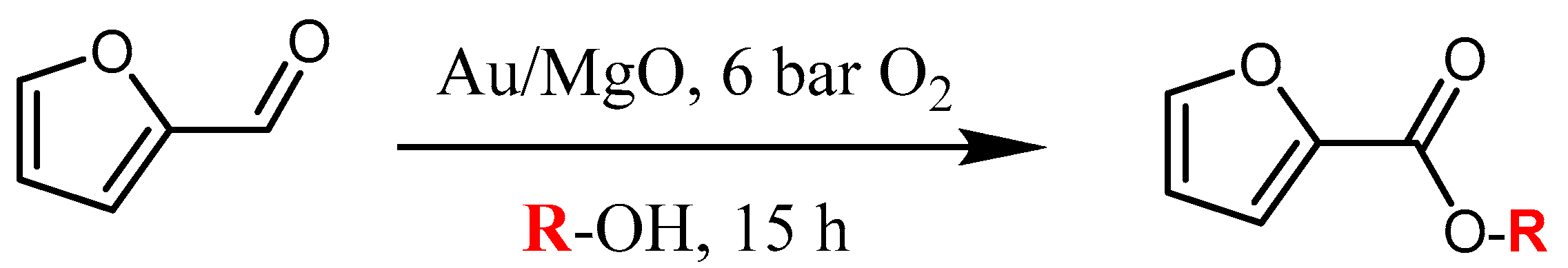1. Introduction
Currently, special attention has been given to the oxidation of biomass-derived substrates due to their potential to replace petroleum products [
1,
2,
3,
4,
5,
6,
7]. When considering the high extent of functionality and hydrophilic properties, biomass-derived substrates should be processed in liquid phase, typically in an aqueous phase [
1,
2,
3,
4]. Thus, the development of efficient liquid phase technologies is desired, as well as catalysts working under mild conditions [
1,
2,
3,
4]. Several routes for the conversion of bio-based compounds into fuels and highly valued chemicals involve the production of furfural and 5-hydroxymethylfurfural (HMF) (
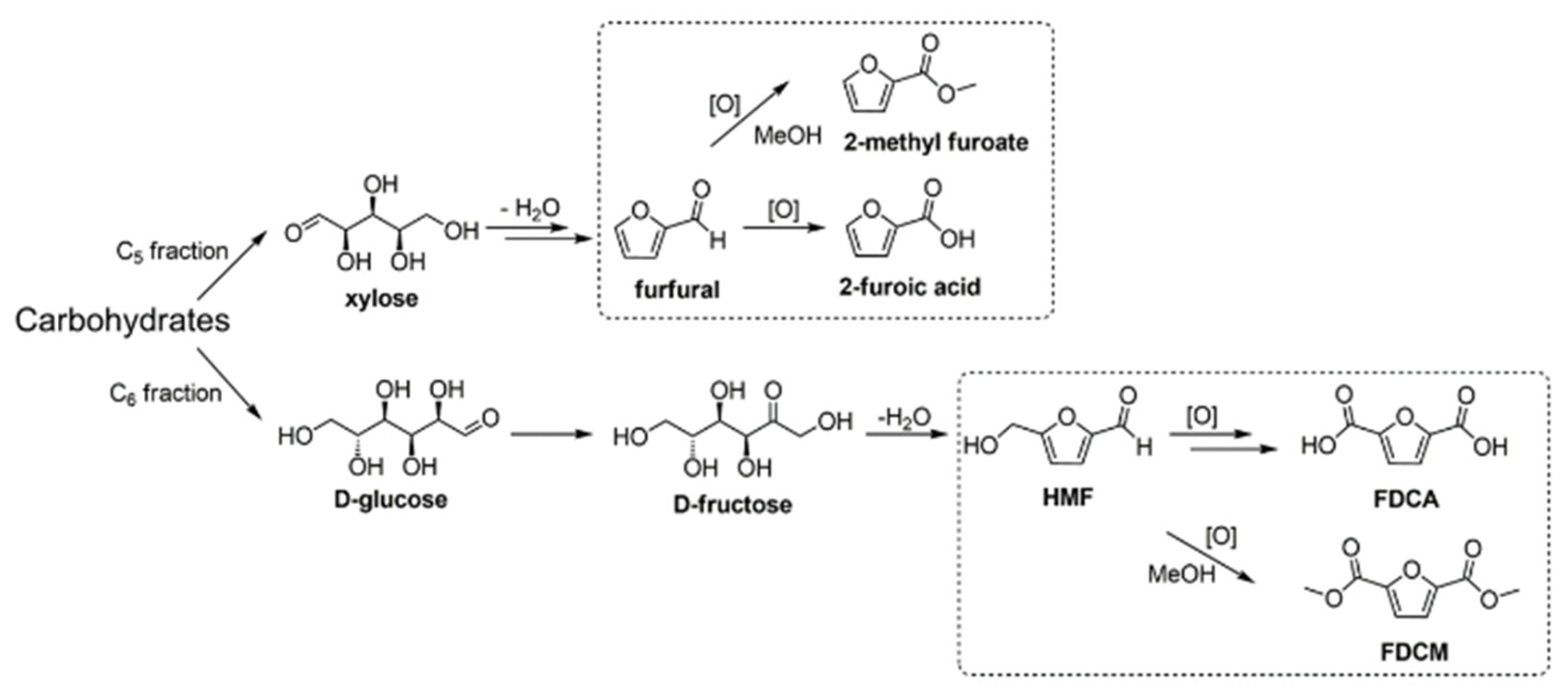
Scheme 1) [
8]. These are some of the products that are considered to be strategic for the development of biofuels and chemicals from biomass [
8,
9,
10]. HMF is considered to be one of the most important platform compounds in biorefineries of the future [
8,
9,
10]. Several products that are obtained
via HMF oxidation are of great interest for the polymer industry because their derivatives can be used as C6 monomers to replace petrochemical monomers [
1,
2,
3,
4,
5,
6,
7,
8,
9,
10]. For example, 2,5-furandicarboxylic acid (FDCA) is a monomer in the production of polyethylene 2,5-furandicarboxylate (PEF), being a green alternative to polyethylene terephthalate (PET) [
11,
12,
13]. The direct use of FDCA in the industry is difficult due to its low solubility in most commonly used solvents. The furan-2,5-dimethylcarboxylate ester (FDMC) is actually more suitable for the subsequent polymerization reaction, thanks to its better solubility. For this reason, the development of catalytic systems that are capable of producing FDMC directly from the HMF has received much attention today. One alternative is the production of FDCA can be obtained from furoic acid (furfural oxidation product) via the Henkel reaction [
13]. However, the use of Au catalysts can also be a viable alternative. FDCA can be obtained from furoic acid (furfural oxidation product)
via the Henkel reaction [
13]. Recently, Au catalysts have been successfully applied for the oxidative esterification of alcohols [
14,
15,
16,
17] and they have also been explored for the esterification of furfural and HMF [
18,
19,
20,
21,
22,
23,
24,
25,
26,
27,
28]. The first example of HMF esterification to produce dimethyl-2-furoate (FDMC) required the use of a base (NaOMe) [
29]. Although other catalytic systems have then been reported, there are few examples of successful Au catalytic systems for the production of baseless FDMC. Corma
et al. reported an Au/CeO
2 catalyst that was capable of performing the reaction in the absence of a base [
8]. In comparison with other catalysts, where total conversion was observed, the observed FDMC yield followed the order Au/Fe
2O
3 < Au/AC < Au/TiO
2 ~ Au/CeO
2. The high performance of Au/CeO
2 catalyst under base free conditions inspired the development of Au catalysts for oxidative furfural esterification in the absence of a base [
8]. Titania, ceria, and zirconia were studied by different research groups; however, Signoretto
et al. revealed that the performance of catalysts in the absence of a base followed the order: Au/ZrO
2 > Au/CeO
2 >> Au/TiO
2 [
30].
