HKUST-1-Supported Cerium Catalysts for CO Oxidation
Abstract
1. Introduction
2. Results and Discussion
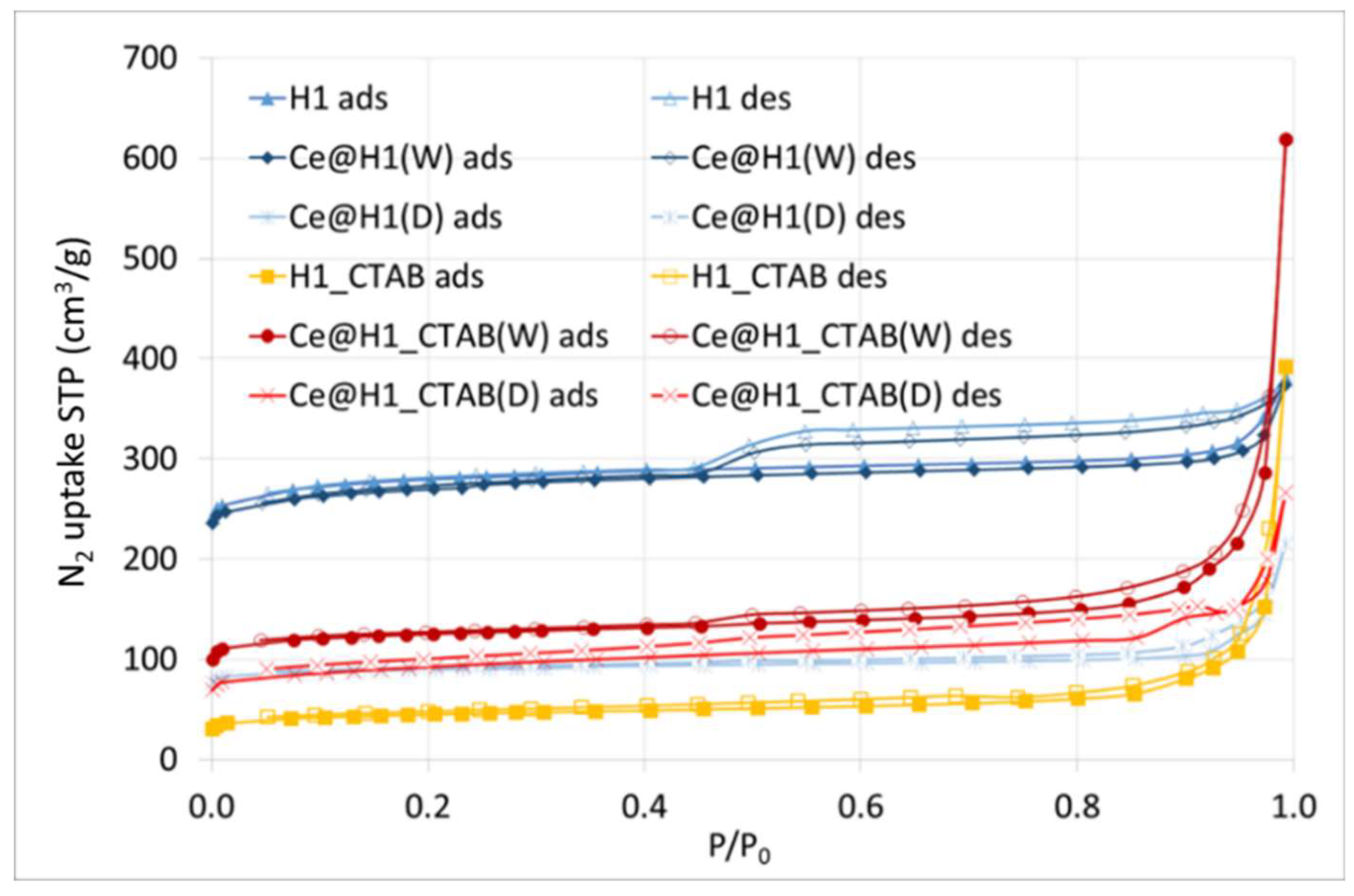
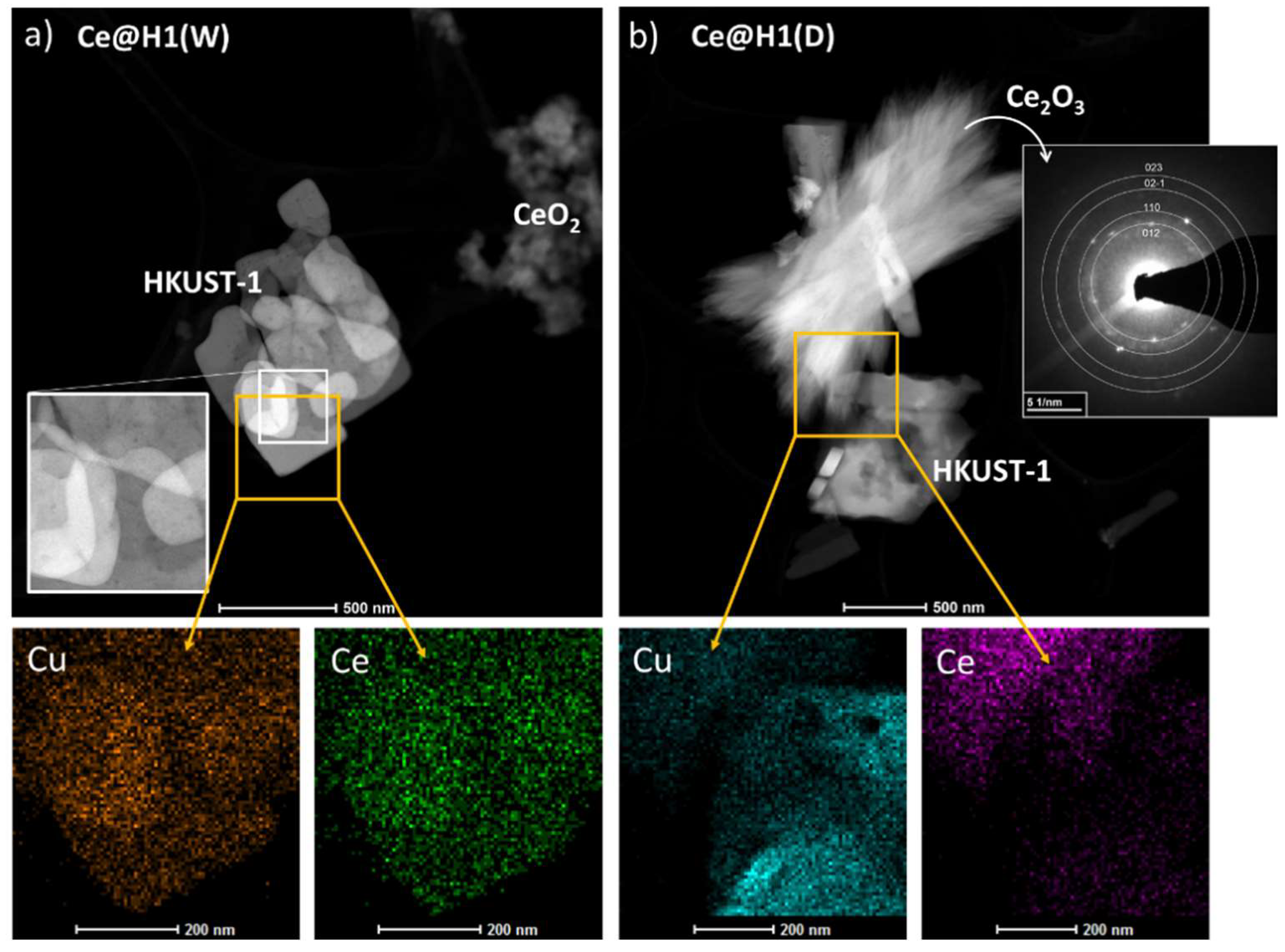
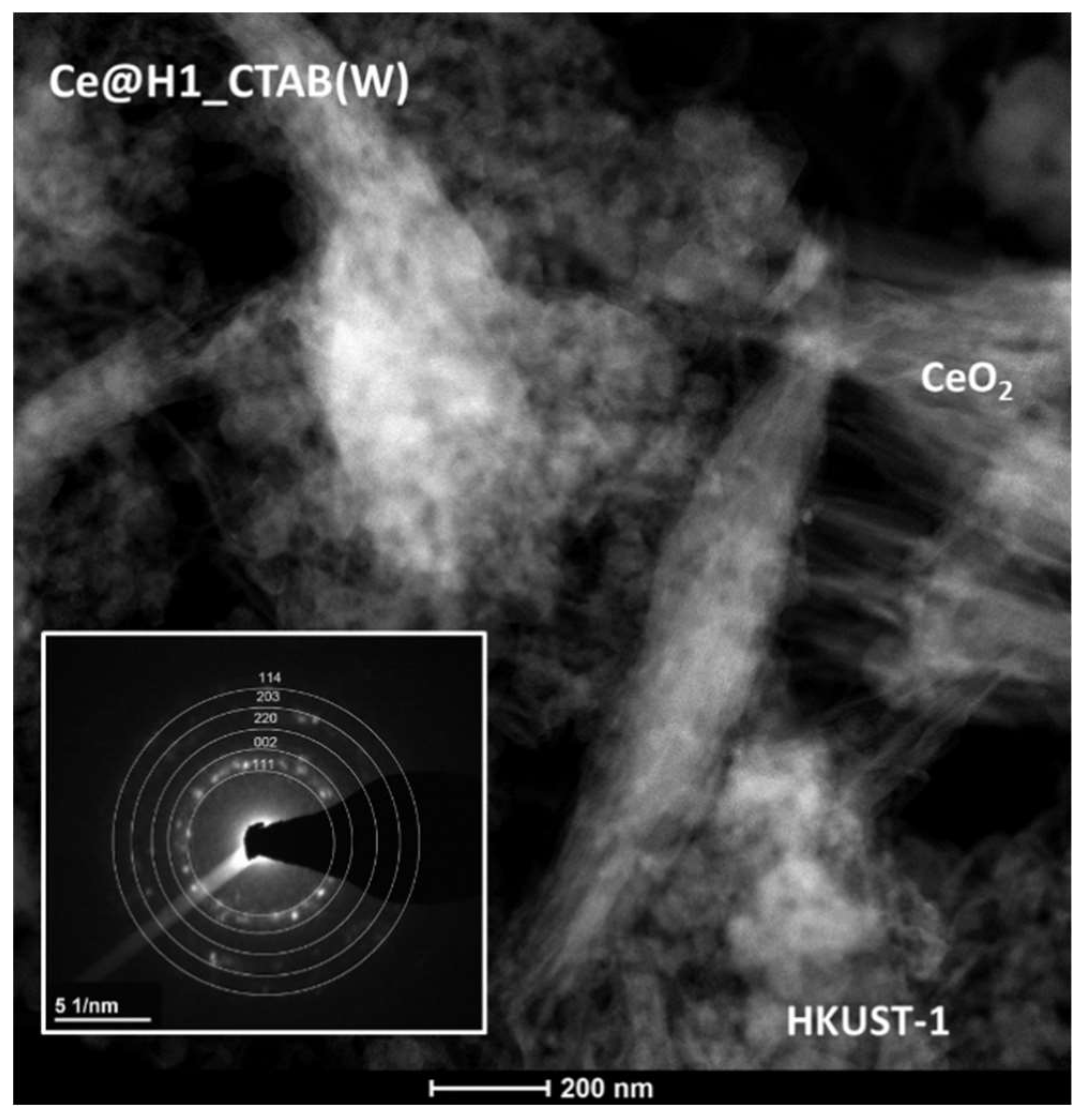
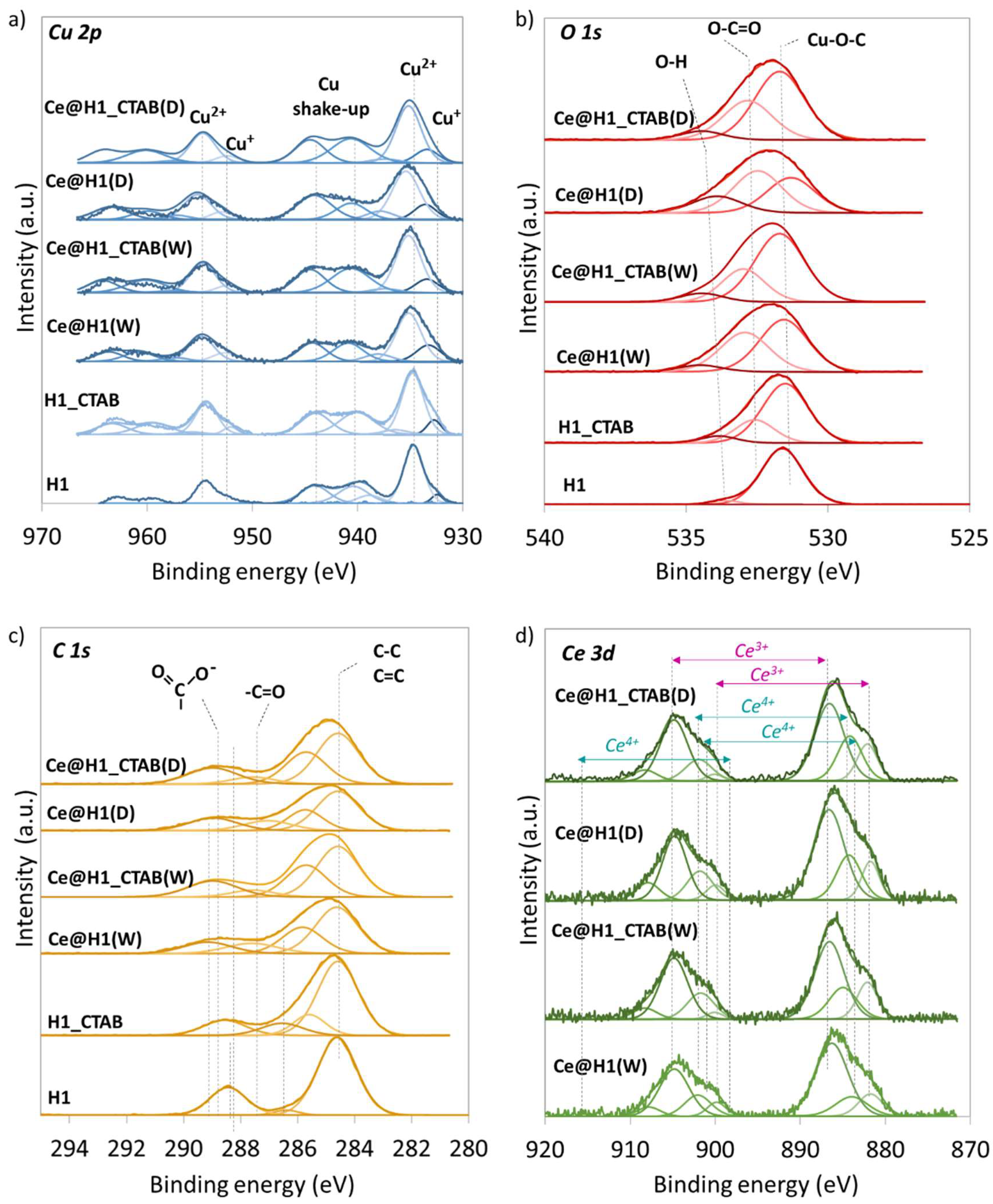
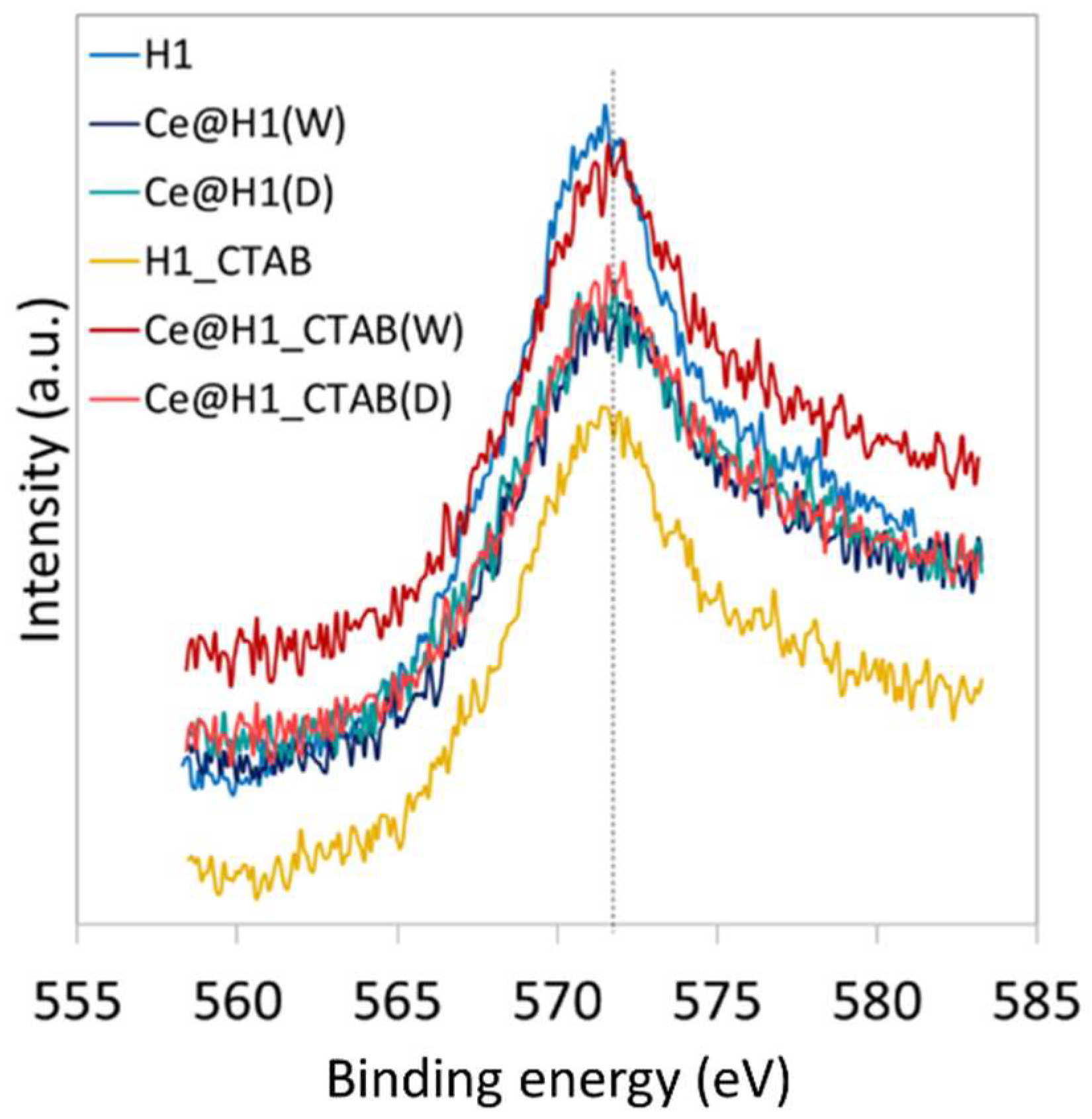
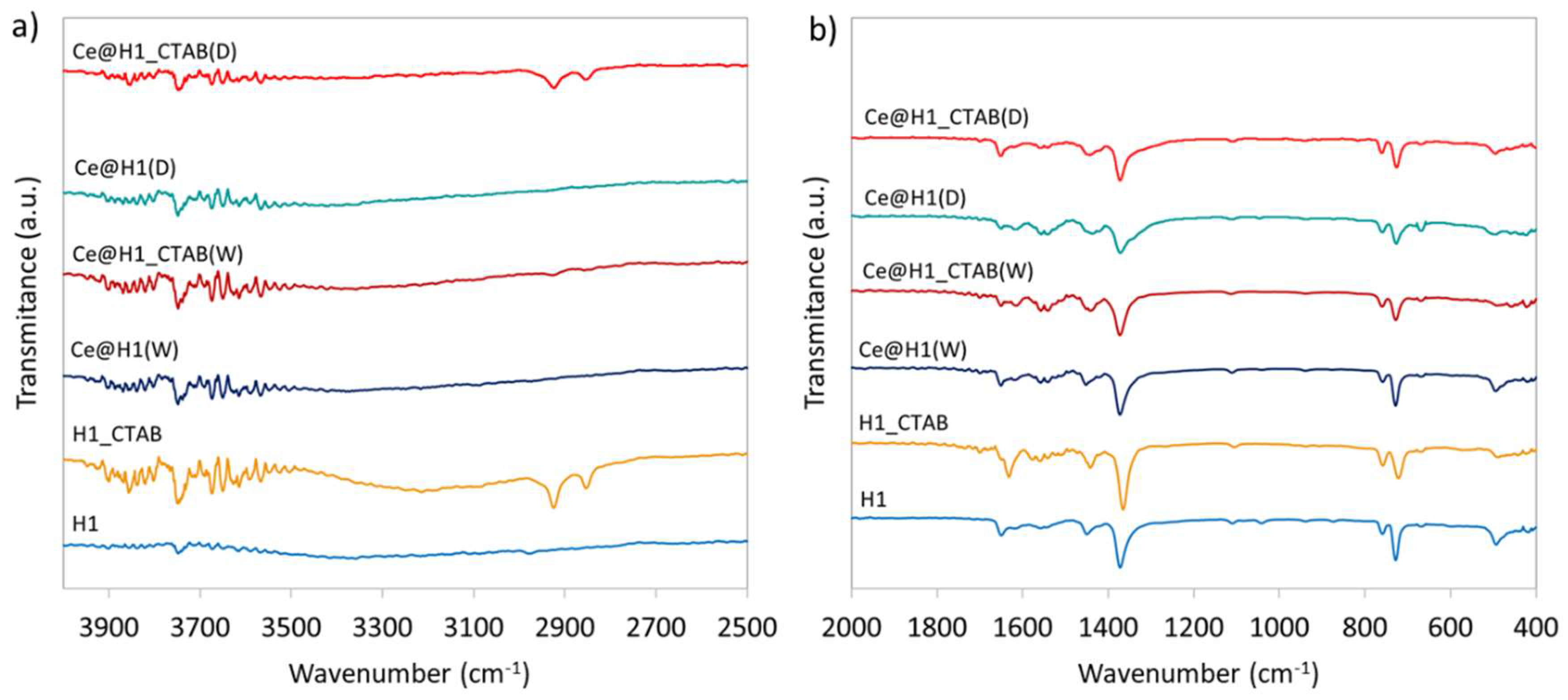
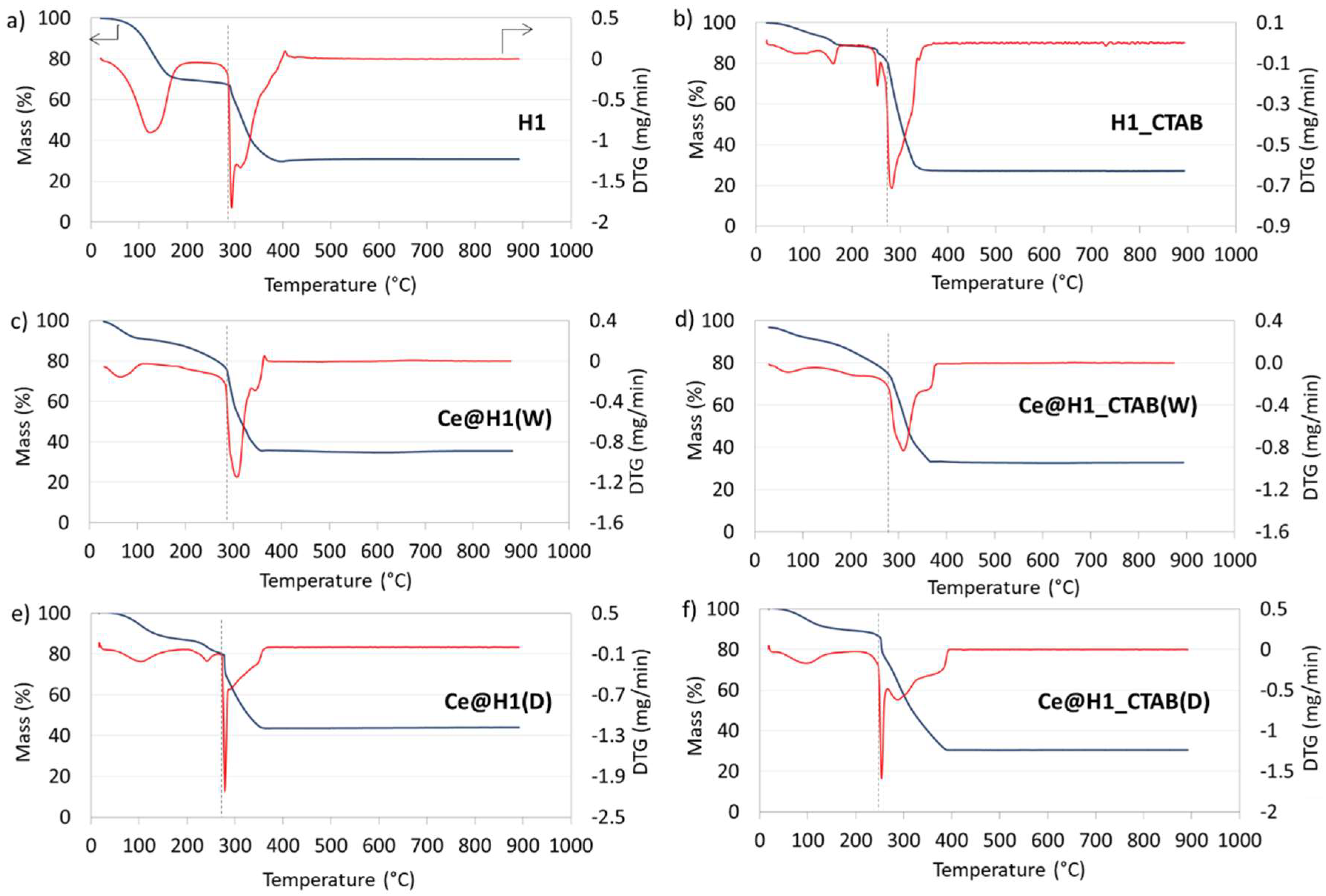
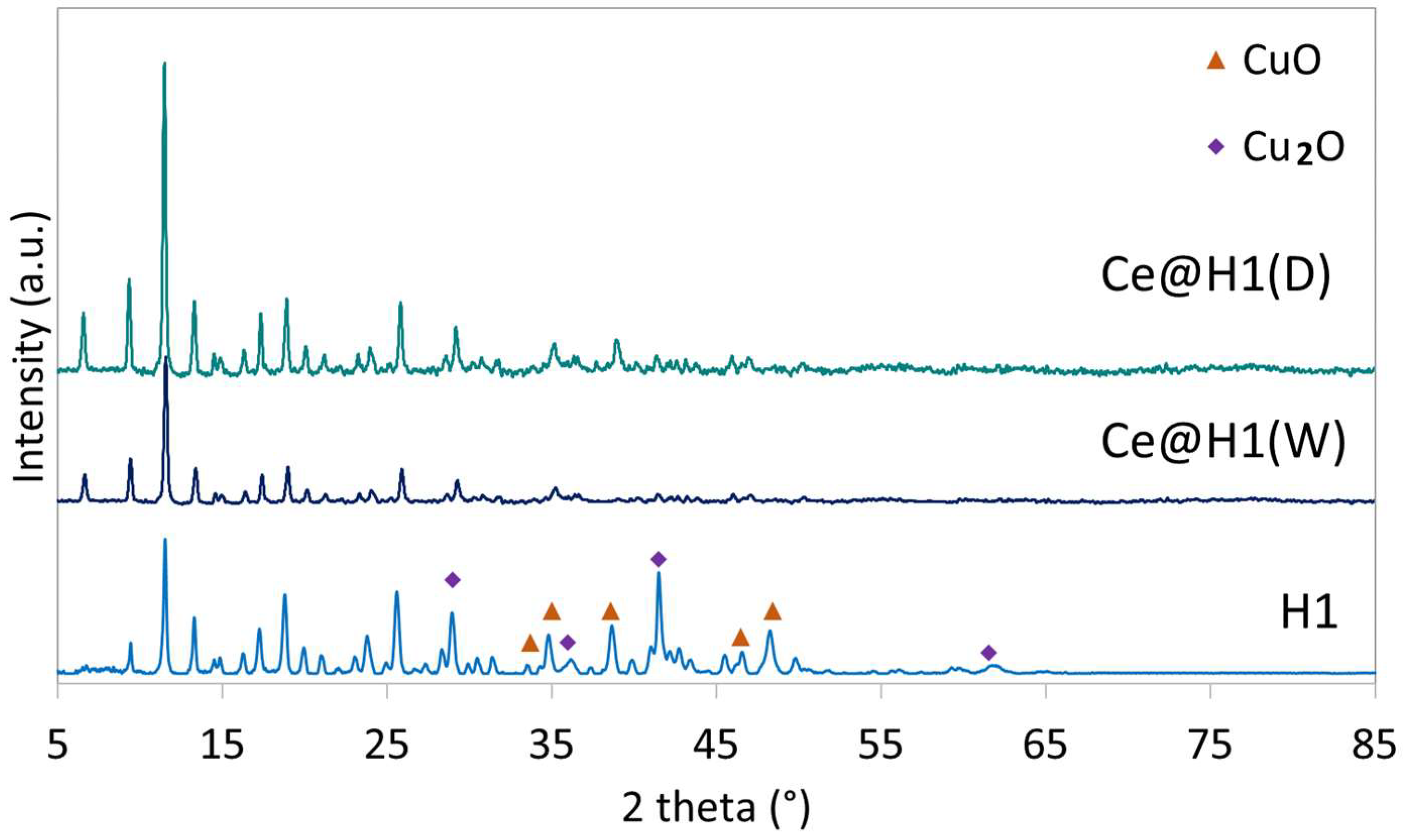
2.1. Characterisation
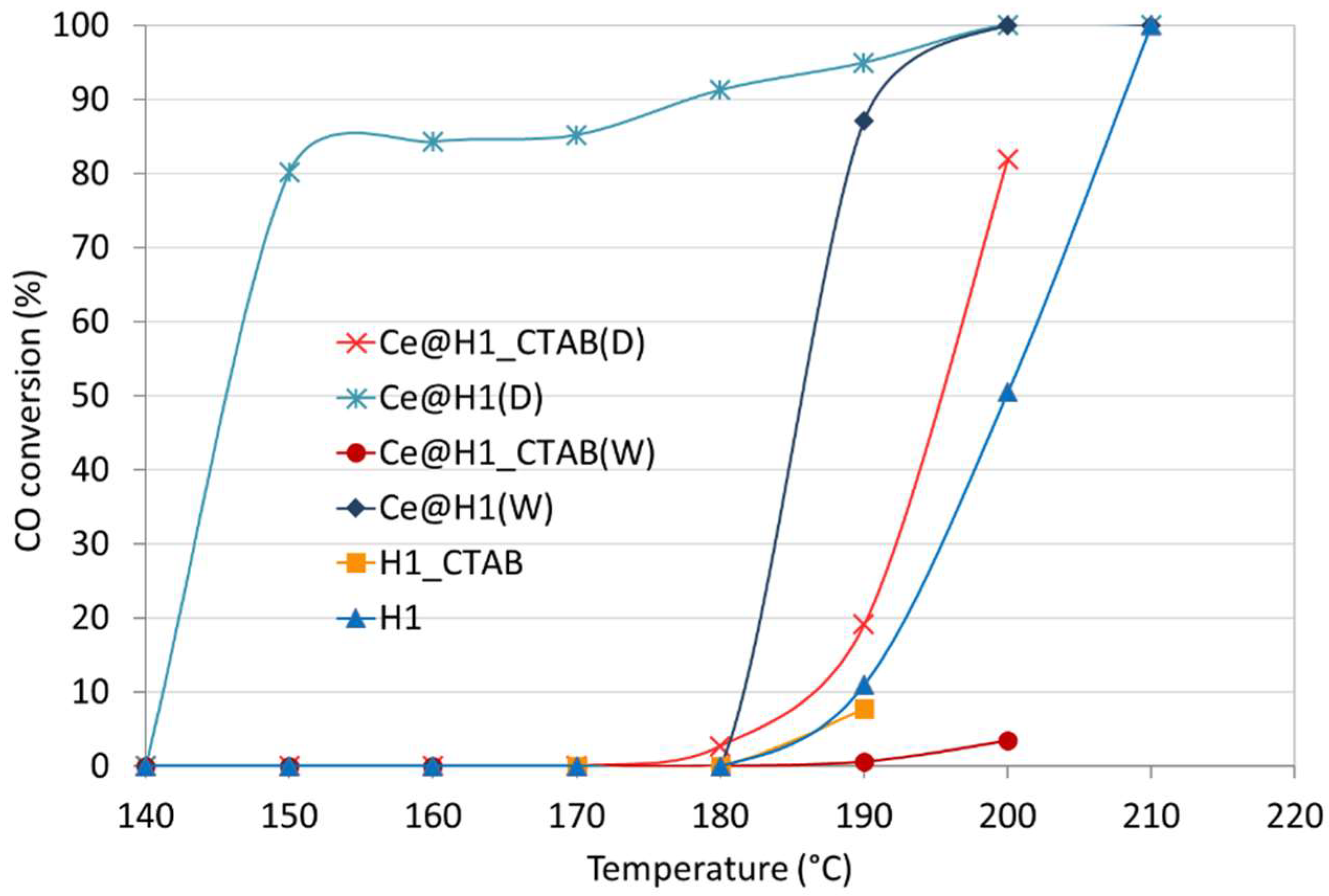
2.2. Catalytic Tests of CO Oxidation
3. Materials and Methods
3.1. Synthesis of HKUST-1
3.2. Synthesis of Ce@HKUST-1
3.3. Characterisation
3.4. Catalytic Tests of CO Oxidation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chui, S.S.; Lo, S.M.; Charmant, J.P.; Orpen, A.G.; Williams, I.D. A Chemically Functionalizable Nanoporous Material [Cu3(TMA)2(H2O)3]n. Science 1999, 283, 1148–1150. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.-S.; Adhikari, A.K.; Ku, C.-N.; Chiang, C.-L.; Kuo, H. Synthesis and characterization of porous HKUST-1 metal organic frameworks for hydrogen storage. Int. J. Hydrog. Energy 2012, 37, 13865–13871. [Google Scholar] [CrossRef]
- Rungtaweevoranit, B.; Zhao, Y.; Choi, K.M.; Yaghi, O.M. Cooperative effects at the interface of nanocrystalline metal–organic frameworks. Nano Res. 2016, 9, 47–58. [Google Scholar] [CrossRef]
- Tsuruoka, T.; Furukawa, S.; Takashima, Y.; Yoshida, K.; Isoda, S.; Kitagawa, S. Nanoporous Nanorods Fabricated by Coordination Modulation and Oriented Attachment Growth. Angew. Chem. Int. Ed. 2009, 48, 4739–4743. [Google Scholar] [CrossRef] [PubMed]
