Photoelectrochemical Water Oxidation by Cobalt Cytochrome C Integrated-ATO Photoanode
Abstract
1. Introduction
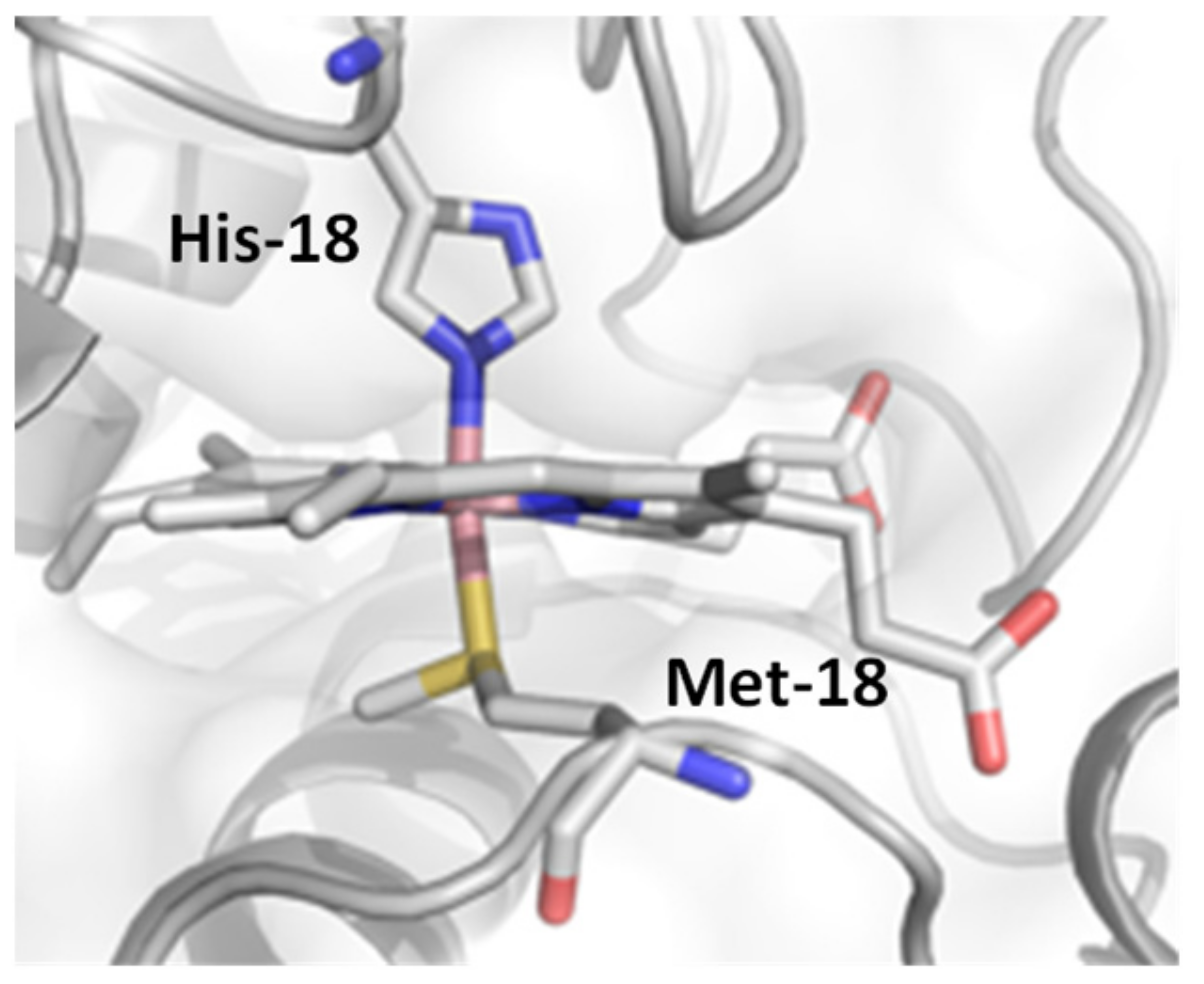
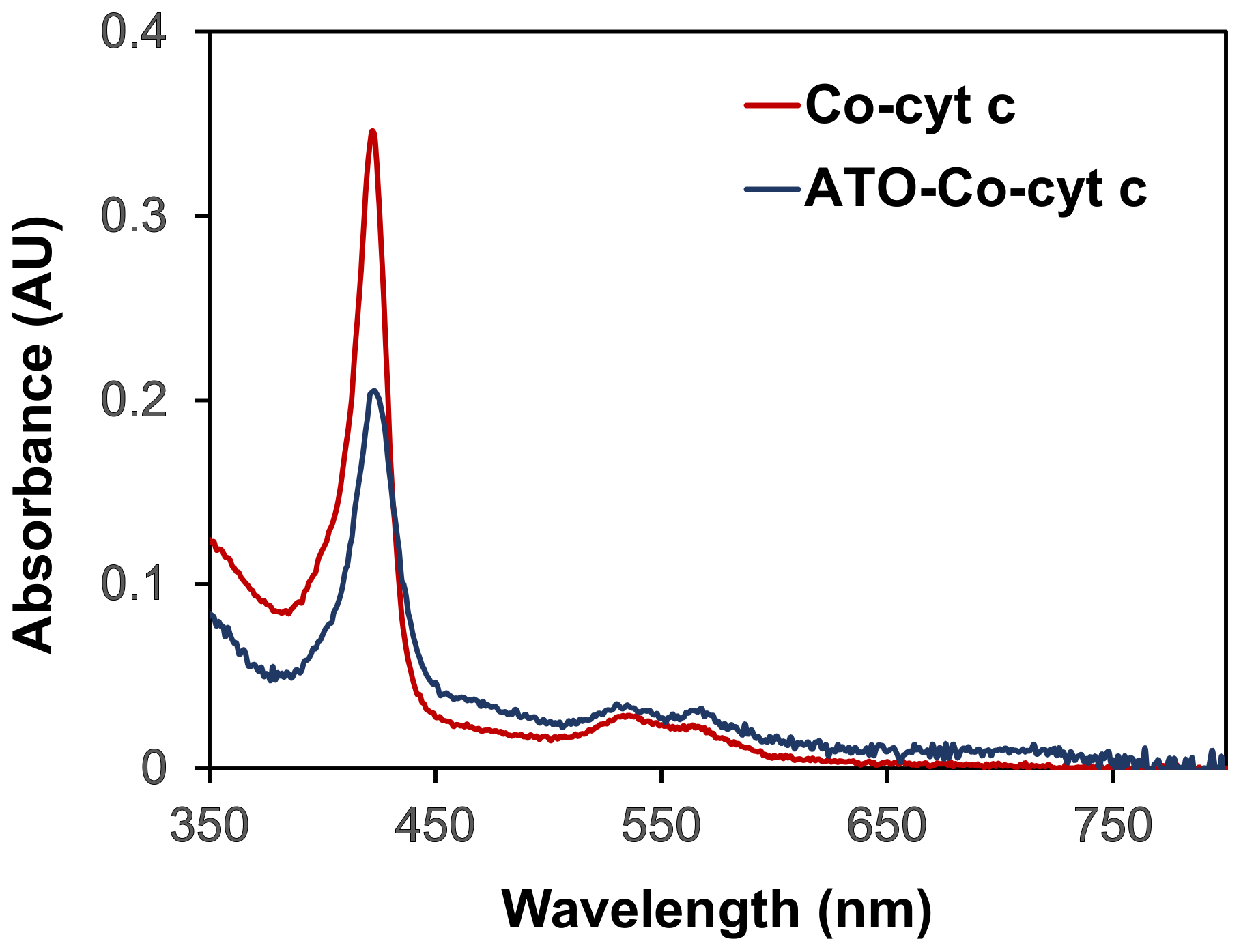
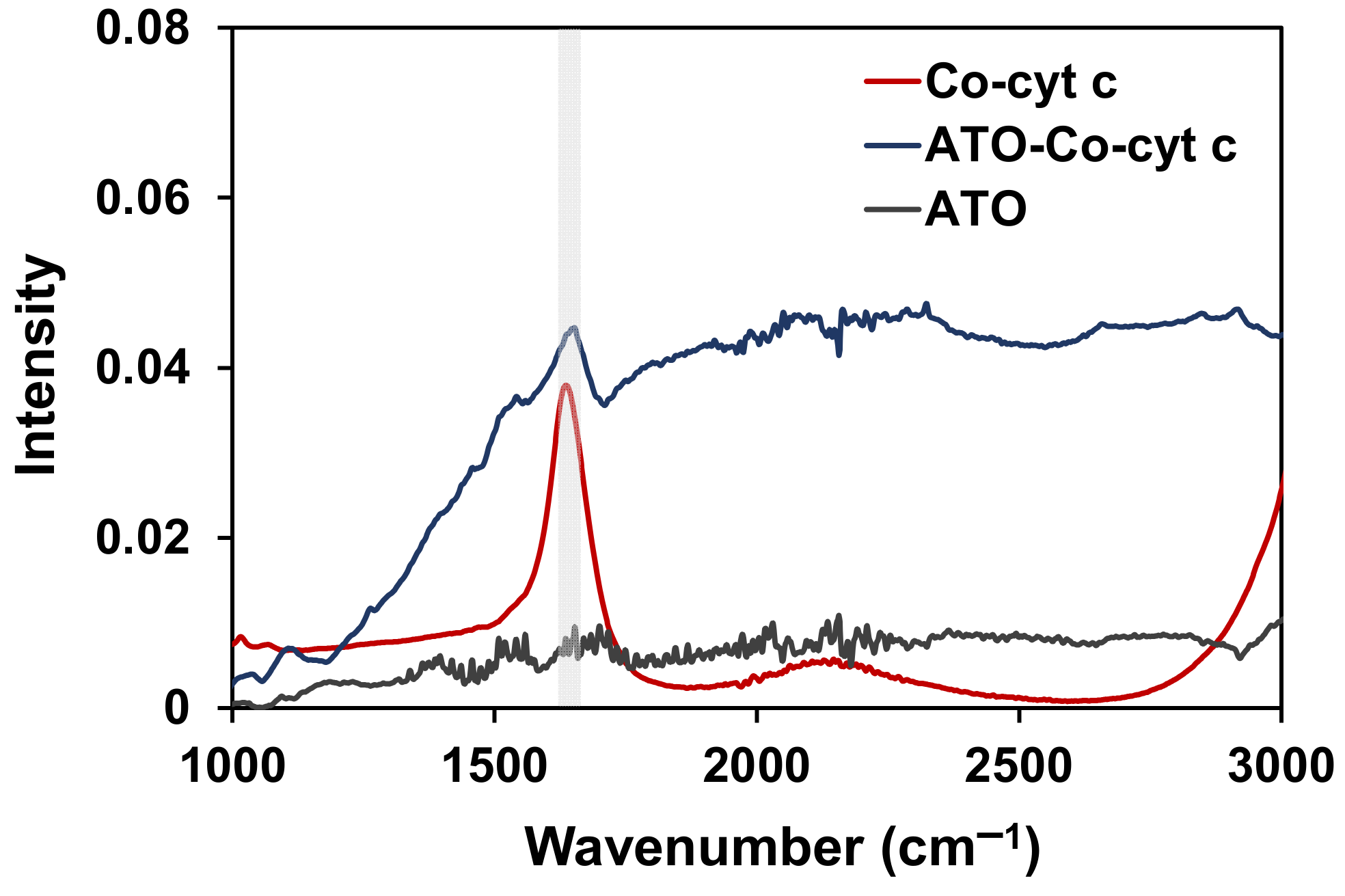
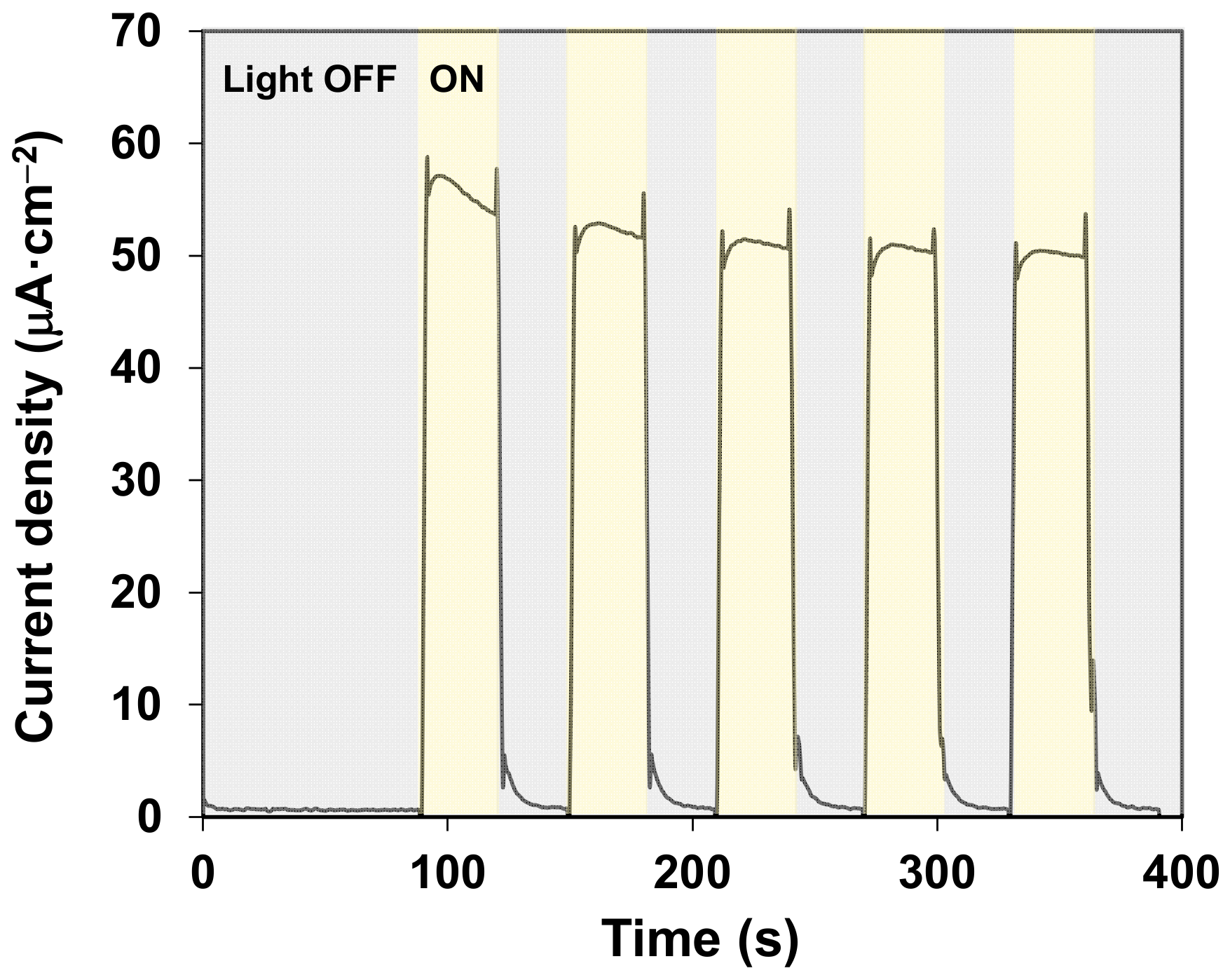
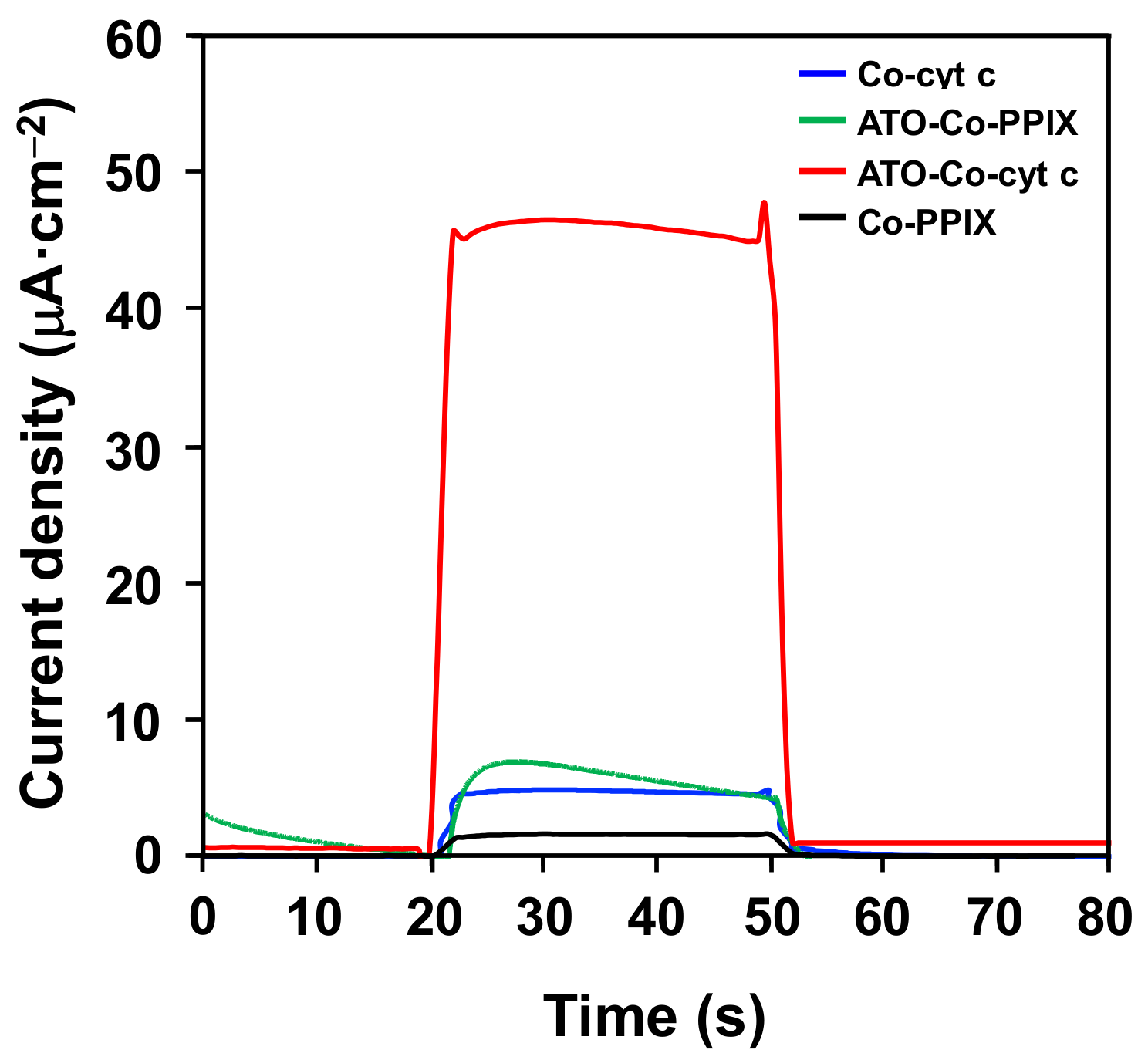
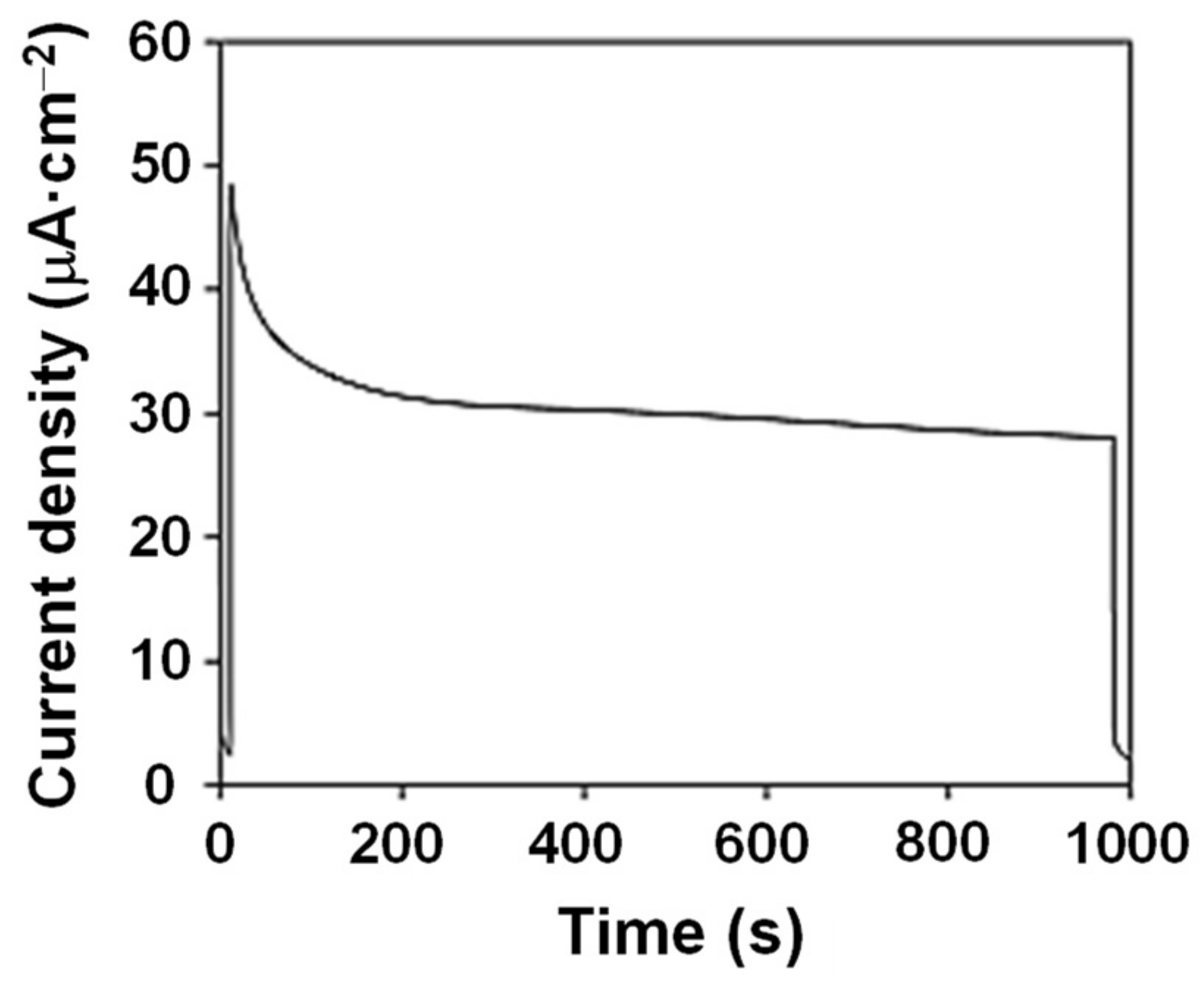
2. Results and Discussion
3. Materials and Methods
3.1. Preparation of ATO Film on FTO Glass
3.2. Metal Substitution Procedure
3.3. Cobalt Insertion
3.4. Incorporation of Co-cyt C into ATO Films
3.5. Photocurrent Measurements and Cyclic Voltammetry
3.6. ICP-MS
3.7. Photoelectrochemical Water Splitting
3.8. FTIR
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar Water Splitting Cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- Reece, S.Y.; Hamel, J.A.; Sung, K.; Jarvi, T.D.; Esswein, A.J.; Pijpers, J.J.H.; Nocera, D.G. Wireless Solar Water Splitting Using Silicon-Based Semiconductors and Earth-Abundant Catalysts. Science 2011, 334, 645. [Google Scholar] [CrossRef] [PubMed]
- Sokol, K.P.; Robinson, W.E.; Warnan, J.; Kornienko, N.; Nowaczyk, M.M.; Ruff, A.; Zhang, J.Z.; Reisner, E. Bias-free photoelectrochemical water splitting with photosystem II on a dye-sensitized photoanode wired to hydrogenase. Nat. Energy 2018, 3, 944–951. [Google Scholar] [CrossRef]
- Dalle, K.E.; Warnan, J.; Leung, J.J.; Reuillard, B.; Karmel, I.S.; Reisner, E. Electro- and Solar-Driven Fuel Synthesis with First Row Transition Metal Complexes. Chem. Rev. 2019, 119, 2752–2875. [Google Scholar] [CrossRef]
