Focal Therapy Using High-Intensity Focused Ultrasound for Low- and Intermediate-Risk Prostate Cancer: Results from a Prospective, Multicenter Feasibility Trial
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Pre-Treatment
2.2. Treatment Protocol
2.3. Adverse Events and Salvage Therapy
2.4. Follow-Up
2.5. Statistical Analysis
3. Results
3.1. Safety
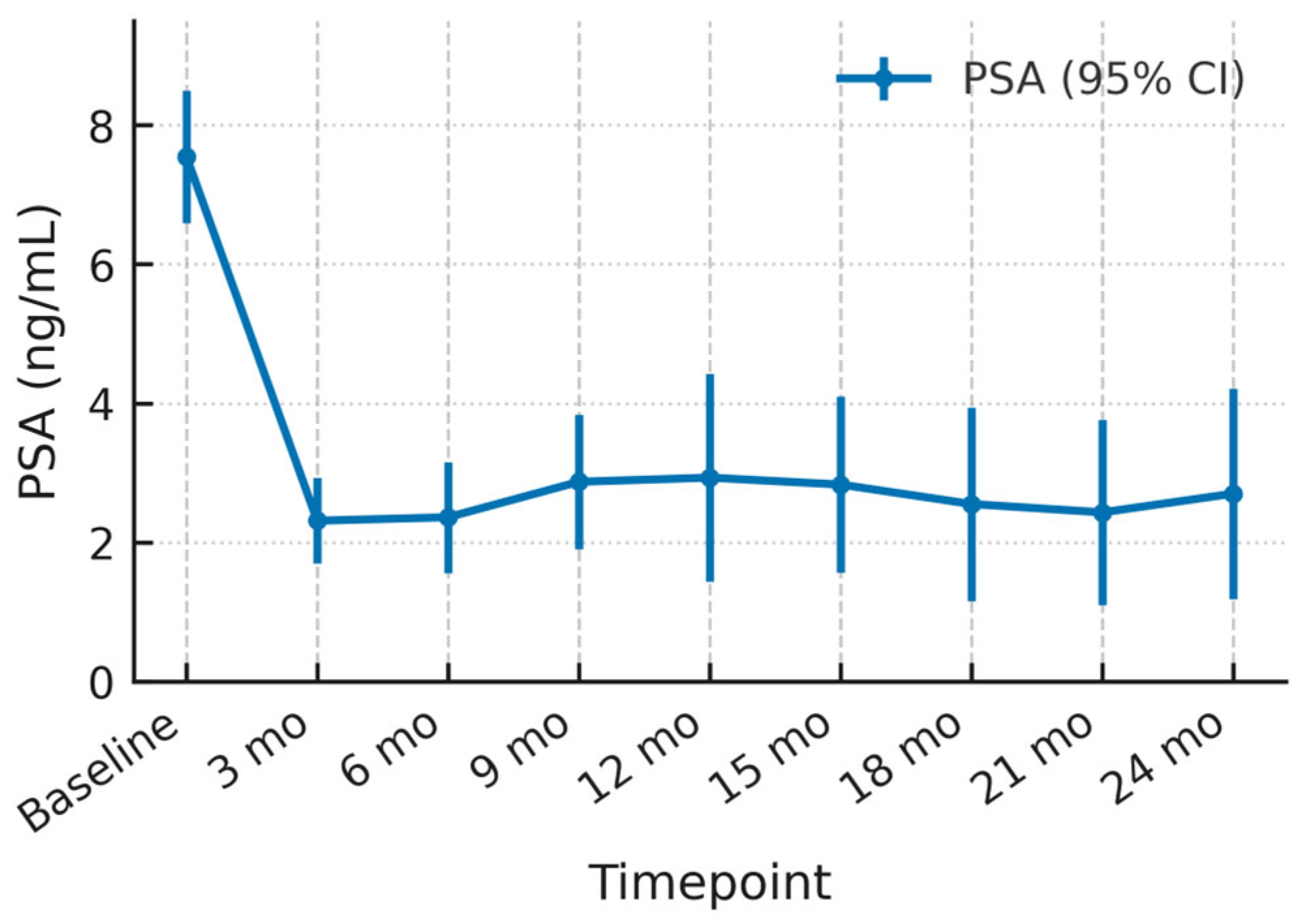
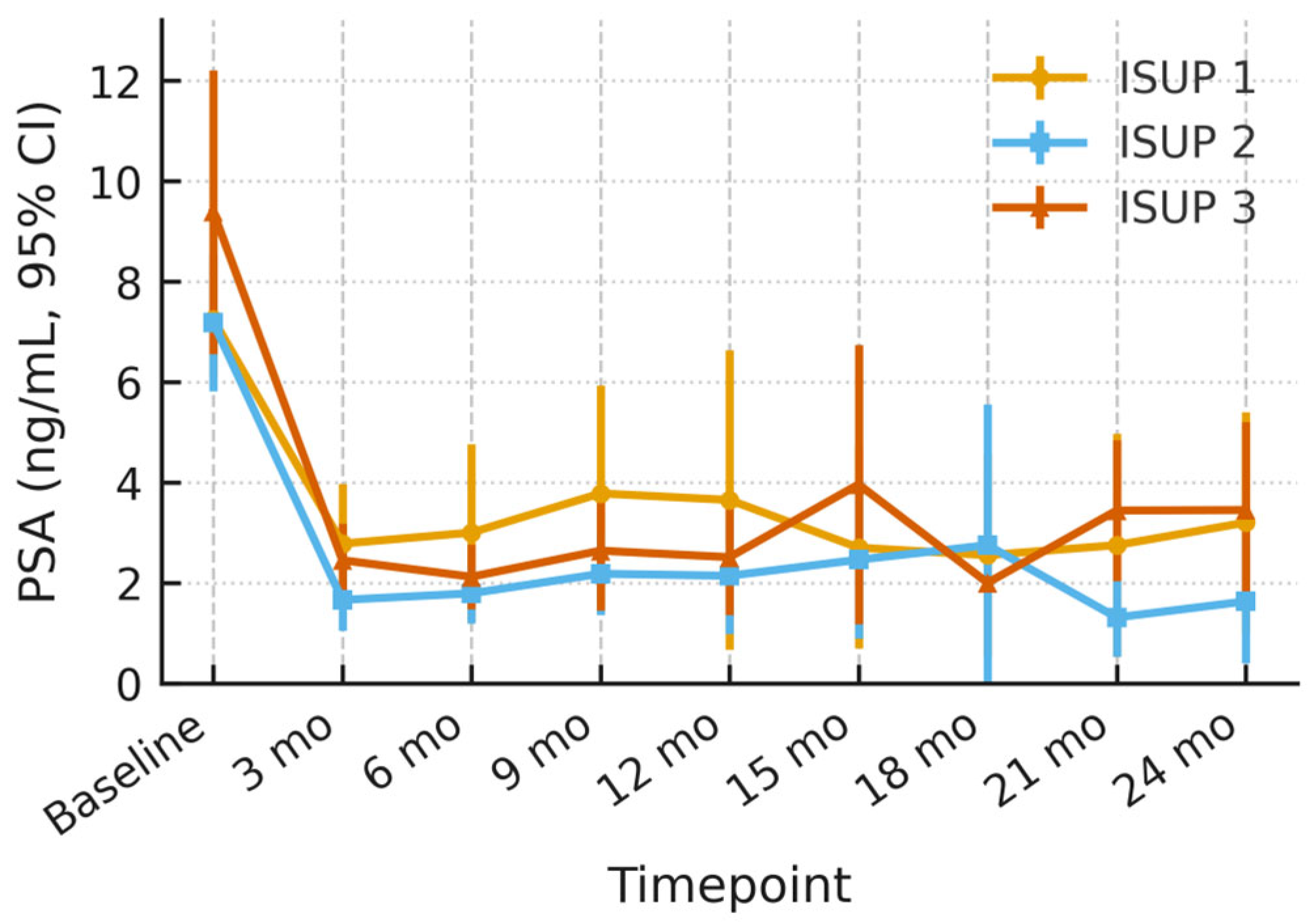
3.2. PSA Dynamics
3.3. Imaging Follow-Up
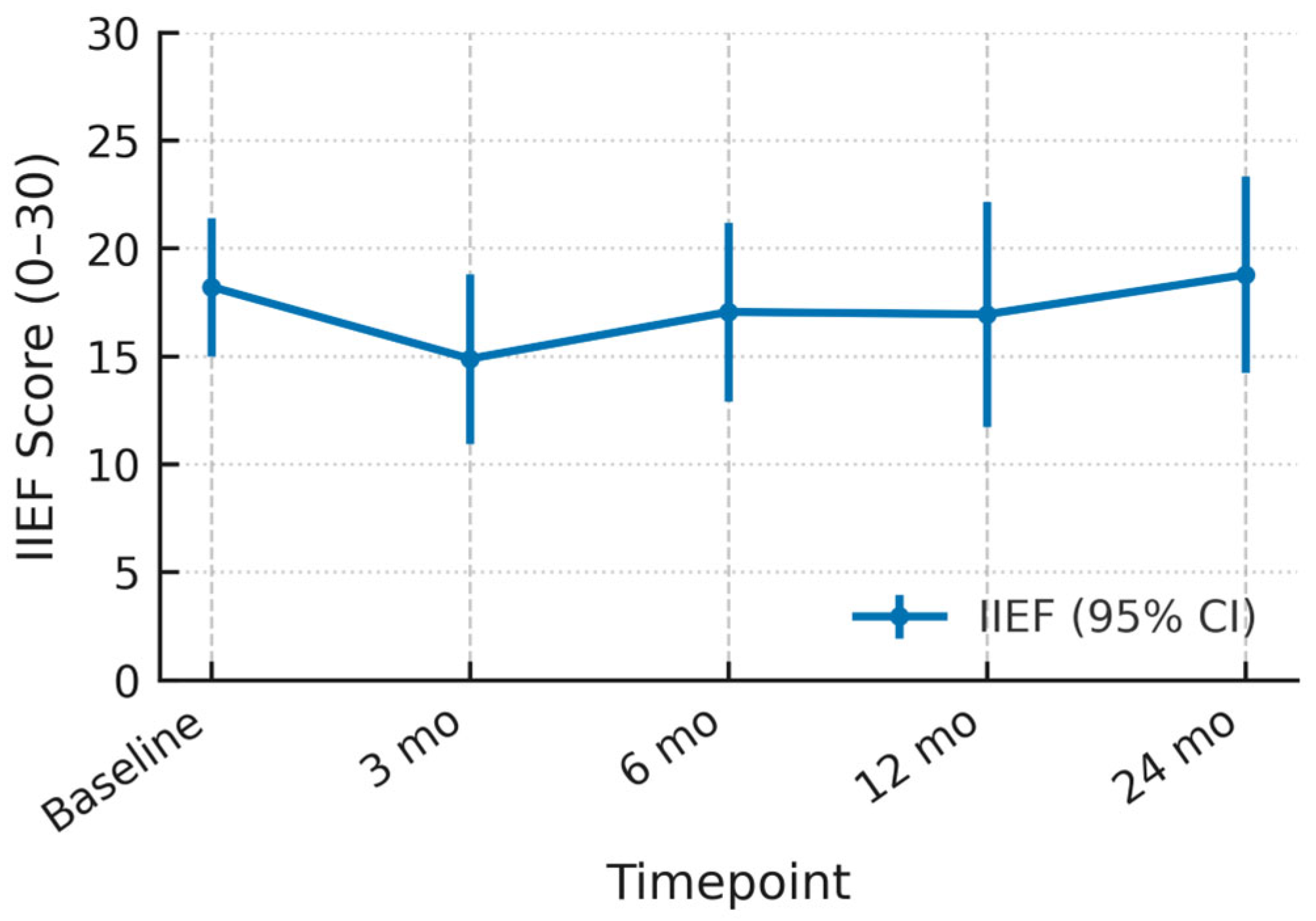
3.4. Functional Outcomes
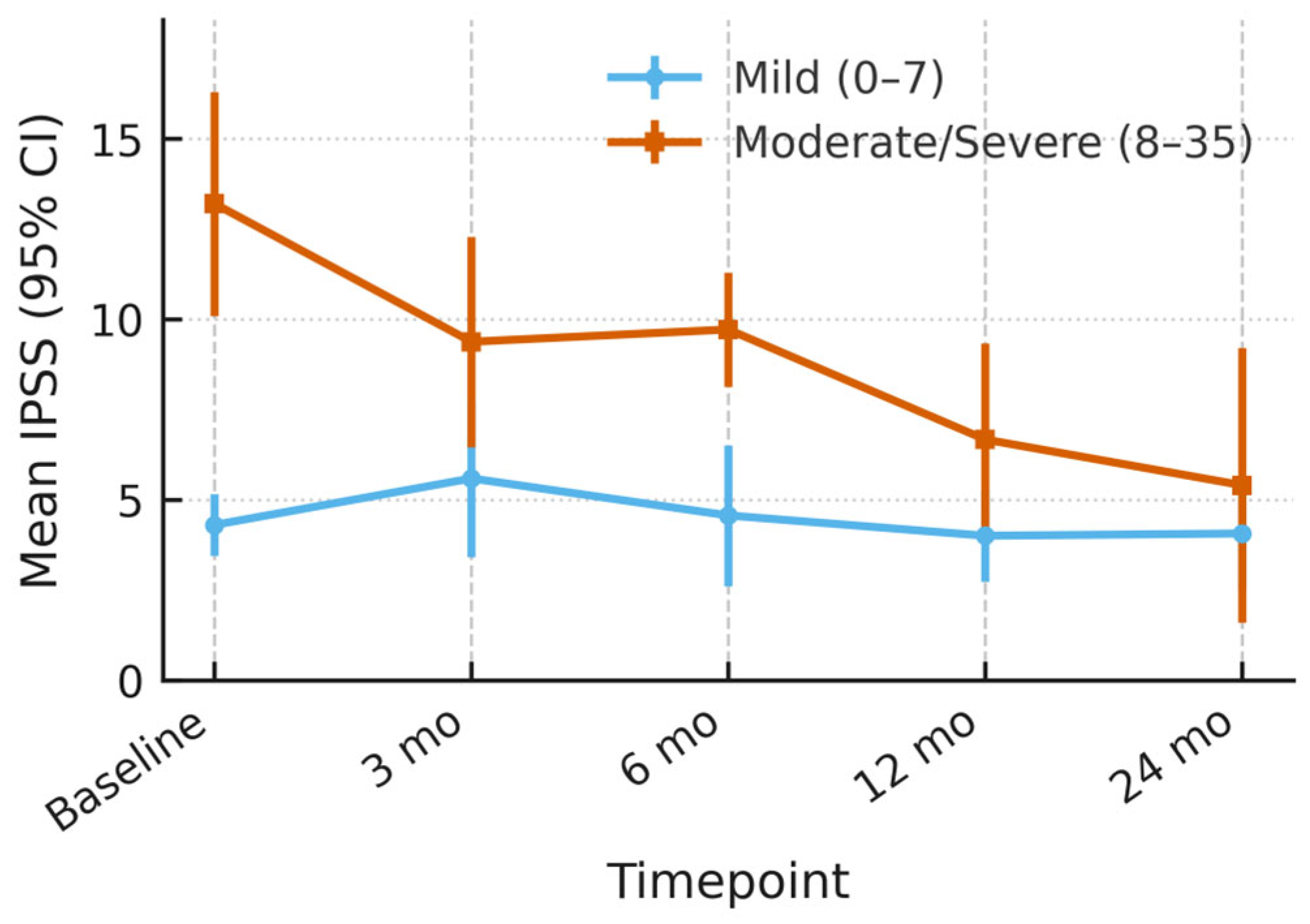
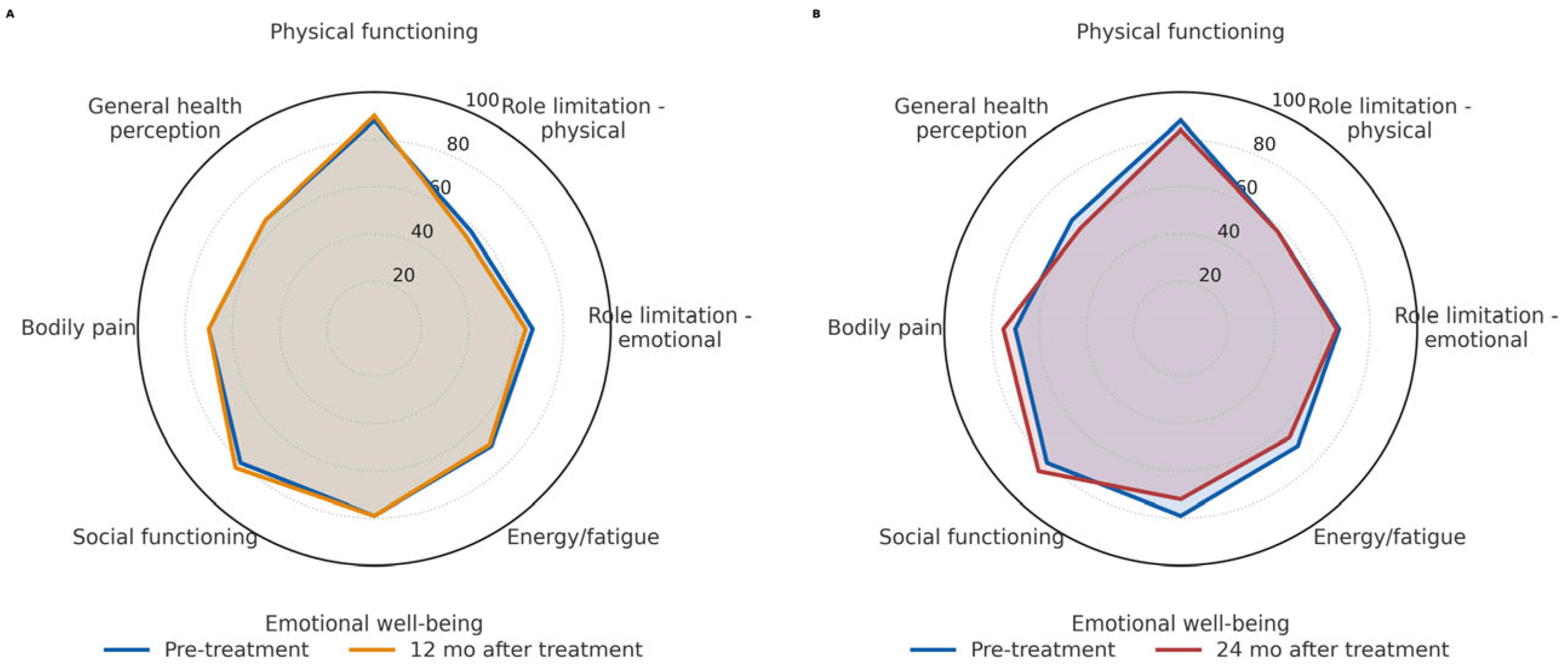
3.5. Outcome of Quality of Life
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HIFU | High-Intensity Focused Ultrasound |
| PCa | Prostate Cancer |
| mpMRI | Multiparametric Magnet Resonance Imaging |
| FFS | Failure Free Survival |
| IIEF | International Index of Erectile Function |
| IPSS | International Prostate Symptom Score |
| AEs | Adverse Event |
| FT | Focal Therapy |
| RP | Radical Prostatectomy |
| Rt | Radiotherapy |
| IQR | Interquartile Range |
| PI-RADS | Prostate Imaging Reporting and Data System |
| SF-36 | Medical Outcomes Study Short Form-36 |
| 5ARI | 5-alpha reductase inhibitor |
| CEUS | Contrast-Enhanced Ultrasound |