- Umemura, A.; Diring, S.; Furukawa, S.; Uehara, H.; Tsuruoka, T.; Kitagawa, S. Morphology Design of Porous Coordination Polymer Crystals by Coordination Modulation. J. Am. Chem. Soc. 2011, 133, 15506–15513. [Google Scholar] [CrossRef] [PubMed]
- Schaate, A.; Roy, P.; Godt, A.; Lippke, J.; Waltz, F.; Wiebcke, M.; Behrens, P. Modulated Synthesis of Zr-Based Metal-Organic Frameworks: From Nano to Single Crystals. Chem. Eur. J. 2011, 17, 6643–6651. [Google Scholar] [CrossRef]
- Lu, G.; Cui, C.; Zhang, W.; Liu, Y.; Huo, F. Synthesis and Self-Assembly of Monodispersed Metal-Organic Framework Microcrystals. Chem. Asian J. 2012, 8, 69–72. [Google Scholar] [CrossRef]
- Wißmann, G.; Schaate, A.; Lilienthal, S.; Bremer, I.; Schneider, A.M.; Behrens, P. Modulated synthesis of Zr-fumarate MOF. Microporous Mesoporous Mater. 2012, 152, 64–70. [Google Scholar] [CrossRef]
- Cravillon, J.; Nayuk, R.; Springer, S.; Feldhoff, A.; Huber, K.; Wiebcke, M. Controlling Zeolitic Imidazolate Framework Nano- and Microcrystal Formation: Insight into Crystal Growth by Time-Resolved In Situ Static Light Scattering. Chem. Mater. 2011, 23, 2130–2141. [Google Scholar] [CrossRef]
- Xin, C.; Zhan, H.; Huang, X.; Li, H.; Zhao, N.; Xiao, F.; Wei, W.; Sun, Y. Effect of various alkaline agents on the size and morphology of nano-sized HKUST-1 for CO2 adsorption. RSC Adv. 2015, 5, 27901–27911. [Google Scholar] [CrossRef]
- Mu, X.; Chen, Y.; Lester, E.; Wu, T. Optimized synthesis of nano-scale high quality HKUST-1 under mild conditions and its application in CO2 capture. Microporous Mesoporous Mater. 2018, 270, 249–257. [Google Scholar] [CrossRef]