Later, Au/ZrO
2 was widely explored for the oxidative esterification reactions of furfural and HMF, due to the ability of ZrO
2 to provide a high dispersion of Au and its appropriate acid-base properties. Many parameters were also evaluated, such as the addition of dopants in the support (such as sulfates), the effect of the calcination of the support before catalyst preparation, and the calcination of the catalyst as a post-treatment step [
30]. However, there is still a lack of deeper understanding of which catalytic characteristics or sites of the support are directing the reaction selectivity to ester formation at high rates. Gupta
et al. [
25] reported the efficient oxidative esterification of furfural with propanol to high yield methyl-2-furoate in the presence of Au hydrotalcite supported catalyst, O
2, and a base (K
2CO
3). Moreover, Kegnæs
et al. studied the oxidative coupling of alcohols with N -hexylamine to form N-hexylamides [
15]. The authors found that methyl ester were formed in high yield by the furfural oxidation while using Au/TiO
2 and KOMe as a homogeneous base. However, this process requires a high quantity of base and it would be much greener and economic if no base would be used. The furfural oxidative esterification with methanol and without the addition of a base (NaCH
3O) was also reported [
31]. Gold that was supported on sulfated zirconia catalyst showed very good catalytic properties, if compared to Au/TiO
2 catalyst. The authors proposed that the increase in the activity of these materials was due to the presence of very small clusters of Au, which are able to dissociate O
2. This forms atomic oxygen with strong basic properties that could easily activate CH
3OH and increase the reaction rates [
31]. The application of gold catalysis in oxidations and oxidative esterification has grown a lot, but the issue of the use of base remains unsolved [
32].
Here, we seek to evaluate Au catalysts that are supported on alkaline-earth metal oxides (MgO, CaO, SrO, BaO) for the oxidative esterification of furfural without the addition of a base. The results will be compared to the gold supported on non-basic supports, such as TiO2, CeO2, and ZrO2. Furfural esters are of high industrial importance and, moreover, the studies with furfural can also serve as a model for the oxidative esterification of HMF, which is a molecule of greater complexity.
2. Results and Discussion
Au-based catalysts were prepared by the pre-formed nanoparticle immobilization method (sol immobilization method) because of its reproducibility. AuPVA NPs of about 3 nm, as determined by TEM [
32], were first prepared and then supported on different metal oxides. As expected, the Au nanoparticles size was preserved after immobilization on different metal oxides, e.g. an average size of 4 ± 0.8 nm was determined for all samples, as could be seen in
Figure 1. When considering the same Au loading (2 wt. %) and comparable Au particle size, it was expected that the catalytic performances would be mainly governed by the physicochemical properties and morphology of the support.
Specific surface areas of the prepared catalysts were measured while using the nitrogen physisorption method (BET). MgO showed the highest surface area of 50 m
2/g. CaO has a surface area of 8 m
2/g. BaO and SrO oxide supports have a very low surface area of 1 and 3.6 m
2/g (
Table 1). The specific surface area increased after the gold deposition due to the formation of polymorphous phases as confirmed by XRD studies.
Table 1 depicts the catalysts characterization regarding the Au content and specific surface area. Despite the nominal amount of Au was 2% wt., the real Au % that was obtained by Flame Atomic Absorption Spectroscopy (FAAS) was different. This is because of the properties of these oxides that form respective hydroxides during the catalyst synthesis. Alkaline earth oxides are known to hydrate in water forming M(OH)
2 species, which was also observed by XRD (
Figure 2). However, the M(OH)
2 are partially soluble and their solubility increase by decreasing the earth alkaline (EA) group. This could explain the increase in the Au content from Mg to Ba-catalyst, meaning that part of the support was dissolved during the synthesis. Being aware of that, pH corrections were performed during the synthesis, in which the pH of solution was adjusted to 7 prior the addition of the EA oxides for immobilization. Still, Au contents higher than 2% were observed.
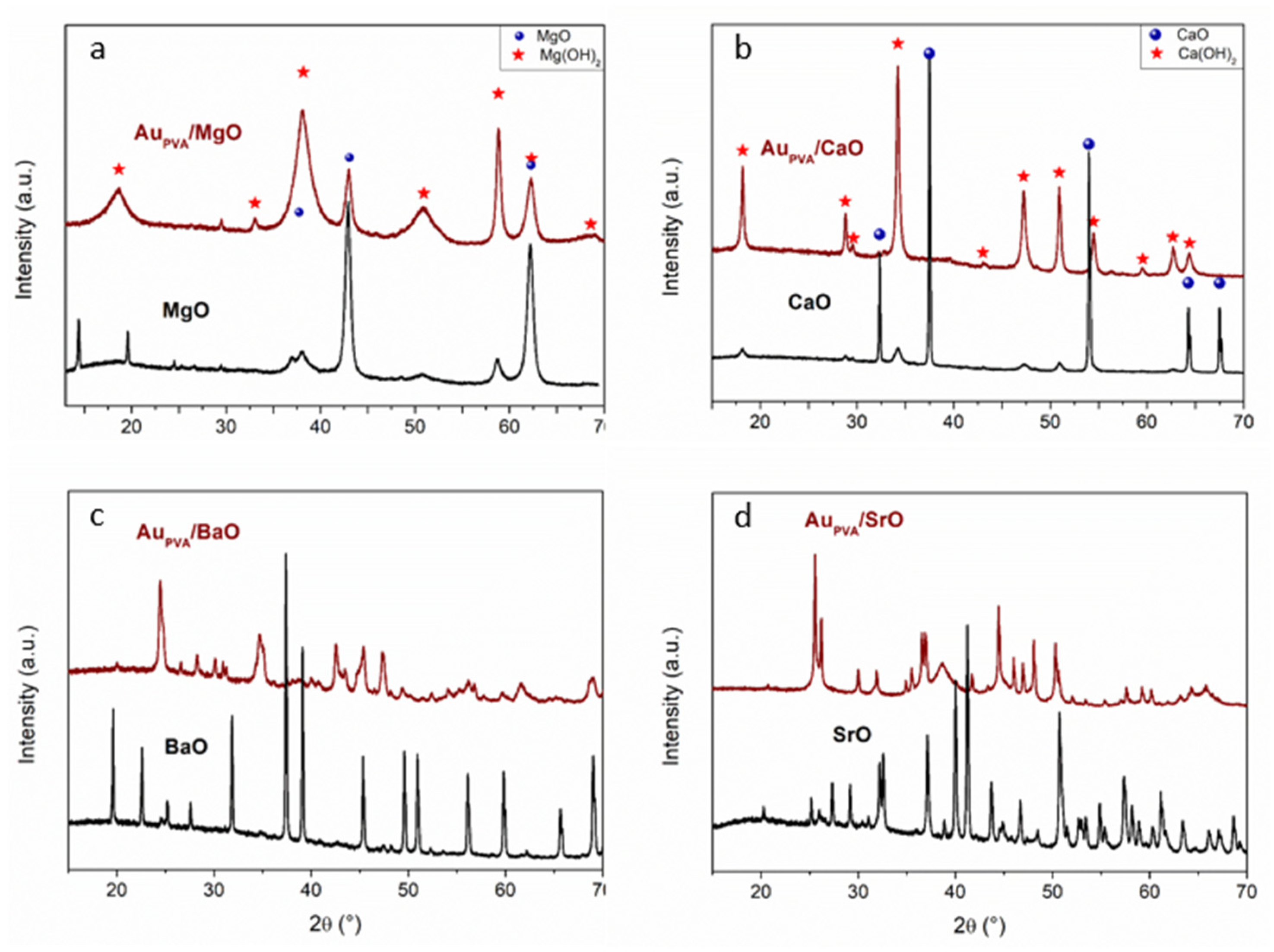
XRD analysis revealed that the sol immobilization method induces changes in the support structure. Indeed, the diffraction peaks originating from hydroxides are present, as could be seen from the
Figure 2 after preparation step. This is the case for all catalysts, but it is more pronounced for MgO and CaO oxides (
Figure 2a,b). XRD patterns of bare MgO support show diffraction peaks that can be attributed to MgO (periclase, Ref. code 01-075-0447). The catalyst prepared by the sol immobilization method presents low crystallinity (broad reflections) of the MgO peaks, whereas those that originated from Mg(OH)
2 are characterized by a more ordered structure (more intense peaks). It could be concluded that the preparation method does not result in the formation of a pure MgO phase. This is also the case of the Au/CaO sample where the presence of the hydroxide phase after catalyst preparation is well visible.
