Photocatalytic Activity of n-Alkylamine and n-Alkoxy Derivatives of Layered Perovskite-like Titanates H2Ln2Ti3O10 (Ln = La, Nd) in the Reaction of Hydrogen Production from an Aqueous Solution of Methanol
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification of the Protonated Titanates
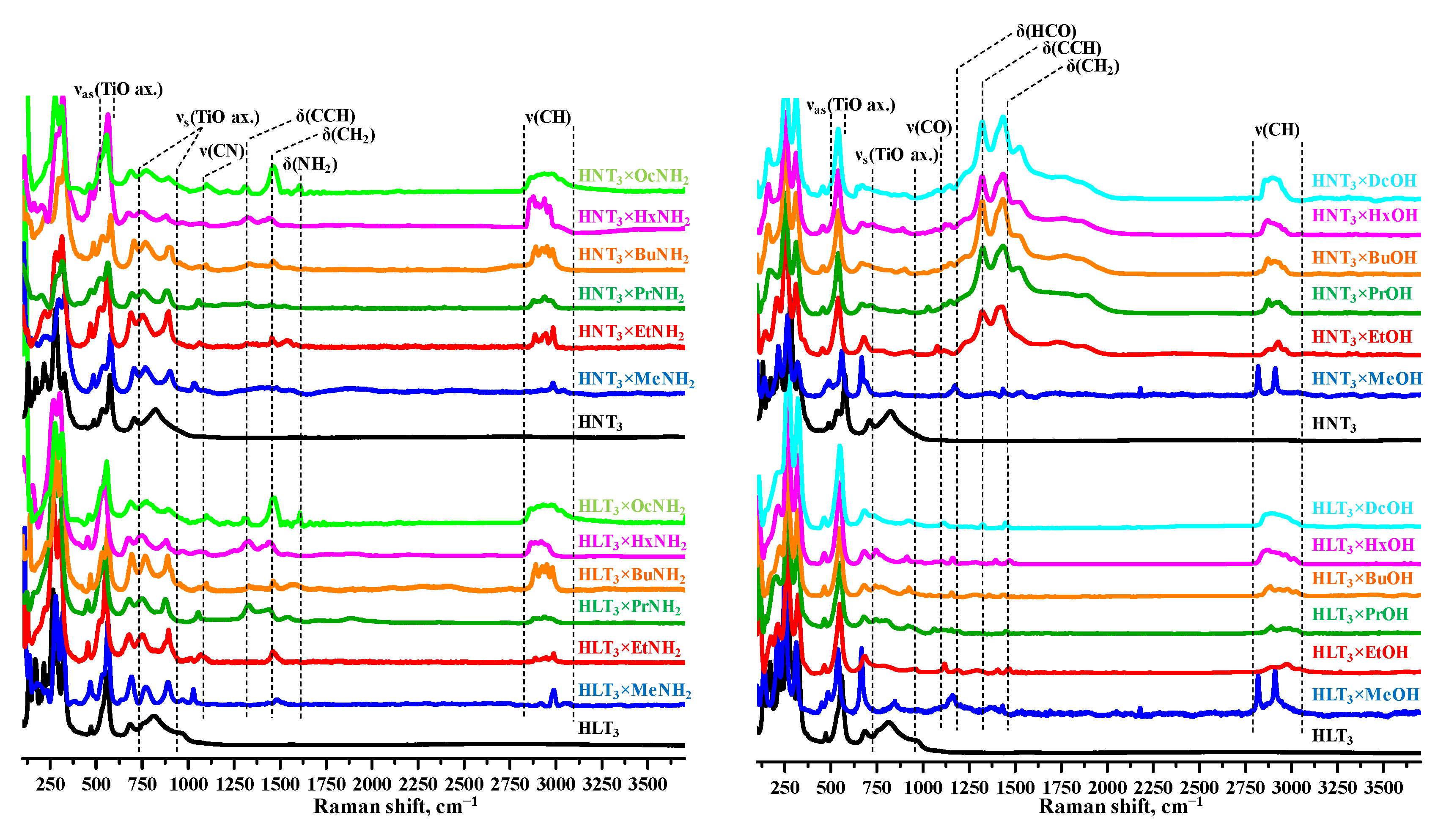
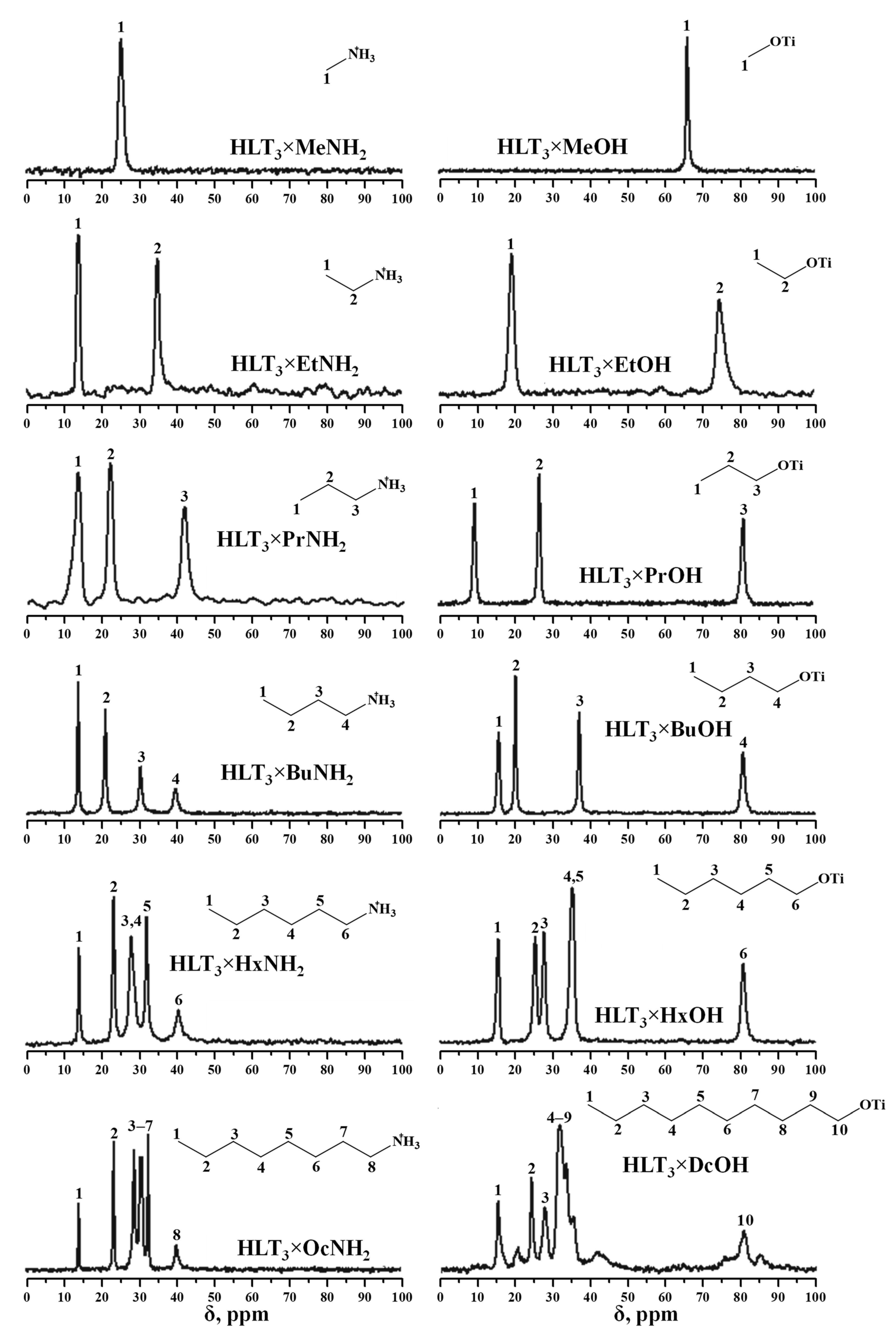
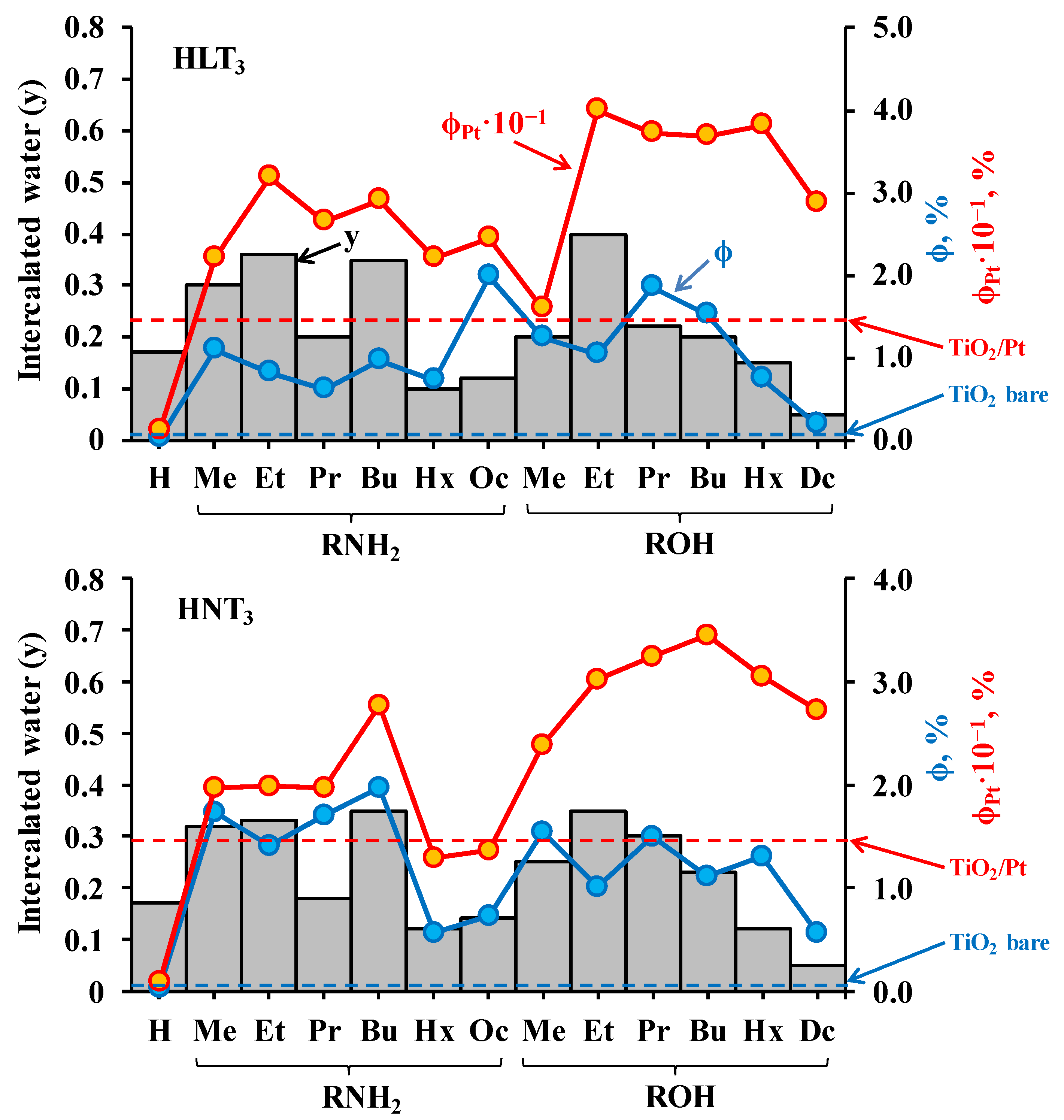
2.2. Analysis of the Inorganic-Organic Derivatives
2.3. Thermal, Vacuum and Hydrolytic Stability of the Inorganic-Organic Derivatives
2.4. Photocatalytic Activity of the Hybrid Inorganic-Organic Titanates
2.5. Analysis of the Hybrid Inorganic-Organic Titanates after the Photocatalytic Experiment
2.6. Possible Explanations of Photocatalytic Properties of the Hybrid Inorganic-Organic Titanates
3. Materials and Methods
3.1. Synthesis of Initial Protonated Titanates
3.2. Synthesis of Inorganic-Organic Derivatives
3.3. Investigation of Vacuum and Hydrolytic Stability
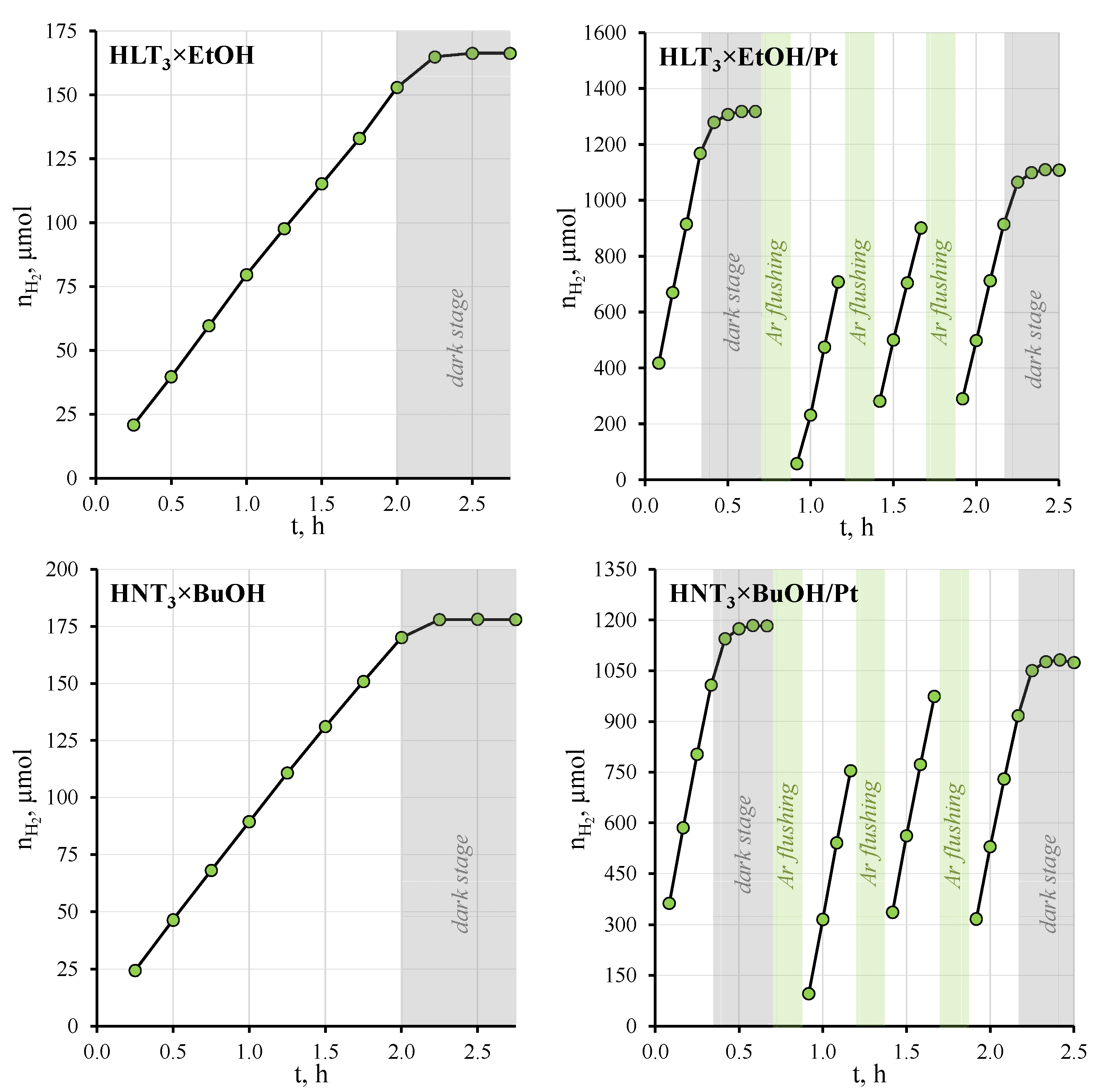
3.4. Investigation of Photocatalytic Activity
3.5. Instrumentation
3.5.1. XRD
3.5.2. Raman Spectroscopy
3.5.3. NMR Spectroscopy
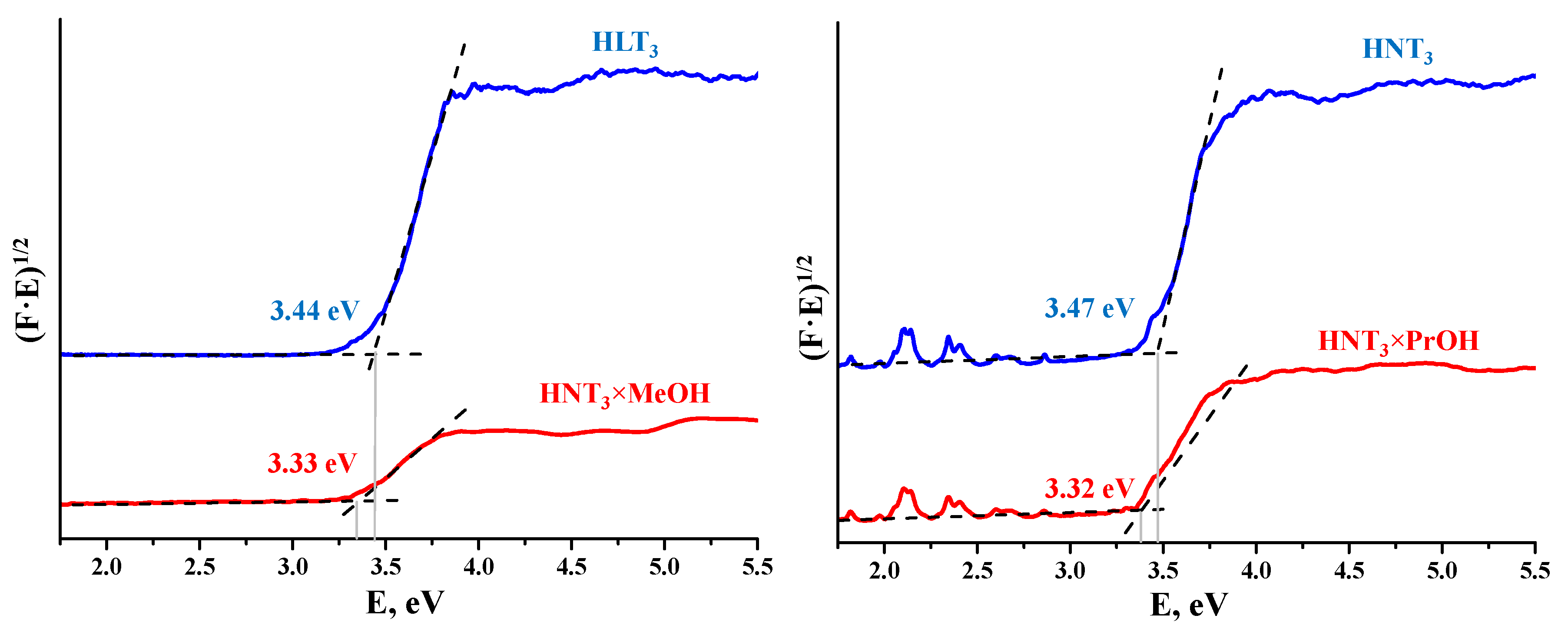
3.5.4. DRS
3.5.5. CHN-Analysis
3.5.6. TG
3.5.7. SEM
3.5.8. BET
3.5.9. ICP-AES
3.5.10. Spectrophotometry
3.5.11. pH Measurement
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moiseev, I.I. Green chemistry: Development trajectory. Russ. Chem. Rev. 2013, 82, 616–623. [Google Scholar] [CrossRef]
- Kozlova, E.A.; Parmon, V.N. Heterogeneous semiconductor photocatalysts for hydrogen production from aqueous solutions of electron donors. Russ. Chem. Rev. 2017, 86, 870–906. [Google Scholar] [CrossRef]
- Rozhkova, E.; Ariga, K. (Eds.) From Molecules to Materials Pathways to Artificial Photosynthesis; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Ismail, A.A.; Bahnemann, D.W. Photochemical splitting of water for hydrogen production by photocatalysis: A review. Sol. Energy Mater. Sol. Cells 2014, 128, 85–101. [Google Scholar] [CrossRef]
- Ahmad, H.; Kamarudin, S.K.; Minggu, L.J.; Kassim, M. Hydrogen from photo-catalytic water splitting process: A review. Renew. Sustain. Energy Rev. 2015, 43, 599–610. [Google Scholar] [CrossRef]
- Takanabe, K. Photocatalytic Water Splitting: Quantitative Approaches toward Photocatalyst by Design. ACS Catal. 2017, 7, 8006–8022. [Google Scholar] [CrossRef]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar water splitting cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- Christoforidis, K.C.; Fornasiero, P. Photocatalytic Hydrogen Production: A Rift into the Future Energy Supply. ChemCatChem 2017, 9, 1523–1544. [Google Scholar] [CrossRef] [Green Version]
- Puga, A.V. Photocatalytic production of hydrogen from biomass-derived feedstocks. Coord. Chem. Rev. 2016, 315, 1–66. [Google Scholar] [CrossRef]
- Liu, R.; Yoshida, H.; Fujita, S.-i.; Arai, M. Photocatalytic hydrogen production from glycerol and water with NiOx/TiO2 catalysts. Appl. Catal. B Environ. 2014, 144, 41–45. [Google Scholar] [CrossRef]
- Taboada, E.; Angurell, I.; Llorca, J. Hydrogen photoproduction from bio-derived alcohols in an optical fiber honeycomb reactor loaded with Au/TiO2. J. Photochem. Photobiol. A Chem. 2014, 281, 35–39. [Google Scholar] [CrossRef]
- Al-Azri, Z.H.N.; Chen, W.T.; Chan, A.; Jovic, V.; Ina, T.; Idriss, H.; Waterhouse, G.I.N. The roles of metal co-catalysts and reaction media in photocatalytic hydrogen production: Performance evaluation of M/TiO2 photocatalysts (M = Pd, Pt, Au) in different alcohol-water mixtures. J. Catal. 2015, 329, 355–367. [Google Scholar] [CrossRef]
- Dosado, A.G.; Chen, W.T.; Chan, A.; Sun-Waterhouse, D.; Waterhouse, G.I.N. Novel Au/TiO2 photocatalysts for hydrogen production in alcohol-water mixtures based on hydrogen titanate nanotube precursors. J. Catal. 2015, 330, 238–254. [Google Scholar] [CrossRef]
