Pd on the Composite of Perlite and Allylamine-N-isopropylacrylamide Copolymer: A Thermo-Responsive Catalyst for Hydrogenation of Nitroarenes under Mild Reaction Condition
Abstract
:1. Introduction
2. Results and Discussion
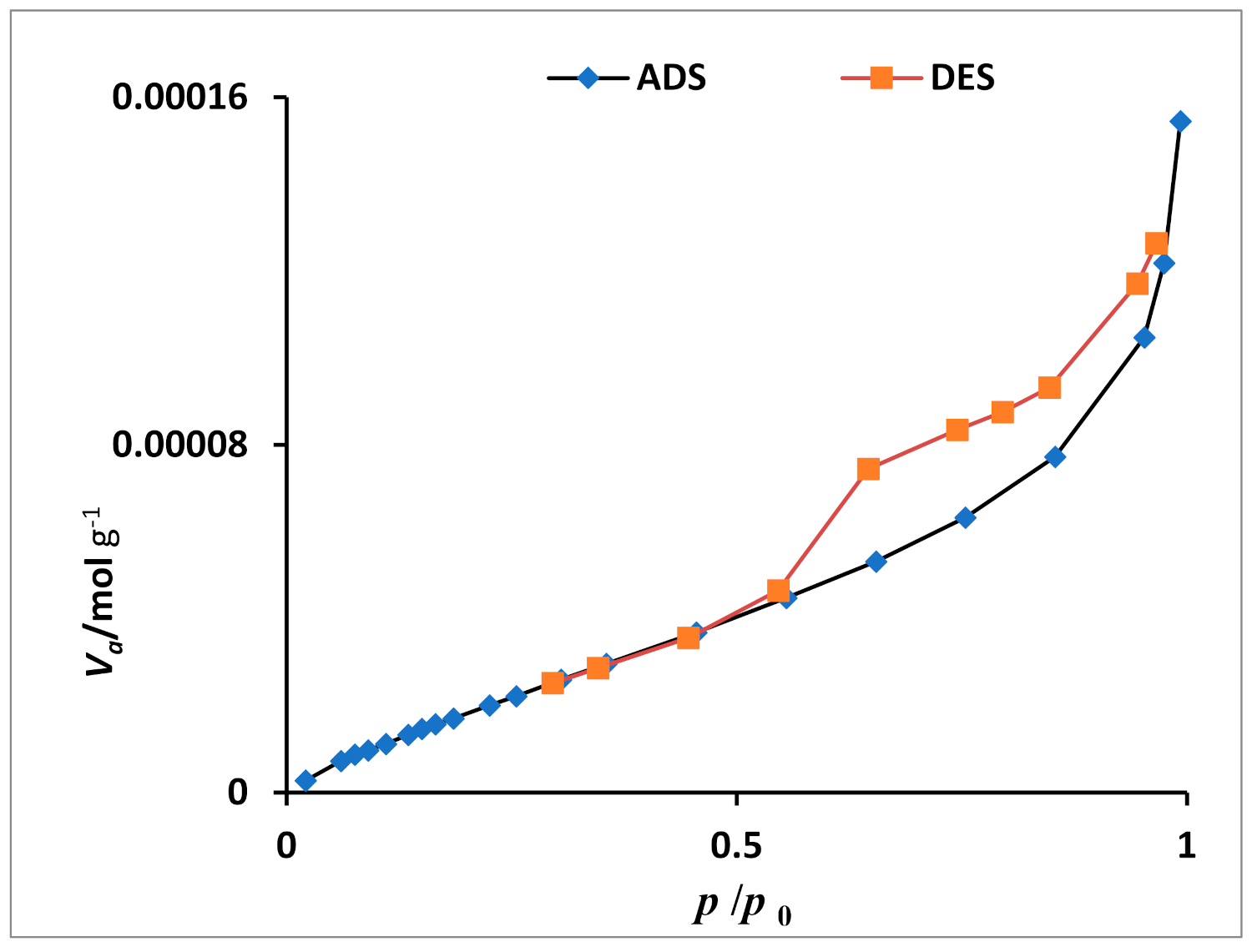
2.1. Catalyst Characterization
2.2. Activity
2.3. Comparison with Control Catalysts
2.4. Substrate Scope
2.5. Selectivity of Pd@Per-P
2.6. Comparative Study
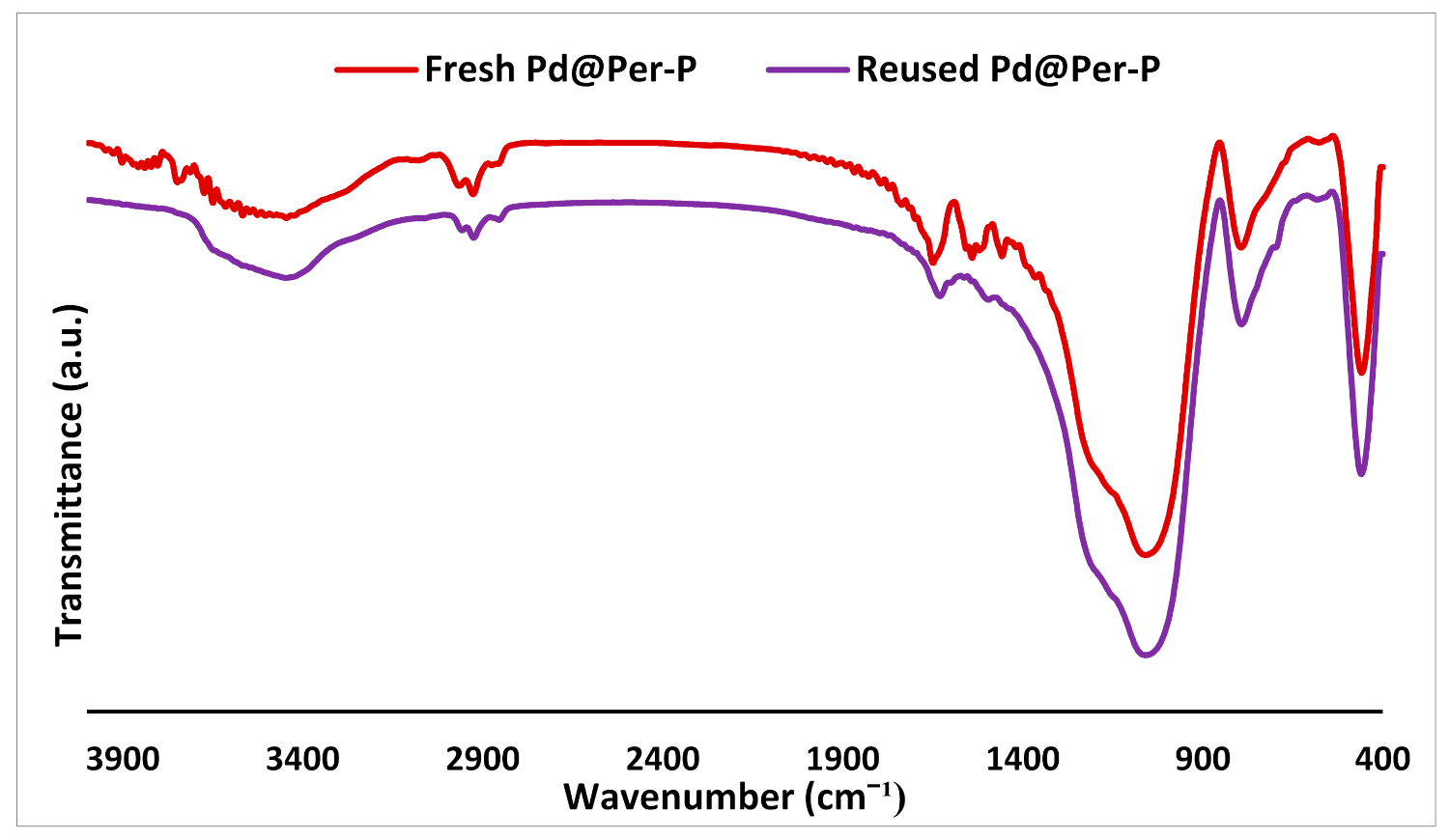
2.7. Recyclability
2.8. Hot Filtration Test
3. Materials and Methods
3.1. Materials and Instruments
3.2. Catalyst Preparation
3.2.1. Synthesis of Poly(NIPAM-co-AAm): P
3.2.2. Chemical Modification of Per: Synthesis of Per-Cl
3.2.3. Formation of Per–P Nanocomposite
3.2.4. Immobilization of Pd Nanoparticles on Per–P Nanocomposite: Pd@Per-P
3.3. Typical Procedure for Hydrogenation of Nitroarenes
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kolvari, E.; Koukabi, N.; Hosseini, M.M. Perlite: A cheap natural support for immobilization of sulfonic acid as a heterogeneous solid acid catalyst for the heterocyclic multicomponent reaction. J. Mol. Catal. A Chem. 2015, 397, 68–75. [Google Scholar] [CrossRef]
- Kolvari, E.; Koukabi, N.; Hosseini, M.M.; Khandani, Z. Perlite: An inexpensive natural support for heterogenization of HBF 4. RSC Adv. 2015, 5, 36828–36836. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Sajadi, S.M.; Rostami-Vartooni, A.; Bagherzadeh, M.; Safari, R. Immobilization of copper nanoparticles on perlite: Green synthesis, characterization and catalytic activity on aqueous reduction of 4-nitrophenol. J. Mol. Catal. A Chem. 2015, 400, 22–30. [Google Scholar] [CrossRef]
- Kara, G.K.; Rahimi, J.; Niksefat, M.; Taheri-Ledari, R.; Rabbani, M.; Maleki, A. Preparation and characterization of perlite/V2O5 nano-spheres via a novel green method: Applied for oxidation of benzyl alcohol derivatives. Mater. Chem. Phys. 2020, 250, 122991. [Google Scholar] [CrossRef]
