Covalently Bonded Ir(IV) on Conducted Blue TiO2 for Efficient Electrocatalytic Oxygen Evolution Reaction in Acid Media
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material Characterization
2.2. Synthesis of the IrO2 Nanocluster-Doped Blue TiO2 Nanoparticles
2.3. Electrochemical Measurements
3. Results and Discussion
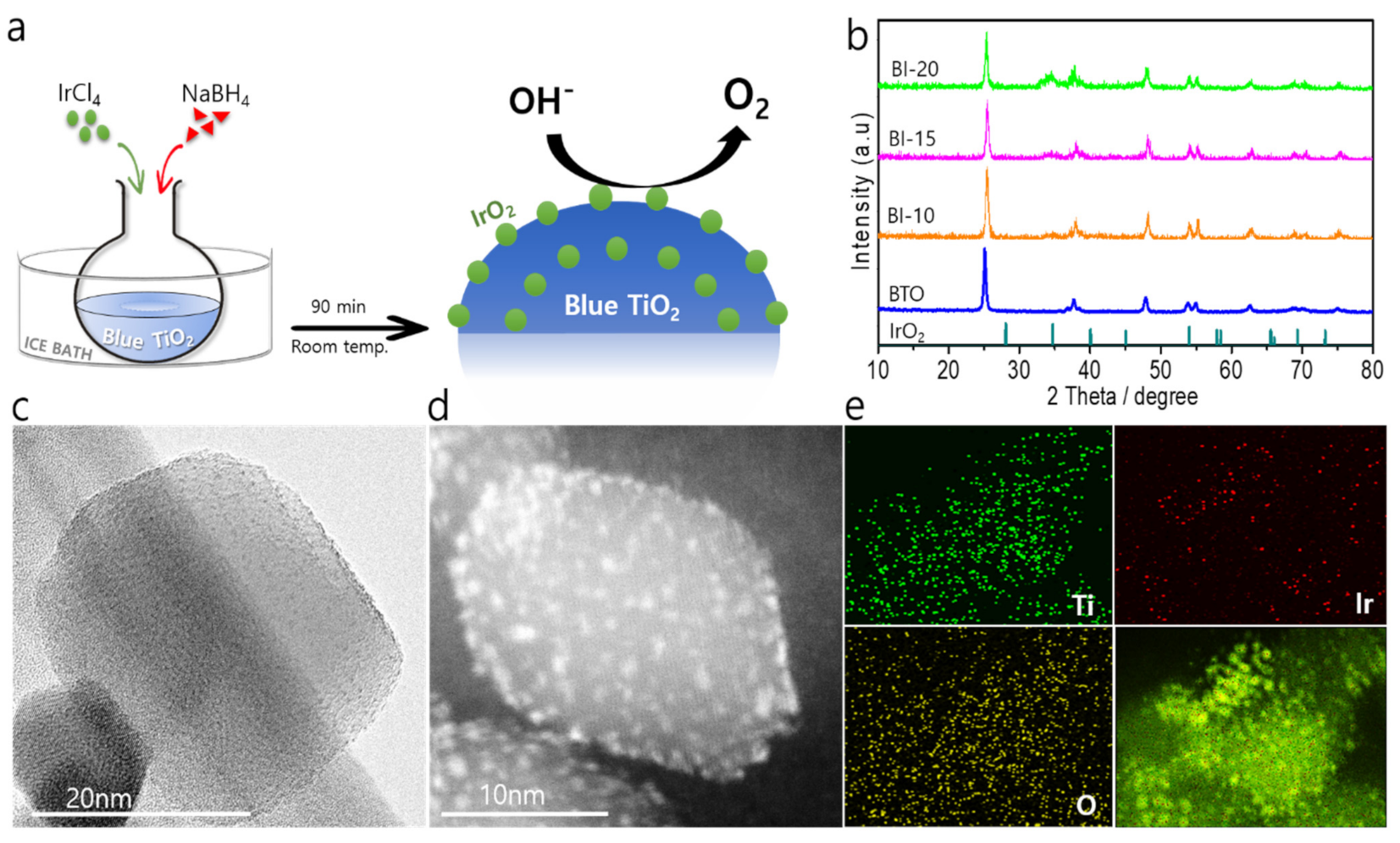
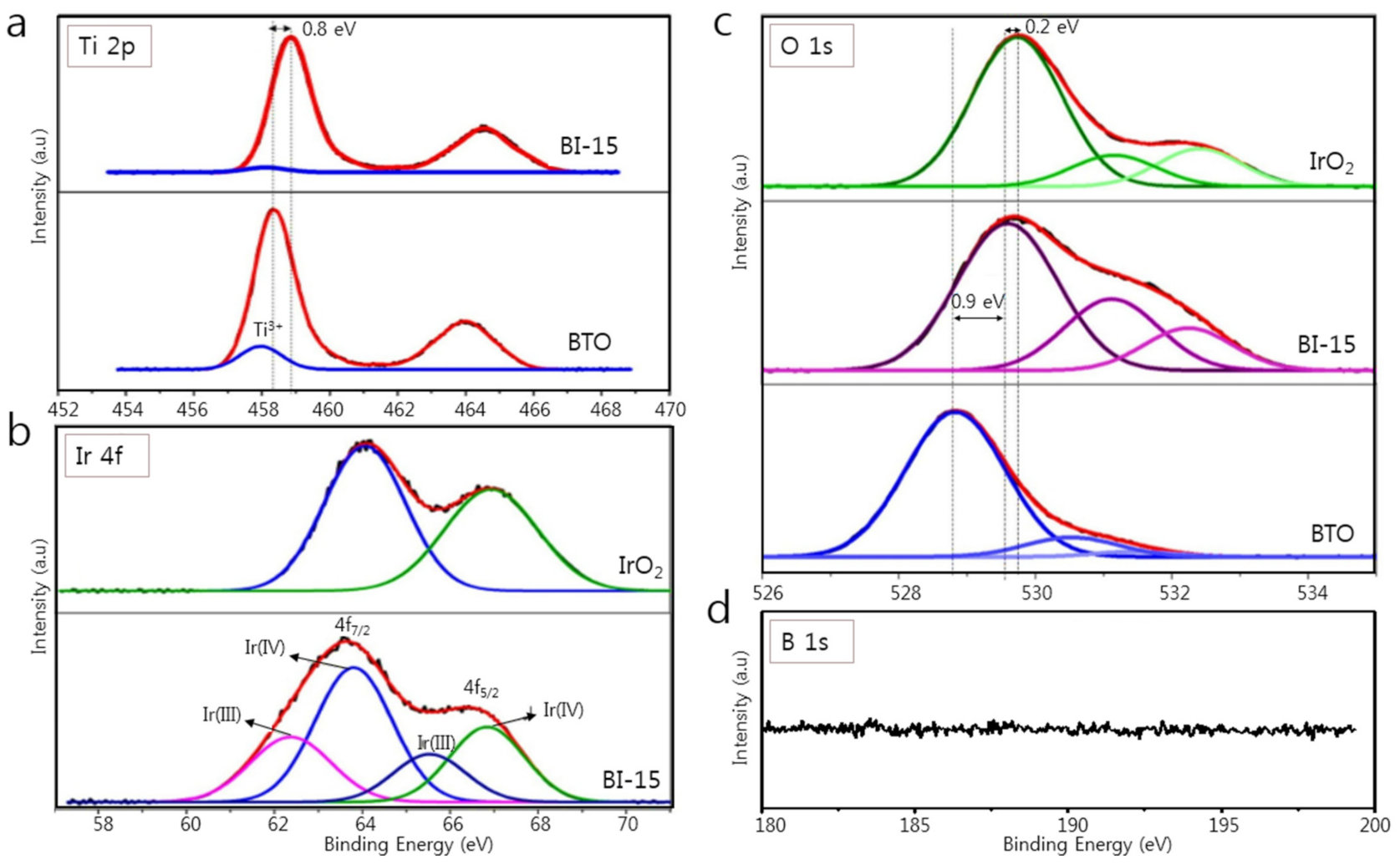
3.1. Structural Characterization
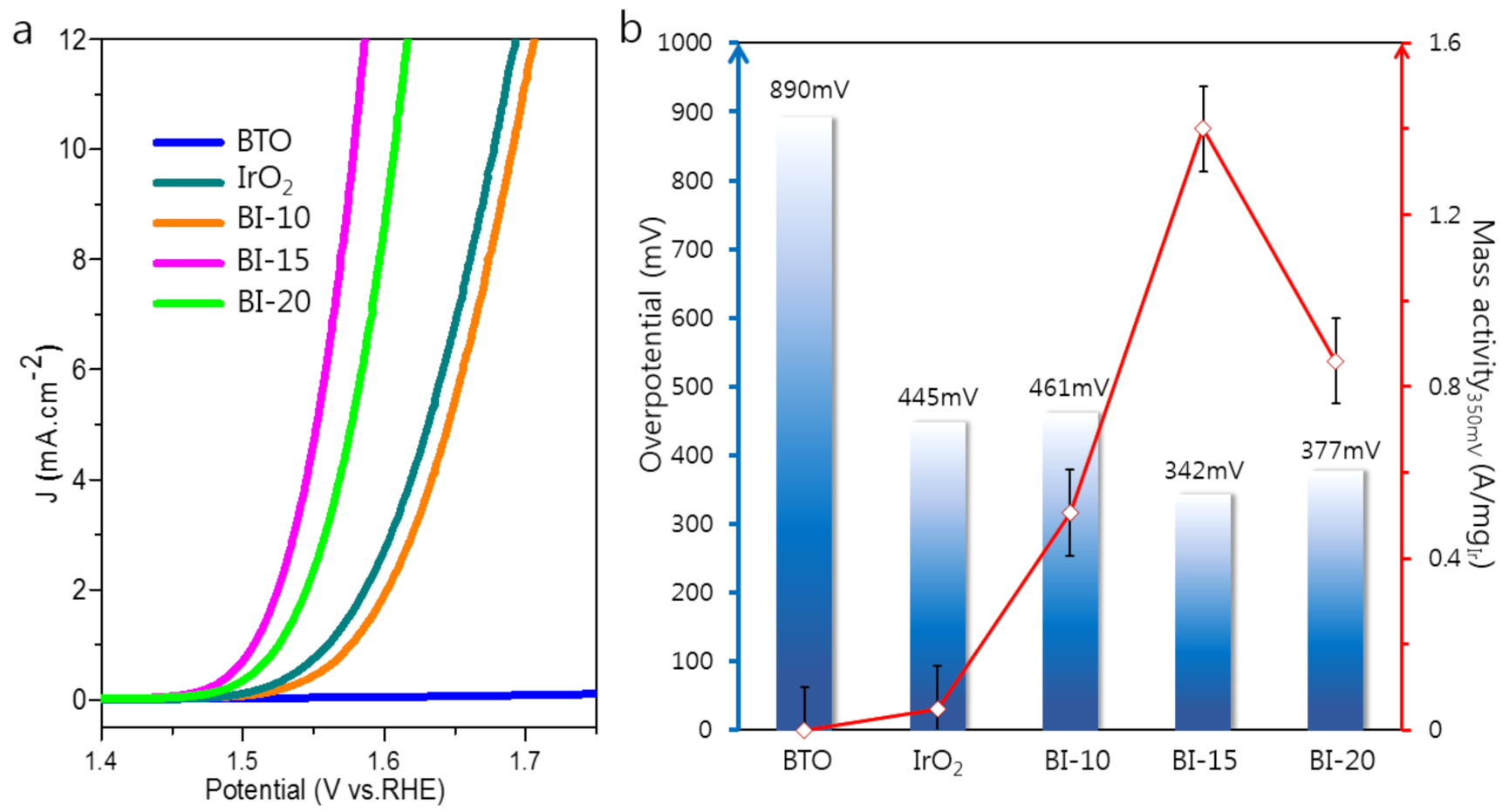
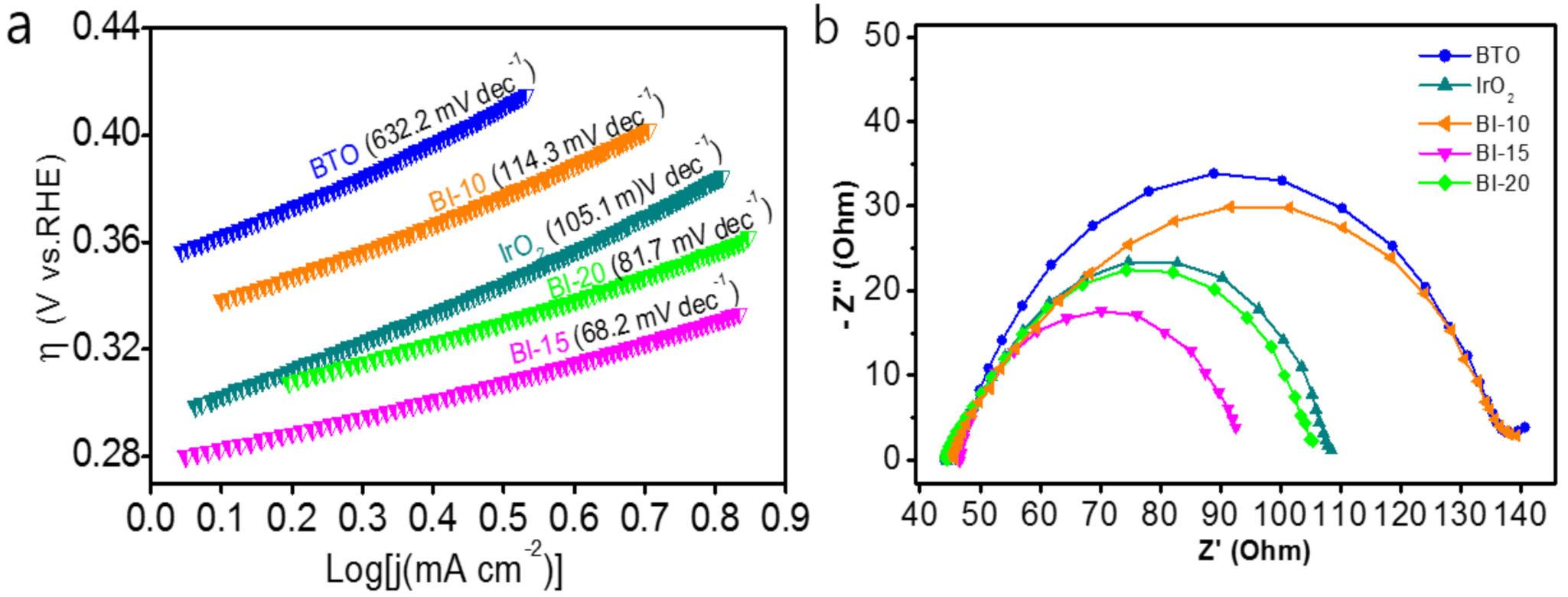
3.2. Evaluation of OER Electrochemical Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ahicart, M.B.; López, J.S.; Carbó, J.J.; Poblet, J.M.; Mascaros, J.R.G. Polyoxometalate electrocatalysts based on earth-abundant metals for efficient water oxidation in acidic media. Nat. Chem. 2018, 10, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.T.K.; Tran, N.Q.; Seo, S.; Hwang, H.; Oh, S.; Yu, J.; Lee, J.; Le, T.A.; Hwang, J.; Kim, M.; et al. Highly efficient nanostructured metal-decorated hybrid semiconductors for solar conversion of CO2 with almost complete CO selectivity. Mater. Today 2019, 35, 25–33. [Google Scholar] [CrossRef]
- Tran, N.Q.; Le, T.A.; Kim, H.; Hong, Y.; Cho, Y.; Park, G.H.; Kim, H.; Kim, M.; Lee, J.; Yoo, W.S.; et al. Low Iridium Content Confined inside a Co3O4 Hollow Sphere for Superior Acidic Water Oxidation. ACS Sustain. Chem. Eng. 2019, 7, 16640–16650. [Google Scholar] [CrossRef]
- Yu, J.; Seo, S.; Luo, Y.; Sun, Y.; Oh, S.; Nguyen, C.T.K.; Seo, C.; Kim, J.H.; Kim, J.; Lee, H. Efficient and Stable Solar Hydrogen Generation of Hydrophilic Rhenium-Disulfide-Based Photocatalysts via Chemically Controlled Charge Transfer Paths. ACS Nano 2020, 14, 1715–1726. [Google Scholar] [CrossRef]
