2.1. Characterization of Catalyst Precursors and Catalysts
The zeolites were obtained from two layered precursors (PREFER) with the intended molar Si/Al ratios of 30 (PREFER_30) and 50 (PREFER_50). The calcination of such precursors resulted in the condensation of layers with the formation three dimensional (3D) microporous ferrierite zeolites (FER_30 and FER_50). The second part of the zeolite precursors was swollen with alkylammonium surfactants to open interlayer space for the intercalation of the silica pillars precursor (tetraethyl orthosilicate, TEOS), which after calcination resulted in ITQ-36_30 and ITQ-36_50 (depending on the intended Si/Al ratio in the zeolite framework). On the other hand, swollen PREFER_30 and PREFER_50, after sonication and calcination, resulted in delaminated ITQ-6_30 and ITQ-6_50 zeolites, respectively.
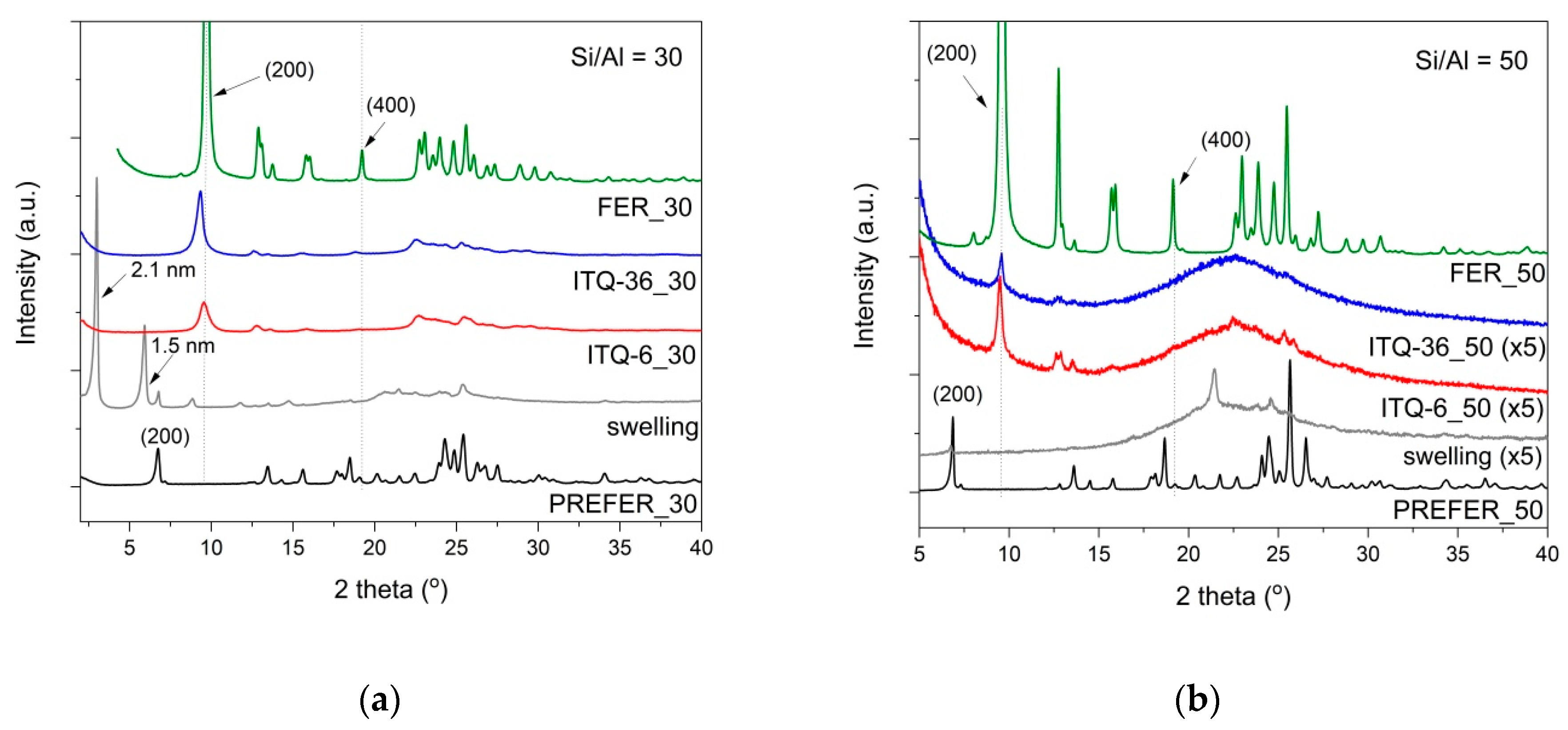
The process of PREFER swelling, condensation, delamination and pillaring was monitored by the X-ray diffraction method, as shown in
Figure 1. In the diffractograms of the layered precursors, the (200) reflection at 2Θ = 6.8°, corresponding to the interlayer distance of about 1.3 nm, was identified [
20]. After the swelling of PREFER_30 with surfactant, the main (200) reflection was shifted to 2Θ at about 3.0°, indicating the successful intercalation of the surfactants into the interlayer space of zeolites and resulting in the extending of the interlayer distance to about 2.1 nm. This interlayer distance is very close to the length of the hexadecyltrimethylammonium cations (CTMA
+), indicating that the surfactant molecules are oriented perpendicularly to the zeolite layers. Apart from the main (200) reflections, the diffraction peaks at 2Θ at about 6.0, 6.8 and 8.9°, characteristic of the parallel ordering of the zeolite layers, are also present in the diffractogram of the swollen PREFER_30 sample. The first diffraction peak indicates the extension of the interlayer distance to about 1.5 nm and shows that part of the zeolite was intercalated with the surfactant molecules oriented non-perpendicularly to the zeolite layers [
15]. The diffraction peak at 6.8° corresponds to a small fraction of the PREFER_30 sample, which was not swollen, while the diffraction peak at 8.9° is possibly related to the partially condensed zeolite layers. The swelling process causes a structural disorder in PREFER_30 indicated by broadening peaks and the loss of their intensities. The calcination of PREFER_30 resulted in a shift in the (200) reflection to 9.7°. The high intensity of this basal reflection, as well as the diffraction peaks characteristic of the 3-dimensional (3D) structure of ferrierite, indicates the successful synthesis of this zeolite (FER_30). The sonication of swelled PREFER_30 in acidic conditions, followed by calcination, resulted in a nearly completely delaminated structure (non-parallel ordering of the zeolite layers). The low-intensive reflection at 9.7° indicates that part of the zeolite layers was condensed with the formation of ferrierite. However, the comparison of this reflection intensity in diffractograms of FER_30 and ITQ-6_30 shows that the contribution of the ferrierite in the delaminated ITQ-6_30 sample is relatively small. The intercalation of swollen PREFER_30 with silica pillars resulted in ITQ-36_30. The diffractogram of this sample is characteristic of the delaminated structure, due to the lack of the basal reflections typical of the pillared samples with the parallel ordering of the zeolite layers. The (200) diffraction peak at about 9.3° is only slightly shifted in relation to the (200) reflection in the diffractogram of FER_30 (9.7°), indicating the presence of small micropores, with a diameter of about 0.5 nm located in the interlayer space of the zeolite. However, the very low intensity of this reflection, in comparison to the (200) reflection recorded for FER_30, shows only a small contribution of this fraction of material in the ITQ-36_30 sample. The comparison of diffractograms recorded for FER_30 and FER_50, with respect to the intensity of the reflections, shows that the calcination of PREFER_50 resulted in the ferrierite zeolite with better crystallinity, as shown in
Figure 1. However, the disappearance of the (200) reflection in the diffractogram of the swollen PREFER_50 sample indicates the delamination of its layered structure. The very small diffraction peak, in the position characteristic of the (200) reflection, in the diffractogram of PREFER_50 indicates that only a small fraction of this sample was not swollen. Moreover, the swelling process, conducted in strong basic media, resulted in the significant destruction of the zeolite structure, which corresponded to decreased intensity or the disappearance of the reflections characteristic of the intralayer ordering of the zeolite structure. The broad reflection in the range of 15–30° indicates the presence of amorphous silica [
21], which was possibly formed by the extraction of silica from the zeolite precursor in strong basic media used for the swelling of PREFER_50. Possibly part of the dissolved silica was precipitated in a solution and deposited on the external zeolite surface in the form of amorphous silica aggregates. Thus, it seems that the stability of the ferrierite precursors (PREFER) in a basic media strongly depends on the Si/Al ratio. The high-silica ferrierite precursors are significantly less stable in basic media than the low-silica PREFER samples. The delamination and intercalation of the swollen PREFER_50 sample resulted in delaminated materials, consisting of chaotically organized defected zeolite layers with a significant contribution of amorphous silica. The low intensive (200) reflection at 9.7° indicates the presence of a ferrierite phase in these samples.
