Photocatalytic Performance of Sn–Doped TiO2 Nanopowders for Photocatalytic Degradation of Methyl Orange Dye
Abstract
1. Introduction
2. Results and Discussion
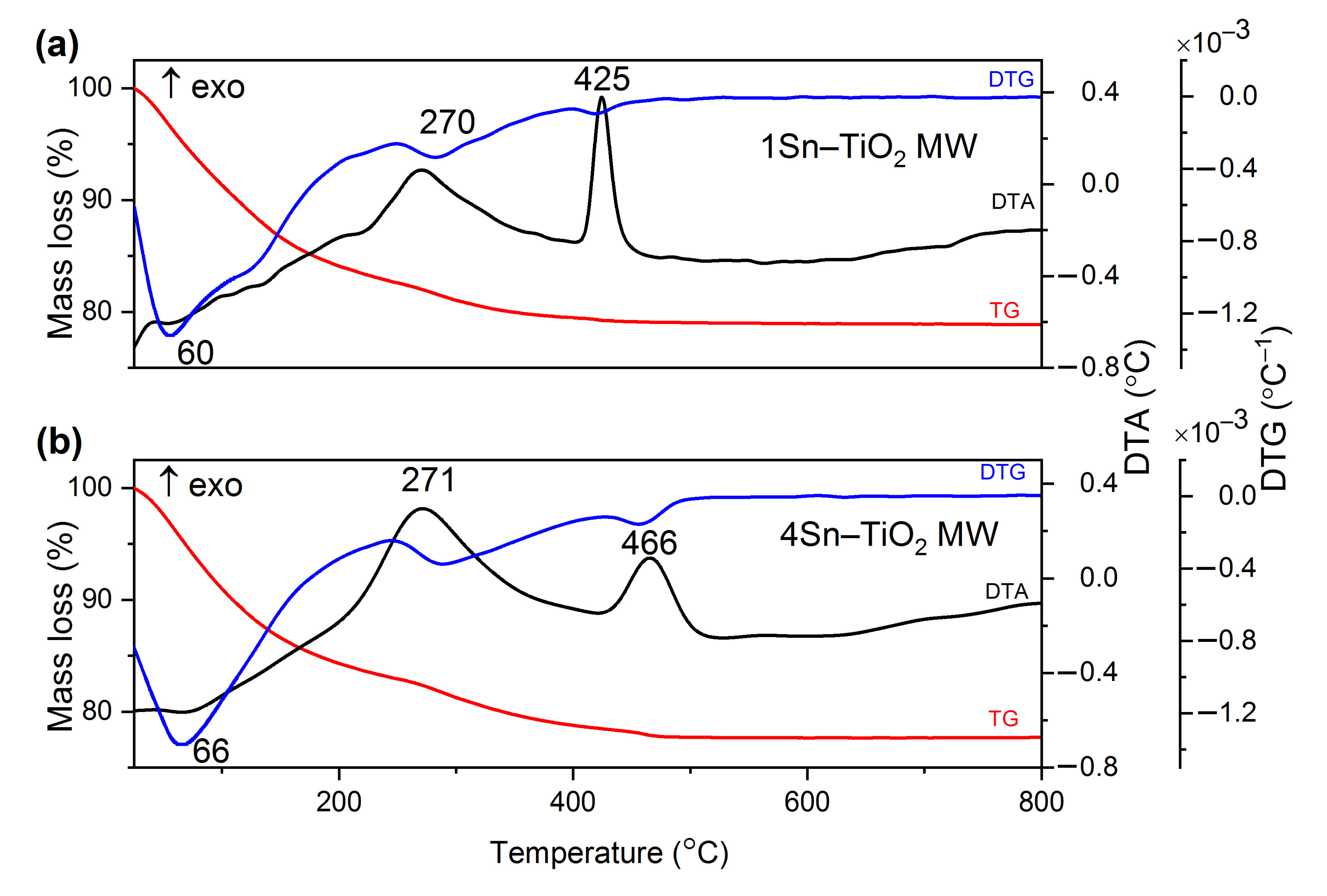
2.1. Thermal Treatment
2.2. X-ray Diffraction
2.3. X-ray Fluorescence (XRF)
2.4. SEM Results
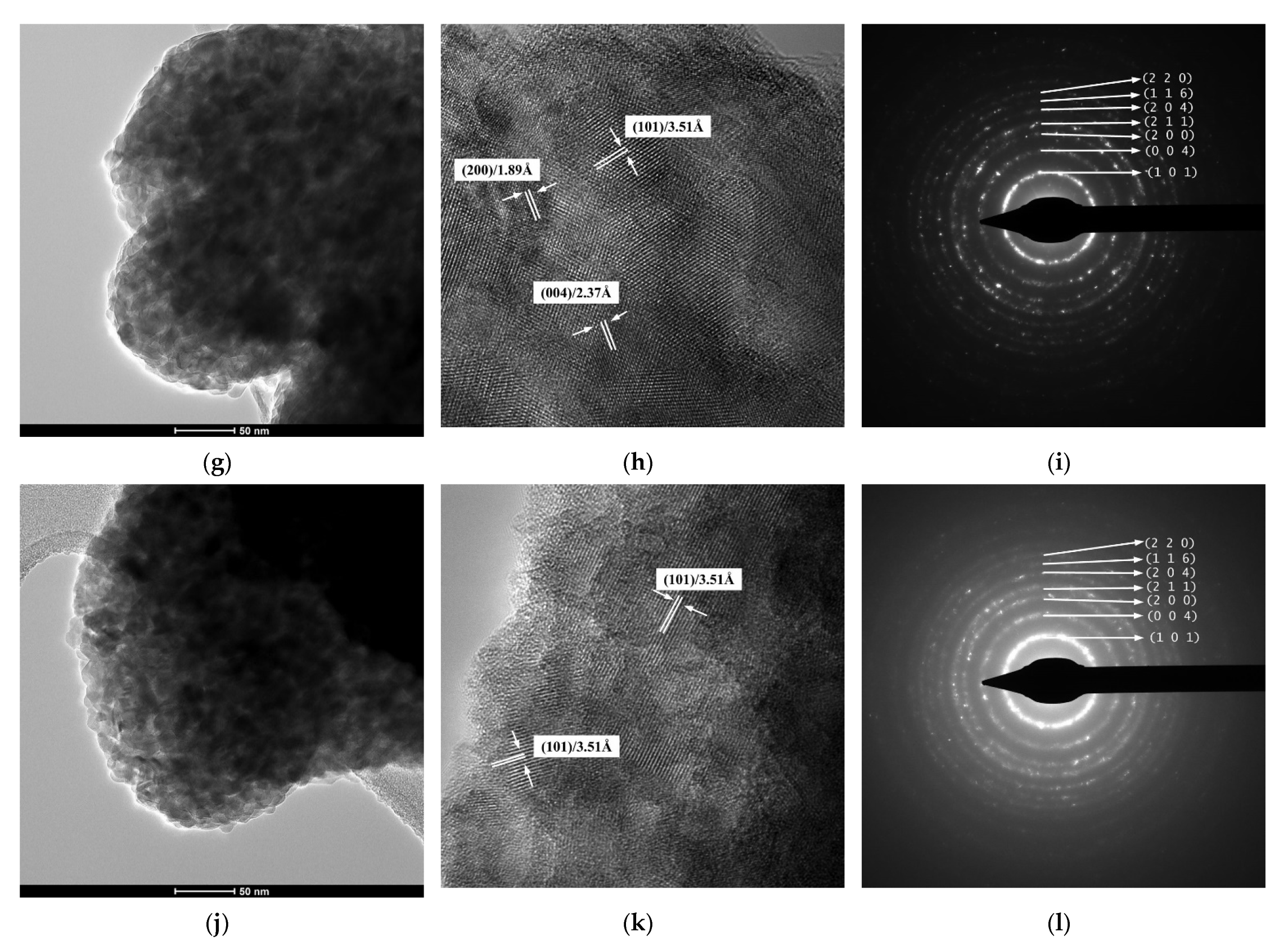
2.5. TEM/HRTEM/SAED Investigations
2.6. STEM/EDX Investigations
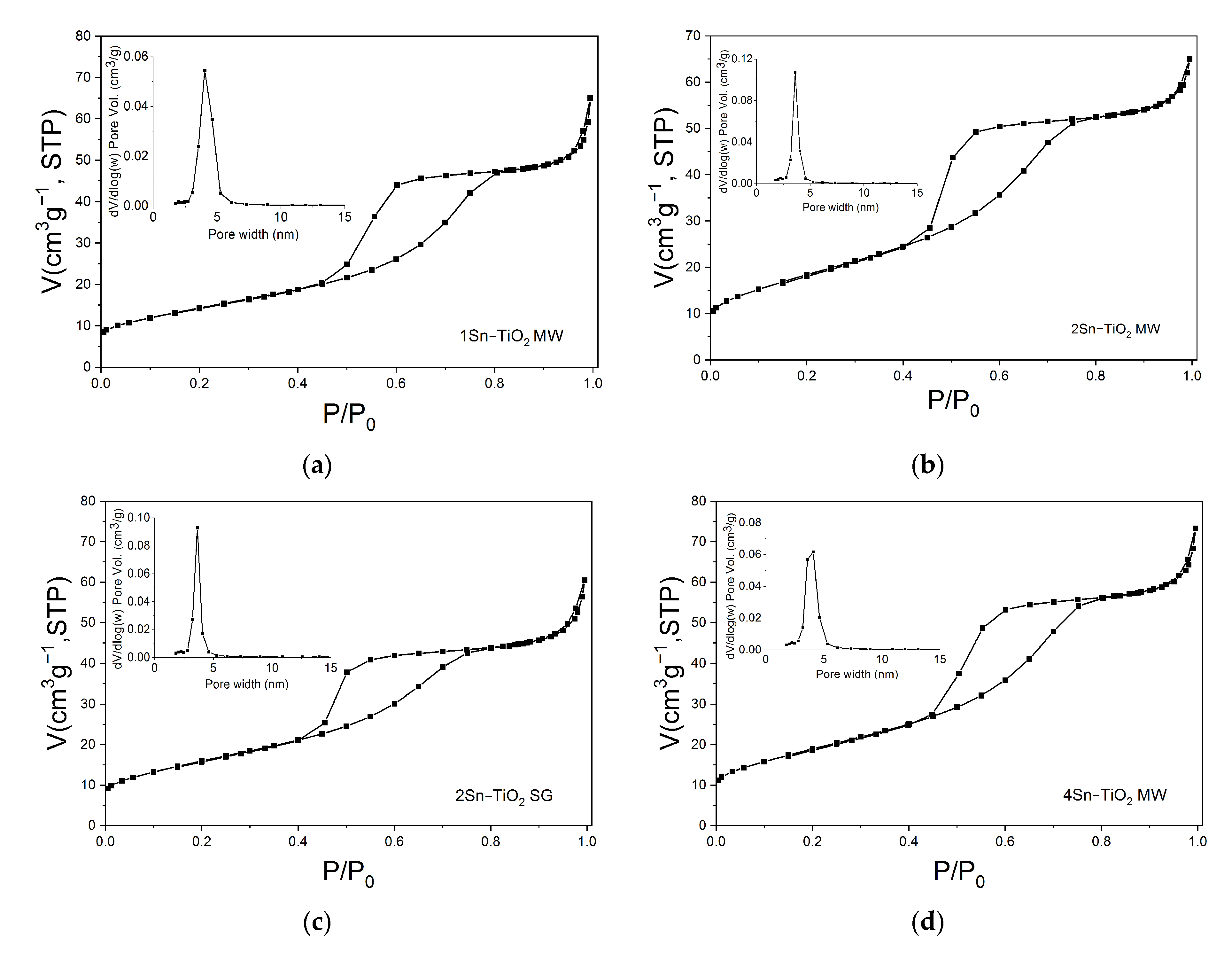
2.7. Textural Characterization
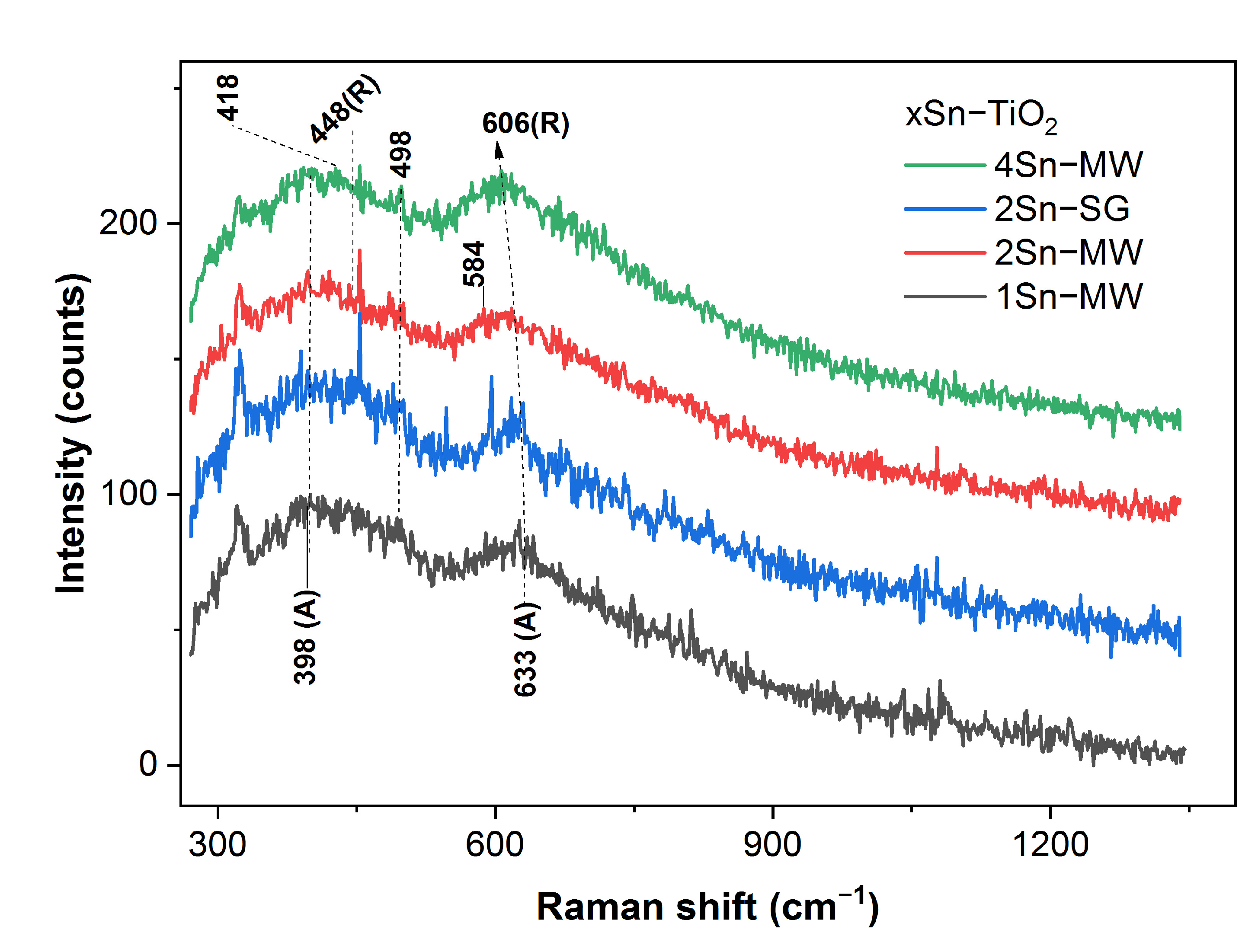
2.8. Raman Spectroscopy Analysis
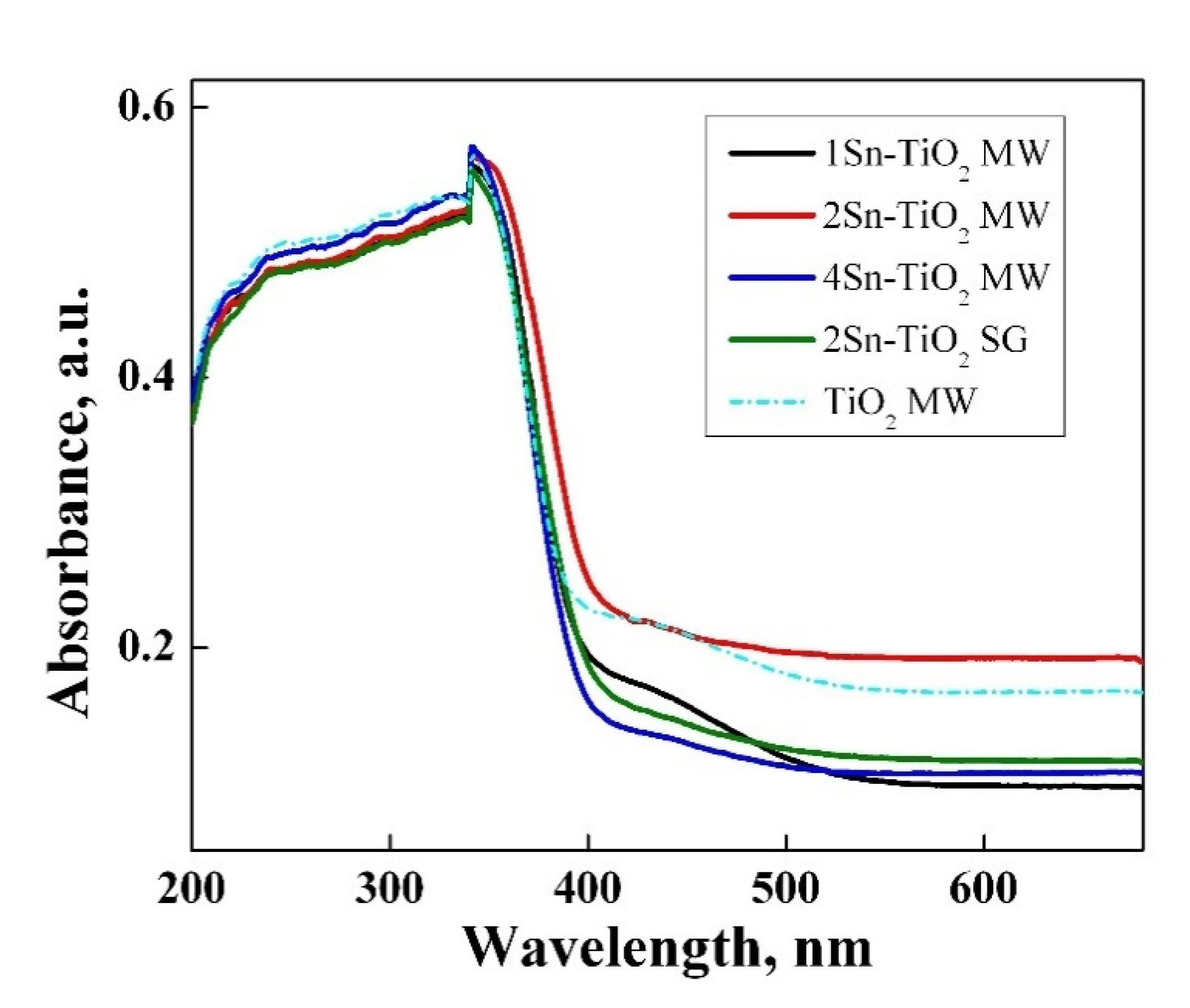
2.9. UV–Vis Spectroscopy Analysis
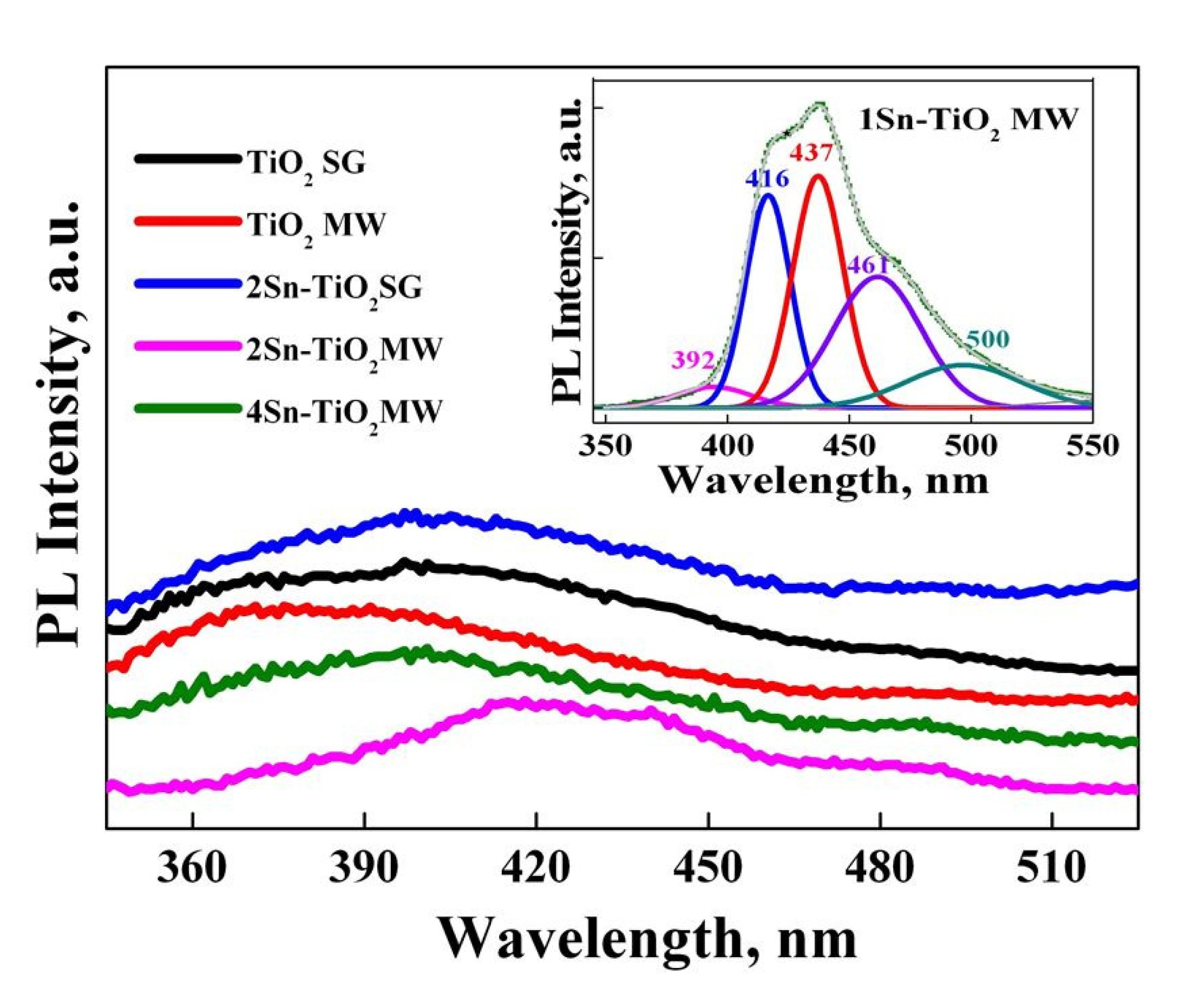
2.10. Photoluminescence (PL) Analysis
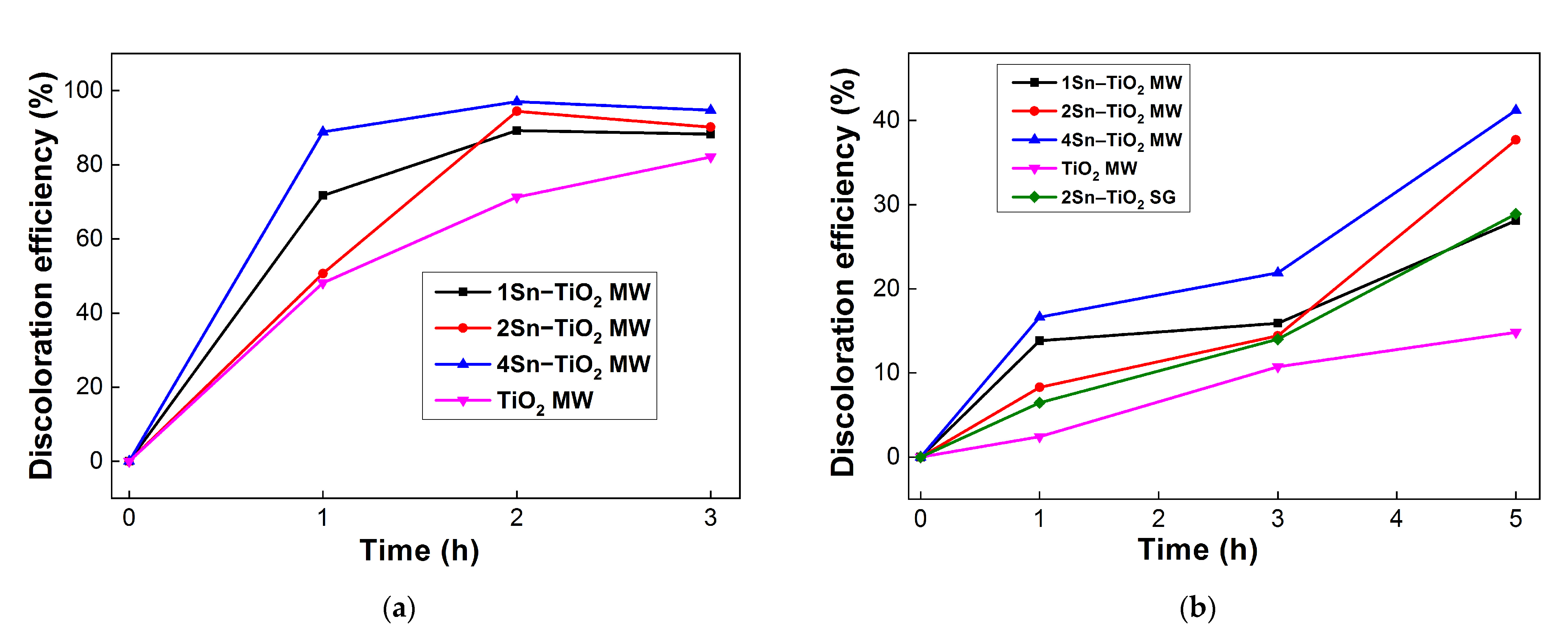
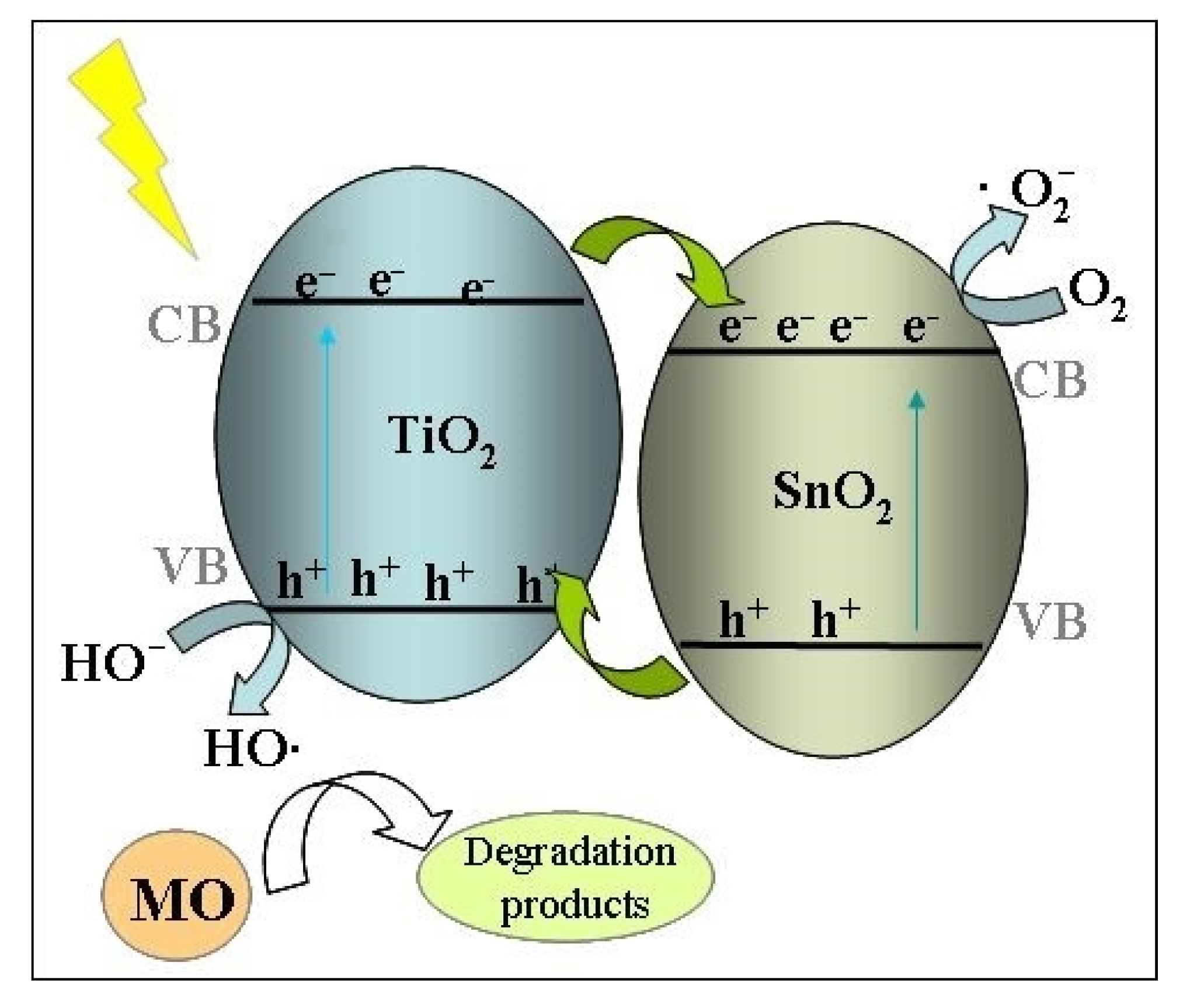
2.11. Photocatalytic Activity
3. Materials and Methods
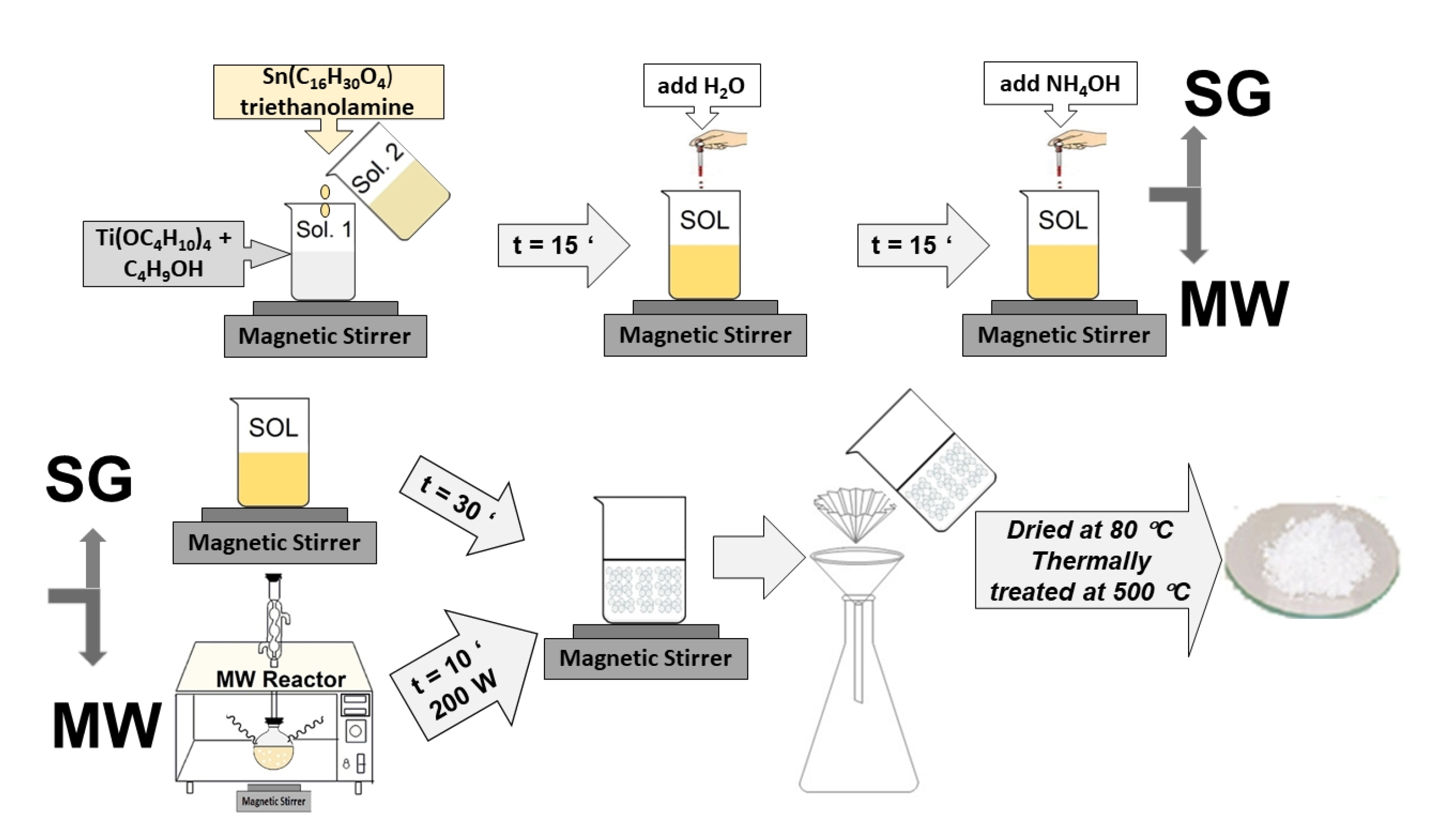
3.1. Gels and Powders Preparation
3.2. Methods of Gels and Powders Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raliya, R.; Avery, C.; Chakrabarti, S.; Biswas, P. Photocatalytic Degradation of Methyl Orange Dye by Pristine Titanium Dioxide, Zinc Oxide, and Graphene Oxide Nanostructures and Their Composites under Visible Light Irradiation. Appl. Nanosci. 2017, 7, 253–259. [Google Scholar] [CrossRef]
- Gürses, A.; Güneş, K.; Şahin, E. Chapter 5—Removal of Dyes and Pigments from Industrial Effluents. In Green Chemistry and Water Remediation: Research and Applications; Sharma, S.K., Ed.; Advances in Green and Sustainable Chemistry; Elsevier: Amsterdam, The Netherlands, 2021; pp. 135–187. ISBN 978-0-12-817742-6. [Google Scholar]
- Wang, J.; Wang, Z.; Zhao, D.; Liang, Y.; Wang, H.; Wang, N.; Jiang, W.; Liu, S.; Liu, C.; Ding, W.; et al. Preparation, Structural and Photocatalytic Activity of Sn/Fe Co-Doped TiO2 Nanoparticles by Sol-Gel Method. Ceram. Int. 2022, 48, 8297–8305. [Google Scholar] [CrossRef]
- Gaur, N.; Dutta, D.; Singh, A.; Dubey, R.; Kamboj, D.V. Recent Advances in the Elimination of Persistent Organic Pollutants by Photocatalysis. Front. Environ. Sci. 2022, 10, 2076. [Google Scholar] [CrossRef]
- Anwer, H.; Mahmood, A.; Lee, J.; Kim, K.-H.; Park, J.-W.; Yip, A.C.K. Photocatalysts for Degradation of Dyes in Industrial Effluents: Opportunities and Challenges. Nano Res. 2019, 12, 955–972. [Google Scholar] [CrossRef]
- Thamaraiselvi, K.; Sivakumar, T. Photocatalytic Reduction of Carbon Dioxide by Using Bare and Copper Oxide Impregnated Nano Titania Catalysts. J. Nanosci. Nanotechnol. 2017, 17, 313–322. [Google Scholar] [CrossRef]
- Stride, J.A.; Tuong, N.T. Controlled Synthesis of Titanium Dioxide Nanostructures. Solid State Phenom. 2010, 162, 261–294. [Google Scholar] [CrossRef]
