C-,N- and S-Doped TiO2 Photocatalysts: A Review
Abstract
1. Introduction
2. TiO2 Photocatalysis
3. C-Doped TiO2
4. N-Doped TiO2
5. S-Doped TiO2
6. C,N-Co-Doped TiO2
7. C,S-Co-Doped TiO2
8. N,S-Co-Doped TiO2
9. C,N,S-Tri-Doped TiO2
10. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations and Symbols
| 2ATO | anionic S-doped TiO2 nanorods |
| 2CTO | cationic S-doped TiO2 nanorods |
| 4-NP | 4-nitrophenol |
| 5-FU | 5-fluorouracil |
| Asc-TiO2 | TiO2 modified with citric acid as carbon source |
| BE | binding energy |
| BPA | bisphenol A |
| BQ | benzoquinone |
| CB | conduction band |
| CBD | chemical bath deposition |
| C/C0 | normalized concentration |
| CDs | carbon dots |
| CIP | ciprofloxacin |
| Cit-TiO2 | TiO2 modified with citric acid as carbon source |
| CTCNF | C-doped TiO2/carbon nanofibrous film |
| CTF | C-doped TiO2 flakes |
| CTS | C-doped TiO2 sheets |
| CVD | chemical vapor deposition |
| DC | doxycycline |
| DCF | diclofenac |
| DEA | diethanolamine |
| DFT | density functional theory |
| DMF | N,N-dimethylformamide |
| DMSO | dimethyl sulfoxide |
| DP | diphenhydramine hydrochloride |
| dTiO2 | crystallite size estimated from XRD |
| e− | electron |
| Ebg | band gap energy |
| EDA | ethylenediamine |
| EDT | 1,2-ethanedithiol |
| EDTA-Na2 | ethylenediaminetetraacetic acid disodium salt |
| EPF | extrapallial fluid |
| ETA | ethanolamine |
| FB | flat-band |
| h+ | hole |
| HA | humic acids |
| HDA | 1,6-diaminohexane |
| HDPE | high-density polyethylene |
| HMT | hexamethylenetetramine |
| IB | intra band gap states |
| IBP | ibuprofen |
| k | pseudo-first order rate constant |
| MB | methylene blue |
| MCP | monocrotophos |
| MO | methyl orange |
| MWCNT | multi-walled carbon nanotubes |
| Ni | interstitial nitrogen state |
| Ns | substitutional nitrogen state |
| NHE | normal hydrogen electrode |
| NPs | nanoparticles |
| NPX | naproxen |
| NWs | nanowires |
| Olat | lattice oxygen |
| Osur | surface adsorbed oxygen |
| Ov | oxygen vacancy |
| OPM | oxidant peroxide method |
| PAN | polyacrylonitrile |
| PLD | pulsed laser deposition |
| Ph | phenol |
| PS | polystyrene |
| PVP | polyvinylpyrrolidone |
| QP | quinalphos |
| r-GO | reduced graphene oxide |
| RB19 | reactive blue 19 |
| Rh B | rhodamine b |
| RO16 | reactive orange 16 |
| ROS | reactive oxygen species |
| RR198 | reactive red 198 |
| RR76 | reactive red 76 |
| RT | room temperature |
| SBET | specific surface area estimated with the Brunauer–Emmett–Teller (BET) method |
| (S)O | substitutional sulfur |
| (SO)O | interstitial sulfur |
| (S2)O | interstitial sulfur sharing a lattice site with substitutional sulfur |
| ST | safranin T |
| TAA | anatase TiO2 from Sigma Aldrich |
| TBAH | tetrabutylammonium hydroxide |
| TBOT | titanium(IV) butoxide |
| TC | tetracycline |
| TCH | tetracycline hydrochloride |
| TCPP | tris(1-chloro-2-propyl)phosphate |
| TDMAT | tetrakis(dimethylamino)titanium |
| TEA | triethylamine |
| TELA | triethanolamine |
| TEM | transmission electron microscopy |
| TiO2 P25 | Aeroxide® TiO2 P25 from Evonik, Germany |
| TOC | total organic carbon |
| TTIP | titanium isopropoxide |
| UV-vis/DR | UV-vis diffuse reflectance (spectroscopy) |
| VB | valence band |
| VOCs | volatile organic compound |
| XPS | X-ray photoelectron spectroscopy |
| XRD | X-ray diffraction |
References
- Mittal, A.; Mari, B.; Sharma, S.; Kumari, V.; Maken, S.; Kumari, K.; Kumar, N. Non-metal Modified TiO2: A Step towards Visible Light Photocatalysis. J. Mater. Sci. Mater. Electron. 2019, 30, 3186–3207. [Google Scholar] [CrossRef]
- Cuerda-Correa, E.M.; Alexandre-Franco, M.F.; Fernández-González, C. Advanced Oxidation Processes for the Removal of Antibiotics from Water. An Overview. Water 2019, 12, 102. [Google Scholar] [CrossRef]
- Saibu, S.; Adebusoye, S.A.; Oyetibo, G.O. Aerobic Bacterial Transformation and Biodegradation of Dioxins: A Review. Bioresour. Bioprocess. 2020, 7. [Google Scholar] [CrossRef]
- Kumari, V.; Mittal, A.; Jindal, J.; Yadav, S.; Kumar, N. S-, N- and C-doped ZnO as Semiconductor Photocatalysts: A Review. Front. Mater. Sci. 2019, 13, 1–22. [Google Scholar] [CrossRef]
- Din, M.I.; Khalid, R.; Hussain, Z. Recent Research on Development and Modification of Nontoxic Semiconductor for Environmental Application. Sep. Purif. Rev. 2020, 1–18. [Google Scholar] [CrossRef]
- Al-Mamun, M.R.; Kader, S.; Islam, M.S.; Khan, M.Z.H. Photocatalytic Activity Improvement and Application of UV-TiO2 Photocatalysis in Textile Wastewater Treatment: A Review. J. Environ. Chem. Eng. 2019, 7, 103248. [Google Scholar] [CrossRef]
- Ramandi, S.; Entezari, M.H.; Ghows, N. Sono-Synthesis of Solar Light Responsive S–N–C–Tri Doped TiO2 Photo-Catalyst under Optimized Conditions for Degradation and Mineralization of Diclofenac. Ultrason. Sonochem. 2017, 38, 234–245. [Google Scholar] [CrossRef]
- Khedr, T.M.; El-Sheikh, S.M.; Hakki, A.; Ismail, A.A.; Badawy, W.A.; Bahnemann, D.W. Highly Active Non-metals Doped Mixed-Phase TiO2 for Photocatalytic Oxidation of Ibuprofen under Visible Light. J. Photoch. Photobiol. A. 2017, 346, 530–540. [Google Scholar] [CrossRef]
- Nasirian, M.; Lin, Y.P.; Bustillo-Lecompte, C.F.; Mehrvar, M. Enhancement of Photocatalytic Activity of Titanium Dioxide Using Non-metal Doping Methods under Visible Light: A Review. Int. J. Environ. Sci. Technol. 2017, 15, 2009–2032. [Google Scholar] [CrossRef]
- Islam, S.; Nagpure, S.; Kim, D.; Rankin, S. Synthesis and Catalytic Applications of Non-metal Doped Mesoporous Titania. Inorganics 2017, 5, 15. [Google Scholar] [CrossRef]
- Sanchez-Martinez, A.; Ceballos-Sanchez, O.; Koop-Santa, C.; López-Mena, E.R.; Orozco-Guareño, E.; García-Guaderrama, M. N-doped TiO2 Nanoparticles Obtained by a Facile Coprecipitation Method at Low Temperature. Ceram. Int. 2018, 44, 5273–5283. [Google Scholar] [CrossRef]
- Lazar, M.; Varghese, S.; Nair, S. Photocatalytic Water Treatment by Titanium Dioxide: Recent Updates. Catalysts 2012, 2, 572–601. [Google Scholar] [CrossRef]
- Chen, J.; Qiu, F.; Xu, W.; Cao, S.; Zhu, H. Recent Progress in Enhancing Photocatalytic Efficiency of TiO2 -based Materials. Appl. Catal. A-Gen. 2015, 495, 131–140. [Google Scholar] [CrossRef]
- Devi, L.G.; Kavitha, R. A Review on Non Metal Ion Doped Titania for the Photocatalytic Degradation of Organic Pollutants under UV/Solar Light: Role of Photogenerated Charge Carrier Dynamics in Enhancing the Activity. Appl. Catal. B-Environ. 2013, 140–141, 559–587. [Google Scholar] [CrossRef]
- Al Jitan, S.; Palmisano, G.; Garlisi, C. Synthesis and Surface Modification of TiO2-based Photocatalysts for the Conversion of CO2. Catalysts 2020, 10, 227. [Google Scholar] [CrossRef]
- Basavarajappa, P.S.; Patil, S.B.; Ganganagappa, N.; Reddy, K.R.; Raghu, A.V.; Reddy, C.V. Recent Progress in Metal-doped TiO2, Non-metal Doped/Codoped TiO2 and TiO2 Nanostructured Hybrids for Enhanced Photocatalysis. Int. J. Hydrogen Energ. 2020, 45, 7764–7778. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Irie, H.; Ohwaki, T. Nitrogen-Doped Titanium Dioxide as Visible-Light-Sensitive Photocatalyst: Designs, Developments, and Prospects. Chem. Rev. 2014, 114, 9824–9852. [Google Scholar] [CrossRef]
- Rammohan, G.; Nadagouda, M. Green Photocatalysis for Degradation of Organic Contaminants: A Review. COC 2013, 17, 2338–2348. [Google Scholar] [CrossRef]
- Teh, C.M.; Mohamed, A.R. Roles of Titanium Dioxide and Ion-Doped Titanium Dioxide on Photocatalytic Degradation of Organic Pollutants (Phenolic Compounds and Dyes) in Aqueous Solutions: A Review. J. Alloys Compd. 2011, 509, 1648–1660. [Google Scholar] [CrossRef]
- Ismail, A.A.; Bahnemann, D.W. Mesoporous Titania Photocatalysts: Preparation, Characterization and Reaction Mechanisms. J. Mater. Chem. 2011, 21, 11686. [Google Scholar] [CrossRef]
- Akpan, U.G.; Hameed, B.H. The Advancements in Sol–Gel Method of Doped-TiO2 Photocatalysts. Appl. Catal. A Gen. 2010, 375, 1–11. [Google Scholar] [CrossRef]
- Ajmal, A.; Majeed, I.; Malik, R.N.; Idriss, H.; Nadeem, M.A. Principles and Mechanisms of Photocatalytic Dye Degradation on TiO2based Photocatalysts: A Comparative Overview. RSC Adv. 2014, 4, 37003–37026. [Google Scholar] [CrossRef]
- Ahmed, S.; Rasul, M.G.; Martens, W.N.; Brown, R.; Hashib, M.A. Heterogeneous Photocatalytic Degradation of Phenols in Wastewater: A Review on Current Status and Developments. Desalination 2010, 261, 3–18. [Google Scholar] [CrossRef]
- Wang, W.; Chen, M.; Huang, D.; Zeng, G.; Zhang, C.; Lai, C.; Zhou, C.; Yang, Y.; Cheng, M.; Hu, L.; et al. An Overview on Nitride and Nitrogen-doped Photocatalysts for Energy and Environmental Applications. Compos. Part B Eng. 2019, 172, 704–723. [Google Scholar] [CrossRef]
- Kaur, N.; Shahi, S.K.; Shahi, J.S.; Sandhu, S.; Sharma, R.; Singh, V. Comprehensive Review and Future Perspectives of Efficient N-doped, Fe-doped and (N,Fe)-co-doped Titania as Visible Light Active Photocatalysts. Vacuum 2020, 178, 109429. [Google Scholar] [CrossRef]
- Samokhvalov, A. Hydrogen by Photocatalysis with Nitrogen Codoped Titanium Dioxide. Renew. Sustain. Energ. Rev. 2017, 72, 981–1000. [Google Scholar] [CrossRef]
- Ansari, S.A.; Khan, M.M.; Ansari, M.O.; Cho, M.H. Nitrogen-doped Titanium Dioxide (N-doped TiO2) for Visible Light Photocatalysis. New J. Chem. 2016, 40, 3000–3009. [Google Scholar] [CrossRef]
- Bakar, S.A.; Ribeiro, C. Nitrogen-doped Titanium Dioxide: An Overview of Material Design and Dimensionality Effect over Modern Applications. J. Photoch. Photobiol. C 2016, 27, 1–29. [Google Scholar] [CrossRef]
- Gomes, J.; Lincho, J.; Domingues, E.; Quinta-Ferreira, R.; Martins, R. N–TiO2 Photocatalysts: A Review of Their Characteristics and Capacity for Emerging Contaminants Removal. Water 2019, 11, 373. [Google Scholar] [CrossRef]
- Zhang, W.; Jia, B.; Wang, Q.; Dionysiou, D. Visible-light Sensitization of TiO2 Photocatalysts via Wet Chemical N-doping for the Degradation of Dissolved Organic Compounds in Wastewater Treatment: A Review. J. Nanopart. Res. 2015, 17. [Google Scholar] [CrossRef]
- Spadavecchia, F.; Ceotto, M.; Presti, L.L.; Aieta, C.; Biraghi, I.; Meroni, D.; Ardizzone, S.; Cappelletti, G. Second Generation Nitrogen Doped Titania Nanoparticles: A Comprehensive Electronic and Microstructural Picture. Chin. J. Chem. 2014, 32, 1195–1213. [Google Scholar] [CrossRef]
- Gomathi Devi, L.; Kavitha, R. Review on Modified N–TiO2 for Green Energy Applications under UV/Visible Light: Selected Results and Reaction Mechanisms. RSC Adv. 2014, 4, 28265–28299. [Google Scholar] [CrossRef]
- Dunnill, C.W.; Parkin, I.P. Nitrogen-Doped TiO2 thin Films: Photocatalytic Applications for Healthcare Environments. Dalton Trans. 2011, 40, 1635–1640. [Google Scholar] [CrossRef] [PubMed]
- Thompson, T.L.; Yates, J.T., Jr. TiO2-Based Photocatalysis: Surface Defects, Oxygen and Charge Transfer. Top. Catal. 2005, 35, 197–210. [Google Scholar] [CrossRef]
- Lamo, M.P.B.; Nowotny, J. Water Purification Using Solar Energy: Effect of Sulphur on Photocatalytic Properties of TiO2. Energy Mater. 2009, 4, 150–158. [Google Scholar] [CrossRef]
- Shi, Z.-J.; Ma, M.-G.; Zhu, J.-F. Recent Development of Photocatalysts Containing Carbon Species: A Review. Catalysts 2018, 9, 20. [Google Scholar] [CrossRef]
- Mestre, A.S.; Carvalho, A.P. Photocatalytic Degradation of Pharmaceuticals Carbamazepine, Diclofenac, and Sulfamethoxazole by Semiconductor and Carbon Materials: A Review. Molecules 2019, 24, 3702. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, G.; Zhang, Z. TiO2-Based Catalysts for Photocatalytic Reduction of Aqueous Oxyanions: State-of-the-Art and Future Prospects. Environ. Int. 2020, 136, 105453. [Google Scholar] [CrossRef]
- Sushma, C.; Kumar, S.G. C–N–S Tridoping into TiO2 matrix for Photocatalytic Applications: Observations, Speculations and Contradictions in the Codoping Process. Inorg. Chem. Front. 2017, 4, 1250–1267. [Google Scholar] [CrossRef]
- Linsebigler, A.L.; Lu, G.; Yates, J.T. Photocatalysis on TiO2 Surfaces: Principles, Mechanisms, and Selected Results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Banerjee, S.; Pillai, S.C.; Falaras, P.; O’Shea, K.E.; Byrne, J.A.; Dionysiou, D.D. New Insights into the Mechanism of Visible Light Photocatalysis. J. Phys. Chem. Lett. 2014, 5, 2543–2554. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Zhang, X.; Tryk, D. TiO2 Photocatalysis and Related Surface Phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium Dioxide Photocatalysis. J. Photoch. Photobiol. C 2000, 1, 1–21. [Google Scholar] [CrossRef]
- Ahmed, S.; Rasul, M.G.; Brown, R.; Hashib, M.A. Influence of Parameters on the Heterogeneous Photocatalytic Degradation of Pesticides and Phenolic Contaminants in Wastewater: A Short Review. J. Environ. Manag. 2011, 92, 311–330. [Google Scholar] [CrossRef]
- Ramanathan, R.; Bansal, V. Ionic Liquid Mediated Synthesis of Nitrogen, Carbon and Fluorine-codoped Rutile TiO2 Nanorods for Improved UV and Visible Light Photocatalysis. RSC Adv. 2015, 5, 1424–1429. [Google Scholar] [CrossRef]
- Chen, M.; Chu, J.-W. NOx Photocatalytic Degradation on Active Concrete Road Surface—From Experiment to Real-Scale Application. J. Clean. Prod. 2011, 19, 1266–1272. [Google Scholar] [CrossRef]
- Su, T.; Shao, Q.; Qin, Z.; Guo, Z.; Wu, Z. Role of Interfaces in Two-Dimensional Photocatalyst for Water Splitting. ACS Catal. 2018, 8, 2253–2276. [Google Scholar] [CrossRef]
- Razzaq, A.; Sinhamahapatra, T.-H.; Kang, C.A.; Grimes, J.-S.; Yu, S.-I. Efficient Solar Light Photoreduction of CO2 to Hydrocarbon Fuels via Magnesiothermally Reduced TiO2 Photocatalyst. Appl. Catal. B-Environ. 2017, 215, 28–35. [Google Scholar] [CrossRef]