- Wang, X.; Dong, H.; Hu, Z.; Qi, Z.; Li, L. Fabrication of a Cu2O/Au/TiO2 composite film for efficient photocatalytic hydrogen production from aqueous solution of methanol and glucose. Mater. Sci. Eng. B Solid State Mater. Adv. Technol. 2017, 219, 10–19. [Google Scholar] [CrossRef]
- Li, C.; Wang, H.; Ming, J.; Liu, M.; Fang, P. Hydrogen generation by photocatalytic reforming of glucose with heterostructured CdS/MoS2 composites under visible light irradiation. Int. J. Hydrogen Energy 2017, 42, 16968–16978. [Google Scholar] [CrossRef]
- Jaswal, R.; Shende, R.; Nan, W.; Shende, A. Photocatalytic reforming of pinewood (Pinus ponderosa) acid hydrolysate for hydrogen generation. Int. J. Hydrogen Energy 2017, 42, 2839–2848. [Google Scholar] [CrossRef]
- Udom, I.; Myers, P.D.; Ram, M.K.; Hepp, A.F.; Archibong, E.; Stefanakos, E.K.; Goswami, D.Y. Optimization of Photocatalytic Degradation of Phenol Using Simple Photocatalytic Reactor. Am. J. Anal. Chem. 2014, 5, 743–750. [Google Scholar] [CrossRef] [Green Version]
- Long, Z.; Li, Q.; Wei, T.; Zhang, G.; Ren, Z. Historical development and prospects of photocatalysts for pollutant removal in water. J. Hazard. Mater. 2020, 395, 122599. [Google Scholar] [CrossRef] [PubMed]
- Rueda-Marquez, J.J.; Levchuk, I.; Fernández Ibañez, P.; Sillanpää, M. A critical review on application of photocatalysis for toxicity reduction of real wastewaters. J. Clean. Prod. 2020, 258, 120694. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, X.; Meng, Y.; Pan, G.; Ni, Z.; Xia, S. Layered double hydroxides-based photocatalysts and visible-light driven photodegradation of organic pollutants: A review. Chem. Eng. J. 2020, 392, 123684. [Google Scholar] [CrossRef]
- Karthikeyan, C.; Arunachalam, P.; Ramachandran, K.; Al-Mayouf, A.M.; Karuppuchamy, S. Recent advances in semiconductor metal oxides with enhanced methods for solar photocatalytic applications. J. Alloys Compd. 2020, 828, 154281. [Google Scholar] [CrossRef]
- Schaak, R.E.; Mallouk, T.E. Perovskites by Design: A Toolbox of Solid-State Reactions. Chem. Mater. 2002, 14, 1455–1471. [Google Scholar] [CrossRef]
- Uppuluri, R.; Sen Gupta, A.; Rosas, A.S.; Mallouk, T.E. Soft chemistry of ion-exchangeable layered metal oxides. Chem. Soc. Rev. 2018, 47, 2401–2430. [Google Scholar] [CrossRef]
- Tani, S.; Komori, Y.; Hayashi, S.; Sugahara, Y. Local environments and dynamics of hydrogen atoms in protonated forms of ion-exchangeable layered perovskites estimated by solid-state 1H NMR. J. Solid State Chem. 2006, 179, 3357–3364. [Google Scholar] [CrossRef]
- Silyukov, O.; Chislov, M.; Burovikhina, A.; Utkina, T.; Zvereva, I. Thermogravimetry study of ion exchange and hydration in layered oxide materials. J. Therm. Anal. Calorim. 2012, 110, 187–192. [Google Scholar] [CrossRef]
- Rodionov, I.A.; Zvereva, I.A. Photocatalytic activity of layered perovskite-like oxides in practically valuable chemical reactions. Russ. Chem. Rev. 2016, 85, 248–279. [Google Scholar] [CrossRef]
- Silyukov, O.I.; Kurnosenko, S.A.; Zvereva, I.A. Intercalation of Methylamine into the Protonated Forms of Layered Perovskite-Like Oxides HLnTiO4 (Ln = La and Nd). Glass Phys. Chem. 2018, 44, 428–432. [Google Scholar] [CrossRef]
- Kurnosenko, S.A.; Silyukov, O.I.; Mazur, A.S.; Zvereva, I.A. Synthesis and thermal stability of new inorganic-organic perovskite-like hybrids based on layered titanates HLnTiO4 (Ln = La, Nd). Ceram. Int. 2019, 46, 5058–5068. [Google Scholar] [CrossRef]
- Shelyapina, M.G.; Lushpinskaya, I.P.; Kurnosenko, S.A.; Silyukov, O.I.; Zvereva, I.A. Identification of Intercalates and Grafted Organic Derivatives of H2La2Ti3O10 by Multinuclear NMR. Russ. J. Gen. Chem. 2020, 90, 760–761. [Google Scholar] [CrossRef]
- Kurnosenko, S.A.; Silyukov, O.I.; Minich, I.A.; Zvereva, I.A. Exfoliation of methylamine and n-butylamine derivatives of layered perovskite-like oxides HLnTiO4 and H2Ln2Ti3O10 (Ln = La, Nd) into nanolayers. Glass Phys. Chem. 2021, 47, 372–381. [Google Scholar] [CrossRef]
- Silyukov, O.I.; Minich, I.A.; Zvereva, I.A. Synthesis of Protonated Derivatives of Layered Perovskite-Like Bismuth Titanates. Glass Phys. Chem. 2018, 44, 115–119. [Google Scholar] [CrossRef]
