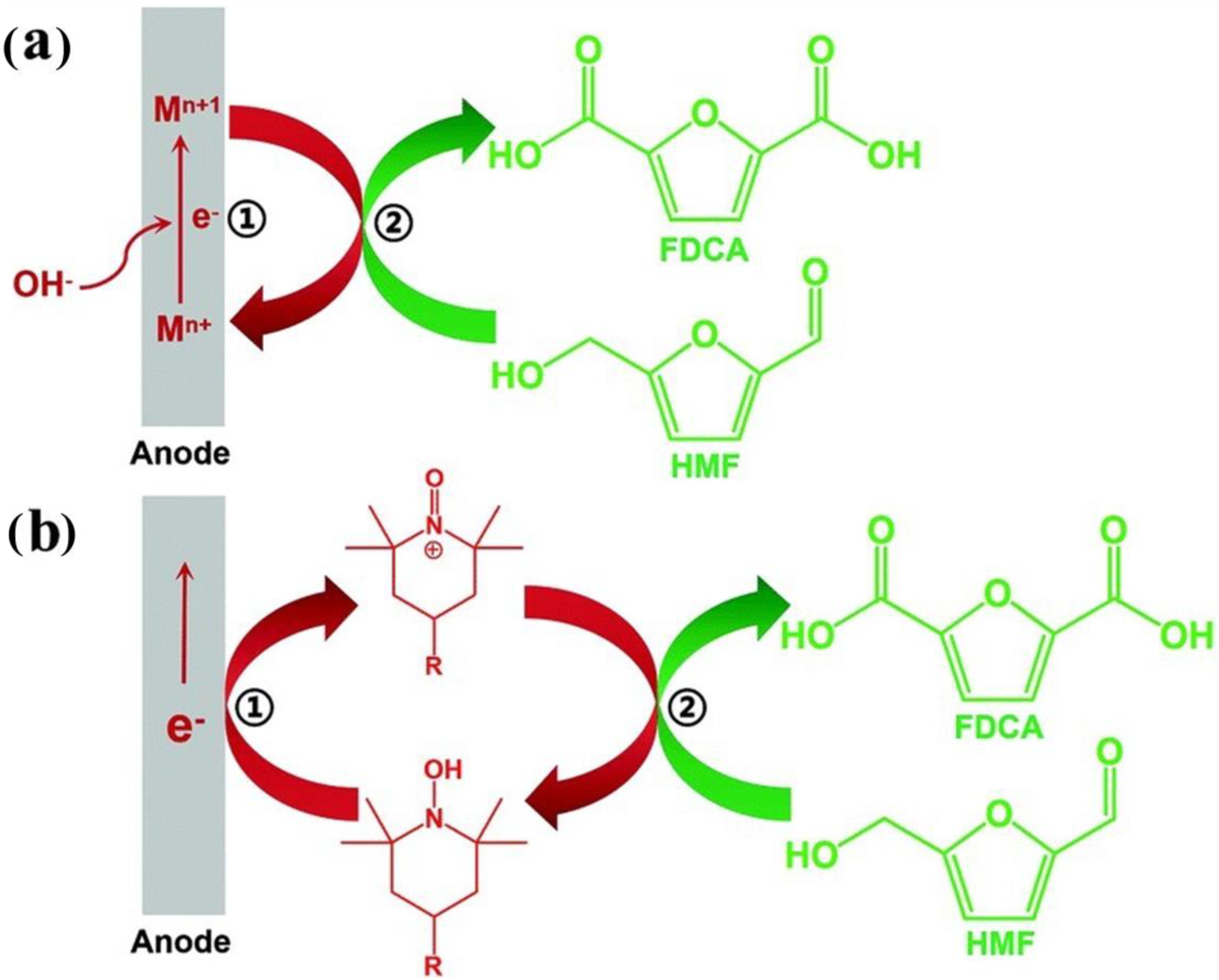
2. Reaction Pathway and Mechanism of HMFOR
2.1. Reaction Pathway
2.2. Reaction Mechanism
2.2.1. Direct Oxidation
2.2.2. Indirect Oxidation
3. Heterogeneous Catalysts for HMFOR to FDCA
3.1. Noble Metal Catalysts
| Entry | Catalyst | HMF (mM) | Potential (V vs. RHE) | HMF Conversion (%) | FDCA Yield (%) | FE (%) | Ref. |
|---|---|---|---|---|---|---|---|
| 1 b | Pt/C | 5 | - | 29 | <1 | - | [16] |
| 2 c | Ru(III)-PEI@ MWCNTs | 1 | 1.34 | - | - | 94 | [18] |
| 3 c | Au/C | 20 | 0.9 | 100 | 1 | - | [17] |
| 4 c | Pd/C | 20 | 0.9 | 97 | 29 | - | [17] |
| 5 c | Pd2Au1/C | 20 | 0.9 | 100 | 64 | - | [17] |
| 6 c | Pd1Au2/C | 20 | 0.9 | 100 | 83 | - | [17] |
| 7 | (AuPd)7 | 5 | 0.82 | 49.3 | 11.1 | 72.8 | [19] |
| 8 | Au7/Pd7 | 5 | 0.82 | 33 | 6.8 | 83.8 | [19] |
| 9 | Pd7/Au7 | 5 | 0.82 | 42.4 | 10.1 | 85.8 | [19] |
| 10 | Pd-NiCo | 50 | 1.38 | 99.6 | 96.5 | 95.9 | [20] |
| 11 | Ir-Co3O4 | 50 | 1.42 | - | 98 | 98 | [21] |
| 12 | Ru1/CoOx | 5 | 1.45 | - | 55 | 55.2 | [22] |
| 13 | Rh-SA/NiFe NMLDH | 50 | 1.30 | 98 | 99.8 | 98.5 | [23] |
| 14 | Ru0.3/NiFe | 5 | 1.48 | 99.43 | 98.68 | - | [24] |
3.2. Non-Noble Metal Catalysts
3.2.1. Non-Noble Metal Phosphides, Sulfides, Borides, and Nitrides
Phosphides
| Type | Catalyst | HMF (mM) | Potential (V vs. RHE) | HMF Conversion (%) | FDCA Yield (%) | FE (%) | Ref. |
|---|---|---|---|---|---|---|---|
| Phosphides | Co-P/CF | 50 | 1.423 | ~100 | ~90 | 93 | [33] |
| Ni2P NPA/NF | 10 | 1.423 | ~100 | 98 | >98 | [34] | |
| NiP-Al2O3/NF | 0.3 | 1.45 | 97.8 | 99.6 | - | [35] | |
| Ni-PA | 10 | 1.60 | 100 | 99.1 | 90 | [36] | |
| NiP/NF | 100 | 1.49 | 96 | 96 | 96 | [37] | |
| NiP@Ni/C | 15 | 1.42 | >90 | >90 | 97 | [38] | |
| LN-400-P-350 | 10 | 1.49 | ~100 | ~100 | >90 | [39] | |
| Ce–CoP | 10 | 1.44 | 100 | 98 | 96.4 | [40] | |
| Ce-Co2P@NC | 10 | 1.20 | 99.5 | 99.3 | 98.5 | [41] | |
| CoNiP-NIE | 10 | 1.50 | - | - | 87.2 | [32] | |
| Mn-5Ni2P | 10 | 1.43 | 100 | 98 | 97.8 | [42] | |
| Mn-FePSe3/NS | 10 | 1.30 | 98.8 | - | 92 | [43] | |
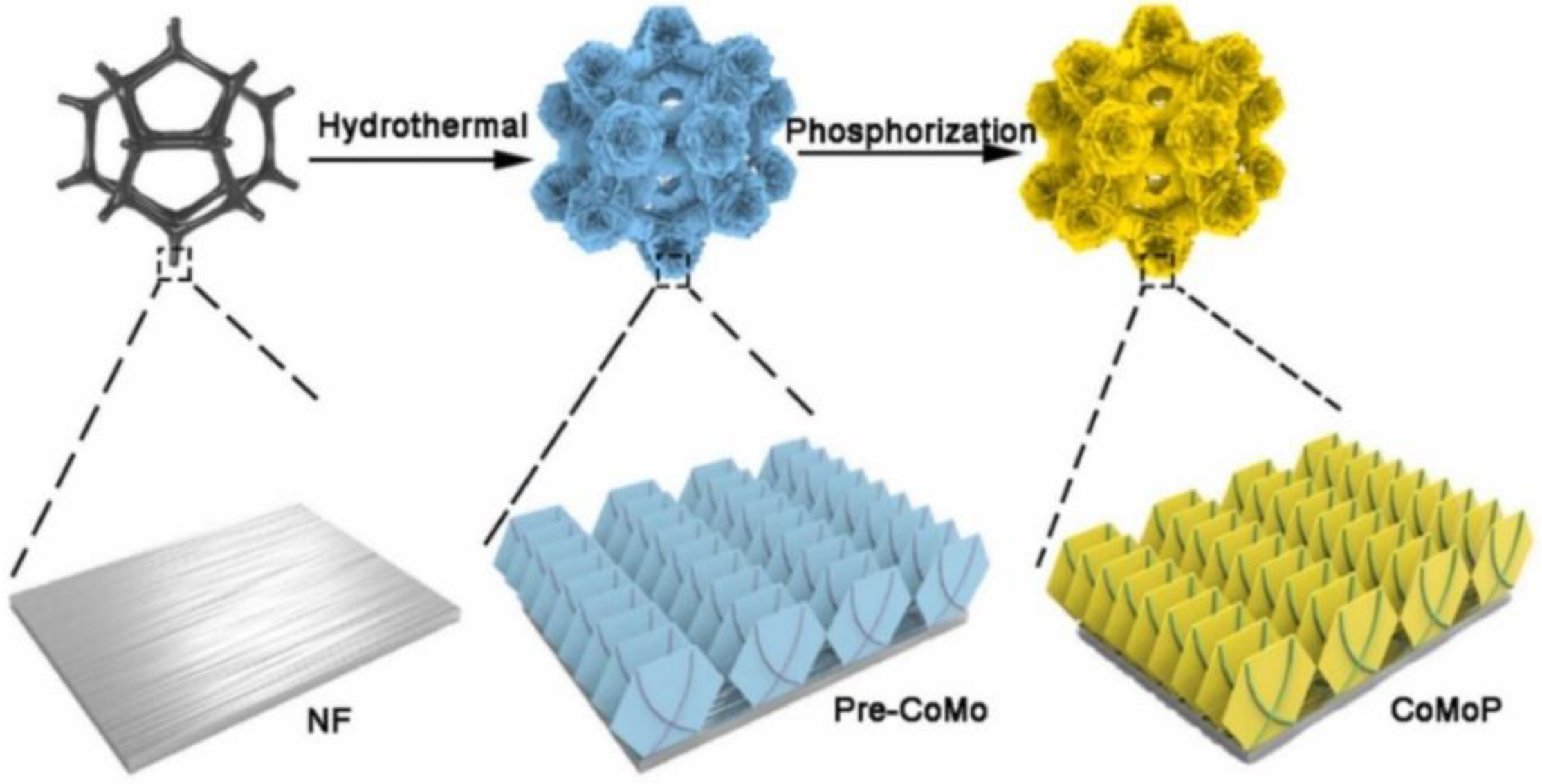
| CoMoP | 100 | 1.36 | 99.9 | 95.8 | 93 | [44] | |
| Sulfides | Ni3S2/NF | 10 | 1.423 | 98 | 98 | 100 | [45] |
| Ni2S3/NF | 10 | 1.498 | 100 | 98 | 94 | [46] | |
| S−Ni@C | 10 | 1.473 | ~100 | 96 | 96 | [47] | |
| NiCo–S | 10 | 1.45 | 99.1 | 97.1 | 96.4 | [48] | |
| Co0.4NiS@NF | 10 | 1.45 | 100 | >99 | >99 | [49] | |
| Co2NiS | 10 | 1.45 | 84.5 b | 54 b | - | [50] | |
| Mn0.2NiS/GF | 100 | 1.48 | 99.3 | 97.6 | 94.2 | [51] | |
| Cu-Ni3S2-R | 20 | 1.40 | ~100 | ~100 | ~100 | [52] | |
| W20-Ni3S2@NF | 10 | 1.42 | 100 | 99.2 | 97.3 | [53] | |
| Borides | NixB/NF | 10 | 1.45 | 100 | 98.5 | ~100 | [54] |
| NiBx | 10 | 1.426 | ≥99 | ≥99 | ≥99 | [55] | |
| NiBx–P0.07 | 10 | 1.464 | >99 b | 90.6 b | 92.5 b | [56] | |
| Nitrides | Ni3N@C | 10 | 1.45 | - | 98 | 99 | [57] |
| Ni3N | 50 | 1.47 | 92 | 92 | - | [58] | |
| Co4N/NC@CC | 10 | 1.38 | 99.2 | 98.6 | 97.8 | [59] |
Sulfides
Borides
Nitrides
3.2.2. Non-Noble Metal Hydroxides and Oxides
Hydroxides
| Type | Catalyst | HMF (mM) | Potential (V vs. RHE) | HMF Conversion (%) | FDCA Yield (%) | FE (%) | Ref. |
|---|---|---|---|---|---|---|---|
| hydroxides | NiFe LDH | 10 | 1.23 | 99 | 98 | 99.4 | [64] |
| NiCoFe LDHs | 10 | 1.54 | 95.5 b | 84.9 b | ~90 b | [65] | |
| d-NiFe LDH/CP | 10 | 1.48 | 97.35 | 96.8 | 84.47 | [66] | |
| NixSey–NiFe LDH@NF | 10 | 1.423 | 99.6 | 99.3 | 98.9 | [67] | |
| NiFe LDH/CoCH/NF | 5 | 1.58 | 98.8 | 98.6 | 98.1 | [68] | |
| Ni3V1-LDHs | 100 | - | 83.5 | 79.5 | - | [69] | |
| CF-Cu(OH)2 | 100 | 0.8 | ~100 | 98.7 | ~100 | [70] | |
| 14%Ce-Ni(OH)2 | 10 | 0.45 | 100 | - | 86.6 | [71] | |
| Cr-Ni(OH)2/NF | 10 | 1.47 | ~100 | >98 | - | [72] | |
| oxides | CoNW/NF | 100 | 1.504 | 100 | 96.8 | 96.6 | [73] |
| mesoporous δ-MnO2 | 10 | 1.35 | 100 | 98 | 98 | [74] | |
| CuMn2O4 | 10 | 1.31 | 100 | - | 96 | [75] | |
| CuCo2O4 | 50 | 1.45 | - | 93.7 | 94 | [76] | |
| BiCoO-NA/NF | 10 | 1.30 | ~100 | - | 97.7 | [77] | |
| Co3O4-VO | 5 | 1.52 | 90 | 61 | 56 | [78] | |
| N-Co3O4/NF-2 | 10 | 1.423 | 99.5 | 96.4 | 97.3 | [79] | |
| CoOxHy-MA | 5 | 1.52 | - | 98 | 83 | [80] | |
| NiO-N/C | 10 | 1.473 | 99 | 84 | 96 | [81] |
Oxides
3.2.3. Heterostructures
| Catalyst | HMF (mM) | Potential (V vs. RHE) | HMF Conversion (%) | FDCA Yield (%) | FE (%) | Ref. |
|---|---|---|---|---|---|---|
| MoO2–FeP@C | 10 | 1.424 | 99.4 | 98.6 | 97.8 | [83] |
| NiO-Co3O4 | 10 | 1.45 | ~100 | 98 | 96 | [84] |
| NiSe@NiOx | 10 | 1.423 | 98 | 96 | 97 | [85] |
| Ni3N-V2O3 | 10 | 1.40 | 97.4 | 96.1 | - | [86] |
| CoP-CoOOH | 150 | 1.42 | 98.3 | 96.3 | 96.3 | [87] |
| Y-Co–CoSx@CN | 5 | 1.29 | 100 b | 96 b | 93.5 b | [88] |
| CoP/Ni2P-NiCoP@NC-600 | 5 | 1.32 | - | 98.1 b | 97.6 b | [89] |
| Ni(OH)2-NiOOH/NiFeP | 10 | 1.435 | 99.4 | 99.4 | 94.62 | [90] |
| Ni3N−NiMoN/CC | 10 | 1.40 | 100 | 98 | ~100 | [91] |