- Spadaro, L.; Arena, F.; Negro, P.; Palella, A. Sunfuels from CO2 Exhaust Emissions: Insights Into the Role of Photoreactor Configuration by the Study in Laboratory and Industrial Environment. J. CO2 Util. 2018, 26, 445–453. [Google Scholar] [CrossRef]
- Tahir, B.; Tahir, M.; Amin, N.S. Gold-Indium Modified TiO2 Nanocatalysts for Photocatalytic CO2 Reduction with H2 as Reductant in a Monolith Photoreactor. Appl. Surf. Sci. 2015, 338, 1–14. [Google Scholar] [CrossRef]
- Hu, B.; Guild, C.; Suib, S.L. Thermal, Electrochemical, and Photochemical Conversion of CO2 to Fuels and Value-Added Products. J. CO2 Util. 2013, 1, 18–27. [Google Scholar] [CrossRef]
- Tahir, M.; Amin, N.S. Advances in Visible Light Responsive Titanium Oxide-Based Photocatalysts for CO2 Conversion to Hydrocarbon Fuels. Energ. Convers. Manag. 2013, 76, 194–214. [Google Scholar] [CrossRef]
- Spadaro, L.; Arena, F.; Palella, A. Which Future Route in the Methanol Synthesis? Photocatalytic Reduction of CO2, the New Challenge in the Solar Energy Exploitation. In Methanol: Science and Engineering; Dalena, F., Basile, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar] [CrossRef]
- Abdullah, H.; Khan, M.M.; Ong, H.R.; Yaakob, Z. Modified TiO2 Photocatalyst for CO2 Photocatalytic Reduction: An Overview. J. CO2 Util. 2017, 22, 15–32. [Google Scholar] [CrossRef]
- Carp, O.; Huisman, C.L.; Reller, A. Photoinduced Reactivity of Titanium Dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Kočí, K.; Obalová, L.; Matějová, L.; Plachá, D.; Lacný, Z.; Jirkovský, J.; Šolcová, O. Effect of TiO2 Particle Size on the Photocatalytic Reduction of CO2. Appl. Catal. B-Environ. 2009, 89, 494–502. [Google Scholar] [CrossRef]
- Adekoya, D.; Tahir, M.; Amin, N.A. Recent Trends in Photocatalytic Materials for Reduction of Carbon Dioxide to Methanol. Renew. Sustain. Energy Rev. 2019, 116, 109389. [Google Scholar] [CrossRef]
- Wang, P.; Yin, G.; Bi, Q.; Huang, X.; Du, X.; Zhao, W.; Huang, F.-Q. Efficient Photocatalytic Reduction of CO2 Using Carbon-Doped Amorphous Titanium Oxide. Chem. Cat. Chem. 2018, 10, 3854–3861. [Google Scholar] [CrossRef]
- Akple, M.S.; Low, J.; Qin, Z.; Wageh, S.; Al-Ghamdi, A.A.; Yu, J.; Liu, S. Nitrogen-Doped TiO2 Microsheets with Enhanced Visible Light Photocatalytic Activity for CO2 Reduction. Chin. J. Catal. 2015, 36, 2127–2134. [Google Scholar] [CrossRef]
- Habisreutinger, S.N.; Schmidt-Mende, L.; Stolarczyk, J.K. Photocatalytic Reduction of CO2 on TiO2 and Other Semiconductors. Angew. Chem. Int. Ed. 2013, 52, 7372–7408. [Google Scholar] [CrossRef]
- Friedmann, D.; Hakki, A.; Kim, H.; Choi, W.; Bahnemann, D. Heterogeneous Photocatalytic Organic Synthesis: State-of-the-Art and Future Perspectives. Green Chem. 2016, 18, 5391–5411. [Google Scholar] [CrossRef]
- Qiu, H.; Fang, S.; Huang, G.; Bi, J. A Novel Application of In2S3 for Visible-Light-Driven Photocatalytic Inactivation of Bacteria: Kinetics, Stability, Toxicity and Mechanism. Environ. Res. 2020, 190, 110018. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Shen, S.; Zhou, W.; Tang, J.; Ding, H.; Wang, J.; Chen, L.; Au, C.-T.; Yin, S.-F. Recent Progress in Photocatalytic Hydrogen Evolution. Acta Phys. Chim. Sin. 2020, 36, 1–9. [Google Scholar] [CrossRef]
- Sakar, M.; Mithun Prakash, R.; Do, T.-O. Insights into the TiO2-Based Photocatalytic Systems and Their Mechanisms. Catalysts 2019, 9, 680. [Google Scholar] [CrossRef]
- Fang, Y.; Zheng, Y.; Fang, T.; Chen, Y.; Zhu, Y.; Liang, Q.; Sheng, H.; Li, Z.; Chen, C.; Wang, X. Photocatalysis: An Overview of Recent Developments and Technological Advancements. Sci. China Chem. 2019, 63, 149–181. [Google Scholar] [CrossRef]
- Matos, J.; Miralles-Cuevas, S.; Ruíz-Delgado, A.; Oller, I.; Malato, S. Development of TiO2-C Photocatalysts for Solar Treatment of Polluted Water. Carbon 2017, 122, 361–373. [Google Scholar] [CrossRef]
- Shi, J.-W.; Wang, Z.; He, C.; Li, G.; Niu, C. Carbon-Doped Titania Flakes with an Octahedral Bipyramid Skeleton Structure for the Visible-Light Photocatalytic Mineralization of Ciprofloxacin. RSC Adv. 2015, 5, 98361–98365. [Google Scholar] [CrossRef]
- Shi, J.-W.; Liu, C.; He, C.; Li, J.; Xie, C.; Yang, S.; Chen, J.-W.; Li, S.; Niu, C. Carbon-doped Titania Nanoplates with Exposed {001} Facets: Facile Synthesis, Characterization and Visible-Light Photocatalytic Performance. RSC Adv. 2015, 5, 17667–17675. [Google Scholar] [CrossRef]
- Chauhan, A.; Sharma, M.; Kumar, S.; Thirumalai, S.; Kumar, R.V.; Vaish, R. TiO2@C Core@shell Nanocomposites: A Single Precursor Synthesis of Photocatalyst for Efficient Solar Water Treatment. J. Hazard. Mater. 2020, 381, 120883. [Google Scholar] [CrossRef]
- Kuang, L.; Zhang, W. Enhanced Hydrogen Production by Carbon-doped TiO2 Decorated with Reduced Graphene Oxide (RGO) under Visible Light Irradiation. RSC Adv. 2016, 6, 2479–2488. [Google Scholar] [CrossRef]
- Jia, G.; Wang, Y.; Cui, X.; Zheng, W. Highly Carbon-doped TiO2 Derived from MXene Boosting the Photocatalytic Hydrogen Evolution. ACS Sustain. Chem. Eng. 2018, 6, 13480–13486. [Google Scholar] [CrossRef]
- Shim, J.; Seo, Y.-S.; Oh, B.-T.; Cho, M. Microbial Inactivation Kinetics and Mechanisms of Carbon-doped TiO2 (C-TiO2) under Visible Light. J. Hazard. Mater. 2016, 306, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Marszewski, M.; Marszewska, J.; Pylypenko, S.; Jaroniec, M. Synthesis of Porous Crystalline Doped Titania Photocatalysts Using Modified Precursor Strategy. Chem. Mater. 2016, 28, 7878–7888. [Google Scholar] [CrossRef]
- Yuan, Y.; Qian, X.; Han, H.; Chen, Y. Synthesis of Carbon Modified TiO2 Photocatalysts with High Photocatalytic Activity by a Facile Calcinations Assisted Solvothermal Method. J. Mater. Sci. Mater. Electron. 2017, 28, 10028–10034. [Google Scholar] [CrossRef]
- Shaban, Y.A.; Fallata, H.M. Sunlight-Induced Photocatalytic Degradation of Acetaminophen over Efficient Carbon Doped TiO2 (CTiO2) Nanoparticles. Res. Chem. Intermed. 2019, 45, 2529–2547. [Google Scholar] [CrossRef]
- de Luna, M.D.G.; Lin, J.C.-T.; Gotostos, M.J.N.; Lu, M.-C. Photocatalytic Oxidation of Acetaminophen Using Carbon Self-doped Titanium Dioxide. Sustain. Environ. Res. 2016, 26, 161–167. [Google Scholar] [CrossRef]
- Purbia, R.; Borah, R.; Paria, S. Carbon-Doped Mesoporous Anatase TiO2 Multi-Tubes Nanostructures for Highly Improved Visible Light Photocatalytic Activity. Inorg. Chem. 2017, 56, 10107–10116. [Google Scholar] [CrossRef]
- Nawawi, W.I.; Ani, A.Y.; Ishak, M.A.M.; Ramli, A.; Azami, M.S.; Zaid, F.; Bakar, F.; Zaharudin, R. Modification and Characterization of C-doped TiO2 Photocatalyst for Photodegradation of Reactive Red (RR4). Desalin. Water Treat. 2018, 113, 254–261. [Google Scholar] [CrossRef]
- Rajamanickam, A.T.; Thirunavukkarasu, P.; Dhanakodi, K. A Simple Route to Synthesis of Carbon Doped TiO2 Nanostructured Thin Film for Enhanced Visible-Light Photocatalytic Activity. J. Mater. Sci. Mater. Electron. 2015, 26, 4038–4045. [Google Scholar] [CrossRef]
- Huang, Y.; Ho, W.; Lee, S.; Zhang, L.; Li, G.; Yu, J.C. Effect of Carbon Doping on the Mesoporous Structure of Nanocrystalline Titanium Dioxide and Its Solar-Light-Driven Photocatalytic Degradation of NOx. Langmuir 2008, 24, 3510–3516. [Google Scholar] [CrossRef]
- Ding, Y.; Nagpal, P. Standalone Anion- and Co-doped Titanium Dioxide Nanotubes for Photocatalytic and Photoelectrochemical Solar-to-Fuel Conversion. Nanoscale 2016, 8, 17496–17505. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Huang, Y.; Liu, T. Enhanced Visible-Light Photocatalytic Performance of Electrospun Carbon-doped TiO2/Halloysite Nanotube Hybrid Nanofibers. J. Colloid Interf. Sci. 2015, 439, 62–68. [Google Scholar] [CrossRef] [PubMed]
- An, N.; Ma, Y.; Liu, J.; Ma, H.; Yang, J.; Zhang, Q. Enhanced Visible-light Photocatalytic Oxidation Capability of Carbon-doped TiO2 via Coupling with Fly Ash. Chin. J. Catal. 2018, 39, 1890–1900. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, G.-F.; Li, D.; Zhou, B.-X.; Chang, S.; Pan, A.; Huang, W.-Q. Facile Route to Fabricate Carbon-doped TiO2 Nanoparticles and Its Mechanism of Enhanced Visible Light Photocatalytic Activity. Appl. Phys. A 2016, 122. [Google Scholar] [CrossRef]
- Qiu, B.; Zhong, C.; Xing, M.; Zhang, J. Facile Preparation of C-modified TiO2 Supported on MCF for High Visible-Light-Driven Photocatalysis. RSC Adv. 2015, 5, 17802–17808. [Google Scholar] [CrossRef]
- Birben, N.C.; Uyguner-Demirel, C.S.; Sen-Kavurmaci, S.; Gürkan, Y.Y.; Türkten, N.; Kılıç, M.; Çınar, Z.; Bekbolet, M. Photocatalytic Performance of Anion Doped TiO2 on the Degradation of Complex Organic Matrix. J. Adv. Oxid. Technol. 2016, 19. [Google Scholar] [CrossRef]
- Choi, Y.; Umebayashi, T.; Yoshikawa, M. Fabrication and Characterization of C-Doped Anatase TiO2 photocatalysts. J. Mater. Sci. 2004, 39, 1837–1839. [Google Scholar] [CrossRef]
- Ananpattarachai, J.; Seraphin, S.; Kajitvichyanukul, P. Formation of Hydroxyl Radicals and Kinetic Study of 2-Chlorophenol Photocatalytic Oxidation Using C-doped TiO2, N-doped TiO2, and C,N Co-doped TiO2 under Visible Light. Environ. Sci. Pollut. Res. 2015, 23, 3884–3896. [Google Scholar] [CrossRef]
- Tijani, J.O.; Mouele, M.E.S.; Tottito, T.C.; Fatoba, O.O.; Petrik, L.F. Degradation of 2-Nitrophenol by Dielectric Barrier Discharge System: The Influence of Carbon Doped TiO2 Photocatalyst Supported on Stainless Steel Mesh. Plasma Chem. Plasma P 2017, 37, 1343–1373. [Google Scholar] [CrossRef]
- Tijani, J.O.; Fatoba, O.O.; Totito, T.C.; Roos, W.D.; Petrik, L.F. Synthesis and Characterization of Carbon Doped TiO2 Photocatalysts Supported on Stainless Steel Mesh by Sol-Gel Method. Carbon Lett. 2017, 22, 48–59. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Z.; Chen, J.; Cheng, L.; Chang, J.; Sheng, W.; Hu, C.; Cao, S. C-doped Hollow TiO2 Spheres: In Situ Synthesis, Controlled Shell Thickness, and Superior Visible-Light Photocatalytic Activity. Appl. Catal. B-Environ. 2015, 165, 715–722. [Google Scholar] [CrossRef]
- Li, J.; Gao, L.; Gan, W. Bioinspired C/TiO2 Photocatalyst for Rhodamine B Degradation under Visible Light Irradiation. Front. Agr. Sci. Eng. 2017, 4, 459. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Salleh, W.N.W.; Jaafar, J.; Mohd Hir, Z.A.; Rosmi, M.S.; Mutalib, M.A.; Ismail, A.F.; Tanemura, M. Regenerated Cellulose Membrane as Bio-Template for in-Situ Growth of Visible-light Driven C-modified Mesoporous Titania. Carbohyd. Polym. 2016, 146, 166–173. [Google Scholar] [CrossRef]
- Mani, A.D.; Reddy, P.M.K.; Srinivaas, M.; Ghosal, P.; Xanthopoulos, N.; Subrahmanyam, C. Facile Synthesis of Efficient Visible Active C-doped TiO2 Nanomaterials with High Surface Area for the Simultaneous Removal of Phenol and Cr(VI). Mater. Res. Bull. 2015, 61, 391–399. [Google Scholar] [CrossRef]
- Yang, Y.; Ni, D.; Yao, Y.; Zhong, Y.; Ma, Y.; Yao, J. High Photocatalytic Activity of Carbon Doped TiO2 Prepared by Fast Combustion of Organic Capping Ligands. RSC Adv. 2015, 5, 93635–93643. [Google Scholar] [CrossRef]
- Habibi, S.; Jamshidi, M. Sol–Gel Synthesis of Carbon-doped TiO2 Nanoparticles Based on Microcrystalline Cellulose for Efficient Photocatalytic Degradation of Methylene Blue under Visible Light. Environ. Technol. 2019, 41, 3233–3247. [Google Scholar] [CrossRef]
- Payormhorm, J.; Idem, R. Synthesis of C-Doped TiO2 by Sol-Microwave Method for Photocatalytic Conversion of Glycerol to Value-Added Chemicals under Visible Light. Appl. Catal. A Gen. 2020, 590, 117362. [Google Scholar] [CrossRef]
- Ouyang, W.; Santiago, A.R.P.; Cerdán-Gómez, K.; Luque, R. Nanoparticles within Functional Frameworks and Their Applications in Photo(Electro)Catalysis. In Photoactive Inorganic Nanoparticles; Elsevier: Amsterdam, The Netherlands, 2019; pp. 109–138. [Google Scholar] [CrossRef]
- Song, L.; Jing, W.; Chen, J.; Zhang, S.; Zhu, Y.; Xiong, J. High Reusability and Durability of Carbon-doped TiO2/Carbon Nanofibrous Film as Visible-Light-Driven Photocatalyst. J. Mater. Sci. 2018, 54, 3795–3804. [Google Scholar] [CrossRef]
- Qian, H.; Hou, Q.; Duan, E.; Niu, J.; Nie, Y.; Bai, C.; Bai, X.; Ju, M. Honeycombed Au@C-TiO2-X Catalysts for Enhanced Photocatalytic Mineralization of Acid Red 3R under Visible Light. J. Hazard. Mater. 2020, 391, 122246. [Google Scholar] [CrossRef]
- El Nemr, A.; Helmy, E.T.; Gomaa, E.A.; Eldafrawy, S.; Mousa, M. Photocatalytic and Biological Activities of Undoped and Doped TiO2 Prepared by Green Method for Water Treatment. J. Environ. Chem. Eng. 2019, 7, 103385. [Google Scholar] [CrossRef]
- Xie, C.; Yang, S.; Shi, J.; Niu, C. Highly Crystallized C-doped Mesoporous Anatase TiO2 with Visible Light Photocatalytic Activity. Catalysts 2016, 6, 117. [Google Scholar] [CrossRef]
- Xie, C.; Yang, S.; Li, B.; Wang, H.; Shi, J.-W.; Li, G.; Niu, C. C-Doped Mesoporous Anatase TiO2 Comprising 10 nm Crystallites. J. ColloidInterf. Sci. 2016, 476, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Jiang, X.; Zhang, Y.; Fu, Q.; Pan, C. Preparation of High-concentration Substitutional Carbon-doped TiO2 Film via a Two-step Method for High-performance Photocatalysis. RSC Adv. 2018, 8, 36691–36696. [Google Scholar] [CrossRef]
- Shaban, Y.A. Solar Light-Induced Photodegradation of Chrysene in Seawater in the Presence of Carbon-modified n-TiO2 Nanoparticles. Arab. J. Chem. 2019, 12, 652–663. [Google Scholar] [CrossRef]
- Di Valentin, C.; Pacchioni, G.; Selloni, A. Theory of Carbon Doping of Titanium Dioxide. Chem. Mater. 2005, 17, 6656–6665. [Google Scholar] [CrossRef]