- Minich, I.A.; Silyukov, O.I.; Gak, V.V.; Borisov, E.V.; Zvereva, I.A. Synthesis of Organic–Inorganic Hybrids Based on Perovskite-like Bismuth Titanate H2K0.5Bi2.5Ti4O13·H2O and n-Alkylamines. ACS Omega 2020, 5, 8158–8168. [Google Scholar] [CrossRef]
- Shelyapina, M.G.; Silyukov, O.I.; Lushpinskaia, I.P.; Kurnosenko, S.A.; Mazur, A.S.; Shenderovich, I.G.; Zvereva, I.A. NMR study of intercalates and grafted organic derivatives of H2La2Ti3O10. Molecules 2020, 25, 5229. [Google Scholar] [CrossRef] [PubMed]
- Silyukov, O.I.; Kurnosenko, S.A.; Minich, I.A.; Rodionov, I.A.; Zvereva, I.A. Protonated Forms of Layered Perovskite-Like Titanate NaNdTiO4: Neutron and X-ray Diffraction Structural Analysis. Solids 2021, 2, 265–277. [Google Scholar] [CrossRef]
- Rodionov, I.A.; Silyukov, O.I.; Utkina, T.D.; Chislov, M.V.; Sokolova, Y.P.; Zvereva, I.A. Photocatalytic properties and hydration of perovskite-type layered titanates A2Ln2Ti3O10 (A = Li, Na, K.; Ln = La, Nd). Russ. J. Gen. Chem. 2012, 82, 1191–1196. [Google Scholar] [CrossRef]
- Takata, T.; Furumi, Y.; Shinohara, K.; Tanaka, A.; Hara, M.; Kondo, J.N.; Domen, K. Photocatalytic Decomposition of Water on Spontaneously Hydrated Layered Perovskites. Chem. Mater. 1997, 9, 1063–1064. [Google Scholar] [CrossRef]
- Zou, Z.; Ye, J.; Arakawa, H. Substitution effects of In3+ by Fe3+ on photocatalytic and structural properties of Bi2InNbO7 photocatalysts. J. Mol. Catal. 2001, 168, 289–297. [Google Scholar] [CrossRef]
- Reddy, V.; Hwang, D.; Lee, J. Effect of Zr substitution for Ti in KLaTiO4 for photocatalytic water splitting. Catal. Lett. 2003, 90, 39–44. [Google Scholar] [CrossRef]
- Kumar, V.; Uma, S. Investigation of cation (Sn2+) and anion (N3−) substitution in favor of visible light photocatalytic activity in the layered perovskite K2La2Ti3O10. J. Hazard. Mater. 2011, 189, 502–508. [Google Scholar] [CrossRef]
- Zhou, Y.; Wen, T.; Guo, Y.; Yang, B.; Wang, Y. Controllable doping of nitrogen and tetravalent niobium affords yellow and black calcium niobate nanosheets for enhanced photocatalytic hydrogen evolution. RSC Adv. 2016, 6, 64930–64936. [Google Scholar] [CrossRef]
- Kawashima, K.; Hojamberdiev, M.; Chen, S.; Yubuta, K.; Wagata, H.; Domen, K.; Teshima, K. Understanding the effect of partial N3−-to-O2− substitution and H+-to-K+ exchange on photocatalytic water reduction activity of Ruddlesden-Popper layered perovskite KLaTiO4. Mol. Catal. 2017, 432, 250–258. [Google Scholar] [CrossRef]
- Huang, Y.; Li, J.; Wei, Y.; Li, Y.; Lin, J.; Wu, J. Fabrication and photocatalytic property of Pt-intercalated layered perovskite niobates H1−xLaNb2−xMoxO7 (x = 0−0.15). J. Hazard. Mater. 2009, 166, 103–108. [Google Scholar] [CrossRef]
- Huang, Y.; Li, Y.; Wei, Y.; Huang, M.; Wu, J. Photocatalytic property of partially substituted Pt-intercalated layered perovskite, ASr2TaxNb3−xO10 (A = K, H; x = 0, 1, 1.5, 2 and 3). Sol. Energy Mater. Sol. Cells 2011, 95, 1019–1027. [Google Scholar] [CrossRef]
- Oshima, T.; Wang, Y.; Lu, D.; Yokoi, T.; Maeda, K. Photocatalytic overall water splitting on Pt nanocluster-intercalated, restacked KCa2Nb3O10 nanosheets: The promotional effect of co-existing ions. Nanoscale Adv. 2019, 1, 189–194. [Google Scholar] [CrossRef] [Green Version]
- Cui, W.; Qi, Y.; Liu, L.; Rana, D.; Hu, J.; Liang, Y. Synthesis of PbS–K2La2Ti3O10 composite and its photocatalytic activity for hydrogen production. Prog. Nat. Sci. Mater. Int. 2012, 22, 120–125. [Google Scholar] [CrossRef] [Green Version]
- Cui, W.; Liu, L.; Ma, S.; Liang, Y.; Zhang, Z. CdS-sensitized K2La2Ti3O10 composite: A new photocatalyst for hydrogen evolution under visible light irradiation. Catal. Today 2013, 207, 44–49. [Google Scholar] [CrossRef]
- Cui, W.; Guo, D.; Liu, L.; Hu, J.; Rana, D.; Liang, Y. Preparation of ZnIn2S4/K2La2Ti3O10 composites and their photocatalytic H2 evolution from aqueous Na2S/Na2SO3 under visible light irradiation. Catal. Commun. 2014, 48, 55–59. [Google Scholar] [CrossRef]