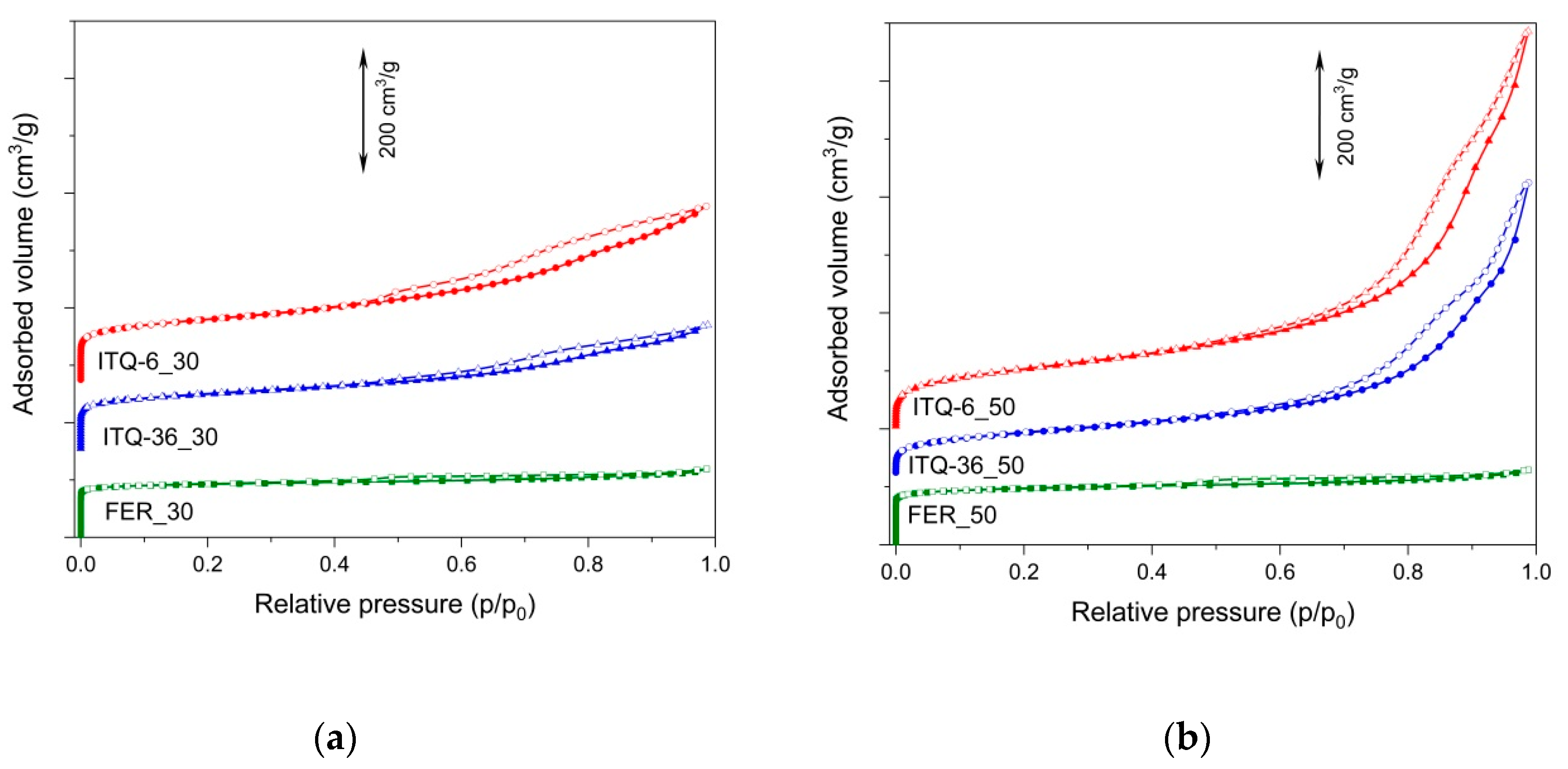
The dinitrogen adsorption–desorption isotherms of the studied samples are presented in
Figure 2, while their textural parameters are compared in
Table 1. The isotherms of FER_30 and FER_50 are type I, according to the IUPAC classification [
22], characteristic of microporous materials. The microporous character of the samples is indicated by a steep adsorption at low relative pressure, assigned to dinitrogen capillary condensation in micropores [
23]. The isotherms of the ITQ-6_30 and ITQ-6_50, as well as the ITQ-36_30 and ITQ-36_50 samples, presented in
Figure 2, are type IV, according to the IUPAC classification [
22], characteristic for mesoporous materials. However, an increase in adsorbed dinitrogen volume, observed at very low relative dinitrogen pressure, also indicates a significant contribution of micropores in these series of the zeolitic samples. Micropores are located in the zeolitic layers, while mesopores are the spaces between intercalated and delaminated zeolite layers in ITQ-36 and ITQ-6, respectively. As it was shown by XRD analysis, the swelling of high-silica PREFER_50 resulted in the partial extraction of silicon from the ferrierite layers with the formation of amorphous silica, identified in ITQ-6_50 and ITQ-36_50, as shown in
Figure 1. Thus, an increase in adsorbed dinitrogen volume, observed at higher relative pressure for these samples, indicates not only the presence of interlayer mesopores but also mesopores of different size in amorphous silica [
24]. The hysteresis loops present in the adsorption–desorption isotherms of ITQ-6_30 and ITQ-36_30 are type H4, typical for mesoporous zeolites and micro-mesoporous materials, while the hysteresis loops recorded for ITQ-6_50 and ITQ-36_50 are type H3, characteristic of non-rigid aggregates of plate-like particles [
22]. The characteristic step down in the desorption isotherms, associated with hysteresis loop observed for ITQ-6_50 and ITQ-36_50, indicates the presence of partially plugged mesopores [
25].