| t-Ni-P@POC | 10 | 1.42 | 100 | 99.9 | 99.7 | [92] |
| Co-NixP@C | 10 | 1.38 | ~100 | ~100 | 98.9 | [93] |
| NF@Co3O4/CeO2 | 50 | 1.40 | 98 | 94.5 | 97.5 | [94] |
| NF/Co4N@CeO2 | 10 | 1.425 | 91.1 | 93.6 | 84.5 | [95] |
| Cu2P7-CoP | 10 | 1.43 | 100 | 98.8 | 98 | [96] |
| NiSx/Ni2P | 10 | 1.46 | ~100 | 98.5 | 95.1 | [97] |
| NiFeP@NiFe(OH)x | 10 | 1.45 | - | - | 94.42 | [98] |
| a-Ni(OH)2–Cu2O/NF-6 | 50 | 1.424 | - | 91 | 98 | [99] |
| Ni/Ni0.2Mo0.8N/NF | 50 | 1.423 | ~100 | 98.5 | ~100 | [100] |
| Ni-VN/NF | 10 | 1.402 | >99 | 99 | >98 | [101] |
| NiCo2@MoO2-NF | 10 | 1.40 | ~100 | 99.6 | 99.4 | [102] |
| NiOOH-coated Cu(OH)2 | 5 | 1.40 | 100 c | 98.3 c | 98.3 c | [103] |
3.2.4. Other Catalysts
MOFs
| Type | Catalyst | HMF (mM) | Potential (V vs. RHE) | HMF Conversion (%) | FDCA Yield (%) | FE (%) | Ref. |
|---|---|---|---|---|---|---|---|
| MOFs | NiCoBDC-NF | 10 | 1.55 | - | 99 b | 78.8 b | [108] |
| Co(OH)2@ZIF-67 | 10 | 1.42 | 90.9 | 81.8 | 83.6 | [109] | |
| CoNiFe-MOFs/NF | 10 | 1.40 | 100 | 99.76 | 100 | [110] | |
| Rbf-Ni-MOF | 10 | 0.8 | - | - | 95 | [111] | |
| Co-CAT | 10 | 1.42 | - | 99.3 | 97.6 | [112] | |
| Ni-CAT | 10 | 1.42 | - | 98.7 | 86.8 | [112] | |
| CF-Ni-MOF/Ag | 10 | 1.623 | ~100 | - | 98.6 | [113] | |
| CF-CuO/Ni-BTC MOF | 10 | 0.55 | ~100 | 99.9 | 91 | [114] | |
| NiCoFeS-MOF | 50 | 1.39 | 100 | 99 | 99 | [115] | |
| COFs | TpBpy-Ni@FTO | 0.5 | 1.55 | 96 c | 58 c | - | [116] |
| non-metal | B–N codoped porous carbons (BNC-2) | 5 | 1.90 | 71 b | 57 b | - | [117] |
COFs
Non-Metal Catalysts
4. Conclusions and Prospects
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Su, T.; Zhao, D.; Wang, Y.; Lü, H.; Varma, R.S.; Len, C. Innovative Protocols in the Catalytic Oxidation of 5-Hydroxymethylfurfural. ChemSusChem 2021, 14, 266–280. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Mu, T. Electrochemical oxidation of biomass derived 5-hydroxymethylfurfural (HMF): Pathway, mechanism, catalysts and coupling reactions. Green Chem. 2021, 23, 4228–4254. [Google Scholar] [CrossRef]
- Zhao, Y.; Cai, M.; Xian, J.; Sun, Y.; Li, G. Recent advances in the electrocatalytic synthesis of 2,5-furandicarboxylic acid from 5-(hydroxymethyl)furfural. J. Mater. Chem. A 2021, 9, 20164–20183. [Google Scholar] [CrossRef]
- Guo, M.; Lu, X.; Xiong, J.; Zhang, R.; Li, X.; Qiao, Y.; Ji, N.; Yu, Z. Alloy-Driven Efficient Electrocatalytic Oxidation of Biomass-Derived 5-Hydroxymethylfurfural towards 2,5-Furandicarboxylic Acid: A Review. ChemSusChem 2022, 15, e202201074. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, X.; Gan, L.; Pan, L.; Shi, C.; Huang, Z.-F.; Zhang, X.; Zou, J.-J. Advances in Selective Electrochemical Oxidation of 5-Hydroxymethylfurfural to Produce High-Value Chemicals. Adv. Sci. 2023, 10, 2205540. [Google Scholar] [CrossRef]
- Jing, Y.; Guo, Y.; Xia, Q.; Liu, X.; Wang, Y. Catalytic Production of Value-Added Chemicals and Liquid Fuels from Lignocellulosic Biomass. Chem 2019, 5, 2520–2546. [Google Scholar] [CrossRef]
- Lin, Z.; Chen, X.; Lu, L.; Yao, X.; Zhai, C.; Tao, H. Recent advances in electrocatalytic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid: Mechanism, catalyst, coupling system. Nanotechnol. Rev. 2023, 12, 20220518. [Google Scholar] [CrossRef]
- Ge, R.; Li, J.; Duan, H. Recent advances in non-noble electrocatalysts for oxidative valorization of biomass derivatives. Sci. China Mater. 2022, 65, 3273–3301. [Google Scholar] [CrossRef]
- Totaro, G.; Sisti, L.; Marchese, P.; Colonna, M.; Romano, A.; Gioia, C.; Vannini, M.; Celli, A. Current Advances in the Sustainable Conversion of 5-Hydroxymethylfurfural into 2,5-Furandicarboxylic Acid. ChemSusChem 2022, 15, e202200501. [Google Scholar] [CrossRef]
- Jiang, X.; Li, W.; Liu, Y.; Zhao, L.; Chen, Z.; Zhang, L.; Zhang, Y.; Yun, S. Electrocatalytic oxidation of 5-hydroxymethylfurfural for sustainable 2,5-furandicarboxylic acid production—From mechanism to catalysts design. SusMat 2023, 3, 21–43. [Google Scholar] [CrossRef]
- Meng, Y.; Yang, S.; Li, H. Electro- and Photocatalytic Oxidative Upgrading of Bio-based 5-Hydroxymethylfurfural. ChemSusChem 2022, 15, e202102581. [Google Scholar] [CrossRef]
- German, D.; Pakrieva, E.; Kolobova, E.; Carabineiro, S.A.C.; Stucchi, M.; Villa, A.; Prati, L.; Bogdanchikova, N.; Cortés Corberán, V.; Pestryakov, A. Oxidation of 5-Hydroxymethylfurfural on Supported Ag, Au, Pd and Bimetallic Pd-Au Catalysts: Effect of the Support. Catalysts 2021, 11, 115. [Google Scholar] [CrossRef]
- Chen, W.; Xie, C.; Wang, Y.; Zou, Y.; Dong, C.-L.; Huang, Y.-C.; Xiao, Z.; Wei, Z.; Du, S.; Chen, C.; et al. Activity Origins and Design Principles of Nickel-Based Catalysts for Nucleophile Electrooxidation. Chem 2020, 6, 2974–2993. [Google Scholar] [CrossRef]
