Facile Synthesis of Poly(o-anisidine)/Graphitic Carbon Nitride/Zinc Oxide Composite for Photo-Catalytic Degradation of Congo Red Dye
Abstract
1. Introduction
2. Results and Discussion
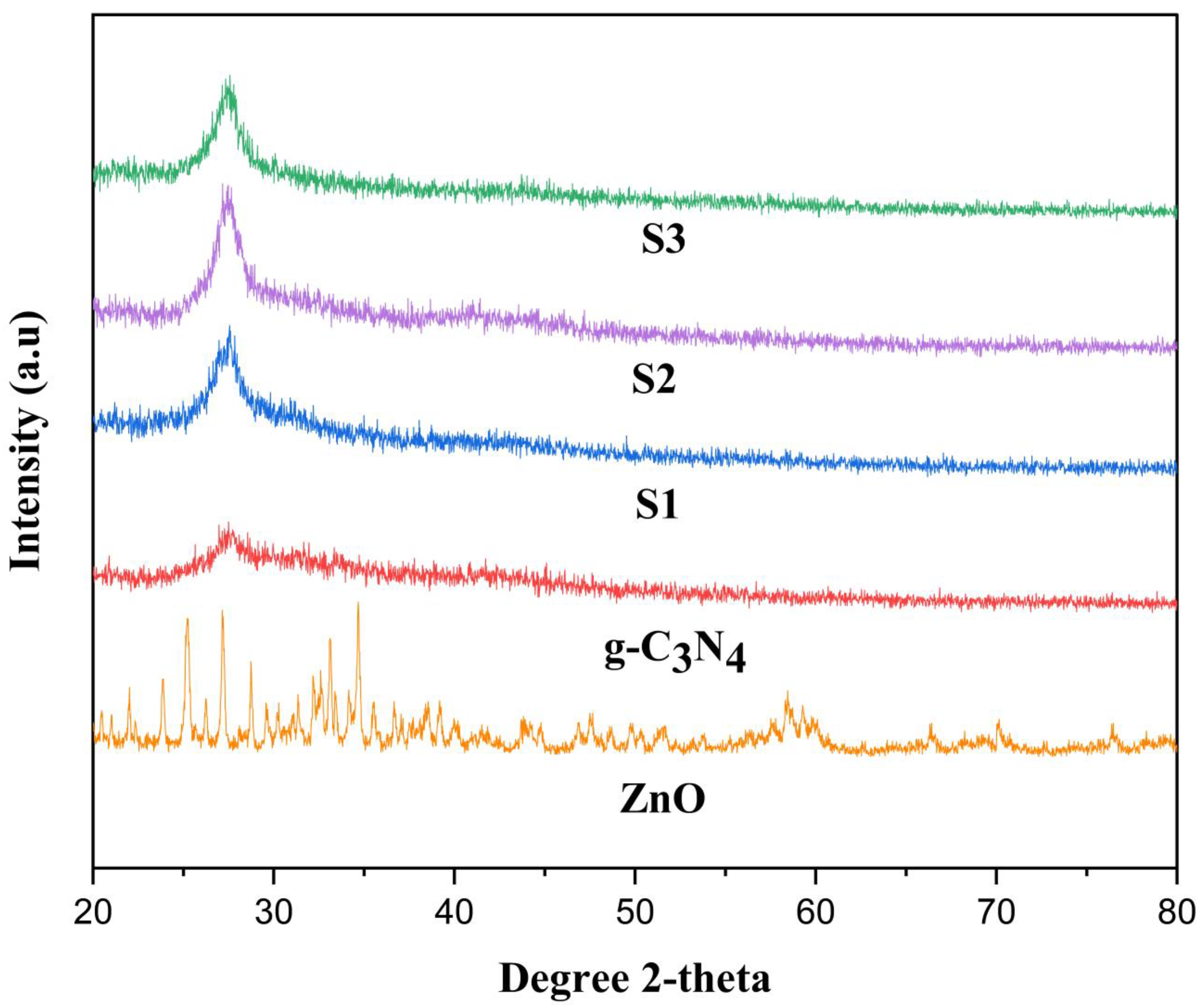
2.1. X-ray Diffraction (XRD)
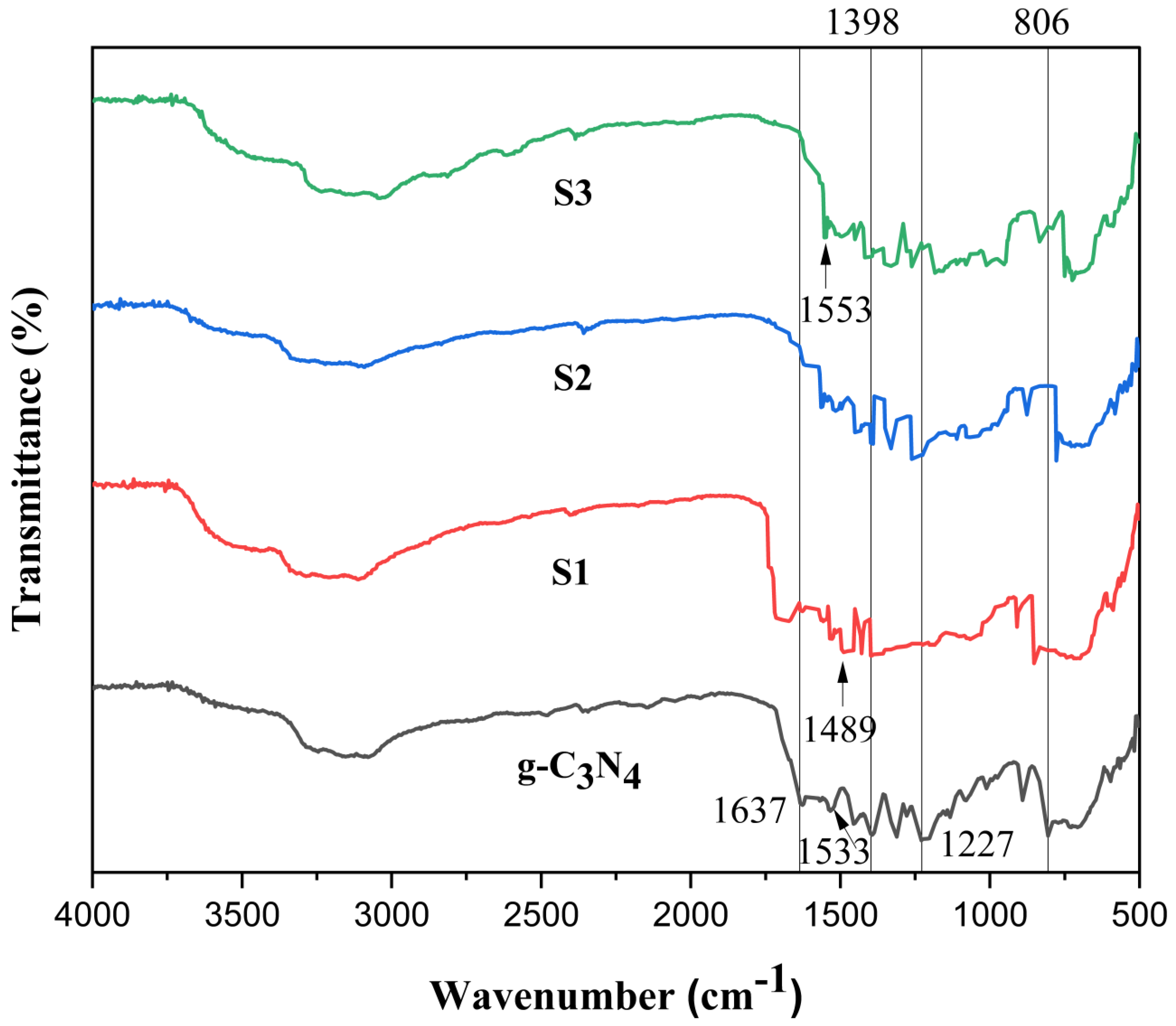
2.2. Fourier Transform Infrared Spectroscopy (FTIR)
2.3. Scanning Electron Microscope (SEM)
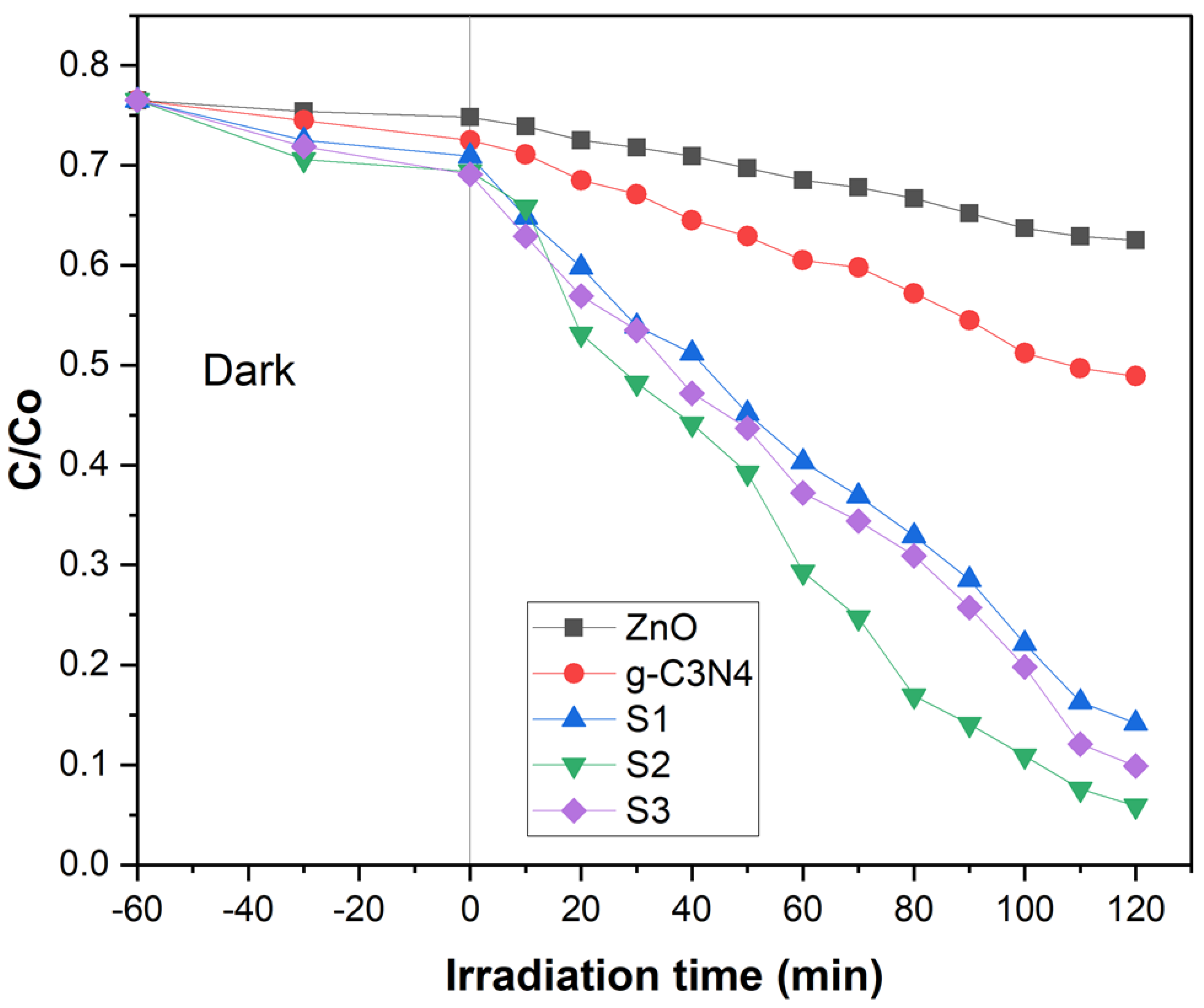
2.4. Photo-Catalytic Activity
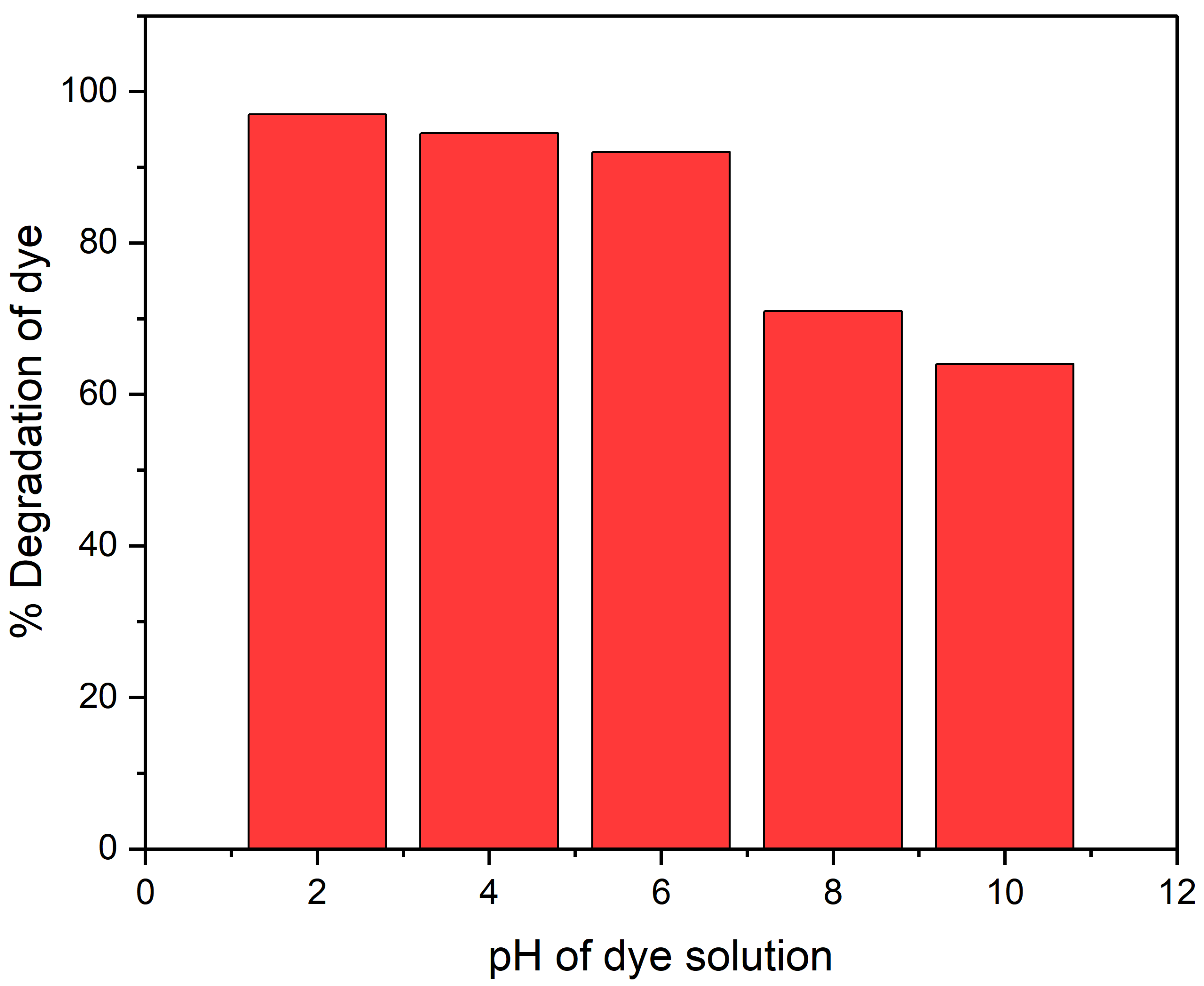
2.4.1. pH Effect on CR Dye Degradation
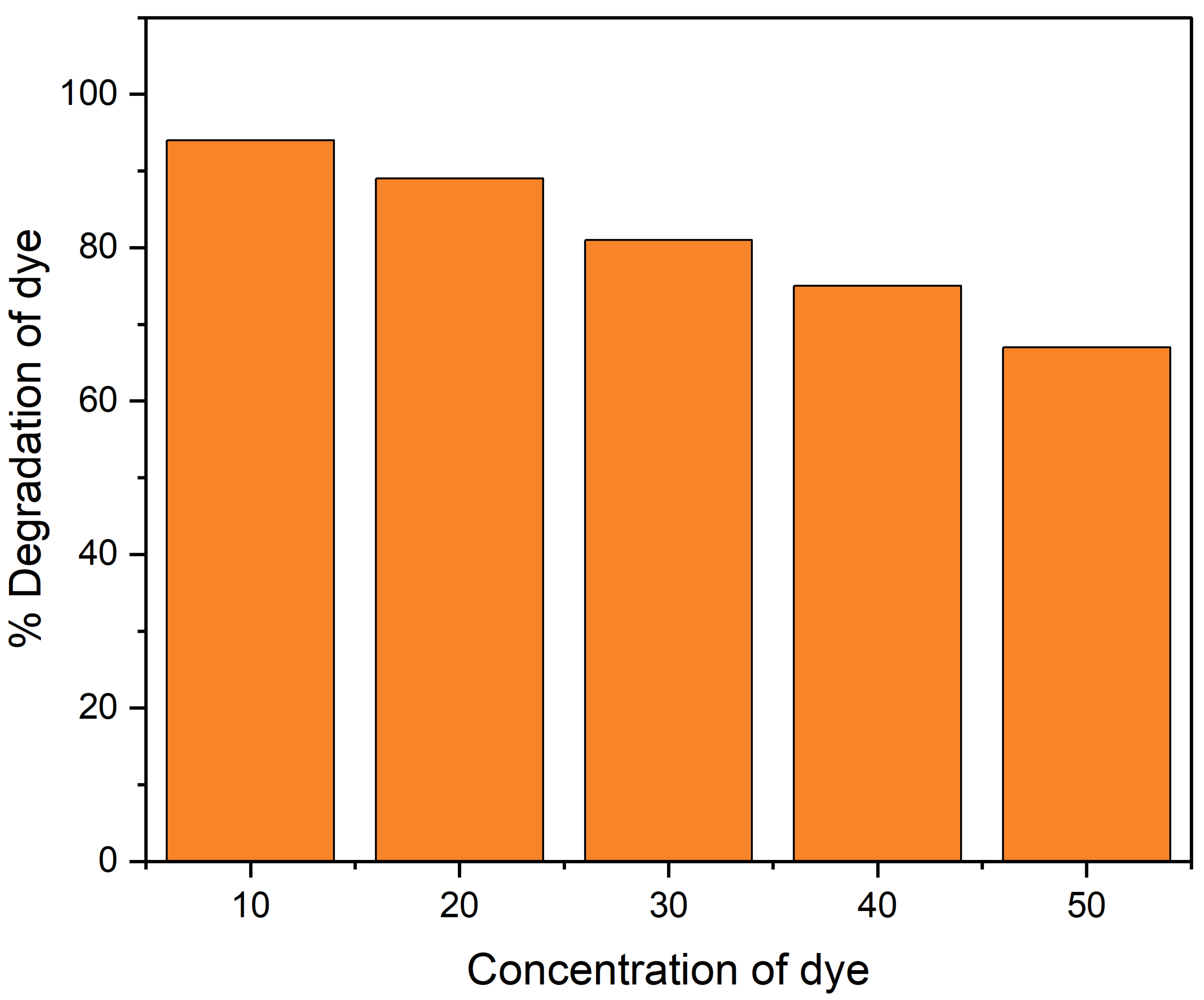
2.4.2. Effect of Dye Concentration
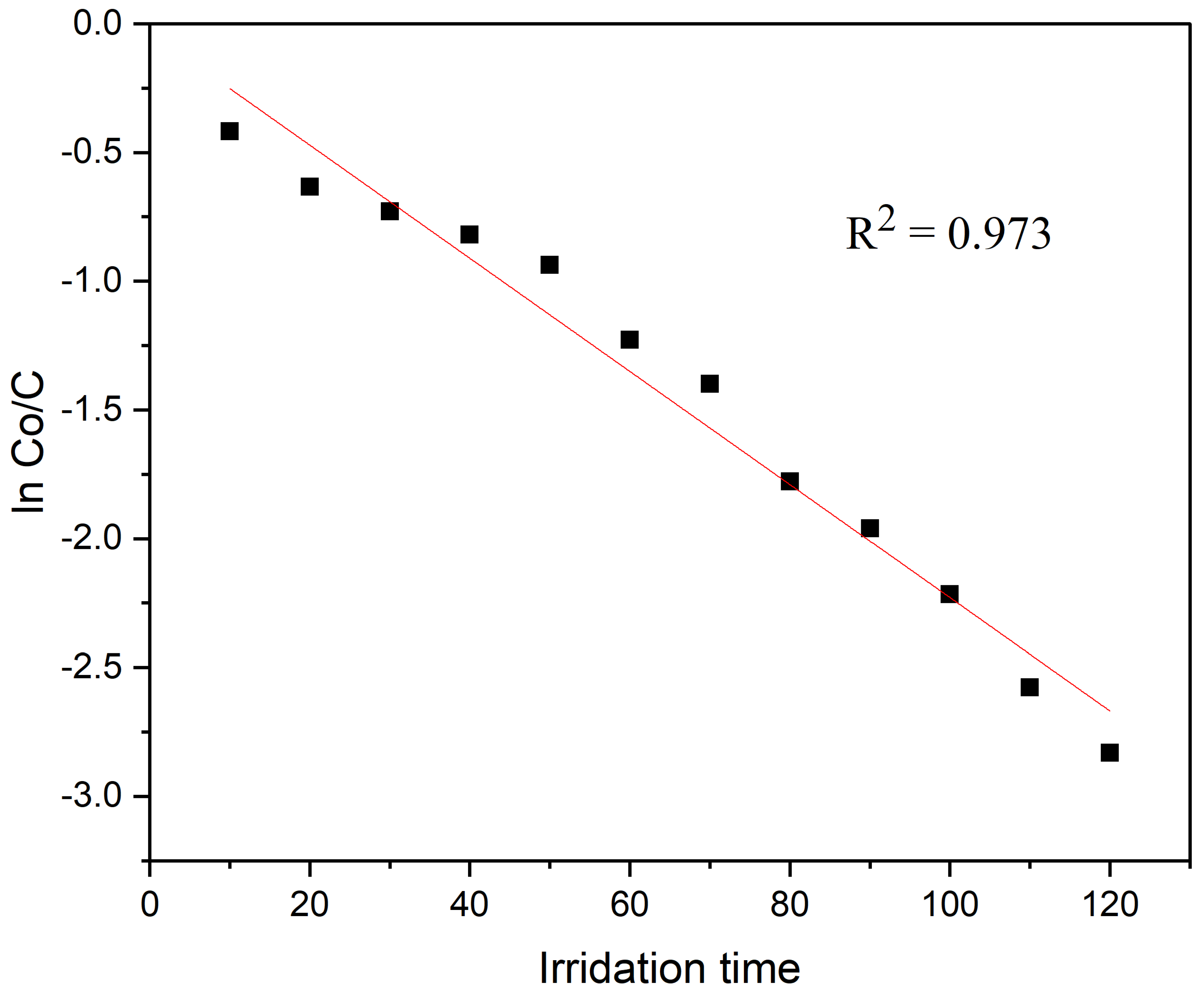
2.4.3. Kinetic Analysis
3. Photo-Catalytic Mechanism
4. Experimental Section
4.1. Materials
4.2. Synthesis of g-C3N4
4.3. Synthesis of ZnO NPs
4.4. Regeneration of ZnO NPs
4.5. Synthesis of POA/g-C3N4/ZnO Composites
4.6. Test of Photo-Catalytic Activity
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, C.; Wu, X.; Shan, J.; Liu, J.; Huang, X. Preparation, Characterization of Graphitic Carbon Nitride Photo-Catalytic Nanocomposites and Their Application in Wastewater Remediation: A Review. Crystals 2021, 11, 723. [Google Scholar] [CrossRef]
- Zarei, M.; Bahrami, J.; Zarei, M. Zirconia nanoparticle-modified graphitic carbon nitride nanosheets for effective photocatalytic degradation of 4-nitrophenol in water. Appl. Water Sci. 2019, 9, 175. [Google Scholar] [CrossRef]
- Buthelezi, S.P.; Olaniran, A.O.; Pillay, B. Textile dye removal from wastewater effluents using bioflocculants produced by indigenous bacterial isolates. Molecules 2012, 17, 14260–14274. [Google Scholar] [CrossRef] [PubMed]