- Saharudin, K.A.; Sreekantan, S.; Lai, C.W. Fabrication and Photocatalysis of Nanotubular C-doped TiO2 Arrays: Impact of Annealing Atmosphere on the Degradation Efficiency of Methyl Orange. Mat. Sci. Semicon. Proc. 2014, 20, 1–6. [Google Scholar] [CrossRef]
- Shao, J.; Sheng, W.; Wang, M.; Li, S.; Chen, J.; Zhang, Y.; Cao, S. In Situ Synthesis of Carbon-doped TiO2 Single-Crystal Nanorods with a Remarkably Photocatalytic Efficiency. Appl. Catal. B-Environ. 2017, 209, 311–319. [Google Scholar] [CrossRef]
- Ohtani, B. Photocatalysis A to Z—What We Know and What We Do Not Know in a Scientific Sense. J. Photoch. Photobiol. C. 2010, 11, 157–178. [Google Scholar] [CrossRef]
- Dong, F.; Guo, S.; Wang, H.; Li, X.; Wu, Z. Enhancement of the Visible Light Photocatalytic Activity of C-Doped TiO2 Nanomaterials Prepared by a Green Synthetic Approach. J. Phys. Chem. C 2011, 115, 13285–13292. [Google Scholar] [CrossRef]
- Bergamonti, L.; Predieri, G.; Paz, Y.; Fornasini, L.; Lottici, P.P.; Bondioli, F. Enhanced Self-cleaning Properties of N-doped TiO2 Coating for Cultural Heritage. Microchem. J. 2017, 133, 1–12. [Google Scholar] [CrossRef]
- Preethi, L.K.; Antony, R.P.; Mathews, T.; Walczak, L.; Gopinath, C.S. A Study on Doped Heterojunctions in TiO2 Nanotubes: An Efficient Photocatalyst for Solar Water Splitting. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Calisir, M.D.; Gungor, M.; Demir, A.; Kilic, A.; Khan, M.M. Nitrogen-doped TiO2 Fibers for Visible-Light-Induced Photocatalytic Activities. Ceram. Int. 2020, 46, 16743–16753. [Google Scholar] [CrossRef]
- Liu, T.; Chen, W.; Liu, X.; Zhu, J.; Lu, L. Well-Dispersed Ultrafine Nitrogen-doped TiO2 with Polyvinylpyrrolidone (PVP) Acted as N-source and Stabilizer for Water Splitting. J. Energy Chem. 2016, 25, 1–9. [Google Scholar] [CrossRef]
- Wang, T.; Yan, X.; Zhao, S.; Lin, B.; Xue, C.; Yang, G.; Ding, S.; Yang, B.; Ma, C.; Yang, G.; et al. A Facile One-Step Synthesis of Three-Dimensionally Ordered Macroporous N-Doped TiO2 with Ethanediamine as the Nitrogen Source. J. Mater. Chem. A 2014, 2, 15611–15619. [Google Scholar] [CrossRef]
- Sirivallop, A.; Areerob, T.; Chiarakorn, S. Enhanced Visible Light Photocatalytic Activity of N and Ag Doped and Co-doped TiO2 Synthesized by Using an In-Situ Solvothermal Method for Gas Phase Ammonia Removal. Catalysts 2020, 10, 251. [Google Scholar] [CrossRef]
- Breeson, A.C.; Sankar, G.; Goh, G.K.L.; Palgrave, R.G. Rutile to Anatase Phase Transition Induced by N Doping in Highly Oriented TiO2 Films. Phys. Chem. Chem. Phys. 2016, 18, 24722–24728. [Google Scholar] [CrossRef]
- Siuzdak, K.; Szkoda, M.; Sawczak, M.; Lisowska-Oleksiak, A. Novel Nitrogen Precursors for Electrochemically Driven Doping of Titania Nanotubes Exhibiting Enhanced Photoactivity. New J. Chem. 2015, 39, 2741–2751. [Google Scholar] [CrossRef]
- May Ix, L.A.; Estrella González, A.; Cipagauta-Díaz, S.; Gómez, R. Effective Electron–Hole Separation over N-doped TiO2 Materials for Improved Photocatalytic Reduction of 4-nitrophenol Using Visible Light. J. Chem. Technol. Biotechnol. 2020. [Google Scholar] [CrossRef]
- Cheng, Z.-L.; Han, S. Preparation and Photoelectrocatalytic Performance of N-doped TiO2/NaY Zeolite Membrane Composite Electrode Material. Water Sci. Technol. 2015, 73, 486–492. [Google Scholar] [CrossRef]
- Huang, L.; Fu, W.; Fu, X.; Zong, B.; Liu, H.; Bala, H.; Wang, X.; Sun, G.; Cao, J.; Zhang, Z. Facile and Large-Scale Preparation of N doped TiO2 Photocatalyst with High Visible Light Photocatalytic Activity. Mater. Lett. 2017, 209, 585–588. [Google Scholar] [CrossRef]
- Cheng, X.; Yu, X.; Xing, Z.; Yang, L. Synthesis and Characterization of N-doped TiO2 and Its Enhanced Visible-Light Photocatalytic Activity. Arab. J. Chem. 2016, 9, S1706–S1711. [Google Scholar] [CrossRef]
- Zeng, H.; Xie, J.; Xie, H.; Su, B.-L.; Wang, M.; Ping, H.; Wang, W.; Wang, H.; Fu, Z. Bioprocess-Inspired Synthesis of Hierarchically Porous Nitrogen-Doped TiO2 with High Visible-Light Photocatalytic Activity. J. Mater. Chem. A 2015, 3, 19588–19596. [Google Scholar] [CrossRef]
- Ariza-Tarazona, M.C.; Villarreal-Chiu, J.F.; Barbieri, V.; Siligardi, C.; Cedillo-González, E.I. New Strategy for Microplastic Degradation: Green Photocatalysis Using a Protein-Based Porous N-TiO2 Semiconductor. Ceram. Int. 2019, 45, 9618–9624. [Google Scholar] [CrossRef]
- Xu, X.; Song, W. Enhanced H2 Production Activity under Solar Irradiation over N-doped TiO2 Prepared Using Pyridine as a Precursor: A Typical Sample of N-doped TiO2 Series. Mater. Technol. 2016, 32, 52–63. [Google Scholar] [CrossRef]
- Zhou, X.; Peng, F.; Wang, H.; Yu, H.; Yang, J. Preparation of Nitrogen Doped TiO2 Photocatalyst by Oxidation of Titanium Nitride with H2O2. Mater. Res. Bull. 2011, 46, 840–844. [Google Scholar] [CrossRef]
- Huang, J.-G.; Zhao, X.-G.; Zheng, M.-Y.; Li, S.; Wang, Y.; Liu, X.-J. Preparation of N-Doped TiO2 by Oxidizing TiN and Its Application on Phenol Degradation. Water Sci. Technol. 2013, 68, 934–939. [Google Scholar] [CrossRef]
- Nguyen-Le, M.-T.; Lee, B.-K. Novel Fabrication of a Nitrogen-doped Mesoporous TiO2-Nanorod Titanate Heterojunction to Enhance the Photocatalytic Degradation of Dyes under Visible Light. RSC Adv. 2016, 6, 31347–31350. [Google Scholar] [CrossRef]
- Assayehegn, E.; Solaiappan, A.; Chebude, Y.; Alemayehu, E. Fabrication of Tunable Anatase/Rutile Heterojunction N/TiO2 Nanophotocatalyst for Enhanced Visible Light Degradation Activity. Appl. Surf. Sci. 2020, 515, 145966. [Google Scholar] [CrossRef]
- Assayehegn, E.; Solaiappan, A.; Chebudie, Y.; Alemayehu, E. Influence of Temperature on Preparing Mesoporous Mixed Phase N/TiO2 Nanocomposite with Enhanced Solar Light Photocatalytic Activity. Front. Mater. Sci. 2019, 13, 352–366. [Google Scholar] [CrossRef]
- Fang, Q.; Tang, J.; Zou, H.; Cai, T.; Deng, Q. Preparation of N-doped Mesoporous TiO2 Using Nitromethane as Nitrogen Source and Their High Photocatalytic Performance. Synth. React. Inorg. Met. Chem. 2015, 46, 766–774. [Google Scholar] [CrossRef]
- Lee, S.-H.; Yamasue, E.; Okumura, H.; Ishihara, K.N. Effect of Oxygen and Nitrogen Concentration of Nitrogen Doped TiOx Film as Photocatalyst Prepared by Reactive Sputtering. Appl. Catal. A-Gen. 2009, 371, 179–190. [Google Scholar] [CrossRef]
- Marques, J.; Gomes, T.D.; Forte, M.A.; Silva, R.F.; Tavares, C.J. A New Route for the Synthesis of Highly-Active N-doped TiO2 Nanoparticles for Visible Light Photocatalysis Using Urea as Nitrogen Precursor. Catal. Today 2019, 326, 36–45. [Google Scholar] [CrossRef]
- Pu, X.; Hu, Y.; Cui, S.; Cheng, L.; Jiao, Z. Preparation of N-doped and Oxygen-deficient TiO2 Microspheres via a Novel Electron Beam-Assisted Method. Solid State Sci. 2017, 70, 66–73. [Google Scholar] [CrossRef]
- Quesada-Cabrera, R.; Sotelo-Vázquez, C.; Quesada-González, M.; Melián, E.P.; Chadwick, N.; Parkin, I.P. On the Apparent Visible-Light and Enhanced UV-Light Photocatalytic Activity of Nitrogen-doped TiO2 Thin Films. J. Photoch. Photobiol. A. 2017, 333, 49–55. [Google Scholar] [CrossRef][Green Version]
- Karim, A.V.; Shriwastav, A. Degradation of Ciprofloxacin Using Photo, Sono, and Sonophotocatalytic Oxidation with Visible Light and Low-Frequency Ultrasound: Degradation Kinetics and Pathways. Chem. Eng. J. 2020, 392, 124853. [Google Scholar] [CrossRef]
- Huang, W.; Cheng, H.; Feng, J.; Shi, Z.; Bai, D.; Li, L. Synthesis of Highly Water-Dispersible N-doped Anatase Titania Based on Low Temperature Solvent-Thermal Method. Arab. J. Chem. 2018, 11, 871–879. [Google Scholar] [CrossRef]
- Delavari, S.; Amin, N.A.S.; Ghaedi, M. Photocatalytic Conversion and Kinetic Study of CO2 and CH4 over Nitrogen-doped Titania Nanotube Arrays. J. Clean. Prod. 2016, 111, 143–154. [Google Scholar] [CrossRef]
- Islam, S.Z.; Rankin, S.E. Hydrazine-Based Synergistic Ti(III)/N Doping of Surfactant-Templated TiO2 Thin Films for Enhanced Visible Light Photocatalysis. Mater. Chem. Phys. 2016, 182, 382–393. [Google Scholar] [CrossRef]
- Pikuda, O.; Garlisi, C.; Scandura, G.; Palmisano, G. Micro-Mesoporous N-doped Brookite-Rutile TiO2 as Efficient Catalysts for Water Remediation under UV-free Visible LED Radiation. J. Catal. 2017, 346, 109–116. [Google Scholar] [CrossRef]
- Fiorenza, R.; Di Mauro, A.; Cantarella, M.; Gulino, A.; Spitaleri, L.; Privitera, V.; Impellizzeri, G. Molecularly Imprinted N-doped TiO2 Photocatalysts for the Selective Degradation of o-Phenylphenol Fungicide from Water. Mat. Sci. Semicon. Proc. 2020, 112, 105019. [Google Scholar] [CrossRef]
- Ananpattarachai, J.; Kajitvichyanukul, P. Photocatalytic Degradation of p,p′-DDT under UV and Visible Light Using Interstitial N-doped TiO2. J. Environ. Sci. Heal. B 2015, 50, 247–260. [Google Scholar] [CrossRef]
- Ananpattarachai, J.; Boonto, Y.; Kajitvichyanukul, P. Visible Light Photocatalytic Antibacterial Activity of Ni-doped and N-doped TiO2 on Staphylococcus aureus and Escherichia coli Bacteria. Environ. Sci. Pollut. Res. 2015, 23, 4111–4119. [Google Scholar] [CrossRef]
- Nursam, N.M.; Tan, J.Z.Y.; Wang, X.; Li, W.; Xia, F.; Caruso, R.A. Mesoporous Nitrogen-Modified Titania with Enhanced Dye Adsorption Capacity and Visible Light Photocatalytic Activity. Chem. Sel. 2016, 1, 4868–4878. [Google Scholar] [CrossRef]
- Szkoda, M.; Siuzdak, K.; Lisowska-Oleksiak, A. Non-Metal Doped TiO2 Nanotube Arrays for High Efficiency Photocatalytic Decomposition of Organic Species in Water. Phys. E 2016, 84, 141–145. [Google Scholar] [CrossRef]
- Pablos, C.; Marugán, J.; van Grieken, R.; Dunlop, P.; Hamilton, J.; Dionysiou, D.; Byrne, J. Electrochemical Enhancement of Photocatalytic Disinfection on Aligned TiO2 and Nitrogen doped TiO2 Nanotubes. Molecules 2017, 22, 704. [Google Scholar] [CrossRef] [PubMed]
- Mehdizadeh, P.; Tavangar, Z.; Shabani, N.; Hamadanian, M. Visible Light Activity of Nitrogen-doped TiO2 by Sol-Gel Method Using Various Nitrogen Sources. J. Nanostruct. 2020, 10, 307–316. [Google Scholar] [CrossRef]
- Kamaludin, R.; Mohamad Puad, A.S.; Othman, M.H.D.; Kadir, S.H.S.A.; Harun, Z. Incorporation of N-doped TiO2 into Dual Layer Hollow Fiber (DLHF) Membrane for Visible Light-Driven Photocatalytic Removal of Reactive Black 5. Polym. Test. 2019, 78, 105939. [Google Scholar] [CrossRef]
- Ramchiary, A.; Samdarshi, S.K. Hydrogenation Based Disorder-Engineered Visible Active N-doped Mixed Phase Titania. Sol. Energ. Mat. Sol. C 2015, 134, 381–388. [Google Scholar] [CrossRef]
- Tobaldi, D.M.; Pullar, R.C.; Gualtieri, A.F.; Otero-Irurueta, G.; Singh, M.K.; Seabra, M.P.; Labrincha, J.A. Nitrogen-modified Nano-titania: True Phase Composition, Microstructure and Visible-Light Induced Photocatalytic NO Abatement. J. Solid State Chem. 2015, 231, 87–100. [Google Scholar] [CrossRef]
- Choi, A.Y.; Han, C.-H. FTIR Investigation of Sonochemically N-doped TiO2 Nano Powder. J. Nanosci. Nanotechnol. 2016, 16, 11109–11112. [Google Scholar] [CrossRef]
- Esteban Florez, F.L.; Hiers, R.D.; Larson, P.; Johnson, M.; O’Rear, E.; Rondinone, A.J.; Khajotia, S.S. Antibacterial Dental Adhesive Resins Containing Nitrogen-doped Titanium Dioxide Nanoparticles. Mater. Sci. Eng. C 2018, 93, 931–943. [Google Scholar] [CrossRef] [PubMed]
- Khore, S.K.; Tellabati, N.V.; Apte, S.K.; Naik, S.D.; Ojha, P.; Kale, B.B.; Sonawane, R.S. Green Sol–Gel Route for Selective Growth of 1D Rutile N–TiO2: A Highly Active Photocatalyst for H2 Generation and Environmental Remediation under Natural Sunlight. RSC Adv. 2017, 7, 33029–33042. [Google Scholar] [CrossRef]
- Li, X.; Zhang, G.; Wang, X.; Liu, W.; Yu, K.; Liang, C. Facile Synthesis of Nitrogen-doped Titanium Dioxide with Enhanced Photocatalytic Properties. Mater. Res. Express 2019, 6, 115019. [Google Scholar] [CrossRef]
- Ivanov, S.; Barylyak, A.; Besaha, K.; Bund, A.; Bobitski, Y.; Wojnarowska-Nowak, R.; Yaremchuk, I.; Kus-Liśkiewicz, M. Synthesis, Characterization, and Photocatalytic Properties of Sulfur- and Carbon-Codoped TiO2 Nanoparticles. Nanoscale Res. Lett. 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Khalyavka, T.; Bondarenko, M.; Shcherban, N.; Petrik, I.; Melnyk, A. Effect of the C and S Additives on Structural, Optical, and Photocatalytic Properties of TiO2. Appl. Nanosci. 2018, 9, 695–702. [Google Scholar] [CrossRef]
- Zou, X.; Shang, X.; Wang, R.; Zhang, J.; Pan, Q.; Guo, Y. Sulfur and Carbon Co-doped TiO2 Composite Fabricated by Lignosulphonate and Its Suitability for Removal of Cadmium. Clean Soil Air Water 2019, 47, 1800446. [Google Scholar] [CrossRef]
- Minnekhanov, A.A.; Le, N.T.; Konstantinova, E.A.; Kashkarov, P.K. Influence of Defects on Photoconductivity and Photocatalytic Activity of Nitrogen-doped Titania. Appl. Magn. Reson. 2017, 48, 335–345. [Google Scholar] [CrossRef]
- Chen, Y.; Luo, X.; Luo, Y.; Xu, P.; He, J.; Jiang, L.; Li, J.; Yan, Z.; Wang, J. Efficient Charge Carrier Separation in L-Alanine Acids Derived N-TiO2 Nanospheres: The Role of Oxygen Vacancies in Tetrahedral Ti4+ Sites. Nanomaterials 2019, 9, 698. [Google Scholar] [CrossRef]
- Batra, R.; Sharotri, N.; Sud, D. Ultrasound Assisted Synthesis of Visible Light Responsive Nitrogen Doped TiO2 Nano Photocatalyst. AMR 2013, 856, 22–24. [Google Scholar] [CrossRef]
- Wang, X.-K.; Wang, C.; Guo, W.-L.; Wang, J.-G. A Novel Single-Step Synthesis of N-Doped TiO2 via a Sonochemical Method. Mater. Res. Bull. 2011, 46, 2041–2044. [Google Scholar] [CrossRef]
- Wang, H.; Gao, X.; Duan, G.; Yang, X.; Liu, X. Facile Preparation of Anatase–Brookite–Rutile Mixed-Phase N-doped TiO2 with High Visible-Light Photocatalytic Activity. J. Environ. Chem. Eng. 2015, 3, 603–608. [Google Scholar] [CrossRef]