- Ye, S.; Ding, C.; Liu, M.; Wang, A.; Huang, Q.; Li, C. Water Oxidation Catalysts for Artificial Photosynthesis. Adv. Mater. 2019, 31, 1902069. [Google Scholar] [CrossRef]
- Hisatomi, T.; Kubota, J.; Domen, K. Recent advances in semiconductors for photocatalytic and photoelectrochemical water splitting. Chem. Soc. Rev. 2014, 43, 7520–7535. [Google Scholar] [CrossRef]
- Zhang, B.; Sun, L. Artificial photosynthesis: Opportunities and challenges of molecular catalysts. Chem. Soc. Rev. 2019, 48, 2216–2264. [Google Scholar] [CrossRef]
- Pannwitz, A.; Klein, D.M.; Rodríguez-Jiménez, S.; Casadevall, C.; Song, H.; Reisner, E.; Hammarström, L.; Bonnet, S. Roadmap towards solar fuel synthesis at the water interface of liposome membranes. Chem. Soc. Rev. 2021, 50, 4833–4855. [Google Scholar] [CrossRef]
- Ferreira, K.N.; Iverson, T.M.; Maghlaoui, K.; Barber, J.; Iwata, S. Architecture of the photosynthetic oxygen-evolving center. Science 2004, 303, 1831–1838. [Google Scholar] [CrossRef]
- Lubitz, W.; Chrysina, M.; Cox, N. Water oxidation in photosystem II. Photosynth Res. 2019, 142, 105–125. [Google Scholar] [CrossRef]
- Abdi, F.F.; van de Krol, R. Nature and Light Dependence of Bulk Recombination in Co-Pi-Catalyzed BiVO4 Photoanodes. J. Phys. Chem. C 2012, 116, 9398–9404. [Google Scholar] [CrossRef]
- Zhong, D.K.; Choi, S.; Gamelin, D.R. Near-complete suppression of surface recombination in solar photoelectrolysis by "Co-Pi" catalyst-modified W:BiVO4. J. Am. Chem. Soc. 2011, 133, 18370–18377. [Google Scholar] [CrossRef]
- Blakemore, J.D.; Crabtree, R.H.; Brudvig, G.W. Molecular Catalysts for Water Oxidation. Chem. Rev. 2015, 115, 12974–13005. [Google Scholar] [CrossRef]
