Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings
Abstract
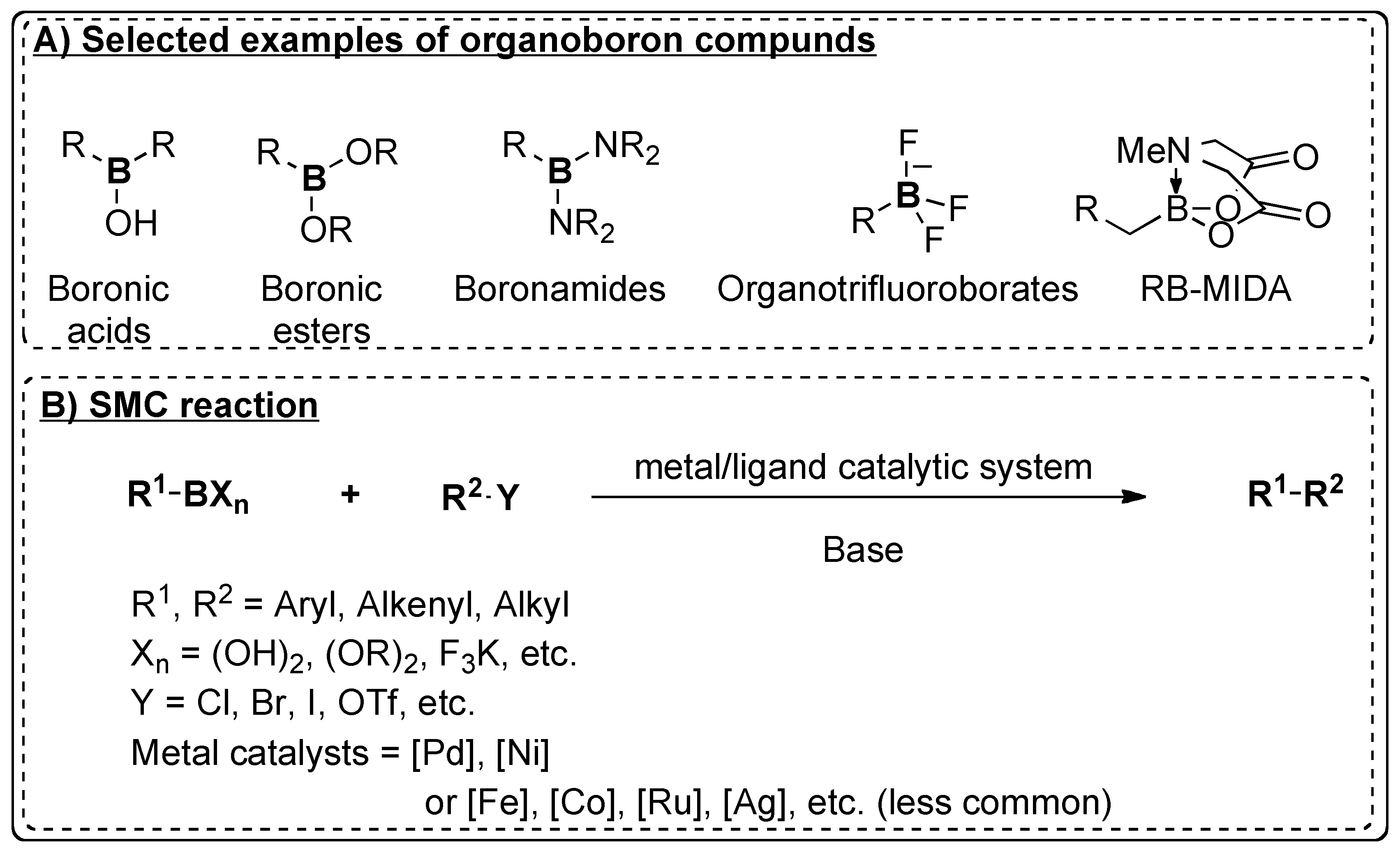
1. Introduction
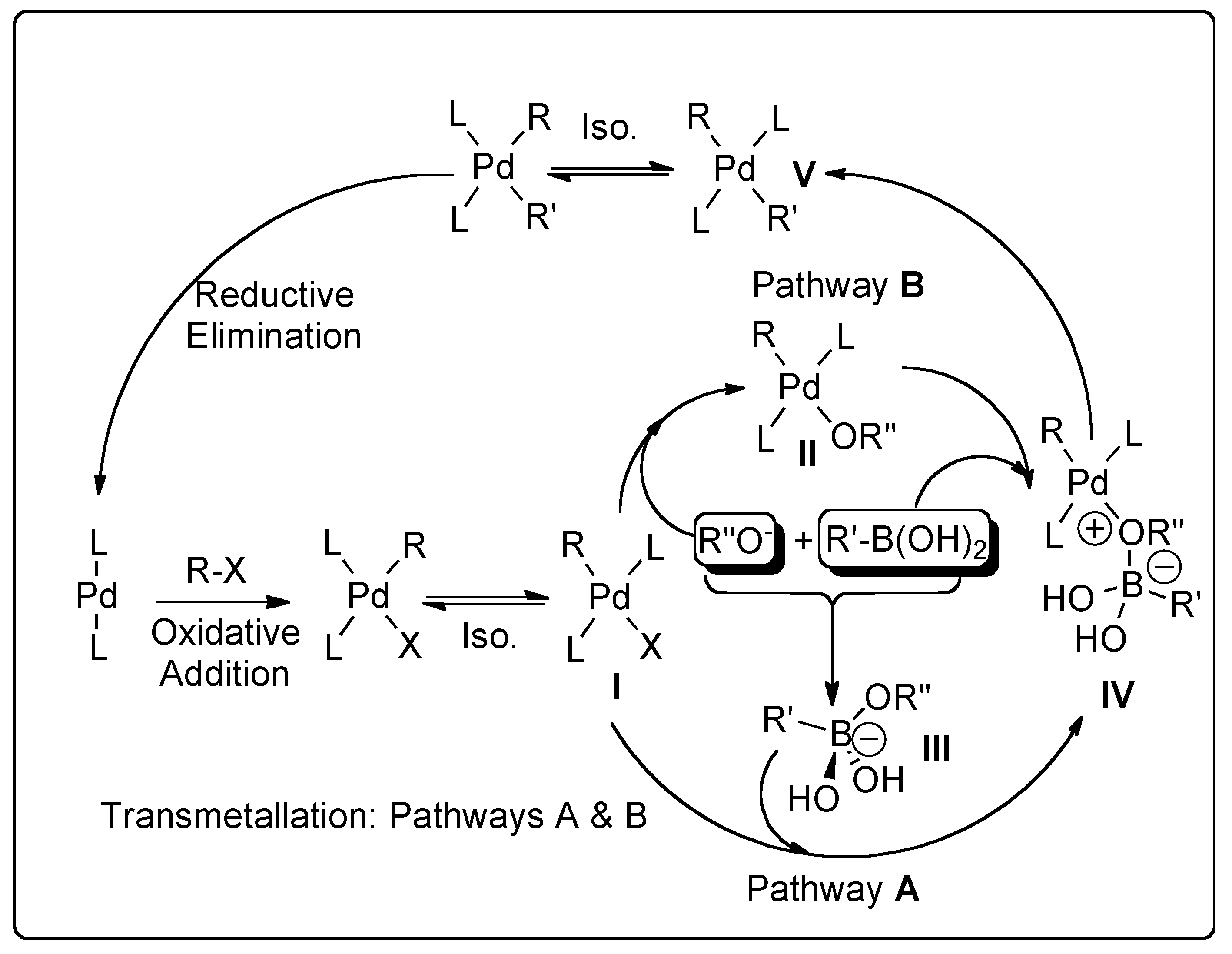
2. Suzuki–Miyaura Cross-Coupling (SMC)
3. First Reports of B-alkyl SMC and Methods Employing 9-BBN Derivatives
4. Organotrifluoroborates in sp3–sp2 SMCs
5. Other Alkylboranes in sp3–sp2 SMCs
6. Alkylboronic Acids in sp3–sp2 SMCs
7. Boronic Esters and MIDA Boronates in sp3–sp2 SMCs
8. B–Alkyl SMCs Using BBN Variants (9-MeO-9-BBN and OBBD Derivatives)
9. Selected Examples of Applications of SMCs and B–alkyl SMC in the Synthesis of Target Molecules
10. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Frenking, G. Peculiar boron startles again. Nature 2015, 522, 297–298. [Google Scholar] [CrossRef]
- Kumar, R.; Karkamkar, A.; Bowden, M.; Autrey, T. Solid-state hydrogen rich boron-nitrogen compounds for energy storage. Chem. Soc. Rev. 2019, 48, 5350–5380. [Google Scholar] [CrossRef]
- William Lipscomb Biographical. Available online: https://www.nobelprize.org/prizes/chemistry/1976/lipscomb/biographical/ (accessed on 22 February 2020).
- Brown, H.C. From Little Acorns to Tall Oaks: From Boranes through Organoboranes. Available online: https://www.nobelprize.org/prizes/chemistry/1979/summary/ (accessed on 22 February 2020).