Most complicated XRD patterns were observed for other oxides. Indeed, different polymorphic structures are present. The main reflections of the SrO sample can be attributed to strontianite (Ref. Code 01-084-1778) [
33]. The diffraction patterns of the AU modified material exhibit reflections peaks that originate from strontianite as well as the reflections peaks originated from Sr(OH)
2 (Ref. Code 00-019-1276) and to Sr(OH)
2(H
2O) (Ref.Code 01-077-2336) [
33]. The very high reactivity of SrO toward water could explain the presence of these hydrated compounds [
33]. The same trend is observed for barium oxide. The presence of several polymorphus phases is observed, which also confirm that the calcination temperature is not high enough to obtain pure BaO phase. As expected in all cases, the reflections peaks from gold nanoparticles are not observed, which confirm the small particle size, as already observed by TEM analysis.
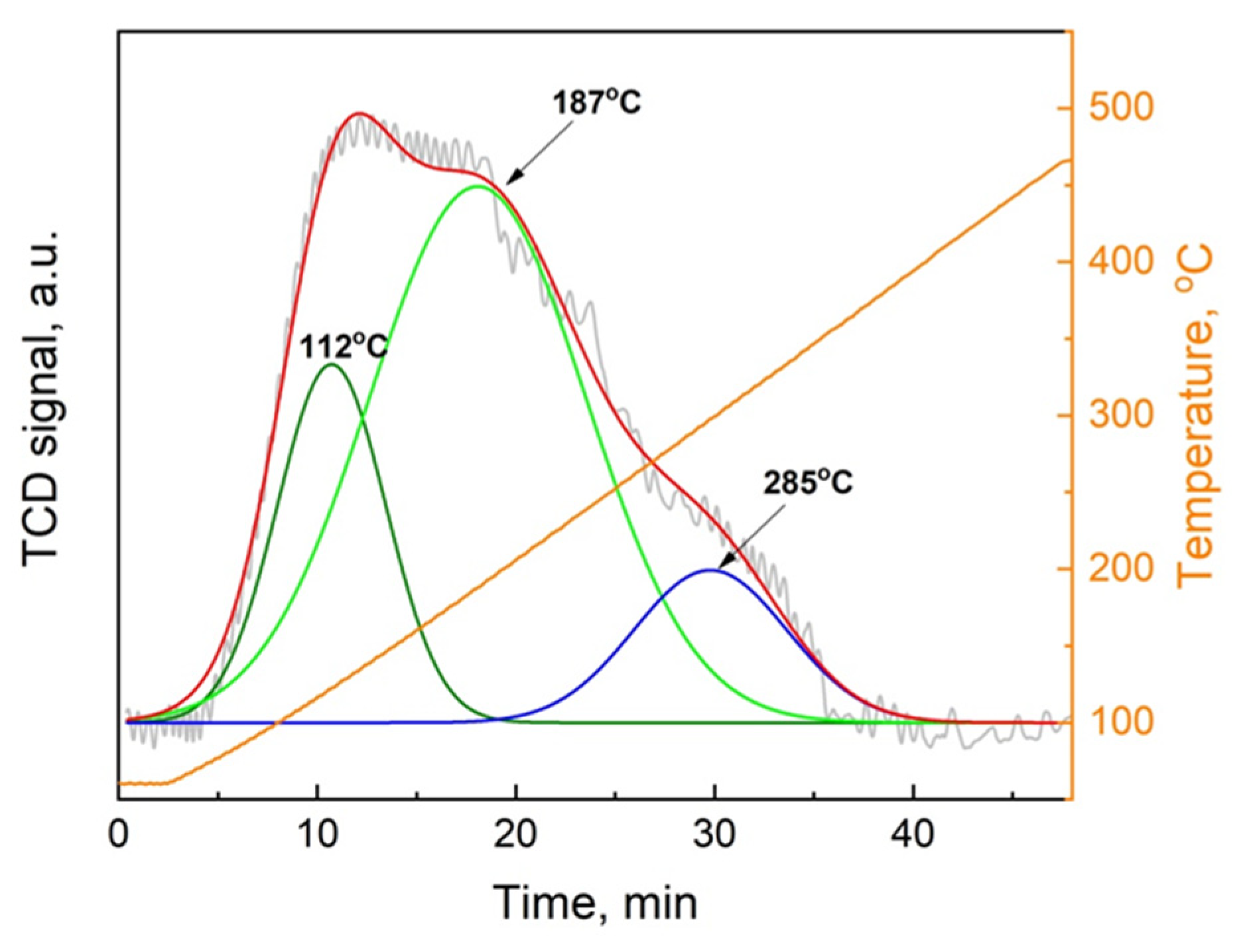
The TPD-CO
2 analysis was performed for all of the samples. However, only MgO oxide gave quantifiable results, as presented in
Figure 3. The total quantity of basic sites was evaluated at 0.265 mmol
CO2·g
−1. The desorption profile was decomposed into three components (
Figure 3), giving three well resolved peaks with the maximum at 112°C, 187°C, and 285°C. These positions corresponds to week, medium, and strong basic sites, respectively. Comparable quantity of week and strong basic sites were observed (0.058 and 0.035 mmol
CO2·g
−1, receptivity). Contrary to that, the quantity of the medium basic sites was almost three times higher (0.173 mmol
CO2·g
−1) than that of week basic sites. The results that are obtained for other oxides are difficult to quantify. In the case of CaO, the majority of the basic sites are with the desorption peak in the range of 550–700 °C. In the case of BaO and SrO, the desorption of CO
2 only starts at temperatures over 800 °C with the agreement with the literature data [
33]. This strongly indicates that both BaO and SrO oxides have high predominance of the very strong basic sites. This confirms the well-known basicity strength order for alkaline earth oxides: BaO > SrO > CaO > MgO [
33].