- Macchioni, A. The Middle-Earth between Homogeneous and Heterogeneous Catalysis in Water Oxidation with Iridium. Eur. J. Inorg. Chem. 2019, 2019, 7–17. [Google Scholar] [CrossRef]
- Lloret-Fillol, J.; Costas, M. Chapter One—Water oxidation at base metal molecular catalysts. In Advances in Organometallic Chemistry; Pérez, P.J., Ed.; Academic Press/Elsevier: Cambridge, MA, USA, 2019; Volume 71, pp. 1–52. [Google Scholar]
- Casadevall, C.; Bucci, A.; Costas, M.; Lloret-Fillol, J. Chapter Four—Water oxidation catalysis with well-defined molecular iron complexes. In Advances in Inorganic Chemistry; Van Eldik, R., Hubbard, C.D., Eds.; Academic Press/Elsevier: Cambridge, MA, USA, 2019; Volume 74, pp. 151–196. [Google Scholar]
- Sayama, K.; Nomura, A.; Arai, T.; Sugita, T.; Abe, R.; Yanagida, M.; Oi, T.; Iwasaki, Y.; Abe, Y.; Sugihara, H. Photoelectrochemical decomposition of water into H2 and O2 on porous BiVO4 thin-film electrodes under visible light and significant effect of Ag ion treatment. J. Phys. Chem. B 2006, 110, 11352–11360. [Google Scholar] [CrossRef]
- Carey, A.-M.; Zhang, H.; Mieritz, D.; Volosin, A.; Gardiner, A.T.; Cogdell, R.J.; Yan, H.; Seo, D.-K.; Lin, S.; Woodbury, N.W. Photocurrent Generation by Photosynthetic Purple Bacterial Reaction Centers Interfaced with a Porous Antimony-Doped Tin Oxide (ATO) Electrode. ACS Appl. Mater. Interfaces 2016, 8, 25104–25110. [Google Scholar] [CrossRef]
- Jo, W.; Jang, J.-W.; Kong, K.-J.; Kang, H.J.; Kim, J.; Jun, H.; Parmar, K.; Lee, J.S. Phosphate Doping into Monoclinic BiVO4 for Enhanced Photoelectrochemical Water Oxidation Activity. Angew. Chem. Int. Ed. Engl. 2012, 124, 3201–3205. [Google Scholar] [CrossRef]