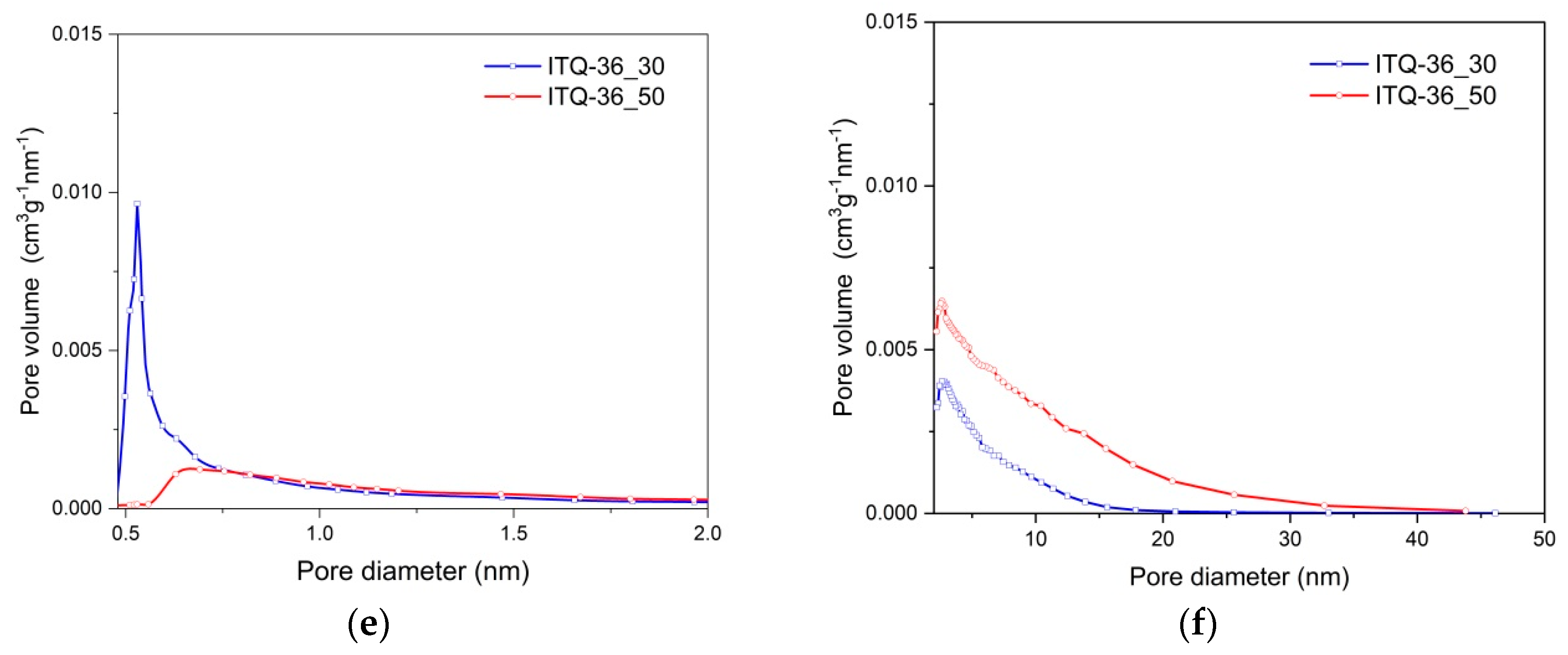
Profiles of pore size distribution in the micropore and mesopore ranges for ferrierites and their delaminated and intercalated forms are shown in
Figure 3. In the case of FER_30 and FER_50, the maximum micropore size distribution, located at about 0.53–0.55 nm, fits very well to the diameter of 10 MR (member ring) channels in ferrierite [
16,
26]. In the mesopore range, the only small contribution of pores with the diameter below 15 nm was found for both ferrierite samples. The delamination and intercalation of PREFER_30, resulting in ITQ-6_30 and ITQ-36_30, respectively, reduced the intensity of micropore size distribution profile, as shown in
Figure 3. This effect is related to the ordered microporous structure limited only to the zeolite layers in the case of ITQ-6_30 and ITQ-36_30, and the three dimensional microporous structure of FER_30 and FER_50. In the mesopore range, the maxima of pore size distribution are at about 3.6 and 2.6 nm for ITQ-6_30 and ITQ-36_30, respectively. For both these samples, the tails up to about 18 nm indicate the high heterogeneity of pore sizes, which is characteristic of delaminated materials. The microporous character of ITQ-6_50 and ITQ-36_50 is significantly different than the ITQ-6_30 and ITQ-36_30 samples, as shown in
Figure 3. The maximum characteristic of the ferrierite 10 MR channels disappeared, while broad and low-intensive peaks at about 0.6–0.7 nm with long tails from the side of larger pores indicate the high heterogeneity of micropore sizes. These results are consistent with the XRD studies, which showed the partial destruction of the ferrierite layers during PREFER_50 swelling. The mesopore size distribution in ITQ-6_50 and ITQ-36_50 is much broader than for the analogous zeolitic samples with the lower Si/Al ratio. The maxima are located at about 4.5 and 2.6 nm for ITQ-6_50 and ITQ-36_50, respectively. It was shown by the XRD analysis of these samples that part of the silicon was extracted from the zeolite layers during PREFER_50 swelling and was then deposited in the form of amorphous silica aggregates on the zeolite external surface, as shown in
Figure 1. Thus, possibly the mesopores of various size, present in amorphous silica, contribute in the mesopore size distribution profiles of ITQ-6_50 and ITQ-36_50. The textural parameters of all the studied samples are presented in
Table 1. Micropores dominate in FER_30 and FER_50, while in their delaminated and intercalated forms the contribution of micropores is reduced, while the mesopore volume significantly increased. The introduction of copper into these zeolitic samples decreased their textural parameters.