- Ma, Z.; Liu, S.; Tang, N.; Song, T.; Motokura, K.; Shen, Z.; Yang, Y. Coexistence of Fe Nanoclusters Boosting Fe Single Atoms to Generate Singlet Oxygen for Efficient Aerobic Oxidation of Primary Amines to Imines. ACS Catal. 2022, 12, 5595–5604. [Google Scholar] [CrossRef]
- Ma, Z.; Song, T.; Yuan, Y.; Yang, Y. Synergistic catalysis on Fe–Nx sites and Fe nanoparticles for efficient synthesis of quinolines and quinazolinones via oxidative coupling of amines and aldehydes. Chem. Sci. 2019, 10, 10283–10289. [Google Scholar] [CrossRef]
- Vuyyuru, K.R.; Strasser, P. Oxidation of biomass derived 5-hydroxymethylfurfural using heterogeneous and electrochemical catalysis. Catal. Today 2012, 195, 144–154. [Google Scholar] [CrossRef]
- Chadderdon, D.J.; Xin, L.; Qi, J.; Qiu, Y.; Krishna, P.; More, K.L.; Li, W. Electrocatalytic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid on supported Au and Pd bimetallic nanoparticles. Green Chem. 2014, 16, 3778–3786. [Google Scholar] [CrossRef]
- Xu, G.-R.; Batmunkh, M.; Donne, S.; Jin, H.; Jiang, J.-X.; Chen, Y.; Ma, T. Ruthenium(iii) polyethyleneimine complexes for bifunctional ammonia production and biomass upgrading. J. Mater. Chem. A 2019, 7, 25433–25440. [Google Scholar] [CrossRef]
- Park, M.; Gu, M.; Kim, B.-S. Tailorable Electrocatalytic 5-Hydroxymethylfurfural Oxidation and H2 Production: Architecture–Performance Relationship in Bifunctional Multilayer Electrodes. ACS Nano 2020, 14, 6812–6822. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Nie, T.; Song, Z.; Sun, X.; Shen, T.; Bai, S.; Zheng, L.; Song, Y.-F. Pd Loaded NiCo Hydroxides for Biomass Electrooxidation: Understanding the Synergistic Effect of Proton Deintercalation and Adsorption Kinetics. Angew. Chem. Int. Ed. 2023, 62, e202311696. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Liu, T.; Dong, C.-L.; Huang, Y.-C.; Li, Y.; Chen, J.; Zou, Y.; Wang, S. Tuning the Selective Adsorption Site of Biomass on Co3O4 by Ir Single Atoms for Electrosynthesis. Adv. Mater. 2021, 33, 2007056. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Pei, A.; Zhang, S.; Jiang, F.; Jia, Y.; Qin, Q.; Du, R.; Li, Z.; Liu, R.; Qiu, Y.; et al. Atomic-Interface Effect of Single-Atom Ru/CoOx for Selective Electrooxidation of 5-Hydroxymethylfurfural. ACS Appl. Mater. Interfaces 2023, 15, 28036–28043. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Chen, Y.; Sun, M.; Huang, Q.; Sun, K.; Ma, J.; Li, J.; Tan, H.; Li, M.; Pan, Y.; et al. Cooperative Rh-O5/Ni(Fe) Site for Efficient Biomass Upgrading Coupled with H2 Production. J. Am. Chem. Soc. 2023, 145, 17577–17587. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Xin, G.; Hu, W.; Zhang, Z.; Si, C.; Chen, J.; Lu, L.; Peng, Y.; Li, X. Single-atoms Ru/NiFe layered double hydroxide electrocatalyst: Efficient for oxidation of selective oxidation of 5-hydroxymethylfurfural and oxygen evolution reaction. Appl. Catal. B 2023, 339, 123157. [Google Scholar] [CrossRef]
- Han, X.; Zhang, T.; Wang, X.; Zhang, Z.; Li, Y.; Qin, Y.; Wang, B.; Han, A.; Liu, J. Hollow mesoporous atomically dispersed metal-nitrogen-carbon catalysts with enhanced diffusion for catalysis involving larger molecules. Nat. Commun. 2022, 13, 2900. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Jia, C.; Zhao, Y.; Zhao, C. Single atom-based catalysts for electrochemical CO2 reduction. Chin. J. Catal. 2022, 43, 1547–1597. [Google Scholar] [CrossRef]
- Kim, J.; Yoo, J.M.; Lee, H.S.; Sung, Y.-E.; Hyeon, T. Single-atom M–N–C catalysts for oxygen reduction electrocatalysis. Trends Chem. 2021, 3, 779–794. [Google Scholar] [CrossRef]
- Shang, Y.; Xu, X.; Gao, B.; Wang, S.; Duan, X. Single-atom catalysis in advanced oxidation processes for environmental remediation. Chem. Soc. Rev. 2021, 50, 5281–5322. [Google Scholar] [CrossRef]
- Yuan, L.-J.; Sui, X.-L.; Liu, C.; Zhuo, Y.-L.; Li, Q.; Pan, H.; Wang, Z.-B. Electrocatalysis Mechanism and Structure–Activity Relationship of Atomically Dispersed Metal-Nitrogen-Carbon Catalysts for Electrocatalytic Reactions. Small Methods 2023, 7, 2201524. [Google Scholar] [CrossRef]
- Zhang, L.; Meng, Q.; Zheng, R.; Wang, L.; Xing, W.; Cai, W.; Xiao, M. Microenvironment regulation of M-N-C single-atom catalysts towards oxygen reduction reaction. Nano Res. 2023, 16, 4468–4487. [Google Scholar] [CrossRef]
- Yang, G.; Jiao, Y.; Yan, H.; Tian, C.; Fu, H. Electronic Structure Modulation of Non-Noble-Metal-Based Catalysts for Biomass Electrooxidation Reactions. Small Struct. 2021, 2, 2100095. [Google Scholar] [CrossRef]
- Song, Y.; Xie, W.; Song, Y.; Li, H.; Li, S.; Jiang, S.; Lee, J.Y.; Shao, M. Bifunctional integrated electrode for high-efficient hydrogen production coupled with 5-hydroxymethylfurfural oxidation. Appl. Catal. B 2022, 312, 121400. [Google Scholar] [CrossRef]
- Jiang, N.; You, B.; Boonstra, R.; Terrero Rodriguez, I.M.; Sun, Y. Integrating Electrocatalytic 5-Hydroxymethylfurfural Oxidation and Hydrogen Production via Co–P-Derived Electrocatalysts. ACS Energy Lett. 2016, 1, 386–390. [Google Scholar] [CrossRef]