- Akcam, O.; Karaca, E. Development of water vapor resistance feature of polyester woven fabrics by using perlite additive. J. Text. Appar. Tekst. Konfeksiyon 2013, 23, 233–240. [Google Scholar]
- Talip, Z.; Eral, M.; Hiçsönmez, Ü. Adsorption of thorium from aqueous solutions by perlite. J. Environ. Radioact. 2009, 100, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Yilmazer, S.; Ozdeniz, M.B. The effect of moisture content on sound absorption of expanded perlite plates. Build. Environ. 2005, 40, 311–318. [Google Scholar] [CrossRef] [Green Version]
- Bektas, F.; Turanli, L.; Monteiro, P. Use of perlite powder to suppress the alkali–silica reaction. Cem. Concr. Res. 2005, 35, 2014–2017. [Google Scholar] [CrossRef]
- Sengul, O.; Azizi, S.; Karaosmanoglu, F.; Tasdemir, M.A. Effect of expanded perlite on the mechanical properties and thermal conductivity of lightweight concrete. Energy Build. 2011, 43, 671–676. [Google Scholar] [CrossRef]
- Hosseini, S.; Borghei, S.; Vossoughi, M.; Taghavinia, N. Immobilization of TiO2 on perlite granules for photocatalytic degradation of phenol. Appl. Catal. B Environ. 2007, 74, 53–62. [Google Scholar] [CrossRef]
- Erdem, T.; Meral, Ç.; Tokyay, M.; Erdoğan, T. Use of perlite as a pozzolanic addition in producing blended cements. Cem. Concr. Compos. 2007, 29, 13–21. [Google Scholar] [CrossRef]
- Lee, W.; Van Deventer, J. Use of infrared spectroscopy to study geopolymerization of heterogeneous amorphous aluminosilicates. Langmuir 2003, 19, 8726–8734. [Google Scholar] [CrossRef]
- Shavisi, Y.; Sharifnia, S.; Hosseini, S.; Khadivi, M. Application of TiO2/perlite photocatalysis for degradation of ammonia in wastewater. J. Ind. Eng. Chem. 2014, 20, 278–283. [Google Scholar] [CrossRef]
- Maryami, M.; Nasrollahzadeh, M.; Sajadi, S.M. Green synthesis of the Pd/perlite nanocomposite using Euphorbia neriifolia L. leaf extract and evaluation of its catalytic activity. Sep. Purif. Technol. 2017, 184, 298–307. [Google Scholar] [CrossRef]