- Gu, X.K.; Camayang, J.C.A.; Samira, S.; Nikolla, E. Oxygen evolution electrocatalysis using mixed metal oxides under acidic conditions: Challenges and opportunities. J. Catal. 2020, 388, 130–140. [Google Scholar] [CrossRef]
- Kumar. S., S.; Himabindu., V. Hydrogen production by PEM water electrolysis – A review. Mater. Sci. Energy Technol. 2019, 2, 442–454. [Google Scholar]
- Chandrasekaran, S.; Ma, D.; Ge, Y.; Deng, L.; Bowen, C.; Roscow, J.; Zhang, Y.; Lin, Z.; Misra, R.D.K.; Li, J.; et al. Electronic structure engineering on two-dimensional (2D) electrocatalytic materials for oxygen reduction, oxygen evolution, and hydrogen evolution reactions. Nano Energy 2020, 77, 105080. [Google Scholar] [CrossRef]
- Shi, Q.; Zhu, C.; Du, D.; Lin, Y. Robust noble metal-based electrocatalysts for oxygen evolution reaction. Chem. Soc. Rev. 2019, 48, 3181–3192. [Google Scholar] [CrossRef]
- Alia, S.M.; Shulda, S.; Ngo, C.; Pylypenko, S.; Pivovar, B.S. Iridium-Based Nanowires as Highly Active, Oxygen Evolution Reaction Electrocatalysts. ACS Catal. 2018, 8, 2111–2120. [Google Scholar] [CrossRef]
- Oakton, E.; Lebedev, D.; Povia, M.; Abbott, D.F.; Fabbri, E.; Fedorov, A.; Nachtegaal, M.; Coperet, C.; Schmidt, T.J. IrO2-TiO2: A High-Surface-Area, Active, and Stable Electrocatalyst for the Oxygen Evolution Reaction. ACS Catal. 2017, 7, 2346–2352. [Google Scholar] [CrossRef]
- Cheng, J.; Yang, J.; Kitano, S.; Juhasz, G.; Higashi, M.; Sadakiyo, M.; Kato, K.; Yoshioka, S.; Sugiyama, T.; Yamauchi, M.; et al. Impact of Ir-Valence Control and Surface Nanostructure on Oxygen Evolution Reaction over a Highly Efficient Ir–TiO2 Nanorod Catalyst. ACS Catal. 2019, 9, 6974–6986. [Google Scholar] [CrossRef]