- You, B.; Jiang, N.; Liu, X.; Sun, Y. Simultaneous H2 Generation and Biomass Upgrading in Water by an Efficient Noble-Metal-Free Bifunctional Electrocatalyst. Angew. Chem. Int. Ed. 2016, 55, 9913–9917. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Chen, L.; Ye, S.; Fan, G.; Yang, L.; Zhang, X.; Li, F. Dispersive non-noble metal phosphide embedded in alumina arrays derived from layered double hydroxide precursor toward efficient oxygen evolution reaction and biomass upgrading. J. Mater. Chem. A 2019, 7, 13695–13704. [Google Scholar] [CrossRef]
- Liu, S.; Yuan, X.; Huang, X.; Huang, Y.; Sun, C.; Qian, K.; Zhang, W. Nickel-phytic acid hybrid for highly efficient electrocatalytic upgrading of HMF. Front. Chem. 2023, 11, 1199921. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, B.; Hausmann, J.N.; Beltrán-Suito, R.; Kalra, S.; Laun, K.; Zebger, I.; Driess, M.; Menezes, P.W. A Facile Molecular Approach to Amorphous Nickel Pnictides and Their Reconstruction to Crystalline Potassium-Intercalated γ-NiOOHx Enabling High-Performance Electrocatalytic Water Oxidation and Selective Oxidation of 5-Hydroxymethylfurfural. Small 2023, 19, 2301258. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Salehi, M.; Guo, J.; Seifitokaldani, A. High oxidation state enabled by plated Ni-P achieves superior electrocatalytic performance for 5-hydroxymethylfurfural oxidation reaction. iScience 2022, 25, 104744. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Li, J.; Ma, J.; Liu, D.; Long, R.; Xiong, Y. Highly efficient electrocatalytic biomass valorization over a perovskite-derived nickel phosphide catalyst. Nanoscale Horiz. 2023, 8, 69–74. [Google Scholar] [CrossRef]
- Bi, J.; Ying, H.; Xu, H.; Zhao, X.; Du, X.; Hao, J.; Li, Z. Phosphorus vacancy-engineered Ce-doped CoP nanosheets for the electrocatalytic oxidation of 5-hydroxymethylfurfural. Chem. Commun. 2022, 58, 7817–7820. [Google Scholar] [CrossRef]
- Xie, S.; Fu, H.; Chen, L.; Li, Y.; Shen, K. Carbon-based nanoarrays embedded with Ce-doped ultrasmall Co2P nanoparticles enable efficient electrooxidation of 5-hydroxymethylfurfural coupled with hydrogen production. Sci. China Chem. 2023, 66, 2141–2152. [Google Scholar] [CrossRef]
- Xu, H.; Bi, J.; Sang, T.; Wang, W.; Hao, J.; Li, Z. Mn-doped Ni2P: Nanocrystal-decorated amorphous nanosheets for efficient electrooxidation of 5-hydroxymethylfurfural. Chem. Commun. 2023, 59, 8440–8443. [Google Scholar] [CrossRef]
- Zhang, H.; Qi, G.; Liu, W.; Zhang, S.; Liu, Q.; Luo, J.; Liu, X. Bimetallic phosphoselenide nanosheets as bifunctional catalysts for 5-hydroxymethylfurfural oxidation and hydrogen evolution. Inorg. Chem. Front. 2023, 10, 2423–2429. [Google Scholar] [CrossRef]
- Wang, H.; Niu, C.; Liu, W.; Tao, S. d-Electron tuned CoMoP for enhance 5-hydroxymethylfurfural oxidation and HER. Appl. Catal. B 2024, 340, 123249. [Google Scholar] [CrossRef]
- You, B.; Liu, X.; Jiang, N.; Sun, Y. A General Strategy for Decoupled Hydrogen Production from Water Splitting by Integrating Oxidative Biomass Valorization. J. Am. Chem. Soc. 2016, 138, 13639–13646. [Google Scholar] [CrossRef]
- Wang, W.; Kong, F.; Zhang, Z.; Yang, L.; Wang, M. Sulfidation of nickel foam with enhanced electrocatalytic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid. Dalton Trans. 2021, 50, 10922–10927. [Google Scholar] [CrossRef]
- Kong, F.; Wang, M. Preparation of Sulfur-Modulated Nickel/Carbon Composites from Lignosulfonate for the Electrocatalytic Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid. ACS Appl. Energy Mater. 2021, 4, 1182–1188. [Google Scholar] [CrossRef]
- Zhao, Z.; Guo, T.; Luo, X.; Qin, X.; Zheng, L.; Yu, L.; Lv, Z.; Ma, D.; Zheng, H. Bimetallic sites and coordination effects: Electronic structure engineering of NiCo-based sulfide for 5-hydroxymethylfurfural electrooxidation. Catal. Sci. Technol. 2022, 12, 3817–3825. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, J.; Qi, Y.; Li, W.; Wang, C. Efficient Electrooxidation of 5-Hydroxymethylfurfural Using Co-Doped Ni3S2 Catalyst: Promising for H2 Production under Industrial-Level Current Density. Adv. Sci. 2022, 9, 2200957. [Google Scholar] [CrossRef]
- Guo, C.; Huo, Y.; Zhang, Q.; Wan, K.; Yang, G.; Liu, Z.; Peng, F. MOF Material-Derived Bimetallic Sulfide CoxNiyS for Electrocatalytic Oxidation of 5-Hydroxymethylfurfural. Nanomaterials 2023, 13, 2318. [Google Scholar] [CrossRef]
- Li, S.; Wang, S.; Wang, Y.; He, J.; Li, K.; Xu, Y.; Wang, M.; Zhao, S.; Li, X.; Zhong, X.; et al. Doped Mn Enhanced NiS Electrooxidation Performance of HMF into FDCA at Industrial-Level Current Density. Adv. Funct. Mater. 2023, 33, 2214488. [Google Scholar] [CrossRef]
- Xu, P.; Bao, Z.; Zhao, Y.; Zheng, L.; Lv, Z.; Shi, X.; Wang, H.-E.; Fang, X.; Zheng, H. Anionic Regulation and Heteroatom Doping of Ni-Based Electrocatalysts to Boost Biomass Valorization Coupled with Hydrogen Production. Adv. Energy Mater. 2024, 14, 2303557. [Google Scholar] [CrossRef]