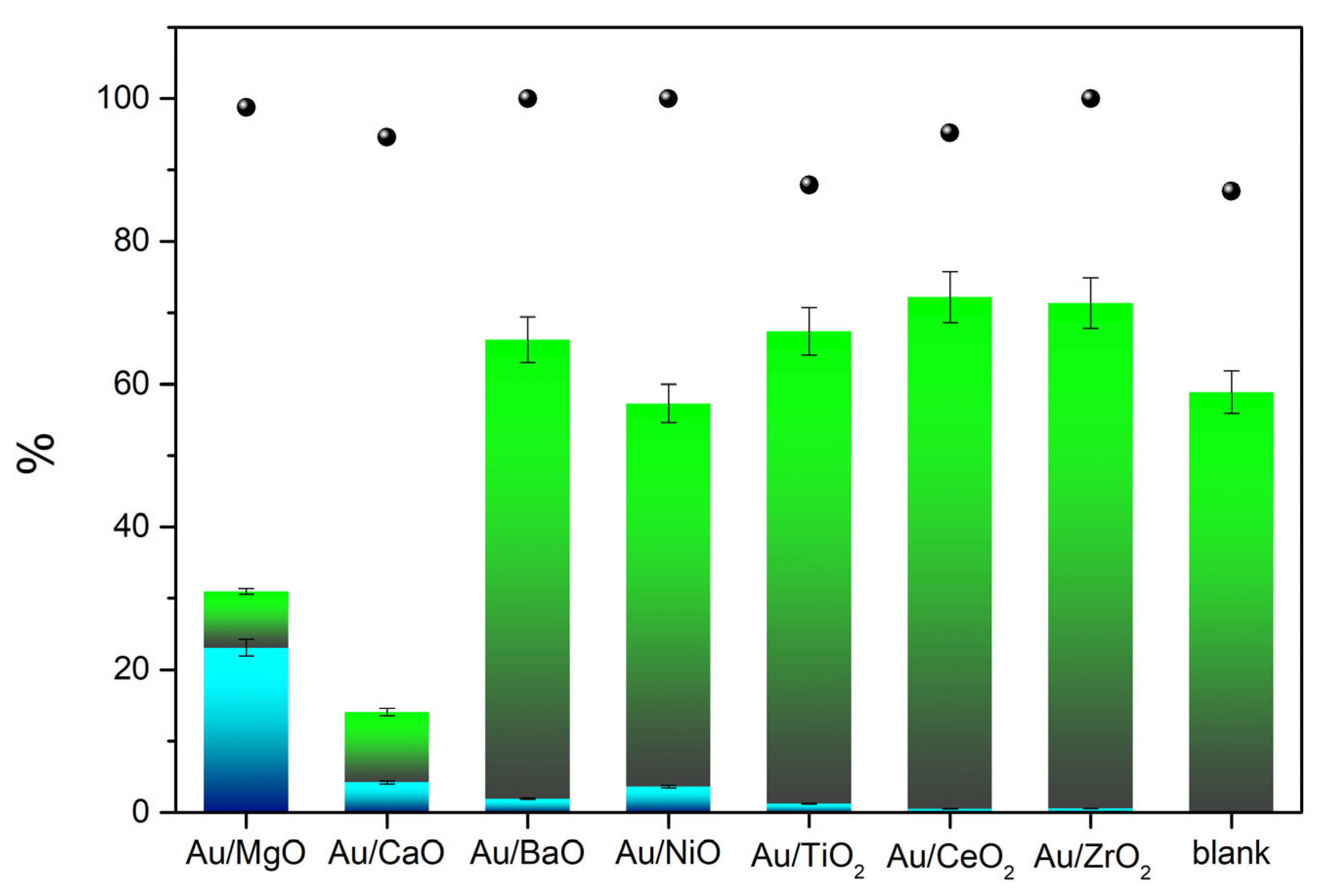
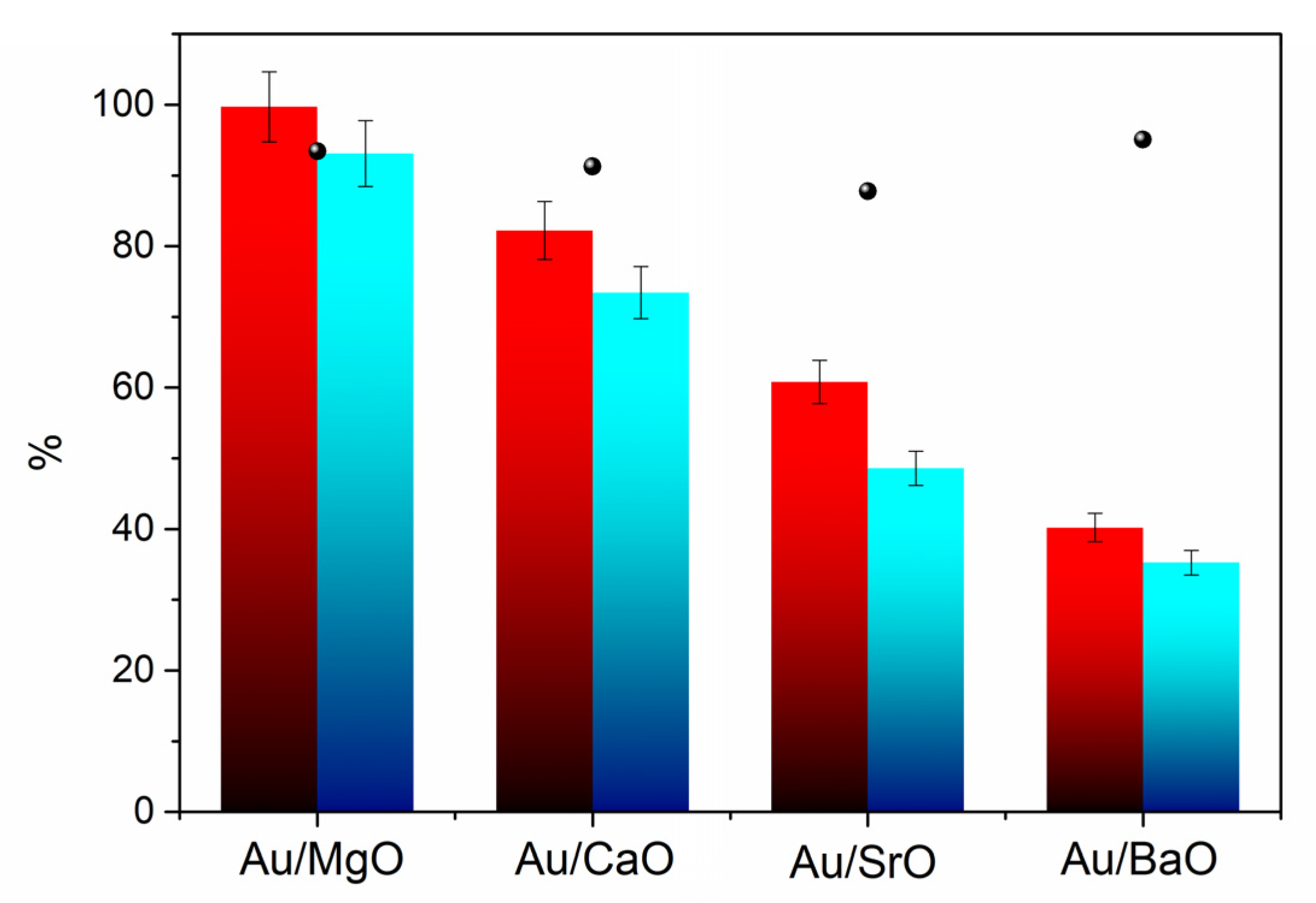
The synthesized Au catalysts were tested in the oxidative esterification of furfural in air (reactions performed in the SPR reactor), as shown in the
Figure 4. High conversions were observed for most of the Au catalysts and even the blank test showed more than 80% of furfural conversion. However, the yield of methyl furoate (MF) was negligible, in which dimethyl acetal furfural was the only by-product detected. With Au/MgO, Au/CaO, Au/NiO, and Au/BaO catalysts, MF was formed and up to 71% selectivity to MF was observed for Au/MgO sample. The presence of basic sites seem to be necessary for this reaction, since only catalysts containing basic supports (MgO, CaO, BaO, NiO) were effective for the oxidative esterification. Non-basic supports (TiO
