Roles of Integrin α6β4 Glycosylation in Cancer
Abstract
:1. Introduction
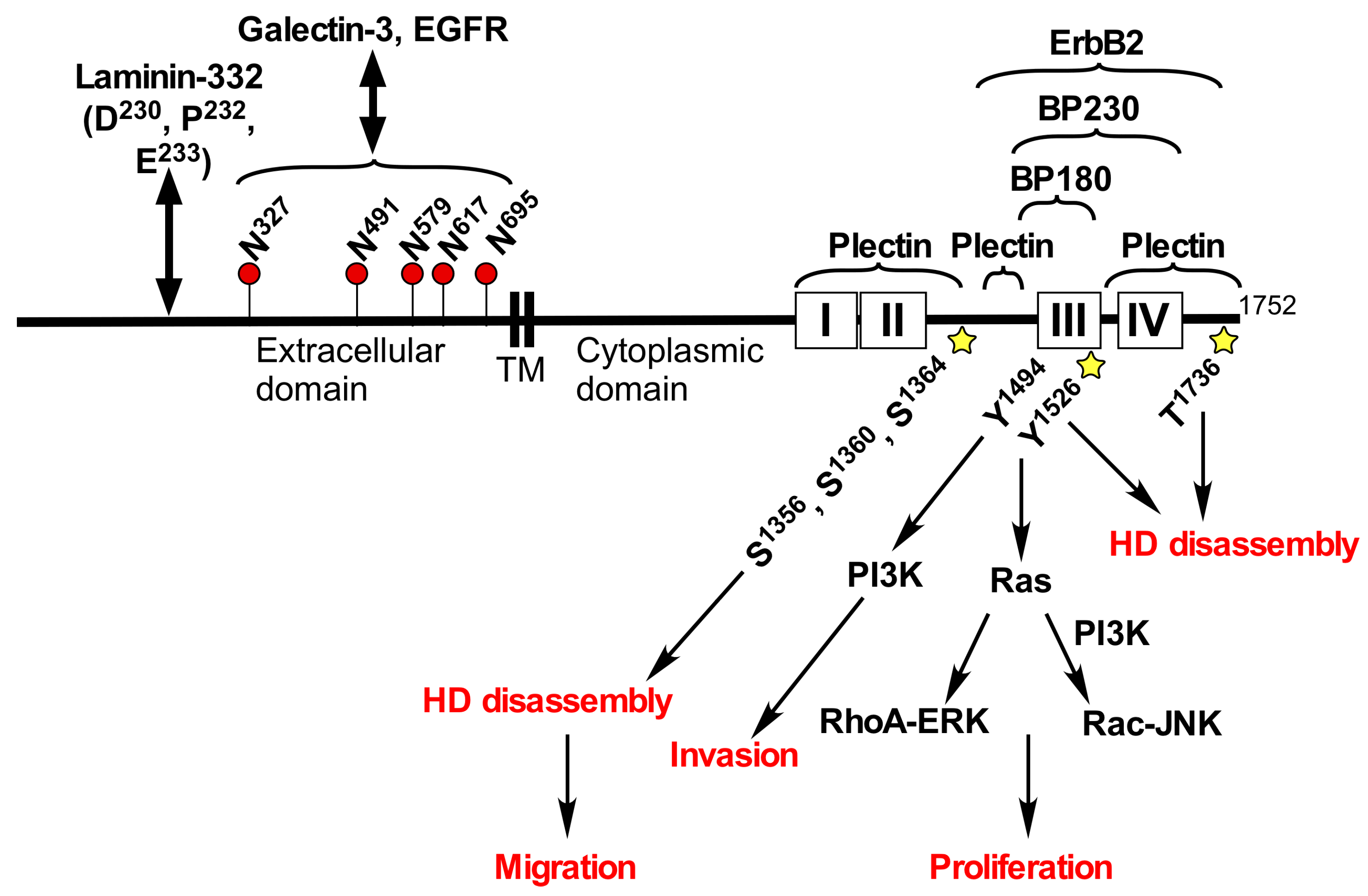
2. Structure and Functions of Integrin α6β4
3. Integrin α6β4 in Cancer
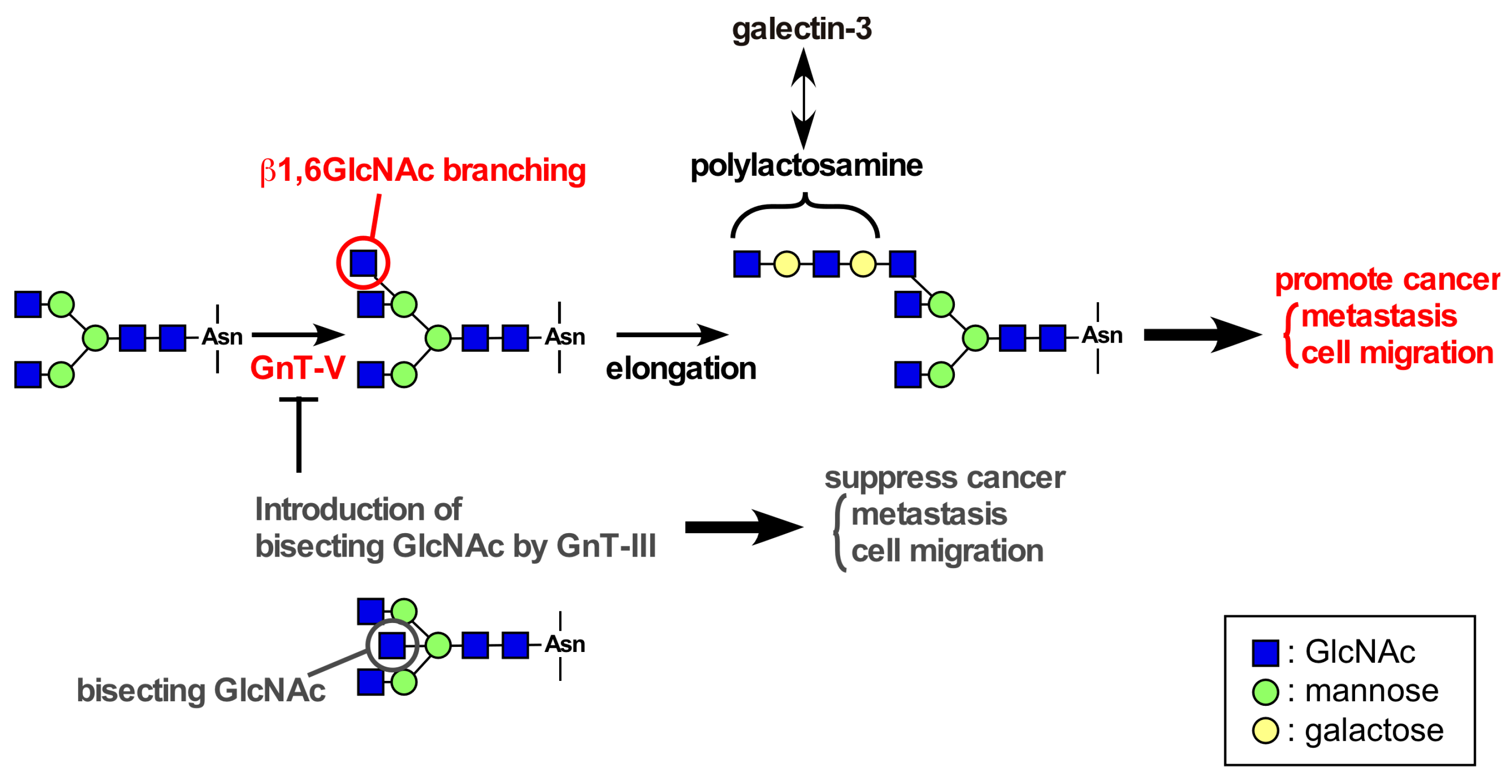
4. Roles of Glycans in Integrin β4 Function
5. Conclusions and Perspective
Acknowledgments
Conflicts of Interest
References
- Gu, J.; Taniguchi, N. Regulation of integrin functions by N-glycans. Glycoconj. J. 2004, 21, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Isaji, T.; Gu, J.; Nishiuchi, R.; Zhao, Y.; Takahashi, M.; Miyoshi, E.; Honke, K.; Sekiguchi, K.; Taniguchi, N. Introduction of bisecting GlcNAc into integrin α5β1 reduces ligand binding and downregulates cell adhesion and cell migration. J. Biol. Chem. 2004, 279, 19747–19754. [Google Scholar] [CrossRef] [PubMed]
- Isaji, T.; Sato, Y.; Fukuda, T.; Gu, J. N-glycosylation of the I-like domain of β1 integrin is essential for β1 integrin expression and biological function: Identification of the minimal N-glycosylation requirement for α5β1. J. Biol. Chem. 2009, 284, 12207–12216. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Kizuka, Y.; Ohtsubo, K.; Gu, J.; Taniguchi, N. Disease-associated glycans on cell surface proteins. Mol. Asp. Med. 2016, 51, 56–70. [Google Scholar] [CrossRef] [PubMed]
- Janik, M.E.; Litynska, A.; Vereecken, P. Cell migration-the role of integrin glycosylation. Biochim. Biophys. Acta 2010, 1800, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Hang, Q.; Isaji, T.; Hou, S.; Im, S.; Fukuda, T.; Gu, J. Integrin α5 suppresses the phosphorylation of epidermal growth factor receptor and its cellular signaling of cell proliferation via N-Glycosylation. J. Biol. Chem. 2015, 290, 29345–29360. [Google Scholar] [CrossRef] [PubMed]
- Hang, Q.; Isaji, T.; Hou, S.; Wang, Y.; Fukuda, T.; Gu, J. A key regulator of cell adhesion: Identification and characterization of important N-Glycosylation Sites on Integrin α5 for Cell Migration. Mol. Cell Biol. 2017, 37. [Google Scholar] [CrossRef] [PubMed]
- Sonnenberg, A.; Calafat, J.; Janssen, H.; Daams, H.; van der Raaij-Helmer, L.M.; Falcioni, R.; Kennel, S.J.; Aplin, J.D.; Baker, J.; Loizidou, M.; et al. Integrin alpha 6/beta 4 complex is located in hemidesmosomes, suggesting a major role in epidermal cell-basement membrane adhesion. J. Cell Biol. 1991, 113, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Litjens, S.H.; de