- Prakash, J.; Sun, S.; Swart, H.C.; Gupta, R.K. Noble Metals-TiO2 Nanocomposites: From Fundamental Mechanisms to Photocatalysis, Surface Enhanced Raman Scattering and Antibacterial Applications. Appl. Mater. Today 2018, 11, 82–135. [Google Scholar] [CrossRef]
- Muniandy, S.S.; Mohd Kaus, N.H.; Jiang, Z.-T.; Altarawneh, M.; Lee, H.L. Green Synthesis of Mesoporous Anatase TiO2 Nanoparticles and Their Photocatalytic Activities. RSC Adv. 2017, 7, 48083–48094. [Google Scholar] [CrossRef]
- Mathew, S.; Ganguly, P.; Kumaravel, V.; Harrison, J.; Hinder, S.J.; Bartlett, J.; Pillai, S.C. Effect of Chalcogens (S, Se, and Te) on the Anatase Phase Stability and Photocatalytic Antimicrobial Activity of TiO2. Mater. Today Proc. 2020, 33, 2458–2464. [Google Scholar] [CrossRef]
- Kanan, S.; Moyet, M.A.; Arthur, R.B.; Patterson, H.H. Recent Advances on TiO2-Based Photocatalysts toward the Degradation of Pesticides and Major Organic Pollutants from Water Bodies. Catal. Rev. 2020, 62, 1–65. [Google Scholar] [CrossRef]
- Chen, X.; Mao, S.S. Titanium Dioxide Nanomaterials: Synthesis, Properties, Modifications, and Applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef] [PubMed]
- Štengl, V.; Grygar, T.M.; Henych, J.; Kormunda, M. Hydrogen Peroxide Route to Sn-Doped Titania Photocatalysts. Chem. Cent. J. 2012, 6, 113. [Google Scholar] [CrossRef]
- Bjelajac, A.; Petrović, R.; Vujancevic, J.; Veltruska, K.; Matolin, V.; Siketic, Z.; Provatas, G.; Jaksic, M.; Stan, G.E.; Socol, G.; et al. Sn-Doped TiO2 Nanotubular Thin Film for Photocatalytic Degradation of Methyl Orange Dye. J. Phys. Chem. Solids 2020, 147, 109609. [Google Scholar] [CrossRef]
- Wang, J.; Cai, Q.; Li, H.; Cui, Y.; Wang, H. A Review on Nanotube Film Photocatalysts Prepared by Liquid-Phase Deposition. Int. J. Photoenergy 2012, 2012, 702940. [Google Scholar] [CrossRef]
- Buchalska, M.; Kobielusz, M.; Matuszek, A.; Pacia, M.; Wojtyła, S.; Macyk, W. On Oxygen Activation at Rutile- and Anatase-TiO2. ACS Catal. 2015, 5, 7424–7431. [Google Scholar] [CrossRef]
- Athar, T. Chapter 14—Metal Oxide Nanopowder. In Emerging Nanotechnologies for Manufacturing, 2nd ed.; Ahmed, W., Jackson, M.J., Eds.; Micro and Nano Technologies; William Andrew Publishing: Boston, MA, USA, 2015; pp. 343–401. ISBN 978-0-323-28990-0. [Google Scholar]
- Athar, T. Chapter 17—Smart Precursors for Smart Nanoparticles. In Emerging Nanotechnologies for Manufacturing, 2nd ed.; Ahmed, W., Jackson, M.J., Eds.; Micro and Nano Technologies; William Andrew Publishing: Boston, MA, USA, 2015; pp. 444–538. ISBN 978-0-323-28990-0. [Google Scholar]