- Saito, K.; Kozeni, M.; Sohmiya, M.; Komaguchi, K.; Ogawa, M.; Sugahara, Y.; Ide, Y. Unprecedentedly enhanced solar photocatalytic activity of a layered titanate simply integrated with TiO2 nanoparticles. Phys. Chem. Chem. Phys. 2016, 18, 30920–30925. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhou, Y.; Lv, C.; Zhang, C.; Jin, X.; Meng, Q.; Chen, G. Construction of 2D-composite HCa2Nb3O10/CaNb2O6 heterostructured photocatalysts with enhanced hydrogen production performance. New J. Chem. 2018, 42, 681–687. [Google Scholar] [CrossRef]
- Chen, X.; Shen, S.; Guo, L.; Mao, S.S. Semiconductor-based photocatalytic hydrogen generation. Chem. Rev. 2010, 110, 6503–6570. [Google Scholar] [CrossRef]
- Zhang, L.; Wong, K.H.; Chen, Z.; Yu, J.C.; Zhao, J.; Hu, C.; Chan, C.Y.; Wong, P.K. AgBr-Ag-Bi2WO6 nanojunction system: A novel and efficient photocatalyst with double visible-light active components. Appl. Catal. A Gen. 2009, 363, 221–229. [Google Scholar] [CrossRef]
- Kim, H.G.; Jeong, E.D.; Borse, P.H.; Jeon, S.; Yong, K.; Lee, J.S.; Li, W.; Oh, S.H. Photocatalytic Ohmic layered nanocomposite for efficient utilization of visible light photons. Appl. Phys. Lett. 2006, 89, 2012–2015. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.G.; Borse, P.H.; Choi, W.; Lee, J.S. Photocatalytic Nanodiodes for Visible-Light Photocatalysis. Angew. Chem. 2005, 117, 4661–4665. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, G.; Xiong, Z.; Tang, H.; Jiang, C. Fabrication of flower-like direct Z-scheme β-Bi2O3/g-C3N4 photocatalyst with enhanced visible light photoactivity for Rhodamine B degradation. Appl. Surf. Sci. 2018, 436, 162–171. [Google Scholar] [CrossRef]
- Youngblood, W.J.; Anna Lee, S.H.; Maeda, K.; Mallouk, T.E. Visible light water splitting using dye-sensitized oxide semiconductors. Acc. Chem. Res. 2009, 42, 1966–1973. [Google Scholar] [CrossRef] [Green Version]
- Maeda, K.; Mallouk, T.E. Two-Dimensional Metal Oxide Nanosheets as Building Blocks for Artificial Photosynthetic Assemblies. Bull. Chem. Soc. Jpn. 2018, 92, 38–54. [Google Scholar] [CrossRef]
- Gómez-Romero, P.; Sanchez, C. Functional Hybrid Materials; Wiley: Weinheim, Germany, 2006; ISBN 3527303596. [Google Scholar]
- Kickelbick, G. Hybrid Materials: Synthesis, Characterization, and Applications; Wiley: Weinheim, Germany, 2007; ISBN 9783527312993. [Google Scholar]
- Mir, S.H.; Nagahara, L.A.; Thundat, T.; Mokarian-Tabari, P.; Furukawa, H.; Khosla, A. Review—Organic-Inorganic Hybrid Functional Materials: An Integrated Platform for Applied Technologies. J. Electrochem. Soc. 2018, 165, B3137. [Google Scholar] [CrossRef]
- Constantino, V.R.L.; Barbosa, C.A.S.; Bizeto, M.A.; Dias, P.M. Intercalation compounds involving inorganic layered structures. An. Acad. Bras. Cienc. 2000, 72, 45–50. [Google Scholar] [CrossRef] [Green Version]
- Tahara, S.; Sugahara, Y. Interlayer Surface Modification of the Protonated Triple-Layered Perovskite HCa2Nb3O10·xH2O with n-Alcohols. Langmuir 2003, 19, 9473–9478. [Google Scholar] [CrossRef]
- Tahara, S.; Ichikawa, T.; Kajiwara, G.; Sugahara, Y. Reactivity of the Ruddlesden−Popper Phase H2La2Ti3O10 with Organic Compounds: Intercalation and Grafting Reactions. Chem. Mater. 2007, 19, 2352–2358. [Google Scholar] [CrossRef]
- Wang, C.; Tang, K.; Wang, D.; Liu, Z.; Wang, L.; Zhu, Y.; Qian, Y. A new carbon intercalated compound of Dion–Jacobson phase HLaNb2O7. J. Mater. Chem. 2012, 22, 11086. [Google Scholar] [CrossRef]
- Aznar, A.J.; Sanz, J.; Ruiz-Hitzky, E. Mechanism of the grafting of organosilanes on mineral surfaces. IV. Phenylderivatives of sepiolite and poly (organosiloxanes). Colloid Polym. Sci. 1992, 270, 165–176. [Google Scholar] [CrossRef]
- Shimada, A.; Yoneyama, Y.; Tahara, S.; Mutin, P.H.; Sugahara, Y. Interlayer surface modification of the protonated ion-exchangeable layered perovskite HLaNb2O7 xH2O with organophosphonic acids. Chem. Mater. 2009, 21, 4155–4162. [Google Scholar] [CrossRef]