Pereda, J.M.; Sonnenberg, A. Current insights into the formation and breakdown of hemidesmosomes. Trends Cell Biol. 2006, 16, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Kariya, Y.; Gu, J. Laminin-332 and integrins: Signaling platform for cell adhesion and migration and its regulation by N-glycosylation. In Laminins: Structure, Biological Activity and Role in Disease; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2013; pp. 29–51. [Google Scholar]
- Takizawa, Y.; Shimizu, H.; Nishikawa, T.; Hatta, N.; Pulkkinen, L.; Uitto, J. Novel ITGB4 mutations in a patient with junctional epidermolysis bullosa-pyloric atresia syndrome and altered basement membrane zone immunofluorescence for the α6β4 integrin. J. Investig. Dermatol. 1997, 108, 943–946. [Google Scholar] [CrossRef] [PubMed]
- Ko, M.S.; Marinkovich, M.P. Role of dermal-epidermal basement membrane zone in skin, cancer, and developmental disorders. Dermatol. Clin. 2010, 28, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Pulkkinen, L.; Rouan, F.; Bruckner-Tuderman, L.; Wallerstein, R.; Garzon, M.; Brown, T.; Smith, L.; Carter, W.; Uitto, J. Novel ITGB4 mutations in lethal and nonlethal variants of epidermolysis bullosa with pyloric atresia: Missense versus nonsense. Am. J. Hum. Genet. 1998, 63, 1376–1387. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.J.; Fincher, E.F.; Millman, L.; Smith, R.; Vela, V.; Waterman, E.A.; Dey, C.N.; Guide, S.; Weaver, V.M.; Marinkovich, M.P. α6β4 integrin regulates keratinocyte chemotaxis through differential GTPase activation and antagonism of α3β1 integrin. J. Cell Sci. 2003, 116, 3543–3556. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Tsubota, Y.; Hirosaki, T.; Mizushima, H.; Puzon-McLaughlin, W.; Takada, Y.; Miyazaki, K. Differential regulation of cellular adhesion and migration by recombinant laminin-5 forms with partial deletion or mutation within the G3 domain of α3 chain. J. Cell Biochem. 2003, 88, 506–520. [Google Scholar] [CrossRef] [PubMed]
- Hogervorst, F.; Kuikman, I.; von dem Borne, A.E.; Sonnenberg, A. Cloning and sequence analysis of β4 cDNA: An integrin subunit that contains a unique 118 kd cytoplasmic domain. EMBO J. 1990, 9, 765–770. [Google Scholar] [PubMed]
- Murgia, C.; Blaikie, P.; Kim, N.; Dans, M.; Petrie, H.T.; Giancotti, F.G. Cell cycle and adhesion defects in mice carrying a targeted deletion of the integrin β4 cytoplasmic domain. EMBO J. 1998, 17, 3940–3951. [Google Scholar] [CrossRef] [PubMed]