- Wang, S.; Yang, G.; Jiao, Y.; Liu, Y.; Tian, C.; Wu, A.; Yan, H. Tuning electronic structure of Ni3S2 with tungsten doping for high-performance electrooxidation of 5-hydroxymethylfurfural. Sci. China Chem. 2023, 66, 3636–3644. [Google Scholar] [CrossRef]
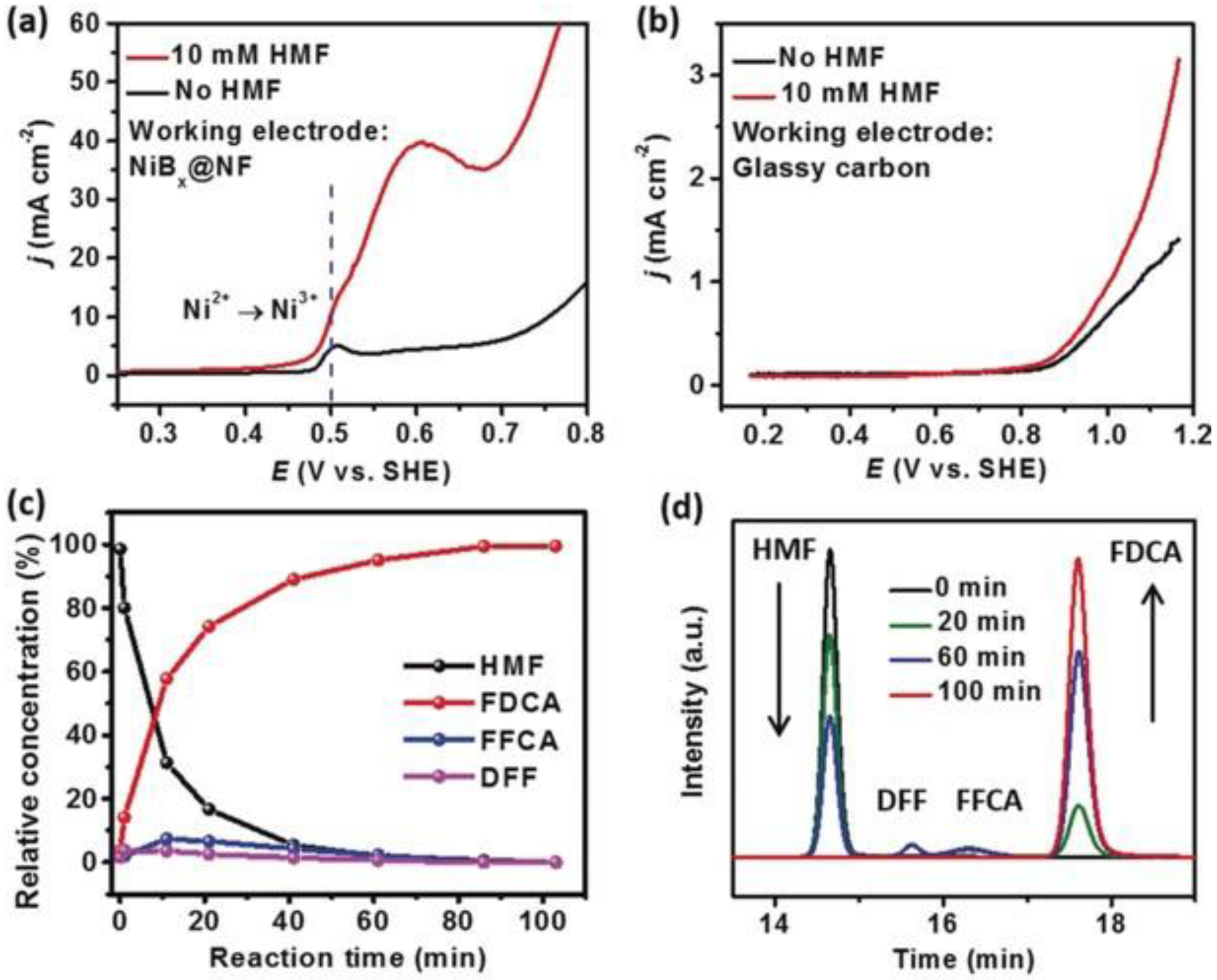
- Barwe, S.; Weidner, J.; Cychy, S.; Morales, D.M.; Dieckhöfer, S.; Hiltrop, D.; Masa, J.; Muhler, M.; Schuhmann, W. Electrocatalytic Oxidation of 5-(Hydroxymethyl)furfural Using High-Surface-Area Nickel Boride. Angew. Chem. Int. Ed. 2018, 57, 11460–11464. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Sheng, X.; Chen, X.; Fang, Z.; Jiang, J.; Wang, M.; Li, F.; Fan, L.; Ren, Y.; Zhang, B.; et al. Paired Electrocatalytic Oxygenation and Hydrogenation of Organic Substrates with Water as the Oxygen and Hydrogen Source. Angew. Chem. Int. Ed. 2019, 58, 9155–9159. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Liu, X.; Wang, H.; Guo, Y.; Wang, Y. Improved Performance of Nickel Boride by Phosphorus Doping as an Efficient Electrocatalyst for the Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid. Ind. Eng. Chem. Res. 2020, 59, 17348–17356. [Google Scholar] [CrossRef]
- Zhang, N.; Zou, Y.; Tao, L.; Chen, W.; Zhou, L.; Liu, Z.; Zhou, B.; Huang, G.; Lin, H.; Wang, S. Electrochemical Oxidation of 5-Hydroxymethylfurfural on Nickel Nitride/Carbon Nanosheets: Reaction Pathway Determined by In Situ Sum Frequency Generation Vibrational Spectroscopy. Angew. Chem. Int. Ed. 2019, 58, 15895–15903. [Google Scholar] [CrossRef]
- Zhou, B.; Dong, C.-L.; Huang, Y.-C.; Zhang, N.; Wu, Y.; Lu, Y.; Yue, X.; Xiao, Z.; Zou, Y.; Wang, S. Activity origin and alkalinity effect of electrocatalytic biomass oxidation on nickel nitride. J. Energy Chem. 2021, 61, 179–185. [Google Scholar] [CrossRef]
- Zhang, D.; Xing, M.; Mou, X.; Song, C.; Wang, D. Deep eutectic solvent induced ultrathin Co4N/N-doped carbon nanosheets self-supporting electrode for boosting hydrogen evolution integrated with biomass electrooxidation. Appl. Surf. Sci. 2023, 608, 155283. [Google Scholar] [CrossRef]
- Xiao, Y.; Shen, C.; Xiong, Z.; Ding, Y.; Liu, L.; Zhang, W.; Wu, Y.A. Electrocatalytic upgrading biomass approach to address oxidation of 5-(Hydroxymethyl)furfural using Mo2B2 MBene active surface. Mater. Today Phys. 2023, 35, 101122. [Google Scholar] [CrossRef]
- Chen, H.; Lv, X.; Li, S.; Hu, Y.; Li, F. Preparation and performance of phosphorus-doped NiCo LDH for oxygen evolution reaction. J. Shenyang Ligong Univ. 2023, 42, 61–67. Available online: https://kns.cnki.net/kcms2/article/abstract?v=BS8_DD2Uwa4Z0uIE8AWWa6Q4dwaaLsTyWB3VbTU5czEMU2uVPuqKV8ioBRzzgS4E4m5y5F2-oByJPYYOb7tZEjJ0g4kMh1spANU3fkmRdbzXUHi-vYAIMjCPZltkxZEs65JvyU-y4E-WoAgRU8_saK5hdMo-NTv3&uniplatform=NZKPT&language=CHS (accessed on 1 April 2023).
- Zhang, Y.; Lv, X.; Li, S.; Chen, H.; Quan, X.; Zhou, X.; Dong, Z. Preparation and properties of graphene supported NiCoMn LDH catalysts. J. Shenyang Ligong Univ. 2022, 41, 47–52. Available online: https://kns.cnki.net/kcms2/article/abstract?v=BS8_DD2Uwa4iwj6y0c2gAeym2zlqE79t4l-iwUPVKbicgefKtDwqXKJXG0JftN8yVnopc23ziBQH71c-3U0-d2d1LhG9XVUu4hlwIGiTrvlm1obiAQ5gQIgD3CjrCrr6KZEbsctE_6Hcy9L8K_qkUhtf2Xp7wair&uniplatform=NZKPT&language=CHS (accessed on 1 October 2022).
- Song, Y.; Ji, K.; Duan, H.; Shao, M. Hydrogen production coupled with water and organic oxidation based on layered double hydroxides. Exploration 2021, 1, 20210050. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-J.; Dang, L.; Xu, Z.; Yu, H.-Q.; Jin, S.; Huber, G.W. Electrochemical Oxidation of 5-Hydroxymethylfurfural with NiFe Layered Double Hydroxide (LDH) Nanosheet Catalysts. ACS Catal. 2018, 8, 5533–5541. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, Y.; Liu, B.; Chen, Z.; Xu, H.; Yan, K. Trimetallic NiCoFe-Layered Double Hydroxides Nanosheets Efficient for Oxygen Evolution and Highly Selective Oxidation of Biomass-Derived 5-Hydroxymethylfurfural. ACS Catal. 2020, 10, 5179–5189. [Google Scholar] [CrossRef]
- Qi, Y.-F.; Wang, K.-Y.; Sun, Y.; Wang, J.; Wang, C. Engineering the Electronic Structure of NiFe Layered Double Hydroxide Nanosheet Array by Implanting Cationic Vacancies for Efficient Electrochemical Conversion of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid. ACS Sustain. Chem. Eng. 2022, 10, 645–654. [Google Scholar] [CrossRef]
- Zhong, Y.; Ren, R.-Q.; Wang, J.-B.; Peng, Y.-Y.; Li, Q.; Fan, Y.-M. Grass-like NixSey nanowire arrays shelled with NiFe LDH nanosheets as a 3D hierarchical core–shell electrocatalyst for efficient upgrading of biomass-derived 5-hydroxymethylfurfural and furfural. Catal. Sci. Technol. 2022, 12, 201–211. [Google Scholar] [CrossRef]