- Miyaura, N.; Suzuki, A. Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds. Chem. Rev. 1995, 95, 2457–2483. [Google Scholar] [CrossRef]
- Maluenda, I.; Navarro, O. Recent developments in the Suzuki-Miyaura reaction: 2010–2014. Molecules 2015, 20, 7528–7557. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Yamamoto, Y. Cross-coupling Reactions of Organoboranes: An Easy Method for C–C Bonding. Chem. Lett. 2011, 40, 894–901. [Google Scholar] [CrossRef]
- Defrancesco, H.; Dudley, J.; Coca, A. Boron Chemistry: An Overview. ACS Symp. Ser. 2016, 1236, 1–25. [Google Scholar]
- Brown, H.C.; Cole, T.E. Organoboranes. 31 A Simple Preparation of Boronic Esters from Organolithium Reagents and Selected Trialkoxyboranes. Organometallics 1983, 2, 1316–1319. [Google Scholar] [CrossRef]
- Fyfe, J.W.B.; Watson, A.J.B. Recent Developments in Organoboron Chemistry: Old Dogs, New Tricks. Chem 2017, 3, 31–55. [Google Scholar] [CrossRef]
- Dimitrijević, E.; Taylor, M.S. Organoboron acids and their derivatives as catalysts for organic synthesis. ACS Catal. 2013, 3, 945–962. [Google Scholar] [CrossRef]
- Akira Suzuki Nobel Lecture Nobel Prizes 2019. Available online: https://www.nobelprize.org/prizes/chemistry/2010/suzuki/lecture/ (accessed on 22 February 2020).
- Das, B.C.; Thapa, P.; Karki, R.; Schinke, C.; Das, S.; Kambhampati, S.; Banerjee, S.K.; Van Veldhuizen, P.; Verma, A.; Weiss, L.M.; et al. Boron chemicals in diagnosis and therapeutics. Future Med. Chem. 2013, 5, 653–676. [Google Scholar] [CrossRef]
- Klotz, J.H.; Moss, J.I.; Zhao, R.; Davis, L.R.; Patterson, R.S. Oral toxicity of boric acid and other boron compounds to immature cat fleas (Siphonaptera: Pulicidae). J. Econ. Entomol. 1994, 87, 1534–1536. [Google Scholar] [CrossRef] [PubMed]
- Ban, X.; Jiang, W.; Sun, K.; Xie, X.; Peng, L.; Dong, H.; Sun, Y.; Huang, B.; Duan, L.; Qiu, Y. Bipolar host with multielectron transport benzimidazole units for low operating voltage and high power efficiency solution-processed phosphorescent OLEDs. ACS Appl. Mater. Interfaces 2015, 7, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Jäkle, F. Recent Advances in the Synthesis and Applications of Organoborane Polymers. In Synthesis and Application of Organoboron Compounds; Fernández, E., Whiting, A., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 297–325. ISBN 978-3-319-13053-8. [Google Scholar]
- Matsumi, N.; Sugai, K.; Sakamoto, K.; Mizumo, T.; Ohno, H. Direct synthesis of poly(lithium organoborate)s and their ion conductive properties. Macromolecules 2005, 38, 4951–4954. [Google Scholar] [CrossRef]
- Liu, L.; Corma, A. Metal Catalysts for Heterogeneous Catalysis: From Single Atoms to Nanoclusters and Nanoparticles. Chem. Rev. 2018, 118, 4981–5079. [Google Scholar] [CrossRef]
- Toffoli, D.; Stredansky, M.; Feng, Z.; Balducci, G.; Furlan, S.; Stener, M.; Ustunel, H.; Cvetko, D.; Kladnik, G.; Morgante, A.; et al. Electronic properties of the boroxine–gold interface: Evidence of ultra-fast charge delocalization. Chem. Sci. 2017, 8, 3789–3798. [Google Scholar] [CrossRef]
- Chen, C.C.; Fan, H.J.; Shaya, J.; Chang, Y.K.; Golovko, V.B.; Toulemonde, O.; Huang, C.H.; Song, Y.X.; Lu, C.S. Accelerated ZnMoO4 photocatalytic degradation of pirimicarb under UV light mediated by peroxymonosulfate. Appl. Organomet. Chem. 2019, 33, 1–15. [Google Scholar]
- Chardon, A.; Mohy El Dine, T.; Legay, R.; De Paolis, M.; Rouden, J.; Blanchet, J. Borinic Acid Catalysed Reduction of Tertiary Amides with Hydrosilanes: A Mild and Chemoselective Synthesis of Amines. Chem. Eur. J. 2017, 23, 2005–2009. [Google Scholar] [CrossRef]
- Shaya, J.; Deschamps, M.A.; Michel, B.Y.; Burger, A. Air-Stable Pd Catalytic Systems for Sequential One-Pot Synthesis of Challenging Unsymmetrical Aminoaromatics. J. Org. Chem. 2016, 81, 7566–7573. [Google Scholar] [CrossRef]
- Papageridis, K.N.; Siakavelas, G.; Charisiou, N.D.; Avraam, D.G.; Tzounis, L.; Kousi, K.; Goula, M.A. Comparative study of Ni, Co, Cu supported on γ-alumina catalysts for hydrogen production via the glycerol steam reforming reaction. Fuel Process. Technol. 2016, 152, 156–175. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Costa, C.N.; Efstathiou, A.M. The steam reforming of phenol reaction over supported-Rh catalysts. Appl. Catal. A Gen. 2004, 272, 37–52. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Papageridis, K.N.; Baklavaridis, A.; Tzounis, L.; Polychronopoulou, K.; Goula, M.A. Hydrogen production via the glycerol steam reforming reaction over nickel supported on alumina and lanthana-alumina catalysts. Int. J. Hydrog. Energy 2017, 42, 13039–13060. [Google Scholar] [CrossRef]
- Karamé, I.; Shaya, J.; Srour, H. Carbon Dioxide Chemistry, Capture and Oil Recovery; IntechOpen: London, UK, 2018; ISBN 9781789235753. [Google Scholar]