The elemental compositions of the zeolitic samples, presented in
Table 2, show that the real Si/Al ratios, determined by the ICP-OES method, are 22 for FER_30 and 64 for FER_50, so slightly different from the intended values of 30 and 50, respectively. The delamination of PRERER_30 did not change the Si/Al ratio in ITQ-6_30. However, in the case of ITQ-6_50, the Si/Al ratio increased to 214, indicating a decrease in aluminum content in this sample. As previously discussed, the swelling conducted in strong basic conditions resulted in the partial leaching of silicon from the zeolite layers of PREFER_50, which in solution possibly formed amorphous silica aggregates deposited on the zeolite surface. This effect was observed only for high-silica zeolite, which is possibly less stable in basic conditions. In the next step of ITQ-6_50 synthesis, the swollen PREFER_50 was treated in acidic conditions, which possibly resulted in the effective leaching of aluminum from the partially degraded zeolite layers. Additionally, the partial dealumination of PREFER_50 in swelling conditions, as the process assisting silicon leaching, cannot be excluded. The intercalation of the zeolite precursors with silica to obtain ITQ-36_30 and ITQ-36_50 resulted in an increase in the Si/Al ratio. The content of copper introduced to the zeolitic samples is in the range of 0.9–2.7 wt %, as shown in
Table 2. Taking into account that one Al
+3 cation in the zeolite framework generates one negative charge in such a framework, as well as taking into account that cooper is deposited in the form of Cu
2+ cations, the number of copper cations introduced by the ion-exchange mechanism should be two-times lower than the Al
3+ cations in the zeolite framework. The Cu/Al molar ratio for FER_30 and its delaminated and pillared forms, as well as for FER_50, are in the range of 0.55–0.64, thus it seems that, in these cases, the ion-exchange mechanism dominated in copper deposition. Much larger Cu/Al ratios, determined for ITQ-6_50 and ITQ-36_50 indicate a contribution, aside from ion-exchange, also other mechanisms of copper species deposition.
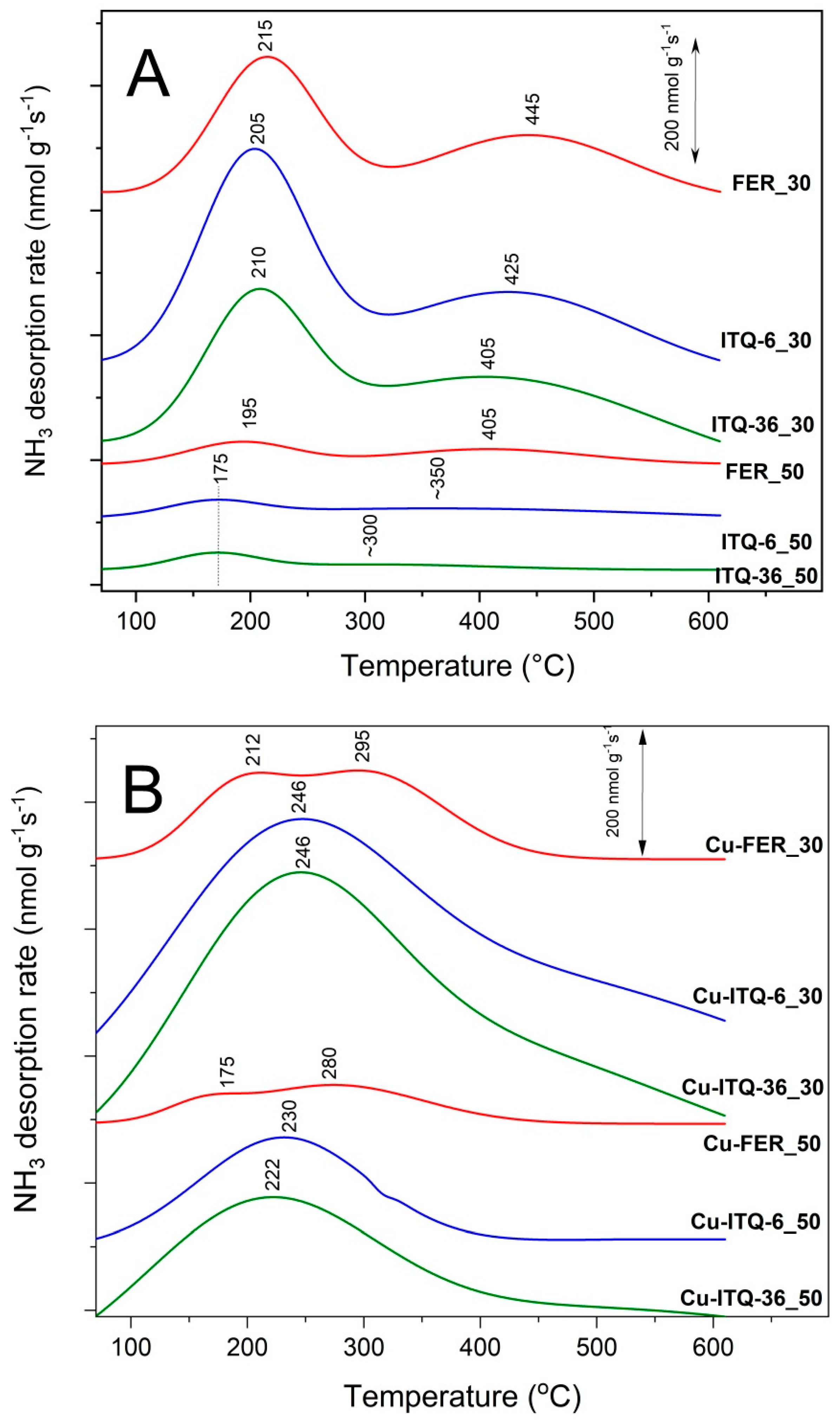
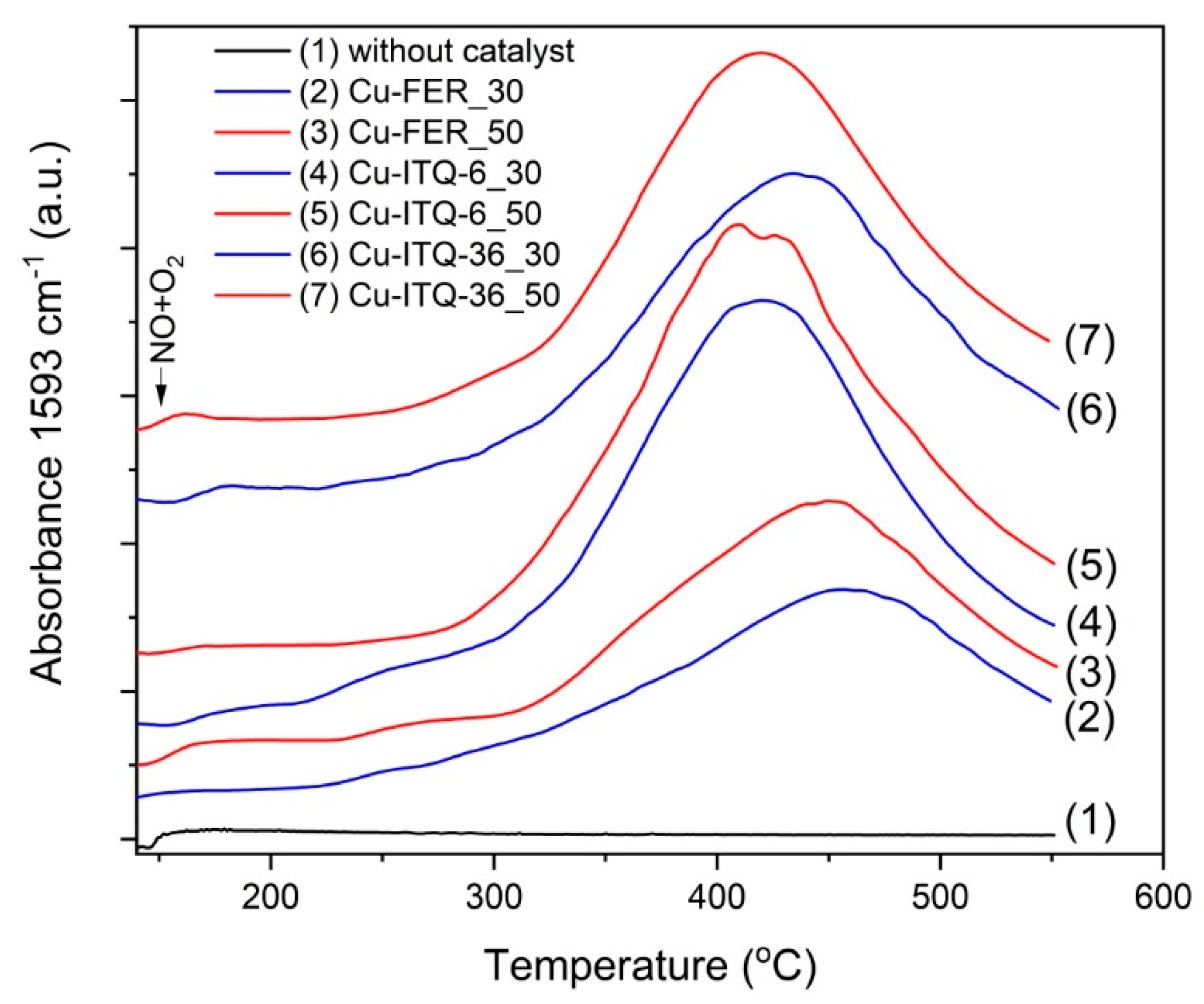
The surface acidity of the zeolitic samples was studied by the temperature-programmed desorption of ammonia (NH
3-TPD). Ammonia is a basic molecule with respect to both Brønsted and Lewis theory and therefore may interact with both these types of acid sites. Moreover, the diameter of the ammonia molecule is about 0.3 nm and therefore may easily penetrate all microporous channels in ferrierite [
27,
28]. The ammonia desorption profiles, presented in
Figure 4A, consist of two desorption peaks and are characteristic of ferrierite zeolites [