- Doornbusch, G.; van der Wal, M.; Tedesco, M.; Post, J.; Nijmeijer, K.; Borneman, Z. Multistage electrodialysis for desalination of natural seawater. Desalination 2021, 505, 114973. [Google Scholar] [CrossRef]
- Saleh, I.A.; Zouari, N.; Al-Ghouti, M.A. Removal of pesticides from water and wastewater: Chemical, physical and biological treatment approaches. Environ. Technol. Innov. 2020, 19, 101026. [Google Scholar] [CrossRef]
- Van Tran, V.; Park, D.; Lee, Y.-C. Hydrogel applications for adsorption of contaminants in water and wastewater treatment. Environ. Sci. Pollut. Res. 2018, 25, 24569–24599. [Google Scholar] [CrossRef]
- Zuo, K.; Wang, K.; DuChanois, R.M.; Fang, Q.; Deemer, E.M.; Huang, X.; Xin, R.; Said, I.A.; He, Z.; Feng, Y. Selective membranes in water and wastewater treatment: Role of advanced materials. Mater. Today 2021, 50, 516–532. [Google Scholar] [CrossRef]
- Kumar, A.; Nidheesh, P.; Kumar, M.S. Composite wastewater treatment by aerated electrocoagulation and modified peroxi-coagulation processes. Chemosphere 2018, 205, 587–593. [Google Scholar] [CrossRef]
- Ejraei, A.; Aroon, M.A.; Saravani, A.Z. Wastewater treatment using a hybrid system combining adsorption, photocatalytic degradation and membrane filtration processes. J. Water Process Eng. 2019, 28, 45–53. [Google Scholar] [CrossRef]
- Wang, A.; Wang, Y.; Yu, W.; Huang, Z.; Fang, Y.; Long, L.; Song, Y.; Cifuentes, M.P.; Humphrey, M.G.; Zhang, L. TiO2–multi-walled carbon nanotube nanocomposites: Hydrothermal synthesis and temporally-dependent optical properties. RSC Adv. 2016, 6, 20120–20127. [Google Scholar] [CrossRef]