- Liu, Q.; Jin, L.-N.; Sun, W.-Y. Facile fabrication and adsorption property of a nano/microporous coordination polymer with controllable size and morphology. Chem. Commun. 2012, 48, 8814–8816. [Google Scholar] [CrossRef] [PubMed]
- Nune, S.K.; Thallapally, P.K.; Dohnalkova, A.; Wang, C.; Liu, J.; Exarhos, G.J. Synthesis and properties of nano zeolitic imidazolate frameworks. Chem. Commun. 2010, 46, 4878–4880. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Amsler, M.; Goedecker, S.; Caravella, A.; Yoshida, M.; Kato, M. Surfactant-assisted synthesis of large Cu-BTC MOFs single crystals and the potential utilization as photodetectors. CrystEngComm 2019, 21, 3948–3953. [Google Scholar] [CrossRef]
- Ranft, A.; Betzler, S.B.; Haase, F.; Lotsch, B.V. Additive-mediated size control of MOF nanoparticles. CrystEngComm 2013, 15, 9296–9300. [Google Scholar] [CrossRef]
- Goyal, P.S.; Aswal, V.K. Micellar structure and inter-micelle interactions in micellar solutions: Results of small angle neutron scattering studies. Curr. Sci. 2001, 80, 972–979. [Google Scholar]
- Tan, Y.C.; Zeng, H.C. Defect Creation in HKUST-1 via Molecular Imprinting: Attaining Anionic Framework Property and Mesoporosity for Cation Exchange Applications. Adv. Funct. Mater. 2017, 27, 1703765. [Google Scholar] [CrossRef]
- Wang, F.; Guo, H.; Chai, Y.; Li, Y.; Liu, C. The controlled regulation of morphology and size of HKUST-1 by coordination modulation method. Microporous Mesoporous Mater. 2013, 173, 181–188. [Google Scholar] [CrossRef]
- Isaeva, V.I.; Kustov, L.M. The application of metal-organic frameworks in catalysis (Review). Pet. Chem. 2010, 50, 167–180. [Google Scholar] [CrossRef]