| LUTS | Lower Urinary Tract Symptoms |
| TPLA | Transperineal Laser Ablation of the Prostate |
References
- James, N.D.; Tannock, I.; N’Dow, J.; Feng, F.; Cillessen, S.; Ali, S.A.; Trujillo, B.; Al-Lazikani, B.; Attard, G.; Bray, F.; et al. The Lancet Commission on prostate cancer: Planning for the surge in cases. Lancet 2024, 403, 1683–1722. [Google Scholar] [CrossRef]
- Hopstaken, J.S.; Bomers, J.G.R.; Sedelaar, M.J.P.; Valerio, M.; Fütterer, J.J.; Rovers, M.M. An Updated Systematic Review on Focal Therapy in Localized Prostate Cancer: What Has Changed over the Past 5 Years? Eur. Urol. 2022, 81, 5–33. [Google Scholar] [CrossRef]
- Mouraviev, V.; Mayes, J.M.; Polascik, T.J. Pathologic basis of focal therapy for early-stage prostate cancer. Nat. Rev. Urol. 2009, 6, 205–215. [Google Scholar] [CrossRef]
- de Rooij, M.; Hamoen, E.H.; Witjes, J.A.; Barentsz, J.O.; Rovers, M.M. Accuracy of Magnetic Resonance Imaging for Local Staging of Prostate Cancer: A Diagnostic Meta-analysis. Eur. Urol. 2016, 70, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.U.; Freeman, A.; Kirkham, A.; Sahu, M.; Scott, R.; Allen, C.; Van der Meulen, J.; Emberton, M. Focal therapy for localized prostate cancer: A phase I/II trial. J. Urol. 2011, 185, 1246–1254. [Google Scholar] [CrossRef] [PubMed]