2, CeO
2, ZrO
2) give rise to the acetalization reaction. Although oxidation occurs on the metal surface, the support also plays an important role by forming more reactive intermediates. According to Casanova et al., the hemiacetal can be converted to acetal (acetalization) or ester (oxidation), as shown in
Scheme 2 [
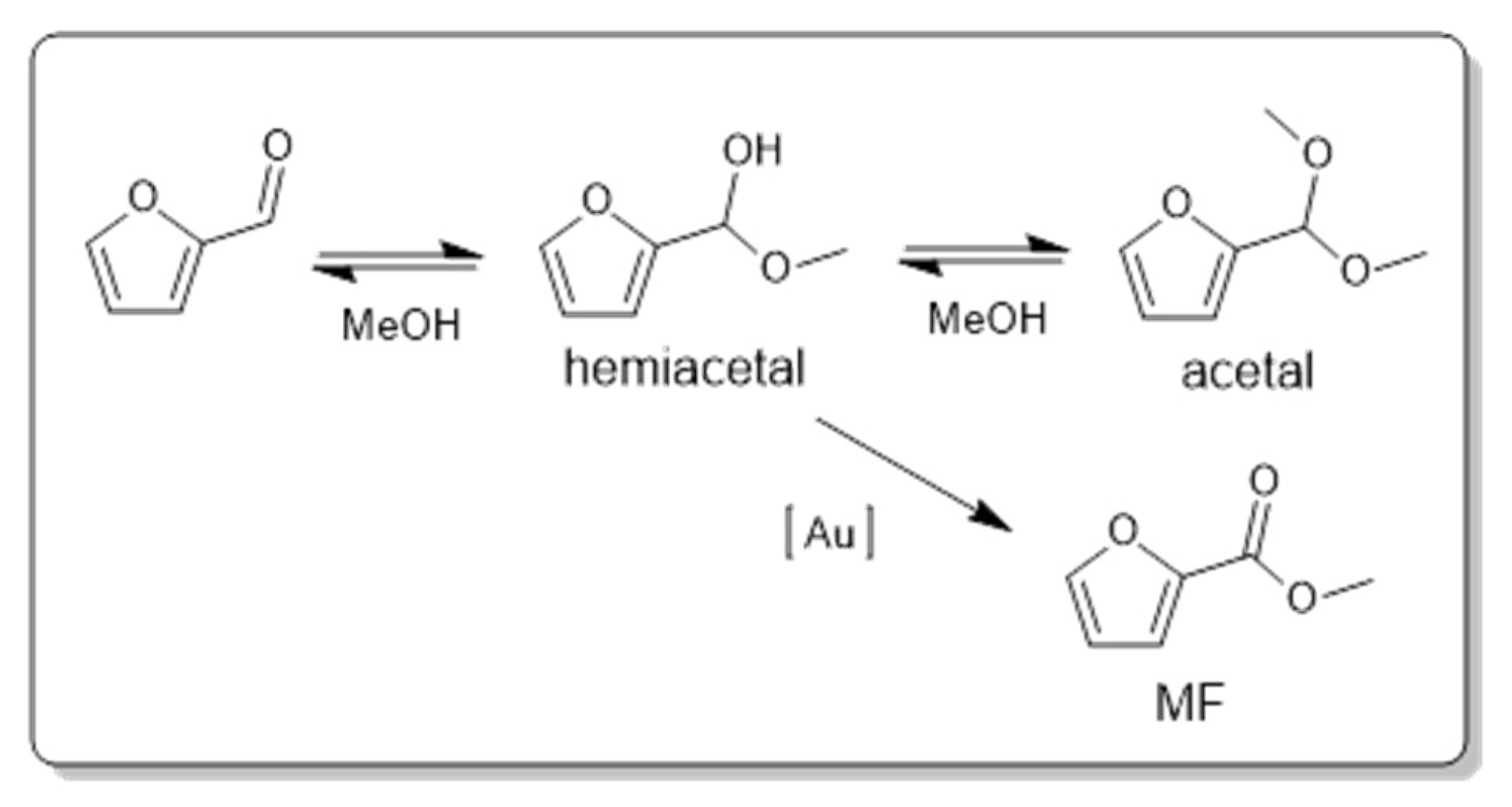
8]. A hemiacetal is formed as an intermediate that is later converted to an acetal when considering the general reaction mechanism for aldehydes reactivity. However, this is only valid when the esterification is conducted under acid conditions, otherwise the hemiacetal remains in equilibrium with the initial aldehyde (basic conditions).