- Manea, Y.K.; Khan, A.M.; Nabi, S.A. Facile synthesis of Mesoporous Sm@ POA/TP and POA/TP nanocomposites with excellent performance for the photocatalytic degradation of MB and MG dyes. J. Alloys Compd. 2019, 791, 1046–1062. [Google Scholar] [CrossRef]
- Khamngoen, K.; Paradee, N.; Sirivat, A. Chemical oxidation polymerization and characterization of poly ortho-anisidine nanoparticles. J. Polym. Res. 2016, 23, 172. [Google Scholar] [CrossRef]
- Xu, B.; Ahmed, M.B.; Zhou, J.L.; Altaee, A.; Xu, G.; Wu, M. Graphitic carbon nitride based nanocomposites for the photocatalysis of organic contaminants under visible irradiation: Progress, limitations and future directions. Sci. Total Environ. 2018, 633, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Palanivel, B.; Mani, A. Conversion of a type-II to a Z-scheme heterojunction by intercalation of a 0D electron mediator between the integrative NiFe2O4/g-C3N4 composite nanoparticles: Boosting the radical production for photo-fenton degradation. ACS Omega 2020, 5, 19747–19759. [Google Scholar] [CrossRef]
- Ong, W.-J.; Tan, L.-L.; Ng, Y.H.; Yong, S.-T.; Chai, S.-P. Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: Are we a step closer to achieving sustainability? Chem. Rev. 2016, 116, 7159–7329. [Google Scholar] [CrossRef]
- Vinothkumar, V.; Kesavan, G.; Chen, S.-M. Graphitic carbon nitride nanosheets incorporated with polypyrrole nanocomposite: A sensitive metal-free electrocatalyst for determination of antibiotic drug nitrofurantoin. Colloids Surf. A Physicochem. Eng. Asp. 2021, 629, 127433. [Google Scholar] [CrossRef]
- Paul, D.R.; Gautam, S.; Panchal, P.; Nehra, S.P.; Choudhary, P.; Sharma, A. ZnO-modified g-C3N4: A potential photocatalyst for environmental application. ACS Omega 2020, 5, 3828–3838. [Google Scholar] [CrossRef]