- Xiao, Y.-Y.; Gong, X.-L.; Kang, Y.; Jiang, Z.-C.; Zhang, S.; Li, B.-J. Light-, pH-and thermal-responsive hydrogels with the triple-shape memory effect. Chem. Commun. 2016, 52, 10609–10612. [Google Scholar] [CrossRef]
- Li, J.; Zhou, M.; Ye, Z.; Wang, H.; Ma, C.; Huo, P.; Yan, Y. Enhanced photocatalytic activity of g-C3N4-ZnO/HNTs composite heterostructure photocatalysts for degradation of tetracycline under visible light irradiation. RSC Adv. 2015, 5, 91177–91189. [Google Scholar] [CrossRef]
- Roy, D.; Brooks, W.L.; Sumerlin, B.S. New directions in thermoresponsive polymers. Chem. Soc. Rev. 2013, 42, 7214–7243. [Google Scholar] [CrossRef]
- Jiang, H.; Kelch, S.; Lendlein, A. Polymers move in response to light. Adv. Mater. 2006, 18, 1471–1475. [Google Scholar] [CrossRef]
- Ahn, Y.; Jang, Y.; Selvapalam, N.; Yun, G.; Kim, K. Supramolecular velcro for reversible underwater adhesion. Angew. Chem. Int. Ed. 2013, 52, 3140–3144. [Google Scholar] [CrossRef]
- Guo, J.; Yuan, C.; Guo, M.; Wang, L.; Yan, F. Flexible and voltage-switchable polymer velcro constructed using host–guest recognition between poly (ionic liquid) strips. Chem. Sci. 2014, 5, 3261–3266. [Google Scholar] [CrossRef]
- Chen, F.; Ren, Y.; Guo, J.; Yan, F. Thermo-and electro-dual responsive poly (ionic liquid) electrolyte based smart windows. Chem. Commun. 2017, 53, 1595–1598. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.C.; Lodge, T.P.; Frisbie, C.D. Solution processable, electrochromic ion gels for sub-1 V, flexible displays on plastic. Chem. Mater. 2015, 27, 1420–1425. [Google Scholar] [CrossRef]
- Beaujuge, P.M.; Reynolds, J.R. Color control in π-conjugated organic polymers for use in electrochromic devices. Chem. Rev. 2010, 110, 268–320. [Google Scholar] [CrossRef] [PubMed]
- Massaro, M.; Schembri, V.; Campisciano, V.; Cavallaro, G.; Lazzara, G.; Milioto, S.; Noto, R.; Parisi, F.; Riela, S. Design of PNIPAAM covalently grafted on halloysite nanotubes as a support for metal-based catalysts. RSC Adv. 2016, 6, 55312–55318. [Google Scholar] [CrossRef]