- Yepez, R.; García, S.; Schachat, P.; Sánchez-Sánchez, M.; González-Estefan, J.H.; González-Zamora, E.; Ibrara, I.A.; Aguilar-Pliego, J. Catalytic activity of HKUST-1 in the oxidation of trans-ferulic acid to vanillin. New J. Chem. 2015, 39, 5112–5115. [Google Scholar] [CrossRef]
- Arzehgar, Z.; Sajjadifar, S.; Arandiyan, H. HKUST-1 as an efficient and reusable heterogeneous catalyst for synthesis of 1,4-dihydropyridine at room temperature. Asian J. Green Chem. 2018, 3, 43–52. [Google Scholar] [CrossRef]
- Toyao, T.; Styles, M.J.; Yago, T.; Sadiq, M.M.; Riccò, R.; Suzuki, K.; Horiuchi, Y.; Takahashi, M.; Falcaro, P. Fe3O4@HKUST-1 and Pd/Fe3O4@HKUST-1 as magnetically recyclable catalysts prepared via conversion from a Cu-based ceramic. CrystEngComm 2017, 19, 4201–4210. [Google Scholar] [CrossRef]
- Guo, P.; Froese, C.; Fu, Q.; Chen, Y.-T.; Peng, B.; Kleist, W.; Fischer, R.A.; Muhler, M.; Wang, Y. CuPd Mixed-Metal HKUST-1 as Catalyst for Aerobic Alcohol Oxidation. J. Phys. Chem. C 2018, 122, 21433–21440. [Google Scholar] [CrossRef]
- Ye, J.; Liu, C. Cu3(BTC)2: CO oxidation over MOF based catalysts. Chem. Commun. 2011, 47, 2167–2169. [Google Scholar] [CrossRef] [PubMed]
- Zamaro, J.M.; Pérez, N.C.; Miró, E.E.; Casado, C.; Seoane, B.; Téllez, C.; Coronas, J. HKUST-1 MOF: A matrix to synthesize CuO and CuO–CeO2 nanoparticle catalysts for CO oxidation. Chem. Eng. J. 2012, 195-196, 180–187. [Google Scholar] [CrossRef]
- Zheng, X.-C.; Wu, S.-H.; Wang, S.-P.; Wang, S.-R.; Zhang, S.-M.; Huang, W.-P. The preparation and catalytic behavior of copper–cerium oxide catalysts for low-temperature carbon monoxide oxidation. Appl. Catal. A Gen. 2005, 283, 217–223. [Google Scholar] [CrossRef]