- Moafi, H.F.; Zanjanchi, M.A.; Shojaie, A.F. Tungsten-doped ZnO nanocomposite: Synthesis, characterization, and highly active photocatalyst toward dye photodegradation. Mater. Chem. Phys. 2013, 139, 856–864. [Google Scholar] [CrossRef]
- Fang, J.; Fan, H.; Ma, Y.; Wang, Z.; Chang, Q. Surface defects control for ZnO nanorods synthesized by quenching and their anti-recombination in photocatalysis. Appl. Surf. Sci. 2015, 332, 47–54. [Google Scholar] [CrossRef]
- Saravanan, R.; Agarwal, S.; Gupta, V.K.; Khan, M.M.; Gracia, F.; Mosquera, E.; Narayanan, V.; Stephen, A. Line defect Ce3+ induced Ag/CeO2/ZnO nanostructure for visible-light photocatalytic activity. J. Photochem. Photobiol. A Chem. 2018, 353, 499–506. [Google Scholar] [CrossRef]
- Yim, K.; Lee, J.; Lee, D.; Lee, M.; Cho, E.; Lee, H.S.; Nahm, H.-H.; Han, S. Property database for single-element doping in ZnO obtained by automated first-principles calculations. Sci. Rep. 2017, 7, 40907. [Google Scholar] [CrossRef] [PubMed]
- Mohd, S.; Wani, A.A.; Khan, A.M. ZnO/POA functionalized metal-organic framework ZIF-8 nanomaterial for dye removal. Clean. Chem. Eng. 2022, 3, 100047. [Google Scholar] [CrossRef]
- Nie, Y.-C.; Yu, F.; Wang, L.-C.; Xing, Q.-J.; Liu, X.; Pei, Y.; Zou, J.-P.; Dai, W.-L.; Li, Y.; Suib, S.L. Photocatalytic degradation of organic pollutants coupled with simultaneous photocatalytic H2 evolution over graphene quantum dots/Mn-N-TiO2/g-C3N4 composite catalysts: Performance and mechanism. Appl. Catal. B Environ. 2018, 227, 312–321. [Google Scholar] [CrossRef]
- Liu, W.; Wang, M.; Xu, C.; Chen, S. Facile synthesis of g-C3N4/ZnO composite with enhanced visible light photooxidation and photoreduction properties. Chem. Eng. J. 2012, 209, 386–393. [Google Scholar] [CrossRef]