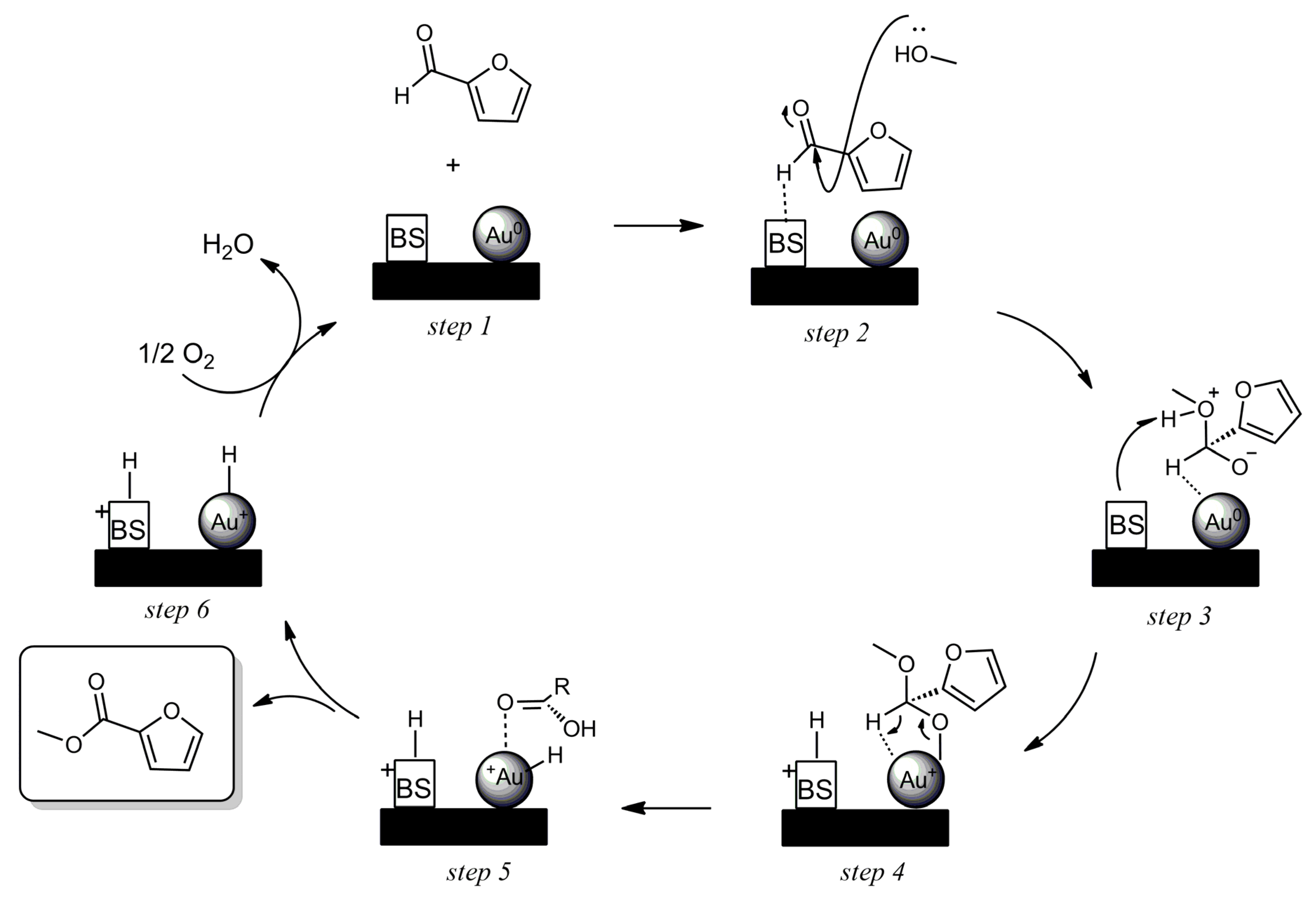
The reactivity of the different Au catalysts supported on basic oxides followed the same rule; when the intermediate hemiacetal was formed, the MF was almost exclusively produced, since the basic sites do not allow for the formation of the acetal For acid sites, both pathways are possible, so the high yield of furfural acetal obtained indicates that the catalyst is more prone to acetalization than oxidation. Although conversion from acetal to ester is also possible, some authors report that this is unlikely to occur. When considering the homogeneous mechanism, well known for this reaction, and the obtained results, a mechanism was proposed for the reaction occurring on the surface of the Au catalyst (
Scheme 3). The presence of basic sites favor the adsorption of furfural through the H
aldehyde, the resulting in the polarization of H
aldehyde–C bond. The electrophilic character of carbonyl allow for the nucleophilic attack of a molecule of methanol (step 2). Subsequently, the basic site is protonated and an alkoxyde group is formed (step 3). The Au-alkoxyde bond is formed, favoring then the β-hydrogenation (step 4). Further the regeneration of the catalyst in presence of oxygen (step 5–6) is well discussed by Zope
et al [
7,
26]. The presence of acid sites can also activate the H
aldehyde–C bond and favor the nucleophilic attack of methanol. However, in this case, the formation of the Au
alkoxyde bond, which is crucial for the mechanism, seems unlikely. Once formed, the O
alkoxyde can more probably bond to other acid sites than to Au.
These first results were encouraging and led us to study the effect of basic supports, when considering that possibly the variation of alkaline earth oxide basicity would lead us to the most active system. The higher activity that was observed for Au/MgO motivated us to optimize the reaction conditions, where different catalyst:substrate:MeOH ratios were studied (autoclave batch reactor, as in
Table 2). A higher furfural:MeOH ratio favors ester formation, since methanol is a reagent. However, the increase in the furfural:Au ratio has a more pronounced effect on activity. In all cases, the selectivity is quite high, but the furfural:Au ratio condition was chosen when considering the total conversion in the studied reaction time.
A more in-depth study was made with Au catalysts that were supported on basic oxides under these conditions (
Figure 5). The Sr-based catalyst was also synthesized and tested, as a decrease in activity had already been detected when going down in the alkaline earth group (Au/MgO > Au/CaO > Au/BaO) for furfural oxidation [
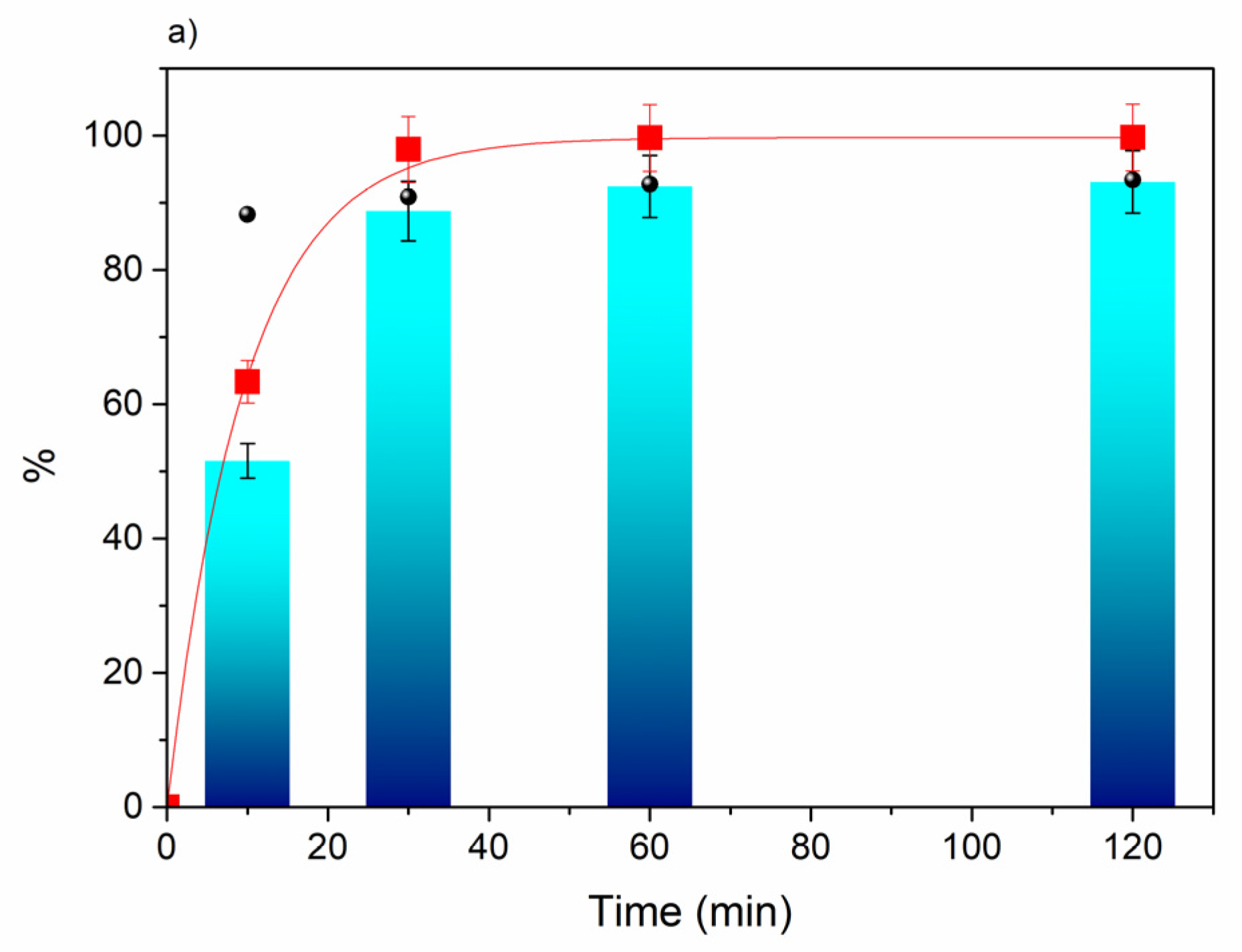