- Kwon, T.; Hwang, H.; Sa, Y.J.; Park, J.; Baik, H.; Joo, S.H.; Lee, K. Cobalt Assisted Synthesis of IrCu Hollow Octahedral Nanocages as Highly Active Electrocatalysts toward Oxygen Evolution Reaction. Adv. Funct. Mater. 2017, 27, 1604688. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, L.; Kim, J.K.; Ma, M.; Veerappan, G.; Lee, C.L.; Kong, K.J.; Lee, H.; Park, J.H. An order/disorder/water junction system for highly efficient co-catalyst-free photocatalytic hydrogen generation. Energy Environ. Sci. 2016, 9, 499. [Google Scholar] [CrossRef]
- Yang, L.; Yu, G.; Ai, X.; Yan, W.; Duan, H.; Chen, W.; Li, X.; Wang, T.; Zhang, C.; Huang, X.; et al. Efficient oxygen evolution electrocatalysis in acid by a perovskite with face-sharing IrO6 octahedral dimers. Nat. Commun. 2018, 9, 5236. [Google Scholar] [CrossRef] [Green Version]
- Park, J.; Sa, Y.J.; Baik, H.; Kwon, T.; Joo, S.H.; Lee, K. Iridium-Based Multimetallic Nanoframe@Nanoframe Structure: An Efficient and Robust Electrocatalyst toward Oxygen Evolution Reaction. ACS Nano 2017, 11, 5500–5509. [Google Scholar] [CrossRef]
- Chakrapani, K.; Sampath, S. The dual role of borohydride depending on reaction temperature: Synthesis of iridium and iridium oxide. Chem. Commun. 2015, 51, 9690. [Google Scholar] [CrossRef]
- Tachikawa, T.; Beniya, A.; Shigetoh, K.; Higashi, S. Relationship between OER Activity and Annealing Temperature of Sputter-Deposited Flat IrO2 Thin Films. Catal. Lett. 2020, 150, 1976–1984. [Google Scholar] [CrossRef]
- Seitz, L.C.; Dickens, C.F.; Nishio, K.; Hikita, Y.; Montoya, J.; Doyle, A.; Kirk, C.; Vojvodic, A.; Hwang, H.Y.; Norskov, J.K.; et al. A highly active and stable IrOx/SrIrO3 catalyst for the oxygen evolution reaction. Science 2016, 353, 1011–1014. [Google Scholar] [CrossRef]