- Sugaya, T.; Guégan, R.; Idota, N.; Tsukahara, T.; Sugahara, Y. Highly Efficient Surface Modification of Layered Perovskite Nanosheets with a Phosphorus Coupling Reagent Making Use of Microchannels. Langmuir 2020, 36, 7252–7258. [Google Scholar] [CrossRef] [PubMed]
- Sugaya, T.; Ozaki, M.; Guégan, R.; Idota, N.; Sugahara, Y. Surface Modification of Layered Perovskite Nanosheets with a Phosphorus Coupling Reagent in a Biphasic System. Langmuir 2019, 35, 6594–6601. [Google Scholar] [CrossRef]
- Idota, N.; Fukuda, S.; Tsukahara, T.; Sugahara, Y. Preparation of thermoresponsive nanosheets exhibiting phase transitions in water via surface modification of layered perovskite nanosheets with poly(N-isopropylacrylamide) (PNIPAAm). Chem. Lett. 2015, 44, 203–205. [Google Scholar] [CrossRef]
- Sato, S.; Shintani, K.; Idota, N.; Nishino, T.; Sugahara, Y. Effect of the graft density of cellulose diacetate-modified layered perovskite nanosheets on mechanical properties of the transparent organic–inorganic hybrids bearing covalent bonds at the interface. Cellulose 2017, 24, 5463–5473. [Google Scholar] [CrossRef] [Green Version]
- Akbarian-Tefaghi, S.; Teixeira Veiga, E.; Amand, G.; Wiley, J.B. Rapid Topochemical Modification of Layered Perovskites via Microwave Reactions. Inorg. Chem. 2016, 55, 1604–1612. [Google Scholar] [CrossRef]
- Akbarian-Tefaghi, S.; Rostamzadeh, T.; Brown, T.T.; Davis-Wheeler, C.; Wiley, J.B. Rapid Exfoliation and Surface Tailoring of Perovskite Nanosheets via Microwave-Assisted Reactions. ChemNanoMat 2017, 3, 538–550. [Google Scholar] [CrossRef]
- Akbarian-Tefaghi, S.; Wiley, J.B. Microwave-assisted routes for rapid and efficient modification of layered perovskites. Dalt. Trans. 2018, 47, 2917–2924. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Chen, X.; Hao, Y.; Wei, Z.; Zhang, T.; Cai, H. A trilayered organic-inorganic hybrid perovskite material with low bandgap. Chem. Phys. Lett. 2020, 747, 137313. [Google Scholar] [CrossRef]
- Kaiba, A.; Geesi, M.H.; Guionneau, P.; Aljohani, T.A.; Bih, L.; Bih, H.; Kassou, S. Synthesis, structural and Raman spectroscopic in organic−inorganic halide perovskites based on β-Alanine. J. Mol. Struct. 2020, 1204, 127380. [Google Scholar] [CrossRef]
- Wang, Y.; Luo, L.; Wang, Z.; Tawiah, B.; Liu, C.; Xin, J.H.; Fei, B.; Wong, W.Y. Growing Poly(norepinephrine) Layer over Individual Nanoparticles to Boost Hybrid Perovskite Photocatalysts. ACS Appl. Mater. Interfaces 2020, 12, 27578–27586. [Google Scholar] [CrossRef]
- Liu, F.; Wang, M.; Liu, X.; Wang, B.; Li, C.; Liu, C.; Lin, Z.; Huang, F. A rapid and robust light-and-solution-triggered in situ crafting of organic passivating membrane over metal halide perovskites for markedly improved stability and photocatalysis. Nano Lett. 2021, 21, 1643–1650. [Google Scholar] [CrossRef] [PubMed]
- Gélvez-Rueda, M.C.; Ahlawat, P.; Merten, L.; Jahanbakhshi, F.; Mladenović, M.; Hinderhofer, A.; Dar, M.I.; Li, Y.; Dučinskas, A.; Carlsen, B.; et al. Formamidinium-Based Dion-Jacobson Layered Hybrid Perovskites: Structural Complexity and Optoelectronic Properties. Adv. Funct. Mater. 2020, 30, 2003428. [Google Scholar] [CrossRef]
- Machida, M.; Mitsuyama, T.; Ikeue, K.; Matsushima, S.; Arai, M. Photocatalytic property and electronic structure of triple-layered perovskite tantalates, MCa2Ta3O10 (M = Cs, Na, H, and C6H13NH3). J. Phys. Chem. B 2005, 109, 7801–7806. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, C.; Wang, L.; Hao, Q.; Zhu, X.; Chen, X.; Tang, K. Preparation of interlayer surface tailored protonated double-layered perovskite H2CaTa2O7 with n-alcohols, and their photocatalytic activity. RSC Adv. 2014, 4, 4047–4054. [Google Scholar] [CrossRef]
- Rodionov, I.A.; Maksimova, E.A.; Pozhidaev, A.Y.; Kurnosenko, S.A.; Silyukov, O.I.; Zvereva, I.A. Layered Titanate H2Nd2Ti3O10 Intercalated With n-Butylamine: A New Highly Efficient Hybrid Photocatalyst for Hydrogen Production From Aqueous Solutions of Alcohols. Front. Chem. 2019, 7, 863. [Google Scholar] [CrossRef] [PubMed]