- Lin, K.-S.; Chowdhury, S. Synthesis, Characterization, and Application of 1-D Cerium Oxide Nanomaterials: A Review. Int. J. Mol. Sci. 2010, 11, 3226–3251. [Google Scholar] [CrossRef]
- Goworek, J.; Kierys, A.; Gac, W.; Borówka, A.; Kusak, R. Thermal degradation of CTAB in as-synthesized MCM-41. J. Therm. Anal. Calorim. 2009, 96, 375–382. [Google Scholar] [CrossRef]
- Chitsaz, A.; Jalilpour, M.; Fathalilou, M. Effects of PVP and CTAB surfactants on the morphology of cerium oxide nanoparticles synthesized via co-precipitation method. Int. J. Mater. Res. 2013, 104, 511–514. [Google Scholar] [CrossRef]
- Kim, Y.-Y.; Neudeck, C.; Walsh, D. Biopolymer templating as synthetic route to functional metal oxide nanoparticles and porous sponges. Polym. Chem. 2010, 1, 272–275. [Google Scholar] [CrossRef]
- Pan, C.; Zhang, D.; Shi, L. CTAB assisted hydrothermal synthesis, controlled conversion and CO oxidation properties of CeO2 nanoplates, nanotubes, and nanorods. J. Solid State Chem. 2008, 181, 1298–1306. [Google Scholar] [CrossRef]
- Peng, B.; Feng, C.; Liu, S.; Zhang, R. Synthesis of CuO catalyst derived from HKUST-1 temple for the low-temperature NH3-SCR process. Catal. Today 2018, 314, 122–128. [Google Scholar] [CrossRef]
- Fan, C.; Dong, H.; Liang, Y.; Yang, J.; Tang, G.; Zhang, W.; Cao, Y. Sustainable synthesis of HKUST-1 and its composite by biocompatible ionic liquid for enhancing visible-light photocatalytic performance. J. Clean. Prod. 2019, 208, 353–362. [Google Scholar] [CrossRef]