- Mohy El Dine, T.; Evans, D.; Rouden, J.; Blanchet, J. Mild Formamide Synthesis through Borinic Acid Catalysed Transamidation. Chem. Eur. J. 2016, 22, 5894–5901. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Shaya, J.; Fan, H.J.; Chang, Y.K.; Chi, H.T.; Lu, C.S. Silver vanadium oxide materials: Controlled synthesis by hydrothermal method and efficient photocatalytic degradation of atrazine and CV dye. Sep. Purif. Technol. 2018, 206, 226–238. [Google Scholar] [CrossRef]
- Holstein, P.M.; Dailler, D.; Vantourout, J.; Shaya, J.; Millet, A.; Baudoin, O. Synthesis of Strained γ-Lactams by Palladium(0)-Catalyzed C(sp3)-H Alkenylation and Application to Alkaloid Synthesis. Angew. Chem. Int. Ed. 2016, 55, 2805–2809. [Google Scholar] [CrossRef]
- Karamé, I.; Zaher, S.; Eid, N.; Christ, L. New zinc/tetradentate N4 ligand complexes: Efficient catalysts for solvent-free preparation of cyclic carbonates by CO2/epoxide coupling. Mol. Catal. 2018, 456, 87–95. [Google Scholar] [CrossRef]
- Ridgway, B.H.; Woerpel, K.A. Transmetalation of Alkylboranes to Palladium in the Suzuki Coupling Reaction Proceeds with Retention of Stereochemistry. J. Org. Chem. 1998, 63, 458–460. [Google Scholar] [CrossRef]
- Johnson, C.R.; Braun, M.P. A Two-step, Three-Component Synthesis of PGE1: Utilization of a-Iodoenones in Pd(0)-Catalyzed Cross-Couplings of Organoboranest. J. Am. Chem. Soc. 1993, 115, 11014–11015. [Google Scholar] [CrossRef]
- Chemler, S.R.; Trauner, D.; Danishefsky, S.J. The B -alkyl Suzuki−Miyaura cross-coupling reaction: Development, mechanistic study, and applications in natural product synthesis. Angew. Chem. Int. Ed. 2001, 40, 4544–4568. [Google Scholar] [CrossRef]
- Dreher, S.D.; Dormer, P.G.; Sandrock, D.L.; Molander, G.A. Efficient cross-coupling of secondary alkyltrifluoroborates with aryl chlorides-reaction discovery using parallel microscale experimentation. J. Am. Chem. Soc. 2008, 130, 9257–9259. [Google Scholar] [CrossRef]
- Molander, G.A.; Wisniewski, S.R. Stereospecific cross-coupling of secondary organotrifluoroborates: Potassium 1-(benzyloxy)alkyltrifluoroborates. J. Am. Chem. Soc. 2012, 134, 16856–16868. [Google Scholar] [CrossRef]
- González-Bobes, F.; Fu, G.C. Amino alcohols as ligands for nickel-catalyzed Suzuki reactions of unactivated alkyl halides, including secondary alkyl chlorides, with arylboronic acids. J. Am. Chem. Soc. 2006, 128, 5360–5361. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Wilsily, A.; Fu, G.C. Stereoconvergent amine-directed alkyl-alkyl Suzuki reactions of unactivated secondary alkyl chlorides. J. Am. Chem. Soc. 2011, 133, 8154–8157. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.-Y.; Hall, D.G. At the Forefront of the Suzuki–Miyaura Reaction: Advances in Stereoselective Cross-Couplings BT—Synthesis and Application of Organoboron Compounds; Fernández, E., Whiting, A., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 221–242. ISBN 978-3-319-13054-5. [Google Scholar]
- Lennox, A.J.J.; Lloyd-Jones, G.C. Selection of boron reagents for Suzuki–Miyaura coupling. Chem. Soc. Rev. 2014, 43, 412–443. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhang, S.; Li, P. Boron-selective reactions as powerful tools for modular synthesis of diverse complex molecules. Chem. Soc. Rev. 2015, 44, 8848–8858. [Google Scholar] [CrossRef]
- Suzuki, A. Organoboranes in organic syntheses including Suzuki coupling reaction. Heterocycles 2010, 80, 15–43. [Google Scholar] [CrossRef]
- Nave, S.; Sonawane, R.P.; Elford, T.G.; Aggarwal, V.K. Protodeboronation of tertiary boronic esters: Asymmetric synthesis of tertiary alkyl stereogenic centers. J. Am. Chem. Soc. 2010, 132, 17096–17098. [Google Scholar] [CrossRef]
- St. Denis, J.D.; Scully, C.C.G.; Lee, C.F.; Yudin, A.K. Development of the Direct Suzuki–Miyaura Cross-Coupling of Primary B -Alkyl MIDA-boronates and Aryl Bromides. Org. Lett. 2014, 16, 1338–1341. [Google Scholar] [CrossRef]
- Molander, G.A.; Yun, C.S.; Ribagorda, M.; Biolatto, B. B-alkyl suzuki-miyaura cross-coupling reactions with air-stable potassium alkyltrifluoroborates. J. Org. Chem. 2003, 68, 5534–5539. [Google Scholar] [CrossRef]
- Choi, J.; Fu, G.C. Transition metal-catalyzed alkyl-alkyl bond formation: Another dimension in cross-coupling chemistry. Science 2017, 356, 1. [Google Scholar] [CrossRef]
- Crudden, C.M.; Glasspoole, B.W.; Lata, C.J. Expanding the scope of transformations of organoboron species: Carbon–carbon bond formation with retention of configuration. Chem. Commun. 2009, 44, 6704–6716. [Google Scholar] [CrossRef]
- Ohmura, T.; Awano, T.; Suginome, M. Stereospecific Suzuki-Miyaura coupling of chiral α-(Acylamino) benzylboronic esters with inversion of configuration. J. Am. Chem. Soc. 2010, 132, 13191–13193. [Google Scholar] [CrossRef] [PubMed]
- Buchspies, J.; Szostak, M. Recent advances in acyl suzuki cross-coupling. Catalysts 2019, 9, 53. [Google Scholar] [CrossRef]