- Dhanasmoro, L.; Li, O.L. Highly active NiFe LDH anchoring on cobalt carbonate hydroxide for efficient electrocatalytic 5-hydroxymethylfurfural oxidation towards 2,5-furandicarboxylic acid. New J. Chem. 2023, 47, 14282–14288. [Google Scholar] [CrossRef]
- Liu, B.; Zheng, Z.; Liu, Y.; Zhang, M.; Wang, Y.; Wan, Y.; Yan, K. Efficient electrooxidation of biomass-derived aldehydes over ultrathin NiV-layered double hydroxides films. J. Energy Chem. 2023, 78, 412–421. [Google Scholar] [CrossRef]
- Pang, X.; Bai, H.; Zhao, H.; Fan, W.; Shi, W. Efficient Electrocatalytic Oxidation of 5-Hydroxymethylfurfural Coupled with 4-Nitrophenol Hydrogenation in a Water System. ACS Catal. 2022, 12, 1545–1557. [Google Scholar] [CrossRef]
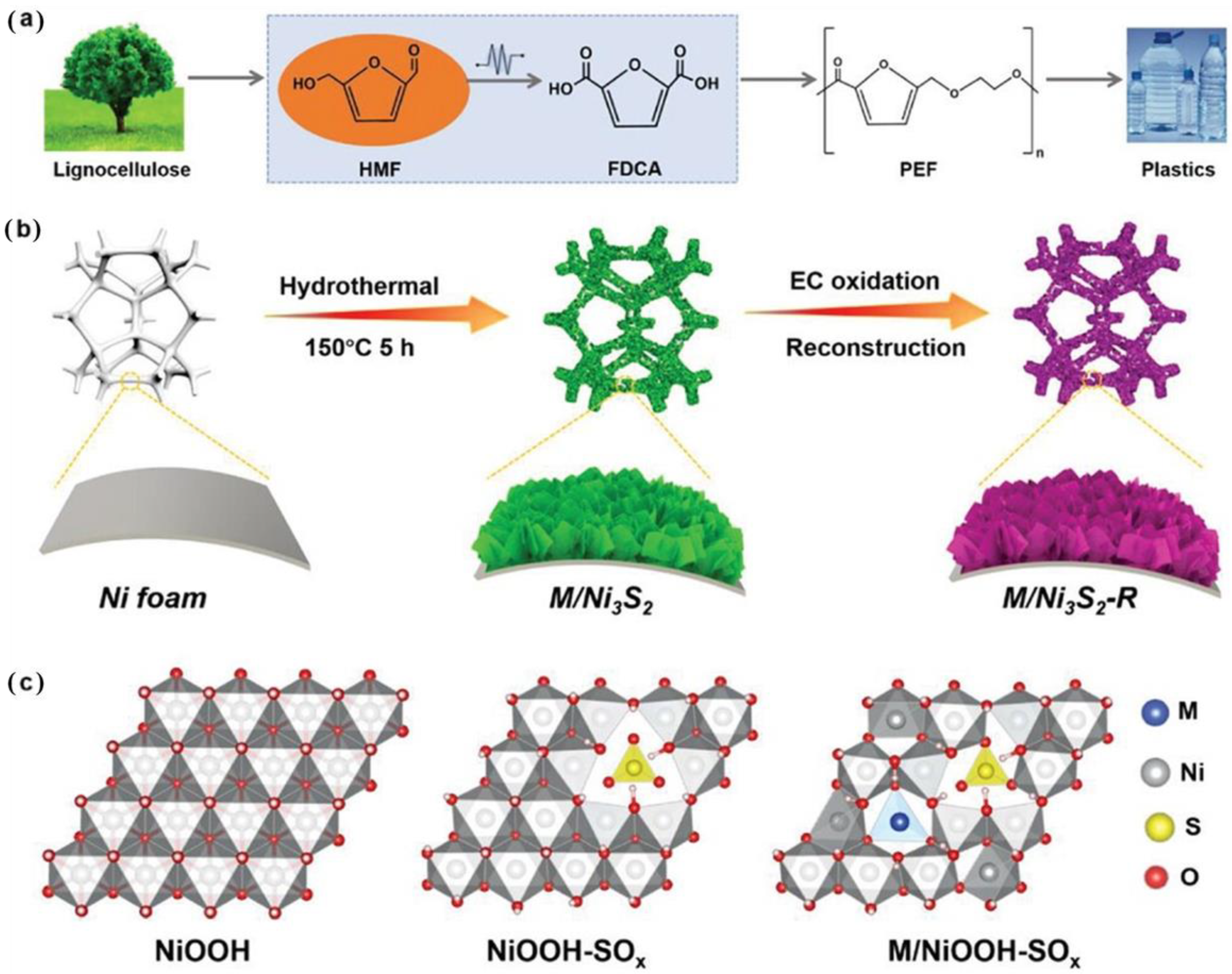
- Huang, Y.; Pang, X.; Cui, J.; Huang, Z.; Wang, G.; Zhao, H.; Bai, H.; Fan, W. Strengthening the Stability of the Reconstructed NiOOH Phase for 5-Hydroxymethylfurfural Oxidation. Inorg. Chem. 2023, 62, 6499–6509. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, B.; Yan, C.; Xue, Z.; Mu, T. The pivot to achieve high current density for biomass electrooxidation: Accelerating the reduction of Ni3+ to Ni2+. Appl. Catal. B 2023, 330, 122590. [Google Scholar] [CrossRef]
- Zhou, Z.; Chen, C.; Gao, M.; Xia, B.; Zhang, J. In situ anchoring of a Co3O4 nanowire on nickel foam: An outstanding bifunctional catalyst for energy-saving simultaneous reactions. Green Chem. 2019, 21, 6699–6706. [Google Scholar] [CrossRef]
- Wang, C.; Bongard, H.-J.; Weidenthaler, C.; Wu, Y.; Schüth, F. Design and Application of a High-Surface-Area Mesoporous δ-MnO2 Electrocatalyst for Biomass Oxidative Valorization. Chem. Mater. 2022, 34, 3123–3132. [Google Scholar] [CrossRef]
- Zhu, B.; Qin, Y.; Du, J.; Zhang, F.; Lei, X. Ammonia Etching to Generate Oxygen Vacancies on CuMn2O4 for Highly Efficient Electrocatalytic Oxidation of 5-Hydroxymethylfurfural. ACS Sustain. Chem. Eng. 2021, 9, 11790–11797. [Google Scholar] [CrossRef]
- Lu, Y.; Dong, C.-L.; Huang, Y.-C.; Zou, Y.; Liu, Z.; Liu, Y.; Li, Y.; He, N.; Shi, J.; Wang, S. Identifying the Geometric Site Dependence of Spinel Oxides for the Electrooxidation of 5-Hydroxymethylfurfural. Angew. Chem. Int. Ed. 2020, 59, 19215–19221. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Liu, W.; Zhang, S.; Liu, Q.; Luo, J.; Liu, X. A dual-functional Bi-doped Co3O4 nanosheet array towards high efficiency 5-hydroxymethylfurfural oxidation and hydrogen production. Chem. Commun. 2023, 59, 442–445. [Google Scholar] [CrossRef] [PubMed]
- Zhong, R.; Wang, Q.; Du, L.; Pu, Y.; Ye, S.; Gu, M.; Conrad Zhang, Z.; Huang, L. Ultrathin polycrystalline Co3O4 nanosheets with enriched oxygen vacancies for efficient electrochemical oxygen evolution and 5-hydroxymethylfurfural oxidation. Appl. Surf. Sci. 2022, 584, 152553. [Google Scholar] [CrossRef]
- Sun, M.; Wang, Y.; Sun, C.; Qi, Y.; Cheng, J.; Song, Y.; Zhang, L. Nitrogen-doped Co3O4 nanowires enable high-efficiency electrochemical oxidation of 5-hydroxymethylfurfural. Chin. Chem. Lett. 2022, 33, 385–389. [Google Scholar] [CrossRef]
- Zhong, R.; Wu, P.; Wang, Q.; Zhang, X.; Du, L.; Liu, Y.; Yang, H.; Gu, M.; Zhang, Z.C.; Huang, L.; et al. Room-temperature fabrication of defective CoOxHy nanosheets with abundant oxygen vacancies and high porosity as efficient 5-hydroxymethylfurfural oxidation electrocatalysts. Green Chem. 2023, 25, 4674–4684. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, Z.; Wang, M. Preparation of NiO-N/C composites for electrochemical oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid. Biomass Convers. Biorefin. 2022, 13, 17247–17254. [Google Scholar] [CrossRef]
- Guo, Z.-Y.; Li, C.-X.; Gao, M.; Han, X.; Zhang, Y.-J.; Zhang, W.-J.; Li, W.-W. Mn-O Covalency Governs the Intrinsic Activity of Co-Mn Spinel Oxides for Boosted Peroxymonosulfate Activation. Angew. Chem. Int. Ed. 2021, 60, 274–280. [Google Scholar] [CrossRef]
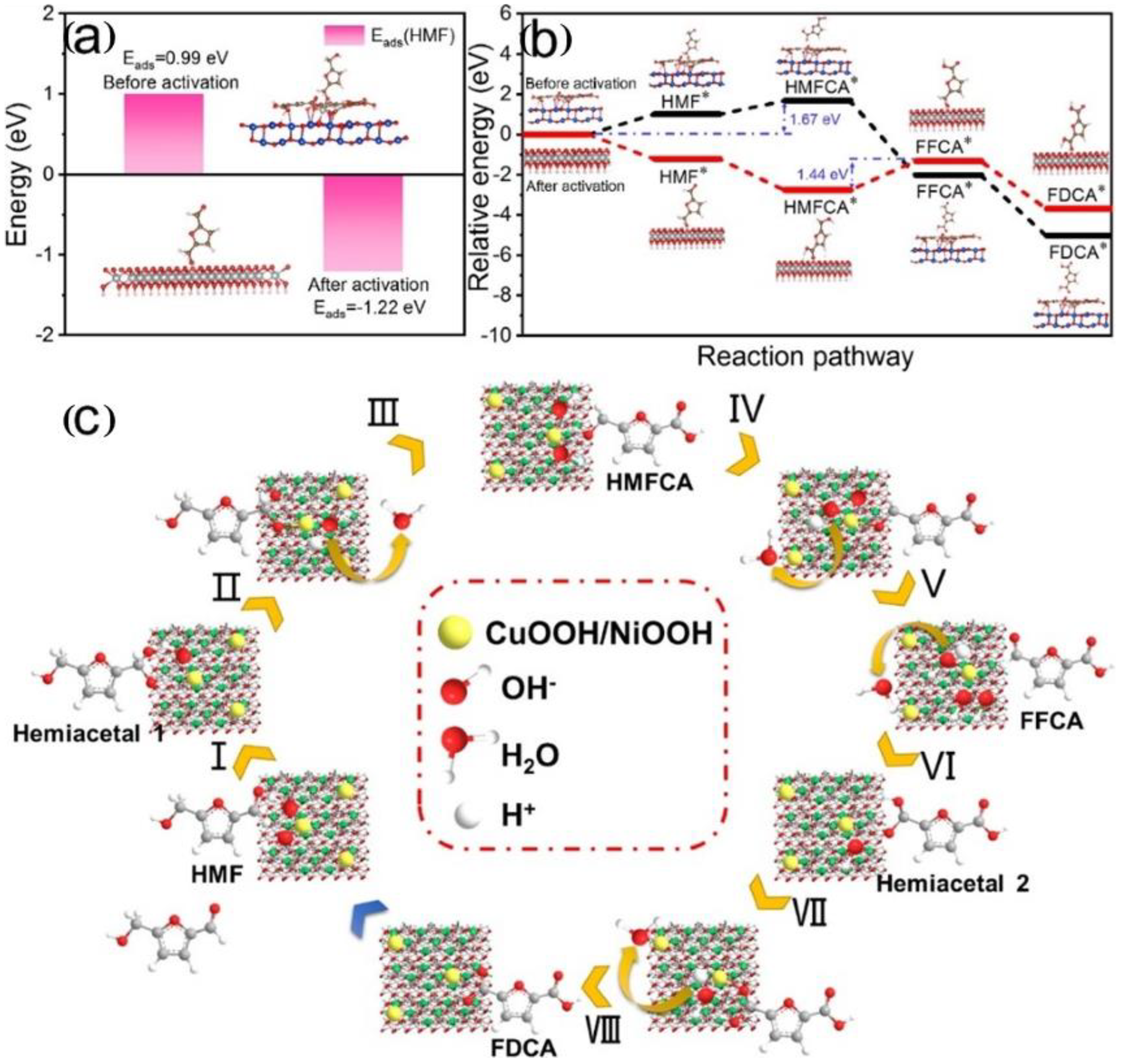
- Yang, G.; Jiao, Y.; Yan, H.; Xie, Y.; Wu, A.; Dong, X.; Guo, D.; Tian, C.; Fu, H. Interfacial Engineering of MoO2-FeP Heterojunction for Highly Efficient Hydrogen Evolution Coupled with Biomass Electrooxidation. Adv. Mater. 2020, 32, 2000455. [Google Scholar] [CrossRef]
- Lu, Y.; Dong, C.-L.; Huang, Y.-C.; Zou, Y.; Liu, Y.; Li, Y.; Zhang, N.; Chen, W.; Zhou, L.; Lin, H.; et al. Hierarchically nanostructured NiO-Co3O4 with rich interface defects for the electro-oxidation of 5-hydroxymethylfurfural. Sci. China Chem. 2020, 63, 980–986. [Google Scholar] [CrossRef]
- Gao, L.; Liu, Z.; Ma, J.; Zhong, L.; Song, Z.; Xu, J.; Gan, S.; Han, D.; Niu, L. NiSe@NiOx core-shell nanowires as a non-precious electrocatalyst for upgrading 5-hydroxymethylfurfural into 2,5-furandicarboxylic acid. Appl. Catal. B 2020, 261, 118235. [Google Scholar] [CrossRef]