- Shibayama, M.; Suetoh, Y.; Nomura, S. Structure relaxation of hydrophobically aggregated poly (N-isopropylacrylamide) in water. Macromolecules 1996, 29, 6966–6968. [Google Scholar] [CrossRef]
- Gao, C.; Möhwald, H.; Shen, J. Thermosensitive poly (allylamine)-g-poly (N-isopropylacrylamide): Synthesis, phase separation and particle formation. Polymer 2005, 46, 4088–4097. [Google Scholar] [CrossRef]
- Latypova, A.R.; Lebedev, M.D.; Tarasyuk, I.A.; Sidorov, A.I.; Rumyantsev, E.V.; Vashurin, A.S.; Marfin, Y.S. Sol-Gel Synthesis of Organically Modified Silica Particles as Efficient Palladium Catalyst Supports to Perform Hydrogenation Process. Catalysts 2021, 11, 1175. [Google Scholar] [CrossRef]
- Khajonvittayakul, C.; Tongnan, V.; Amornraksa, S.; Laosiripojana, N.; Hartley, M.; Hartley, U.W. CO2 Hydrogenation to Synthetic Natural Gas over Ni, Fe and Co–Based CeO2–Cr2O3. Catalysts 2021, 11, 1159. [Google Scholar] [CrossRef]
- Metin, Ö.; Can, H.; Şendil, K.; Gültekin, M.S. Monodisperse Ag/Pd core/shell nanoparticles assembled on reduced graphene oxide as highly efficient catalysts for the transfer hydrogenation of nitroarenes. J. Colloid Interface Sci. 2017, 498, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Parida, K.; Varadwaj, G.B.B.; Sahu, S.; Sahoo, P.C. Schiff base Pt (II) complex intercalated montmorillonite: A robust catalyst for hydrogenation of aromatic nitro compounds at room temperature. Ind. Eng. Chem. Res. 2011, 50, 7849–7856. [Google Scholar] [CrossRef]
- Song, J.; Huang, Z.-F.; Pan, L.; Li, K.; Zhang, X.; Wang, L.; Zou, J.-J. Review on selective hydrogenation of nitroarene by catalytic, photocatalytic and electrocatalytic reactions. Appl. Catal. B 2018, 227, 386–408. [Google Scholar] [CrossRef]
- Dehghani, S.; Sadjadi, S.; Bahri-Laleh, N.; Nekoomanesh-Haghighi, M.; Poater, A. Study of the effect of the ligand structure on the catalytic activity of Pd@ ligand decorated halloysite: Combination of experimental and computational studies. Appl. Organomet. Chem. 2019, 33, e4891. [Google Scholar] [CrossRef]