- Hübner, N.; Shariat, S.F.; Remzi, M. Focal therapy of prostate cancer. Curr. Opin. Urol. 2018, 28, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Ghai, S.; Finelli, A.; Corr, K.; Chan, R.; Jokhu, S.; Li, X.; McCluskey, S.; Konukhova, A.; Hlasny, E.; van der Kwast, T.H.; et al. MRI-guided Focused Ultrasound Ablation for Localized Intermediate-Risk Prostate Cancer: Early Results of a Phase II Trial. Radiology 2021, 298, 695–703. [Google Scholar] [CrossRef]
- Polascik, T.J.; Mayes, J.M.; Sun, L.; Madden, J.F.; Moul, J.W.; Mouraviev, V. Pathologic stage T2a and T2b prostate cancer in the recent prostate-specific antigen era: Implications for unilateral ablative therapy. Prostate 2008, 68, 1380–1386. [Google Scholar] [CrossRef]
- Reddy, D.; Peters, M.; Shah, T.T.; van Son, M.; Tanaka, M.B.; Huber, P.M.; Lomas, D.; Rakauskas, A.; Miah, S.; Eldred-Evans, D.; et al. Cancer Control Outcomes Following Focal Therapy Using High-intensity Focused Ultrasound in 1379 Men with Nonmetastatic Prostate Cancer: A Multi-institute 15-year Experience. Eur. Urol. 2022, 81, 407–413. [Google Scholar] [CrossRef]
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2021, 79, 243–262. [Google Scholar] [CrossRef]