- Pandele-Cusu, J.; Petrescu, S.; Preda, S.; Petcu, G.; Ciobanu, M.; Predoana, L. Comparative Study of the TiO2 Nanopowders Prepared from Different Precursors and Chemical Methods for Heterogeneous Photocatalysis Application. J. Therm. Anal. Calorim. 2022, 147, 13111–13124. [Google Scholar] [CrossRef]
- Kumar, S.G.; Rao, K.S.R.K. Polymorphic Phase Transition among the Titania Crystal Structures Using a Solution-Based Approach: From Precursor Chemistry to Nucleation Process. Nanoscale 2014, 6, 11574–11632. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.-W.; Cao, Y.; Li, H.; Webb, C.; Pan, W.-P. Synthesis of TiO2 Based on Hydrothermal Methods Using Elevated Pressures and Microwave Conditions. J. Therm. Anal. Calorim. 2014, 116, 1241–1248. [Google Scholar] [CrossRef]
- Bregadiolli, B.A.; Fernandes, S.L.; de Graeff, C.F.O. Easy and Fast Preparation of TiO2—Based Nanostructures Using Microwave Assisted Hydrothermal Synthesis. Mater. Res. 2017, 20, 912–919. [Google Scholar] [CrossRef]
- Mehraz, S.; Kongsong, P.; Taleb, A.; Dokhane, N.; Sikong, L. Large Scale and Facile Synthesis of Sn Doped TiO2 Aggregates Using Hydrothermal Synthesis. Sol. Energy Mater. Sol. Cells 2019, 189, 254–262. [Google Scholar] [CrossRef]
- Vinogradov, A.V.; Vinogradov, V.V. Low-Temperature Sol–Gel Synthesis of Crystalline Materials. RSC Adv. 2014, 4, 45903–45919. [Google Scholar] [CrossRef]
- Jongprateep, O.; Puranasamriddhi, R.; Palomas, J. Nanoparticulate Titanium Dioxide Synthesized by Sol–Gel and Solution Combustion Techniques. Ceram. Int. 2015, 41, S169–S173. [Google Scholar] [CrossRef]
- Abdel-Messih, M.F.; Ahmed, M.A.; El-Sayed, A.S. Photocatalytic Decolorization of Rhodamine B Dye Using Novel Mesoporous SnO2–TiO2 Nano Mixed Oxides Prepared by Sol–Gel Method. J. Photochem. Photobiol. Chem. 2013, 260, 1–8. [Google Scholar] [CrossRef]
- Rangel-Vázquez, I.; Angel, G.D.; Bertin, V.; González, F.; Vázquez-Zavala, A.; Arrieta, A.; Padilla, J.M.; Barrera, A.; Ramos-Ramirez, E. Synthesis and Characterization of Sn Doped TiO2 Photocatalysts: Effect of Sn Concentration on the Textural Properties and on the Photocatalytic Degradation of 2,4-Dichlorophenoxyacetic Acid. J. Alloys Compd. 2015, 643, S144–S149. [Google Scholar] [CrossRef]
- Wei, X.; Cai, H.; Feng, Q.; Liu, Z.; Ma, D.; Chen, K.; Huang, Y. Synthesis of Co-Existing Phases Sn-TiO2 Aerogel by Ultrasonic-Assisted Sol-Gel Method without Calcination. Mater. Lett. 2018, 228, 379–383. [Google Scholar] [CrossRef]
- Falk, G.S.; Borlaf, M.; López-Muñoz, M.J.; Fariñas, J.C.; Rodrigues Neto, J.B.; Moreno, R. Microwave-Assisted Synthesis of TiO2 Nanoparticles: Photocatalytic Activity of Powders and Thin Films. J. Nanopart. Res. 2018, 20, 23. [Google Scholar] [CrossRef]