- Sadjadi, S.; Lazzara, G.; Heravi, M.M.; Cavallaro, G. Pd supported on magnetic carbon coated halloysite as hydrogenation catalyst: Study of the contribution of carbon layer and magnetization to the catalytic activity. Appl. Clay Sci. 2019, 182, 105299. [Google Scholar] [CrossRef] [Green Version]
- Sadjadi, S. Halloysite-based hybrids/composites in catalysis. Appl. Clay Sci. 2020, 189, 105537. [Google Scholar] [CrossRef]
- Leonhardt, S.E.S.; Stolle, A.; Ondruschka, B.; Cravotto, G.; Leo, C.D.; Jandt, K.D.; Keller, T.F. Chitosan as a support for heterogeneous Pd catalysts in liquid phase catalysis. Appl. Catal. A Gen. 2010, 379, 30–37. [Google Scholar] [CrossRef]
- Tabrizi, M.; Sadjadi, S.; Pareras, G.; Nekoomanesh-Haghighi, M.; Bahri-Laleh, N.; Poater, A. Efficient hydro-finishing of polyalfaolefin based lubricants under mild reaction condition using Pd on ligands decorated halloysite. J. Colloid Interface Sci. 2021, 581, 939–953. [Google Scholar] [CrossRef]
- Karimi, S.; Bahri-Laleh, N.; Pareras, G.; Sadjadi, S.; Nekoomanesh-Haghighi, M.; Poater, A. Pd on nitrogen rich polymer–halloysite nanocomposite as an environmentally benign and sustainable catalyst for hydrogenation of polyalfaolefin based lubricants. J. Ind. Eng. Chem. 2021, 97, 441–451. [Google Scholar] [CrossRef]
- Saufi, H.; El Alouani, M.; Alehyen, S.; El Achouri, M.; Aride, J. Photocatalytic degradation of methylene blue from aqueous medium onto perlite-based geopolymer. Int. J. Chem. Eng. 2020, 2020, 9498349. [Google Scholar] [CrossRef] [Green Version]
- Zujovic, Z.; Wheelwright, W.V.K.; Kilmartin, P.A.; Hanna, J.V.; Cooney, R.P. Structural investigations of perlite and expanded perlite using 1H, 27Al and 29Si solid-state NMR. Ceram. Int. 2018, 44, 2952–2958. [Google Scholar] [CrossRef]
- Sadjadi, S.; Heravi, M.M.; Malmir, M.; Masoumi, B. HPA decorated Halloysite Nanoclay: An efficient catalyst for the green synthesis of Spirooxindole derivatives. Appl. Organomet. Chem. 2018, 32, e4113. [Google Scholar] [CrossRef]