- Huang, W.C.; Ting, J.-M. Novel Nitrogen-Doped Anatase TiO2 Mesoporous Bead Photocatalysts for Enhanced Visible Light Response. Ceram. Int. 2017, 43, 9992–9997. [Google Scholar] [CrossRef]
- Mahy, J.; Cerfontaine, V.; Poelman, D.; Devred, F.; Gaigneaux, E.; Heinrichs, B.; Lambert, S. Highly Efficient Low-Temperature N-doped TiO2 Catalysts for Visible Light Photocatalytic Applications. Materials 2018, 11, 584. [Google Scholar] [CrossRef] [PubMed]
- Makropoulou, T.; Panagiotopoulou, P.; Venieri, D. N-doped TiO2 Photocatalysts for Bacterial Inactivation in Water. J. Chem. Technol. Biotechnol. 2018, 93, 2518–2526. [Google Scholar] [CrossRef]
- Chen, X.; Kuo, D.-H.; Lu, D. N-doped Mesoporous TiO2 Nanoparticles Synthesized by Using Biological Renewable Nanocrystalline Cellulose as Template for the Degradation of Pollutants under Visible and Sun Light. Chem. Eng. J. 2016, 295, 192–200. [Google Scholar] [CrossRef]
- Reddy, P.A.K.; Reddy, P.V.L.; Kim, K.-H.; Kumar, M.K.; Manvitha, C.; Shim, J.-J. Novel Approach for the Synthesis of Nitrogen-Doped Titania with Variable Phase Composition and Enhanced Production of Hydrogen under Solar Irradiation. J. Ind. Eng. Chem. 2017, 53, 253–260. [Google Scholar] [CrossRef]
- Li, X.; Liu, P.; Mao, Y.; Xing, M.; Zhang, J. Preparation of Homogeneous Nitrogen-Doped Mesoporous TiO2 Spheres with Enhanced Visible-light Photocatalysis. Appl. Catal. B-Environ. 2015, 164, 352–359. [Google Scholar] [CrossRef]
- Kowalska, K.; Maniakova, G.; Carotenuto, M.; Sacco, O.; Vaiano, V.; Lofrano, G.; Rizzo, L. Removal of Carbamazepine, Diclofenac and Trimethoprim by Solar Driven Advanced Oxidation Processes in a Compound Triangular Collector Based Reactor: A Comparison between Homogeneous and Heterogeneous Processes. Chemosphere 2020, 238, 124665. [Google Scholar] [CrossRef] [PubMed]
- Singha, K.; Ghosh, S.C.; Panda, A.B. N-Doped Yellow TiO2 Hollow Sphere-Mediated Visible-Light-Driven Efficient Esterification of Alcohol and N -Hydroxyimides to Active Esters. Chem. Asian J. 2019, 14, 3205–3212. [Google Scholar] [CrossRef]
- Suwannaruang, T.; Kamonsuangkasem, K.; Kidkhunthod, P.; Chirawatkul, P.; Saiyasombat, C.; Chanlek, N.; Wantala, K. Influence of Nitrogen Content Levels on Structural Properties and Photocatalytic Activities of Nanorice-like N-doped TiO2 with Various Calcination Temperatures. Mater. Res. Bull. 2018, 105, 265–276. [Google Scholar] [CrossRef]
- Ibukun, O.; Jeong, H.K. Enhancement of Photocatalytic Activities of Nitrogen-doped Titanium Dioxide by Ambient Plasma. Chem. Phys. Lett. 2020, 744, 137234. [Google Scholar] [CrossRef]
- Xing, X.; Du, Z.; Zhuang, J.; Wang, D. Removal of Ciprofloxacin from Water by Nitrogen Doped TiO2 Immobilized on Glass Spheres: Rapid Screening of Degradation Products. J. Photoch. Photobiol. A. 2018, 359, 23–32. [Google Scholar] [CrossRef]
- Pandiangan, I.F.D.; Sutanto, H.; Nurhasanah, I. Effect of Annealing Temperature on Optical Properties and Photocatalytic Properties of TiO2:N 8% Thin Film for Rhodamine B Degradation. Mater. Res. Express 2018, 5, 086404. [Google Scholar] [CrossRef]
- Fernandes, E.; Contreras, S.; Medina, F.; Martins, R.C.; Gomes, J. N-doped Titanium Dioxide for Mixture of Parabens Degradation Based on Ozone Action and Toxicity Evaluation: Precursor of Nitrogen and Titanium Effect. Process Saf. Environ. 2020, 138, 80–89. [Google Scholar] [CrossRef]
- Kusano, D.; Emori, M.; Sakama, H. Influence of Electronic Structure on Visible Light Photocatalytic Activity of Nitrogen-doped TiO2. RSC Adv. 2017, 7, 1887–1898. [Google Scholar] [CrossRef]
- Konstantinova, E.A.; Zaitsev, V.B.; Minnekhanov, A.A.; Le, N.T.; Kashkarov, P.K. The Effect of Spin Center Parameters on the Photoactivity of Nanocrystalline Titanium Dioxide in the Visible Spectral Range. Crystallogr. Rep. 2020, 65, 130–137. [Google Scholar] [CrossRef]
- Than, L.D.; Luong, N.S.; Ngo, V.D.; Tien, N.M.; Dung, T.N.; Nghia, N.M.; Loc, N.T.; Thu, V.T.; Lam, T.D. Highly Visible Light Activity of Nitrogen Doped TiO2 Prepared by Sol–Gel Approach. J. Electron. Mater. 2016, 46, 158–166. [Google Scholar] [CrossRef]
- Fornasini, L.; Bergamonti, L.; Bondioli, F.; Bersani, D.; Lazzarini, L.; Paz, Y.; Lottici, P.P. Photocatalytic N-doped TiO2 for Self-cleaning of Limestones. Eur. Phys. J. Plus 2019, 134. [Google Scholar] [CrossRef]
- Jang, I.; Leong, H.J.; Noh, H.; Kang, T.; Kong, S.; Oh, S.-G. Preparation of N-functionalized TiO2 Particles Using One-Step Sol–Gel Method and Their Photocatalytic Activity. J. Ind. Eng. Chem. 2016, 37, 380–389. [Google Scholar] [CrossRef]
- Kamaludin, R.; Othman, M.H.D.; Kadir, S.H.S.A.; Ismail, A.F.; Rahman, M.A.; Jaafar, J. Visible-Light-Driven Photocatalytic N-doped TiO2 for Degradation of Bisphenol A (BPA) and Reactive Black 5 (RB5) Dye. Water Air Soil Pollut. 2018, 229. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Salleh, W.N.W.; Jaafar, J.; Ismail, A.F.; Nor, N.A.M. Photodegradation of Phenol by N-doped TiO2 Anatase/Rutile Nanorods Assembled Microsphere under UV and Visible Light Irradiation. Mater. Chem. Phys. 2015, 162, 113–123. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Salleh, W.N.W.; Jaafar, J.; Ismail, A.F. Structural Characterization of N-doped Anatase–Rutile Mixed Phase TiO2 Nanorods Assembled Microspheres Synthesized by Simple Sol–Gel Method. J. Sol. Gel. Sci. Technol. 2015, 74, 513–520. [Google Scholar] [CrossRef]
- Zhang, W.; Zou, L.; Dionysio, D. A Parametric Study of Visible-Light Sensitive TiO2 photocatalysts Synthesis via a Facile Sol–Gel N-doping Method. J. Exp. Nanosci. 2014, 10, 1153–1165. [Google Scholar] [CrossRef]
- Samsudin, E.M.; Abd Hamid, S.B. Effect of Band Gap Engineering in Anionic-doped TiO2 Photocatalyst. Appl. Surf. Sci. 2017, 391, 326–336. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, J. Modification of the Optical and Electronic Properties of TiO2 by N Anion-doping for Augmentation of the Visible Light Assisted Photocatalytic Performance. J. Struct. Chem. 2018, 59, 1353–1361. [Google Scholar] [CrossRef]
- Zhao, B.; Wang, X.; Zhang, Y.; Gao, J.; Chen, Z.; Lu, Z. Synergism of Oxygen Vacancies, Ti3+ and N Dopants on the Visible-Light Photocatalytic Activity of N-doped TiO2. J. Photochem. Photobiol. A 2019, 382, 111928. [Google Scholar] [CrossRef]
- Kim, T.H.; Go, G.-M.; Cho, H.-B.; Song, Y.; Lee, C.-G.; Choa, Y.-H. A Novel Synthetic Method for N doped TiO2 Nanoparticles Through Plasma-Assisted Electrolysis and Photocatalytic Activity in the Visible Region. Front. Chem. 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Pichugin, V.F.; Evdokimov, K.E.; Konishchev, M.E.; Syrtanov, M.S.; Kudiiarov, V.N.; Li, K.; Tverdokhlebov, S.I. Effect of Nitrogen-Doping and Post Annealing on Wettability and Band Gap Energy of TiO2 Thin Film. Appl. Surf. Sci. 2020, 500, 144048. [Google Scholar] [CrossRef]
- Jin, Y.J.; Linghu, J.; Chai, J.; Chua, C.S.; Wong, L.M.; Feng, Y.P.; Yang, M.; Wang, S. Defect Evolution Enhanced Visible-Light Photocatalytic Activity in Nitrogen-doped Anatase TiO2 Thin Films. J. Phys. Chem. C 2018, 122, 16600–16606. [Google Scholar] [CrossRef]
- Farkas, B.; Heszler, P.; Budai, J.; Oszkó, A.; Ottosson, M.; Geretovszky, Z. Optical, Compositional and Structural Properties of Pulsed Laser Deposited Nitrogen-Doped Titanium-Dioxide. Appl. Surf. Sci. 2018, 433, 149–154. [Google Scholar] [CrossRef]
- Zhao, Z.; Kou, T.; Zhang, L.; Zhai, S.; Wang, W.; Wang, Y. Dealloying Induced N-doping in Spindle-like Porous Rutile TiO2 for Enhanced Visible Light Photocatalytic Activity. Corros. Sci. 2018, 137, 204–211. [Google Scholar] [CrossRef]
- Krivtsov, I.; Ilkaeva, M.; Salas-Colera, E.; Amghouz, Z.; García, J.R.; Díaz, E.; Ordóñez, S.; Villar-Rodil, S. Consequences of Nitrogen Doping and Oxygen Enrichment on Titanium Local Order and Photocatalytic Performance of TiO2 Anatase. J. Phys. Chem. C 2017, 121, 6770–6780. [Google Scholar] [CrossRef]
- Kovalevskiy, N.; Selishchev, D.; Svintsitskiy, D.; Selishcheva, S.; Berezin, A.; Kozlov, D. Synergistic Effect of Polychromatic Radiation on Visible Light Activity of N-doped TiO2 Photocatalyst. Catal. Commun. 2020, 134, 105841. [Google Scholar] [CrossRef]
- Cao, Y.; Xing, Z.; Shen, Y.; Li, Z.; Wu, X.; Yan, X.; Zou, J.; Yang, S.; Zhou, W. Mesoporous Black Ti3+ /N-TiO2 Spheres for Efficient Visible-Light-Driven Photocatalytic Performance. Chem. Eng. J. 2017, 325, 199–207. [Google Scholar] [CrossRef]
- Di Valentin, C.; Pacchioni, G.; Selloni, A.; Livraghi, S.; Giamello, E. Characterization of Paramagnetic Species in N-doped TiO2 Powders by EPR Spectroscopy and DFT Calculations. J. Phys. Chem. B 2005, 109, 11414–11419. [Google Scholar] [CrossRef]
- Boningari, T.; Inturi, S.N.R.; Suidan, M.; Smirniotis, P.G. Novel One-Step Synthesis of Nitrogen-doped TiO2 by Flame Aerosol Technique for Visible-Light Photocatalysis: Effect of Synthesis Parameters and Secondary Nitrogen (N) Source. Chem. Eng. J. 2018, 350, 324–334. [Google Scholar] [CrossRef]
- Dong, F.; Zhao, W.; Wu, Z.; Guo, S. Band Structure and Visible Light Photocatalytic Activity of Multi-Type Nitrogen Doped TiO2 Nanoparticles Prepared by Thermal Decomposition. J. Hazard. Mater. 2009, 162, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Dawson, M.; Soares, G.B.; Ribeiro, C. Influence of Calcination Parameters on the Synthesis of N-doped TiO2 by the Polymeric Precursors Method. J. Solid State Chem. 2014, 215, 211–218. [Google Scholar] [CrossRef]
- Bjelajac, A.; Petrović, R.; Popović, M.; Rakočević, Z.; Socol, G.; Mihailescu, I.N.; Janaćković, D. Doping of TiO2 Nanotubes with Nitrogen by Annealing in Ammonia for Visible Light Activation: Influence of Pre- and Post-Annealing in Air. Thin Solid Films 2019, 692, 137598. [Google Scholar] [CrossRef]
- Peng, F.; Cai, L.; Yu, H.; Wang, H.; Yang, J. Synthesis and Characterization of Substitutional and Interstitial Nitrogen-doped Titanium Dioxides with Visible Light Photocatalytic Activity. J. Solid State Chem. 2008, 181, 130–136. [Google Scholar] [CrossRef]
- Azami, M.S.; Nawawi, W.I.; Shukri, D.S.M. Formation of Predominant Interstitial N-TiO2 Using Physical Preparation under Microwave Irradiation for Reactive Red 4 Dye Removal. Desalin. Water Treat. 2017, 92, 172–180. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, Y.; Liu, P.; Zhang, W.; Xing, M.; Zhang, J. A Facile Approach to Further Improve the Substitution of Nitrogen into Reduced TiO2− with an Enhanced Photocatalytic Activity. Appl. Catal. B-Environ. 2015, 170–171, 66–73. [Google Scholar] [CrossRef]
- Lee, S.; Cho, I.-S.; Lee, D.K.; Kim, D.W.; Noh, T.H.; Kwak, C.H.; Park, S.; Hong, K.S.; Lee, J.-K.; Jung, H.S. Influence of Nitrogen Chemical States on Photocatalytic Activities of Nitrogen-doped TiO2 Nanoparticles under Visible Light. J. Photoch. Photobiol. A 2010, 213, 129–135. [Google Scholar] [CrossRef]
- Huang, J.; Song, H.; Chen, C.; Yang, Y.; Xu, N.; Ji, X.; Li, C.; You, J.-A. Facile Synthesis of N-doped TiO2 Nanoparticles Caged in MIL-100(Fe) for Photocatalytic Degradation of Organic Dyes under Visible Light Irradiation. J. Environ. Chem. Eng. 2017, 5, 2579–2585. [Google Scholar] [CrossRef]
- Borges, K.A.; Santos, L.M.; Paniago, R.M.; Barbosa Neto, N.M.; Schneider, J.; Bahnemann, D.W.; Patrocinio, A.O.T.; Machado, A.E.H. Characterization of a Highly Efficient N-doped TiO2 Photocatalyst Prepared via Factorial Design. New J. Chem. 2016, 40, 7846–7855. [Google Scholar] [CrossRef]
- Le, P.; Hieu, L.; Lam, T.-N.; Hang, N.; Truong, N.; Tuyen, L.; Phong, P.; Leu, J. Enhanced Photocatalytic Performance of Nitrogen-doped TiO2 Nanotube Arrays Using a Simple Annealing Process. Micromachines 2018, 9, 618. [Google Scholar] [CrossRef]
- Monteiro, R.A.R.; Miranda, S.M.; Vilar, V.J.P.; Pastrana-Martínez, L.M.; Tavares, P.B.; Boaventura, R.A.R.; Faria, J.L.; Pinto, E.; Silva, A.M.T. N-Modified TiO2 Photocatalytic Activity towards Diphenhydramine Degradation and Escherichia coli Inactivation in Aqueous Solutions. Appl. Catal. B Environ. 2015, 162, 66–74. [Google Scholar] [CrossRef]
- Kuźmiński, K.; Morawski, A.W.; Janus, M. Adsorption and Photocatalytic Degradation of Anionic and Cationic Surfactants on Nitrogen-Modified TiO2. J. Surfactants Deterg. 2018, 21, 909–921. [Google Scholar] [CrossRef]
- Veisi, F.; Zazouli, M.A.; Ebrahimzadeh, M.A.; Charati, J.Y.; Dezfoli, A.S. Photocatalytic Degradation of Furfural in Aqueous Solution by N-doped Titanium Dioxide Nanoparticles. Environ. Sci. Pollut. Res. 2016, 23, 21846–21860. [Google Scholar] [CrossRef]
- Janus, M.; Mądraszewski, S.; Zając, K.; Kusiak-Nejman, E.; Morawski, A.W.; Stephan, D. Photocatalytic Activity and Mechanical Properties of Cements Modified with TiO2/N. Materials 2019, 12, 3756. [Google Scholar] [CrossRef]
- Mungondori, H.H.; Tichagwa, L.; Katwire, D.M.; Aoyi, O. Preparation of Photo-Catalytic Copolymer Grafted Asymmetric Membranes (N-TiO2-PMAA-g-PVDF/PAN) and Their Application on the Degradation of Bentazon in Water. Iran. Polym. J. 2016, 25, 135–144. [Google Scholar] [CrossRef]
- Khalilzadeh, A.; Fatemi, S. Spouted Bed Reactor for VOC Removal by Modified Nano-TiO2 Photocatalytic Particles. Chem. Eng. Res. Des. 2016, 115, 241–250. [Google Scholar] [CrossRef]
- Sun, P.; Zhang, J.; Liu, W.; Wang, Q.; Cao, W. Modification to L-H Kinetics Model and Its Application in the Investigation on Photodegradation of Gaseous Benzene by Nitrogen-doped TiO2. Catalysts 2018, 8, 326. [Google Scholar] [CrossRef]
- Kamaei, M.; Rashedi, H.; Dastgheib, S.; Tasharrofi, S. Comparing Photocatalytic Degradation of Gaseous Ethylbenzene Using N-doped and Pure TiO2 Nano-catalysts Coated on Glass Beads under Both UV and Visible Light Irradiation. Catalysts 2018, 8, 466. [Google Scholar] [CrossRef]