- Liang, S.; Pan, L.; Thomas, T.; Zhu, B.; Chen, C.; Zhang, J.; Shen, H.; Liu, J.; Yang, M. Ni3N-V2O3 enables highly efficient 5-(Hydroxymethyl) furfural oxidation enabling membrane free hydrogen production. Chem. Eng. J. 2021, 415, 128864. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, Y.; Tao, S. CoP-CoOOH heterojunction with modulating interfacial electronic structure: A robust biomass-upgrading electrocatalyst. Appl. Catal. B 2022, 315, 121588. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Y.; Zhou, M.; Li, Y. Boosting the electro-oxidation of 5-hydroxymethyl-furfural on a Co–CoSx heterojunction by intensified spin polarization. Chem. Sci. 2022, 13, 4647–4653. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Chen, J.; Li, Y. CoP nanorods anchored on Ni2P-NiCoP nanosheets with abundant heterogeneous interfaces boosting the electrocatalytic oxidation of 5-hydroxymethyl-furfural. Catal. Sci. Technol. 2022, 12, 4288–4297. [Google Scholar] [CrossRef]
- Luo, R.; Li, Y.; Xing, L.; Wang, N.; Zhong, R.; Qian, Z.; Du, C.; Yin, G.; Wang, Y.; Du, L. A dynamic Ni(OH)2-NiOOH/NiFeP heterojunction enabling high-performance E-upgrading of hydroxymethylfurfural. Appl. Catal. B 2022, 311, 121357. [Google Scholar] [CrossRef]
- Zeng, J.; Chen, W.; Zhang, G.; Yu, L.; Zhong, L.; Liu, Y.; Zhao, S.; Qiu, Y. Heterostructured Ni3N–NiMoN Nanowires as Bifunctional Electrocatalysts for Hydrogen Evolution and 5-Hydroxymethylfurfural Oxidation. ACS Appl. Nano Mater. 2022, 5, 7321–7330. [Google Scholar] [CrossRef]
- Xu, D.; Yang, Y.; Zhang, B.; Yang, Z.; Liu, S.; Mu, T. Deep Eutectic Solvent-Induced In Situ Etching and Phosphorization to Form Nickel Phosphides for Electrooxidation of 5-Hydroxymethylfurfural. ChemSusChem 2022, 15, e202200822. [Google Scholar] [CrossRef] [PubMed]
- Xing, M.; Zhang, D.; Liu, D.; Song, C.; Wang, D. Surface engineering of carbon-coated cobalt-doped nickel phosphides bifunctional electrocatalyst for boosting 5-hydroxymethylfurfural oxidation coupled with hydrogen evolution. J. Colloid Interface Sci. 2023, 629, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Hai, G.; Zhou, P.; Liu, Z.; Zhang, Y.; Peng, B.; Xia, W.; Huang, X.; Wang, G. Electrochemical Oxidation of 5-Hydroxymethylfurfural on CeO2-Modified Co3O4 with Regulated Intermediate Adsorption and Promoted Charge Transfer. Adv. Funct. Mater. 2023, 33, 2213170. [Google Scholar] [CrossRef]
- Zhou, P.; Hai, G.; Zhao, G.; Li, R.; Huang, X.; Lu, Y.; Wang, G. CeO2 as an “electron pump” to boost the performance of Co4N in electrocatalytic hydrogen evolution, oxygen evolution and biomass oxidation valorization. Appl. Catal. B 2023, 325, 122364. [Google Scholar] [CrossRef]
- Bi, J.; Xu, H.; Wang, W.; Sang, T.; Jiang, A.; Hao, J.; Li, Z. Cu2P7-CoP Heterostructure Nanosheets Enable High-Performance of 5-Hydroxymethylfurfural Electrooxidation. Chemistry 2023, 29, e202300973. [Google Scholar] [CrossRef]
- Zhang, B.; Fu, H.; Mu, T. Hierarchical NiSx/Ni2P nanotube arrays with abundant interfaces for efficient electrocatalytic oxidation of 5-hydroxymethylfurfural. Green Chem. 2022, 24, 877–884. [Google Scholar] [CrossRef]
- Luo, R.; Li, Y.; Wang, N.; Zhong, R.; Xing, L.; Zhu, L.; Wang, Y.; Du, L.; Ye, S. A Bimetallic Phosphide@Hydroxide Interface for High-Performance 5-Hydroxymethylfurfural Electro-Valorization. J. Phys. Chem. C 2023, 127, 4967–4974. [Google Scholar] [CrossRef]
- Li, Y.; Fu, Y.; Cao, Y.; Lei, F.; Zhao, J.; Wang, R.; Qiu, S.; Zhang, Z. Construction of hierarchical NiCu-based bimetallic electrocatalysts for promoting the electrooxidation of biomass derivatives. Sustain. Energy Fuels 2023, 7, 4505–4513. [Google Scholar] [CrossRef]
- Sun, M.; Yang, J.; Huang, J.; Wang, Y.; Liu, X.; Qi, Y.; Zhang, L. Interfacial Engineering of Ni/Ni0.2Mo0.8N Heterostructured Nanorods Realizes Efficient 5-Hydroxymethylfurfural Electrooxidation and Hydrogen Evolution. Langmuir 2023, 39, 3762–3769. [Google Scholar] [CrossRef]
- Jia, W.; Liu, B.; Gong, R.; Bian, X.; Du, S.; Ma, S.; Song, Z.; Ren, Z.; Chen, Z. Electronic Modulation Induced by Ni-VN Heterojunction Reinforces Electrolytic Hydrogen Evolution Coupled with Biomass Upgrade. Small 2023, 19, 2302025. [Google Scholar] [CrossRef]
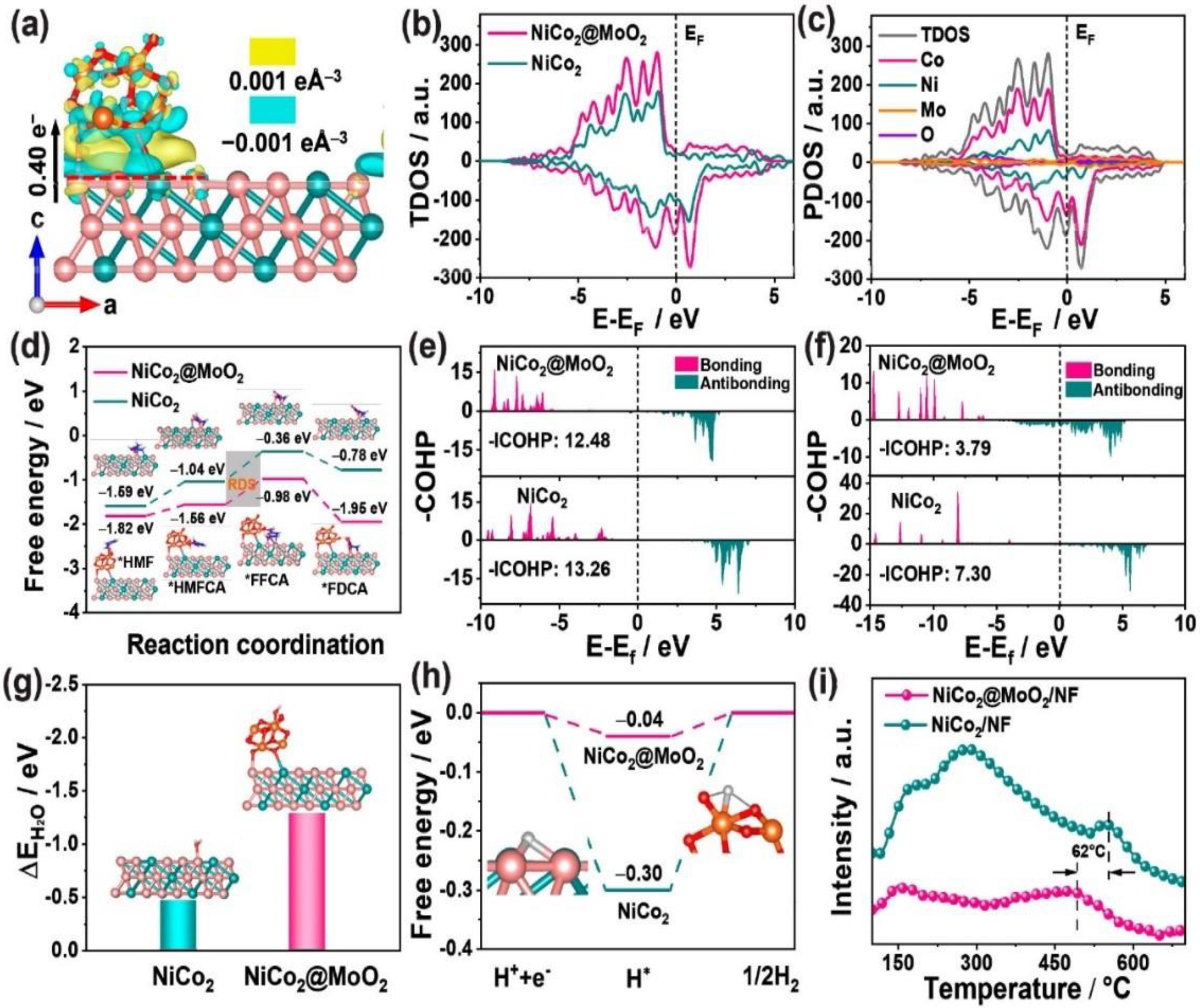
- Wu, J.; Chen, J.; Yu, T.; Zhai, Z.; Zhu, Y.; Wu, X.; Yin, S. Boosting Electrochemical Kinetics of NiCo2 via MoO2 Modification for Biomass Upgrading Assisted Hydrogen Evolution. ACS Catal. 2023, 13, 13257–13266. [Google Scholar] [CrossRef]
- Woo, J.; Moon, B.C.; Lee, U.; Oh, H.-S.; Chae, K.H.; Jun, Y.; Min, B.K.; Lee, D.K. Collaborative Electrochemical Oxidation of the Alcohol and Aldehyde Groups of 5-Hydroxymethylfurfural by NiOOH and Cu(OH)2 for Superior 2,5-Furandicarboxylic Acid Production. ACS Catal. 2022, 12, 4078–4091. [Google Scholar] [CrossRef]