- Simonsen, M.E.; Li, Z.; Søgaard, E.G. Influence of the OH Groups on the Photocatalytic Activity and Photoinduced Hydrophilicity of Microwave Assisted Sol–Gel TiO2 Film. Appl. Surf. Sci. 2009, 255, 8054–8062. [Google Scholar] [CrossRef]
- Andrade-Guel, M.; Díaz-Jiménez, L.; Cortés-Hernández, D.; Cabello-Alvarado, C.; Ávila-Orta, C.; Bartolo-Pérez, P.; Gamero-Melo, P. Microwave Assisted Sol–Gel Synthesis of Titanium Dioxide Using Hydrochloric and Acetic Acid as Catalysts. Bol. Soc. Esp. Cerámica Vidr. 2019, 58, 171–177. [Google Scholar] [CrossRef]
- Reda, S.M.; Khairy, M.; Mousa, M.A. Photocatalytic Activity of Nitrogen and Copper Doped TiO2 Nanoparticles Prepared by Microwave-Assisted Sol-Gel Process. Arab. J. Chem. 2020, 13, 86–95. [Google Scholar] [CrossRef]
- Carp, O.; Huisman, C.L.; Reller, A. Photoinduced Reactivity of Titanium Dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Preda, S.; Pandele-Cușu, J.; Petrescu, S.V.; Ciobanu, E.M.; Petcu, G.; Culiță, D.C.; Apostol, N.G.; Costescu, R.M.; Raut, I.; Constantin, M.; et al. Photocatalytic and Antibacterial Properties of Doped TiO2 Nanopowders Synthesized by Sol-Gel Method. Gels 2022, 8, 673. [Google Scholar] [CrossRef]
- Li, X.; Wang, L.; Lu, X. Preparation of Silver-Modified TiO2 via Microwave-Assisted Method and Its Photocatalytic Activity for Toluene Degradation. J. Hazard. Mater. 2010, 177, 639–647. [Google Scholar] [CrossRef]
- Tongon, W.; Chawengkijwanich, C.; Chiarakorn, S. Visible Light Responsive Ag/TiO2/MCM-41 Nanocomposite Films Synthesized by a Microwave Assisted Sol–Gel Technique. Superlattices Microstruct. 2014, 69, 108–121. [Google Scholar] [CrossRef]
- Baghbanzadeh, M.; Carbone, L.; Cozzoli, P.D.; Kappe, C.O. Microwave-Assisted Synthesis of Colloidal Inorganic Nanocrystals. Angew. Chem. Int. Ed. 2011, 50, 11312–11359. [Google Scholar] [CrossRef]
- Das, S.; Mukhopadhyay, A.K.; Datta, S.; Basu, D. Prospects of Microwave Processing: An Overview. Bull. Mater. Sci. 2009, 32, 1–13. [Google Scholar] [CrossRef]
- Li, Y.; Yang, W. Microwave Synthesis of Zeolite Membranes: A Review. J. Membr. Sci. 2008, 316, 3–17. [Google Scholar] [CrossRef]
- Wang, X.; Shen, J.; Pan, Q. Raman Spectroscopy of Sol–Gel Derived Titanium Oxide Thin Films. J. Raman Spectrosc. 2011, 42, 1578–1582. [Google Scholar] [CrossRef]
- Iliev, M.N.; Hadjiev, V.G.; Litvinchuk, A.P. Raman and Infrared Spectra of Brookite (TiO2): Experiment and Theory. Vib. Spectrosc. 2013, 64, 148–152. [Google Scholar] [CrossRef]