- Mattinen, M.; Hämäläinen, J.; Vehkamäki, M.; Heikkilä, M.J.; Mizohata, K.; Jalkanen, P.; Räisänen, J.; Ritala, M.; Leskelä, M. Atomic Layer Deposition of Iridium Thin Films Using Sequential Oxygen and Hydrogen Pulses. J. Phys. Chem. C 2016, 28, 15235–15243. [Google Scholar] [CrossRef]
- Matienzo, D.D.; Settipani, D.; Instuli, E.; Kallio, T. Active IrO2 and NiO Thin Films Prepared by Atomic Layer Deposition for Oxygen Evolution Reaction. Catalysts 2020, 10, 92. [Google Scholar] [CrossRef] [Green Version]
- Anantharaj, S.; Karthick, K.; Kundu, S. Evolution of layered double hydroxides (LDH) as high performance water oxidation electrocatalysts: A review with insights on structure, activity and mechanism. Mater. Today Energy 2017, 6, 1–26. [Google Scholar] [CrossRef]
- Mattinen, M.; Hämäläinen, J.; Gao, F.; Jalkanen, P.; Mizohata, K.; Räisänen, J.; Puurunen, R.L.; Ritala, M.; Leskelä, M. Nucleation and Conformality of Iridium and Iridium Oxide Thin Films Grown by Atomic Layer Deposition. Langmuir 2016, 32, 10559–10569. [Google Scholar] [CrossRef] [PubMed]
- Lettenmeier, P.; Majchel, J.; Wang, L.; Saveleva, V.A.; Zafeiratos, S.; Savinova, E.R.; Gallet, J.J.; Bournel, F.; Gago, A.S.; Friedrich, K.A. Highly active nano-sized iridium catalysts: Synthesis and operando spectroscopy in a proton exchange membrane electrolyzer. Chem. Sci. 2018, 9, 3570–3579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, K.; Luo, M.; Peng, M.; Yu, Y.; Lu, Y.R.; Chan, T.S.; Liu, P.; Groot, F.M.F.; Tan, Y. Dynamic active-site generation of atomic iridium stabilized on nanoporous metal phosphides for water oxidation. Nat. Commun. 2020, 11, 2701. [Google Scholar] [CrossRef]