- Nakazono, T.; Parent, A.R.; Sakai, K. Cobalt porphyrins as homogeneous catalysts for water oxidation. Chem. Commun. 2013, 49, 6325–6327. [Google Scholar] [CrossRef]
- Wang, D.; Groves, J.T. Efficient water oxidation catalyzed by homogeneous cationic cobalt porphyrins with critical roles for the buffer base. Proc. Natl. Acad. Sci. USA 2013, 110, 15579–15584. [Google Scholar] [CrossRef]
- Dogutan, D.K.; McGuire, R.; Nocera, D.G. Electocatalytic Water Oxidation by Cobalt(III) Hangman β-Octafluoro Corroles. J. Am. Chem. Soc. 2011, 133, 9178–9180. [Google Scholar] [CrossRef]
- Han, A.; Jia, H.; Ma, H.; Ye, S.; Wu, H.; Lei, H.; Han, Y.; Cao, R.; Du, P. Cobalt porphyrin electrode films for electrocatalytic water oxidation. Phys. Chem. Chem. Phys. 2014, 16, 11224–11232. [Google Scholar] [CrossRef]
- Liu, B.; Li, J.; Wu, H.-L.; Liu, W.-Q.; Jiang, X.; Li, Z.-J.; Chen, B.; Tung, C.-H.; Wu, L.-Z. Improved Photoelectrocatalytic Performance for Water Oxidation by Earth-Abundant Cobalt Molecular Porphyrin Complex-Integrated BiVO4 Photoanode. Acs Appl. Mater. Interfaces 2016, 8, 18577–18583. [Google Scholar] [CrossRef]
- Sun, Z.; Li, J.; Zheng, H.; Liu, X.; Ye, S.; Du, P. Pyrolyzed cobalt porphyrin-modified carbon nanomaterial as an active catalyst for electrocatalytic water oxidation. Int. J. Hydrog. Energy 2015, 40, 6538–6545. [Google Scholar] [CrossRef]
- Daniel, Q.; Ambre, R.B.; Zhang, B.; Philippe, B.; Chen, H.; Li, F.; Fan, K.; Ahmadi, S.; Rensmo, H.; Sun, L. Re-Investigation of Cobalt Porphyrin for Electrochemical Water Oxidation on FTO Surface: Formation of CoOx as Active Species. ACS Catal. 2017, 7, 1143–1149. [Google Scholar] [CrossRef]