- Bindhu, M.R.; Willington, T.D.; Hatshan, M.R.; Chen, S.-M.; Chen, T.-W. Environmental Photochemistry with Sn/F Simultaneously Doped TiO2 Nanoparticles: UV and Visible Light Induced Degradation of Thiazine Dye. Environ. Res. 2022, 207, 112108. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Yu, J.C.; Lo, D.; Lam, S.K. Photocatalytic Activity of Rutile Ti1−xSnxO2 Solid Solutions. J. Catal. 1999, 183, 368–372. [Google Scholar] [CrossRef]
- Fruth, V.; Todan, L.; Codrea, C.I.; Poenaru, I.; Petrescu, S.; Aricov, L.; Ciobanu, M.; Jecu, L.; Ion, R.M.; Predoana, L. Multifunctional Composite Coatings Based on Photoactive Metal-Oxide Nanopowders (MgO/TiO2) in Hydrophobic Polymer Matrix for Stone Heritage Conservation. Nanomaterials 2021, 11, 2586. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, P.; Wright, J.D.; Sommedijk, N.A.J.M.; Phillips, D.; Roberts, S. Sol-Gel Materials: Chemistry and Applications, 1st ed.; CRC Press: London, UK, 2017; ISBN 978-1-315-27380-8. [Google Scholar]
- Zhang, Y.; Fang, Z.Z.; Sun, P.; Huang, Z.; Zheng, S. A Study on the Synthesis of Coarse TiO2 Powder with Controlled Particle Sizes and Morphology via Hydrolysis. Powder Technol. 2021, 393, 650–658. [Google Scholar] [CrossRef]
- Crişan, M.; Răileanu, M.; Drăgan, N.; Crişan, D.; Ianculescu, A.; Niţoi, I.; Oancea, P.; Şomăcescu, S.; Stănică, N.; Vasile, B.; et al. Sol–Gel Iron-Doped TiO2 Nanopowders with Photocatalytic Activity. Appl. Catal. Gen. 2015, 504, 130–142. [Google Scholar] [CrossRef]
- Drăgan, N.; Crişan, M.; Răileanu, M.; Crişan, D.; Ianculescu, A.; Oancea, P.; Şomăcescu, S.; Todan, L.; Stănică, N.; Vasile, B. The Effect of Co Dopant on TiO2 Structure of Sol–Gel Nanopowders Used as Photocatalysts. Ceram. Int. 2014, 40, 12273–12284. [Google Scholar] [CrossRef]
- Răileanu, M.; Crișan, M.; Ianculescu, A.; Crișan, D.; Drăgan, N.; Osiceanu, P.; Şomăcescu, S.; Stănică, N.; Todan, L.; Nițoi, I. The Influence of Ni Dopant on the Structure and Photocatalytic Properties of Sol-Gel TiO2 Nanopowders. Water Air Soil Pollut. 2013, 224, 1773. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Beldjebli, O.; Bensaha, R.; Panneerselvam, P. Effect of Both Sn Doping and Annealing Temperature on the Properties of Dip-Coated Nanostructured TiO2 Thin Films. J. Inorg. Organomet. Polym. Mater. 2022, 32, 1624–1636. [Google Scholar] [CrossRef]
- Wang, W.; Xu, C.; Wang, G.; Liu, Y.; Zheng, C. Synthesis and Raman Scattering Study of Rutile SnO2 Nanowires. J. Appl. Phys. 2002, 92, 2740–2742. [Google Scholar] [CrossRef]
- Kernazhitsky, L.; Shymanovska, V.; Gavrilko, T.; Naumov, V.; Fedorenko, L.; Kshnyakin, V.; Baran, J. Photoluminescence of Cr-Doped TiO2 Induced by Intense UV Laser Excitation. J. Lumin. 2015, 166, 253–258. [Google Scholar] [CrossRef]