- Mainiero, F.; Pepe, A.; Yeon, M.; Ren, Y.; Giancotti, F.G. The intracellular functions of α6β4 integrin are regulated by EGF. J. Cell Biol. 1996, 134, 241–253. [Google Scholar] [CrossRef] [PubMed]
- Rabinovitz, I.; Tsomo, L.; Mercurio, A.M. Protein kinase C-α phosphorylation of specific serines in the connecting segment of the β4 integrin regulates the dynamics of type II hemidesmosomes. Mol. Cell Biol. 2004, 24, 4351–4360. [Google Scholar] [CrossRef] [PubMed]
- Falcioni, R.; Kennel, S.J.; Giacomini, P.; Zupi, G.; Sacchi, A. Expression of tumor antigen correlated with metastatic potential of Lewis lung carcinoma and B16 melanoma clones in mice. Cancer Res. 1986, 46, 5772–5778. [Google Scholar] [PubMed]
- Kimmel, K.A.; Carey, T.E. Altered expression in squamous carcinoma cells of an orientation restricted epithelial antigen detected by monoclonal antibody A9. Cancer Res. 1986, 46, 3614–3623. [Google Scholar] [PubMed]
- Savoia, P.; Trusolino, L.; Pepino, E.; Cremona, O.; Marchisio, P.C. Expression and topography of integrins and basement membrane proteins in epidermal carcinomas: Basal but not squamous cell carcinomas display loss of α6β4 and BM-600/nicein. J. Investig. Dermatol. 1993, 101, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Mariani Costantini, R.; Falcioni, R.; Battista, P.; Zupi, G.; Kennel, S.J.; Colasante, A.; Venturo, I.; Curio, C.G.; Sacchi, A. Integrin (alph 6/beta 4) expression in human lung cancer as monitored by specific monoclonal antibodies. Cancer Res. 1990, 50, 6107–6112. [Google Scholar] [PubMed]
- Wolf, G.T.; Carey, T.E.; Schmaltz, S.P.; McClatchey, K.D.; Poore, J.; Glaser, L.; Hayashida, D.J.; Hsu, S. Altered antigen expression predicts outcome in squamous cell carcinoma of the head and neck. J. Natl. Cancer Inst. 1990, 82, 1566–1572. [Google Scholar] [CrossRef] [PubMed]
- Carico, E.; French, D.; Bucci, B.; Falcioni, R.; Vecchione, A.; Mariani-Costantini, R. Integrin β4 expression in the neoplastic progression of cervical epithelium. Gynecol. Oncol. 1993, 49, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Stewart, R.L.; O’Connor, K.L. Clinical significance of the integrin α6β4 in human malignancies. Lab. Investig. 2015, 95, 976–986. [Google Scholar] [CrossRef] [PubMed]