- van der Poel, H.G.; van den Bergh, R.C.N.; Briers, E.; Cornford, P.; Govorov, A.; Henry, A.M.; Lam, T.B.; Mason, M.D.; Rouvière, O.; De Santis, M.; et al. Focal Therapy in Primary Localised Prostate Cancer: The European Association of Urology Position in 2018. Eur. Urol. 2018, 74, 84–91. [Google Scholar] [CrossRef]
- Ramsay, C.R.; Adewuyi, T.E.; Gray, J.; Hislop, J.; Shirley, M.D.; Jayakody, S.; MacLennan, G.; Fraser, C.; MacLennan, S.; Brazzelli, M.; et al. Ablative therapy for people with localised prostate cancer: A systematic review and economic evaluation. Health Technol. Assess. 2015, 19, 1–490. [Google Scholar] [CrossRef] [PubMed]
- Marra, G.; Ploussard, G.; Ost, P.; De Visschere, P.J.L.; Briganti, A.; Gandaglia, G.; Tilki, D.; Surcel, C.I.; Tsaur, I.; Van Den Bergh, R.C.N.; et al. Focal therapy in localised prostate cancer: Real-world urological perspective explored in a cross-sectional European survey. Urol. Oncol. 2018, 36, 529.e11–529.e22. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, A.V.; Whittington, R.; Malkowicz, S.B.; Schultz, D.; Blank, K.; Broderick, G.A.; Tomaszewski, J.E.; Renshaw, A.A.; Kaplan, I.; Beard, C.J.; et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 1998, 280, 969–974. [Google Scholar] [CrossRef]