- Rokicka, P.; Markowska-Szczupak, A.; Kowalczyk, Ł.; Kowalska, E.; Morawski, A.W. Influence of Titanium Dioxide Modification on the Antibacterial Properties. Pol. J. Chem. Technol. 2016, 18, 56–64. [Google Scholar] [CrossRef]
- Kawashita, M.; Endo, N.; Watanabe, T.; Miyazaki, T.; Furuya, M.; Yokota, K.; Abiko, Y.; Kanetaka, H.; Takahashi, N. Formation of Bioactive N-doped TiO2 on Ti with Visible Light-Induced Antibacterial Activity Using NaOH, Hot Water, and Subsequent Ammonia Atmospheric Heat Treatment. Colloid. Surf. B. 2016, 145, 285–290. [Google Scholar] [CrossRef]
- Kassahun, S.K.; Kiflie, Z.; Kim, H.; Gadisa, B.T. Effects of Operational Parameters on Bacterial Inactivation in Vis-LEDs Illuminated N-doped TiO2 Based Photoreactor. J. Environ. Chem. Eng. 2020, 8, 104374. [Google Scholar] [CrossRef]
- Surmacki, J. Nitrogen-Doped Titanium Dioxide Nanoparticles Modified by an Electron Beam for Improving Human Breast Cancer Detection by Raman Spectroscopy: A Preliminary Study. Diagnostics 2020, 10, 757. [Google Scholar] [CrossRef]
- Mohammadalipour, Z.; Rahmati, M.; Khataee, A.; Moosavi, M.A. Differential Effects of N-TiO2 Nanoparticle and Its Photo-activated Form on Autophagy and Necroptosis in Human Melanoma A375 Cells. J. Cell Physiol. 2020, 235, 8246–8259. [Google Scholar] [CrossRef]
- Preethi, L.K.; Antony, R.P.; Mathews, T.; Loo, S.C.J.; Wong, L.H.; Dash, S.; Tyagi, A.K. Nitrogen Doped Anatase-Rutile Heterostructured Nanotubes for Enhanced Photocatalytic Hydrogen Production: Promising Structure for Sustainable Fuel Production. Int. J. Hydrogen Energ. 2016, 41, 5865–5877. [Google Scholar] [CrossRef]
- Shvadchina, Y.O.; Cherepivskaya, M.K.; Vakulenko, V.F.; Sova, A.N.; Stolyarova, I.V.; Prikhodko, R.V. The Study of Properties and Catalytic Activity of Titanium Dioxide Doped with Sulphur. J. Water Chem. Technol. 2015, 37, 283–288. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Hsueh, H.-T.; Chang, C.-W.; Chu, H. The Visible Light-Driven Photodegradation of Dimethyl Sulfide on S-doped TiO2: Characterization, Kinetics, and Reaction Pathways. Appl. Catal. B-Environ. 2016, 199, 1–10. [Google Scholar] [CrossRef]
- Hosseinzadeh, G.; Rasoulnezhad, H.; Ghasemian, N.; Hosseinzadeh, R. Ultrasonic-Assisted Spray Pyrolysis Technique for Synthesis of Transparent S-doped TiO2 Thin Film. J. Aust. Ceram. Soc. 2018, 55, 387–394. [Google Scholar] [CrossRef]
- Sun, X.-Y.; Sun, X.; Zhang, X.; Qian, N.-X.; Wang, M.; Ma, Y.-Q. Improved Adsorption and Degradation Performance by S-doping of (001)-TiO2. Beilstein J. Nanotechnol. 2019, 10, 2116–2127. [Google Scholar] [CrossRef] [PubMed]
- Taherinia, M.; Nasiri, M.; Abedini, E.; Pouretedal, H.R. Comparing the Photocatalytic Activity of N-doped and S-doped Titanium Dioxide Nanoparticles for Water Splitting under Sunlight Radiation. J. Iran. Chem. Soc. 2018, 15, 1301–1310. [Google Scholar] [CrossRef]
- Esquivel-Escalante, K.; Nava-Mendoza, R.; Velázquez-Castillo, R. Crystal structure determination of the S/TiO2 system and the correlation with its photocatalytic properties. J. Nanosci. Nanotechnol. 2016, 16, 967–972. [Google Scholar] [CrossRef]
- Ramacharyulu, P.V.R.K.; Nimbalkar, D.B.; Kumar, J.P.; Prasad, G.K.; Ke, S.-C. N-doped, S-doped TiO2 nanocatalysts: Synthesis, Characterization and Photocatalytic Activity in the Presence of Sunlight. RSC Adv. 2015, 5, 37096–37101. [Google Scholar] [CrossRef]
- Baeissa, E.S. Synthesis and Characterization of Sulfur-Titanium Dioxide Nanocomposites for Photocatalytic Oxidation of Cyanide Using Visible Light Irradiation. Chin. J. Catal. 2015, 36, 698–704. [Google Scholar] [CrossRef]
- Birben, N.C.; Uyguner-Demirel, C.S.; Sen-Kavurmaci, S.; Gurkan, Y.Y.; Turkten, N.; Cinar, Z.; Bekbolet, M. Comparative Evaluation of Anion Doped Photocatalysts on the Mineralization and Decolorization of Natural Organic Matter. Catal. Today 2015, 240, 125–131. [Google Scholar] [CrossRef]
- Chen, X.; Sun, H.; Zhang, J.; Guo, Y.; Kuo, D.-H. Cationic S-doped TiO2/SiO2 Visible-Light Photocatalyst Synthesized by Co-hydrolysis Method and Its Application for Organic Degradation. J. Mol. Liq. 2019, 273, 50–57. [Google Scholar] [CrossRef]
- McManamon, C.; O’Connell, J.; Delaney, P.; Rasappa, S.; Holmes, J.D.; Morris, M.A. A Facile Route to Synthesis of S-doped TiO2 Nanoparticles for Photocatalytic Activity. J. Mol. Catal. A-Chem. 2015, 406, 51–57. [Google Scholar] [CrossRef]
- Yi, C.; Liao, Q.; Deng, W.; Huang, Y.; Mao, J.; Zhang, B.; Wu, G. The Preparation of Amorphous TiO2 Doped with Cationic S and Its Application to the Degradation of DCFs under Visible Light Irradiation. Sci. Total Environ. 2019, 684, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Abu Bakar, S.; Ribeiro, C. An Insight Toward the Photocatalytic Activity of S Doped 1-D TiO2 Nanorods Prepared via Novel Route: As Promising Platform for Environmental Leap. J. Mol. Catal. A-Chem. 2016, 412, 78–92. [Google Scholar] [CrossRef]
- Bakar, S.A.; Ribeiro, C. A Comparative Run for Visible-Light-Driven Photocatalytic Activity of Anionic and Cationic S-doped TiO2 Photocatalysts: A Case Study of Possible Sulfur Doping through Chemical Protocol. J. Mol. Catal. A-Chem. 2016, 421, 1–15. [Google Scholar] [CrossRef]
- Chen, X.; Sun, H.; Zelekew, O.A.; Zhang, J.; Guo, Y.; Zeng, A.; Kuo, D.-H.; Lin, J. Biological Renewable Hemicellulose-Template for Synthesis of Visible Light Responsive Sulfur-doped TiO2 for Photocatalytic Oxidation of Toxic Organic and As(III) Pollutants. Appl. Surf. Sci. 2020, 525, 146531. [Google Scholar] [CrossRef]
- Meng, Q.; Liu, B.; Liu, H.; Cai, Y.; Dong, L. Effects of S and Ta Codoping on Photocatalytic Activity of Rutile TiO2. J. Sol-Gel Sci. Technol. 2018, 86, 631–639. [Google Scholar] [CrossRef]
- Anitha, B.; Ravidhas, C.; Venkatesh, R.; Raj, A.M.E.; Ravichandran, K.; Subramanian, B.; Sanjeeviraja, C. Self Assembled Sulfur Induced Interconnected Nanostructure TiO2 Electrode for Visible Light Photoresponse and Photocatalytic Application. Phys. E 2017, 91, 148–160. [Google Scholar] [CrossRef]
- Chen, X.; Kuo, D.-H.; Lu, D. Visible Light Response and Superior Dispersed S-doped TiO2 Nanoparticles Synthesized via Ionic Liquid. Adv. Powder Technol. 2017, 28, 1213–1220. [Google Scholar] [CrossRef]
- Nam, S.-H.; Kim, T.K.; Boo, J.-H. Physical Property and Photo-Catalytic Activity of Sulfur Doped TiO2 Catalysts Responding to Visible Light. Catal. Today 2012, 185, 259–262. [Google Scholar] [CrossRef]
- Ramacharyulu, P.V.R.K.; Praveen Kumar, J.; Prasad, G.K.; Sreedhar, B. Sulphur Doped Nano TiO2: Synthesis, Characterization and Photocatalytic Degradation of a Toxic Chemical in Presence of Sunlight. Mater. Chem. Phys. 2014, 148, 692–698. [Google Scholar] [CrossRef]
- Yu, J.C.; Ho, W.; Yu, J.; Yip, H.; Wong, P.K.; Zhao, J. Efficient Visible-Light-Induced Photocatalytic Disinfection on Sulfur-Doped Nanocrystalline Titania. Environ. Sci. Technol. 2005, 39, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Zhongchen, Y.; Song, W.; Lu, H.; Zhuan, L.; Yuanlin, N. Photocatalytic Property of Nanostructured S Doped TiO2 Films Prepared by the Micro Plasma Method. Rare Met. Mater. Eng. 2015, 44, 1629–1632. [Google Scholar] [CrossRef]
- Randeniya, L.K.; Murphy, A.B.; Plumb, I.C. A Study of S-Doped TiO2 for Photoelectrochemical Hydrogen Generation from Water. J. Mater. Sci. 2007, 43, 1389–1399. [Google Scholar] [CrossRef]
- Bu, X.; Wang, Y.; Li, J.; Zhang, C. Improving the Visible Light Photocatalytic Activity of TiO2 by Combining Sulfur Doping and Rectorite Carrier. J. Alloys Compd. 2015, 628, 20–26. [Google Scholar] [CrossRef]
- Albrbar, A.J.; Djokić, V.; Bjelajac, A.; Kovač, J.; Ćirković, J.; Mitrić, M.; Janaćković, D.; Petrović, R. Visible-Light Active Mesoporous, Nanocrystalline N,S-doped and Co-doped Titania Photocatalysts Synthesized by Non-Hydrolytic Sol-Gel Route. Ceram. Int. 2016, 42, 16718–16728. [Google Scholar] [CrossRef]
- Zhu, M.; Zhai, C.; Qiu, L.; Lu, C.; Paton, A.S.; Du, Y.; Goh, M.C. New Method to Synthesize S-doped TiO2 with Stable and Highly Efficient Photocatalytic Performance under Indoor Sunlight Irradiation. ACS Sustain. Chem. Eng. 2015, 3, 3123–3129. [Google Scholar] [CrossRef]
- Sharotri, N.; Sud, D. A Greener Approach to Synthesize Visible Light Responsive Nanoporous S-doped TiO2 with Enhanced Photocatalytic Activity. New J. Chem. 2015, 39, 2217–2223. [Google Scholar] [CrossRef]
- Kovačić, M.; Perović, K.; Papac, J.; Tomić, A.; Matoh, L.; Žener, B.; Brodar, T.; Capan, I.; Surca, A.K.; Kušić, H.; et al. One-Pot Synthesis of Sulfur-doped TiO2/Reduced Graphene Oxide Composite (S-TiO2/RGO) with Improved Photocatalytic Activity for the Removal of Diclofenac from Water. Materials 2020, 13, 1621. [Google Scholar] [CrossRef]
- Huang, Z.; Gao, Z.; Gao, S.; Wang, Q.; Wang, Z.; Huang, B.; Dai, Y. Facile Synthesis of S-doped Reduced TiO2-x with Enhanced Visible-Light Photocatalytic Performance. Chin. J. Catal. 2017, 38, 821–830. [Google Scholar] [CrossRef]
- Xing, Z.; Li, Z.; Wu, X.; Wang, G.; Zhou, W. In-Situ S-doped Porous Anatase TiO2 Nanopillars for High-Efficient Visible-Light Photocatalytic Hydrogen Evolution. Int. J. Hydrog. Energy 2016, 41, 1535–1541. [Google Scholar] [CrossRef]
- Li, M.; Xing, Z.; Jiang, J.; Li, Z.; Kuang, J.; Yin, J.; Wan, N.; Zhu, Q.; Zhou, W. In-Situ Ti3+/S Doped High Thermostable Anatase TiO2 Nanorods as Efficient Visible-Light-Driven Photocatalysts. Mater. Chem. Phys. 2018, 219, 303–310. [Google Scholar] [CrossRef]
- Mohan Kumar, K.; Godavarthi, S.; Karthik, T.V.K.; Mahendhiran, M.; Hernandez-Eligio, A.; Hernandez-Como, N.; Agarwal, V.; Martinez Gomez, L. Green Synthesis of S-doped Rod Shaped Anatase TiO2 Microstructures. Mater. Lett. 2016, 183, 211–214. [Google Scholar] [CrossRef]
- Bakar, S.A.; Ribeiro, C. Rapid and Morphology Controlled Synthesis of Anionic S-doped TiO2 Photocatalysts for the Visible-Light-Driven Photodegradation of Organic Pollutants. RSC Adv. 2016, 6, 36516–36527. [Google Scholar] [CrossRef]
- Wang, F.; Li, F.; Zhang, L.; Zeng, H.; Sun, Y.; Zhang, S.; Xu, X. S-TiO2 with Enhanced Visible-Light Photocatalytic Activity Derived from TiS2 in Deionized Water. Mater. Res. Bull. 2017, 87, 20–26. [Google Scholar] [CrossRef]
- Ho, W.; Yu, J.C.; Lee, S. Low-Temperature Hydrothermal Synthesis of S-Doped TiO2 with Visible Light Photocatalytic Activity. J. Solid State Chem. 2006, 179, 1171–1176. [Google Scholar] [CrossRef]
- Umebayashi, T.; Yamaki, T.; Itoh, H.; Asai, K. Band Gap Narrowing of Titanium Dioxide by Sulfur Doping. Appl. Phys. Lett. 2002, 81, 454–456. [Google Scholar] [CrossRef]
- Shin, S.W.; Lee, J.Y.; Ahn, K.-S.; Kang, S.H.; Kim, J.H. Visible Light Absorbing TiO2 Nanotube Arrays by Sulfur Treatment for Photoelectrochemical Water Splitting. J. Phys. Chem. C 2015, 119, 13375–13383. [Google Scholar] [CrossRef]
- Bento, R.T.; Correa, O.V.; Pillis, M.F. Photocatalytic Activity of Undoped and Sulfur-doped TiO2 Films Grown by MOCVD for Water Treatment under Visible Light. J. Eur. Ceram. Soc. 2019, 39, 3498–3504. [Google Scholar] [CrossRef]
- Boningari, T.; Inturi, S.N.R.; Suidan, M.; Smirniotis, P.G. Novel One-Step Synthesis of Sulfur Doped-TiO2 by Flame Spray Pyrolysis for Visible Light Photocatalytic Degradation of Acetaldehyde. Chem. Eng. J. 2018, 339, 249–258. [Google Scholar] [CrossRef]
- Momeni, M.M.; Ghayeb, Y.; Ghonchegi, Z. Visible Light Activity of Sulfur-doped TiO2 Nanostructure Photoelectrodes Prepared by Single-Step Electrochemical Anodizing Process. J. Solid State Electrochem. 2015, 19, 1359–1366. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, X.; Li, W.; Wang, H.; Li, H. Enhancing the Photocatalytic Degradation of Salicylic Acid by Using Molecular Imprinted S-Doped TiO2 under Simulated Solar Light. Ceram. Int. 2014, 40, 8863–8867. [Google Scholar] [CrossRef]
- Sun, S.; Zhang, J.; Gao, P.; Wang, Y.; Li, X.; Wu, T.; Wang, Y.; Chen, Y.; Yang, P. Full Visible-Light Absorption of TiO2 Nanotubes Induced by Anionic S22− Doping and Their Greatly Enhanced Photocatalytic Hydrogen Production Abilities. Appl. Catal. B Environ. 2017, 206, 168–174. [Google Scholar] [CrossRef]
- Adiansyah, A.; Purba, I.E.; Tarigan, M.; Lisnadiyanti, L.; Yusnaidar, Y.; Mahendra, I.P. Preparation of S-doped TiO2 via Sol-Gel/Hydrothermal Method and Its Activity as Photodegradation of RB05. RJC 2020, 13, 757–760. [Google Scholar] [CrossRef]
- Olowoyo, J.O.; Kumar, M.; Jain, S.L.; Shen, S.; Zhou, Z.; Mao, S.S.; Vorontsov, A.V.; Kumar, U. Reinforced Photocatalytic Reduction of CO2 to Fuel by Efficient S-TiO2: Significance of Sulfur Doping. Int. J. Hydrog. Energy 2018, 43, 17682–17695. [Google Scholar] [CrossRef]
- Pillai, V.V.; Lonkar, S.P.; Alhassan, S.M. Template-Free, Solid-State Synthesis of Hierarchically Macroporous S-doped TiO2 Nano-photocatalysts for Efficient Water Remediation. ACS Omega 2020, 5, 7969–7978. [Google Scholar] [CrossRef] [PubMed]
- Sraw, A.; Kaur, T.; Pandey, Y.; Verma, A.; Sobti, A.; Wanchoo, R.K.; Toor, A.P. Photocatalytic Degradation of Monocrotophos and Quinalphos Using Solar-Activated S-Doped TiO2. Int. J. Environ. Sci. Technol. 2020, 17, 4895–4908. [Google Scholar] [CrossRef]
- Liu, C.; Chen, W.; Sheng, Y.; Li, L. Atrazine Degradation in Solar Irradiation/S-Doped Titanium Dioxide Treatment. In Proceedings of the 2009 3rd International Conference on Bioinformatics and Biomedical Engineering, Beijing, China, 11–13 June 2009. [Google Scholar] [CrossRef]