- Dajee, M.; Lazarov, M.; Zhang, J.Y.; Cai, T.; Green, C.L.; Russell, A.J.; Marinkovich, M.P.; Tao, S.; Lin, Q.; Kubo, Y.; et al. NF-κB blockade and oncogenic Ras trigger invasive human epidermal neoplasia. Nature 2003, 421, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Miyazaki, K. The basement membrane protein laminin-5 acts as a soluble cell motility factor. Exp. Cell Res. 2004, 297, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Nikolopoulos, S.N.; Blaikie, P.; Yoshioka, T.; Guo, W.; Puri, C.; Tacchetti, C.; Giancotti, F.G. Targeted deletion of the integrin β4 signaling domain suppresses laminin-5-dependent nuclear entry of mitogen-activated protein kinases and NF-κB, causing defects in epidermal growth and migration. Mol. Cell Biol. 2005, 25, 6090–6102. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.M.; Rabinovitz, I.; Wang, H.H.; Toker, A.; Mercurio, A.M. Activation of phosphoinositide 3-OH kinase by the α6β4 integrin promotes carcinoma invasion. Cell 1997, 91, 949–960. [Google Scholar] [CrossRef]
- Mainiero, F.; Murgia, C.; Wary, K.K.; Curatola, A.M.; Pepe, A.; Blumemberg, M.; Westwick, J.K.; Der, C.J.; Giancotti, F.G. The coupling of α6β4 integrin to Ras-MAP kinase pathways mediated by Shc controls keratinocyte proliferation. EMBO J. 1997, 16, 2365–2375. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Kariya, Y.; Gu, J. Roles of laminin-332 and α6β4 integrin in tumor progression. Mini Rev. Med. Chem. 2009, 9, 1284–1291. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.M. Identification of insulin receptor substrate 1 (IRS-1) and IRS-2 as signaling intermediates in the α6β4 integrin-dependent activation of phosphoinositide 3-OH kinase and promotion of invasion. Mol. Cell Biol. 2001, 21, 5082–5093. [Google Scholar] [CrossRef] [PubMed]
- Sacchi, A.; Falcioni, R.; Piaggio, G.; Gianfelice, M.A.; Perrotti, N.; Kennel, S.J. Ligand-induced phosphorylation of a murine tumor surface protein (TSP-180) associated with metastatic phenotype. Cancer Res. 1989, 49, 2615–2620. [Google Scholar] [PubMed]
- Trusolino, L.; Bertotti, A.; Comoglio, P.M. A signaling adapter function for α6β4 integrin in the control of HGF-dependent invasive growth. Cell 2001, 107, 643–654. [Google Scholar] [CrossRef]
- Bertotti, A.; Comoglio, P.M.; Trusolino, L. β4 integrin activates a Shp2-Src signaling pathway that sustains HGF-induced anchorage-independent growth. J. Cell Biol. 2006, 175, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Dans, M.; Gagnoux-Palacios, L.; Blaikie, P.; Klein, S.; Mariotti, A.; Giancotti, F.G. Tyrosine phosphorylation of the β4 integrin cytoplasmic domain mediates Shc signaling to extracellular signal-regulated kinase and antagonizes formation of hemidesmosomes. J. Biol. Chem. 2001, 276, 1494–1502. [Google Scholar] [CrossRef] [PubMed]