- Blangetti, M.; Rosso, H.; Prandi, C.; Deagostino, A.; Venturello, P. Suzuki-miyaura cross-coupling in acylation reactions, scope and recent developments. Molecules 2013, 18, 1188–1213. [Google Scholar] [CrossRef]
- Cheng, H.G.; Chen, H.; Liu, Y.; Zhou, Q. The Liebeskind–Srogl Cross-Coupling Reaction and its Synthetic Applications. Asian J. Org. Chem. 2018, 7, 490–508. [Google Scholar] [CrossRef]
- Takise, R.; Muto, K.; Yamaguchi, J. Cross-coupling of aromatic esters and amides. Chem. Soc. Rev. 2017, 46, 5864–5888. [Google Scholar] [CrossRef]
- Guo, L.; Rueping, M. Transition-Metal-Catalyzed Decarbonylative Coupling Reactions: Concepts, Classifications, and Applications. Chem. Eur. J. 2018, 24, 7794–7809. [Google Scholar] [CrossRef]
- Roy, D.; Uozumi, Y. Recent Advances in Palladium-Catalyzed Cross-Coupling Reactions at ppm to ppb Molar Catalyst Loadings. Adv. Synth. Catal. 2018, 360, 602–625. [Google Scholar] [CrossRef]
- Percec, V.; Bae, J.-Y.; Hill, D.H. Aryl Mesylates in Metal Catalyzed Homocoupling and Cross-Coupling Reactions. 2. Suzuki-Type Nickel-Catalyzed Cross-Coupling of Aryl Arenesulfonates and Aryl Mesylates with Arylboronic Acids. J. Org. Chem. 1995, 60, 1060–1065. [Google Scholar] [CrossRef]
- Han, F.-S. Transition-metal-catalyzed Suzuki–Miyaura cross-coupling reactions: A remarkable advance from palladium to nickel catalysts. Chem. Soc. Rev. 2013, 42, 5270–5298. [Google Scholar] [CrossRef]
- Dong, L.; Wen, J.; Qin, S.; Yang, N.; Yang, H.; Su, Z.; Yu, X.; Hu, C. Iron-Catalyzed Direct Suzuki–Miyaura Reaction: Theoretical and Experimental Studies on the Mechanism and the Regioselectivity. ACS Catal. 2012, 2, 1829–1837. [Google Scholar] [CrossRef]
- Hatakeyama, T.; Hashimoto, T.; Kathriarachchi, K.K.A.D.S.; Zenmyo, T.; Seike, H.; Nakamura, M. Iron-catalyzed alkyl-alkyl Suzuki-Miyaura coupling. Angew. Chem. Int. Ed. 2012, 51, 8834–8837. [Google Scholar] [CrossRef] [PubMed]
- Ansari, R.M.; Bhat, B.R. Schiff base transition metal complexes for Suzuki–Miyaura cross-coupling reaction. J. Chem. Sci. 2017, 129, 1483–1490. [Google Scholar] [CrossRef]
- Miyaura, N.; Yamada, K.; Suzuki, A. A new stereospecific cross-coupling by the palladium-catalyzed reaction of 1-alkenylboranes with 1- alkenyl or 1-alkynyl halides. Tetrahedron Lett. 1979, 20, 3437–3440. [Google Scholar] [CrossRef]
- De Meijere, A.; Brase, S.; Oestreich, M. Metal-Catalyzed Cross-Coupling Reactions and More. In Metal-Catalyzed Cross-Coupling Reactions and More; Wiley: New York, NY, USA, 2014. [Google Scholar]
- Xu, Z.-Y.; Yu, H.-Z.; Fu, Y. Mechanism of Nickel-Catalyzed Suzuki–Miyaura Coupling of Amides. Chem. Asian J. 2017, 12, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Phan, N.T.S.; Van Der Sluys, M.; Jones, C.W. On the nature of the active species in palladium catalyzed Mizoroki-Heck and Suzuki-Miyaura couplings—Homogeneous or heterogeneous catalysis, a critical review. Adv. Synth. Catal. 2006, 348, 609–679. [Google Scholar] [CrossRef]
- Ortuño, M.A.; Lledós, A.; Maseras, F.; Ujaque, G. The transmetalation process in Suzuki-Miyaura reactions: Calculations indicate lower barrier via boronate intermediate. ChemCatChem 2014, 6, 3132–3138. [Google Scholar] [CrossRef]
- Lennox, A.J.J.; Lloyd-Jones, G.C. Transmetalation in the Suzuki-Miyaura coupling: The fork in the trail. Angew. Chem. Int. Ed. 2013, 52, 7362–7370. [Google Scholar] [CrossRef]
- Yunker, L.P.E.; Ahmadi, Z.; Logan, J.R.; Wu, W.; Li, T.; Martindale, A.; Oliver, A.G.; McIndoe, J.S. Real-Time Mass Spectrometric Investigations into the Mechanism of the Suzuki-Miyaura Reaction. Organometallics 2018, 37, 4297–4308. [Google Scholar] [CrossRef]
- Aliprantis, A.O.; Canary, J.W. Observation of Catalytic Intermediates in the Suzuki Reaction by Electrospray Mass Spectrometry. J. Am. Chem. Soc. 1994, 116, 6985–6986. [Google Scholar] [CrossRef]
- Nunes, C.M.; Monteiro, A.L. Pd-Catalyzed Suzuki Cross-Coupling Reaction of Bromostilbene: Insights on the Nature of the Boron Species. J. Braz. Chem. Soc. 2007, 18, 1443–1447. [Google Scholar] [CrossRef]
- Braga, A.A.C.; Ujaque, G.; Maseras, F. A DFT study of the full catalytic cycle of the Suzuki-Miyaura cross-coupling on a model system. Organometallics 2006, 25, 3647–3658. [Google Scholar] [CrossRef]
- Suzuki, A. Cross-coupling reactions via organoboranes. J. Organomet. Chem. 2002, 653, 83–90. [Google Scholar] [CrossRef]
- Chatterjee, A.; Ward, T.R. Recent Advances in the Palladium Catalyzed Suzuki-Miyaura Cross-Coupling Reaction in Water. Catal. Lett. 2016, 146, 820–840. [Google Scholar] [CrossRef]
- Lu, G.P.; Voigtritter, K.R.; Cai, C.; Lipshutz, B.H. Ligand effects on the stereochemical outcome of suzuki-miyaura couplings. J. Org. Chem. 2012, 77, 3700–3703. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Chen, D.; Tang, W. Addressing the Challenges in Suzuki-Miyaura Cross-Couplings by Ligand Design. Synlett 2016, 27, 2183–2200. [Google Scholar]