- Deng, Y.; Liu, J.; Huang, Y.; Ma, M.; Liu, K.; Dou, X.; Wang, Z.; Qu, S.; Wang, Z. Engineering the photocatalytic behaviors of g/C3N4-based metal-free materials for degradation of a representative antibiotic. Adv. Funct. Mater. 2020, 30, 2002353. [Google Scholar] [CrossRef]
- Ahmad, I. Comparative study of metal (Al, Mg, Ni, Cu and Ag) doped ZnO/g-C3N4 composites: Efficient photocatalysts for the degradation of organic pollutants. Sep. Purif. Technol. 2020, 251, 117372. [Google Scholar] [CrossRef]
- Wu, H.; Lin, S.; Chen, C.; Liang, W.; Liu, X.; Yang, H. A new ZnO/rGO/polyaniline ternary nanocomposite as photocatalyst with improved photocatalytic activity. Mater. Res. Bull. 2016, 83, 434–441. [Google Scholar] [CrossRef]
- Li, X.; Wang, B.; Yin, W.; Di, J.; Xia, J.; Zhu, W.; Li, H. Cu2+ modified g-C3N4 photocatalysts for visible light photocatalytic properties. Acta Phys.-Chim. Sin. 2020, 36, 1902001. [Google Scholar] [CrossRef]
- Pandiselvi, K.; Fang, H.; Huang, X.; Wang, J.; Xu, X.; Li, T. Constructing a novel carbon nitride/polyaniline/ZnO ternary heterostructure with enhanced photocatalytic performance using exfoliated carbon nitride nanosheets as supports. J. Hazard. Mater. 2016, 314, 67–77. [Google Scholar] [CrossRef]
- Wan, Z.; Zhang, G.; Wu, X.; Yin, S. Novel visible-light-driven Z-scheme Bi12GeO20/g-C3N4 photocatalyst: Oxygen-induced pathway of organic pollutants degradation and proton assisted electron transfer mechanism of Cr (VI) reduction. Appl. Catal. B Environ. 2017, 207, 17–26. [Google Scholar] [CrossRef]