- Alosnso-Garcia, N.; Garcia-Rubio, I.; Manso, J.A.; Buey, R.M.; Urien, H.; Sonnenberg, A.; Jeschke, G.; de Pereda, J.M. Combination of X-ray crystallography, SAXS and DEER to obtain the structure of the FnIII-3,4 domains of integrin α6β4. Acta Crystallogr. D Biol. Crystallogr. 2015, 71, 969–985. [Google Scholar] [CrossRef] [PubMed]
- Ramovs, V.; Molder, L.T.; Sonnenberg, A. The opposing roles of laminin-binding integrins in cancer. Matrix Biol. 2017, 57–58, 213–243. [Google Scholar] [CrossRef] [PubMed]
- Rabinovitz, I.; Toker, A.; Mercurio, A.M. Protein kinase C-dependent mobilization of the α6β4 integrin from hemidesmosomes and its association with actin-rich cell protrusions drive the chemotactic migration of carcinoma cells. J. Cell Biol. 1999, 146, 1147–1160. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, T.; Germain, E.; Roche, M.; Lyle, S.; Rabinovitz, I. Role of β4 integrin phosphorylation in human invasive squamous cell carcinoma: Regulation of hemidesmosome stability modulates cell migration. Lab. Investig. 2011, 91, 1414–1426. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmsen, K.; Litjens, S.H.; Kuikman, I.; Margadant, C.; van Rheenen, J.; Sonnenberg, A. Serine phosphorylation of the integrin β4 subunit is necessary for epidermal growth factor receptor induced hemidesmosome disruption. Mol. Biol. Cell 2007, 18, 3512–3522. [Google Scholar] [CrossRef] [PubMed]
- Frijns, E.; Kuikman, I.; Litjens, S.; Raspe, M.; Jalink, K.; Ports, M.; Wilhelmsen, K.; Sonnenberg, A. Phosphorylation of threonine 1736 in the C-terminal tail of integrin β4 contributes to hemidesmosome disassembly. Mol. Biol. Cell 2012, 23, 1475–1485. [Google Scholar] [CrossRef] [PubMed]
- Mariotti, A.; Kedeshian, P.A.; Dans, M.; Curatola, A.M.; Gagnoux-Palacios, L.; Giancotti, F.G. EGF-R signaling through Fyn kinase disrupts the function of integrin α6β4 at hemidesmosomes: Role in epithelial cell migration and carcinoma invasion. J. Cell Biol. 2001, 155, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Pylayeva, Y.; Pepe, A.; Yoshioka, T.; Muller, W.J.; Inghirami, G.; Giancotti, F.G. β4 integrin amplifies ErbB2 signaling to promote mammary tumorigenesis. Cell 