26]. The low-temperature maximum was centered at about 205–215 °C for the samples with the intended Si/Al ratio in the zeolite framework of 30, and at about 175–195 °C for the zeolites with the intended framework Si/Al ratio of 50, which indicates the presence of the relatively weak acid sites. The second, much broader and less intensive maxima, centered at about 405–445 °C for the zeolites with the intended framework Si/Al ratio of 30 and at about 300–405 °C for the samples with the Si/Al ratio in the zeolite framework of 50, are related to the stronger acid sites. The most significant difference in desorption profiles is related to their intensity depending on the Si/Al ratio. It is not surprising, assuming that the surface acidity depends on the aluminum content in the zeolitic samples. The deposition of copper into zeolites resulted in a very significant modification of the ammonia desorption profiles, as shown in
Figure 4B. In the case of FER_30, the intensity of the low-temperature maximum was significantly reduced, while the high-temperate maximum was shifted to lower temperature. The similar effect of the high-temperature maximum shift to lower temperature was observed for FER_50; however, in this case the deposition of cooper resulted in an increase in ammonia adsorption. The ammonia desorption profiles of delaminated and intercalated zeolites consist of one broad and asymmetric maximum, centered at about 245 °C in the case of the samples with the lower Si/Al ratio, while for zeolites with the higher Si/Al ratio at a temperature about 15–25 °C lower. Moreover, the surface concentration of adsorbed ammonia increased after copper deposition to these samples. Thus, the introduction of copper into the zeolitic samples very significantly modified their surface acidic properties. It should not be surprising, taking into account that possibly all ion-exchange positions were substituted by copper species, as shown in
Table 2, and in the Cu-ITQ-6_50 and Cu-ITQ-36_50 samples the content of deposited copper is a few times higher than their ion-exchange capacity, as shown in
Table 2. Copper species introduced to zeolites play a role in the Lewis acid sites, which can accommodate a free electron pair of ammonia molecules into unoccupied d-orbital. Of course the content of ammonia chemisorbed in this way depends on the surface availability of the copper species and, therefore, their dispersion. The results of NH
3-TPD should be discussed not only as an evaluation of the surface acidity of the samples, but also as important steps in the NH
3-SCR and AMOx processes, in which chemisorption and the activation of ammonia is one of the main stages [
2,
14]. This topic will be discussed together with the results of the catalytic tests.
The analysis of the pyridine pre-adsorbed samples by FTIR spectroscopy, which is a standard method used for determination of Brønsted and Lewis acid site contribution, is very problematic in the studies of the ferrierite based samples. According to Wichterlova et al. [
29] and Starzyk et al. [
30], the kinetic diameter of pyridine is about 0.57 nm, which is bigger than the diameter of 10 MR channels in ferrierite [
16,
26], and therefore cannot be used for the analysis of acid sites located inside such channels.