- Park, C.-Y.; Lee, Y.-G.; Oh, C.; Oh, S.-G. Preparation of poly-(NIPAM) grafted hybrid silica particles with hollow structure in emulsion. J. Ind. Eng. Chem. 2010, 16, 32–38. [Google Scholar] [CrossRef]
- Sarkar, D.; El Khoury, J.M.; Lopina, S.T.; Hu, J. Grafting poly (t-butyl acrylate) to poly (allylamine) by inverse microemulsion radical polymerization: From comb-polymer to amphiphilic shell crosslinked polymer nanocapsule. J. Appl. Polym. Sci. 2007, 104, 1905–1911. [Google Scholar] [CrossRef]
- Sadjadi, S.; Atai, M. Palladated halloysite hybridized with photo-polymerized hydrogel in the presence of cyclodextrin: An efficient catalytic system benefiting from nanoreactor concept. Appl. Organomet. Chem. 2019, 33, e4776. [Google Scholar] [CrossRef]
- Agrahari, S.; Lande, S.; Balachandran, V.; Kalpana, G.; Jasra, R. Palladium Supported on Mesoporous Alumina Catalyst for Selective Hydrogenation. J. Nanosci. Curr. Sci. 2017, 2, 2572. [Google Scholar] [CrossRef] [Green Version]
- Sadjadi, S.; Koohestani, F.; Bahri-Laleh, N. Pd immobilization on the multi-amine functionalized halloysite as an efficient catalyst for hydrogenation reaction: An experimental and computational study. Appl. Clay Sci. 2020, 192, 105645. [Google Scholar] [CrossRef]
- Samsonu, D.; Brahmayya, M.; Govindh, B.; Murthy, Y. Green synthesis & catalytic study of sucrose stabilized Pd nanoparticles in reduction of nitro compounds to useful amines. S. Afr. J. Chem. Eng. 2018, 25, 110–115. [Google Scholar]
- Esmaeilzadeh, M.; Sadjadi, S.; Salehi, Z. Pd immobilized on hybrid of magnetic graphene quantum dots and cyclodextrin decorated chitosan: An efficient hydrogenation catalyst. Int. J. Biol. Macromol. 2020, 150, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Li, H.; Ren, Y.; Ren, X.; Yang, Q.; Li, C. Improving catalytic hydrogenation performance of Pd nanoparticles by electronic modulation using phosphine ligands. ACS Catal. 2018, 8, 6476–6485. [Google Scholar] [CrossRef]
