Fractionated Follow-Up Chemotherapy Delays the Onset of Resistance in Bone Metastatic Prostate Cancer
Abstract
1. Introduction
2. Model
- The fitness function for OBs is given bywhere the first summand of corresponds to the benefit conferred to OBs by OCs. This benefit can be attributed to the secretion of TGF- by OCs that recruits OBs to the remodeling site. This is dependent on the proportion of OC among healthy cells () and the dearth of bone with respect to its standard equilibrium . In the second summand, is the benefit that an OB receives from interacting with any PCa cell due to their natural secretion of TGF-.
- The fitness function for OCs is given bywhich captures the benefit conferred to OCs by OBs that can be attributed to the secretion of RANKL as a result of bone resorption. This is dependent on the proportion of OBs among healthy cells () and the overabundance of bone with respect to its standard equilibrium . Since both and are functions of the proportions of OCs and OBs, we could rewrite the equations for and as a replicator dynamic model (for a similar model, see Kaznatcheev [23]).
- Finally, the fitness functions for chemotherapy-sensitive and -resistant tumors arewhere is the benefit a PCa cell receives from interacting with OCs. The OC-led resorption of the bone allows the neighboring PCa cells to access nutrients and growth factors previously embedded in the bone; is the benefit that a PCa cell receives from interacting with other PCa cells derived from the secretion of TGF-. The cost of resistance to treatment is r, and the efficacy of the treatment is s. Resistance to chemotherapy is common and