- Turkbey, B.; Rosenkrantz, A.B.; Haider, M.A.; Padhani, A.R.; Villeirs, G.; Macura, K.J.; Tempany, C.M.; Choyke, P.L.; Cornud, F.; Margolis, D.J.; et al. Prostate Imaging Reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur. Urol. 2019, 76, 340–351. [Google Scholar] [CrossRef] [PubMed]
- von Hardenberg, J.; Westhoff, N.; Baumunk, D.; Hausmann, D.; Martini, T.; Marx, A.; Porubsky, S.; Schostak, M.; Michel, M.S.; Ritter, M. Prostate cancer treatment by the latest focal HIFU device with MRI/TRUS-fusion control biopsies: A prospective evaluation. Urol. Oncol. 2018, 36, 401.e1–401.e9. [Google Scholar] [CrossRef]
- Gaudenz, R. A questionnaire with a new urge-score and stress-score for the evaluation of female urinary incontinence (author’s transl). Geburtshilfe Frauenheilkd 1979, 39, 784–792. [Google Scholar]
- Jenkinson, C.; Coulter, A.; Wright, L. Short form 36 (SF36) health survey questionnaire: Normative data for adults of working age. BMJ 1993, 306, 1437–1440. [Google Scholar] [CrossRef]
- Clavien, P.A.; Barkun, J.; de Oliveira, M.L.; Vauthey, J.N.; Dindo, D.; Schulick, R.D.; de Santibañes, E.; Pekolj, J.; Slankamenac, K.; Bassi, C.; et al. The Clavien-Dindo classification of surgical complications: Five-year experience. Ann. Surg. 2009, 250, 187–196. [Google Scholar] [CrossRef]