- Voytovich, V.V.; Kurnosenko, S.A.; Silyukov, O.I.; Rodionov, I.A.; Minich, I.A.; Zvereva, I.A. Study of n-alkylamine intercalated layered perovskite-like niobates HCa2Nb3O10 as photocatalysts for hydrogen production from an aqueous solution of methanol. Front. Chem. 2020, 8, 300. [Google Scholar] [CrossRef] [PubMed]
- Voytovich, V.V.; Kurnosenko, S.A.; Silyukov, O.I.; Rodionov, I.A.; Bugrov, A.N.; Minich, I.A.; Malygina, E.N.; Zvereva, I.A. Synthesis of n-alkoxy derivatives of layered perovskite-like niobate HCa2Nb3O10 and study of their photocatalytic activity for hydrogen production from an aqueous solution of methanol. Catalysts 2021, 11, 897. [Google Scholar] [CrossRef]
- Gopalakrishnan, J.; Bhat, V. A2Ln2Ti3O10 (A = potassium or rubidium; Ln = lanthanum or rare earth): A new series of layered perovskites exhibiting ion exchange. Inorg. Chem. 1987, 26, 4299–4301. [Google Scholar] [CrossRef]
- Takata, T.; Shinohara, K.; Tanaka, A.; Hara, M.; Kondo, J.N.; Domen, K. A highly active photocatalyst for overall water splitting with a hydrated layered perovskite structure. J. Photochem. Photobiol. A Chem. 1997, 106, 45–49. [Google Scholar] [CrossRef]
- Cui, W.; Liu, L.; Feng, L.; Xu, C.; Li, Z.; Lü, S.; Qiu, F. Preparation of Pt/K2La2Ti3O10 and its photo-catalytic activity for hydrogen evolution from methanol water solution. Sci. China Ser. B 2006, 49, 162–168. [Google Scholar] [CrossRef]
- Tai, Y.-W.; Chen, J.-S.; Yang, C.-C.; Wan, B.-Z. Preparation of nano-gold on K2La2Ti3O10 for producing hydrogen from photo-catalytic water splitting. Catal. Today 2004, 97, 95–101. [Google Scholar] [CrossRef]
- Thaminimulla, C. Effect of Chromium Addition for Photocatalytic Overall Water Splitting on Ni–K2La2Ti3O10. J. Catal. 2000, 196, 362–365. [Google Scholar] [CrossRef]
- Ya-Hui, Y.; Qi-Yuan, C.; Zhou-Lan, Y.; Jie, L. Study on the photocatalytic activity of K2La2Ti3O10 doped with zinc(Zn). Appl. Surf. Sci. 2009, 255, 8419–8424. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, J.; Wei, Y.; Hao, S.; Huang, M.; Lin, J. Synthesis and photocatalytic activity of hydrated layered perovskite K2−xLa2Ti3−xNbxO10 (0 ≤ x ≤ 1) and protonated derivatives. Scr. Mater. 2007, 57, 437–440. [Google Scholar] [CrossRef]
- Wang, B.; Li, C.; Hirabayashi, D.; Suzuki, K. Hydrogen evolution by photocatalytic decomposition of water under ultraviolet–visible irradiation over K2La2Ti3−xMxO10+δ perovskite. Int. J. Hydrogen Energy 2010, 35, 3306–3312. [Google Scholar] [CrossRef]
- Kawashima, K.; Hojamberdiev, M.; Wagata, H.; Yubuta, K.; Domen, K.; Teshima, K. Protonated Oxide, Nitrided, and Reoxidized K2La2Ti3O10 Crystals: Visible-Light-Induced Photocatalytic Water Oxidation and Fabrication of Their Nanosheets. ACS Sustain. Chem. Eng. 2017, 5, 232–240. [Google Scholar] [CrossRef]
- Wu, J.; Huang, Y.; Li, T.; Lin, J.; Huang, M.; Wei, Y. Synthesis and photocatalytic properties of layered nanocomposite H2La2Ti3O10/Fe2O3. Scr. Mater. 2006, 54, 1357–1362. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, J.; Li, T.; Hao, S.; Lin, J. Synthesis and photocatalytic properties of H2La2Ti3O10/TiO2 intercalated nanomaterial. J. Porous Mater. 2006, 13, 55–59. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, J.; Wei, Y.; Lin, J.; Huang, M. Hydrothermal synthesis of K2La2Ti3O10 and photocatalytic splitting of water. J. Alloys Compd. 2008, 456, 364–367. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, W.; Zhu, J.; Lu, L.; Yang, X.; Wang, X. Rapid synthesis of ultrafine K2Ln2Ti3O10 (Ln = La, Nd, Sm, Gd, Dy) series and its photoactivity. J. Solid State Chem. 2005, 178, 761–768. [Google Scholar] [CrossRef]
- Yang, Y.-h.; Qiu, G.-z.; Chen, Q.-Y.; Feng, Q.-m.; Yin, Z.-L. Influence of calcination atmosphere on photocatalytic reactivity of K2La2Ti3010 for water splitting. Trans. Nonferrous Met. Soc. China 2007, 17, 836–840. [Google Scholar] [CrossRef]