results from the treatment providing strong selection for PCa cells that can avoid its cyto-toxicity. This resistance often comes through the upregulation of drug exporter pumps on the surface of PCa cells [24,25,26]. Producing and maintaining these pumps is energetically costly to the PCa cells but allows those that have a sufficient number of them to deal with cytotoxic drugs.
Chemotherapy
3. Results
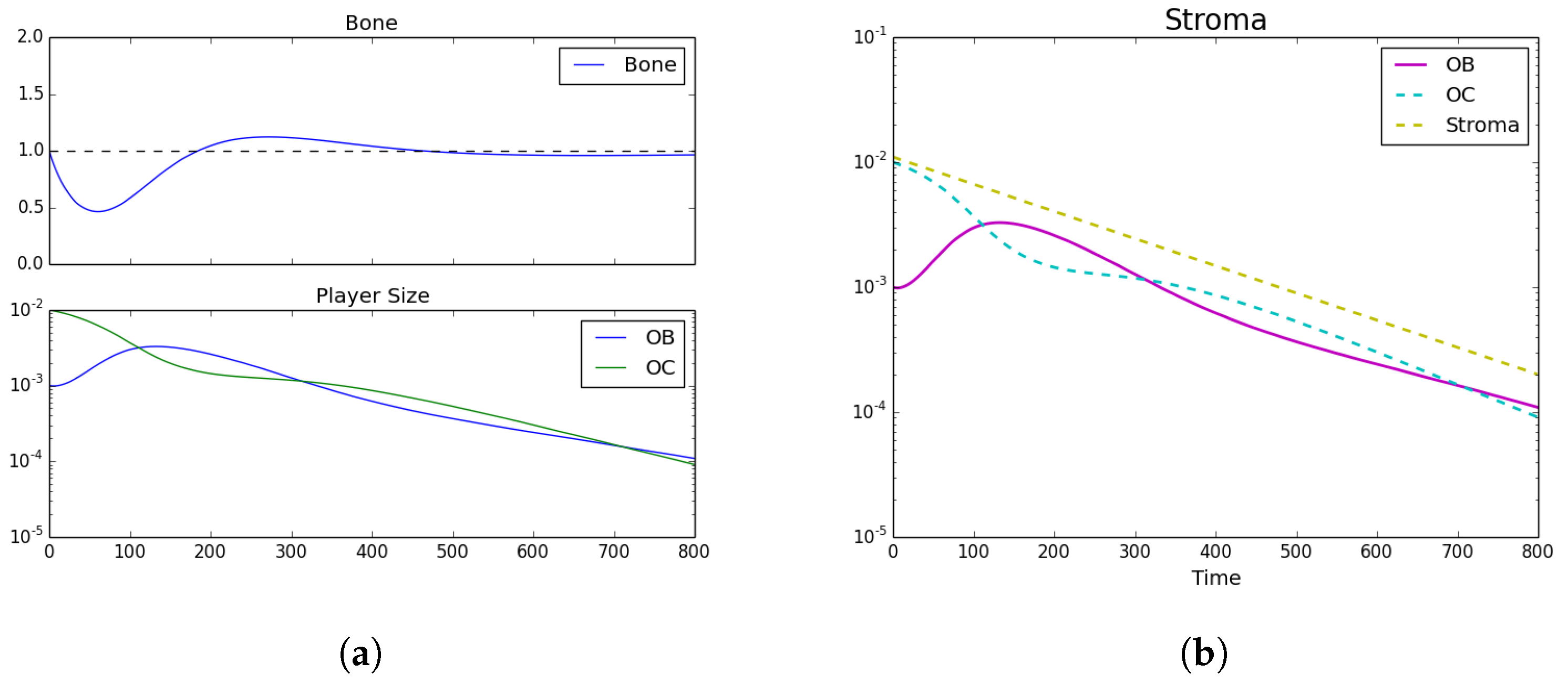
3.1. Tumor Introduction
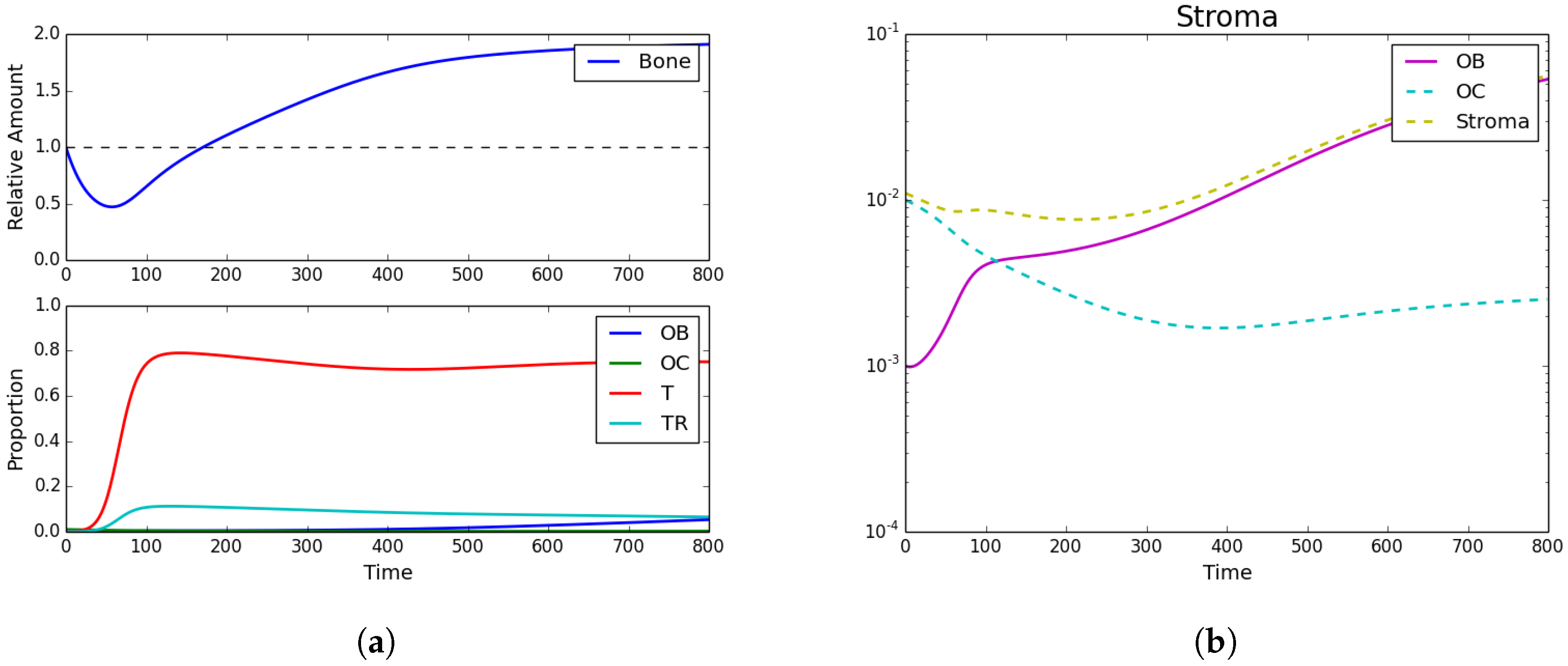
3.2. Standard Chemotherapy
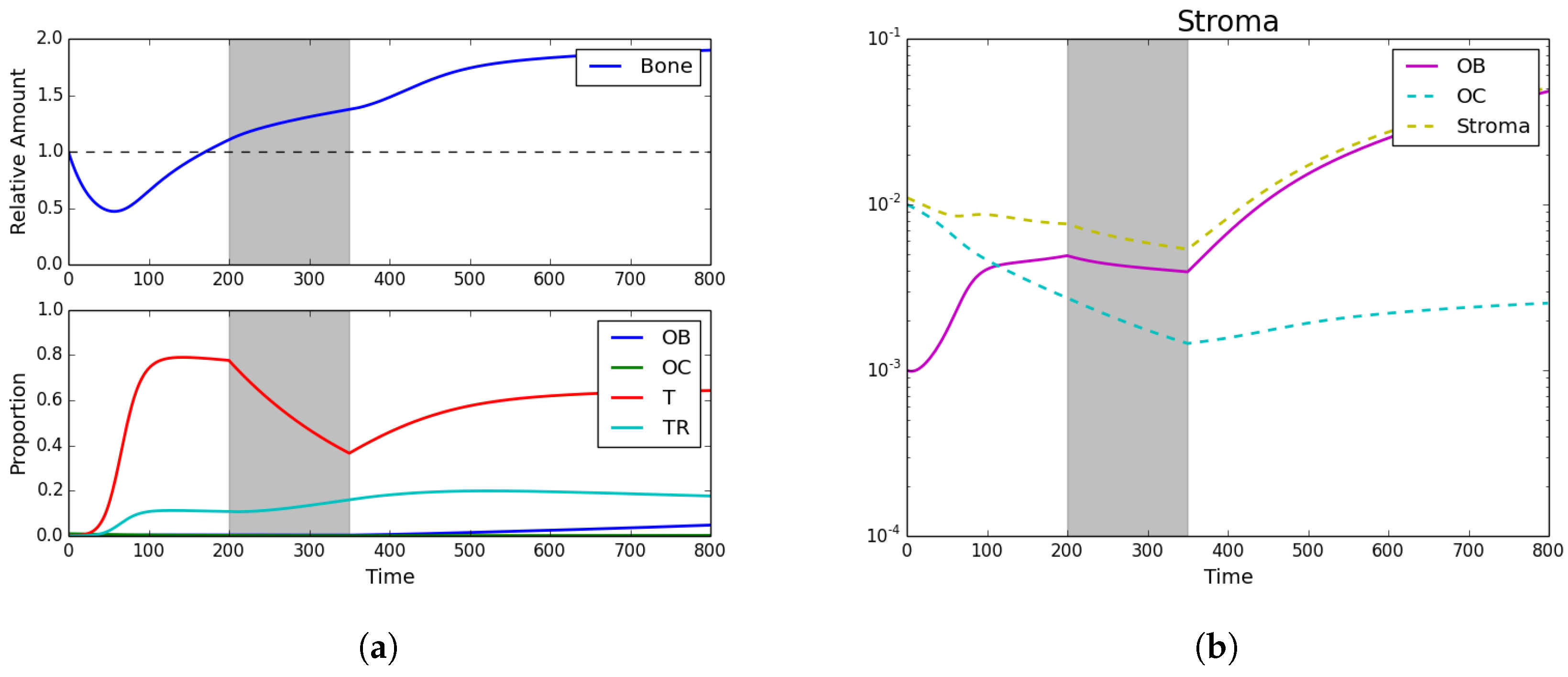
3.3. Fractionated Treatment
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- National Cancer Institute. Cancer Stat Facts: Prostate Cancer. Available online: https://seer.cancer.gov/statfacts/html/prost.html (accessed on 1 August 2017).