32]. The results show the same trend as previously observed, thus confirming the Au/MgO catalyst as the most active in this transformation. However, under these conditions, acetal was not observed and, therefore, selectivities to MF of at least 80% were obtained for alkaline-earth oxide-based catalysts. These results suggest that the basic sites present in these oxides are essential in directing the reaction path to the formation of the ester. Later studies with Au/MgO showed a very fast reaction kinetics, as shown in
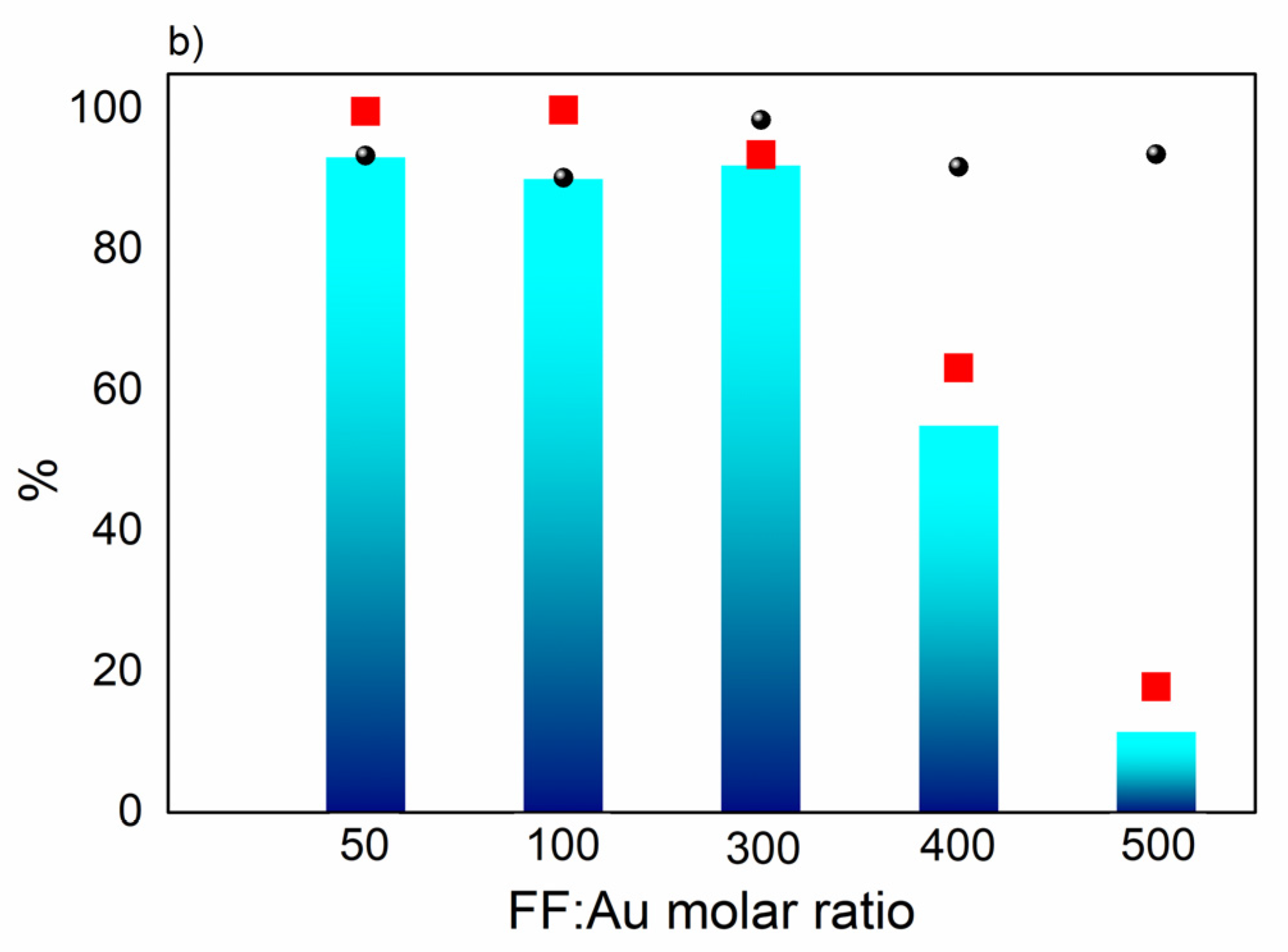
Figure 6a, in which complete conversion was observed in only 30 min. with high MF yield (>90%). Furthermore, regarding the substrate:metal ratio, higher furfural:Au ratios can be used with high MF yields (
Figure 6b), while a decrease of activity was observed for furfural:Au ratios that were higher than 300. However, the selectivity remained high in all ratios studied, highlighting the specificity of this transformation.
Recycling experiments were examined for the oxidative esterification of furfural using the following conditions: furfural: Au = 50 and 15 min. reaction while using the Au/MgO catalyst (
Figure 7). After the first reaction, the catalyst was recovered by centrifugation of the reaction mixture. The products were collected with a syringe and the recovered catalyst was washed with ethanol (2 mL) and then dried at 80 °C for 2 h. The reused catalyst was weighed and used in successive oxidation reactions, adding new portions of solvent and substrate. This procedure was repeated four times, however the Au/MgO catalyst increased its activity after the first cycle, when considering the same reaction time. After the first cycle, the furfural conversion went from 64 to 80%, while the selectivity to MF was maintained at 84%, resulting in 70% MF yield, as shown in
Figure 7. The catalyst did not undergo deactivation process, on the contrary, Au/MgO was more active after reuse and its catalytic activity remains the same, even after the 4
th reuse. This might be related to the removal of part of the PVA that acts as a stabilizer for Au NPs. Stabilizers can block access to specific sites on the catalyst surface and, therefore, some authors report the removal of these stabilizers by several techniques (calcination, O
2 treatment, solvent washing) in order to improve the catalytic performance of catalysts prepared from pre-formed nanoparticles. However, the reaction condition itself might represent a stabilizer washing treatment, since the catalyst remains at a higher temperature of 100 °C in methanol and under agitation. Additionally, the fact that this activity is maintained in the other cycles rules out any leaching or metal loss.