- Rodionov, I.A.; Sokolova, I.P.; Silyukov, O.I.; Burovikhina, A.A.; Fateev, S.A.; Zvereva, I.A. Protonation and Photocatalytic Activity of the Rb2La2Ti3O10 Layered Oxide in the Reaction of Hydrogen Production. Int. J. Photoenergy 2017, 2017, 9628146. [Google Scholar] [CrossRef] [Green Version]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Kurnosenko, S.A.; Silyukov, O.I.; Zvereva, I.A. Preparation of Porous Particles of Layered Perovskite-Like Titanate HLaTiO4. Glass Phys. Chem. 2020, 46, 272–276. [Google Scholar] [CrossRef]
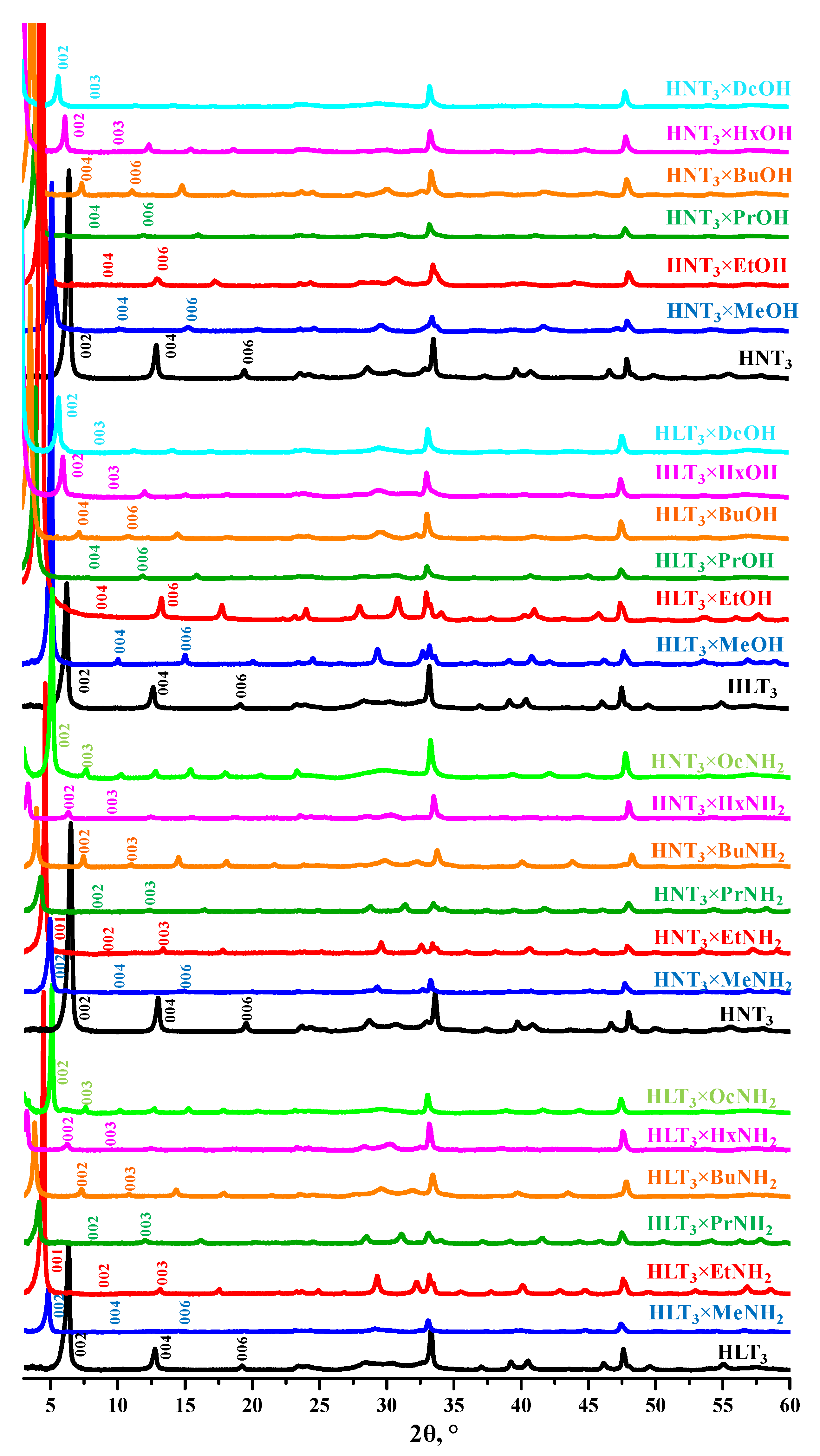
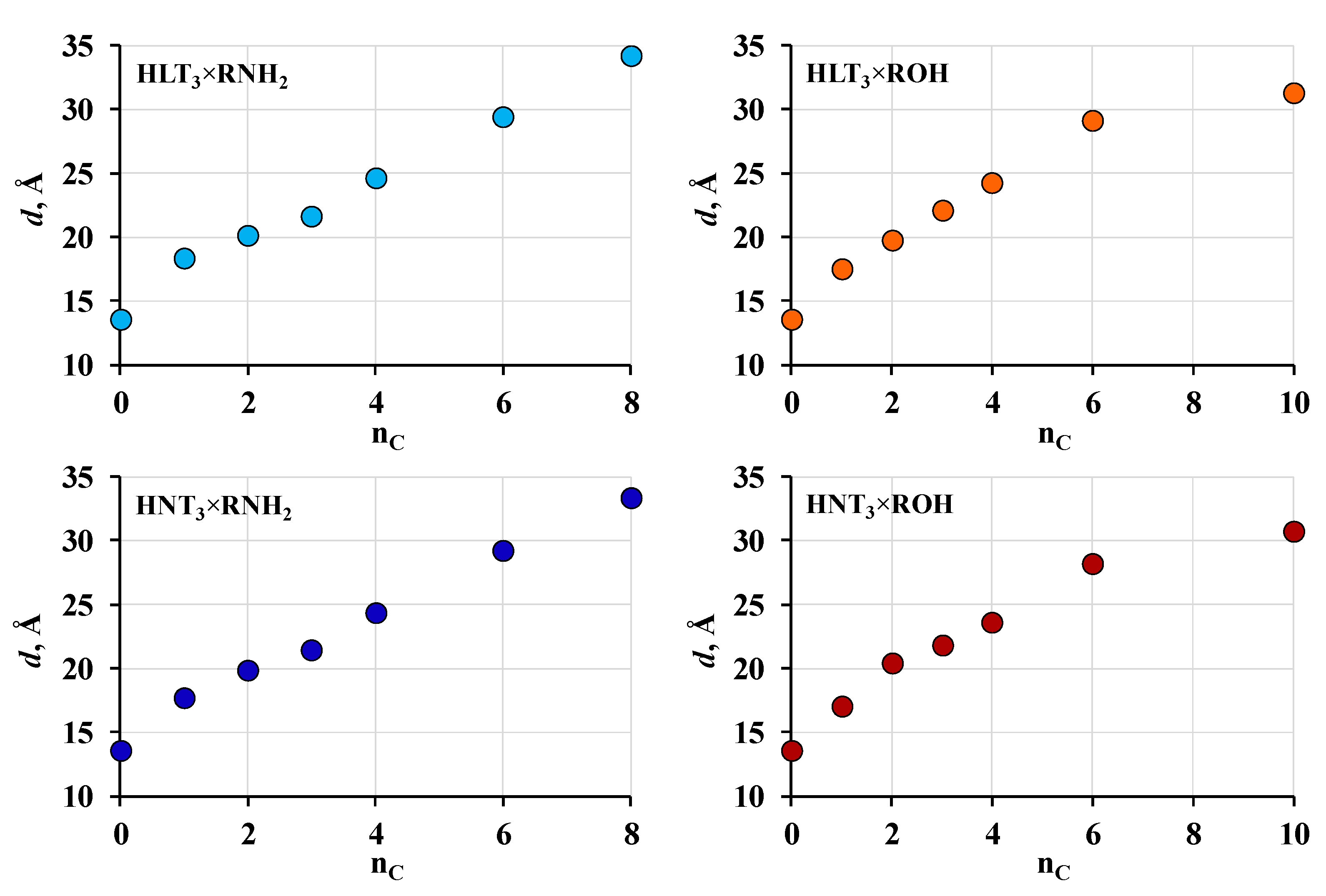

| Sample | a, Å | c, Å | d, Å | α, ° | Sample | a, Å | c, Å | d, Å | |
|---|---|---|---|---|---|---|---|---|---|
| HLT3 | 3.79 | 27.2 | 13.6 | HNT3 | 3.78 | 27.2 | 13.6 | ||
| HLT3 × MeNH2 | 3.83 | 36.7 | 18.4 | 74 | HNT3 × MeNH2 | 3.81 | 35.4 | 17.7 | 71 |
| HLT3 × EtNH2 | 3.82 | 20.2 | 20.2 | HNT3 × EtNH2 | 3.80 | 19.9 | 19.9 | ||
| HLT3 × PrNH2 | 3.82 | 21.7 | 21.7 | HNT3 × PrNH2 | 3.78 | 21.5 | 21.5 | ||
| HLT3 × BuNH2 | 3.81 | 24.7 | 24.7 | HNT3 × BuNH2 | 3.78 | 24.4 | 24.4 | ||
| HLT3 × HxNH2 | 3.87 | 29.4 | 29.4 | HNT3 × HxNH2 | 3.80 | 29.2 | 29.2 | ||
| HLT3 × OcNH2 | 3.85 | 34.2 | 34.2 | HNT3 × OcNH2 | 3.80 | 33.4 | 33.4 | ||
| HLT3 × MeOH | 3.81 | 35.1 | 17.6 | 79 | HNT3 × MeOH | 3.76 | 34.1 | 17.1 | 67 |
| HLT3 × EtOH | 3.83 | 39.5 | 19.8 | HNT3 × EtOH | 3.77 | 40.9 | 20.5 | ||
| HLT3 × PrOH | 3.83 | 44.2 | 22.1 | HNT3 × PrOH | 3.79 | 43.8 | 21.9 | ||
| HLT3 × BuOH | 3.82 | 48.5 | 24.3 | HNT3 × BuOH | 3.78 | 47.3 | 23.7 | ||
| HLT3 × HxOH | 3.82 | 29.2 | 29.2 | HNT3 × HxOH | 3.81 | 28.3 | 28.3 | ||
| HLT3 × DcOH | 3.82 | 31.3 | 31.3 | HNT3 × DcOH | 3.74 | 30.8 | 30.8 |
| Sample | x | y | Sample | x | y |
|---|---|---|---|---|---|
| HLT3 | - | 0.15 | HNT3 | - | 0.15 |
| HLT3 × MeNH2 | 0.70 | 0.30 | HNT3 × MeNH2 | 0.70 | 0.30 |
| HLT3 × EtNH2 | 0.70 | 0.35 | HNT3 × EtNH2 | 0.70 | 0.35 |
| HLT3 × PrNH2 | 0.65 | 0.20 | HNT3 × PrNH2 | 0.70 | 0.20 |
| HLT3 × BuNH2 | 0.70 | 0.35 | HNT3 × BuNH2 | 0.75 | 0.35 |
| HLT3 × HxNH2 | 0.80 | 0.10 | HNT3 × HxNH2 | 0.90 | 0.10 |
| HLT3 × OcNH2 | 0.80 | 0.10 | HNT3 × OcNH2 | 0.85 | 0.15 |
| HLT3 × MeOH | 1.3 | 0.20 | HNT3 × MeOH | 0.95 | 0.25 |
| HLT3 × EtOH | 0.85 | 0.40 | HNT3 × EtOH | 0.85 | 0.35 |
| HLT3 × PrOH | 0.75 | 0.20 | HNT3 × PrOH | 0.75 | 0.30 |
| HLT3 × BuOH | 0.70 | 0.20 | HNT3 × BuOH | 1.1 | 0.25 |
| HLT3 × HxOH | 0.65 | 0.15 | HNT3 × HxOH | 0.80 | 0.10 |
| HLT3 × DcOH | 0.65 | 0.05 | HNT3 × DcOH | 0.65 | 0.05 |
| Sample | Eg, eV | λmax, nm | S, m2/g | Sample | Eg, eV | λmax, nm | S, m2/g |
|---|---|---|---|---|---|---|---|
| HLT3 | 3.44 | 360 | 3.24 | HNT3 | 3.47 | 357 | 3.08 |
| HLT3 × MeNH2 | 3.34 | 371 | 3.88 | HNT3 × MeNH2 | 3.40 | 365 | 3.72 |
| HLT3 × EtNH2 | 3.39 | 366 | − | HNT3 × EtNH2 | 3.32 | 373 | − |
| HLT3 × PrNH2 | 3.40 | 365 | − | HNT3 × PrNH2 | 3.33 | 372 | − |
| HLT3 × BuNH2 | 3.42 | 363 | 4.09 | HNT3 × BuNH2 | 3.33 | 372 | 3.89 |
| HLT3 × HxNH2 | 3.41 | 364 | − | HNT3 × HxNH2 | 3.35 | 370 | − |
| HLT3 × OcNH2 | 3.40 | 365 | 4.38 | HNT3 × OcNH2 | 3.36 | 369 | 4.39 |
| HLT3 × MeOH | 3.33 | 372 | 5.73 | HNT3 × MeOH | 3.33 | 372 | 4.51 |
| HLT3 × EtOH | 3.41 | 364 | − | HNT3 × EtOH | 3.33 | 372 | − |
| HLT3 × PrOH | 3.38 | 367 | − | HNT3 × PrOH | 3.32 | 373 | − |
| HLT3 × BuOH | 3.40 | 365 | 4.19 | HNT3 × BuOH | 3.33 | 372 | 3.87 |
| HLT3 × HxOH | 3.40 | 365 | − | HNT3 × HxOH | 3.35 | 370 | − |
| HLT3 × DcOH | 3.38 | 367 | 5.46 | HNT3 × DcOH | 3.36 | 369 | 4.66 |
| Sample | ω, μmol/h | φ, % | kPt | Sample | ω, μmol/h | φ, % | kPt |
|---|---|---|---|---|---|---|---|
| HLT3 | 1.27 | 0.0170 | 66.9 | HNT3 | 2.01 | 0.0268 | 34.4 |
| HLT3/Pt | 85.1 | 1.14 | HNT3/Pt | 69.0 | 0.922 | ||
| HLT3 × MeNH2 | 81.9 | 1.10 | 20.1 | HNT3 × MeNH2 | 130 | 1.74 | 11.4 |
| HLT3 × MeNH2/Pt | 1647 | 22.0 | HNT3 × MeNH2/Pt | 1476 | 19.7 | ||
| HLT3 × EtNH2 | 61.6 | 0.824 | 38.7 | HNT3 × EtNH2 | 106 | 1.42 | 14.0 |
| HLT3 × EtNH2/Pt | 2385 | 31.9 | HNT3 × EtNH2/Pt | 1482 | 19.8 | ||
| HLT3 × PrNH2 | 45.9 | 0.614 | 43.3 | HNT3 × PrNH2 | 128 | 1.71 | 11.5 |