- Umebayashi, T.; Yamaki, T.; Tanaka, S.; Asai, K. Visible Light-Induced Degradation of Methylene Blue on S-Doped TiO2. Chem. Lett. 2003, 32, 330–331. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Chou, S.-H.; Chu, H. A Kinetic Study for the Degradation of 1,2-Dichloroethane by S-Doped TiO2 under Visible Light. J. Nanopart. Res. 2014, 16. [Google Scholar] [CrossRef]
- Vorontsov, A.V.; Valdés, H. Insights into the Visible Light Photocatalytic Activity of S-doped Hydrated TiO2. Int. J. Hydrogen Energ. 2019, 44, 17963–17973. [Google Scholar] [CrossRef]
- Etghani, S.A.; Ansari, E.; Mohajerzadeh, S. Evolution of Large Area TiS2-TiO2 Heterostructures and S-doped TiO2 Nano-sheets on Titanium Foils. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Basera, P.; Saini, S.; Arora, E.; Singh, A.; Kumar, M.; Bhattacharya, S. Stability of Non-metal Dopants to Tune the Photo-Absorption of TiO2 at Realistic Temperatures and Oxygen Partial Pressures: A Hybrid DFT Study. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Xin, Y.; Gao, M.; Wu, J. Fabrication and Photocatalytic Properties of Cationic and Anionic S-doped TiO2 Nanofibers by Electrospinning. Appl. Catal. B-Environ. 2014, 147, 49–57. [Google Scholar] [CrossRef]
- Syafiuddin, A.; Hadibarata, T.; Zon, N.F. Characterization of Titanium Dioxide Doped with Nitrogen and Sulfur and Its Photocatalytic Appraisal for Degradation of Phenol and Methylene Blue. J. Chin. Chem. Soc. Taip. 2017, 64, 1333–1339. [Google Scholar] [CrossRef]
- Khang, N.C.; Van, D.Q.; Thuy, N.M.; Minh, N.V.; Minh, P.N. Remarkably Enhanced Photocatalytic Activity by Sulfur-doped Titanium Dioxide in Nanohybrids with Carbon Nanotubes. J. Phys. Chem. Solids 2016, 99, 119–123. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, X.; Rao, R.; Qian, N.X.; Ma, Y.Q. Study on Vapor-Thermal Synthesis and Sulfur-doping of TiO2/Graphene Composites. Appl. Surf. Sci. 2020, 507, 144856. [Google Scholar] [CrossRef]
- Dunnill, C.W.; Aiken, Z.A.; Kafizas, A.; Pratten, J.; Wilson, M.; Morgan, D.J.; Parkin, I.P. White Light Induced Photocatalytic Activity of Sulfur-Doped TiO2 Thin Films and Their Potential for Antibacterial Application. J. Mater. Chem. 2009, 19, 8747. [Google Scholar] [CrossRef]
- Osin, O.A.; Yu, T.; Cai, X.; Jiang, Y.; Peng, G.; Cheng, X.; Li, R.; Qin, Y.; Lin, S. Photocatalytic Degradation of 4-Nitrophenol by C,N-TiO2: Degradation Efficiency vs. Embryonic Toxicity of the Resulting Compounds. Front. Chem. 2018, 6. [Google Scholar] [CrossRef]
- Janus, M.; Bubacz, K.; Zatorska, J.; Kusiak-Nejman, E.; Czyżewski, A.; Morawski, A.W. Preliminary Studies of Photocatalytic Activity of Gypsum Plasters Containing TiO2 Co-modified with Nitrogen and Carbon. Pol. J. Chem. Technol. 2015, 17, 96–102. [Google Scholar] [CrossRef]
- Janus, M.; Kusiak-Nejman, E.; Rokicka-Konieczna, P.; Markowska-Szczupak, A.; Zając, K.; Morawski, A.W. Bacterial Inactivation on Concrete Plates Loaded with Modified TiO2 Photocatalysts under Visible Light Irradiation. Molecules 2019, 24, 3026. [Google Scholar] [CrossRef]
- Janus, M.; Zatorska, J.; Czyżewski, A.; Bubacz, K.; Kusiak-Nejman, E.; Morawski, A.W. Self-Cleaning Properties of Cement Plates Loaded with N,C-modified TiO2 Photocatalysts. Appl. Surf. Sci. 2015, 330, 200–206. [Google Scholar] [CrossRef]
- Xiao, Y.; Sun, X.; Li, I.; Chen, J.; Zhao, S.; Jiang, C.; Yang, L.; Cheng, L.; Cao, S. Simultaneous Formation of a C/N-TiO2 Hollow Photocatalyst with Efficient Photocatalytic Performance and Recyclability. Chin. J. Catal. 2019, 40, 765–775. [Google Scholar] [CrossRef]
- Buda, W.; Czech, B. Preparation and Characterization of C,N-Codoped TiO2 Photocatalyst for the Degradation of Diclofenac from Wastewater. Water Sci. Technol. 2013, 68, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, J.; Nerissa, E.; Hadi, A. Synthesis, Characterization and Efficiency of N, C-TiO2 as an Active Visible Light Photocatalyst. AMM 2014, 661, 63–67. [Google Scholar] [CrossRef]
- Ariza-Tarazona, M.C.; Villarreal-Chiu, J.F.; Hernández-López, J.M.; Rivera De la Rosa, J.; Barbieri, V.; Siligardi, C.; Cedillo-González, E.I. Microplastic Pollution Reduction by a Carbon and Nitrogen-doped TiO2: Effect of pH and Temperature in the Photocatalytic Degradation Process. J. Hazard. Mater. 2020, 395, 122632. [Google Scholar] [CrossRef] [PubMed]
- El-Sheikh, S.M.; Khedr, T.M.; Hakki, A.; Ismail, A.A.; Badawy, W.A.; Bahnemann, D.W. Visible Light Activated Carbon and Nitrogen Co-doped Mesoporous TiO2 as Efficient Photocatalyst for Degradation of Ibuprofen. Sep. Purif. Technol. 2017, 173, 258–268. [Google Scholar] [CrossRef]
- Li, F.; Zhou, J.; Du, C.; Li, W.; Wang, Y.; He, G.; He, Q. Preparation and Photocatalytic Properties of Porous C and N Co-doped TiO2 Deposited on Brick by a Fast, One-Step Microwave Irradiation Method. J. Environ. Sci. 2017, 60, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Zhou, J.; Li, F.; Li, W.; Wang, Y.; He, Q. Extremely Fast Dark Adsorption Rate of Carbon and Nitrogen Co-doped TiO2 Prepared by a Relatively Fast, Facile and Low-Cost Microwave Method. Appl. Phys. A 2016, 122. [Google Scholar] [CrossRef]
- Zhou, J.; Li, F.; Du, C.; Liu, J.; Wang, Y.; Li, W.; He, G.; He, Q. Photodegradation Performance and Recyclability of a Porous Nitrogen and Carbon Co-doped TiO2/Activated Carbon Composite Prepared by an Extremely Fast One-Step Microwave Method. RSC Adv. 2016, 6, 84457–84463. [Google Scholar] [CrossRef]
- Al-Hajji, L.A.; Ismail, A.A.; Alseidi, M.; Almutawa, F.; Ahmad, S.; Bumajdad, A. Green Approach and Ease Synthesis of C/N-codoped TiO2 Nanocrystals for Photodegradation of Endocrine. J. Nanopart. Res. 2020, 22. [Google Scholar] [CrossRef]
- Wang, X.; Lim, T.-T. Effect of Hexamethylenetetramine on the Visible-Light Photocatalytic Activity of C–N Codoped TiO2 for Bisphenol A Degradation: Evaluation of Photocatalytic Mechanism and Solution Toxicity. Appl. Catal. A-Gen. 2011, 399, 233–241. [Google Scholar] [CrossRef]
- Wang, X.; Lim, T.-T. Solvothermal Synthesis of C–N Codoped TiO2 and Photocatalytic Evaluation for Bisphenol A Degradation Using a Visible-Light Irradiated LED Photoreactor. Appl. Catal. B-Environ. 2010, 100, 355–364. [Google Scholar] [CrossRef]
- Komatsuda, S.; Asakura, Y.; Vequizo, J.J.M.; Yamakata, A.; Yin, S. Enhanced Photocatalytic NO Decomposition of Visible-Light Responsive F-TiO2/(N,C)-TiO2 by Charge Transfer between F-TiO2 and (N,C)-TiO2 through Their Doping Levels. Appl. Catal. B-Environ. 2018, 238, 358–364. [Google Scholar] [CrossRef]
- Huang, X.; Yang, W.; Zhang, G.; Yan, L.; Zhang, Y.; Jiang, A.; Xu, H.; Zhou, M.; Liu, Z.; Tang, H.; et al. Alternative Synthesis of Nitrogen and Carbon Co-doped TiO2 for Removing Fluoroquinolone Antibiotics in Water under Visible Light. Catal. Today 2019. [Google Scholar] [CrossRef]
- Zhang, J.; Xing, Z.; Cui, J.; Li, Z.; Tan, S.; Yin, J.; Zou, J.; Zhu, Q.; Zhou, W. C,N Co-doped Porous TiO2 hollow Sphere Visible Light Photocatalysts for Efficient Removal of Highly Toxic Phenolic Pollutants. Dalton Trans. 2018, 47, 4877–4884. [Google Scholar] [CrossRef] [PubMed]
- Boscaro, P.; Cacciaguerra, T.; Cot, D.; Fajula, F.; Hulea, V.; Galarneau, A. C,N-doped TiO2 Monoliths with Hierarchical Macro-/Mesoporosity for Water Treatment under Visible Light. Microporous Mesoporous Mater. 2019, 280, 37–45. [Google Scholar] [CrossRef]
- Ji, L.; Zhou, S.; Liu, X.; Gong, M.; Xu, T. Synthesis of Carbon- and Nitrogen-Doped TiO2/Carbon Composite Fibers by a Surface-Hydrolyzed PAN Fiber and Their Photocatalytic Property. J. Mater Sci. 2019, 55, 2471–2481. [Google Scholar] [CrossRef]
- Abdullah, A.M.; Al-Thani, N.J.; Tawbi, K.; Al-Kandari, H. Carbon/Nitrogen-Doped TiO2: New Synthesis Route, Characterization and Application for Phenol Degradation. Arab. J. Chem. 2016, 9, 229–237. [Google Scholar] [CrossRef]
- Wang, M.; Han, J.; Hu, Y.; Guo, R. Mesoporous C, N-codoped TiO2 Hybrid Shells with Enhanced Visible Light Photocatalytic Performance. RSC Adv. 2017, 7, 15513–15520. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Y.; Cao, C.; Xu, J.; Qian, Q.; Luo, Y.; Xue, H.; Xiao, L.; Chen, Y.; Chen, Q. Electrospun Nitrogen and Carbon Co-doped Porous TiO2 Nanofibers with High Visible Light Photocatalytic Activity. New J. Chem. 2015, 39, 6944–6950. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Y.; Zhou, L.; Li, X.; Guo, X. In Situ C,N-codoped Mesoporous TiO2 Nanocrystallites with High Surface Areas and Worm-like Structure for Efficient Photocatalysis. J. Porous Mater. 2017, 25, 571–579. [Google Scholar] [CrossRef]
- Chen, D.; Jiang, Z.; Geng, J.; Wang, Q.; Yang, D. Carbon and Nitrogen Co-Doped TiO2 with Enhanced Visible-Light Photocatalytic Activity. Ind. Eng. Chem. Res. 2007, 46, 2741–2746. [Google Scholar] [CrossRef]
- Zhang, X.; Cai, M.; Cui, N.; Chen, G.; Zou, G.; Zhou, L. One-Step Synthesis of b-N-TiO2/C Nanocomposites with High Visible Light Photocatalytic Activity to Degrade Microcystis Aeruginosa. Catalysts 2020, 10, 579. [Google Scholar] [CrossRef]
- Chen, H.; Chen, K.-F.; Lai, S.-W.; Dang, Z.; Peng, Y.-P. Photoelectrochemical Oxidation of Azo Dye and Generation of Hydrogen via CN Co-doped TiO2 Nanotube Arrays. Sep. Purif. Technol. 2015, 146, 143–153. [Google Scholar] [CrossRef]
- Wu, T.; Niu, P.; Yang, Y.; Yin, L.; Tan, J.; Zhu, H.; Irvine, J.T.S.; Wang, L.; Liu, G.; Cheng, H. Homogeneous Doping of Substitutional Nitrogen/Carbon in TiO2 Plates for Visible Light Photocatalytic Water Oxidation. Adv. Funct. Mater. 2019, 29, 1901943. [Google Scholar] [CrossRef]
- Zhang, D.; Ma, X.; Zhang, H.; Liao, Y.; Xiang, Q. Enhanced Photocatalytic Hydrogen Evolution Activity of Carbon and Nitrogen Self-Doped TiO2 Hollow Sphere with the Creation of Oxygen Vacancy and Ti3+. Mater. Today Energy 2018, 10, 132–140. [Google Scholar] [CrossRef]
- Chaudhary, J.P.; Mahto, A.; Vadodariya, N.; Kholiya, F.; Maiti, S.; Nataraj, S.K.; Meena, R. Fabrication of Carbon and Sulphur-Doped Nanocomposites with Seaweed Polymer Carrageenan as an Efficient Catalyst for Rapid Degradation of Dye Pollutants Using a Solar Concentrator. RSC Adv. 2016, 6, 61716–61724. [Google Scholar] [CrossRef]
- Romanovska, N.I.; Manoryk, P.A.; Selyshchev, O.V.; Ermokhina, N.I.; Yaremov, P.S.; Grebennikov, V.M.; Shcherbakov, S.M.; Zahn, D.R.T. Effect of the Modification of TiO2 with Thiourea on Its Photocatalytic Activity in Doxycycline Degradation. Theor. Exp. Chem. 2020, 56, 183–191. [Google Scholar] [CrossRef]
- Mansingh, S.; Das, K.K.; Behera, A.; Subudhi, S.; Sultana, S.; Parida, K. Bandgap Engineering via Boron and Sulphur Doped Carbon Modified Anatase TiO2: A Visible Light Stimulated Photocatalyst for Photo-Fixation of N2 and TCH Degradation. Nanoscale Adv. 2020, 2, 2004–2017. [Google Scholar] [CrossRef]
- Sun, H.; Bai, Y.; Cheng, Y.; Jin, W.; Xu, N. Preparation and Characterization of Visible-Light-Driven Carbon−Sulfur-Codoped TiO2 Photocatalysts. Ind. Eng. Chem. Res. 2006, 45, 4971–4976. [Google Scholar] [CrossRef]
- Hohol, M.; Sanytsky, M.; Kropyvnytska, T.; Barylyak, A.; Bobitski, Y. The Effect of Sulfur- and Carbon-Codoped TiO2 Nanocomposite on the Photocatalytic and Mechanical Properties of Cement Mortars. EEJET 2020, 4, 6–14. [Google Scholar] [CrossRef]
- Chung, J.; Chung, J.W.; Kwak, S.-Y. Adsorption-Assisted Photocatalytic Activity of Nitrogen and Sulfur Codoped TiO2 under Visible Light Irradiation. Phys. Chem. Chem. Phys. 2015, 17, 17279–17287. [Google Scholar] [CrossRef]
- Akan, P.; Birben, N.C.; Bekbolet, M. Adsorption of Molecular Size Fractions of Humic Acid onto Anion-Doped TiO2 specimens. Desalin. Water Treat. 2015, 57, 2455–2465. [Google Scholar] [CrossRef]
- Bakar, S.A.; Ribeiro, C. Prospective Aspects of Preferential {001} Facets of N,S-co-doped TiO2 Photocatalysts for Visible-Light-Responsive Photocatalytic Activity. RSC Adv. 2016, 6, 89274–89287. [Google Scholar] [CrossRef]
- Ju, J.; Chen, X.; Shi, Y.; Miao, J.; Wu, D. Hydrothermal Preparation and Photocatalytic Performance of N, S-Doped Nanometer TiO2 under Sunshine Irradiation. Powder Technol. 2013, 237, 616–622. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, H.; Liu, P. Enhanced Transformation of Atrazine by High Efficient Visible Light-Driven N, S-Codoped TiO2 Nanowires Photocatalysts. Int. J. Photoenergy 2014, 1–8. [Google Scholar] [CrossRef]
- Rengifo-Herrera, J.A.; Mielczarski, E.; Mielczarski, J.; Castillo, N.C.; Kiwi, J.; Pulgarin, C. Escherichia Coli Inactivation by N, S Co-Doped Commercial TiO2 Powders under UV and Visible Light. Appl. Catal. B-Environ. 2008, 84, 448–456. [Google Scholar] [CrossRef]
- Gao, H.; Liu, Y.; Ding, C.; Dai, D.; Liu, G. Synthesis, Characterization, and Theoretical Study of N, S-Codoped Nano-TiO2 with Photocatalytic Activities. Int. J. Miner. Metall. Mater. 2011, 18, 606–614. [Google Scholar] [CrossRef]
- Antonopoulou, M.; Giannakas, A.; Bairamis, F.; Papadaki, M.; Konstantinou, I. Degradation of Organophosphorus Flame Retardant Tris (1-Chloro-2-Propyl) Phosphate (TCPP) by Visible Light N,S-codoped TiO2 Photocatalysts. Chem. Eng. J. 2017, 318, 231–239. [Google Scholar] [CrossRef]
- Chen, J.; Zhong, J.; Li, J.; Dou, L. Enhanced Sunlight Photocatalytic Performance of N,S-codoped TiO2 Prepared by Sol-Gel Method Using Ammonium Thiocyanate. Synth. Synth. React. Inorg. Met. Chem. 2016, 46, 1596–1604. [Google Scholar] [CrossRef]
- Eslami, A.; Amini, M.M.; Yazdanbakhsh, A.R.; Mohseni-Bandpei, A.; Safari, A.A.; Asadi, A. N,S Co-doped TiO2 nanoparticles and Nanosheets in Simulated Solar Light for Photocatalytic Degradation of Non-Steroidal Anti-Inflammatory Drugs in Water: A Comparative Study. J. Chem. Technol. Biotechnol. 2016, 91, 2693–2704. [Google Scholar] [CrossRef]