- Li, C.; Baek, J.B. Recent Advances in Noble Metal (Pt, Ru, and Ir)-Based Electrocatalysts for Efficient Hydrogen Evolution Reaction. ACS Omega 2020, 5, 31–40. [Google Scholar] [CrossRef] [Green Version]
- Cho, Y.; Le, T.A.; Lee, H. Understanding Surface Modulation to Improve the Photo/Electrocatalysts for Water Oxidation/Reduction. Molecules 2020, 25, 1965. [Google Scholar] [CrossRef] [Green Version]
- Tahir, M.; Pan, L.; Idrees, F.; Zhang, X.; Wang, L.; Zou, J.J.; Wang, Z.L. Electrocatalytic oxygen evolution reaction for energy conversion and storage: A comprehensive review. Nano Energy 2017, 37, 136–157. [Google Scholar] [CrossRef]
- Reier, T.; Nong, H.N.; Teschner, D.; Schlögl, R.; Strasser, P. Electrocatalytic Oxygen Evolution Reaction in Acidic Environments – Reaction Mechanisms and Catalysts. Adv. Energy Mater. 2017, 7, 1601275. [Google Scholar] [CrossRef]
- Tong, J.; Liu, Y.; Peng, Q.; Hu, W.; Wu, Q. An efficient Sb-SnO2-supported IrO2 electrocatalyst for the oxygen evolution reaction in acidic medium. J. Mater. Sci. 2017, 52, 13427–13443. [Google Scholar] [CrossRef]
- Kim, Y.T.; Lopes, P.P.; Park, S.A.; Lee, A.Y.; Lim, J.; Lee, H.; Back, S.; Jung, Y.; Danilovic, N.; Stamenkovic, V.; et al. Balancing activity, stability and conductivity of nanoporous core-shell iridium/iridium oxide oxygen evolution catalysts. Nat. Commun. 2017, 8, 1449. [Google Scholar] [CrossRef]
- Hwang, H.M.; Oh, S.; Shim, J.H.; Kim, Y.M.; Kim, A.; Kim, D.; Kim, J.; Bak, S.; Cho, Y.; Bui, V.Q.; et al. Phase-Selective Disordered Anatase/Ordered Rutile Interface System for Visible-Light-Driven, Metal-Free CO2 Reduction. ACS Appl. Mater. Interfaces 2019, 11, 35693–35701. [Google Scholar] [CrossRef]