2006, 126, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Shibue, T.; Weinberg, R.A. EMT, CSCs, and drug resistance: The mechanistic link and clinical implications. Nat. Rev. Clin. Oncol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Masugi, Y.; Yamazaki, K.; Emoto, K.; Effendi, K.; Tsujikawa, H.; Kitago, M.; Itano, O.; Kitagawa, Y.; Sakamoto, M. Upregulation of integrin β4 promotes epithelial-mesenchymal transition and is a novel prognostic marker in pancreatic ductal adenocarcinoma. Lab. Investig. 2015, 95, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L.; Liu, L.; Li, D.D.; He, Y.P.; Guo, L.H.; Sun, L.P.; Liu, L.N.; Xu, H.X.; Zhang, X.P. Integrin β4 promotes cell invasion and epithelial-mesenchymal transition through the modulation of Slug expression in hepatocellular carcinoma. Sci. Rep. 2017, 7, 40464. [Google Scholar] [CrossRef] [PubMed]
- Bierie, B.; Pierce, S.E.; Kroeger, C.; Stover, D.G.; Pattabiraman, D.R.; Thiru, P.; Liu Donaher, J.; Reinhardt, F.; Chaffer, C.L.; Keckesova, Z.; et al. Integrin β4 identifies cancer stem cell-enriched populations of partially mesenchymal carcinoma cells. Proc. Natl. Acad. Sci. USA 2017, 114, E2337–E2346. [Google Scholar] [CrossRef] [PubMed]
- Drake, J.M.; Barnes, J.M.; Madsen, J.M.; Domann, F.E.; Stipp, C.S.; Henry, M.D. ZEB1 coordinately regulates laminin-332 and β4 integrin expression altering the invasive phenotype of prostate cancer cells. J. Biol. Chem. 2010, 285, 33940–33948. [Google Scholar] [CrossRef] [PubMed]
- Soung, Y.H.; Nguyen, T.; Cao, H.; Lee, J.; Chung, J. Emerging roles of exosomes in cancer invasion and metastasis. BMB Rep. 2016, 49, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, A.; Costa-Silva, B.; Shen, T.L.; Rodrigues, G.; Hashimoto, A.; Tesic Mark, M.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour exosome integrins determine organotropic metastasis. Nature 2015, 527, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Tamura, R.N.; Rozzo, C.; Starr, L.; Chambers, J.; Reichardt, L.F.; Cooper, H.M.; Quaranta, V. Epithelial integrin α6β4: Complete primary structure of α6 and variant forms of β4. J. Cell Biol. 1990, 111, 1593–1604. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Gu, J. N-glycosylation of β4 integrin controls the adhesion and motility of keratinocytes. PLoS ONE 2011, 6, e27084. [Google Scholar] [CrossRef] [PubMed]