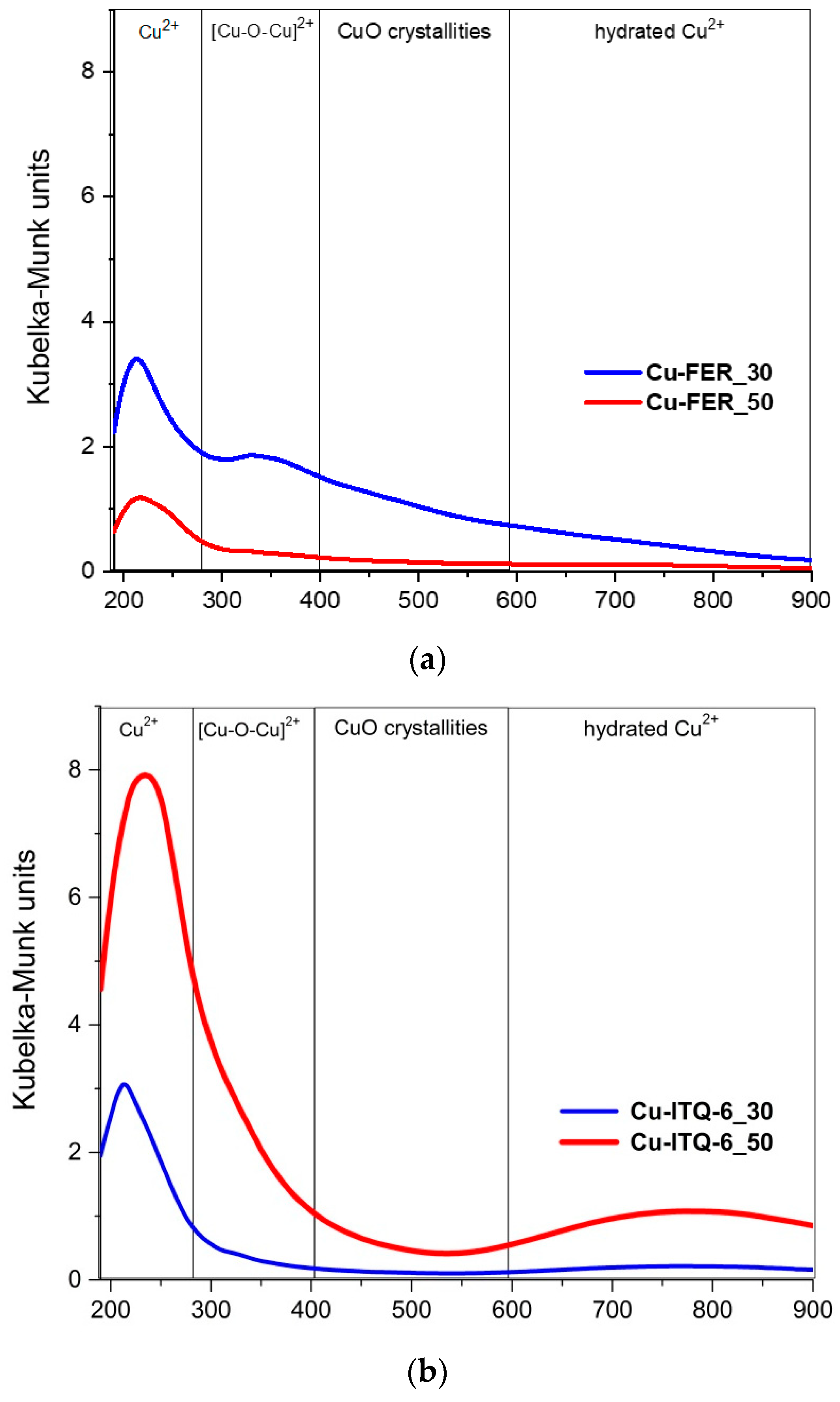
The coordination and aggregation of copper species introduced into the zeolitic samples was studied by UV-vis-DR spectroscopy, as shown in
Figure 5. The intensive band centered at about 220–245 nm, present in the spectra of all copper-containing samples, is related to monomeric Cu
2+ ions interacting with the oxygen of the zeolite framework (O
2−→Cu
2+) [
31,
32,
33]. In the case of Cu-FER_30, the band centered at 340 nm indicates the presence of small oligomeric copper oxide species [
31,
32,
33]. Additionally, the shoulders in the range of 280–400 nm, observed in the spectra of other samples, are attributed to such small copper oxide aggregates. Moreover, the shoulder above 400 nm could be assigned to the presence of small CuO crystallites [
31]. Copper oxide crystallites were not identified by XRD analysis of the samples, which indicates that their size is below the detection level of this experimental method. For the samples of the Cu-ITQ-6 and Cu-ITQ-36 series, the bands above 600 nm are assigned to the d–d transition of Cu
2+ ions in pseudo-octahedral coordination (e.g., Cu(H
2O)
62+) [
31,
32,
33], and therefore indicate, similarly to the bands at 220–245 nm, the significant contribution of copper deposited in the form of monomeric cations.
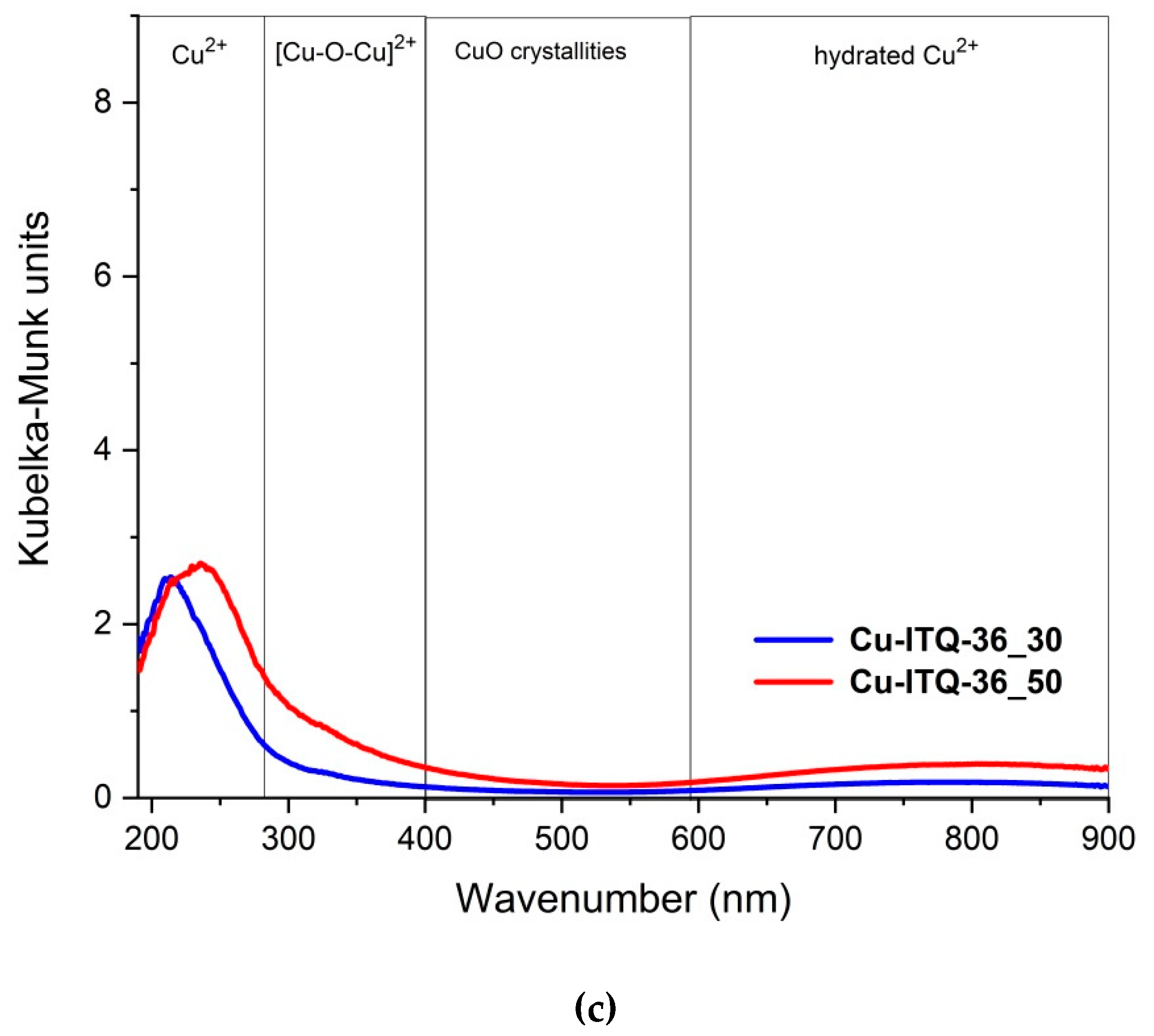
The H
2-TPR profiles of the zeolitic samples modified with copper are shown in
Figure 6. The reduction profiles of copper species depend on their form and aggregation. In the case of Cu
2+ cations in CuO crystallites or bulky oligomeric crystallites their reduction directly precedes to Cu
0 in the temperature range of 200–300 °C [
1,
6]. The reduction of monomeric Cu
2+ cations includes the reduction of Cu
2+ to Cu
+ at temperatures below 300 °C and, at higher temperatures, the reduction of monomeric Cu
+ to Cu
0 [
1,
6]. Thus, the H
2-TPR studies are very informative with respect to the determination of the types of copper species introduced into the zeolitic samples. The reduction profiles of Cu-FER_30 and Cu-FER_50 contain intensive double peaks at temperatures below 300 °C with the only low intensive and broad peaks at higher temperatures, which indicate the presence of copper mainly in the form of aggregated species with the small contribution of monomeric copper cations, as shown in
Figure 6. The reduction profiles of Cu-ITQ-6_30 and Cu-ITQ-6_50 are spread up to temperatures of 650–700 °C and contain at least three unresolved maxima. The low-temperature maxima, centered at about 235–245 °C, as mentioned earlier, could be attributed to the direct reduction of CuO to metallic copper and the reduction of monomeric Cu
2+ cations to Cu
+. However, the intensive high-temperature maxima, centered at about 475–505 °C and related to the reduction of isolated Cu
+ to Cu