- Lovegrove, C.E.; Peters, M.; Guillaumier, S.; Arya, M.; Afzal, N.; Dudderidge, T.; Hosking-Jervis, F.; Hindley, R.G.; Lewi, H.; McCartan, N.; et al. Evaluation of functional outcomes after a second focal high-intensity focused ultrasonography (HIFU) procedure in men with primary localized, non-metastatic prostate cancer: Results from the HIFU Evaluation and Assessment of Treatment (HEAT) registry. BJU Int. 2020, 125, 853–860. [Google Scholar] [CrossRef]
- van den Bos, W.; Muller, B.G.; Ahmed, H.; Bangma, C.H.; Barret, E.; Crouzet, S.; Eggener, S.E.; Gill, I.S.; Joniau, S.; Kovacs, G.; et al. Focal therapy in prostate cancer: International multidisciplinary consensus on trial design. Eur. Urol. 2014, 65, 1078–1083. [Google Scholar] [CrossRef]
- Dickinson, L.; Arya, M.; Afzal, N.; Cathcart, P.; Charman, S.C.; Cornaby, A.; Hindley, R.G.; Lewi, H.; McCartan, N.; Moore, C.M.; et al. Medium-term Outcomes after Whole-gland High-intensity Focused Ultrasound for the Treatment of Nonmetastatic Prostate Cancer from a Multicentre Registry Cohort. Eur. Urol. 2016, 70, 668–674. [Google Scholar] [CrossRef]
- Kaufmann, B.; Raess, E.; Schmid, F.A.; Bieri, U.; Scherer, T.P.; Elleisy, M.; Donati, O.F.; Rupp, N.J.; Moch, H.; Gorin, M.A.; et al. Focal therapy with high-intensity focused ultrasound for prostate cancer: 3-year outcomes from a prospective trial. BJU Int. 2024, 133, 413–424. [Google Scholar] [CrossRef]