- Liu, C.; Li, Y.; Li, Y.; Yang, C.; Wu, H.; Qin, J.; Cao, Y. Efficient Solution-Processed Deep-Blue Organic Light-Emitting Diodes Based on Multibranched Oligofluorenes with a Phosphine Oxide Center. Chem. Mater. 2013, 25, 3320–3327. [Google Scholar] [CrossRef]
- Nishimura, H.; Ishida, N.; Shimazaki, A.; Wakamiya, A.; Saeki, A.; Scott, L.T.; Murata, Y. Hole-Transporting Materials with a Two-Dimensionally Expanded py-System around an Azulene Core for Efficient Perovskite Solar Cells. J. Am. Chem. Soc. 2015, 137, 15656–15659. [Google Scholar] [CrossRef]
- Magano, J.; Dunetz, J.R. Large-scale applications of transition metal-catalyzed couplings for the synthesis of pharmaceuticals. Chem. Rev. 2011, 111, 2177–2250. [Google Scholar] [CrossRef]
- Miyaura, N.; Ishiyama, T.; Ishikawa, M.; Suzuki, A. Palladium-catalyzed cross-coupling reactions of B-alkyl-9-BBN or trialkylboranes with aryl and 1-alkenyl halides. Tetrahedron Lett. 1986, 27, 6369–6372. [Google Scholar] [CrossRef]
- Sato, M.; Miyaura, N.; Suzuki, A. Cross-coupling reaction of alkyl- or arylboronic acid esters with organic halides induced by thallium(I) salts and palladium-catalyst. Chem. Lett. 1989, 8, 1405–1408. [Google Scholar] [CrossRef]
- Saito, B.; Fu, G.C. Alkyl-alkyl Suzuki cross-couplings of unactivated secondary alkyl halides at room temperature. J. Am. Chem. Soc. 2007, 129, 9602–9603. [Google Scholar] [CrossRef] [PubMed]
- Molander, G.A.; Canturk, B. Organotrifluoroborates and monocoordinated palladium complexes as catalysts - A perfect combination for Suzuki-Miyaura coupling. Angew. Chem. Int. Ed. 2009, 48, 9240–9261. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.D.; Barder, T.E.; Martinelli, J.R.; Buchwald, S.L. A rationally designed universal catalyst for Suzuki-Miyaura coupling processes. Angew. Chem. Int. Ed. 2004, 43, 1871–1876. [Google Scholar] [CrossRef] [PubMed]
- Chuang, D.W.; El-Shazly, M.; Chen, C.C.; Chung, Y.M.; D. Barve, B.; Wu, M.J.; Chang, F.R.; Wu, Y.C. Synthesis of 1,5-diphenylpent-3-en-1-yne derivatives utilizing an aqueous B-alkyl Suzuki cross coupling reaction. Tetrahedron Lett. 2013, 54, 5162–5166. [Google Scholar] [CrossRef]
- Yu, D.G.; Li, B.J.; Shi, Z.J. Exploration of new C-O electrophiles in cross-coupling reactions. Acc. Chem. Res. 2010, 43, 1486–1495. [Google Scholar] [CrossRef]
- Tobisu, M.; Chatani, N. Cross-Couplings Using Aryl Ethers via C-O Bond Activation Enabled by Nickel Catalysts. Acc. Chem. Res. 2015, 48, 1717–1726. [Google Scholar] [CrossRef]
- Cornella, J.; Zarate, C.; Martin, R. Metal-catalyzed activation of ethers via C–O bond cleavage: A new strategy for molecular diversity. Chem. Soc. Rev. 2014, 43, 8081–8097. [Google Scholar] [CrossRef]
- Guo, L.; Hsiao, C.C.; Yue, H.; Liu, X.; Rueping, M. Nickel-Catalyzed Csp2-Csp3 Cross-Coupling via C-O Bond Activation. ACS Catal. 2016, 6, 4438–4442. [Google Scholar] [CrossRef]
- Guo, L.; Liu, X.; Baumann, C.; Rueping, M. Nickel-Catalyzed Alkoxy–Alkyl Interconversion with Alkylborane Reagents through C−O Bond Activation of Aryl and Enol Ethers. Angew. Chem. Int. Ed. 2016, 55, 15415–15419. [Google Scholar] [CrossRef]
- Zhang, X.M.; Yang, J.; Zhuang, Q.B.; Tu, Y.Q.; Chen, Z.; Shao, H.; Wang, S.H.; Zhang, F.M. Allylic Arylation of 1,3-Dienes via Hydroboration/Migrative Suzuki-Miyaura Cross-Coupling Reactions. ACS Catal. 2018, 8, 6094–6099. [Google Scholar] [CrossRef]
- Kim, D.E.; Zhu, Y.; Newhouse, T.R. Vinylogous acyl triflates as an entry point to α,β-disubstituted cyclic enones via Suzuki-Miyaura cross-coupling. Org. Biomol. Chem. 2019, 17, 1796–1799. [Google Scholar] [CrossRef] [PubMed]
- Mikagi, A.; Tokairin, D.; Usuki, T. Suzuki-Miyaura cross-coupling reaction of monohalopyridines and L-aspartic acid derivative. Tetrahedron Lett. 2018, 59, 4602–4605. [Google Scholar] [CrossRef]
- Liu, X.; Hsiao, C.C.; Guo, L.; Rueping, M. Cross-Coupling of Amides with Alkylboranes via Nickel-Catalyzed C-N Bond Cleavage. Org. Lett. 2018, 20, 2976–2979. [Google Scholar] [CrossRef] [PubMed]
- Chatupheeraphat, A.; Liao, H.H.; Srimontree, W.; Guo, L.; Minenkov, Y.; Poater, A.; Cavallo, L.; Rueping, M. Ligand-Controlled Chemoselective C(acyl)-O Bond vs C(aryl)-C Bond Activation of Aromatic Esters in Nickel Catalyzed C(sp2)-C(sp3) Cross-Couplings. J. Am. Chem. Soc. 2018, 140, 3724–3735. [Google Scholar] [CrossRef] [PubMed]
- Okuda, Y.; Xu, J.; Ishida, T.; Wang, C.A.; Nishihara, Y. Nickel-Catalyzed Decarbonylative Alkylation of Aroyl Fluorides Assisted by Lewis-Acidic Organoboranes. ACS Omega 2018, 3, 13129–13140. [Google Scholar] [CrossRef] [PubMed]
- Vedejs, E.; Chapman, R.; Fields, S.C.; Lin, S.; Schrimpf, M.R. Conversion of Arylboronic Acids into Potassium Aryltrifluoroborates: Convenient Precursors of Arylboron Difluoride Lewis Acids. J. Org. Chem. 1995, 60, 3020–3027. [Google Scholar] [CrossRef]