- Yan, J.; Zhao, J.; Hao, L.; Hu, Y.; Liu, T.; Guan, S.; Zhao, Q.; Zhu, Z.; Lu, Y. Low-Temperature S-Doping on N-doped TiO2 Films and Remarkable Enhancement on Visible-Light Performance. Mater. Res. Bull. 2019, 120, 110594. [Google Scholar] [CrossRef]
- Farhadian, N.; Akbarzadeh, R.; Pirsaheb, M.; Jen, T.-C.; Fakhri, Y.; Asadi, A. Chitosan Modified N, S-doped TiO2 and N, S-Doped ZnO for Visible Light Photocatalytic Degradation of Tetracycline. Int. J. Biol. Macromol. 2019, 132, 360–373. [Google Scholar] [CrossRef]
- Khalilian, H.; Behpour, M.; Atouf, V.; Hosseini, S.N. Immobilization of S, N-codoped TiO2 Nanoparticles on Glass Beads for Photocatalytic Degradation of Methyl Orange by Fixed Bed Photoreactor under Visible and Sunlight Irradiation. Sol. Energy 2015, 112, 239–245. [Google Scholar] [CrossRef]
- Koltsakidou, Α.; Antonopoulou, M.; Εvgenidou, Ε.; Konstantinou, I.; Giannakas, A.E.; Papadaki, M.; Bikiaris, D.; Lambropoulou, D.A. Photocatalytical Removal of Fluorouracil Using TiO2-P25 and N/S Doped TiO2 Catalysts: A Kinetic and Mechanistic Study. Sci. Total Environ. 2017, 578, 257–267. [Google Scholar] [CrossRef]
- Jiao, W.; Li, N.; Wang, L.; Wen, L.; Li, F.; Liu, G.; Cheng, H.-M. High-Rate Lithium Storage of Anatase TiO2 Crystals Doped with Both Nitrogen and Sulfur. Chem. Commun. 2013, 49, 3461. [Google Scholar] [CrossRef]
- Malini, B.; Allen Gnana Raj, G. C,N and S-doped TiO2—Characterization and Photocatalytic Performance for Rose Bengal Dye Degradation under Day Light. J. Environ. Chem. Eng. 2018, 6, 5763–5770. [Google Scholar] [CrossRef]
- Wang, F.; Ma, Z.; Ban, P.; Xu, X. C, N and S Codoped Rutile TiO2 Nanorods for Enhanced Visible-Light Photocatalytic Activity. Mater. Lett. 2017, 195, 143–146. [Google Scholar] [CrossRef]
- Lei, X.F.; Xue, X.X.; Yang, H.; Chen, C.; Li, X.; Niu, M.C.; Gao, X.Y.; Yang, Y.T. Effect of Calcination Temperature on the Structure and Visible-Light Photocatalytic Activities of (N, S and C) Co-doped TiO2 Nano-Materials. Appl. Surf. Sci. 2015, 332, 172–180. [Google Scholar] [CrossRef]
- Chun, H.-H.; Jo, W.-K. Heterogeneous Decomposition of Volatile Organic Compounds by Visible-Light Activated N, C, S-embedded Titania. J. Nanosci. Nanotechnol. 2016, 16, 4544–4553. [Google Scholar] [CrossRef]
- Amreetha, S.; Dhanuskodi, S.; Nithya, A.; Jothivenkatachalam, K. Photocatalytic Degradation of Rhodamine B by C-N-S Tridoped TiO2 nanoparticles. In Oxide-Based Materials and Devices VI; Teherani, F.H., Look, D.C., Rogers, D.J., Eds.; SPIE: Bellingham, WA, USA, 2015. [Google Scholar] [CrossRef]
- Amreetha, S.; Dhanuskodi, S.; Nithya, A.; Jothivenkatachalam, K. Three Way Electron Transfer of a C–N–S Tri Doped Two-Phase Junction of TiO2 Nanoparticles for Efficient Visible Light Photocatalytic Dye Degradation. RSC Adv. 2016, 6, 7854–7863. [Google Scholar] [CrossRef]
- Yan, X.; Xing, Z.; Cao, Y.; Hu, M.; Li, Z.; Wu, X.; Zhu, Q.; Yang, S.; Zhou, W. In-Situ C-N-S-Tridoped Single Crystal Black TiO2 Nanosheets with Exposed {001} Facets as Efficient Visible-Light-Driven Photocatalysts. Appl. Catal. B Environ. 2017, 219, 572–579. [Google Scholar] [CrossRef]
- Li, D.; Xing, Z.; Yu, X.; Cheng, X. One-Step Hydrothermal Synthesis of C-N-S-Tridoped TiO2-based Nanosheets Photoelectrode for Enhanced Photoelectrocatalytic Performance and Mechanism. Electrochim. Acta 2015, 170, 182–190. [Google Scholar] [CrossRef]
- Guo, Y.; Guo, T.; Chen, J.; Wei, J.; Bai, L.; Ye, X.; Ding, Z.; Xu, W.; Zhou, Z. Synthesis of C–N–S Co-doped TiO2 Mischcrystal with an Isobandgap Characteristic and Its Photocatalytic Activity under Visible Light. Catal. Sci. Technol. 2018, 8, 4108–4121. [Google Scholar] [CrossRef]
- Wang, P.; Yap, P.-S.; Lim, T.-T. C–N–S Tridoped TiO2 for Photocatalytic Degradation of Tetracycline under Visible-Light Irradiation. Appl. Catal. A Gen. 2011, 399, 252–261. [Google Scholar] [CrossRef]
- Folli, A.; Jakobsen, U.H.; Guerrini, G.L.; Macphee, D.E. Rhodamine B Discolouration on TiO2 in the Cement Environment: A Look at Fundamental Aspects of the Self-Cleaning Effect in Concretes. J. Adv. Oxid. Technol. 2009, 12. [Google Scholar] [CrossRef]
- Eun, S.-R.; Mavengere, S.; Cho, B.; Kim, J.-S. Photocatalytic Reactivity of Carbon–Nitrogen–Sulfur-Doped TiO2 Upconversion Phosphor Composites. Catalysts 2020, 10, 1188. [Google Scholar] [CrossRef]
- Dong, F.; Zhao, W.; Wu, Z. Characterization and Photocatalytic Activities of C, N and S Co-Doped TiO2 with 1D Nanostructure Prepared by the Nano-Confinement Effect. Nanotechnology 2008, 19, 365607. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, Y.; Ho, W.; Zhang, L.; Zou, Z.; Lee, S. Biomolecule-Controlled Hydrothermal Synthesis of C–N–S-Tridoped TiO2 Nanocrystalline Photocatalysts for NO Removal under Simulated Solar Light Irradiation. J. Hazard. Mater. 2009, 169, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Lan, N.T.; Anh, V.H.; An, H.D.; Hung, N.P.; Nhiem, D.N.; Van Thang, B.; Lieu, P.K.; Khieu, D.Q. Synthesis of C-N-S-Tridoped TiO2 from Vietnam Ilmenite Ore and Its Visible Light-Driven-Photocatalytic Activity for Tetracyline Degradation. J. Nanomater. 2020, 1–14. [Google Scholar] [CrossRef]
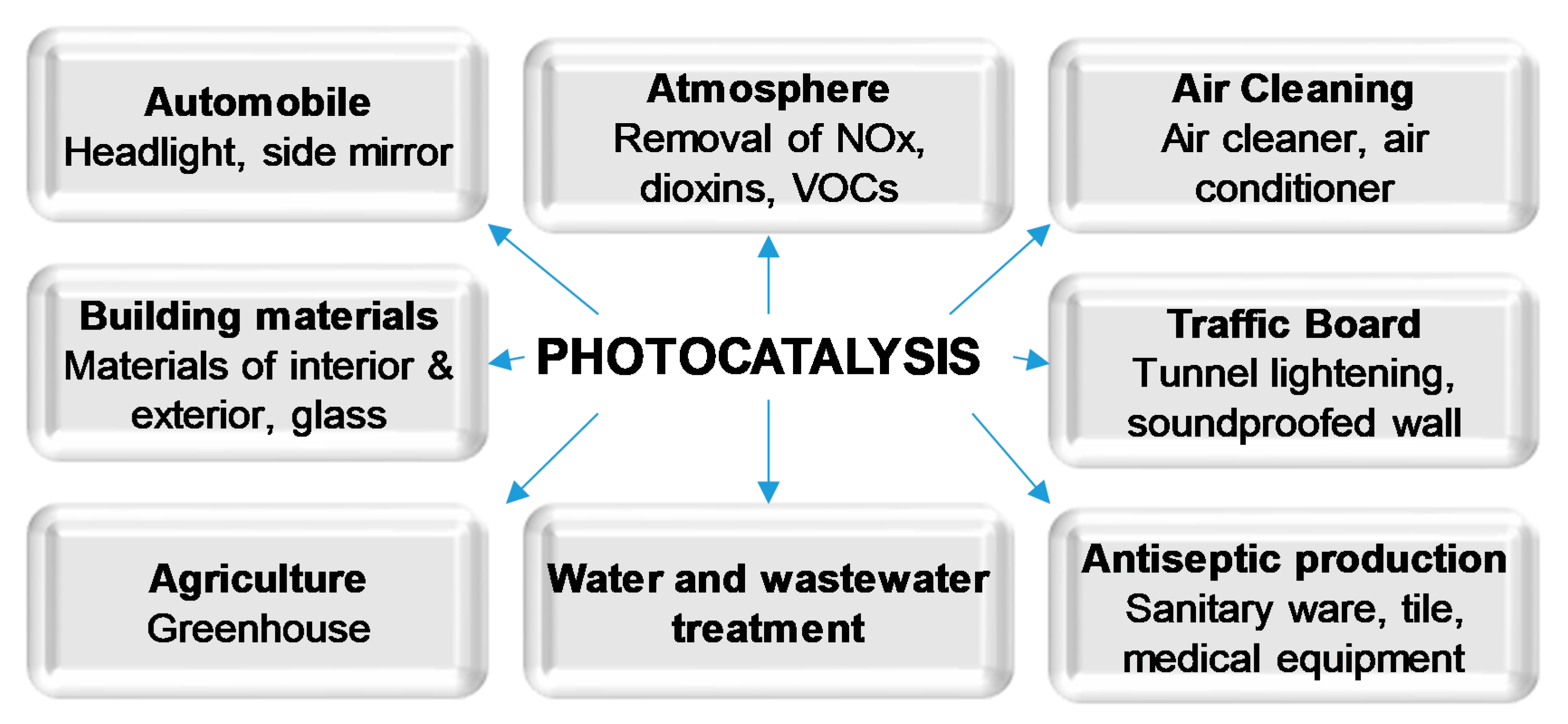
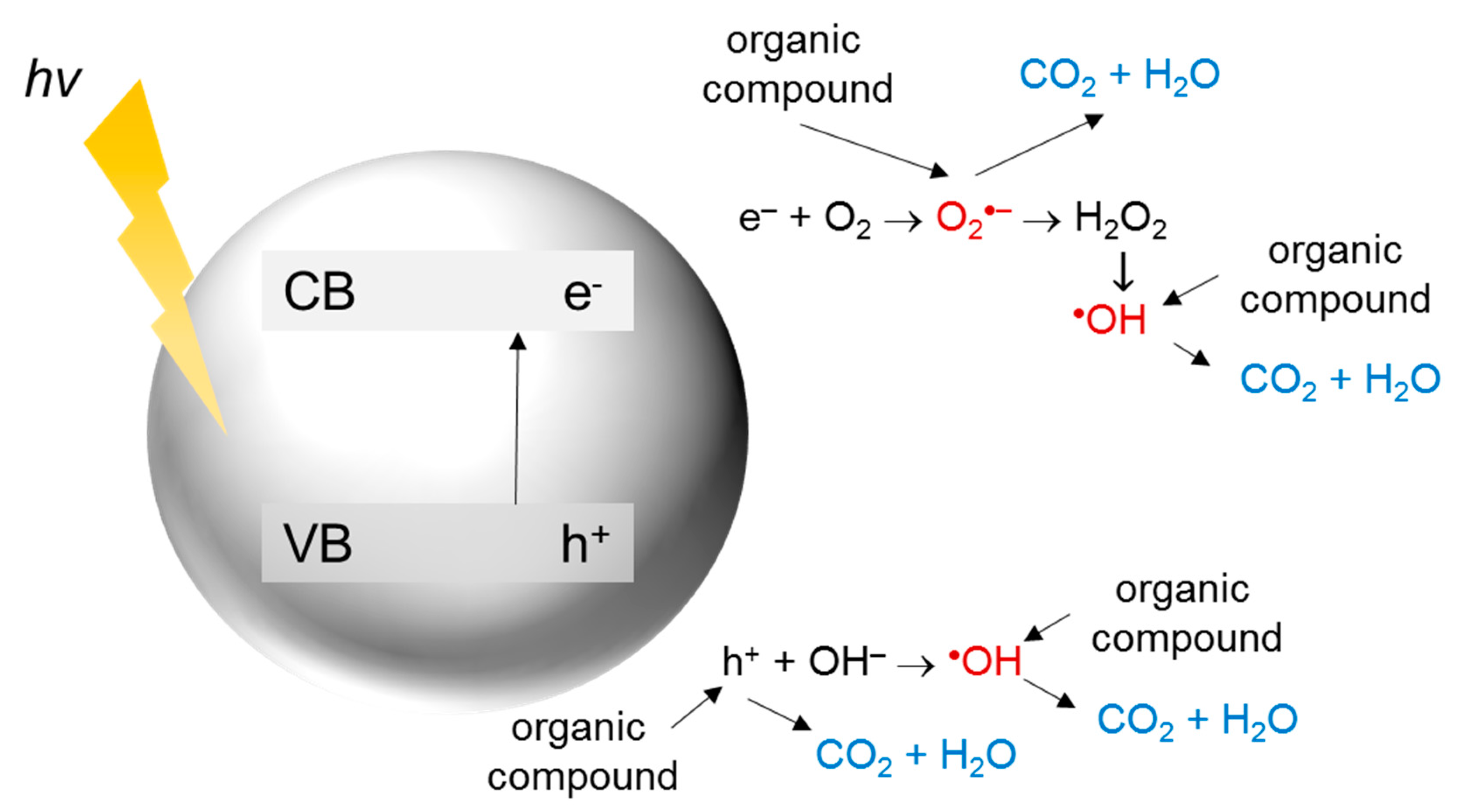
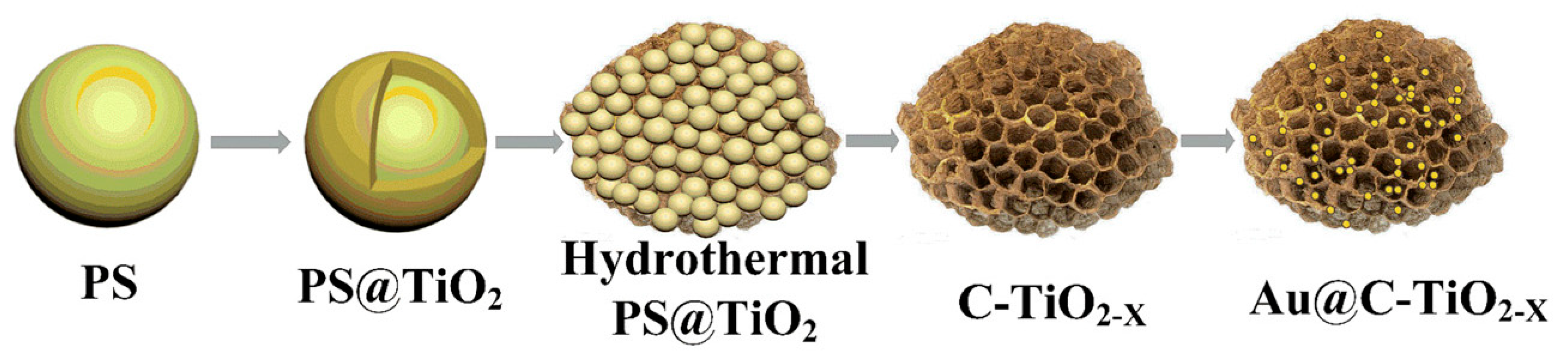
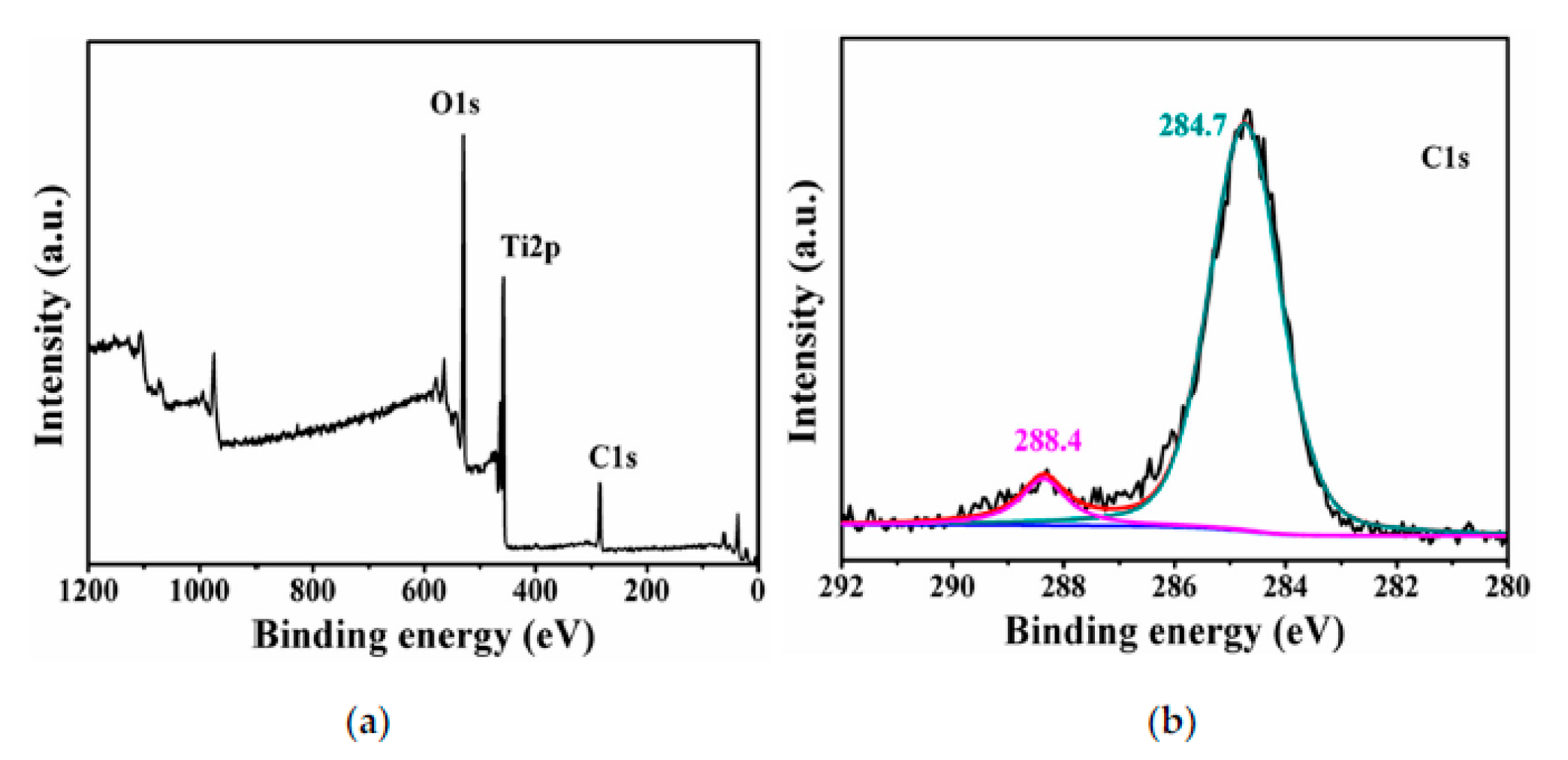
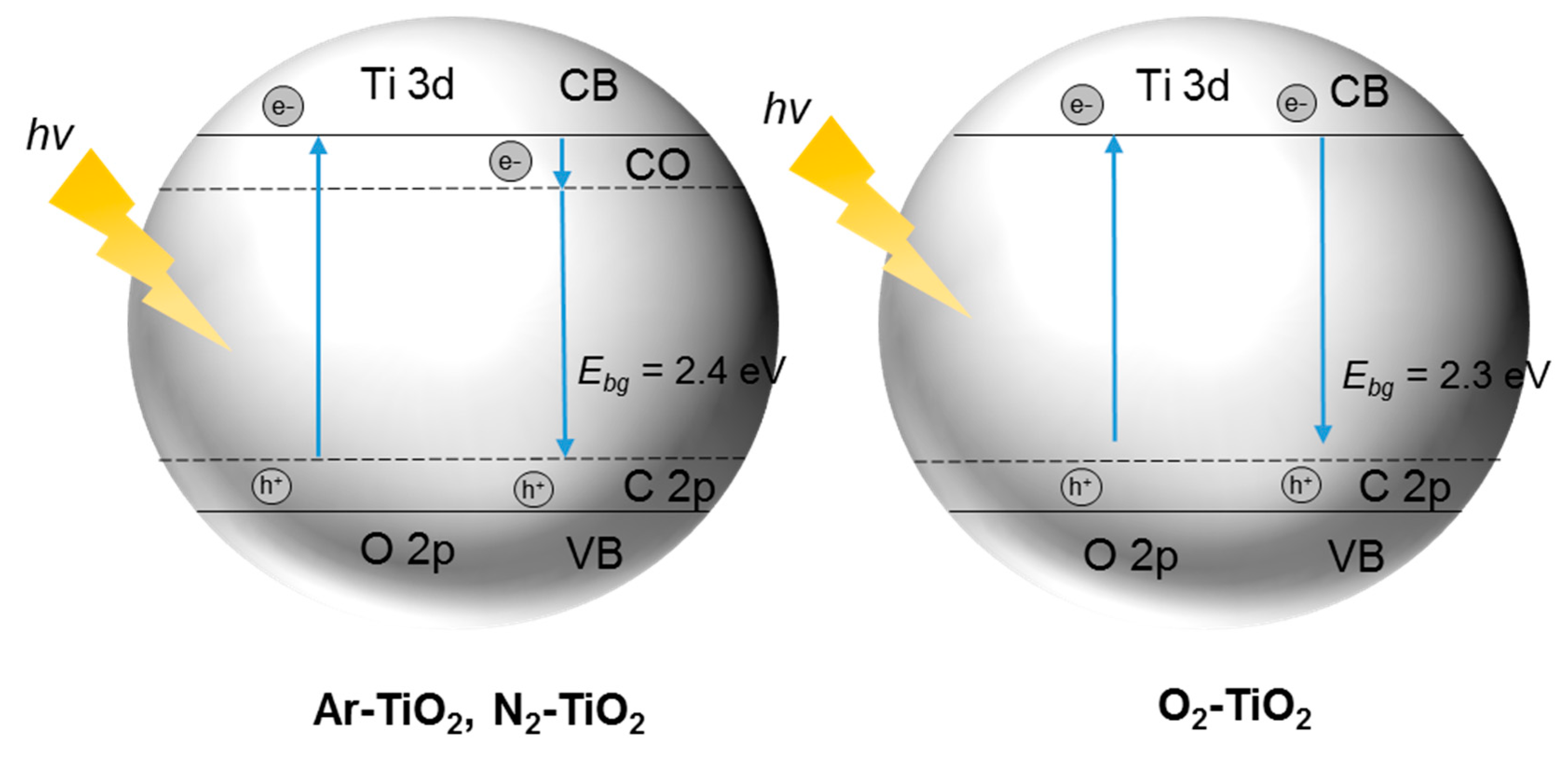
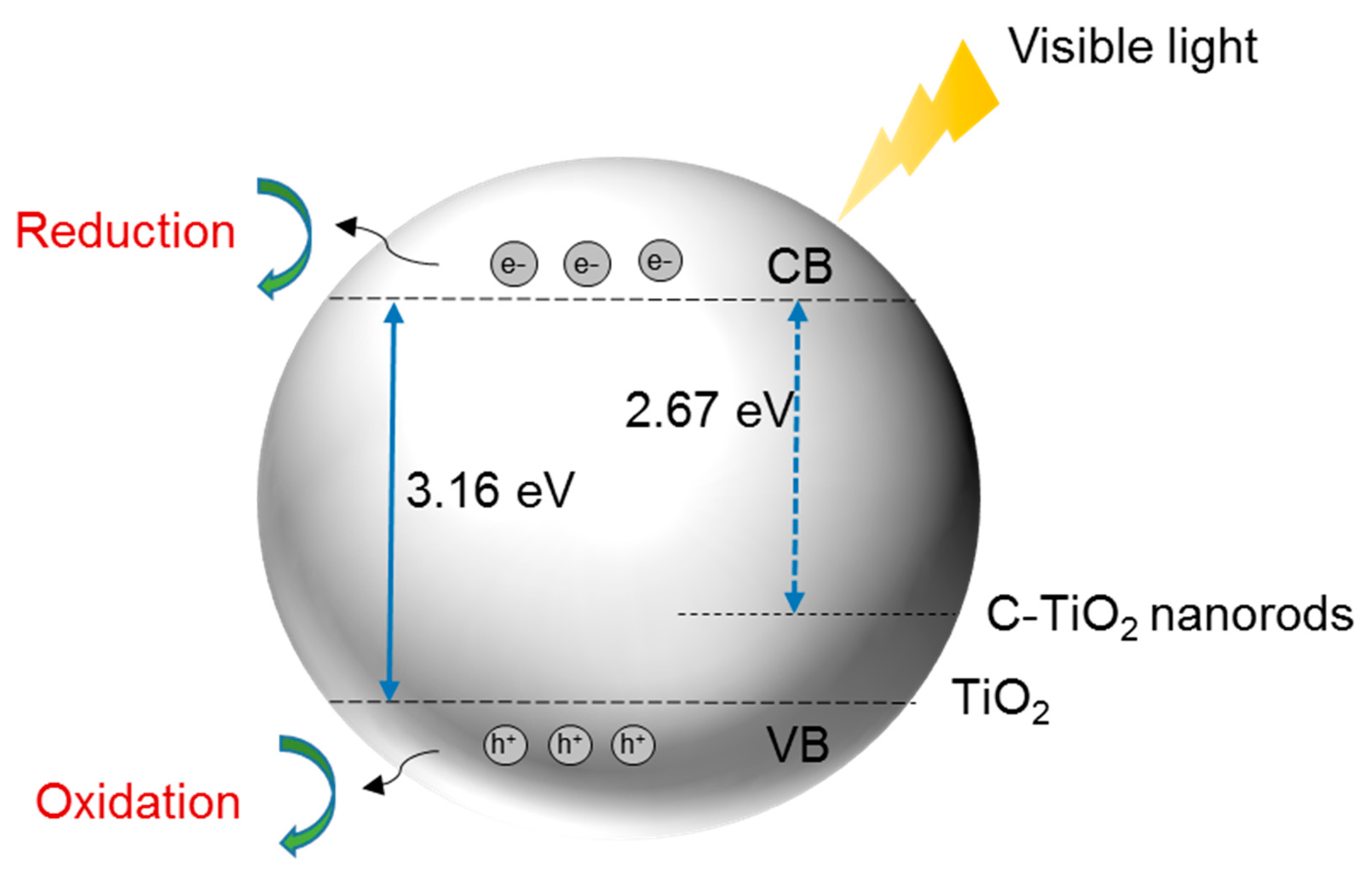
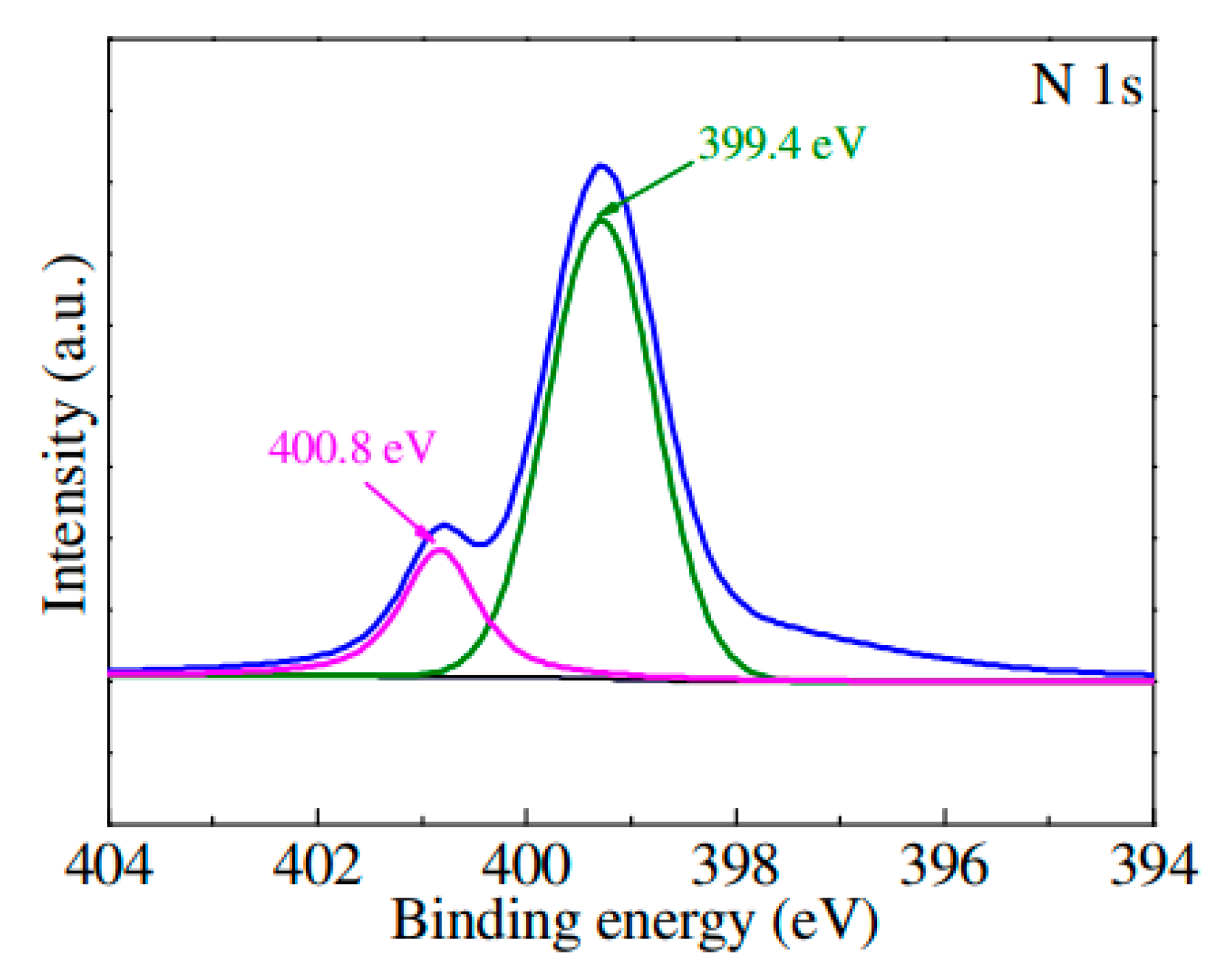
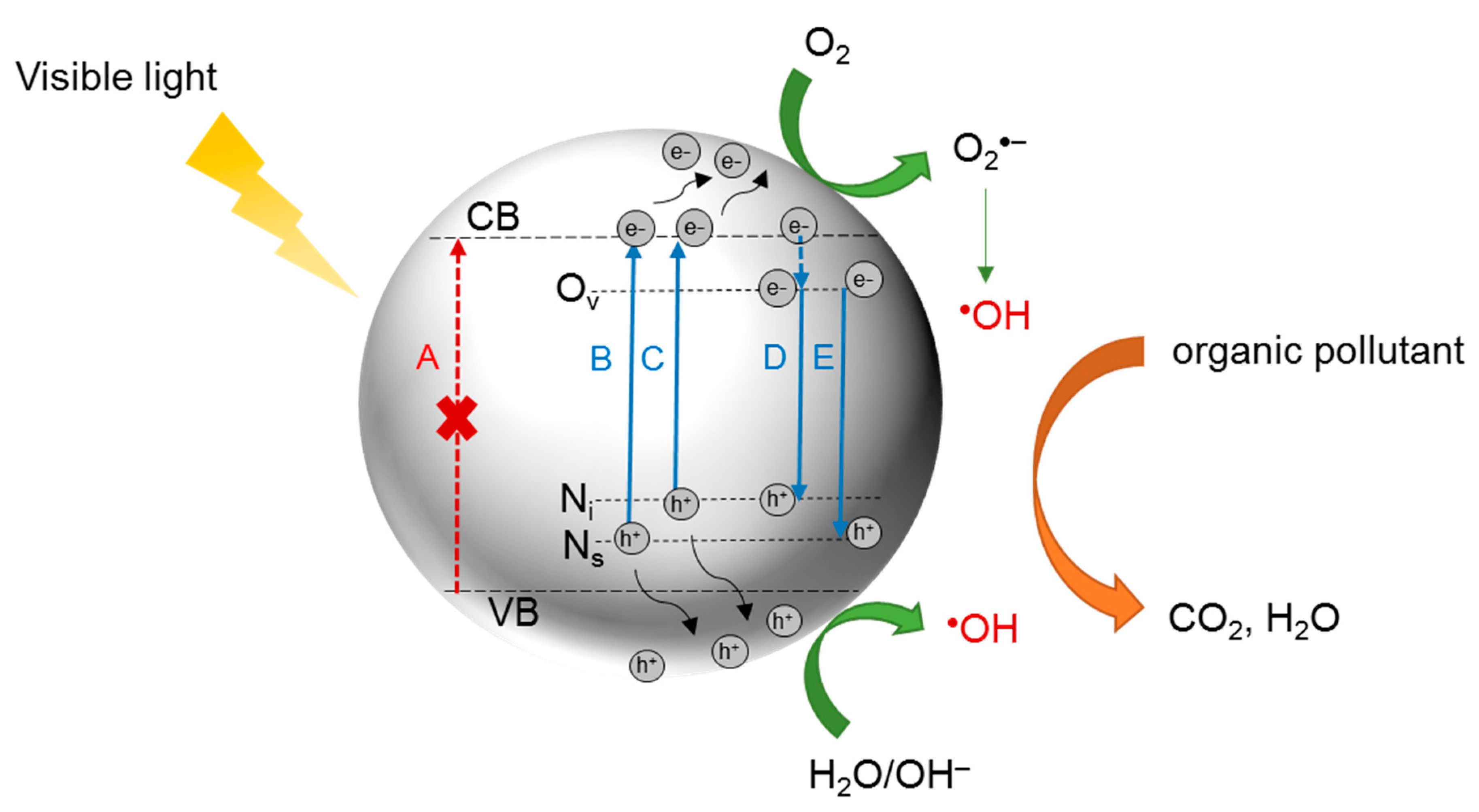
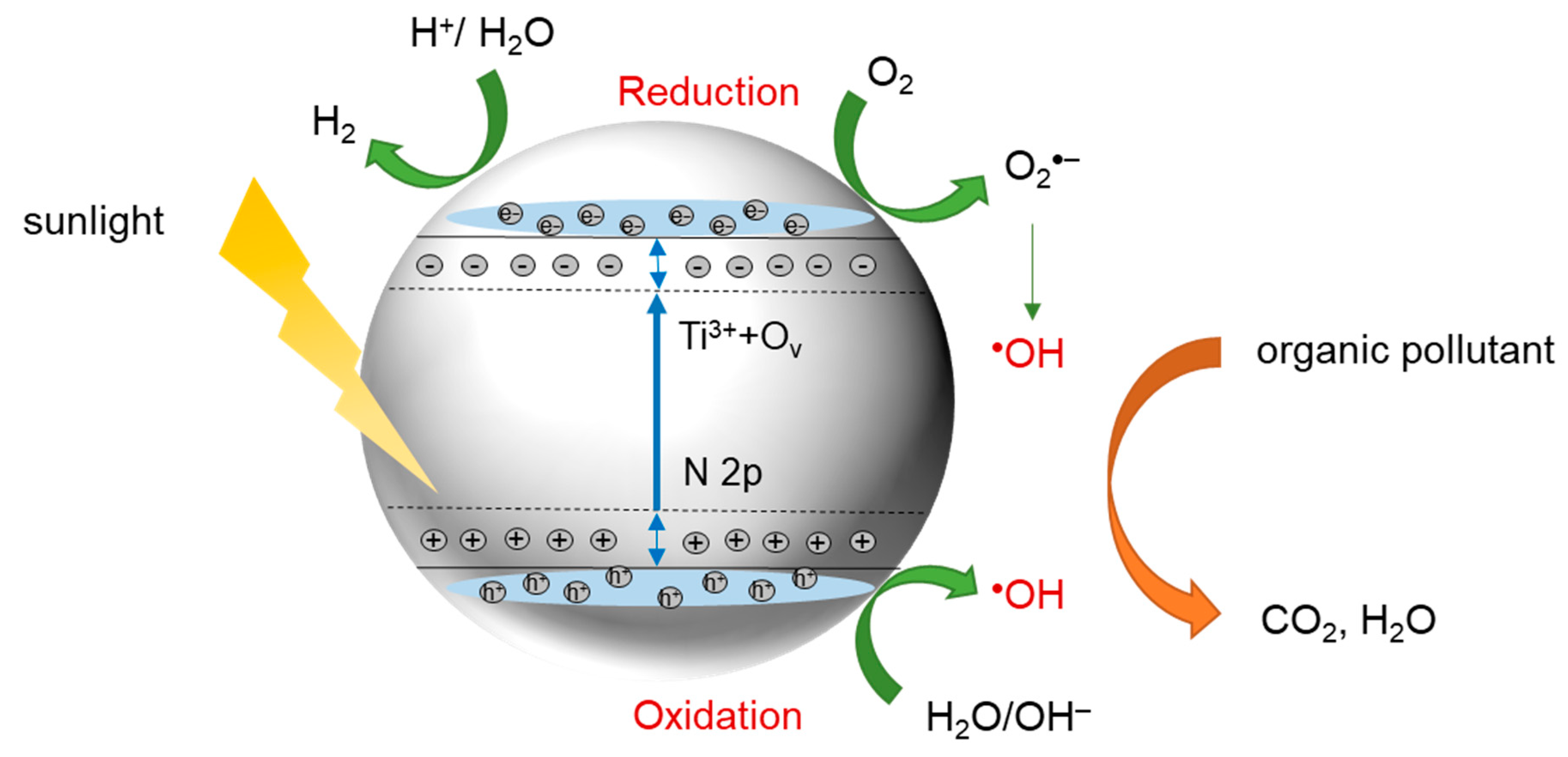
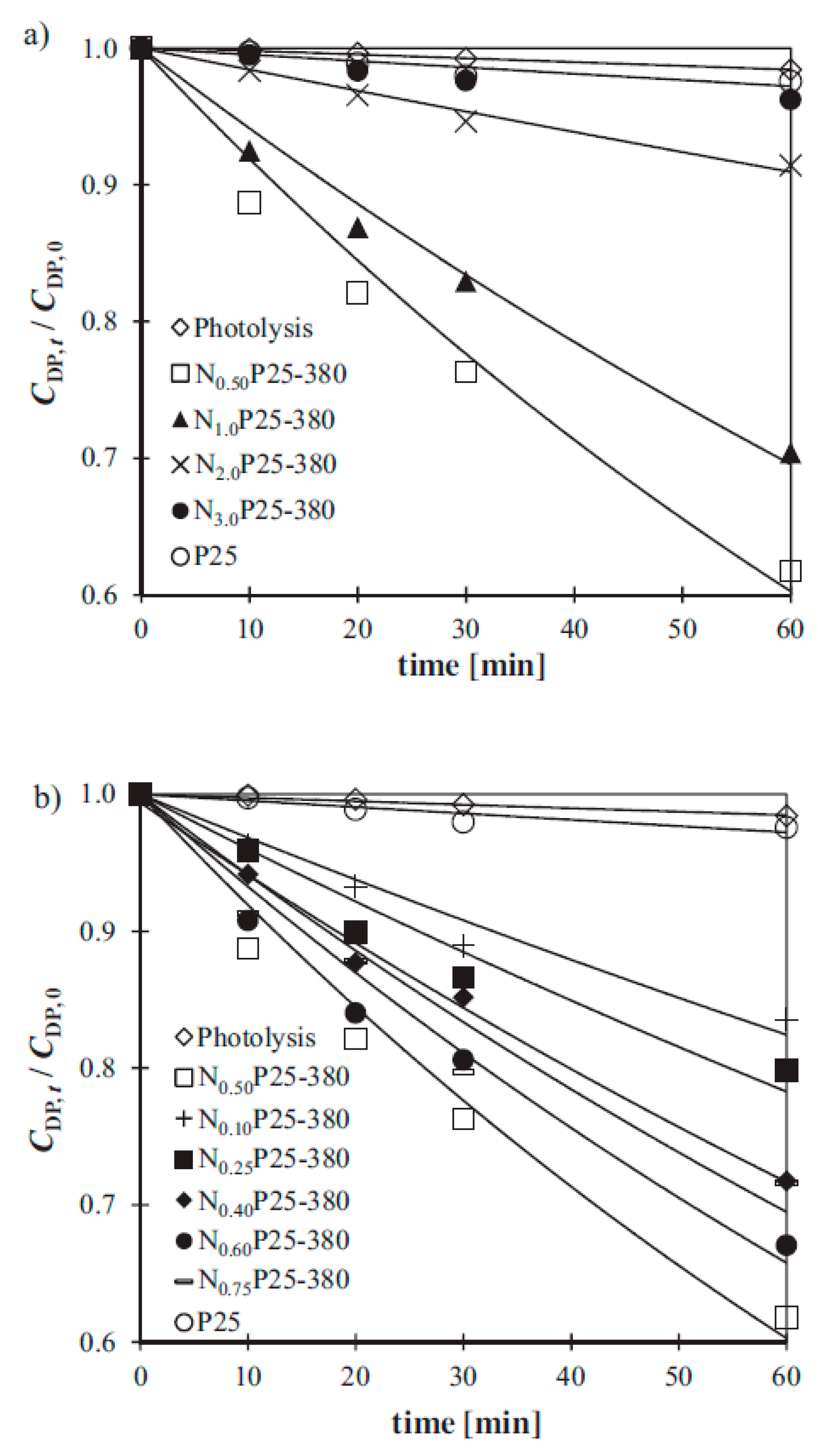
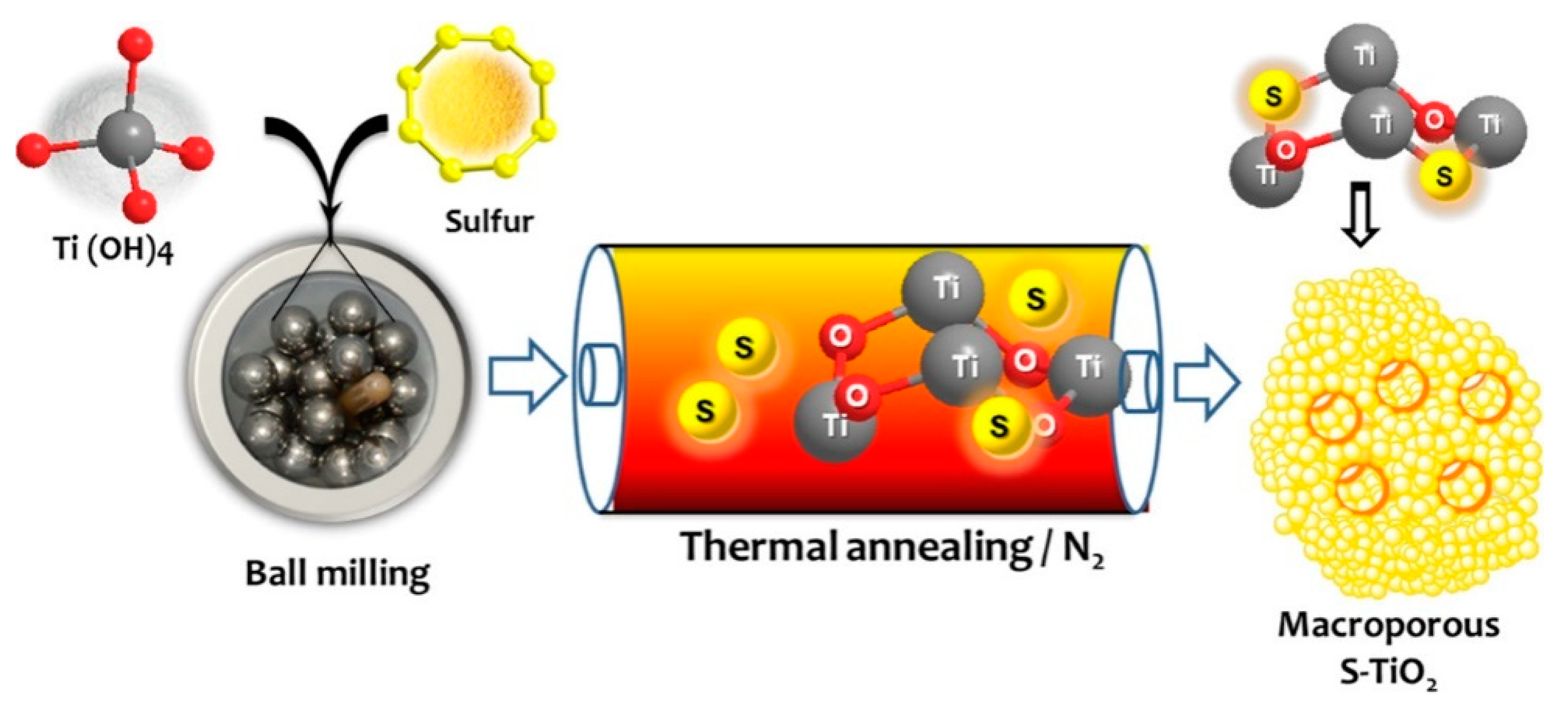
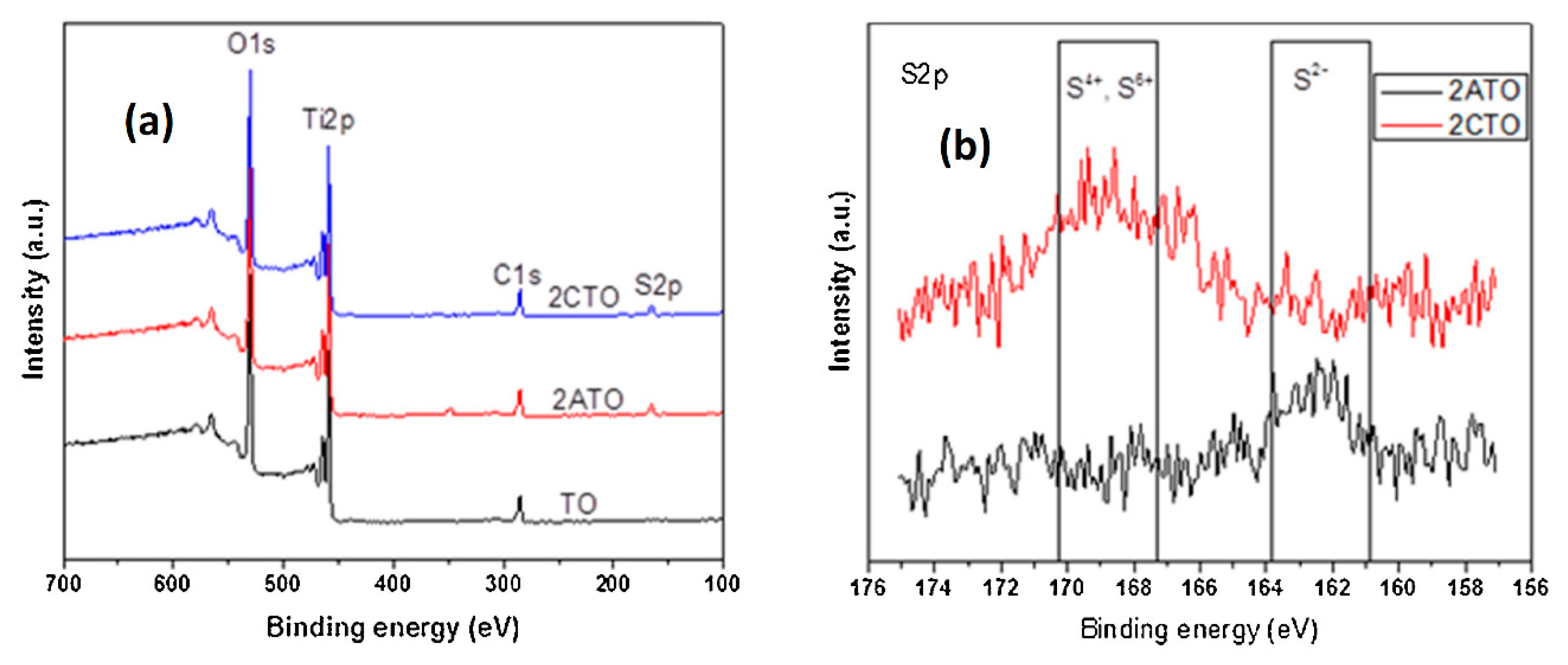
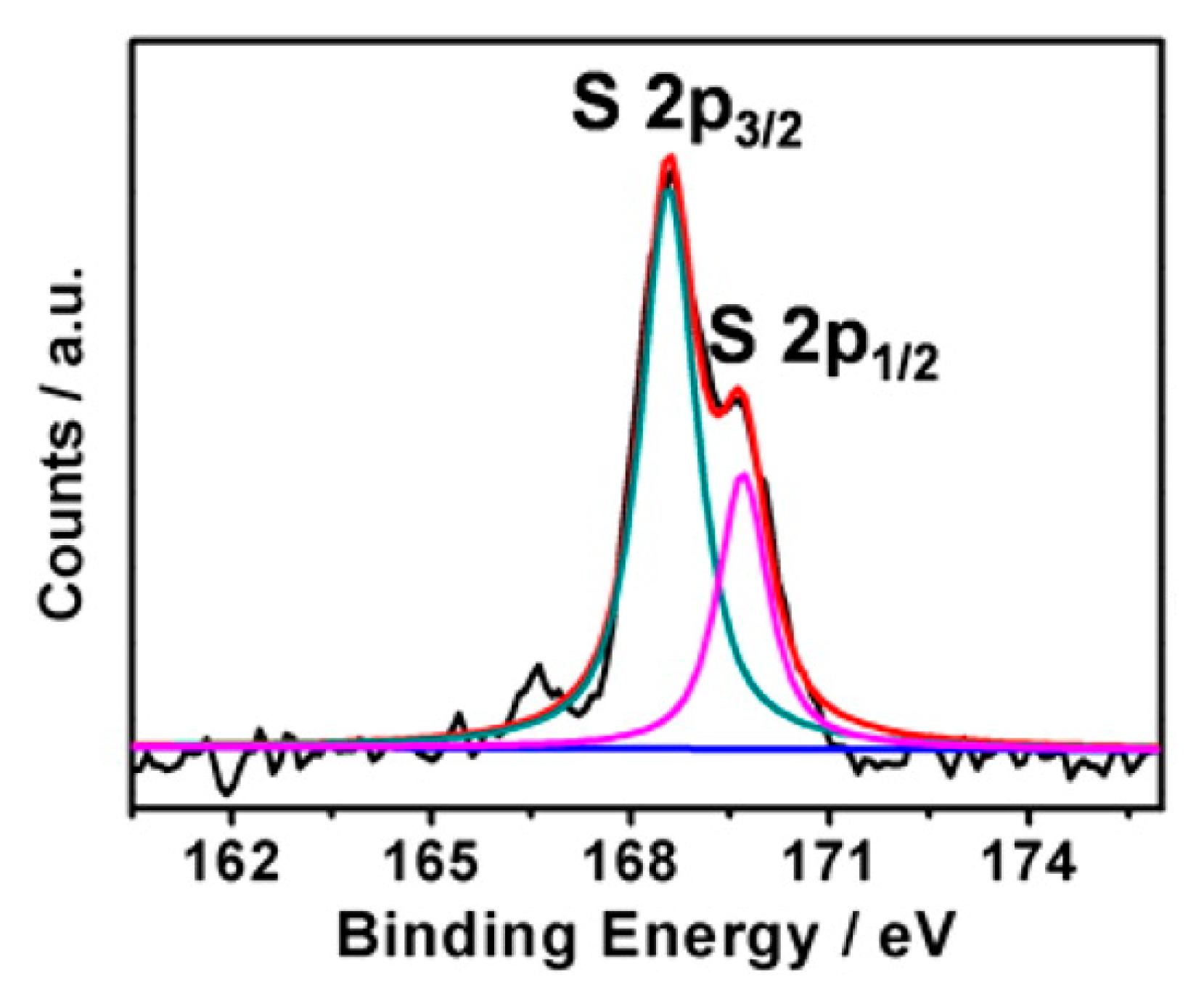
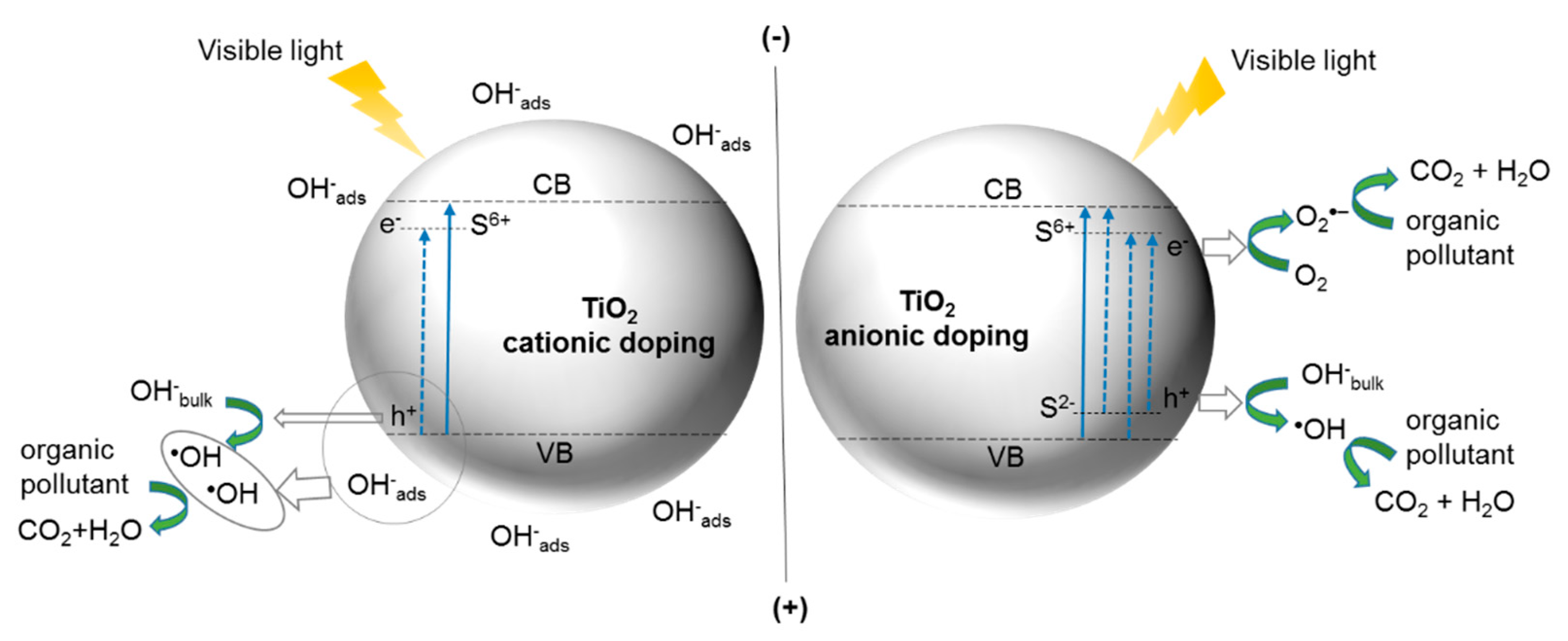
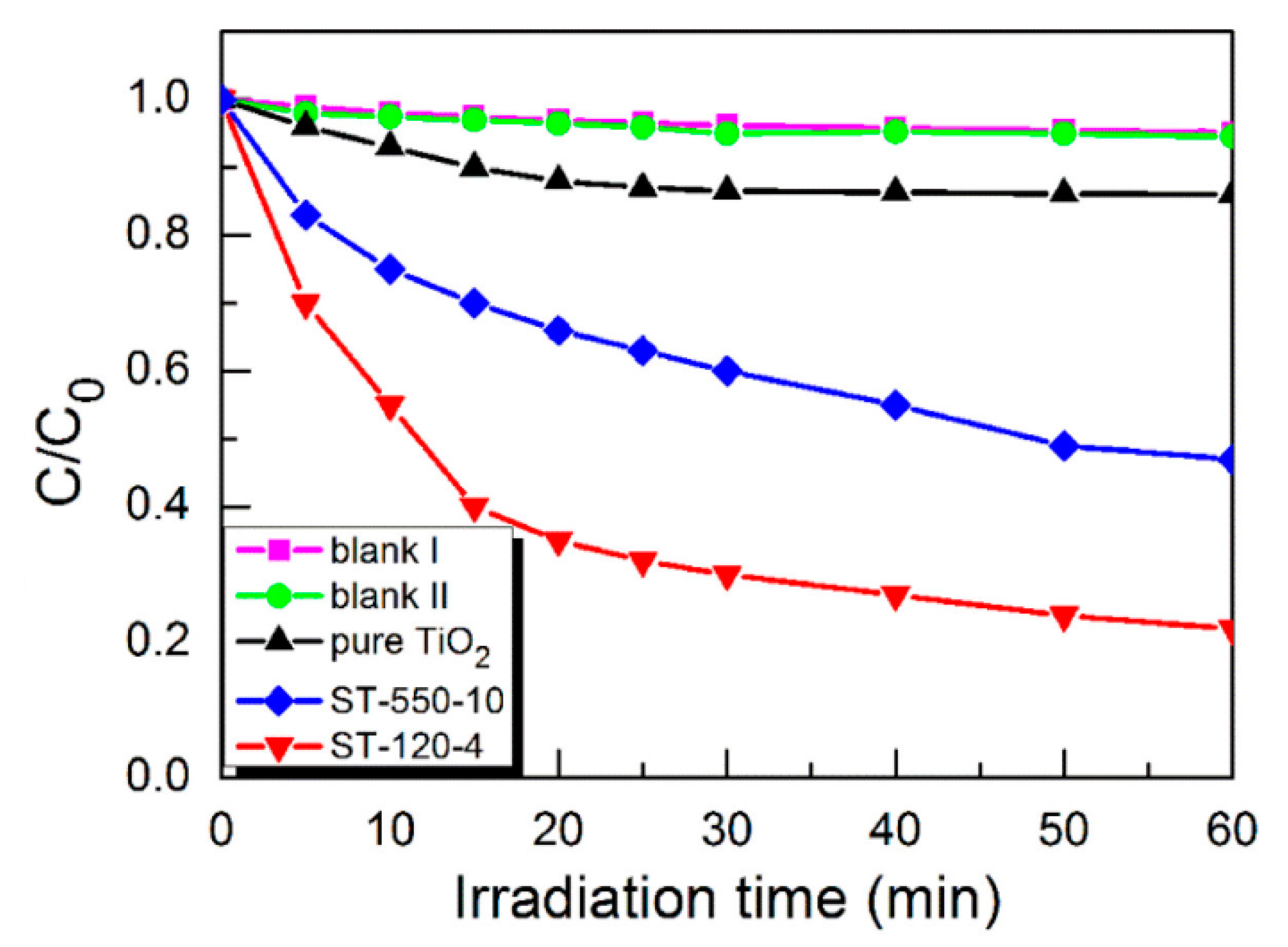
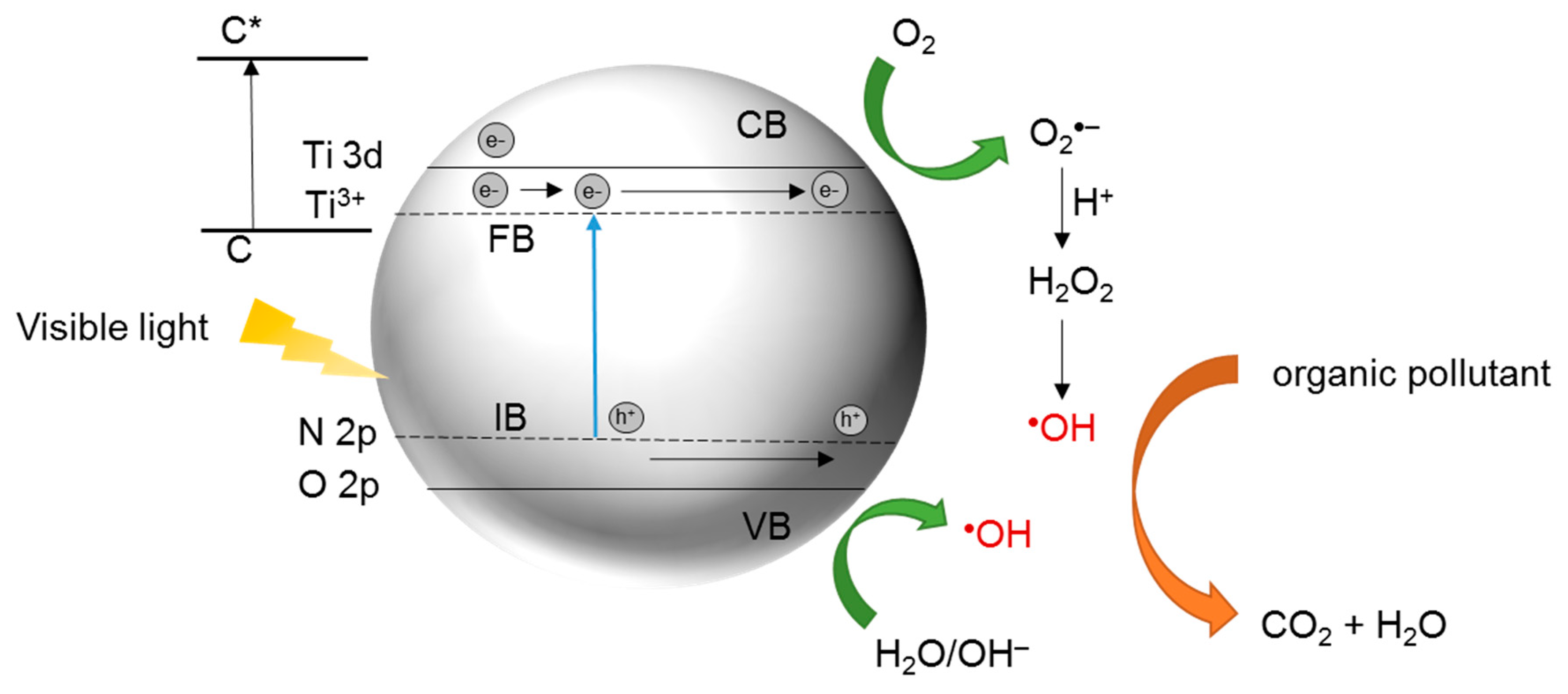
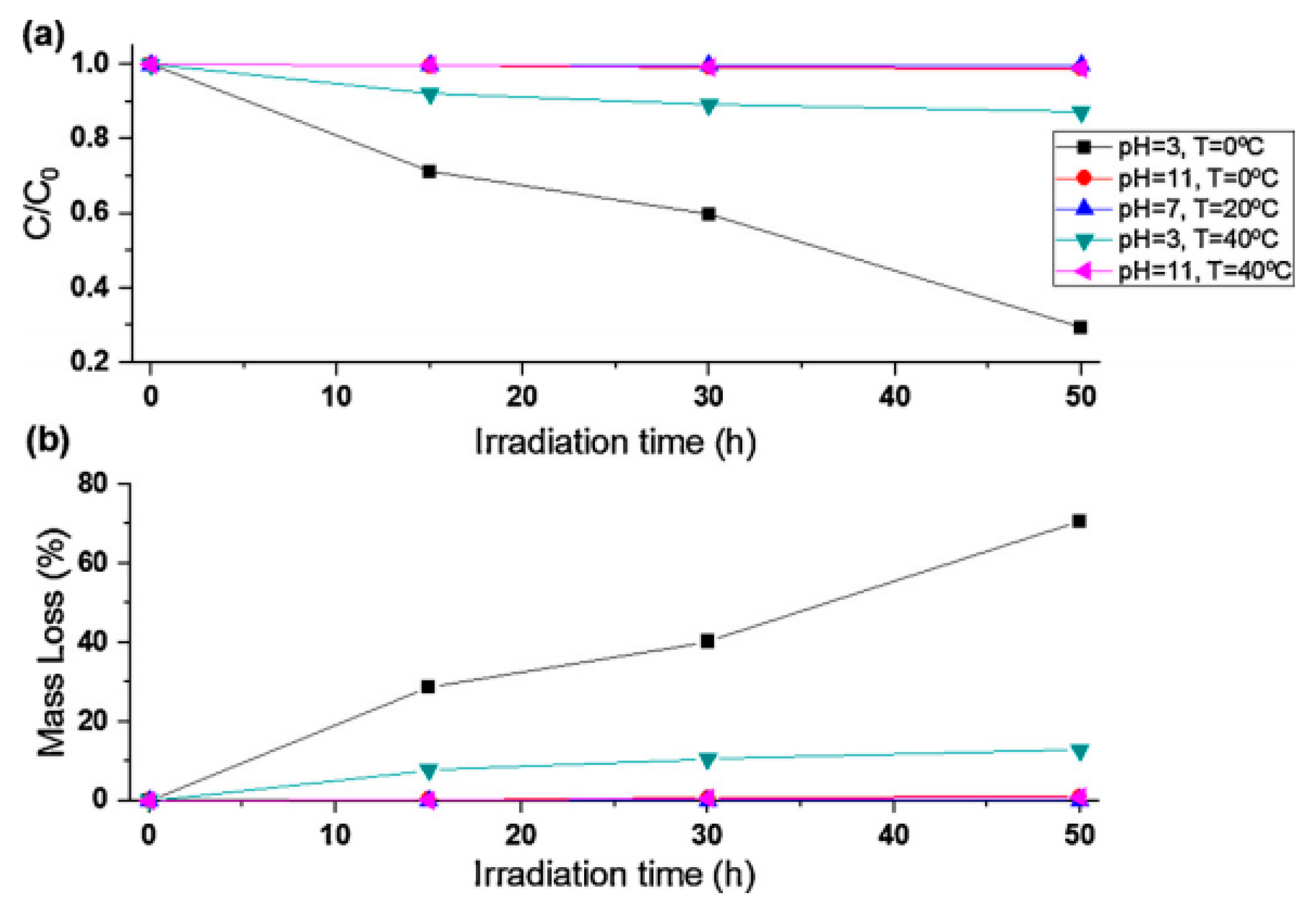
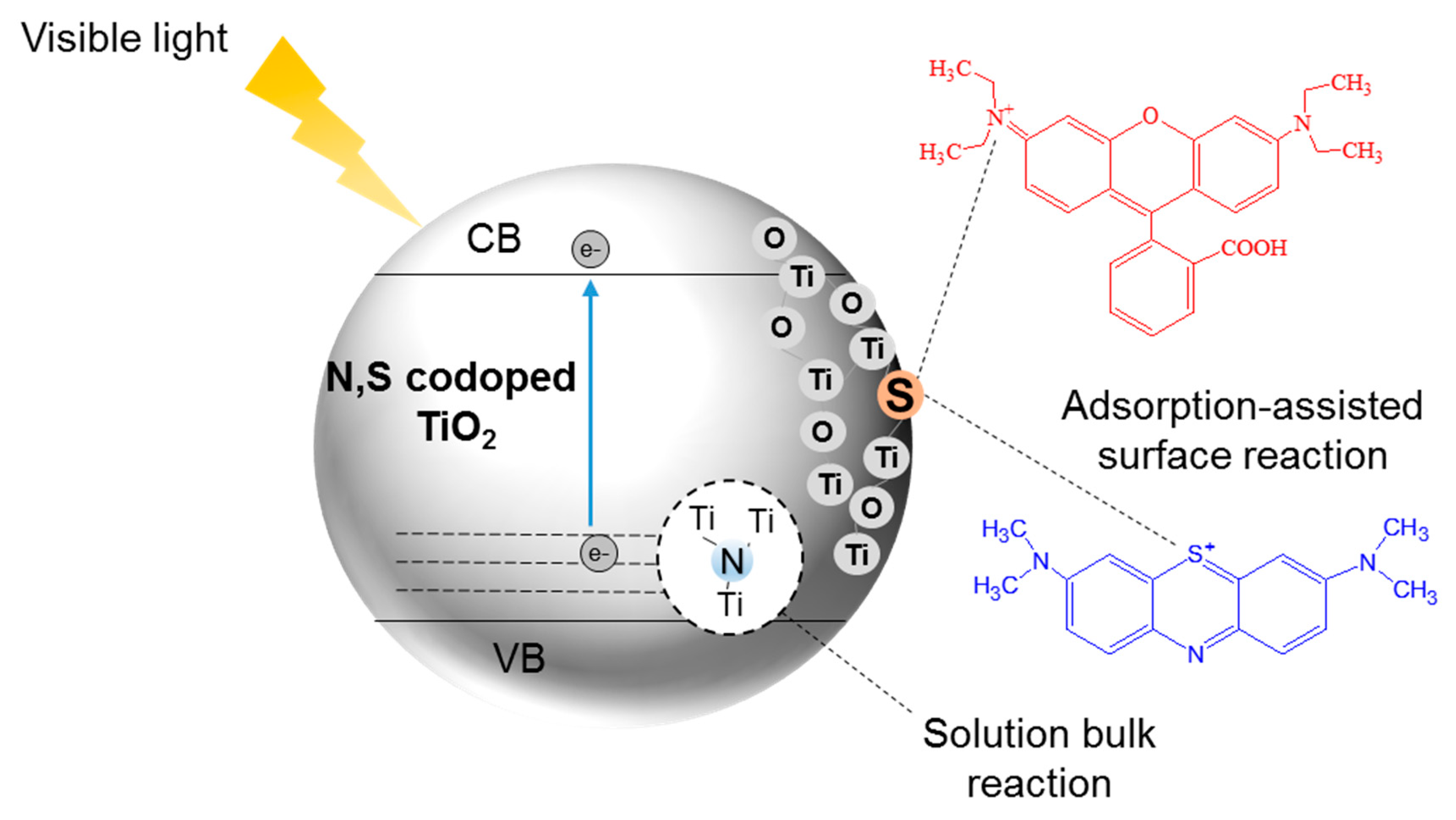
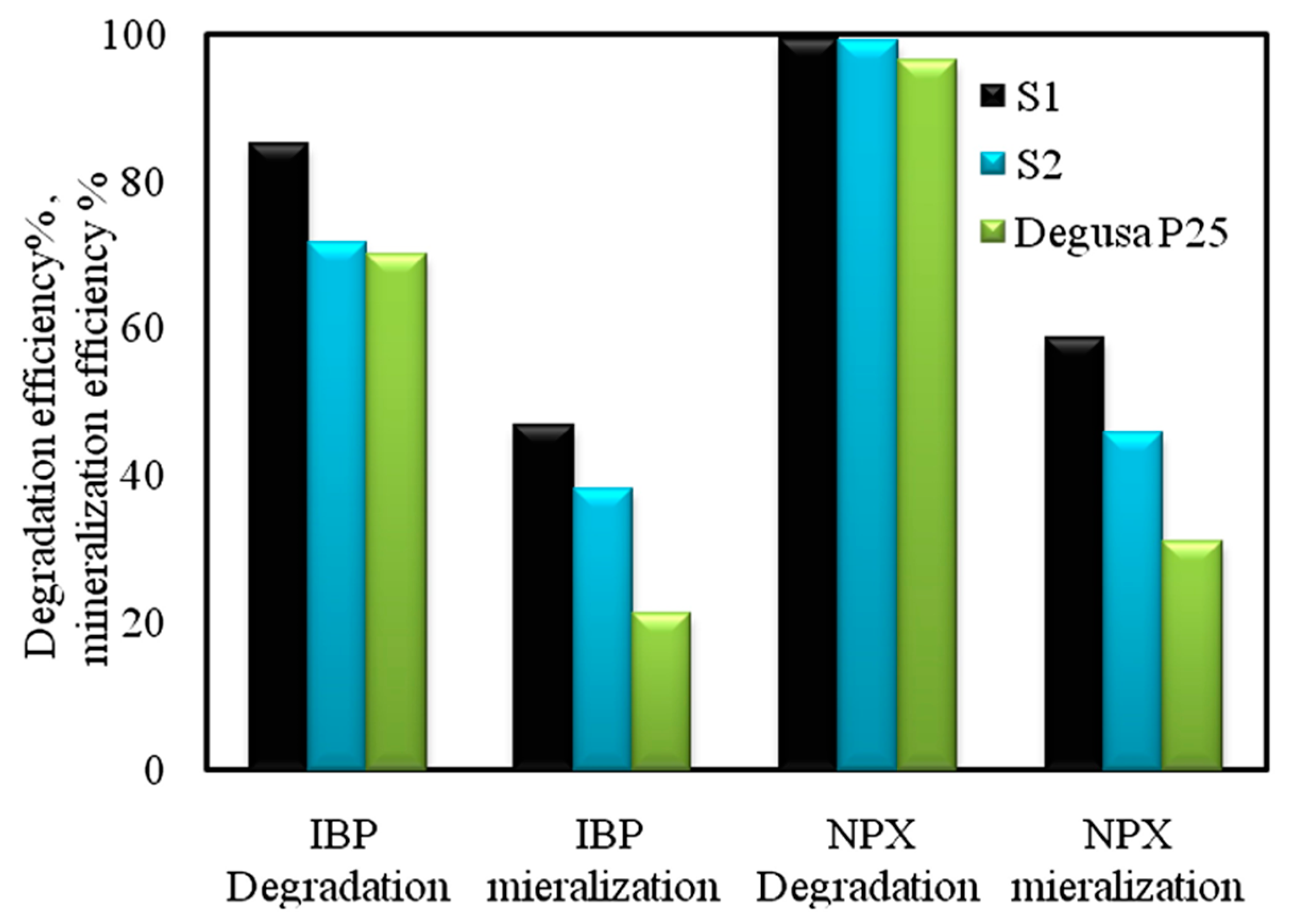
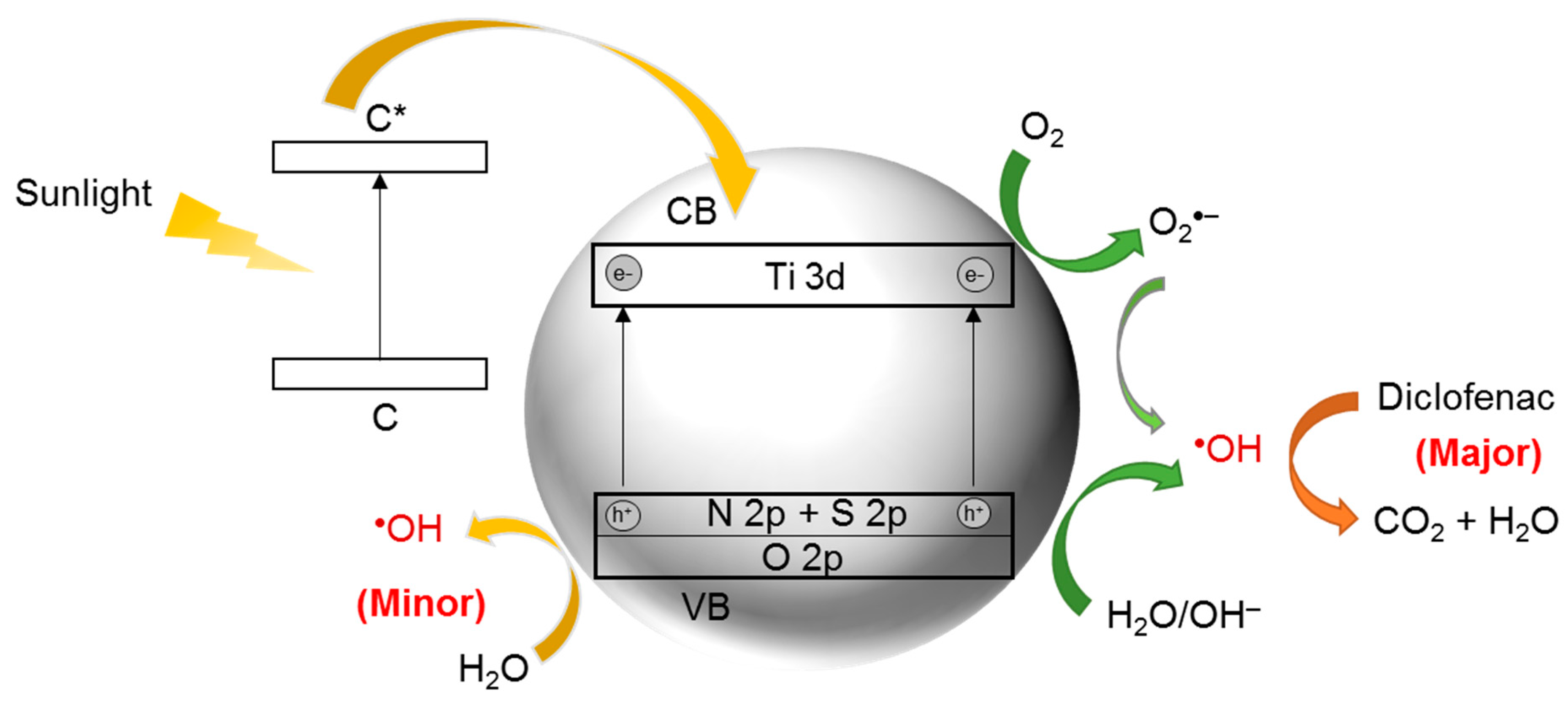
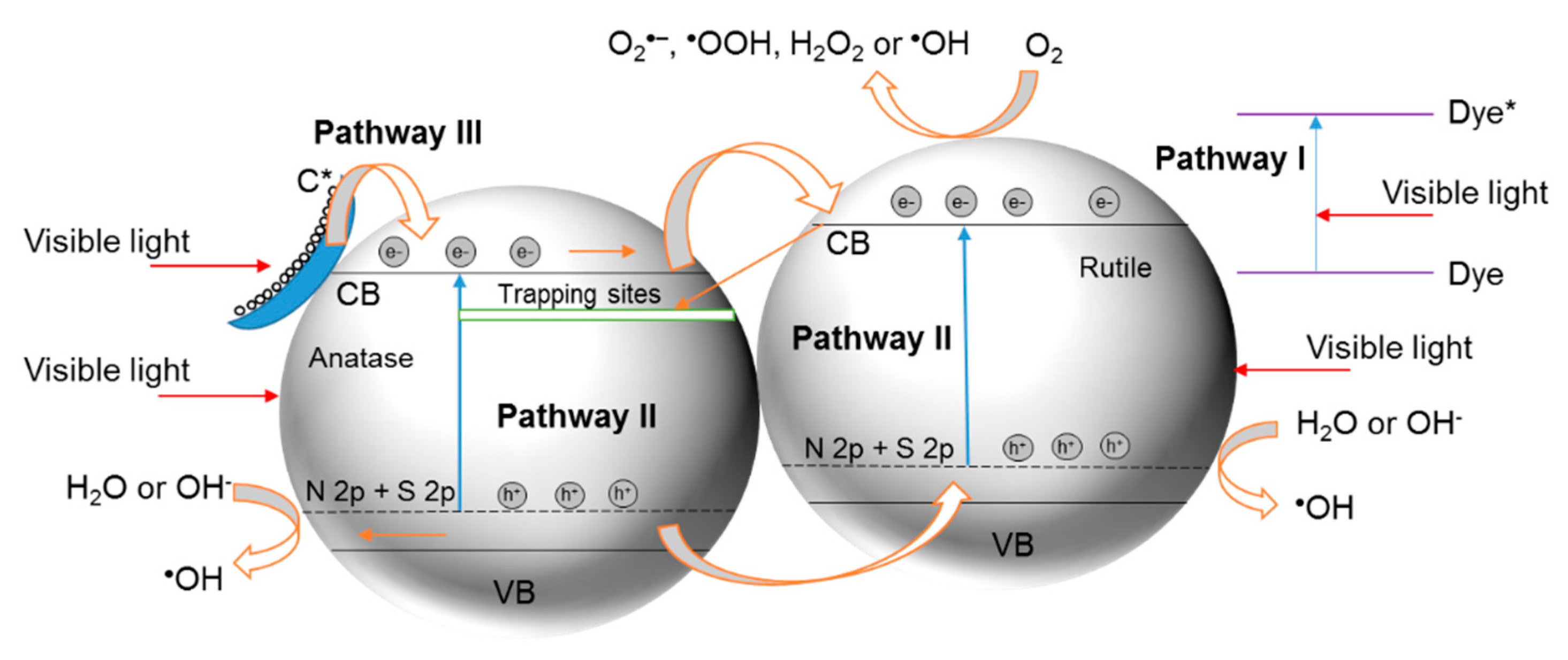
| Method | TiO2 Precursor | Carbon Source | References |
|---|---|---|---|
| Biomimetic template approach | TBOT | Banana stem fibers | [78] |
| Calcination | Aeroxide® TiO2 P25 from Evonik, Germany (TiO2 P25) | Polyethylene glycol | [79] |
| Chemical bath deposition (CBD) | Titanium isopropoxide (TTIP) | Melamine borate | [80] |
| Direct solution-phase carbonization | TiCl4 | Diethanolamine (DEA) | [81] |
| Electrochemical anodization | Ti foils | Ascorbic acid | [82] |
| Electrospinning combining sol-gel | TBOT | Polyvinyl-pyrrolidone (PVP) | [83] |
| Electrospinning followed by heat treatment | TTIP | Acetic acid | [84] |
| Hydrolysis | TBOT | Glucose | [85] |
| Hydrothermal | TiC | TiC | [69] |
| Hydrothermal treatment and calcination | Ti(SO4)2 | Glucose | [86] |
| Incipient wetness impregnation | TiO2 P25 | Glucose | [87] |
| Oxidative annealing | TiC | TiC | [88] |
| Sol impregnation and carbonization | TBOT | Acetic acid | [84] |
| Sol-gel | TTIP, TBOT, TiCl4, TiCl3 | Ethanolamine (ETA), Glycine, Polyacrylonitrile (PAN), Polystyrene (PS), Starch, TBOT | [76,77,82,89,90,91,92] |
| Sol-gel and calcination | TBOT | Butterfly wings | [93] |
| Sol-gel bio-templating | TBOT | Cellulose | [94] |
| Sol-microwave | TBOT | Cellulose | [94] |
| Solution combustion synthesis | TTIP | Citric acid | [95] |
| Solvothermal | TTIP | Acetone | [71] |
| Solvothermal treatment and calcination | TiCl4 | Alcohols (benzyl alcohol and anhydrous ethanol) | [75] |
| Thermal decomposition | TTIP | Oleylamine | [96] |
| Method | TiO2 Precursor | Nitrogen Source | References |
|---|---|---|---|
| Addition of N source to the TiO2 precursor solution | TBOT | Tetramethyl-ethylene-diamine | [113] |
| Bioprocess | TBOT | Extrapallial fluid of fresh blue mussels | [114] |
| Centrifugal spinning | Titanium diisopropoxide bis(acetyl-acetonate) | PVP | [115] |
| Colloidal crystal-templating | TBOT | Ethanediamine | [116] |
| CVD | TiCl4 | tert-butylamine, benzylamine | [117,118] |
| Electrochemical | Titania nanotubes | Diethylenetriamine, ethylenediamine, hydrazine | [113,119,120,121] |
| Electrochemical anodization | Titanium foils | Ammonium fluoride | [122] |
| Hydrolysis | TTIP | NH4Cl, pyridine | [123,124,125] |
| Hydrothermal | TBOT | KNO3 | [126] |
| Hydrothermal treatment of TiN with H2O2 | TiN | TiN | [127] |
| Incomplete oxidation of titanium nitride | TiN | TiN | [128] |
| L-alanine acids assisted process | TTIP | L-alanine acids | [129] |
| Low temperature non-aqueous solvent-thermal method | TiCl4 | NH4Cl | [130,131] |
| One-pot hybridization | TTIP | Ethylenediaminetetraacetic acid disodium salt (EDTA-Na2) | [132] |
| Radio-frequency magnetron reactive sputtering | TiOx film | N(NOx) | [133] |
| Sol-gel | TTIP, TBOT, TiCl4, Titanic acid | Urea, NH3, nitromethane, n-butylamine, N2, hydrazine, HNO3, guanidinium chloride, ethylenediamine, diethanolamine, NH4NO3, NH4Cl, (NH4)2CO3 | [119,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151] |
| Solvent-based ambient condition sol process | TiCl4 | N,N-dimethylformamide (DMF) | [152] |
| Solvothermal | TBOT, TiCl4 | Trimethylamine (TEA), PVP, hydrazine, diethylamine | [102,153,154,155,156,157,158] |
| Sonication of aqueous solution | TiO2 P25 | 4-nitrophenol (4-NP) | [159] |
| Sonochemical | TiO2 P25, TBOT,TTiP | Sodium amide, hydroxylamine hydrochloride, urea | [160,161,162] |
| Surfactant-free solvothermal treatment | TBOT,TiCl4 | Carbamide | [163] |
| Two cycle microwave-assisted hydrothermal | TTIP | Diaminohexane | [164] |
| Photocatalyst | dTiO2 a (nm) | SBET b (m2 g−1) | Ebg c (eV) |
|---|---|---|---|
| N-TiO2 (urea) | 10.8 | 79 | 3.03 |
| N-TiO2 (TEA) | 11.5 | 12 | 2.89 |
| N-TiO2 (NH3) | 18.0 | 29 | 3.01 |
| Photocatalyst | Calcination Temperature (°C) | Anatase (%) | Rutile (%) | Ebg (eV) | Decomposition of Ph after 540 min (%) | |
|---|---|---|---|---|---|---|
| Visible Light | UV Light | |||||
| T75 | - | 95.0 | 5.0 | 2.80 | 81.07 | 68.00 |
| T200 | 200 | 52.8 | 47.2 | 2.54 | 86.92 | 84.00 |
| T400 | 400 | 38.3 | 61.7 | 2.50 | 99.16 | 99.60 |
| T600 | 600 | 12.0 | 88.0 | 2.60 | 95.28 | 92.00 |
| T800 | 800 | 0 | 100 | 3.06 | 53.99 | 44.88 |
| TAA | - | 100 | 0 | 3.85 | 5.21 | 57.86 |
| TiO2 P25 | - | 72 | 28 | 3.24 | 5.21 | 80.64 |
| Method | TiO2 Precursor | Sulfur Source | References |
|---|---|---|---|
| Ball milling | TiO2 P25 | Sulfur powder, thiourea | [245] |
| Electrochemical anodization | Titanium sheet | H2S, K2S2O5 | [259,262] |
| Flame spray pyrolysis | TTIP | H2SO4 | [261] |
| Hydrolysis | TiCl4, TTIP, TiC | Na2SO4, thiourea | [242,263,269] |
| Hydrothermal | TiH2, TiS2, TTIP, TiOSO4 | Thiourea dioxide, thiourea, TiS2, TTIP, TiOSO4 | [242,243,251,254,256,257] |
| Micro-plasma oxidation | Titanium sheet | Thiourea | [244] |
| Oxidant peroxide method (OPM) | Metallic Ti | CS2 | [236] |
| Oxidation annealing | TiS2 | TiS2 | [258,270] |
| Sol-gel | TBOT, TTIP | Thiourea | [102,242,245,271] |
| Solvothermal | TBOT, TTIP | DMSO, thioacetamide | [248,250] |
| Sonothermal | TBOT | Sulfur powder | [266] |
| Thermochemical treatment | TTIP | H2S | [260] |
| Ultrasound application | TTIP, TiOSO4 | EDT, TiOSO4 | [249,254] |
| Wet impregnation | TiO2 P25 | Sodium thiosulfate | [268] |
| EDT/TiO2 Molar Ratio | Phase Composition | |
|---|---|---|
| Anatase (%) | Rutile (%) | |
| 1:0.1 | 11.9 | 88.1 |
| 1:0.5 | 58.2 | 41.8 |
| 1:1 | 89.5 | 10.5 |
| Titania Precursor | Preparation Method | Treatment Temperature (°C) | S Content | Binding Energy (eV) | SBET (m2 g−1) | Ebg (eV) | Irradiation Source | Initial Concentration of MB (mg L−1) | Degradation Rate | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| TTIP | Sol-gel | 600 | - | - | 47.6 | - | Household lamp (2.5 W/m2) | 10 | 71.83%, 6 h | [276] |
| Hydrolysis | 120 | 0.64–0.80 wt.% | - | - | - | 150 W high-pressure Xenon lamp | 10 | Up to 59%, 4 h | [277] | |
| Vapor-thermal | 250 | - | 162.2 (S2−), 164.0 (S), 165.2 (S4+), 168.6 (S6+) | 99.1–125.2 | - | 300 W Xenon lamp | 10 | 78.4–99.9%, 1 h | [278] | |
| TBOT | Ultrasonic-assisted spray pyrolysis | 400 | - | - | - | 2.85 | 570 W Xenon lamp | 3.2 | 80%, 5 h | [225] |
| Thermal CVD | 180 and 250 | S/Ti molar ratio of 0.5, 1, 2, 3, 4, and 5 | 163.5 (S2−), 164.4 (S), 168.0 (S6+) | 81.7–210.9 | 2.4 | 300 W Xenon lamp | 10 | 92.5–99.8%, 2 h | [226] | |
| TiCl4 | Sol-gel | 500 | 4 mol% | 173.1 (S6+) | - | - | 350 W Xenon lamp | 9 | ~60%, 100 min | [238] |
| Ti(OH)2O | Thermal hydrolysis | 600 | Ti(OH)2O/thiourea molar ratio of 1:3 | - | - | 2.56 | Visible light | 8 | ~70%, 6 h | [241] |
| Metallic Ti | Low temperature and template free OPM | 200 | Ti/S molar ratio of 1:1, 1:2 and 1:3 | 168.4 (S6+) | - | 2.89 | 18 W Daylight lamp | 10 | Up to 92%, 4 h | [235] |
| Photocatalyst | Ebg (eV) | Average Particle Size by TEM (nm) |
|---|---|---|
| TiO2 P25 | 3.22 | 33.0 |
| S:Ti = 0.5:1 | 3.18 | 32.2 |
| S:Ti = 0.6:1 | 3.10 | 29.6 |
| S:Ti = 0.7:1 | 2.90 | 27.2 |
| S:Ti = 0.8:1 | 3.14 | 28.6 |
| Nitrogen Precursor | Carbon Precursor | Tio2 Precursor or Type | Synthesis Method | Pollutant (Initial Concentration) | Photodecomposition or Removal Efficiency and Conditions | Ref. |
|---|---|---|---|---|---|---|
| Citric acid | TiCl4 | Sol-gel | 4-NP (7.0 × 10−2 mmol L−1) | 87% in 420 min, visible light | [280] | |
| Ethanol | Amorphous TiO2 | Calcination under gas flow | Reactive Red 198 (RR198) (500 mg L−1) | 96% in 20 h, visible light | [281] | |
| Escherichia coli (1.5 × 108 CFU mL−1) | 100% of bacteria inactivated in 60 min, visible light | [282] | ||||
| Isopropanol | Escherichia coli (1.5 × 108 CFU mL−1) | 100% of bacteria inactivated in 60 min, visible light | [282] | |||