- Sadjadi, S.; Akbari, M.; Leger, B.; Monflier, E.; Heravi, M. Eggplant-derived biochar-halloysite nanocomposite as supports of Pd nanoparticles for the catalytic hydrogenation of nitroarenes in presence of cyclodextrin. ACS Sustain. Chem. Eng. 2019, 7, 6720–6731. [Google Scholar] [CrossRef]
- Sadjadi, S.; Ghoreyshi Kahangi, F.; Heravi, M.M. Pd stabilized on nanocomposite of halloysite and β-cyclodextrin derived carbon: An efficient catalyst for hydrogenation of nitroarene. Polyhedron 2020, 175, 114210. [Google Scholar] [CrossRef]


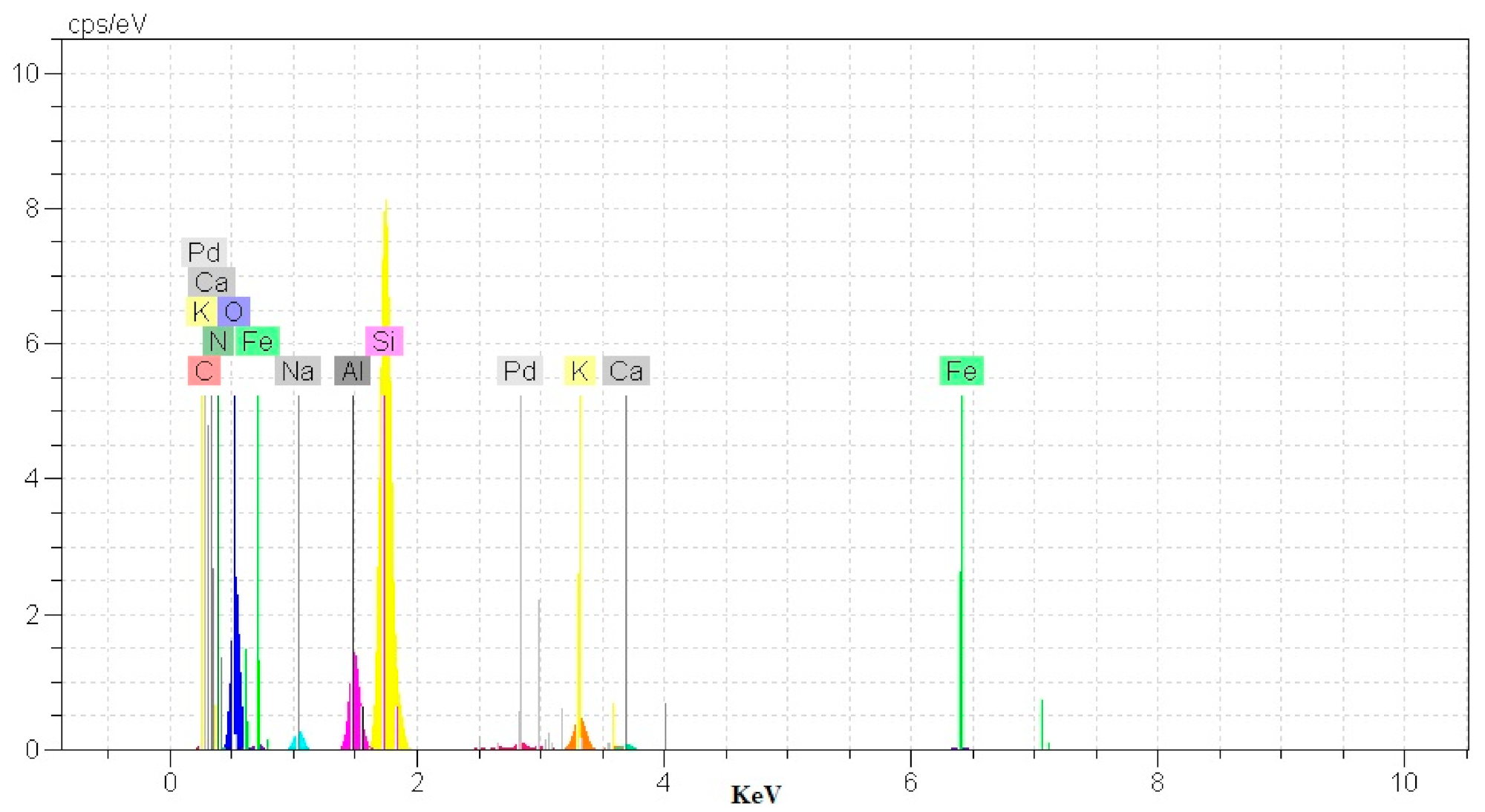


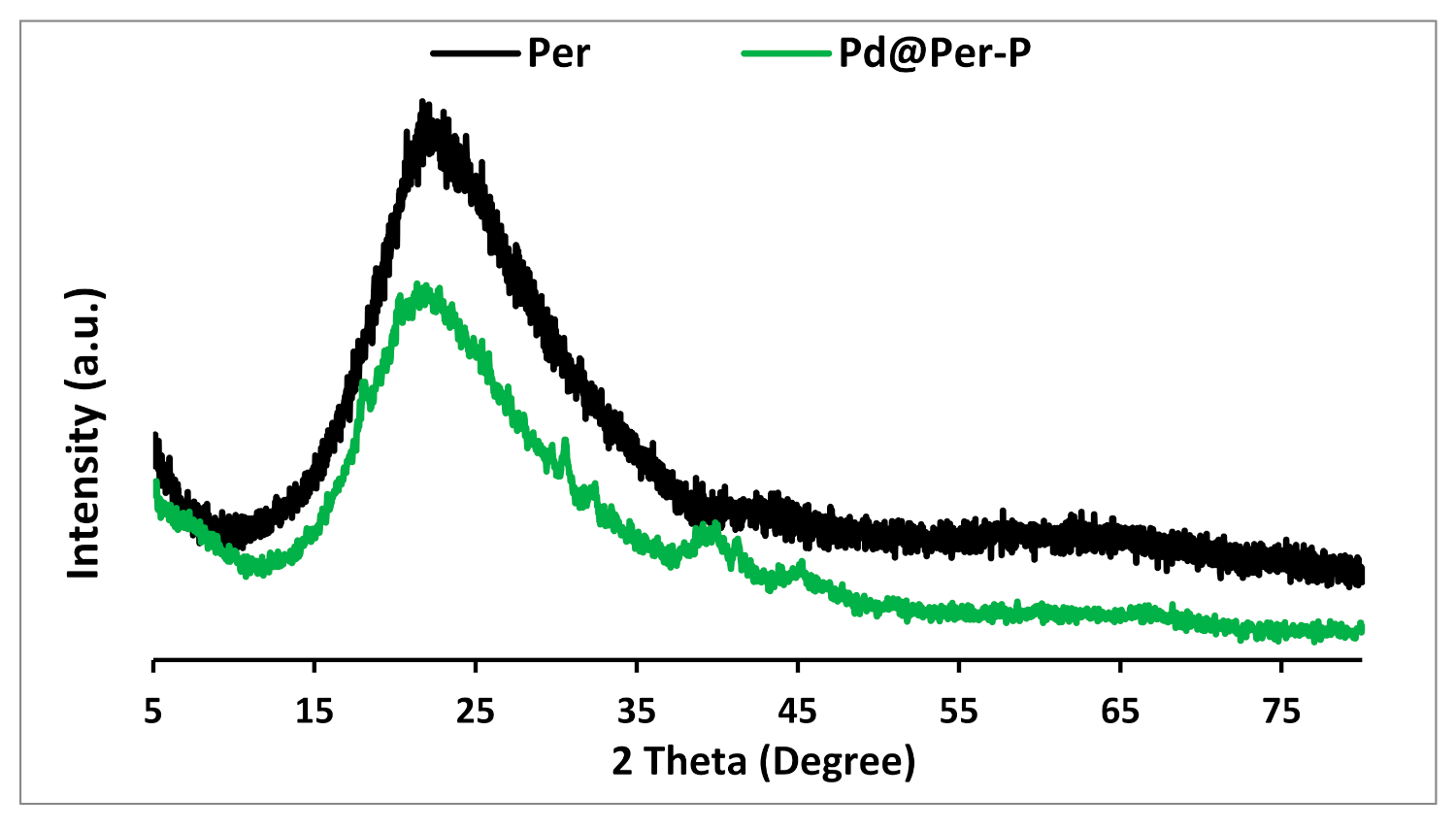
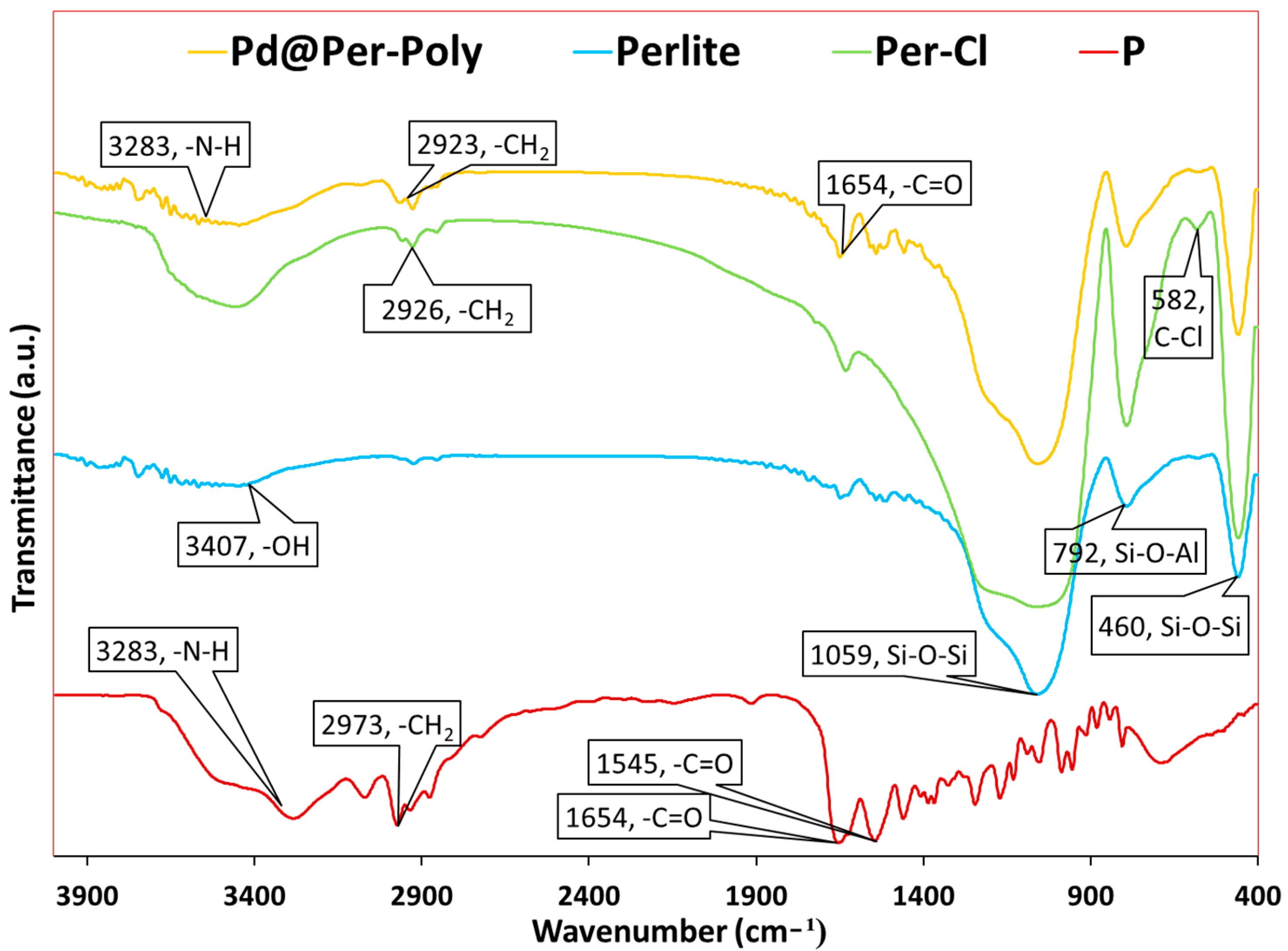
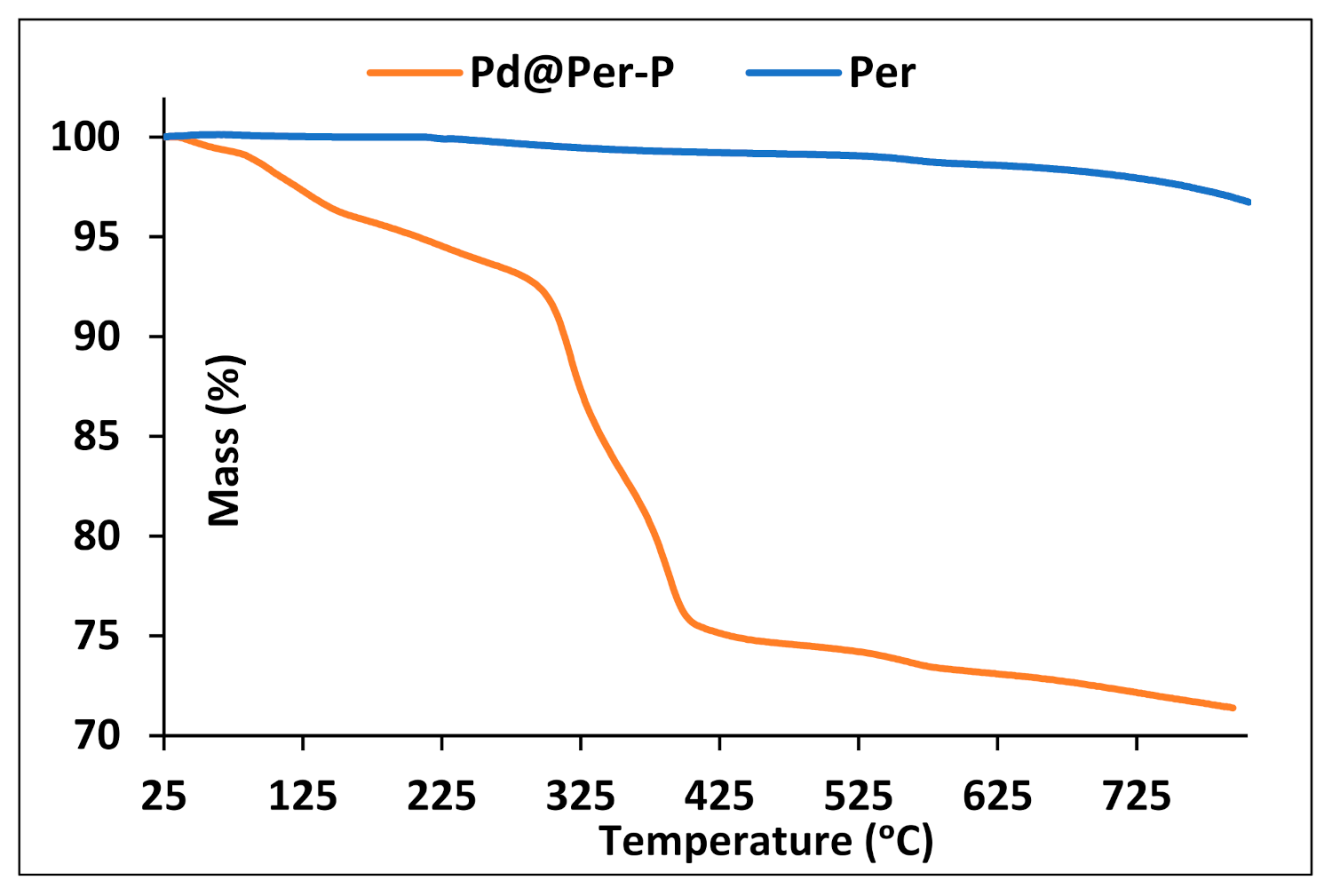




| Entry | Pd@Per-P (mg) | Solvent | Temp. (°C) | Yield (%) a |
|---|---|---|---|---|
| 1 | 20 | H2O | 25 | 60 |
| 2 | 20 | EtOH | 25 | 68 |
| 3 | 20 | CH3CN | 25 | 64 |
| 4 | 20 | THF | 25 | 56 |
| 5 | 20 | H2O:EtOH (1:1) | 25 | 70 |
| 6 | 20 | H2O:EtOH (1:1) | 45 | 85 |
| 7 | 20 | H2O:EtOH (1:1) | 50 | 85 |
| 8 | 30 | H2O:EtOH (1:1) | 45 | 98 |
| Entry | Substrate | Product | Time (min) | Yield (%) |
|---|---|---|---|---|
| 1 | 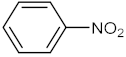 | 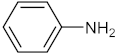 | 90 | 98 |
| 2 | 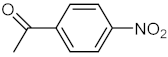 | 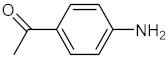 | 120 | 95 |
| 3 | 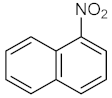 | 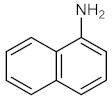 | 150 | 98 |
| 4 | 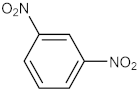 | 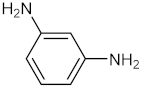 | 120 | 95 |
| 5 | 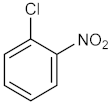 | 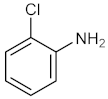 | 150 | 70 |
| 6 | 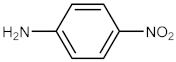 |  | 150 | 95 |
| 7 | 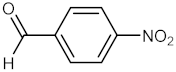 | 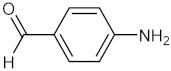 | 110 | 100 |
| 8 | 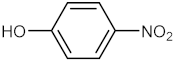 | 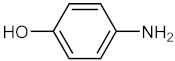 | 135 | 90 |
| Entry | Catalyst | Solvent | Time (min) | Temp. (°C) | H2 Pressure | Yield (%) | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | Pd@Per-P (0.03 g) | H2O: EtOH (1:1) | 90 | 45 | 1 atm | 98 | This work |