- Rajeswari, R.; Venugopal, D.; George, A.; Raj, A.D.; Sundaram, S.J.; Bashir, A.K.H.; Maaza, M.; Kaviyarasu, K. Synthesis and Characterization of Sn-Doped TiO2 Film for Antibacterial Applications. Appl. Phys. A 2021, 127, 498. [Google Scholar] [CrossRef]
- Qin, Q.; Wang, J.; Xia, Y.; Yang, D.; Zhou, Q.; Zhu, X.; Feng, W. Synthesis and Characterization of Sn/Ni Single Doped and Co–Doped Anatase/Rutile Mixed–Crystal Nanomaterials and Their Photocatalytic Performance under UV–Visible Light. Catalysts 2021, 11, 1341. [Google Scholar] [CrossRef]
- Saha, A.; Moya, A.; Kahnt, A.; Iglesias, D.; Marchesan, S.; Wannemacher, R.; Prato, M.; Vilatela, J.J.; Guldi, D.M. Interfacial Charge Transfer in Functionalized Multi-Walled Carbon Nanotube@TiO2 Nanofibres. Nanoscale 2017, 9, 7911–7921. [Google Scholar] [CrossRef]
- Collin, F. Chemical Basis of Reactive Oxygen Species Reactivity and Involvement in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 2407. [Google Scholar] [CrossRef] [PubMed]
- Ani, I.J.; Akpan, U.G.; Olutoye, M.A.; Hameed, B.H. Photocatalytic Degradation of Pollutants in Petroleum Refinery Wastewater by TiO2- and ZnO-Based Photocatalysts: Recent Development. J. Clean. Prod. 2018, 205, 930–954. [Google Scholar] [CrossRef]
- Xiufeng, Z.; Juan, L.; Lianghai, L.; Zuoshan, W. Preparation of Crystalline Sn-Doped TiO2 and Its Application in Visible-Light Photocatalysis. J. Nanomater. 2011, 2011, 432947. [Google Scholar] [CrossRef]
- Bayan, E.M.; Lupeiko, T.G.; Pustovaya, L.E.; Volkova, M.G. Synthesis and Photocatalytic Properties of Sn–TiO2 Nanomaterials. J. Adv. Dielectr. 2020, 10, 2060018. [Google Scholar] [CrossRef]
- Rasouli, H.; Jafarpisheh, F.; Ghorbanpour, M. Synthesis and Characterization of Sn-Doped TiO2 Nanoparticles and the Evaluation of Their Photocatalytic Performance under Vis-Lights. J. Water Environ. Nanotechnol. 2022, 7, 344–350. [Google Scholar] [CrossRef]
- Mohammadi, R.; Massoumi, B. Sn/Cu-TiO2 Nanoparticles Produced via Sol-Gel Method: Synthesis, Characterization, and Photocatalytic Activity. Russ. J. Phys. Chem. A 2014, 88, 1184–1190. [Google Scholar] [CrossRef]
- Deshmukh, S.M.; Patil, S.S.; Babar, S.B.; Alshehri, S.; Ghoneim, M.M.; Tamboli, A.M.; Lam, N.H.; Truong, N.T.N.; Kim, C.D.; Tamboli, M.S.; et al. TiO2-SnO2 Nanocomposites for Photocatalytic Environmental Remediation under UV-Light. Metals 2022, 12, 733. [Google Scholar] [CrossRef]
- Gómez-Obando, V.A.; García-Mora, A.-M.; Basante, J.S.; Hidalgo, A.; Galeano, L.-A. CWPO Degradation of Methyl Orange at Circumneutral PH: Multi-Response Statistical Optimization, Main Intermediates and by-Products. Front. Chem. 2019, 7, 772. [Google Scholar] [CrossRef] [PubMed]