- Hsu, K.; Chen, D. Green synthesis and synergistic catalytic effect of Ag/reduced graphene oxide nanocomposite. Nanoscale Res. Lett. 2014, 9, 484. [Google Scholar] [CrossRef]
- Zhou, L.; Niu, Z.; Jin, X.; Tang, L.; Zhu, L. Effect of Lithium Doping on the Structures and CO2 Adsorption Properties of Metal-Organic Frameworks HKUST-1. ChemistrySelect 2018, 3, 12865–12870. [Google Scholar] [CrossRef]
- Kozachuk, O.; Yusenko, K.; Noei, H.; Wang, Y.; Walleck, S.; Glaser, T.; Fischer, R.A. Solvothermal growth of a ruthenium metal–organic framework featuring HKUST-1 structure type as thin films on oxide surfaces. Chem. Commun. 2011, 47, 8509–8511. [Google Scholar] [CrossRef]
- Kettner, M.; Ševčíková, K.; Homola, P.; Matolín, V.; Nehasil, V. Influence of the Ce–F interaction on cerium photoelectron spectra in CeOXFY layers. Chem. Phys. Lett. 2015, 639, 126–130. [Google Scholar] [CrossRef]
- Seo, Y.-K.; Hundal, G.; Jang, I.T.; Hwang, Y.K.; Jun, C.-H.; Chang, J.-S. Microwave Synthesis of Hybrid Inorganic−Organic Materials Including Porous Cu3(BTC)2 from Cu(II)-Trimesate Mixture. Microporous Mesoporous Mater. 2009, 119, 331–337. [Google Scholar] [CrossRef]
- Zhu, C.; Zhang, Z.; Wang, B.; Chen, Y.; Wang, H.; Chen, X.; Zhang, H.; Sun, N.; Wei, W.; Sun, Y. Synthesis of HKUST-1#MCF compositing materials for CO2 adsorption. Microporous Mesoporous Mater. 2016, 226, 476–481. [Google Scholar] [CrossRef]
- Lin, A.K.-Y.; Hsieh, Y.-T. Copper-based metal organic framework (MOF), HKUST-1, as an efficient adsorbent to remove p-nitrophenol from water. J. Taiwan Inst. Chem. Eng. 2015, 50, 223–228. [Google Scholar] [CrossRef]