| 2 | Pd@Hal-Hydrogel+cyclodextrin (2 wt.%) | H2O | 120 | 50 | 1 bar | 95 | [43] |
| 3 | PdNP (0.5%)/Al2O3 (0.3 g) | THF | 180 | r.t. | 1 atm | 100 | [44] |
| 4 | Pd@Hal-TCT-Met a | H2O | 75 | 65 | 1 bar | 100 | [45] |
| 5 | APSNP b (1 mol%) | EtOH | 120 | r.t. | 40 atm | 100 | [46] |
| 6 | Pd@CS-CD-MGQDs c (0.5 mol%) | H2O | 60 | 50 | 1 atm | 97 | [47] |
| 7 | Pd/PPh3@FDU-12 (8.33 × 10−4 mmol Pd) | EtOH | 60 | 40 | 10 bar | 99 | [48] |
| 8 | Pd@Hal-biochar d (0.03 mol%) | H2O | 60 | r.t. | 1 bar | 75 | [49] |
| 9 | Pd@Hal/di-urea e (1.5 wt%) | H2O | 60 | 50 | 1 atm | 100 | [32] |
| 10 | Pd@Hal-CCD f (1 wt%) | H2O | 90 | r.t. | 1 atm | 100 | [50] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abedian-Dehaghani, N.; Heravi, M.M.; Sadjadi, S. Pd on the Composite of Perlite and Allylamine-N-isopropylacrylamide Copolymer: A Thermo-Responsive Catalyst for Hydrogenation of Nitroarenes under Mild Reaction Condition. Catalysts 2021, 11, 1334. https://doi.org/10.3390/catal11111334
Abedian-Dehaghani N, Heravi MM, Sadjadi S. Pd on the Composite of Perlite and Allylamine-N-isopropylacrylamide Copolymer: A Thermo-Responsive Catalyst for Hydrogenation of Nitroarenes under Mild Reaction Condition. Catalysts. 2021; 11(11):1334. https://doi.org/10.3390/catal11111334
Chicago/Turabian StyleAbedian-Dehaghani, Neda, Majid M. Heravi, and Samahe Sadjadi. 2021. "Pd on the Composite of Perlite and Allylamine-N-isopropylacrylamide Copolymer: A Thermo-Responsive Catalyst for Hydrogenation of Nitroarenes under Mild Reaction Condition" Catalysts 11, no. 11: 1334. https://doi.org/10.3390/catal11111334
APA StyleAbedian-Dehaghani, N., Heravi, M. M., & Sadjadi, S. (2021). Pd on the Composite of Perlite and Allylamine-N-isopropylacrylamide Copolymer: A Thermo-Responsive Catalyst for Hydrogenation of Nitroarenes under Mild Reaction Condition. Catalysts, 11(11), 1334. https://doi.org/10.3390/catal11111334