- Povia, M.; Abbott, D.F.; Herranz, J.; Heinritz, A.; Lebedev, D.; Kim, B.J.; Fabbri, E.; Patru, A.; Kohlbrecher, J.; Schaublin, R.; et al. Operando X-ray characterization of high surface area iridium oxides to decouple their activity losses for the oxygen evolution reaction. Energy Environ. Sci. 2019, 12, 3038–3052. [Google Scholar] [CrossRef]
- Halder, A.; Liu, C.; Liu, Z.; Emery, J.D.; Pellin, M.J.; Curtiss, L.A.; Zapol, P.; Vajda, S.; Martinson, A.B.F. Water Oxidation Catalysis via Size-Selected Iridium Clusters. J. Phys. Chem. C 2018, 122, 9965–9972. [Google Scholar] [CrossRef]
- Chen, L.W.; Tong, L.; Nan, H.; Chu, S.Q.; Liang, H.W. Sub-2 nm Ir Nanoclusters Immobilized on Mesoporous Nitrogen-Doped Carbons as Efficient Catalysts for Selective Hydrogenation. ACS Appl. Nano Mater. 2019, 10, 6546–6553. [Google Scholar] [CrossRef]
- Meekins, B.H.; Kamat, P.V. Role of Water Oxidation Catalyst IrO2 in Shuttling Photogenerated Holes Across TiO2 Interface. J. Phys. Chem. Lett. 2011, 18, 2304–2310. [Google Scholar] [CrossRef]
- Pham, C.V.; Bühler, M.; Knöppel, J.; Bierling, M.; Seeberger, D.; López, D.E.; Mayrhofer, K.J.J.; Cherevko, S.; Thiele, S. IrO2 coated TiO2 core-shell microparticles advance performance of low loading proton exchange membrane water electrolyzers. Appl. Catal. B Environ. 2020, 269, 118762. [Google Scholar] [CrossRef]
- Yuan, M.; Zhu, Y.; Deng, L.; Ming, R.; Zhang, A.; Li, W.; Chaia, B.; Ren, Z. IrO2–TiO2 electrocatalysts for the hydrogen evolution reaction in acidic water electrolysis without activation. New J. Chem. 2017, 41, 6152–6159. [Google Scholar] [CrossRef]
- Zhong, W.; Lin, Z.; Feng, S.; Wang, D.; Shen, S.; Zhang, Q.; Gu, L.; Wang, Z.; Fang, B. Improved oxygen evolution activity of IrO2 by in situ engineering of an ultra-small Ir sphere shell utilizing a pulsed laser. Nanoscale 2019, 11, 4407–4413. [Google Scholar] [CrossRef]