- Guo, Z.; Song, L.; Xu, T.; Gao, D.; Li, C.; Hu, X.; Chen, G. CeO2-CuO Bimetal Oxides Derived from Ce-based MOF and Their Difference in Catalytic Activities for CO Oxidation. Mater. Chem. Phys. 2019, 226, 338–343. [Google Scholar] [CrossRef]
- Zhu, C.; Ding, T.; Gao, W.; Ma, K.; Tian, Y.; Li, X. CuO/CeO2 catalysts synthesized from Ce-UiO-66 metal-organic framework for preferential CO oxidation. Int. J. Hydrog. Energy 2017, 42, 17457–17465. [Google Scholar] [CrossRef]
- Liu, W.; Stephanopoulos, M.F. Total Oxidation of Carbon Monoxide and Methane over Transition Metal Fluorite Oxide Composite Catalysts: I. Catalyst Composition and Activity. J. Catal. 1995, 153, 304. [Google Scholar] [CrossRef]
- Zhao, F.; Li, S.; Wu, X.; Yue, R.; Li, W.; Zha, X.; Deng, Y.; Chen, Y. Catalytic Behaviour of Flame-Made CuO-CeO2 Nanocatalysts in Efficient CO Oxidation. Catalysts 2019, 9, 256. [Google Scholar] [CrossRef]
- Noei, H.; Amirjalayer, S.; Müller, M.; Zhang, X.; Schmid, R.; Muhler, M.; Fischer, R.A.; Wang, Y. Low-Temperature CO Oxidation over Cu-Based Metal-Organic Frameworks Monitored by using FTIR Spectroscopy. ChemCatChem 2012, 4, 755–759. [Google Scholar] [CrossRef]
- Noei, H.; Kozachuk, O.; Amirjalayer, S.; Bureekaew, S.; Kauer, M.; Schmid, R.; Marler, B.; Muhler, M.; Fischer, R.A.; Wang, Y. CO Adsorption on a Mixed-Valence Ruthenium Metal–Organic Framework Studied by UHV-FTIR Spectroscopy and DFT Calculations. J. Phys. Chem. C. 2013, 117, 5658–5666. [Google Scholar] [CrossRef]



| Crystal Plane | Particle Size (nm) | |||||
|---|---|---|---|---|---|---|
| H1 | H1_CTAB | Ce@H1(D) | Ce@H1_CTAB(D) | Ce@H1(W) | Ce@H1_CTAB(W) | |
| (200) | 429.1 | 504.9 | 233.6 | 302.9 | 237.6 | 253.9 |
| (220) | 453.5 | 318.6 | 364.5 | 227.5 | 818.7 | 226.6 |