- Thi, T.A.N.; Vu, A.-T. Nanocomposite ZnO/g-C3N4 for Improved Degradation of Dyes under Visible Light: Facile Preparation, Characterization, and Performance Investigations. Bull. Chem. React. Eng. Catal. 2022, 17, 403–419. [Google Scholar]
- Saravanan, R.; Khan, M.M.; Gupta, V.K.; Mosquera, E.; Gracia, F.; Narayanan, V.; Stephen, A. ZnO/Ag/CdO nanocomposite for visible light-induced photocatalytic degradation of industrial textile effluents. J. Colloid Interface Sci. 2015, 452, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Zhu, F.; Gu, W.; Sun, L.; Shi, W.; Hua, Y. Construction of nitrogen-doped graphene quantum dots-BiVO4/gC3N4 Z-scheme photocatalyst and enhanced photocatalytic degradation of antibiotics under visible light. RSC Adv. 2016, 6, 61162–61174. [Google Scholar] [CrossRef]
- Singh, J.; Kalamdhad, A.S.; Koduru, J.R. Potential degradation of hazardous dye Congo red by nano-metallic particles synthesized from the automobile shredder residue. Nanotechnol. Environ. Eng. 2017, 2, 10. [Google Scholar] [CrossRef]
- Sowmya, S.; Madhu, G.; Hashir, M. Studies on Nano-Engineered TiO2 Photo Catalyst for Effective Degradation of Dye. IOP Conf. Ser. Mater. Sci. Eng. 2018, 310, 012026. [Google Scholar] [CrossRef]
- Khairnar, S.D.; Patil, M.R.; Shrivastava, V.S. Hydrothermally synthesized nanocrystalline Nb2O5 and its visible-light photocatalytic activity for the degradation of congo red and methylene blue. Iran. J. Catal. 2018, 8, 143–150. [Google Scholar]
- Sun, J.-H.; Wang, Y.-K.; Sun, R.-X.; Dong, S.-Y. Photodegradation of azo dye Congo Red from aqueous solution by the WO3–TiO2/activated carbon (AC) photocatalyst under the UV irradiation. Mater. Chem. Phys. 2009, 115, 303–308. [Google Scholar] [CrossRef]