0, may indicate the presence of mainly highly dispersed copper species in these samples. The peak at about 370 °C in the reduction profile of Cu-ITQ-6_30 and the arm at a similar temperature in the profile of Cu-ITQ-6_50 are possibly attributed to the reduction of finely dispersed CuO particles to elemental Cu
0 [
34,
35]. The maxima in the reduction profile of the sample with the higher Si/Al ratio, Cu-ITQ-6_50, are shifted to higher temperatures, compared to Cu-ITQ-6_30. A similar effect was reported in the scientific literature and was explained by the lower electronic density of the samples with lower aluminum content, which makes the reduction of the deposited metal species more difficult [
36,
37]. In the case of Cu-ITQ-36_30 and Cu-ITQ-36_50, the reduction profiles contained low-temperature peaks, centered at about 230–245 °C, and high-temperature maxima at about 445–470 °C, as shown in
Figure 6. The intensities of the high temperature peaks are significantly higher compared to the low-temperature ones, which indicates not only the significant contribution of highly dispersed copper species, but also the presence of Cu
+ cations in these samples. It is possible that part of the highly dispersed copper species was thermally reduced from Cu
2+ to Cu
+ during the treatment of the samples in a flow of pure argon at 500 °C, prior to the H
2-TPR runs. A similar effect was reported and analyzed for the Cu-ZSM-5 samples by Yang et al. [
38]. This effect may indicate a very high lability in Cu
2+ → Cu
+ reduction.
2.2. Catalytic Studies of NH3-SCR
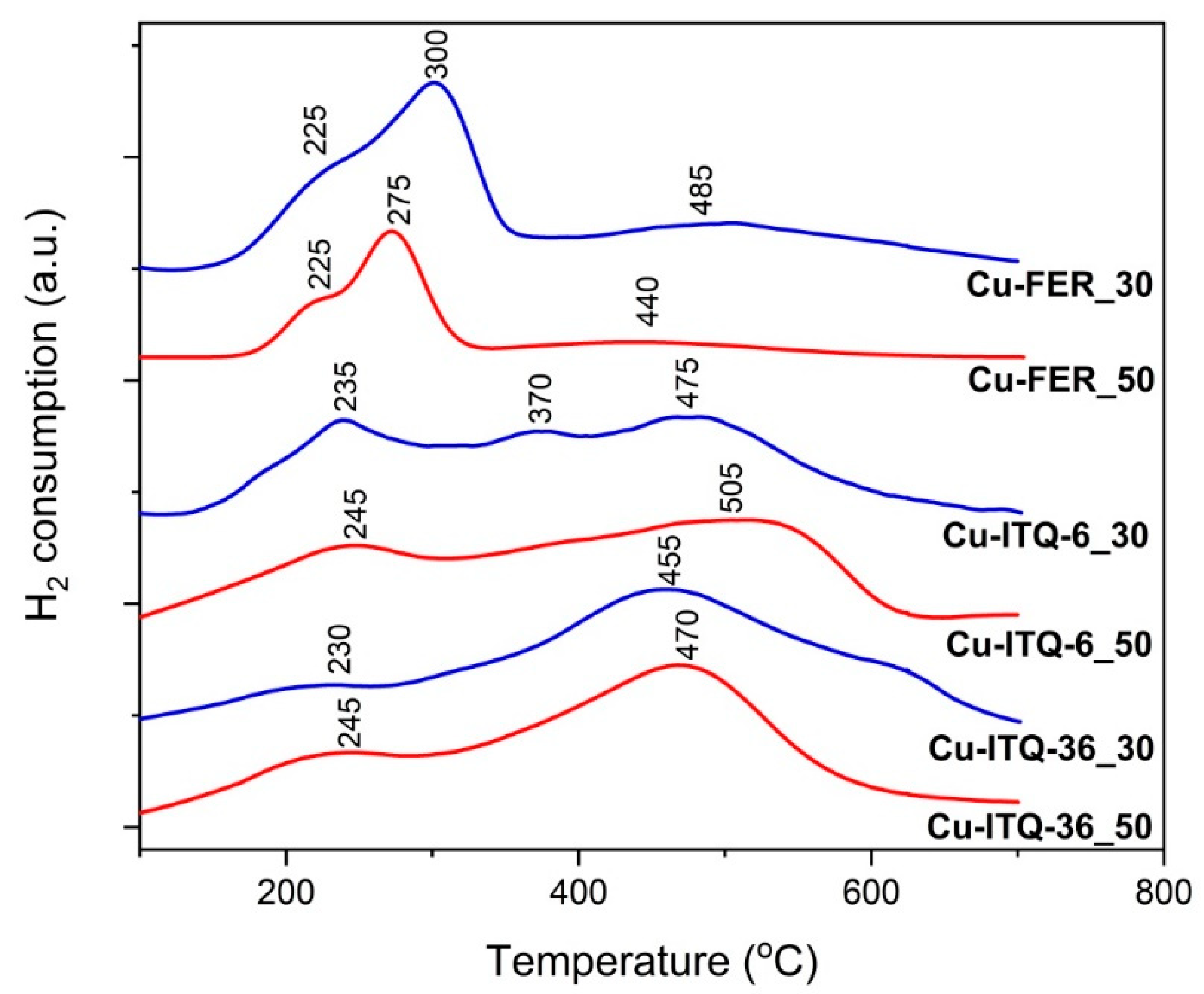
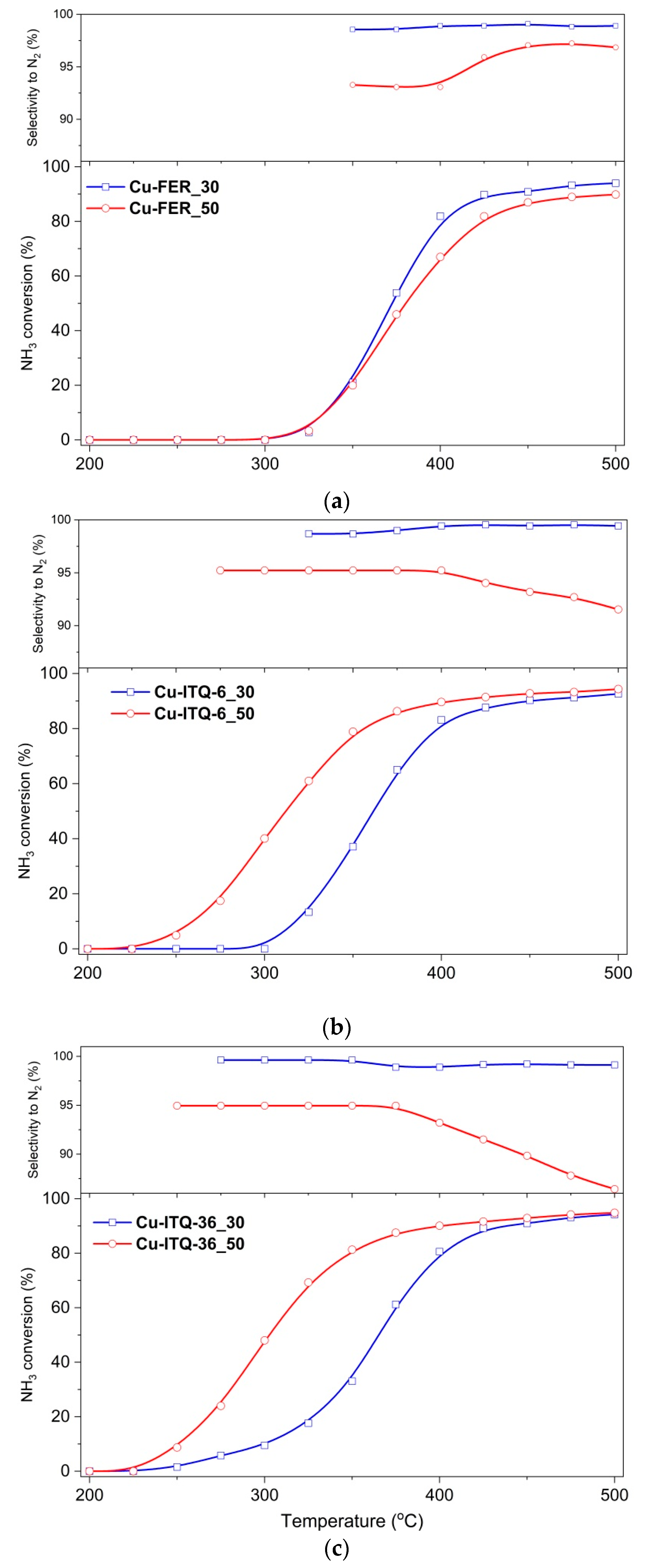
The results of the catalytic tests of copper modified zeolites in the NH
3-SCR process are presented in
Figure 7. The NO conversion in the presence of ferrierite-based catalysts started at about 150–175 °C and increased to about 375–400 °C, reaching 99 and 86% for Cu-FER_30 and Cu-FER_50, respectively. The decrease in the NO conversion, observed at higher temperatures, is related to the side reaction of ammonia oxidation by oxygen present in the reaction mixture. The selectivity to dinitrogen, also in the range characteristics of ammonia oxidation, was very high and did not drop below 97%. Intercalated and delaminated ferrierite zeolites were found to be significantly more catalytically active in the NH
3-SCR process. First of all, the NO conversion in the presence of the Cu-ITQ-6 catalysts started at about 125 °C— so at a temperature lower by about 25–50 °C than in the case of FER based catalysts—and then intensively increased to 275 °C, reaching about 97 and 95% for Cu-ITQ-6_30 and Cu-ITQ-6_50, respectively. Thus, in the low-temperature range the NO conversion profiles for both catalysts of this series are very similar, indicating the limited role of their surface acidity and copper loading. It can be seen that the catalyst with the higher aluminum content, Cu-ITQ-6_30, was more catalytically active at temperatures below 150 °C. As it was shown, the reduction of copper species in the zeolitic catalysts with the higher content of framework aluminum occurs at lower temperatures than for zeolites with the lower aluminum content, as shown in