| (222) | 473.9 | 358.6 | 565.7 | 220.7 | 713.5 | 278.0 |
| (400) | 469.9 | 290.2 | 268.2 | 287.5 | 748.6 | 218.7 |
| Sample | SBET (m2/g) | Vtotal (cm3/g) | d (nm) |
|---|---|---|---|
| H1 | 1003 | 0.584 | 2.3 |
| Ce@H1(W) | 958 | 0.574 | 4.2 |
| Ce@H1(D) | 318 | 0.331 | 4.2 |
| H1_CTAB | 160 | 0.602 | 1.5 |
| Ce@H1_CTAB(W) | 444 | 0.952 | 8.6 |
| Ce@H1_CTAB(D) | 328 | 0.409 | 5.0 |
| Sample | Temperature of Decomposition (C) | Mass Loss (%) |
|---|---|---|
| H1 | 286 | 36.8 |
| Ce@H1(W) | 270 | 36.1 |
| Ce@H1(D) | 271 | 35.8 |
| H1_CTAB | 261 | 56.4 |
| Ce@H1_CTAB(W) | 235 | 52.1 |
| Ce@H1_CTAB(D) | 229 | 57.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stawowy, M.; Jagódka, P.; Matus, K.; Samojeden, B.; Silvestre-Albero, J.; Trawczyński, J.; Łamacz, A. HKUST-1-Supported Cerium Catalysts for CO Oxidation. Catalysts 2020, 10, 108. https://doi.org/10.3390/catal10010108
Stawowy M, Jagódka P, Matus K, Samojeden B, Silvestre-Albero J, Trawczyński J, Łamacz A. HKUST-1-Supported Cerium Catalysts for CO Oxidation. Catalysts. 2020; 10(1):108. https://doi.org/10.3390/catal10010108
Chicago/Turabian StyleStawowy, Michalina, Paulina Jagódka, Krzysztof Matus, Bogdan Samojeden, Joaquin Silvestre-Albero, Janusz Trawczyński, and Agata Łamacz. 2020. "HKUST-1-Supported Cerium Catalysts for CO Oxidation" Catalysts 10, no. 1: 108. https://doi.org/10.3390/catal10010108
APA StyleStawowy, M., Jagódka, P., Matus, K., Samojeden, B., Silvestre-Albero, J., Trawczyński, J., & Łamacz, A. (2020). HKUST-1-Supported Cerium Catalysts for CO Oxidation. Catalysts, 10(1), 108. https://doi.org/10.3390/catal10010108