- Villarreal, R.; Luque-Morales, M.; Chinchillas-Chinchillas, M.; Luque, P. Langmuir-Hinshelwood-Hougen-Watson model for the study of photodegradation properties of zinc oxide semiconductor nanoparticles synthetized by Peumus boldus. Results Phys. 2022, 36, 105421. [Google Scholar] [CrossRef]
- Hanumaiah Anupama, B.; AL-Gunaid, M.Q.; Shivanna Shasikala, B.; Theranya Ereppa, S.; Kavya, R.; Hatna Siddaramaiah, B.; Sangameshwara Madhukar, B. Poly (o-anisidine) Encapsulated K2ZrO3 Nano-core based Gelatin Nano Composites: Investigations of Optical, Thermal, Microcrystalline and Morphological Characteristics. ChemistrySelect 2022, 7, e202201621. [Google Scholar] [CrossRef]
- Faisal, M.; Rashed, M.A.; Ahmed, J.; Alsaiari, M.; Jalalah, M.; Alsareii, S.; Harraz, F.A. Au nanoparticles decorated polypyrrole-carbon black/g-C3N4 nanocomposite as ultrafast and efficient visible light photocatalyst. Chemosphere 2022, 287, 131984. [Google Scholar] [CrossRef]
- Thomas, M.; Naikoo, G.A.; Sheikh, M.U.D.; Bano, M.; Khan, F. Effective photocatalytic degradation of Congo red dye using alginate/carboxymethyl cellulose/TiO2 nanocomposite hydrogel under direct sunlight irradiation. J. Photochem. Photobiol. A Chem. 2016, 327, 33–43. [Google Scholar] [CrossRef]
- Yao, S.; Xue, S.; Peng, S.; Jing, M.; Qian, X.; Shen, X.; Li, T.; Wang, Y. Synthesis of graphitic carbon nitride at different thermal-pyrolysis temperature of urea and it application in lithium–sulfur batteries. J. Mater. Sci. Mater. Electron. 2018, 29, 17921–17930. [Google Scholar] [CrossRef]
- Ghorbani, H.R.; Mehr, F.P.; Pazoki, H.; Rahmani, B.M. Synthesis of ZnO nanoparticles by precipitation method. Orient. J. Chem 2015, 31, 1219–1221. [Google Scholar] [CrossRef]