The high activity of this Au catalyst motivated us to study the efficiency of the oxidative esterification reaction while using other alcohols, from long and branched carbon chains ones. The reaction was slower in the presence of these alcohols and, therefore, the catalytic tests were run for 15 h and furfural ratio: Au = 50 (
Table 3).
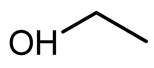
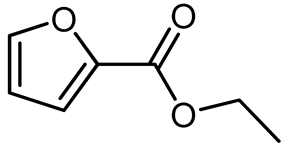
Oxidative esterification in the presence of ethanol (EthOH,
Table 3, entry 1), isopropanol (i-PrOH,
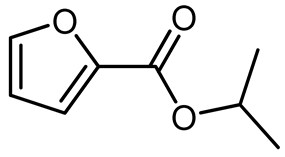
Table 3, entry 2), n-butanol (n-ButOH,
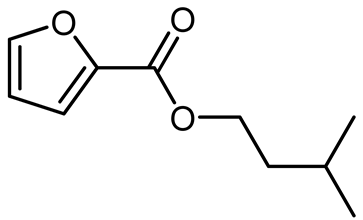
Table 3, entry 3), and isopentanol (i-PentOH,
Table 3, entry 4) was also successful, leading to the formation of the corresponding esters (ethyl-, isopropyl-, n-butyl-, and isopentyl furoates). The respective esters were formed with high selectivity (>99%), and the linear and long carbon chain alcohols provided the highest yields, as is the case of the n-butyl furoate that formed with 92% yield. Branched alcohols represent a higher steric hindrance when compared to linear alcohols and, therefore, esterification in this case is more difficult. Long-chain alcohols also allow for higher conversion, even though they present less nucleophilicity when compared to short-chain alcohols.
3. Materials and Methods
3.1. Materials
Chloroauric acid trihydrate (HAuCl4·3H2O, 99.9%, Sigma–Aldrich, Saint Louis, MO, USA), sodium borohydride (NaBH4, 98%, Sigma–Aldrich, Saint Louis, MO, USA), poly(vinyl alcohol) (PVA, Sigma–Aldrich, Saint Louis, MO, USA, MW 55,000 g/mol), sodium citrate (Na3C6H5O7, 99%, Sigma–Aldrich, Saint Louis, MO, USA), furfural (C5H4O2, >99%, Sigma–Aldrich, Saint Louis, MO, USA), 5-hydroxymethylfurfural (HMF, 99%, Sigma–Aldrich, Saint Louis, MO, USA), furoic acid (FA, 99%, Sigma–Aldrich, Saint Louis, MO, USA), 5-hydroxymethyl-2-furancarboxylic acid (HMFCA, >99%, Sigma–Aldrich, Saint Louis, MO, USA), 2,5-diformylfuran (DFF, >99%, Sigma–Aldrich, Saint Louis, MO, USA), 5-formyl-2-furoic acid (FFCA, >99%, Sigma–Aldrich, Saint Louis, MO, USA), and 2,5-furandicarboxylic acid (FDCA, 99%, Sigma–Aldrich, Saint Louis, MO, USA) were of analytical grade and used as received. Deionized water (18.2 MΩ) was used for the preparation of all the needed solutions. MgO and Cao were commercial oxides that were purchased from Sigma–Aldrich, Saint Louis, MO, USA (<99%). BaO and SrO were synthetized by thermal decomposition of nitrate salts at 700 °C.
3.2. Synthesis of Au Based Catalysts
The Au nanoparticles were prepared according to the literature [
2,
3]. A 2% polyvinyl alcohol (PVA) solution was added to an aqueous solution of HAuCl
4 (5 × 10
−4 mol L
−1) under vigorous stirring PVA/Au (w/w) = 1.2); to this solution a freshly prepared 0.1 mol L
−1 NaBH
4 solution (NaBH
4/Au (mol/mol) = 5) was then added, forming a reddish colloid (Au nanoparticles). After 30 min., the nanoparticles were immobilized by the addition of support (MgO, CaO, SrO, BaO, NiO, TiO
2, CeO
2, and ZrO
2) under vigorous agitation. The amount of support was calculated to result in a final metal loading of 2 wt % (nominal). After 2 h, the mixture was filtered and the solid was washed with 60 °C Milli-Q water (2 × 25 mL) and ethanol (2 × 25 mL) and then dried at 100 °C for 1 h.
3.3. Oxidative Esterification Reactions of Furfural
Some of the reactions were performed using the Freeslate MultiReactor available on the REALCAT EQUIPEX platform at Centrale Lille. It consists of 24 parallel batch reactors for high performance screening, in which each reactor was loaded with a furfural methanolic solution (2 mL, 247 mmol L−1) and the Au based catalyst (10 mg, 0.9 µmol of metal). The reaction was performed at 20-bar of air (final pressure 26 bar), 110 °C, 600 rpm, 2 h. Some reactions were performed in a TopIndustry (Vaux le Penil, France) autoclave reactor. The reactor was loaded with a furfural methanolic solution (20 mL, 24.7 or 247 mmol L−1) and the Au based catalyst (100 mg, 9 µmol of metal). The reaction was performed at 6-bar of O2, 110 °C, 600 rpm, at different reaction times, from two to six hours. In both cases, after the reaction, the catalyst was removed by filtration, the liquid products were diluted in methanol and analyzed by gas chromatography. The analysis of the products was performed in a gas chromatograph that was equipped with a CARBOWAX column for the analysis of furfural and reaction products.
3.4. Characterization
The Flame Atomic Absorption Spectroscopy (FAAS) method was used to determine the amount of metal deposited on the oxides. The samples that were digested in aqua regia analyzed on Shimadzu AA-6300 atomic absorption spectrometer (Columbia, MD, USA).
The ICP-OES (Inductively Coupled Plasma Optical Emission Spectrometry) analysis was performed using Agilent 720-ES ICP-OES equipment (Santa Clara, CA, USA) combined with Vulcan 42S automated digestion system.
The powder X-ray diffraction patterns (XRD) were recorded with a Bruker AXS D8 Advance diffractometer (Billerica, MA, USA). The samples were scanned at 0.014° s−1 over the range 20 ≤ 2θ ≤ 80°.
The (BET) specific surface areas measurements were determined by N2 adsorption at −196 °C while using a Micromeritics ASAP 2010 sorptometer (Norcross, GA, USA).
Transmission Electron Microscopy (TEM) images were recorded placing a drop of the particles dispersion in isopropanol over a carbon film supported on a cooper grid. FEI Tecnai microscope (Hillsboro, Ore. USA) was used for the recording of the images.
TPD-CO2 was carried out on a Micromeritics ChemiSorb 2705 (Norcross, GA, USA). Before the CO2 adsorption, the samples were pre-treated in situ in He at 500 °C for 30 min. CO2 was adsorbed at 50 °C for 1 h. TPD-CO2 measurements were performed in the temperature range of 50–700 °C while using a ramp rate of 10 °C min−1. A thermal conductivity detector (TCD) was used for the quantification of the desorbed CO2.