- Araujo, A.; Cook, L.M.; Lynch, C.C.; Basanta, D. An integrated computational model of the bone microenvironment in bone-metastatic prostate cancer. Cancer Res. 2014, 74, 2391–2401. [Google Scholar] [CrossRef] [PubMed]
- Cook, L.M.; Araujo, A.; Pow-Sang, J.M.; Budzevich, M.M.; Basanta, D.; Lynch, C.C. Predictive computational modeling to define effective treatment strategies for bone metastatic prostate cancer. Sci. Rep. 2016, 6, 29384. [Google Scholar] [CrossRef] [PubMed]
- Heppner, G.H. Tumor Heterogeneity. Cancer Res. 1984, 44, 2259–2265. [Google Scholar] [PubMed]
- Gallaher, J.; Cook, L.M.; Gupta, S.; Araujo, A.; Dhillon, J.; Park, J.Y.; Scott, J.G.; Pow-Sang, J.; Basanta, D.; Lynch, C.C. Improving treatment strategies for patients with metastatic castrate resistant prostate cancer through personalized computational modeling. Clin. Exp. Metastasis 2014, 31, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Kaznatcheev, A. Two conceptions of evolutionary games: Reductive vs effective. BioRxiv 2017, 231993. [Google Scholar] [CrossRef]
- Kaznatcheev, A. Effective games and the confusion over spatial structure. Proc. Natl. Acad. Sci. USA 2018, 115, 201719031. [Google Scholar] [CrossRef] [PubMed]
- Ryser, M.D.; Nigam, N.; Komarova, S.V. Mathematical Modeling of Spatio-Temporal Dynamics of a Single Bone Multicellular Unit. J. Bone Min. Res. 2009, 24, 860–870. [Google Scholar] [CrossRef] [PubMed]
- Ryser, M.D.; Qu, Y.; Komarova, S.V. Osteoprotegerin in Bone Metastases: Mathematical Solution to the Puzzle. PLoS Comput. Biol. 2012, 8, e1002703. [Google Scholar] [CrossRef] [PubMed]
- Buenzli, P.R.; Pivonka, P.; Gardiner, B.S. Modelling the anabolic response of bone using a cell population model. J. Theor. Biol. 2012, 307, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, I.P.M.; Bodmer, W.F. Modelling the consequences of interactions between tumour cells. Br. J. Cancer 1997, 75, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, I.P. Game-theory models of interactions between tumour cells. Eur. J. Cancer 1997, 33, 1495–1500. [Google Scholar] [CrossRef]
- Basanta, D.; Hatzikirou, H.; Deutsch, A. Studying the emergence of invasiveness in tumours using game theory. Eur. Phys. J. B 2008, 63, 393–397. [Google Scholar] [CrossRef]
- Kaznatcheev, A.; Scott, J.G.; Basanta, D. Edge effects in game-theoretic dynamics of spatially structured tumours. J. R. Soc. Interface 2015, 12, 20150154. [Google Scholar] [CrossRef] [PubMed]
- Basanta, D.; Simon, M.; Hatzikirou, H.; Deutsch, A. Evolutionary game theory elucidates the role of glycolysis in glioma progression and invasion. Cell Prolif. 2008, 41, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Basanta, D.; Scott, J.G.; Fishman, M.N.; Ayala, G.; Hayward, S.W.; Anderson, A.R.A. Investigating prostate cancer tumour-stroma interactions: clinical and biological insights from an evolutionary game. Br. J. Cancer 2012, 106, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Kaznatcheev, A.; Vander Velde, R.; Scott, J.G.; Basanta, D. Cancer treatment scheduling and dynamic heterogeneity in social dilemmas of tumour acidity and vasculature. Br. J. Cancer 2017, 116, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Dingli, D.; Chalub, F.A.C.C.; Santos, F.C.; van Segbroeck, S.; Pacheco, J.M. Cancer phenotype as the outcome of an evolutionary game between normal and malignant cells. Br. J. Cancer 2009, 101, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Ma, Y.; Newton, P.K. Capitalizing on competition: An evolutionary model of competitive release in metastatic castration resistant prostate cancer treatment. BioRxiv 2018. [Google Scholar] [CrossRef]
- Bilezikian, J.P.; Raisz, L.G.; Martin, T.J. Principles of Bone Biology, 3rd ed.; Academic Press/Elsevier: San Diego, CA, USA, 2008. [Google Scholar]
- Cook, L.M.; Shay, G.; Araujo, A.; Lynch, C.C. Integrating new discoveries into the “vicious cycle” paradigm of prostate to bone metastases. Cancer Metastasis Rev. 2014, 33, 511–525. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cunningham, J.J.; Brown, J.S.; Gatenby, R.A. Integrating evolutionary dynamics into treatment of metastatic castrate-resistant prostate cancer. Nat. Commun. 2017, 8, 1816. [Google Scholar] [CrossRef] [PubMed]
- Kaznatcheev, A. Evolutionary Dynamics of Cancer in the Bone. Theory, Evolution, and Games Group, 2015. Available online: https://egtheory.wordpress.com/2015/12/06/bone-cancer/ (accessed on 6 December 2015).