- Alenizi, M.; Kumar, R.; Aslam, M.; Alseroury, F.; Barakat, M. Construction of a ternary gC3N4/TiO2@ polyaniline nanocomposite for the enhanced photocatalytic activity under solar light. Sci. Rep. 2019, 9, 12091. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jalalah, M.; Nabi, Z.; Anjum, M.N.; Ahmad, M.N.; Haq, A.U.; Qadir, M.B.; Faisal, M.; Alsaiari, M.; Irfan, M.; Harraz, F.A. Facile Synthesis of Poly(o-anisidine)/Graphitic Carbon Nitride/Zinc Oxide Composite for Photo-Catalytic Degradation of Congo Red Dye. Catalysts 2023, 13, 239. https://doi.org/10.3390/catal13020239
Jalalah M, Nabi Z, Anjum MN, Ahmad MN, Haq AU, Qadir MB, Faisal M, Alsaiari M, Irfan M, Harraz FA. Facile Synthesis of Poly(o-anisidine)/Graphitic Carbon Nitride/Zinc Oxide Composite for Photo-Catalytic Degradation of Congo Red Dye. Catalysts. 2023; 13(2):239. https://doi.org/10.3390/catal13020239
Chicago/Turabian StyleJalalah, Mohammed, Zubair Nabi, Muhammad Naveed Anjum, Mirza Nadeem Ahmad, Atta Ul Haq, Muhammad Bilal Qadir, Mohd Faisal, Mabkhoot Alsaiari, Muhammad Irfan, and Farid A. Harraz. 2023. "Facile Synthesis of Poly(o-anisidine)/Graphitic Carbon Nitride/Zinc Oxide Composite for Photo-Catalytic Degradation of Congo Red Dye" Catalysts 13, no. 2: 239. https://doi.org/10.3390/catal13020239
APA StyleJalalah, M., Nabi, Z., Anjum, M. N., Ahmad, M. N., Haq, A. U., Qadir, M. B., Faisal, M., Alsaiari, M., Irfan, M., & Harraz, F. A. (2023). Facile Synthesis of Poly(o-anisidine)/Graphitic Carbon Nitride/Zinc Oxide Composite for Photo-Catalytic Degradation of Congo Red Dye. Catalysts, 13(2), 239. https://doi.org/10.3390/catal13020239