- Ohno, H.; Nohara, S.; Kakinuma, K.; Uchida, M.; Uchida, H. Effect of Electronic Conductivities of Iridium Oxide/Doped SnO2 Oxygen-Evolving Catalysts on the Polarization Properties in Proton Exchange Membrane Water Electrolysis. Catalysts 2019, 9, 74. [Google Scholar] [CrossRef] [Green Version]
- Grimaud, A.; Demortière, A.; Saubanère, M.; Dachraoui, W.; Duchamp, M.; Doublet, M.L.; Tarascon, J.M. Activation of surface oxygen sites on an iridium-based model catalyst for the oxygen evolution reaction. Nat. Energy 2016, 2, 16189. [Google Scholar] [CrossRef]
- Smith, R.D.L.; Sporinova, B.; Fagan, R.D.; Trudel, S.; Berlinguette, C.P. Facile Photochemical Preparation of Amorphous Iridium Oxide Films for Water Oxidation Catalysis. Chem. Mater. 2014, 26, 1654–1659. [Google Scholar] [CrossRef]
- Wua, X.; Fenga, B.; Lia, W.; Niua, Y.; Yua, Y.; Lua, S.; Zhonga, C.; Liuc, P.; Tianc, Z.; Chenc, L.; et al. Metal-support interaction boosted electrocatalysis of ultrasmall iridium nanoparticles supported on nitrogen doped graphene for highly efficient water electrolysis in acidic and alkaline media. Nano Energy 2019, 62, 117–126. [Google Scholar] [CrossRef]
- Gorlin, Y.; Chung, C.J.; Benck, J.D.; Nordlund, D.; Seitz, L.; Weng, T.C.; Sokaras, D.; Clemens, B.M.; Jaramillo, T.F. Understanding Interactions between Manganese Oxide and Gold That Lead to Enhanced Activity for Electrocatalytic Water Oxidation. J. Am. Chem. Soc. 2014, 136, 4920–4926. [Google Scholar] [CrossRef] [PubMed]