- Isaji, T.; Sato, Y.; Zhao, Y.; Miyoshi, E.; Wada, Y.; Taniguchi, N.; Gu, J. N-glycosylation of the beta-propeller domain of the integrin α5 subunit is essential for α5β1 heterodimerization, expression on the cell surface, and its biological function. J. Biol. Chem. 2006, 281, 33258–33267. [Google Scholar] [CrossRef] [PubMed]
- Dennis, J.W.; Laferte, S. Oncodevelopmental expression of—GlcNAc beta 1-6Man alpha 1-6Man beta 1—Branched asparagine-linked oligosaccharides in murine tissues and human breast carcinomas. Cancer Res. 1989, 49, 945–950. [Google Scholar] [PubMed]
- Gu, J.; Isaji, T.; Sato, Y.; Kariya, Y.; Fukuda, T. Importance of N-glycosylation on α5β1 integrin for its biological functions. Biol. Pharm. Bull. 2009, 32, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Granovsky, M.; Fata, J.; Pawling, J.; Muller, W.J.; Khokha, R.; Dennis, J.W. Suppression of tumor growth and metastasis in Mgat5-deficient mice. Nat. Med. 2000, 6, 306–312. [Google Scholar] [PubMed]
- Zhao, Y.; Nakagawa, T.; Itoh, S.; Inamori, K.; Isaji, T.; Kariya, Y.; Kondo, A.; Miyoshi, E.; Miyazaki, K.; Kawasaki, N.; et al. N-acetylglucosaminyltransferase III antagonizes the effect of N-acetylglucosaminyltransferase V on α3β1 integrin-mediated cell migration. J. Biol. Chem. 2006, 281, 32122–32130. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Kato, R.; Itoh, S.; Fukuda, T.; Shibukawa, Y.; Sanzen, N.; Sekiguchi, K.; Wada, Y.; Kawasaki, N.; Gu, J. N-Glycosylation of laminin-332 regulates its biological functions: A novel function of the bisecting GlcNAc. J. Biol. Chem. 2008, 283, 33036–33045. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.B.; Lee, I.; Kamar, M.; Akiyama, S.K.; Pierce, M. Aberrant N-glycosylation of β1 integrin causes reduced α5β1 integrin clustering and stimulates cell migration. Cancer Res. 2002, 62, 6837–6845. [Google Scholar] [PubMed]
- Takenaka, Y.; Fukumori, T.; Raz, A. Galectin-3 and metastasis. Glycoconj. J. 2004, 19, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.T.; Rabinovich, G.A. Galectins as modulators of tumour progression. Nat. Rev. Cancer 2005, 5, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Wu, Q.; Ge, Y.; Zhang, J.; Sun, L.; Zhang, Y.; Fu, L.; Fan, J.; Wang, Z. Expression of N-acetylglucosaminyltransferase V in gastric cancer correlates with metastasis and prognosis. Int. J. Oncol. 2014, 44, 849–857. [Google Scholar] [PubMed]