| Methanol | RR198 (500 mg L−1) | 54.6% in 100 h, visible light | [283] | |||
| Escherichia coli (1.5 × 108 CFU mL−1) | 100% of bacteria inactivated in 60 min, visible light | [282] | ||||
| Styrene | TBOT | Pyrolysis | Tetracycline hydrochloride (TCH) (10 mg L−1) | 99.6% in 30 min, visible light | [284] | |
| Titanium ethoxide | Titanium ethoxide | Sol-gel | DCF (10, 30 and 50 mg L−1) | 80% in 100 min, UV light | [285] | |
| Ammonium nitrate | Acetylacetone | TTIP | Sol-gel | MB (10 mg L−1) | 91.3% in 3 h, visible light | [286] |
| Extrapallial fluid (EPF) of blue mussels | EPF of blue mussels | TBOT | Green bioinspired synthesis | Microplastic (0.4 w/v%) | 71.77 ± 1.88% in 50 h, visible light | [287] |
| Glycine | Glycine | Titanium(III) sulfate | Hydrothermal | Ibuprofen (IBP) (20 mg L−1) | 100% in 360 min, visible light | [288] |
| Guanidine hydrochloride | Guanidine hydrochloride | Anatase TiO2 | One-step microwave | MO (20 mg L−1) | Up to 94% in 2 h, visible light | [289,290,291] |
| Hexamethylenetetramine (HMT) | HMT | TiCl4 | Solvothermal | Resorcinol (20 mg L−1) | Up to 96% within 90 min, visible light | [292] |
| TTIP | Bisphenol A (BPA) (5 and 0.02 mg L−1) | Over 99% in 5 h and 95% in 2 h, white LED | [293,294] | |||
| Methanol | NOx (1 ppm) | ~10% at 530 nm, ~23% at 445 nm, ~40% at 390 nm | [295] | |||
| Nitric acid | Tween 80 | TTIP | Sol-gel | CIP and levofloxacin | 78.2% and 96.7% in 2 h, visible light | [296] |
| N-lauroyl-L-glutamic acid | N-lauroyl-L-glutamic acid | Titanium (diisopropoxide) bis (2,4-pentanedionate) | Selfassembly soft-template | Ph (10 mg L−1) | 92% in 150 min, visible light | [297] |
| N-methyl-formamide | N-methyl-formamide | TTIP | Sol-gel | Orange G (80 mg L−1) | 99% in 1 h, visible light | [298] |
| PAN | PAN | TBOT | Hydrolysis and calcination | Rh B (20 mg L−1) | About 99% in 4 h, visible light | [299] |
| Polyaniline | Carbon tetrachloride | TiO2 NPs powder | Stirring and carbonization | Ph (20 mg L−1) | 87% in 150 min, UV radiation | [300] |
| Polyaniline | TBOT | Polymerization and sol-gel coating | Rh B (2.0 × 10−5 mol L−1) and Ph (10 mg L−1) | 90% in 100 min and 84% in 150 min, visible light | [301] | |
| PVP | PVP | TTIP | Electrospinning and calcination | MB (2.0 × 10−5 mol L−1) | 95% in 120 min, visible light | [302] |
| Tetrakis(dimethylamino) titanium (TDMAT) | Mesoporous carbon molecular sieves CMK-3 | TDMAT | Stirring in autoclave | Rh B (10 mg L−1) | 99.87% in 60 min, visible light | [303] |
| Triethanolamine (TELA) | TELA | TTIP | Sol-gel | 2-chlorophenol (100 mg L−1) | 94.39% in 90 min, visible light | [89] |
| Urea | Tetrabutylammonium hydroxide (TBAH) | TBOT | Sol-gel | MB (1.8 × 10−5 mol L−1) | Over 90% in 7 h, visible light | [304] |
| Urea | Microcystis aeruginosa (~3.0 × 106 cells mL−1) | Up to 92.7% of bacteria inactivated in 12 h, visible light | [305] | |||
| Ti foil | CBD | MO (5 mg L−1) | 100% in 30 min, visible light | [306] |
| XPS Region | Chemical State | BE [eV] | References |
|---|---|---|---|
| N 1s | N-Ti-N | 395.7 | [297] |
| Ti-N-Ti | 397.98 | [299] | |
| Ti-O-N | 400–400.1 | [289,306] | |
| 401.7 | [303] | ||
| O-Ti-N | 396.3–396.5 | [306] | |
| 397.1–397.7 | [297,307] | ||
| 399.1–400.8 | [89,280,289,296,300,302,303,305,308] | ||
| N-C | 395.7 | [297] | |
| 399.3 | [297] | ||
| ~400 | [298] | ||
| N-N | 399.3 | [297] | |
| C 1s | C-C (usually adventitious carbon) | 284.6–285 | [289,298,301,303,305,307] |
| C-OH | 285.6 | [303] | |
| 284.6 | [280] | ||
| C-N | 284.6 | [280] | |
| 285.9 | [301] | ||
| C=N | 287 | [301] | |
| C-O | 285.9–286.7 | [89,289,296,298,302,303,305] | |
| 288 | [306] | ||
| Ti-C | 281.8 | [307,308] | |
| 282.5 | [292] | ||
| 284.6 | [297] | ||
| C=O | 286 | [306] | |
| 288.5–288.7 | [89,280,289,296,298,302,305] | ||
| Ti-O-C | 288.0–288.9 | [89,296,297,301,303,306] | |
| 289.6 | [289] | ||
| O-Ti-C | 288.0–288.5 | [297,306] |
| Doping Source | TiO2 Precursor or Type | Synthesis Method | Pollutant (Initial Concentration) | Photodecomposition or Removal Efficiency and Conditions | Ref. |
|---|---|---|---|---|---|
| Eichhornia crassipes extract | TTIP | Green modification of sol-gel | Reactive Blue 19 (RB19) (6.10 mg L−1) and Reactive Red 76 (RR76) (4.49 mg L−1) | 72.2% and 73.3% after 30 min, visible light | [102] |
| Carrageenans (kappa (κ-), iota (ι-) and lambda (λ-)) from red seaweeds | TTIP | Hydrothermal followed by calcination | RB5, MB and MO (25 mg L−1) | 100% after <5, <5 and <20 min for κ-, ι- and λ- carrageenans, solar concentrator | [309] |
| Thiourea | Meta-titanic acid | Solid-phase | Rh B and MB | Up to about 60% (Rh B) and 50% (MB) after 30 min of contact with photocatalytic coatings, visible light | [156] |
| TBOT | Solvothermal sol-gel | Doxycycline (DC) | 43.1% after 1 h, visible light | [310] | |
| 1-octadecene and sulfur | TiCl4 | Solvothermal | TCH (10 mg L−1) | 86% after 1 h, visible light | [311] |
| Thiourea, carbon, citric acid and glycerol | Titanium ethoxide | Stirring followed by stepwise heating | Safranin T (ST) (30 mg L−1) | Rate constants: up to 4.04 × 104 s−1 for UV and 0.63 × 104 s−1 for vis | [157] |
| Thiourea, urea | TBOT | Hydrolysis | 4-chlorophenol (1.5 × 10−4 mol L−1 | ~35% in 3 h, visible light | [312] |
| No. | Photocatalyst | Doping Source | Synthesis Method | SBET (m2 g−1) | Crystallite Size of Anatase (nm) | Ebg (eV) | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | N-doped TiO2 | Ammonia | Non-hydrolytic sol-gel | 100.1 | 8.2 | 2.67 | [87,231,315] |
| S-doped TiO2 | DMSO | 69.7 | 10.5 | 3.07 | |||
| N,S-co-doped TiO2 | Ammonia and DMSO | 144.8 | 5.1 | 2.32 | |||
| 2 | N-doped TiO2 | Urea | Wet impregnation | 55.35 | 18.8 | 2.79 | [276] |
| S-doped TiO2 | Thiourea | 50.16 | 18.5 | 2.65 | |||
| N,S-co-doped TiO2 | Urea and thiourea | 45.74 | 16.9 | 2.68 | |||
| 3 | N-doped TiO2 | Ammonium nitrate | Sol-gel | 39.5 | - | - | [231] |
| S-doped TiO2 | Thiourea | 47.6 | - | - | |||
| N,S-co-doped TiO2 | Ammonium nitrate and thiourea | 68.6 | - | - | |||
| 4 | N-doped TiO2 | Dodecylamine | - | 10.32 | 2.95 | [320] | |
| S-doped TiO2 | Thiourea | - | 10.83 | 3.00 | |||
| N,S-co-doped TiO2 | Dodecylamine and thiourea | - | 9.59 | 2.89 | |||
| 5 | N-doped TiO2 | Urea | Wet impregnation | 143.61 | 13.7 | 2.99 | [247] |
| S-doped TiO2 | Thiourea | 158.30 | 14.1 | 3.08 | |||
| N,S-co-doped TiO2 | Urea and thiourea | 97.11 | 13.8 | 2.96 |
| Nitrogen Precursor | Sulfur Precursor | Tio2 Precursor or Type | Synthesis Method | Pollutant (Initial Concentration) | Photo-Decomposition or Removal Efficiency and Conditions | Ref. |
|---|---|---|---|---|---|---|
| Ammonia | DMSO | TiCl4 and TTIP | Non-hydrolytic sol-gel | RO16 (20 mg L−1) | 100% after 30 min, visible light | [247] |
| Ammonium nitrate | Thiourea | TTIP | Sol-gel | MB (10 mg L−1) and Ph (10 mg L−1) | 78.64% after 6 h and 77.87% after 4 h, visible light | [276] |
| Ammonium sulfate | Ammonium sulfate | TBOT | Sol-gel impregnation | 5-FU (10 mg L−1) | Ca. 90% after 90 min, simulated solar light | [327] |
| Thiourea | Thiourea | TBOT | Sol-gel | MO (7 mg L−1) | 96% after 3 h, sunlight | [326] |
| TiO2 P25 | Mixing—calcination | TC (10 mg L−1) | 91% after 20 min, visible light | [325] | ||
| TiO2 powder (Tayca Corp., Japan) | E. coli (104 CFU mL−1) | Total bacteria inactivation after 75 min, visible light | [319] | |||
| Ti(SO4)2 | Hydrothermal | MO (10 mg L−1) | 92% after 6 h, sunlight | [317] | ||
| Urea | Thiourea | Ti(SO4)2 | Hydrothermal | Atrazine (5 mg L−1) | Over 70% after 6 h, visible light | [318] |
| Year | Preparation Method | Precursors | Precursor Ratio (C, N, S Source to Ti Source) | Annealing Temperature, Time | Ebg (eV) | Doping Mode of C, N and S and Respective BE from XPS | Ref. |
|---|---|---|---|---|---|---|---|
| 2015 | Sol-gel | Thiourea, TTIP | 20 mL TTIP, 15 wt.% thiourea | 300, 400, 500 and 600 °C | NR | Carbon: adventitious carbon species (284.6 eV), graphite intercalation compounds (285.3 eV) and C-O (298.6 eV); Nitrogen: substituted oxygen lattice sites as O-Ti-N (396.5 eV) and interstitially doped into TiO2 lattice (400.5 eV); Sulfur: Ti4+ substituted by S6+ (169.4 eV) | [333] |
| 2016 | Sol-gel | Thiourea, TBOT | 1:100 | 450 °C, 1 h | NR | Carbon: XPS peak at 285 eV; Nitrogen: XPS peak 400 eV; Sulfur: XPS peaks at 165 and 233 eV | [332] |
| 2017 | Hydrothermal (120 °C, 2 h) | Thiourea, TiC | 0.63 | - | NR | Carbon: C-O (286.5 eV) and C=O (288.6 eV) bonds; Nitrogen: interstitially doped as Ti-O-N (400.1 eV); Sulfur: formed Ti-O-S bonds due to cationic doping (168.5 eV) | [330] |
| 2017 | Hydrothermal (200 °C, 20 h) | Thiourea, anatase/brookite heterojunction TiO2 | 1:1 | 450 °C, 1 h | 2.9 | Carbon: carbonate species C-O (286.46 eV) and C=O (288.9 eV); Nitrogen: interstitially Ti-O-N and substitutionally O-Ti-N doped (400.9 eV); Sulfur: Ti4+ substituted by S6+ (168.6 eV) | [8] |
| 2017 | Hydrothermal (180 °C, 24 h) | Cystine, TBOT | 1.7 mL TBOT, 0.5 g cystine | 450 °C, 2 h | 2.51–2.95 | Carbon: hydrocarbons from precursor and adventitious carbon (284.6 eV), C-O (286.1 eV) and C=O (288.6 eV) species; Nitrogen: partially substituted oxygen lattice sites as O-Ti-N (399.7 eV) and interstitially doped as Ti-O-N (400.9 eV) Sulfur: Ti4+ substituted by S6+ (168.3 eV) | [335] |
| 2017 | Sonochemical (20 kHz, room temperature (RT)) | Thiourea, TBOT | 0.05:1, 0.2:1 and 0.5:1 | 300, 400 and 500 °C, 3 h | 2.66 | Carbon: elemental C on the surface (287.60 eV), C-O (289.40 eV) and C=O (291.50 eV) bonds; Nitrogen: N doped into the TiO2 lattice (396.00 eV); Sulfur: Ti4+ substituted by S6+ (171.50 eV) | [7] |
| 2018 | Hydrolysis (RT, 6 h) | Thiourea, TTIP | 10 mL TTIP, 5 wt.% thiourea | 400 °C | 2.91 | NR | [329] |
| 2018 | Thermal decomposition | Urea, Ti(SO4)2 | N:Ti = 4:1 | 600 °C, 2 and 4 h | 1.39–3.05 | Carbon: C substituted oxygen atom in the TiO2 lattice forming Ti-C (281.7 eV), C formed complex unsaturated groups and carbonate species C=C or C-C (284.4 eV), C-O (287.5 eV) and C=O (289.1 eV); Nitrogen: N substituted oxygen atom O-Ti-N (∼399 eV) and interstitially doped in the TiO2 lattice (∼400 eV), adsorbed as N2 and NH3 (∼401, ∼402, ∼404 and ∼408 eV); Sulfur: S6+ in TiO2 (168.38 and 169.48 eV) | [337] |
| 2020 | Sol-gel followed by dip coating | Thiourea, TBOT | 10 mL TBOT, 2.5 mol% thiourea | 450 °C, 2 h | NR | NR | [340] |
| 2020 | Hydrothermal (180 °C, 12 h) | Thiourea, TiOSO4 | 2:1 | 400, 500, 600 and 700 °C, 1 h | 2.88 | Carbon: elemental carbon formed by incomplete combustion (284.7 eV), C replaced Ti atoms in lattice forming Ti-O-C structure (286.4 eV), presence of C=O bond (289.0 eV) Nitrogen: N replaced O atoms (400.3 eV) Sulfur: S6+ replaced Ti4+ (168.7 eV and 169.5 eV) | [343] |
| Model Compound | Type of Radiation | Experiment Conditions | Degradation Rate | Ref. |
|---|---|---|---|---|
| Rh B | Visible | 300 W Xenon lamp with a 420 nm cut-off filter Rh B: 10 mg·L−1 Photocatalyst: 1 g L−1 | 68% Rh B decomposed after 10 min, complete decolorization after 60 min | [330] |
| MB | Visible | 500 W Xenon lamp equipped with a UV cutoff filter. MB: 20 mg L−1 Photocatalyst: 1 g L−1 | 91.9% MB decomposed after 60 min | [337] |
| MO | Visible | 300 W Xenon lamp with a 420 nm cut-off filter MO: 10 mg L−1 Photocatalyst: 1 g L−1 | Up to 92.13% MO decomposed within 180 min | [335] |
| Rose Bengal | Sunlight | Sunny day under clear sky conditions at Tirunelveli between 11 a.m. to 2 p.m. Rose Bengal: 20 mg L−1 Photocatalyst: 1 g L−1 | Complete decolorization after 60 min | [329] |
| DCF | Sunlight | Sunny days in April 2015 with the intensity of 750 lx DCF: 25 mg L−1 Photocatalyst: 1 g L−1 | Up to 69.81% DCF decomposed within 90 min | [7] |
| IBP | Visible | LED lamp (λmax = 420 nm, intensity = 1 mW cm−2, height 31.8 cm) IBP: 20 mg L−1 Photocatalyst: 0.5 g L−1 | Mineralization of TOC: 95.2%, complete decomposition after 5 h | [8] |
| TC | Visible | 6 W cold white visible lamp with a 420 nm cut-off filter TC: 5 mg L−1 Photocatalyst: 0.5 g L−1 | 97% TC removed after 3 h | [338] |
| NOx | Visible | Two 15 W fluorescent light tubes with emission between 400–700 nm NO: 400 ± 50 ppb Relative humidity: 60 ± 10% Flow rate: 1.140 ± 0.027 L min−1 | Up to 18% NOx degraded | [339] |
| NO | Simulated solar light | 300 W tungsten halogen lamp NO: 400 ppb Relative humidity: 70% Flow rate: 4 L min−1 | Up to 25% NO degraded after 30 min | [342] |
| VOCs | Visible | 8 W fluorescent daylight lamp VOCs: 0.1 ppm Relative humidity: 45% Flow rate: 1 L min−1 | 70, 87, ∼100, and ∼100% of decomposition for benzene, toluene, ethyl benzene and o-xylene, respectively | [332] |
| Toluene | UV | 8 W fluorescent lamp Toluene: 1 mL toluene vapor Photocatalyst: 6 g dispersed on a borosilicate Petri dish in a Teflon sampling bag | Above 95% of toluene degraded after 4 h | [340] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piątkowska, A.; Janus, M.; Szymański, K.; Mozia, S. C-,N- and S-Doped TiO2 Photocatalysts: A Review. Catalysts 2021, 11, 144. https://doi.org/10.3390/catal11010144
Piątkowska A, Janus M, Szymański K, Mozia S. C-,N- and S-Doped TiO2 Photocatalysts: A Review. Catalysts. 2021; 11(1):144. https://doi.org/10.3390/catal11010144
Chicago/Turabian StylePiątkowska, Aleksandra, Magdalena Janus, Kacper Szymański, and Sylwia Mozia. 2021. "C-,N- and S-Doped TiO2 Photocatalysts: A Review" Catalysts 11, no. 1: 144. https://doi.org/10.3390/catal11010144
APA StylePiątkowska, A., Janus, M., Szymański, K., & Mozia, S. (2021). C-,N- and S-Doped TiO2 Photocatalysts: A Review. Catalysts, 11(1), 144. https://doi.org/10.3390/catal11010144