Figure 6. Thus, the increased catalytic activity in the low-temperatures range is possibly attributed to the better reducibility of copper species deposited into zeolites. The catalytic performance of these samples is significantly different at higher temperatures. In the presence of Cu-ITQ-6_30, the NO conversion on the level 97–100% was observed in the range of 275–400 °C, while for Cu-ITQ-6_50 a decrease in the NO conversion, due to the side reaction of ammonia oxidation, occurred at about 325 °C. The selectivity to dinitrogen is higher for Cu-ITQ-6_30. Thus, the sample with the higher surface acidity, Cu-ITQ-6_30, is a more effective catalyst in the high-temperature NH
3-SCR process. Similar results were obtained for the series of the Cu-ITQ-36 samples. The catalyst with the higher aluminum content, Cu-ITQ-36_30, presented better catalytic activity at temperatures below 175 °C, possibly due to the better reducibility of copper species deposited into this sample compared to the copper species in the catalysts with the lower aluminum content, Cu-ITQ-36_50, as shown in
Figure 6. The NO conversion in the presence of Cu-ITQ-36_30 increased to about 400 °C, reaching 97%, while in the case of Cu-ITQ-6_50, the NO conversion increased to 93% at 300 °C and then decreased due to the side reaction of ammonia oxidation, as shown in
Figure 7. Thus, in this case, the acid properties of the catalysts also play a very important role in the high-temperature NH
3-SCR process. The comparison of the results, presented in
Figure 7, shows that the catalysts based on ITQ-6 are more active and selective than the analogous catalysts of the ITQ-36 series. On the other hand, the comparison of the results obtained for the FER series and their intercalated and delaminated forms shows that the most significant differences are related to the lower NO conversion for Cu-FER_30 and Cu-FER_50 in the low-temperature range, compared to the catalysts of the Cu-ITQ-6 and Cu-ITQ-36 series. The results of UV-vis DRS, as shown in
Figure 5, and H
2-TPR, as shown in
Figure 6, show a significantly higher contribution of highly dispersed copper species, mainly monomeric copper cations, in the catalysts of the Cu-ITQ-6 and Cu-ITQ-36 series, while the presence of more aggregated copper oxide species was postulated for the samples of the Cu-FER series. Such highly dispersed copper species, which can be reduced at relatively low temperatures, are possibly needed for the low-temperature conversion of NO. One of the possible mechanisms of the low-temperature NH
3-SCR is based on fast-SCR (2NH
3 + NO + NO
2 → 2N
2 + 3H
2O); however, for this reaction NO
2 is needed.