- Szeto, W.; Kan, C.W.; Yuen, C.W.M.; Chan, S.-W.; Lam, K.H. Effective Photodegradation of Methyl Orange Using Fluidized Bed Reactor Loaded with Cross-Linked Chitosan Embedded Nano-CdS Photocatalyst. Int. J. Chem. Eng. 2014, 2014, 270946. [Google Scholar] [CrossRef]
- Chen, T.; Zheng, Y.; Lin, J.-M.; Chen, G. Study on the Photocatalytic Degradation of Methyl Orange in Water Using Ag/ZnO as Catalyst by Liquid Chromatography Electrospray Ionization Ion-Trap Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2008, 19, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Gingașu, D.; Mîndru, I.; Culiță, D.C.; Predoană, L.; Petcu, G.; Ciobanu, E.M.; Preda, S.; Pandele-Cușu, J.; Petrescu, S. MgO Obtained by Chemical and Green Synthesis Methods and Applied in Photocatalytic Degradation of Methyl Orange. Rev. Roum. Chim. 2021, 66, 463–473. [Google Scholar] [CrossRef]








| Sample | Lattice Parameters | Crystallite Size, [nm] | |
|---|---|---|---|
| a, [Å] | c, [Å] | ||
| TiO2 SG (450 °C) | 3.787815 ± 0.000310 | 9.506687 ± 0.000819 | 15 |
| 2Sn–TiO2 SG | 3.791404 ± 0.000399 | 9.520856 ± 0.001049 | 13 |
| TiO2 MW (450 °C) | 3.788359 ± 0.000278 | 9.508230 ± 0.000739 | 16 |
| 1Sn–TiO2 MW | 3.790160 ± 0.000314 | 9.520975 ± 0.000826 | 15 |
| 2Sn–TiO2 MW | 3.792443 ± 0.000516 | 9.517577 ± 0.001358 | 11 |
| 4Sn–TiO2 MW | 3.793325 ± 0.000453 | 9.524177 ± 0.001197 | 11 |
| TiO2, anatase (ICDD 21–1272) | 3.7850 | 9.5140 | - |
| Sample | Composition | Values | U.M. | Line |
|---|---|---|---|---|
| 2Sn–TiO2 SG | Ti | 57.7822 | mass% | Ti–KA |
| Sn | 2.4174 | mass% | Sn–KA | |
| O | 38.4619 | mass% | O–KA | |
| C, S, Si, Al (traces) | 1.3385 | mass% | ||
| Ti | 57.0156 | mass% | Ti–KA | |
| 1Sn–TiO2 MW | Sn | 1.2449 | mass% | Sn–KA |
| O | 40.2381 | mass% | O–KA | |
| C, Si, S (traces) | 1.5014 | mass% | ||
| Ti | 56.9732 | mass% | Ti–KA | |
| 2Sn–TiO2 MW | Sn | 3.5977 | mass% | Sn–KA |
| O | 38.0233 | mass% | O–KA | |
| C, Si, S (traces) | 1.4059 | mass% | ||
| Ti | 54.8955 | mass% | Ti–KA | |
| 4Sn–TiO2 MW | Sn | 3.4533 | mass% | Sn–KA |
| O | 39.9122 | mass% | O–KA | |
| C, Si, S (traces) | 1.7391 | mass% |
| Sample TT 500 °C | SBET (m2g−1) | Vtotal (cm3g−1) | dBJH (nm) |
|---|---|---|---|
| 1Sn–TiO2 MW | 51.6 | 0.100 | 5.55 |
| 2Sn–TiO2 MW | 66.9 | 0.100 | 4.39 |
| 2Sn–TiO2 SG | 57.9 | 0.093 | 4.84 |
| 4Sn–TiO2 MW | 68.4 | 0.113 | 4.92 |
| Sample | Band Gap (eV) |
|---|---|
| 1Sn–TiO2 MW | 3.18 |
| 2Sn–TiO2 MW | 3.11 |
| 4Sn–TiO2 MW | 3.16 |
| 2Sn–TiO2 SG | 3.13 |