| HLT3 × PrNH2/Pt | 1989 | 26.6 | HNT3 × PrNH2/Pt | 1474 | 19.7 | ||
| HLT3 × BuNH2 | 72.3 | 0.966 | 30.2 | HNT3 × BuNH2 | 147 | 1.97 | 14.1 |
| HLT3 × BuNH2/Pt | 2181 | 29.2 | HNT3 × BuNH2/Pt | 2078 | 27.8 | ||
| HLT3 × HxNH2 | 53.8 | 0.719 | 30.8 | HNT3 × HxNH2 | 42.0 | 0.562 | 23.0 |
| HLT3 × HxNH2/Pt | 1655 | 22.1 | HNT3 × HxNH2/Pt | 965 | 12.9 | ||
| HLT3 × OcNH2 | 149 | 1.99 | 12.3 | HNT3 × OcNH2 | 54.6 | 0.730 | 18.8 |
| HLT3 × OcNH2/Pt | 1836 | 24.5 | HNT3 × OcNH2/Pt | 1027 | 13.7 | ||
| HLT3 × MeOH | 92.8 | 1.24 | 12.9 | HNT3 × MeOH | 115 | 1.54 | 15.5 |
| HLT3 × MeOH/Pt | 1198 | 16.0 | HNT3 × MeOH/Pt | 1787 | 23.9 | ||
| HLT3 × EtOH | 78.5 | 1.05 | 38.2 | HNT3 × EtOH | 75.8 | 1.01 | 29.9 |
| HLT3 × EtOH/Pt | 2997 | 40.1 | HNT3 × EtOH/Pt | 2268 | 30.3 | ||
| HLT3 × PrOH | 138 | 1.85 | 20.1 | HNT3 × PrOH | 112 | 1.49 | 21.8 |
| HLT3 × PrOH/Pt | 2783 | 37.2 | HNT3 × PrOH/Pt | 2430 | 32.5 | ||
| HLT3 × BuOH | 114 | 1.52 | 24.1 | HNT3 × BuOH | 83.5 | 1.12 | 30.9 |
| HLT3 × BuOH/Pt | 2751 | 36.8 | HNT3 × BuOH/Pt | 2584 | 34.5 | ||
| HLT3 × HxOH | 55.1 | 0.737 | 51.7 | HNT3 × HxOH | 97.9 | 1.31 | 23.4 |
| HLT3 × HxOH/Pt | 2851 | 38.1 | HNT3 × HxOH/Pt | 2285 | 30.6 | ||
| HLT3 × DcOH | 14.9 | 0.199 | 145 | HNT3 × DcOH | 42.0 | 0.561 | 48.6 |
| HLT3 × DcOH/Pt | 2155 | 28.8 | HNT3 × DcOH/Pt | 2039 | 27.3 |
| Synthesis of n-Alkylamine Derivatives | |||||
| Amine | Precursor | Amine Concentration, % | T, °C | D, d | Solvent for Flushing |
| MeNH2 | HLT3 (HNT3) | 38 (in water) | 60 | 10 | acetone |
| EtNH2 | HLT3 (HNT3) × MeNH2 | 70 (in water) | 25 | 1 | ―//― |
| PrNH2 | ―//― | 90 (in water) | ―//― | ―//― | ―//― |
| BuNH2 | ―//― | ―//― | ―//― | ―//― | ―//― |
| HxNH2 | ―//― | 30 (in n-heptane) | 60 | 7 | n-hexane |
| OcNH2 | ―//― | ―//― | ―//― | ―//― | ―//― |
| Synthesis of n-Alkoxy Derivatives | |||||
| Alcohol | Precursor | Alcohol Concentration, % | T, °C | D, d | Solvent for Flushing |
| MeOH | HLT3 (HNT3) × MeNH2 | 100 | 100 | 7 | acetone |
| EtOH | HLT3 (HNT3) × BuNH2 | 96 (in water) | 180 | ―//― | ―//― |
| PrOH | ―//― | 100 | ―//― | ―//― | ―//― |
| BuOH | ―//― | ―//― | ―//― | ―//― | ―//― |
| HxOH | ―//― | ―//― | ―//― | ―//― | n-hexane |
| DcOH | ―//― | 30 (in n-heptane) | ―//― | 10 | ―//― |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kurnosenko, S.A.; Voytovich, V.V.; Silyukov, O.I.; Rodionov, I.A.; Kirichenko, S.O.; Minich, I.A.; Malygina, E.N.; Khramova, A.D.; Zvereva, I.A. Photocatalytic Activity of n-Alkylamine and n-Alkoxy Derivatives of Layered Perovskite-like Titanates H2Ln2Ti3O10 (Ln = La, Nd) in the Reaction of Hydrogen Production from an Aqueous Solution of Methanol. Catalysts 2021, 11, 1279. https://doi.org/10.3390/catal11111279
Kurnosenko SA, Voytovich VV, Silyukov OI, Rodionov IA, Kirichenko SO, Minich IA, Malygina EN, Khramova AD, Zvereva IA. Photocatalytic Activity of n-Alkylamine and n-Alkoxy Derivatives of Layered Perovskite-like Titanates H2Ln2Ti3O10 (Ln = La, Nd) in the Reaction of Hydrogen Production from an Aqueous Solution of Methanol. Catalysts. 2021; 11(11):1279. https://doi.org/10.3390/catal11111279
Chicago/Turabian StyleKurnosenko, Sergei A., Vladimir V. Voytovich, Oleg I. Silyukov, Ivan A. Rodionov, Sergei O. Kirichenko, Iana A. Minich, Ekaterina N. Malygina, Alina D. Khramova, and Irina A. Zvereva. 2021. "Photocatalytic Activity of n-Alkylamine and n-Alkoxy Derivatives of Layered Perovskite-like Titanates H2Ln2Ti3O10 (Ln = La, Nd) in the Reaction of Hydrogen Production from an Aqueous Solution of Methanol" Catalysts 11, no. 11: 1279. https://doi.org/10.3390/catal11111279
APA StyleKurnosenko, S. A., Voytovich, V. V., Silyukov, O. I., Rodionov, I. A., Kirichenko, S. O., Minich, I. A., Malygina, E. N., Khramova, A. D., & Zvereva, I. A. (2021). Photocatalytic Activity of n-Alkylamine and n-Alkoxy Derivatives of Layered Perovskite-like Titanates H2Ln2Ti3O10 (Ln = La, Nd) in the Reaction of Hydrogen Production from an Aqueous Solution of Methanol. Catalysts, 11(11), 1279. https://doi.org/10.3390/catal11111279