- Lloret-Fillol, J.; Costas, M. Water oxidation at base metal molecular catalysts. Adv. Organomet. Chem. 2019, 71, 1–52. [Google Scholar]
- Hu, X.-M.; Rønne, M.H.; Pedersen, S.U.; Skrydstrup, T.; Daasbjerg, K. Enhanced Catalytic Activity of Cobalt Porphyrin in CO2 Electroreduction upon Immobilization on Carbon Materials. Angew. Chem. Int. Ed. 2017, 56, 6468–6472. [Google Scholar] [CrossRef]
- Pasternack, R.F.; Francesconi, L.; Raff, D.; Spiro, E. Aggregation of nickel(II), copper(II), and zinc(II) derivatives of water-soluble porphyrins. Inorg. Chem. 1973, 12, 2606–2611. [Google Scholar] [CrossRef]
- Ow, Y.P.; Green, D.R.; Hao, Z.; Mak, T.W. Cytochrome c: Functions beyond respiration. Nat. Rev. Mol. Cell Biol. 2008, 9, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zheng, J.; Wang, J.; Xu, J.; Hayat, T.; Alharbi, N.S. Direct electrochemistry of cytochrome c immobilized on one dimensional Au nanoparticles functionalized magnetic N-doped carbon nanotubes and its application for the detection of H2O2. Sens. Actuators B Chem. 2019, 282, 85–95. [Google Scholar] [CrossRef]
- Dong, Y.; Ji, X.; Laaksonen, A.; Cao, W.; He, H.; Lu, X. Excellent Protein Immobilization and Stability on Heterogeneous C–TiO2 Hybrid Nanostructures: A Single Protein AFM Study. Langmuir 2020, 36, 9323–9332. [Google Scholar] [CrossRef] [PubMed]
- Ciornii, D.; Kölsch, A.; Zouni, A.; Lisdat, F. A precursor-approach in constructing 3D ITO electrodes for the improved performance of photosystem I-cyt c photobioelectrodes. Nanoscale 2019, 11, 15862–15870. [Google Scholar] [CrossRef]