- Molander, G.A.; Ito, T. Cross-Coupling Reactions of Potassium Alkyltrifluoroborates with Aryl and 1-Alkenyl Trifluoromethanesulfonates. Org. Lett. 2001, 3, 393–396. [Google Scholar] [CrossRef]
- Darses, S.; Genet, J.P. Potassium organotrifluoroborates: New perspectives in organic synthesis. Chem. Rev. 2008, 108, 288–325. [Google Scholar] [CrossRef]
- Molander, G.A.; Ellis, N. Organotrifluoroborates: Protected Boronic Acids That Expand the Versatility of the Suzuki Coupling Reaction Organotrifluoroborates. Acc. Chem. Res. 2007, 40, 275–286. [Google Scholar] [CrossRef]
- Stefani, H.A.; Cella, R.; Vieira, A.S. Recent advances in organotrifluoroborates chemistry. Tetrahedron 2007, 63, 3623–3658. [Google Scholar] [CrossRef]
- Molander, G.A. Organotrifluoroborates: Another Branch of the Mighty Oak. J. Org. Chem. 2015, 80, 7837–7848. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.R.; Li, Q.; Lian, Y.; Xiao, J.; Londregan, A.T. Construction of 1-Heteroaryl-3-azabicyclo[3.1.0]hexanes by sp3-sp2 Suzuki-Miyaura and Chan-Evans-Lam Coupling Reactions of Tertiary Trifluoroborates. Org. Lett. 2017, 19, 2450–2453. [Google Scholar] [CrossRef]
- Tellis, J.C.; Amani, J.; Molander, G.A. Single-Electron Transmetalation: Photoredox/Nickel Dual Catalytic Cross-Coupling of Secondary Alkyl β-Trifluoroboratoketones and -esters with Aryl Bromides. Org. Lett. 2016, 18, 2994–2997. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Nami, R.; Tellis, J.C.; Molander, G.A. Single-Electron Transmetalation: Protecting-Group-Independent Synthesis of Secondary Benzylic Alcohol Derivatives via Photoredox/Nickel Dual Catalysis. Org. Lett. 2016, 18, 2572–2575. [Google Scholar] [CrossRef] [PubMed]
- Primer, D.N.; Molander, G.A. Enabling the Cross-Coupling of Tertiary Organoboron Nucleophiles through Radical-Mediated Alkyl Transfer. J. Am. Chem. Soc. 2017, 139, 9847–9850. [Google Scholar] [CrossRef]
- Brown, H.C.; Racherla, U.S. Organoboranes. 43. A Convenient, Highly Efficient Synthesis of Triorganylboranes via a Modified Organometallic Route. J. Org. Chem 1986, 51, 427–432. [Google Scholar] [CrossRef]
- Miyaura, N.; Ishiyama, T.; Sasaki, H.; Ishikawa, M.; Sato, M.; Suzuki, A. Palladium-Catalyzed Inter- and Intramolecular Cross-Coupling Reactions of B-Alkyl-9-Borabicyclo[3.3.1]nonane Derivatives with 1-Halo-1-alkenes or Haloarenes. Syntheses of Functionalized Alkenes, Arenes, and Cycloalkenes via a Hydroboration-Coupling Sequence. J. Am. Chem. Soc. 1989, 111, 314–321. [Google Scholar]
- Sun, H.X.; Sun, Z.H.; Wang, B. B-Alkyl Suzuki-Miyaura cross-coupling of tri-n-alkylboranes with arylbromides bearing acidic functions under mild non-aqueous conditions. Tetrahedron Lett. 2009, 50, 1596–1599. [Google Scholar] [CrossRef]
- Wang, B.; Sun, H.X.; Sun, Z.H.; Lin, G.Q. Direct B-alkyl Suzuki-Miyaura cross-coupling of trialkyl-boranes with aryl bromides in the presence of unmasked acidic or basic functions and base-labile protections under mild non-aqueous conditions. Adv. Synth. Catal. 2009, 351, 415–422. [Google Scholar] [CrossRef]
- Monot, J.; Brahmi, M.M.; Ueng, S.; Robert, C.; Murr, M.D.; Curran, D.P.; Malacria, M.; Fensterbank, L.; Lacôte, E. Suzuki—Miyaura Coupling of NHC—Boranes: A New Addition to the C-C Coupling Toolbox. Org. Lett. 2009, 11, 4914–4917. [Google Scholar] [CrossRef]
- Li, H.; Zhong, Y.L.; Chen, C.Y.; Ferraro, A.E.; Wang, D. A Concise and Atom-Economical Suzuki-Miyaura Coupling Reaction Using Unactivated Trialkyl- and Triarylboranes with Aryl Halides. Org. Lett. 2015, 17, 3616–3619. [Google Scholar] [CrossRef] [PubMed]
- Gray, M.; Andrews, I.P.; Hook, D.F.; Kitteringham, J.; Voyle, M. Practical methylation of aryl halides by Suzuki ± Miyaura coupling. Tetrahedron Lett. 2000, 41, 6237–6240. [Google Scholar] [CrossRef]
- Doucet, H. Suzuki-Miyaura cross-coupling reactions of alkylboronic acid derivatives or alkyltrifluoroborates with aryl, alkenyl or alkyl halides and triflates. Eur. J. Org. Chem. 2008, 12, 2013–2030. [Google Scholar] [CrossRef]
- Mu, Y.; Gibbs, R.A. Coupling of isoprenoid triflates with organoboron nucleophiles: Synthesis of all-trans-geranylgeraniol. Tetrahedron Lett. 1995, 36, 5669–5672. [Google Scholar] [CrossRef]
- Zou, G.; Reddy, Y.K.; Falck, J.R. Ag(I)-promoted Suzuki-Miyaura cross-couplings of n-alkylboronic acids. Tetrahedron Lett. 2001, 42, 7213–7215. [Google Scholar] [CrossRef]
- Fall, Y.; Doucet, H.; Santelli, M. Palladium-catalysed Suzuki cross-coupling of primary alkylboronic acids with alkenyl halides. Appl. Organomet. Chem. 2008, 22, 503–509. [Google Scholar] [CrossRef]
- Guo, B.; Fu, C.; Ma, S. Application of LB-Phos·HBF4 in the Suzuki Coupling Reaction of 2-Bromoalken-3-ols with Alkylboronic Acids. Eur. J. Org. Chem. 2012, 2012, 4034–4041. [Google Scholar] [CrossRef]