- Lyons, M.E.G.; Floquet, S. Mechanism of oxygen reactions at porous oxideelectrodes. Part 2—Oxygen evolution at RuO2, IrO2 and IrxRu1-xO2 electrodes in aqueous acid and alkaline solution. Phys. Chem. Chem. Phys. 2011, 13, 5314–5335. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Dubouis, N.; Osman, M.B.; Yin, W.; Sougrati, M.T.; Corte, D.A.D.; Giaume, D.; Grimaud, A. A Dissolution/Precipitation Equilibrium on the Surface of Iridium-Based Perovskites Controls Their Activity as Oxygen Evolution Reaction Catalysts in Acidic Media. Angew. Chem. Int. Ed. 2019, 58, 4571–4575. [Google Scholar] [CrossRef] [PubMed]
- Nong, H.N.; Reier, T.; Oh, H.S.; Gliech, M.; Paciok, P.; Vu, T.H.T.; Teschner, D.; Heggen, M.; Petkov, V.; Schlögl, R.; et al. A unique oxygen ligand environment facilitates water oxidation in hole-doped IrNiOx core–shell electrocatalysts. Nat. Catal. 2018, 1, 841–851. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, C.T.K.; Tran, N.Q.; Le, T.A.; Lee, H. Covalently Bonded Ir(IV) on Conducted Blue TiO2 for Efficient Electrocatalytic Oxygen Evolution Reaction in Acid Media. Catalysts 2021, 11, 1176. https://doi.org/10.3390/catal11101176
Nguyen CTK, Tran NQ, Le TA, Lee H. Covalently Bonded Ir(IV) on Conducted Blue TiO2 for Efficient Electrocatalytic Oxygen Evolution Reaction in Acid Media. Catalysts. 2021; 11(10):1176. https://doi.org/10.3390/catal11101176
Chicago/Turabian StyleNguyen, Chau T. K., Ngoc Quang Tran, Thi Anh Le, and Hyoyoung Lee. 2021. "Covalently Bonded Ir(IV) on Conducted Blue TiO2 for Efficient Electrocatalytic Oxygen Evolution Reaction in Acid Media" Catalysts 11, no. 10: 1176. https://doi.org/10.3390/catal11101176
APA StyleNguyen, C. T. K., Tran, N. Q., Le, T. A., & Lee, H. (2021). Covalently Bonded Ir(IV) on Conducted Blue TiO2 for Efficient Electrocatalytic Oxygen Evolution Reaction in Acid Media. Catalysts, 11(10), 1176. https://doi.org/10.3390/catal11101176