- Hou, K.; Puzzo, D.; Helander, M.G.; Lo, S.S.; Bonifacio, L.D.; Wang, W.; Lu, Z.-H.; Scholes, G.D.; Ozin, G.A. Dye-Anchored Mesoporous Antimony-Doped Tin Oxide Electrochemiluminescence Cell. Adv. Mater. 2009, 21, 2492–2496. [Google Scholar] [CrossRef]
- Carey, A.M.; Zhang, H.; Liu, M.; Sharaf, D.; Akram, N.; Yan, H.; Lin, S.; Woodbury, N.W.; Seo, D.K. Enhancing Photocurrent Generation in Photosynthetic Reaction Center-Based Photoelectrochemical Cells with Biomimetic DNA Antenna. ChemSusChem 2017, 10, 4457–4460. [Google Scholar] [CrossRef]
- Kwan, P.; Schmitt, D.; Volosin, A.M.; McIntosh, C.L.; Seo, D.-K.; Jones, A.K. Spectroelectrochemistry of cytochrome c and azurin immobilized in nanoporous antimony-doped tin oxide. Chem. Commun. 2011, 47, 12367–12369. [Google Scholar] [CrossRef]
- Day, N.U.; Wamser, C.C. Poly-tetrakis-5,10,15,20-(4-aminophenyl)porphyrin Films as Two-Electron Oxygen Reduction Photoelectrocatalysts for the Production of H2O2. J. Phys. Chem. C 2017, 121, 11076–11082. [Google Scholar] [CrossRef]
- Wadsworth, B.L.; Khusnutdinova, D.; Urbine, J.M.; Reyes, A.S.; Moore, G.F. Expanding the Redox Range of Surface-Immobilized Metallocomplexes Using Molecular Interfaces. ACS Appl. Mater. Interfaces 2020, 12, 3903–3911. [Google Scholar] [CrossRef]
- Wadsworth, B.L.; Beiler, A.M.; Khusnutdinova, D.; Reyes Cruz, E.A.; Moore, G.F. Interplay between Light Flux, Quantum Efficiency, and Turnover Frequency in Molecular-Modified Photoelectrosynthetic Assemblies. J. Am. Chem. Soc. 2019, 141, 15932–15941. [Google Scholar] [CrossRef]