- Li, C.; Xiao, G.; Zhao, Q.; Liu, H.; Wang, T.; Tang, W. Sterically demanding aryl–alkyl Suzuki–Miyaura coupling. Org. Chem. Front. 2014, 1, 225–229. [Google Scholar] [CrossRef]
- Si, T.; Li, B.; Xiong, W.; Xu, B.; Tang, W. Efficient cross-coupling of aryl/alkenyl triflates with acyclic secondary alkylboronic acids. Org. Biomol. Chem. 2017, 15, 9903–9909. [Google Scholar] [CrossRef]
- Kakiuchi, F.; Usui, M.; Ueno, S.; Chatani, N.; Murai, S. Ruthenium-Catalyzed Functionalization of Aryl Carbon-Oxygen Bonds in Aromatic Ethers with Organoboron Compounds. J. Am. Chem. Soc. 2004, 126, 2706–2707. [Google Scholar] [CrossRef]
- Murai, S.; Kakiuchi, F.; Sekine, S.; Tanaka, Y.; Kamatani, A.; Sonoda, M.; Chatani, N. Efficient catalytic addition of aromatic carbon-hydrogen bonds to olefins. Nature 1993, 366, 529–531. [Google Scholar] [CrossRef]
- Ueno, S.; Mizushima, E.; Chatani, N.; Kakiuchi, F. Direct observation of the oxidative addition of the aryl carbon-oxygen bond to a ruthenium complex and consideration of the relative reactivity between aryl carbon-oxygen and aryl carbon-hydrogen bonds. J. Am. Chem. Soc. 2006, 128, 16516–16517. [Google Scholar] [CrossRef] [PubMed]
- Tobisu, M.; Shimasaki, T.; Chatani, N. Nickel-catalyzed cross-coupling of aryl methyl ethers with aryl boronic esters. Angew. Chem. Int. Ed. 2008, 120, 4944–4947. [Google Scholar] [CrossRef]
- Mancilla, T.; Contreras, R.; Wrackmeyer, B. New bicyclic organylboronic esters derived from iminodiacetic acids. J. Organomet. Chem. 1986, 307, 1–6. [Google Scholar] [CrossRef]
- Li, J.; Grillo, A.S.; Burke, M.D. From Synthesis to Function via Iterative Assembly of N-Methyliminodiacetic Acid Boronate Building Blocks. Acc. Chem. Res. 2015, 48, 2297–2307. [Google Scholar] [CrossRef]
- St. Denis, J.D.; He, Z.; Yudin, A.K. Amphoteric α-Boryl Aldehyde Linchpins in the Synthesis of Heterocycles. ACS Catal. 2015, 5, 5373–5379. [Google Scholar] [CrossRef]
- Duncton, M.A.J.; Singh, R. Synthesis of trans -2-(Trifluoromethyl)cyclopropanes via Suzuki Reactions with an N -Methyliminodiacetic Acid Boronate. Org. Lett. 2013, 15, 4284–4287. [Google Scholar] [CrossRef]
- Seidel, G.; Fürstner, A. Suzuki reactions of extended scope: The ‘9-MeO-9-BBN variant’ as a complementary format for cross-coupling. Chem. Commun. 2012, 48, 2055–2070. [Google Scholar] [CrossRef]
- Ye, N.; Dai, W.M. An Efficient and Reliable Catalyst System Using Hemilabile Aphos for B-Alkyl Suzuki-Miyaura Cross-Coupling Reaction with Alkenyl Halides. Eur. J. Org. Chem. 2013, 5, 831–835. [Google Scholar] [CrossRef]
- Lee, N.R.; Linstadt, R.T.H.; Gloisten, D.J.; Gallou, F.; Lipshutz, B.H. B-Alkyl sp3-sp2 Suzuki-Miyaura Couplings under Mild Aqueous Micellar Conditions. Org. Lett. 2018, 20, 2902–2905. [Google Scholar] [CrossRef]
- Heravi, M.M.; Hashemi, E. Recent applications of the Suzuki reaction in total synthesis. Tetrahedron 2012, 68, 9145–9178. [Google Scholar] [CrossRef]
- Taheri Kal Koshvandi, A.; Heravi, M.M.; Momeni, T. Current Applications of Suzuki-Miyaura Coupling Reaction in The Total Synthesis of Natural Products: An update. Appl. Organomet. Chem. 2018, 32, e4210. [Google Scholar] [CrossRef]
- Wu, Y.D.; Lai, Y.; Dai, W.M. Synthesis of Two Diastereomeric C1–C7 Acid Fragments of Amphidinolactone B Using B-Alkyl Suzuki–Miyaura Cross-Coupling as the Modular Assembly Step. ChemistrySelect 2016, 1, 1022–1027. [Google Scholar] [CrossRef]
- Koch, S.; Schollmeyer, D.; Löwe, H.; Kunz, H. C-Glycosyl amino acids through hydroboration-cross-coupling of exo-glycals and their application in automated solid-phase synthesis. Chem. Eur. J. 2013, 19, 7020–7041. [Google Scholar] [CrossRef]
- Hirai, S.; Utsugi, M.; Iwamoto, M.; Nakada, M. Formal total synthesis of (-)-taxol through Pd-catalyzed eight-membered carbocyclic ring formation. Chem. Eur. J. 2015, 21, 355–359. [Google Scholar] [CrossRef]
- Han, W.; Wu, J. Synthesis of C14–C21 acid fragments of cytochalasin Z 8 via anti -selective aldol condensation and B -alkyl Suzuki–Miyaura cross-coupling. RSC Adv. 2018, 8, 3899–3902. [Google Scholar] [CrossRef]
- Tungen, J.E.; Aursnes, M.; Hansen, T.V. Stereoselective total synthesis of ieodomycin C. Tetrahedron 2014, 70, 3793–3797. [Google Scholar] [CrossRef]
- Tungen, J.E.; Aursnes, M.; Vik, A. Synthesis of Ieodomycin D. Synlett 2016, 27, 2497–2499. [Google Scholar]
- Xu, G.; Fu, W.; Liu, G.; Senanayake, C.H.; Tang, W. Efficient syntheses of korupensamines A, B and michellamine B by asymmetric Suzuki-Miyaura coupling reactions. J. Am. Chem. Soc. 2014, 136, 570–573. [Google Scholar] [CrossRef]
- Yalcouye, B.; Choppin, S.; Panossian, A.; Leroux, F.R.; Colobert, F. A concise atroposelective formal synthesis of (-)-steganone. Eur. J. Org. Chem. 2014, 2014, 6285–6294. [Google Scholar] [CrossRef]