- O’Neill, A.J.; Prencipe, M.; Dowling, C.; Fan, Y.; Mulrane, L.; Gallagher, W.M.; O’Connor, D.; O’Connor, R.; Devery, A.; Corcoran, C.; et al. Characterisation and manipulation of docetaxel resistant prostate cancer cell lines. Mol. Cancer 2011, 10, 126. [Google Scholar]
- Hwang, C. Overcoming docetaxel resistance in prostate cancer: a perspective review. Ther. Adv. Med. Oncol. 2012, 4, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, C.; Rani, S.; O’Brien, K.; O’Neill, A.; Prencipe, M.; Sheikh, R.; Webb, G.; McDermott, R.; Watson, W.; Crown, J.; et al. Docetaxel-resistance in prostate cancer: Evaluating associated phenotypic changes and potential for resistance transfer via exosomes. PLoS ONE 2012, 7, e50999. [Google Scholar] [CrossRef] [PubMed]
- Henríquez, I.; Rodríguez-Antolín, A.; Cassinello, J.; Gonzalez San Segundo, C.; Unda, M.; Gallardo, E.; López-Torrecilla, J.; Juarez, A.; Arranz, J. Consensus statement on definition, diagnosis, and management of high-risk prostate cancer patients on behalf of the spanish groups of uro-oncology societies uroncor, guo, and sogug. Clin. Transl. Oncol. 2018, 20, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Keller, E.T.; Brown, J. Prostate cancer bone metastases promote both osteolytic and osteoblastic activity. J. Cell Biochem. 2004, 91, 718–729. [Google Scholar] [CrossRef] [PubMed]
- Beer, T.M.; Garzotto, M.; Henner, W.D.; Eilers, K.M.; Wersinger, E.M. Multiple cycles of intermittent chemotherapy in metastatic androgen-independent prostate cancer. Br. J. Cancer 2004, 91, 1425. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mountzios, I.; Bournakis, E.; Efstathiou, E.; Varkaris, A.; Wen, S.; Chrisofos, M.; Deliveliotis, C.; Alamanis, C.; Anastasiou, I.; Constantinides, C.; et al. Intermittent docetaxel chemotherapy in patients with castrate-resistant prostate cancer. Urology 2011, 77, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Orlando, P.A.; Gatenby, R.A.; Brown, J.S. Cancer treatment as a game: integrating evolutionary game theory into the optimal control of chemotherapy. Phys. Biol. 2012, 9, 065007. [Google Scholar] [CrossRef] [PubMed]
- Nanda, M.; Durrett, R. Spatial evolutionary games with weak selection. Proc. Natl. Acad. Sci. USA 2017, 114, 6046–6051. [Google Scholar] [CrossRef] [PubMed]
- Bacevic, K.; Noble, R.; Soffar, A.; Ammar, O.W.; Boszonyik, B.; Prieto, S.; Vincent, C.; Hochberg, M.E.; Krasinska, L.; Fisher, D. Spatial competition constrains resistance to targeted cancer therapy. Nat. Commun. 2017, 8, 1995. [Google Scholar] [CrossRef] [PubMed]
- Kaznatcheev, A. Drug Holidays and Losing Resistance with Replicator Dynamics. Theory, Evolution, and Games Group, 2016. Available online: https://egtheory.wordpress.com/2016/09/02/drug-holidays/ (accessed on 2 September 2016).