- Frasca, S.; von Graberg, T.; Feng, J.-J.; Thomas, A.; Smarsly, B.M.; Weidinger, I.M.; Scheller, F.W.; Hildebrandt, P.; Wollenberger, U. Mesoporous Indium Tin Oxide as a Novel Platform for Bioelectronics. ChemCatChem 2010, 2, 839–845. [Google Scholar] [CrossRef]
- Graf, M.; García, R.G.; Wätzig, H. Protein adsorption in fused-silica and polyacrylamide-coated capillaries. Electrophoresis 2005, 26, 2409–2417. [Google Scholar] [CrossRef]
- Kleingardner, J.G.; Kandemir, B.; Bren, K.L. Hydrogen Evolution from Neutral Water under Aerobic Conditions Catalyzed by Cobalt Microperoxidase-11. J. Am. Chem. Soc. 2014, 136, 4–7. [Google Scholar] [CrossRef]
- Primus, J.L.; Boersma, M.G.; Mandon, D.; Boeren, S.; Veeger, C.; Weiss, R.; Rietjens, I.M. The effect of iron to manganese substitution on microperoxidase 8 catalysed peroxidase and cytochrome P450 type of catalysis. J. Biol. Inorg. Chem. 1999, 4, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Dizicheh, Z.B. Design of Redox Proteins as Catalysts for Fuel Production; Arizona State University: Phoenix, AZ, USA, 2019. [Google Scholar]
- Mersch, D.; Lee, C.-Y.; Zhang, J.Z.; Brinkert, K.; Fontecilla-Camps, J.C.; Rutherford, A.W.; Reisner, E. Wiring of Photosystem II to Hydrogenase for Photoelectrochemical Water Splitting. J. Am. Chem. Soc. 2015, 137, 8541–8549. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Mandal, S.; Carey, A.-M.; Liu, M.; Chen, S.; Seo, D.-K.; Yan, H.; Woodbury, N. Interfacing Photosystem I Reaction Centers with a Porous Antimony-Doped Tin Oxide Electrode to Perform Light Driven Redox Chemistry. Biophys. J. 2019, 116, 443a. [Google Scholar] [CrossRef]