- Xue, Y.; Zhao, G.; Yang, R.; Chu, F.; Chen, J.; Wang, L.; Huang, X. 2D metal–organic framework-based materials for electrocatalytic, photocatalytic and thermocatalytic applications. Nanoscale 2021, 13, 3911–3936. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Lv, J.; Huang, X.; Lu, Y.; Wang, G. Strategies for enhancing the catalytic activity and electronic conductivity of MOFs-based electrocatalysts. Coord. Chem. Rev. 2023, 478, 214969. [Google Scholar] [CrossRef]
- Phan, D.-P.; Tran, M.H.; Lee, E.Y. Metal-organic framework-based materials as heterogeneous catalysts for biomass upgrading into renewable plastic precursors. Mater. Today Chem. 2023, 33, 101691. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Y.; Liu, K.; Delbari, S.A.; Kim, A.; Sabahi Namini, A.; Le, Q.V.; Shokouhimehr, M.; Xia, C.; Jang, H.W.; et al. Metal-organic framework-based nanostructured catalysts: Applications in biomass conversion. Fuel 2023, 340, 127482. [Google Scholar] [CrossRef]
- Cai, M.; Zhang, Y.; Zhao, Y.; Liu, Q.; Li, Y.; Li, G. Two-dimensional metal–organic framework nanosheets for highly efficient electrocatalytic biomass 5-(hydroxymethyl)furfural (HMF) valorization. J. Mater. Chem. A 2020, 8, 20386–20392. [Google Scholar] [CrossRef]
- Pila, T.; Nueangnoraj, K.; Ketrat, S.; Somjit, V.; Kongpatpanich, K. Electrochemical Production of 2,5-Furandicarboxylic from 5-Hydroxymethylfurfural Using Ultrathin Co(OH)2 on ZIF-67. ACS Appl. Energy Mater. 2021, 4, 12909–12916. [Google Scholar] [CrossRef]
- Bai, X.-J.; He, W.-X.; Lu, X.-Y.; Fu, Y.; Qi, W. Electrochemical oxidation of 5-hydroxymethylfurfural on ternary metal–organic framework nanoarrays: Enhancement from electronic structure modulation. J. Mater. Chem. A 2021, 9, 14270–14275. [Google Scholar] [CrossRef]
- Sekar, P.; Vasanthakumar, P.; Shanmugam, R.; Senthil Kumar, S.; Agnoli, S.; Deepak, R.J.; Murugan, K.; Bhuvanesh, N.; Karvembu, R. Green synthesis of a redox-active riboflavin-integrated Ni-MOF and its versatile electrocatalytic applications towards oxygen evolution and reduction, and HMF oxidation reactions. Green Chem. 2022, 24, 9233–9244. [Google Scholar] [CrossRef]
- Zhang, Y.; Kornienko, N. Conductive Metal-Organic Frameworks Bearing M−O4 Active Sites as Highly Active Biomass Valorization Electrocatalysts. ChemSusChem 2022, 15, e202101587. [Google Scholar] [CrossRef]
- Pang, X.; Zhao, H.; Huang, Y.; Luo, B.; Bai, H.; Fan, W. Electrochemically induced NiOOH/Ag+ active species for efficient oxidation of 5-hydroxymethylfurfural. Appl. Surf. Sci. 2023, 608, 155152. [Google Scholar] [CrossRef]
- Pang, X.; Bai, H.; Huang, Y.; Zhao, H.; Zheng, G.; Fan, W. Mechanistic insights for dual-species evolution toward 5-hydroxymethylfurfural oxidation. J. Catal. 2023, 417, 22–34. [Google Scholar] [CrossRef]
- Feng, Y.; Yang, K.; Smith, R.L.; Qi, X. Metal sulfide enhanced metal–organic framework nanoarrays for electrocatalytic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid. J. Mater. Chem. A 2023, 11, 6375–6383. [Google Scholar] [CrossRef]
- Cai, M.; Ding, S.; Gibbons, B.; Yang, X.; Kessinger, M.C.; Morris, A.J. Nickel(ii)-modified covalent-organic framework film for electrocatalytic oxidation of 5-hydroxymethylfurfural (HMF). Chem. Commun. 2020, 56, 14361–14364. [Google Scholar] [CrossRef]
- Qin, Q.; Heil, T.; Schmidt, J.; Schmallegger, M.; Gescheidt, G.; Antonietti, M.; Oschatz, M. Electrochemical Fixation of Nitrogen and Its Coupling with Biomass Valorization with a Strongly Adsorbing and Defect Optimized Boron–Carbon–Nitrogen Catalyst. ACS Appl. Energy Mater. 2019, 2, 8359–8365. [Google Scholar] [CrossRef]
- Ma, T.; Kapustin, E.A.; Yin, S.X.; Liang, L.; Zhou, Z.; Niu, J.; Li, L.-H.; Wang, Y.; Su, J.; Li, J.; et al. Single-crystal X-ray diffraction structures of covalent organic frameworks. Science 2018, 361, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Shao, L.; Wu, Z.; Zhan, P.; Zhang, L. Design and Synthesis of Porous Organic Polymers: Promising Catalysts for Lignocellulose Conversion to 5-Hydroxymethylfurfural and Derivates. Polymers 2023, 15, 2630. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Z.; Wang, L.; Li, G.; Song, T. Recent Advances in Electrocatalytic Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid by Heterogeneous Catalysts. Catalysts 2024, 14, 157. https://doi.org/10.3390/catal14020157
Ma Z, Wang L, Li G, Song T. Recent Advances in Electrocatalytic Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid by Heterogeneous Catalysts. Catalysts. 2024; 14(2):157. https://doi.org/10.3390/catal14020157
Chicago/Turabian StyleMa, Zhiming, Lei Wang, Guangyu Li, and Tao Song. 2024. "Recent Advances in Electrocatalytic Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid by Heterogeneous Catalysts" Catalysts 14, no. 2: 157. https://doi.org/10.3390/catal14020157
APA StyleMa, Z., Wang, L., Li, G., & Song, T. (2024). Recent Advances in Electrocatalytic Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid by Heterogeneous Catalysts. Catalysts, 14(2), 157. https://doi.org/10.3390/catal14020157