- Kariya, Y.; Kawamura, C.; Tabei, T.; Gu, J. Bisecting GlcNAc residues on laminin-332 downregulate galectin-3-dependent keratinocyte motility. J. Biol. Chem. 2010, 285, 3330–3340. [Google Scholar] [CrossRef] [PubMed]
- Brockhausen, I. Mucin-type O-glycans in human colon and breast cancer: Glycodynamics and functions. EMBO Rep. 2006, 7, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Uemura, T.; Shiozaki, K.; Yamaguchi, K.; Miyazaki, S.; Satomi, S.; Kato, K.; Sakuraba, H.; Miyagi, T. Contribution of sialidase NEU1 to suppression of metastasis of human colon cancer cells through desialylation of integrin β4. Oncogene 2009, 28, 1218–1229. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Hong, S.; Dong, L.; Cheng, B.; Lin, L.; Zhao, B.; Chen, Y.G.; Chen, X. Dynamic Sialylation in Transforming Growth Factor-β (TGF-β)-induced Epithelial to Mesenchymal Transition. J. Biol. Chem. 2015, 290, 12000–12013. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmsen, K.; Litjens, S.H.; Sonnenberg, A. Multiple functions of the integrin α6β4 in epidermal homeostasis and tumorigenesis. Mol. Cell Biol. 2006, 26, 2877–2886. [Google Scholar] [CrossRef] [PubMed]
- Koster, J.; Geerts, D.; Favre, B.; Borradori, L.; Sonnenberg, A. Analysis of the interactions between BP180, BP230, plectin and the integrin α6β4 important for hemidesmosome assembly. J. Cell Sci. 2003, 116, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Schaapveld, R.Q.; Borradori, L.; Geerts, D.; van Leusden, M.R.; Kuikman, I.; Nievers, M.G.; Niessen, C.M.; Steenbergen, R.D.; Snijders, P.J.; Sonnenberg, A. Hemidesmosome formation is initiated by the β4 integrin subunit, requires complex formation of β4 and HD1/plectin, and involves a direct interaction between β4 and the bullous pemphigoid antigen 180. J. Cell Biol. 1998, 142, 271–284. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kariya, Y.; Kariya, Y.; Gu, J. Roles of Integrin α6β4 Glycosylation in Cancer. Cancers 2017, 9, 79. https://doi.org/10.3390/cancers9070079
Kariya Y, Kariya Y, Gu J. Roles of Integrin α6β4 Glycosylation in Cancer. Cancers. 2017; 9(7):79. https://doi.org/10.3390/cancers9070079
Chicago/Turabian StyleKariya, Yoshinobu, Yukiko Kariya, and Jianguo Gu. 2017. "Roles of Integrin α6β4 Glycosylation in Cancer" Cancers 9, no. 7: 79. https://doi.org/10.3390/cancers9070079
APA StyleKariya, Y., Kariya, Y., & Gu, J. (2017). Roles of Integrin α6β4 Glycosylation in Cancer. Cancers, 9(7), 79. https://doi.org/10.3390/cancers9070079