| Boron Reagent | Substrate | Reaction Conditions (General) | Reference | Section and Scheme |
|---|---|---|---|---|
| B-alkyl-9-BBN and trialkylboranes | Aryl iodides | PdCl2(dppf), NaOH, THF, reflux, 16 h | 76 | 3;3A |
| Alkylboranes | Aryl bromides and iodides | PdCl2(dppf), NaOH, THF, 65 °C | 77–79 | 3;3B |
| B-alkyl-9-BBN and boronic acids | Aryl halides | Pd(OAc)2, SPhos, K3PO4.H2O, THF or toluene | 80 | 3;3C |
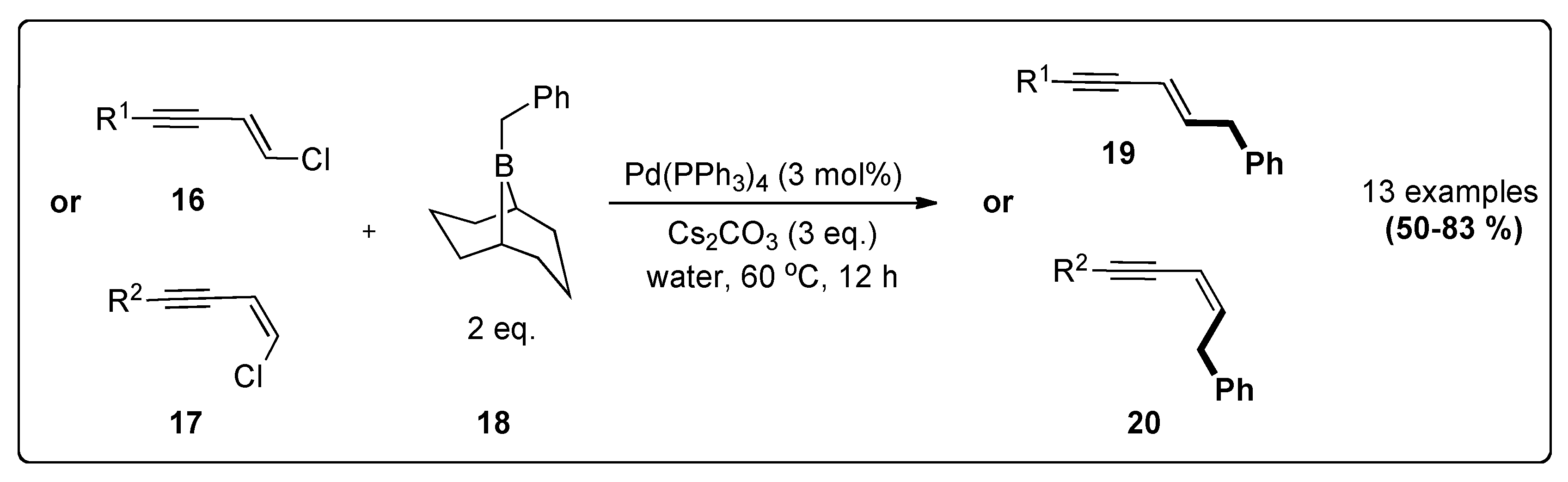
| B-benzyl-9-BBN | Chloroenynes | Pd(PPh3)4, Cs2CO3, water, 60 °C, 12 h | 81 | 3;4 |
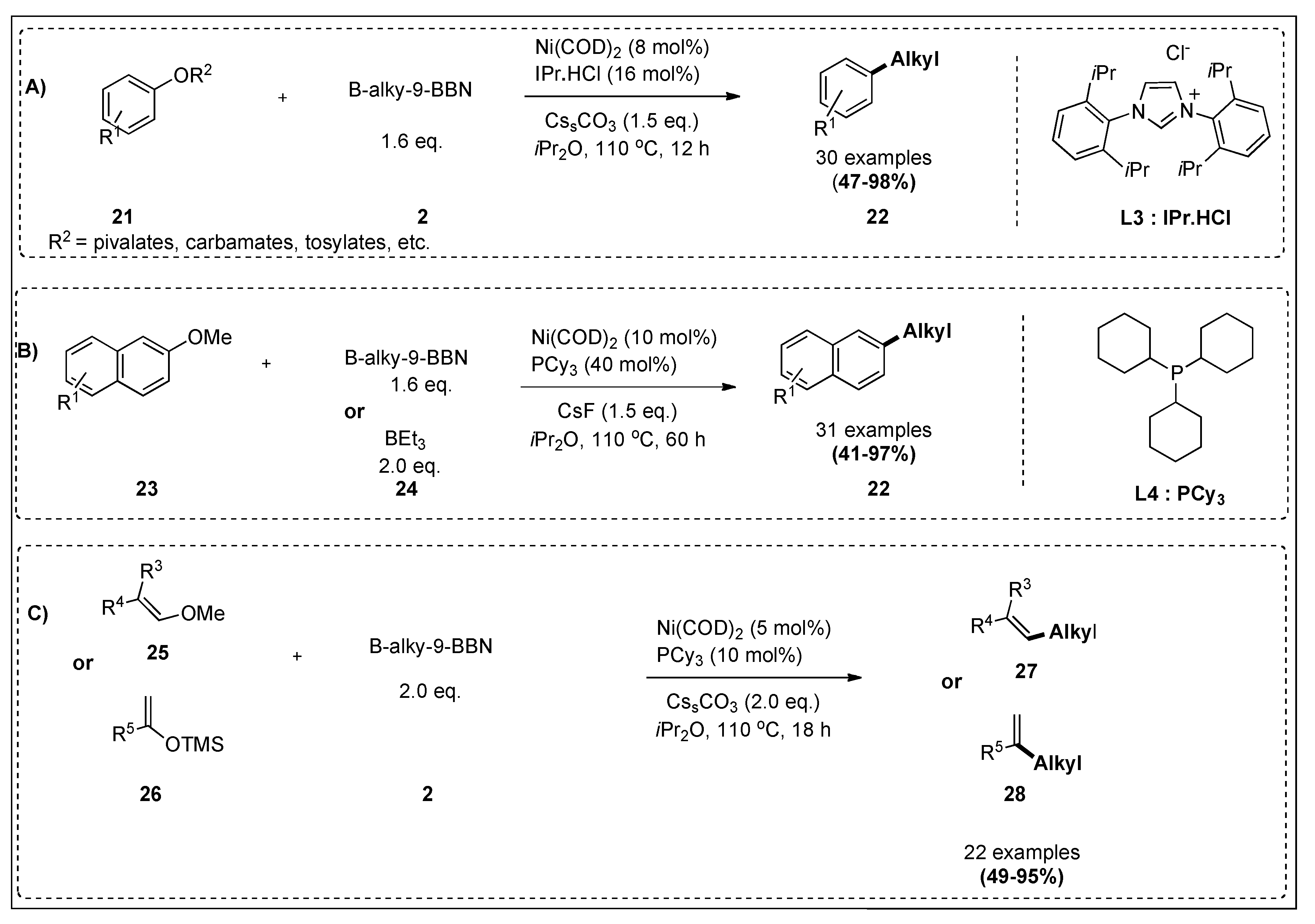
| B-alkyl-9-BBN | CAr-O electrophiles | Ni(COD)2, IPr.HCl, CssCO3, iPr2O, 110 °C, 12 h | 85 | 3;5A |
| B-alkyl-9-BBN | Aromatic and alkenyl ethers | Ni(COD)2, PCy3, base, iPr2O, 110 °C | 86 | 3;5B,C |
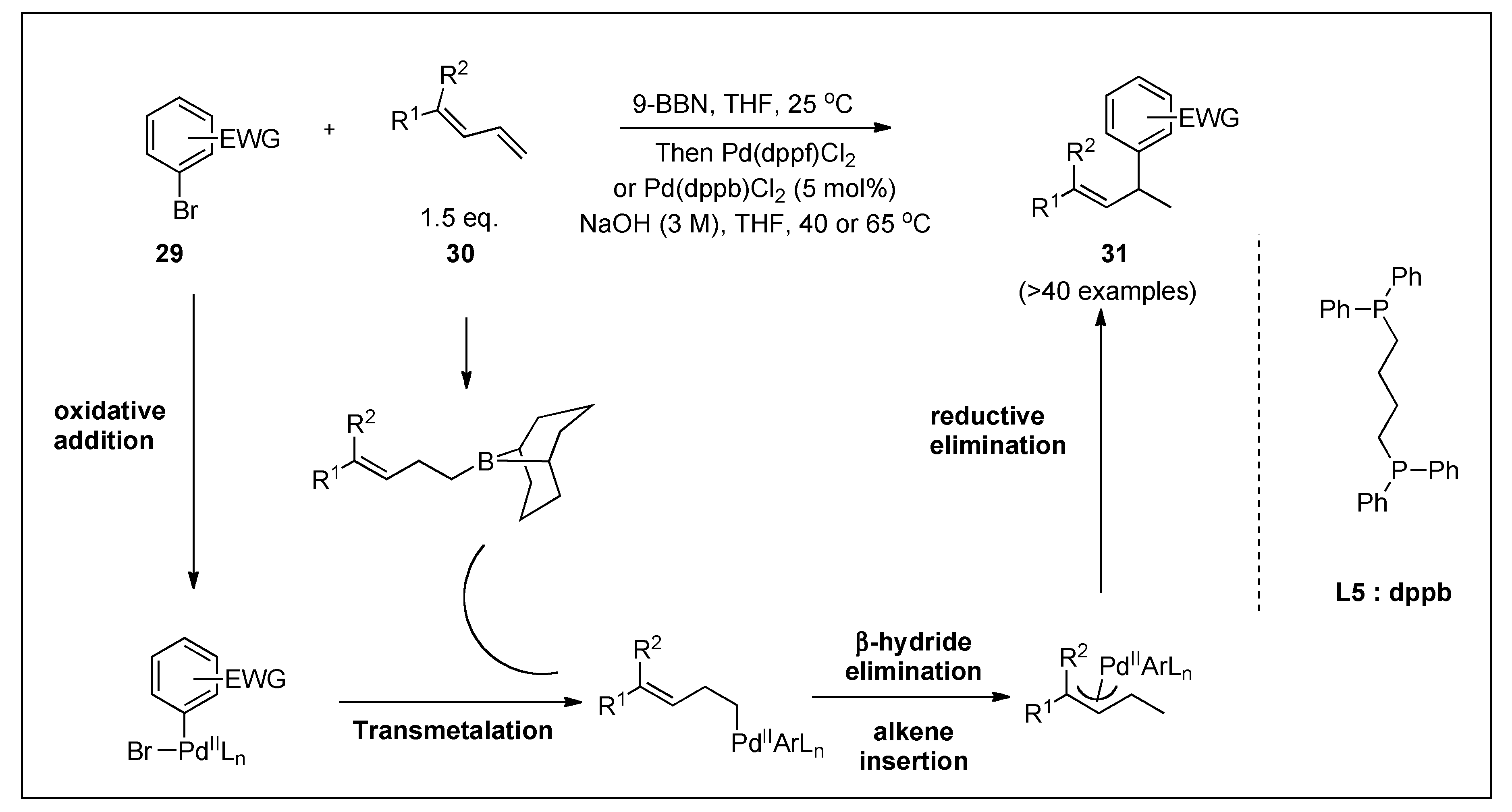
| 1,3-dienes and 9-BBN | Aryl halides | Pd(dppf)Cl2 or Pd(dppb)Cl2, NaOH, THF, 40 or 65 °C | 87 | 3;6 |
| B-alkyl-9-BBN | β-triflyl enones | Pd(dppf)Cl2, Cs2CO3, DMF:THF:H2O, 60 °C, 16 h | 88 | 3;7A |
| 9-BBN derivatives of L-aspartic acid | Halogenated pyridine | Pd(PPh3)4, K3PO4 (aq.), THF, 50 °C, 2 h | 89 | 3;7B |
| Alkyl organoboron reagents | Aromatic esters | Ni(COD)2, dcype, CsF, toluene, 150 °C | 91 | 3; 8A |
| Alkyl organoboron reagents | Aroyl fluorides | Ni(COD)2, dppe, CsF, toluene/hexane, 140 °C | 92 | 3;8B |
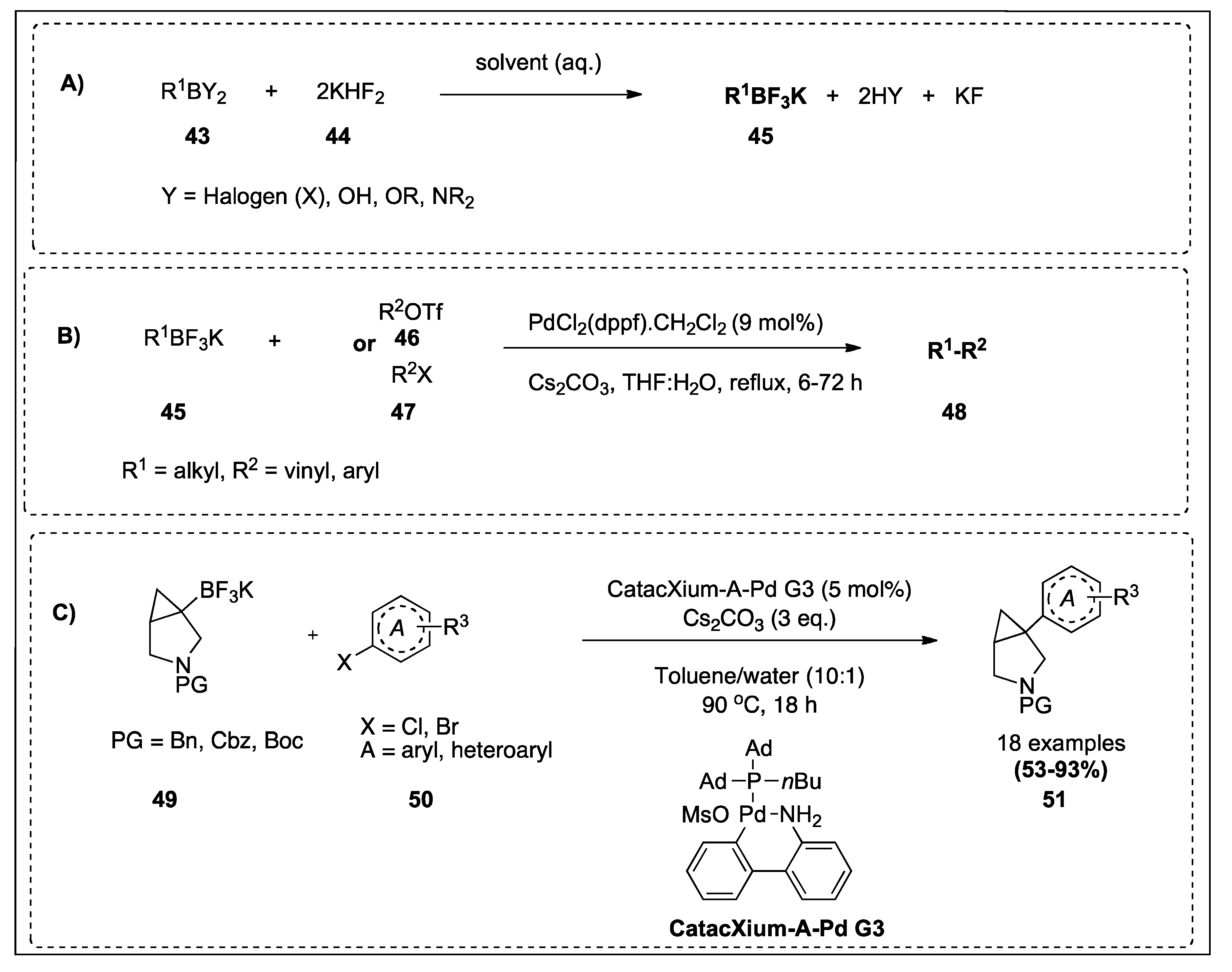
| Potassium alkyltrifluoroborates | Aryl halides/triflates and vinyl triflates | PdCl2(dppf).CH2Cl, Cs2CO3, THF:H2O, reflux, 6-72 h | 44,94 | 4;9B |
| Tertiary trifluoroborate salts | Aryl and heteroaryl chlorides and bromides | CatacXium-A-Pd G3, Cs2CO3, tol/water, 90 °C, 18 h | 99 | 4;9C |
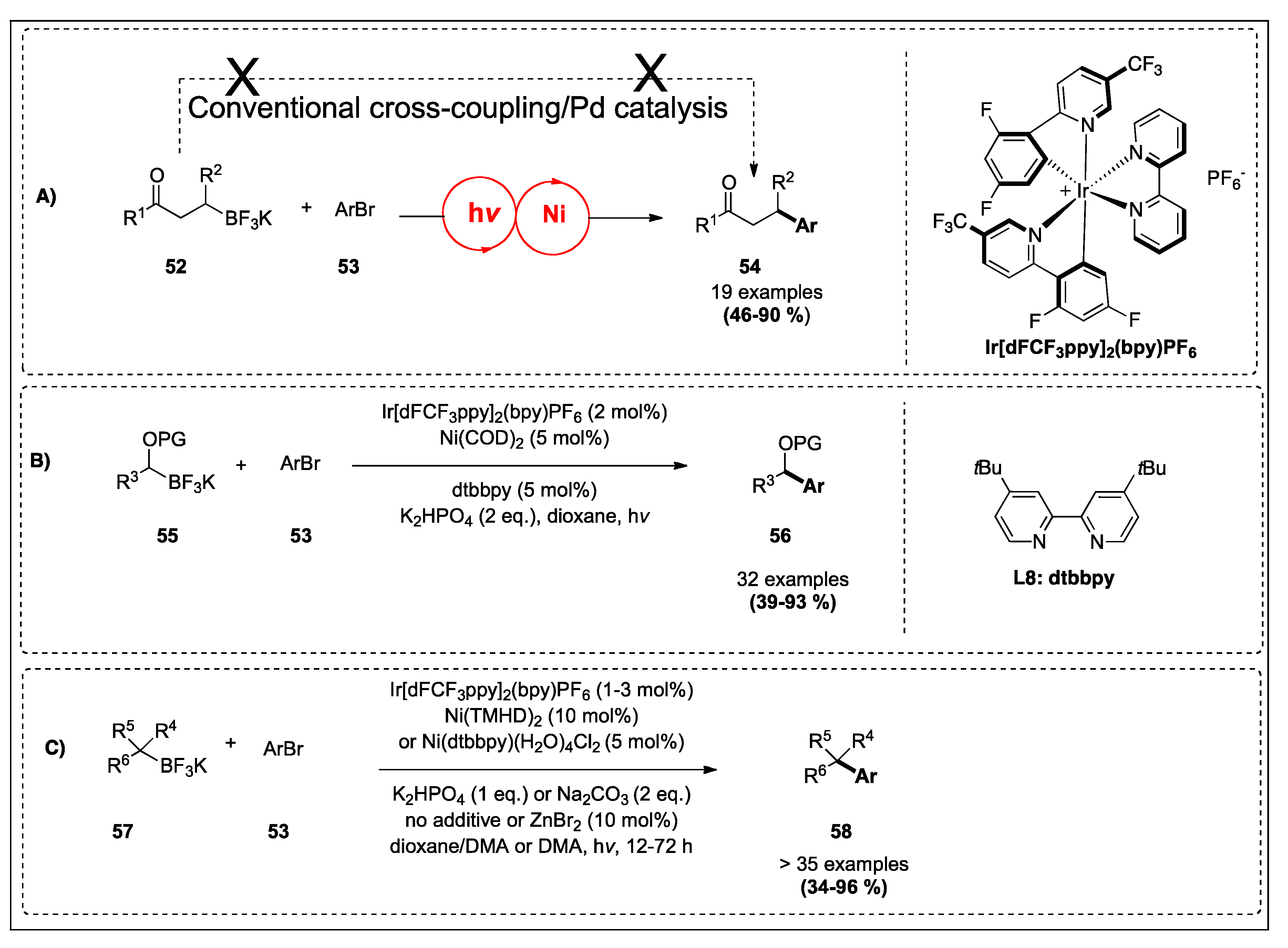
| Secondary alkyl β-trifluoroboratoketones and -esters | Aryl Bromides | Ir[dFCF3ppy]2(bpy)PF6, NiCl2·dme, dtbbpy, Cs2CO3, 2,6-lutidine, 1,4-dioxane, hv | 10 | 4;10A |
| α-alkoxyalkyl- and α-acyloxyalkyltrifluoroborates | Aryl bromides | Ir[dFCF3ppy]2(bpy)PF6, Ni(COD)2, dtbbpy, K2HPO4, dioxane, hv | 101 | 4;10B |
| Tertiary organotrifluoroborates reagents | Aryl bromides | Ir[dFCF3ppy]2(bpy)PF6, Ni(TMHD)2 or Ni(dtbbpy)(H2O)4Cl2, K2HPO4 or Na2CO3, no additive or ZnBr2, dioxane/DMA or DMA, hv, 12–72 h | 102 | 4;10C |
| Trialkylboranes | Aryl bromides | PdCl2(dppf), THF, reflux, 2–6 h | 105,106 | 5;11B |
| NHC-boranes complexes | Aryl halides and triflates | [Pd], Ligand, tol-H2O or THF-H2O, heat or microwave | 107 | 5;11C |
| Trialkyl- and triaryl-boranes (generated in situ) | Alkenyl and aryl halides | Pd(OAc)2, n-BuAd2P or RuPhos, K3PO4, tol-H2O, 100 °C | 108 | 5;11D |
| n-alkylboronic acids | Alkenyl and aryl halides or triflates | PdCl2(dppf), K2CO3, Ag2O, THF, 80 °C, 6–10 h | 112 | 6;12A |
| n-alkylboronic acids | Alkenyl halides | PdCl(C3H5)dppb, Cs2CO3, toluene or xylene, 100–130 °C, 20 h | 113 | 6;12B |
| Primary and secondary alkylboronic acids | 2-bromoalken-3-ol derivatives | Pd(OAc)2, LB-Phos.HBF4, K2CO3, toluene, 110 °C, 3–27 h | 114 | 6;12C |
| Cyclic secondary alkylboronic acids | di-ortho-substituted arylhalides | Pd(OAc)2, AntPhos, K3PO4, toluene, 110 °C, 12–24 h | 115 | 6;12D |
| Acyclic secondary alkylboronic acids | Aryl and alkenyl triflates | [Pd(cinnamyl)Cl]2, Ligand, K3PO4.H2O, toluene, 110 °C, 12 h | 116 | 6;12E |
| Boronic esters | Aryl methyl ethers bearing ortho-carbonyls | RuH2(CO)(PPh3)3, toluene, 110 °C | 117 | 7;13A |
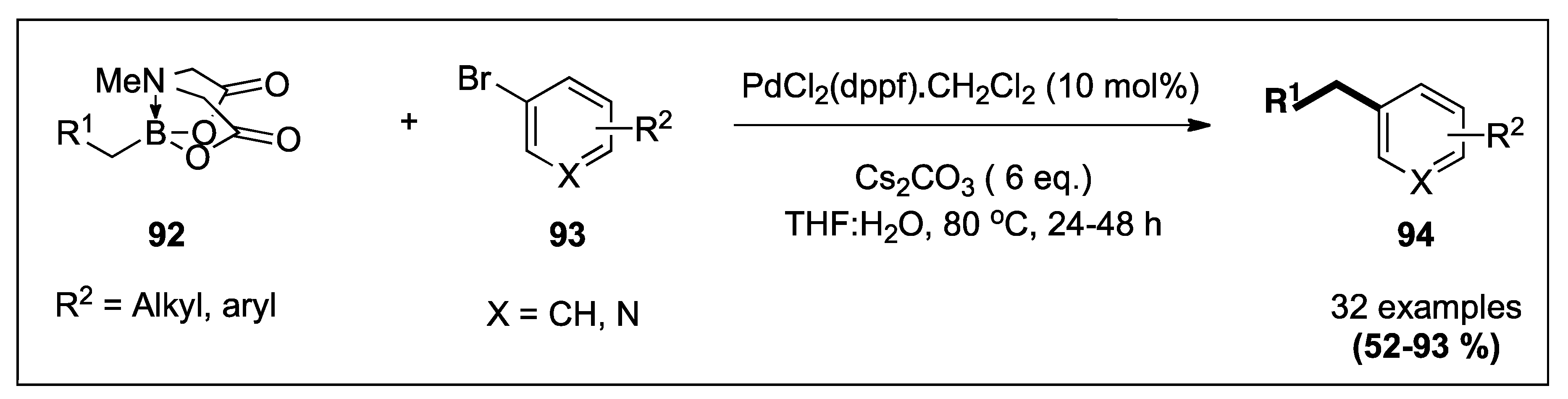
| MIDA boronates | Aryl and heteroarylbromides | PdCl2(dppf).CH2Cl2, Cs2CO3, THF:H2O, 80 °C, 24–48 h | 43 | 7;14 |
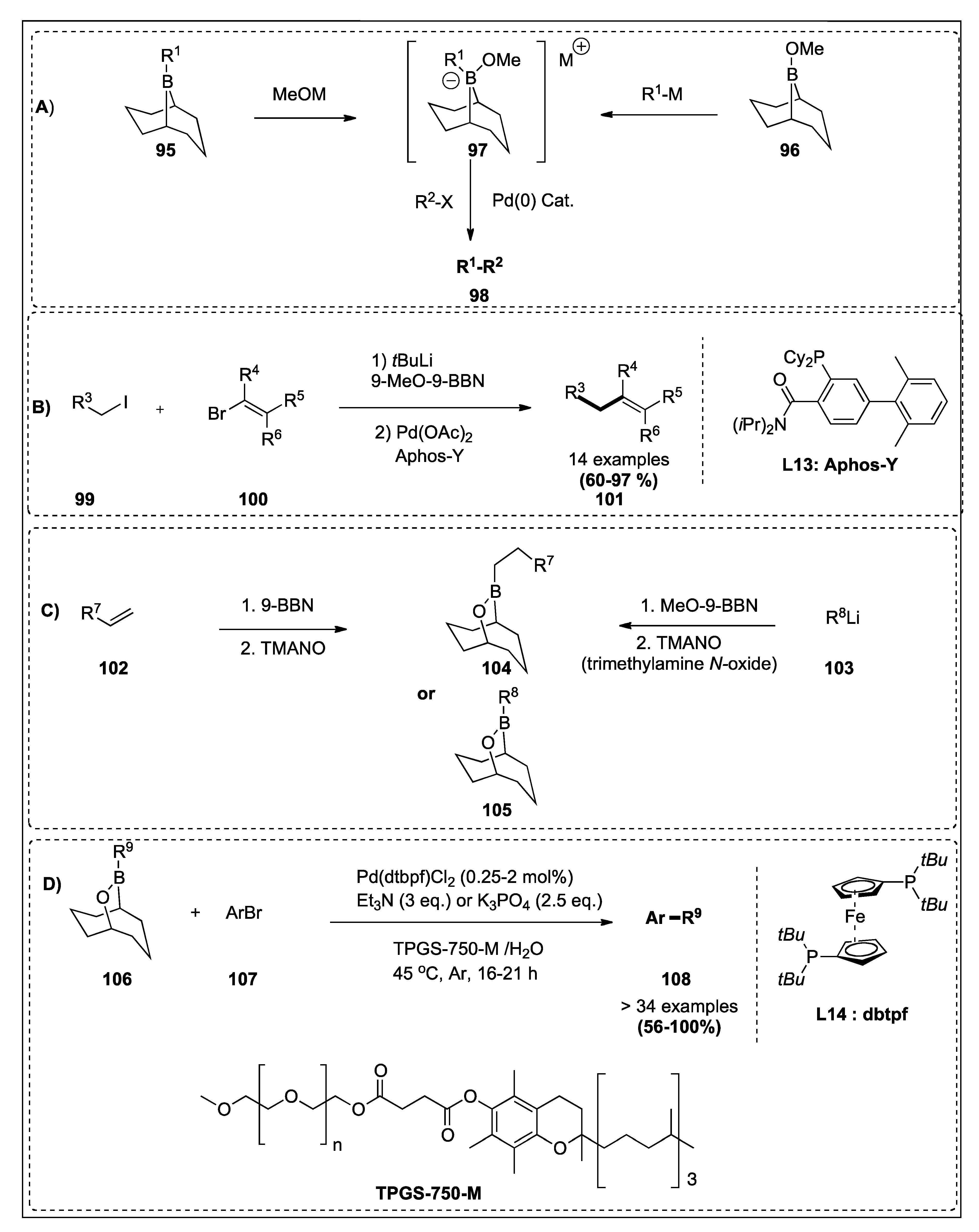
| Alkyl iodide and 9-MeO-9BBN (tBuLi for in situ generation) | Alkenyl bromide | Pd(OAc)2, Aphos-Y | 126 | 8;15B |
| OBBD derivatives | Aryl Bromide | Pd(dtbpf)Cl2, Et3N or K3PO4, TPGS-750-M/H2O 45 °C, Ar, 16–21 h | 127 | 8;15D |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Maiss, J.; Mohy El Dine, T.; Lu, C.-S.; Karamé, I.; Kanj, A.; Polychronopoulou, K.; Shaya, J. Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings. Catalysts 2020, 10, 296. https://doi.org/10.3390/catal10030296
El-Maiss J, Mohy El Dine T, Lu C-S, Karamé I, Kanj A, Polychronopoulou K, Shaya J. Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings. Catalysts. 2020; 10(3):296. https://doi.org/10.3390/catal10030296
Chicago/Turabian StyleEl-Maiss, Janwa, Tharwat Mohy El Dine, Chung-Shin Lu, Iyad Karamé, Ali Kanj, Kyriaki Polychronopoulou, and Janah Shaya. 2020. "Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings" Catalysts 10, no. 3: 296. https://doi.org/10.3390/catal10030296
APA StyleEl-Maiss, J., Mohy El Dine, T., Lu, C.-S., Karamé, I., Kanj, A., Polychronopoulou, K., & Shaya, J. (2020). Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings. Catalysts, 10(3), 296. https://doi.org/10.3390/catal10030296