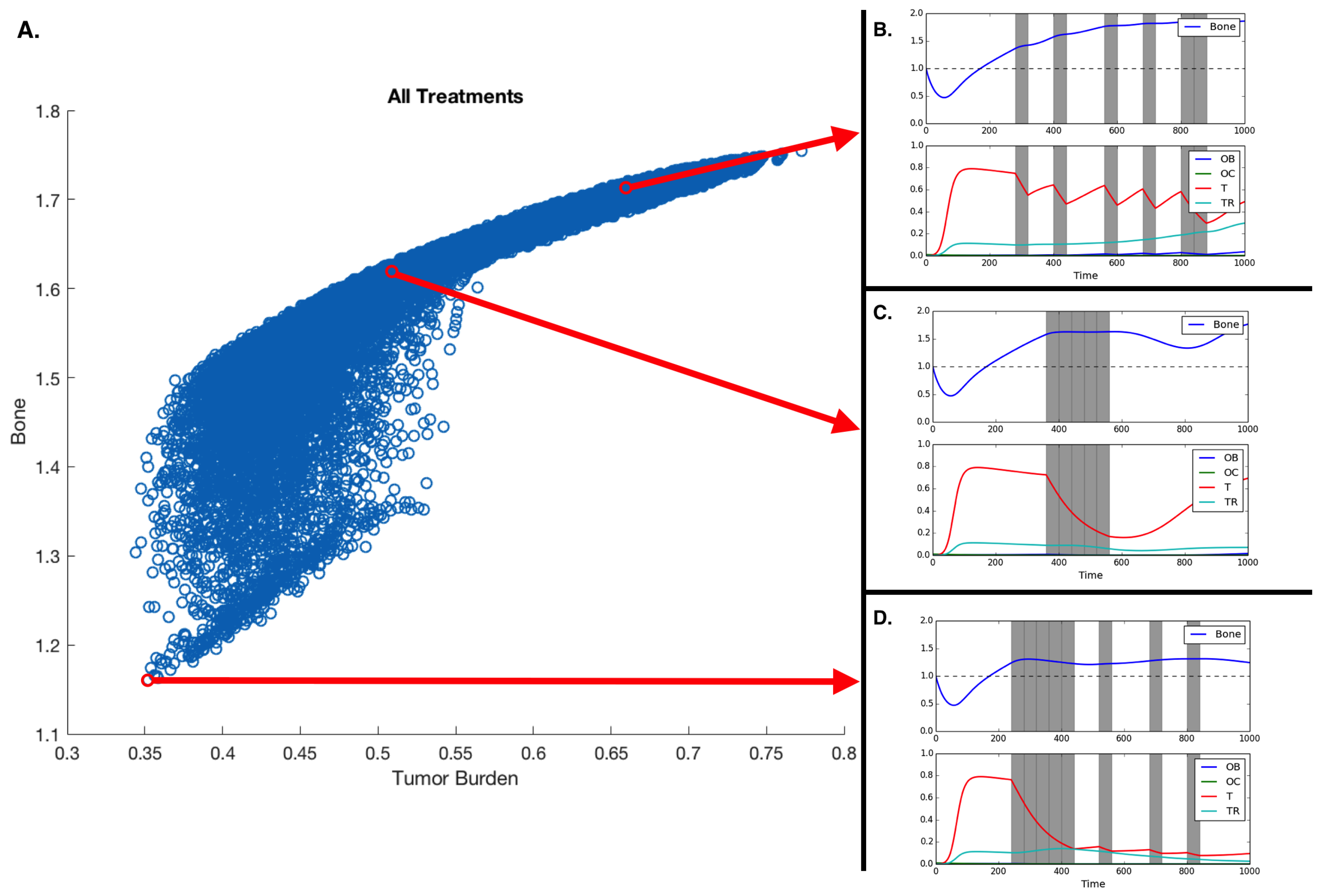
| Treatment Schedule | Bone Size | Average Tumor Burden |
|---|---|---|
| 0000000000000000 | 1.754 | 0.773 |
| 1111100100010010 | 1.160 | 0.352 |
| 1111010101000001 | 1.163 | 0.358 |
| 1111010100100100 | 1.165 | 0.358 |
| 1111011000010010 | 1.166 | 0.356 |
| 1110110101000100 | 1.172 | 0.366 |
| 0001111100000000 | 1.615 | 0.512 |
| 0100100010010011 | 1.709 | 0.666 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warman, P.I.; Kaznatcheev, A.; Araujo, A.; Lynch, C.C.; Basanta, D. Fractionated Follow-Up Chemotherapy Delays the Onset of Resistance in Bone Metastatic Prostate Cancer. Games 2018, 9, 19. https://doi.org/10.3390/g9020019
Warman PI, Kaznatcheev A, Araujo A, Lynch CC, Basanta D. Fractionated Follow-Up Chemotherapy Delays the Onset of Resistance in Bone Metastatic Prostate Cancer. Games. 2018; 9(2):19. https://doi.org/10.3390/g9020019
Chicago/Turabian StyleWarman, Pranav I., Artem Kaznatcheev, Arturo Araujo, Conor C. Lynch, and David Basanta. 2018. "Fractionated Follow-Up Chemotherapy Delays the Onset of Resistance in Bone Metastatic Prostate Cancer" Games 9, no. 2: 19. https://doi.org/10.3390/g9020019
APA StyleWarman, P. I., Kaznatcheev, A., Araujo, A., Lynch, C. C., & Basanta, D. (2018). Fractionated Follow-Up Chemotherapy Delays the Onset of Resistance in Bone Metastatic Prostate Cancer. Games, 9(2), 19. https://doi.org/10.3390/g9020019