- Mieritz, D.; Liang, R.; Zhang, H.; Carey, A.-M.; Chen, S.; Volosin, A.; Lin, S.; Woodbury, N.; Seo, D.-K. Thickness-Dependent Bioelectrochemical and Energy Applications of Thickness-Controlled Meso-Macroporous Antimony-Doped Tin Oxide. Coatings 2018, 8, 128. [Google Scholar] [CrossRef]
- Call, A.; Casadevall, C.; Romero-Rivera, A.; Martin-Diaconescu, V.; Sommer, D.J.; Osuna, S.; Ghirlanda, G.; Lloret-Fillol, J. Improved Electro- and Photocatalytic Water Reduction by Confined Cobalt Catalysts in Streptavidin. ACS Catal. 2019, 9, 5837–5846. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casadevall, C.; Zhang, H.; Chen, S.; Sommer, D.J.; Seo, D.-K.; Ghirlanda, G. Photoelectrochemical Water Oxidation by Cobalt Cytochrome C Integrated-ATO Photoanode. Catalysts 2021, 11, 626. https://doi.org/10.3390/catal11050626
Casadevall C, Zhang H, Chen S, Sommer DJ, Seo D-K, Ghirlanda G. Photoelectrochemical Water Oxidation by Cobalt Cytochrome C Integrated-ATO Photoanode. Catalysts. 2021; 11(5):626. https://doi.org/10.3390/catal11050626
Chicago/Turabian StyleCasadevall, Carla, Haojie Zhang, Shaojiang Chen, Dayn J. Sommer, Dong-Kyun Seo, and Giovanna Ghirlanda. 2021. "Photoelectrochemical Water Oxidation by Cobalt Cytochrome C Integrated-ATO Photoanode" Catalysts 11, no. 5: 626. https://doi.org/10.3390/catal11050626
APA StyleCasadevall, C., Zhang, H., Chen, S., Sommer, D. J., Seo, D.-K., & Ghirlanda, G. (2021). Photoelectrochemical Water Oxidation by Cobalt Cytochrome C Integrated-ATO Photoanode. Catalysts, 11(5), 626. https://doi.org/10.3390/catal11050626