| Method | Sn Content, [mol.%] | TiO2 Phase | Dye Concentration, [M] | Light Irradiation | Degradation Efficiency, [%] | Ref. | |
|---|---|---|---|---|---|---|---|
| Water molecule transport | 0.5 1 5 | A/R | Methylene blue (3 × 10−5 M) | Visible | 96% 97% 84% | [59] | |
| Sol–gel | 5 | A | Methylene blue (6 × 10−5 M) | UV visible | 98% 77% | [60] | |
| Heat treatment | 3 5 7 | A | Methyl orange (9 × 10−5 M) | UV | 83% 85% 90% | [61] | |
| Sol–gel | 0.75 1 | A | Methyl orange (6 × 10−5 M) | UV | 73% 64% | [62] | |
| Sol–gel | 3 | A/R | Methylene blue (3 × 10−5 M) | Visible | 79.5% | [55] | |
| Microwave-assisted sol-gel | 1 2 4 | A A/R/B A/R | Methyl orange (1 × 10−5 M) | UV visible | UV | Vis | Present work |
| 88% 91% 94% | 27% 37% 41% | ||||||
| Sol–gel | 2 | A | Methyl orange (1 × 10−5 M) | Visible | 29% | ||
| Sample | Reagents | Molar Ratio | pH Sol | Experimental Conditions | ||||
|---|---|---|---|---|---|---|---|---|
| TiO2/SnO2 | T (°C) | t (min) | ||||||
| 1Sn–TiO2 MW | Ti(OC4H9)4 + Tin(II) 2-ethylhexanoate | 99/1 | 36.5 | 3 | 0.003 | 10 | 60 | 10 |
| 2Sn–TiO2 MW | 98/2 | 36.5 | 3 | 0.003 | 10 | 60 | 10 | |
| 4Sn–TiO2 MW | 96/4 | 36.5 | 3 | 0.003 | 10 | 60 | 10 | |
| 2Sn–TiO2 SG | 98/2 | 36.5 | 3 | 0.003 | 10 | 25 | 60 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Predoană, L.; Ciobanu, E.M.; Petcu, G.; Preda, S.; Pandele-Cușu, J.; Anghel, E.M.; Petrescu, S.V.; Culiță, D.C.; Băran, A.; Surdu, V.-A.; et al. Photocatalytic Performance of Sn–Doped TiO2 Nanopowders for Photocatalytic Degradation of Methyl Orange Dye. Catalysts 2023, 13, 534. https://doi.org/10.3390/catal13030534
Predoană L, Ciobanu EM, Petcu G, Preda S, Pandele-Cușu J, Anghel EM, Petrescu SV, Culiță DC, Băran A, Surdu V-A, et al. Photocatalytic Performance of Sn–Doped TiO2 Nanopowders for Photocatalytic Degradation of Methyl Orange Dye. Catalysts. 2023; 13(3):534. https://doi.org/10.3390/catal13030534
Chicago/Turabian StylePredoană, Luminița, Elena Mădălina Ciobanu, Gabriela Petcu, Silviu Preda, Jeanina Pandele-Cușu, Elena Maria Anghel, Simona Viorica Petrescu, Daniela Cristina Culiță, Adriana Băran, Vasile-Adrian Surdu, and et al. 2023. "Photocatalytic Performance of Sn–Doped TiO2 Nanopowders for Photocatalytic Degradation of Methyl Orange Dye" Catalysts 13, no. 3: 534. https://doi.org/10.3390/catal13030534
APA StylePredoană, L., Ciobanu, E. M., Petcu, G., Preda, S., Pandele-Cușu, J., Anghel, E. M., Petrescu, S. V., Culiță, D. C., Băran, A., Surdu, V.-A., Vasile, B. Ș., & Ianculescu, A. C. (2023). Photocatalytic Performance of Sn–Doped TiO2 Nanopowders for Photocatalytic Degradation of Methyl Orange Dye. Catalysts, 13(3), 534. https://doi.org/10.3390/catal13030534