- Mala, K.S.; Plage, H.; Mödl, L.; Hofbauer, S.; Friedersdorff, F.; Schostak, M.; Miller, K.; Schlomm, T.; Cash, H. Follow-Up of Men Who Have Undergone Focal Therapy for Prostate Cancer with HIFU-A Real-World Experience. J. Clin. Med. 2023, 12, 7089. [Google Scholar] [CrossRef]
- van Velthoven, R.; Aoun, F.; Marcelis, Q.; Albisinni, S.; Zanaty, M.; Lemort, M.; Peltier, A.; Limani, K. A prospective clinical trial of HIFU hemiablation for clinically localized prostate cancer. Prostate Cancer Prostatic Dis. 2016, 19, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Ladjevardi, S.; Ebner, A.; Femic, A.; Huebner, N.A.; Shariat, S.F.; Kraler, S.; Kubik-Huch, R.A.; Ahlman, R.C.; Häggman, M.; Hefermehl, L.J. Focal high-intensity focused ultrasound therapy for localized prostate cancer: An interim analysis of the multinational FASST study. Eur. J. Clin. Investig. 2024, 54, e14192. [Google Scholar] [CrossRef]
- Babalola, O.; Gebben, D.; Tarver, M.E.; Joyce Lee, T.H.; Wang, S.; Siddiqui, M.M.; Sonn, G.A.; Viviano, C.J. Patient Preferences for Benefit and Risk Associated With High Intensity Focused Ultrasound for the Ablation of Prostate Tissue in Men With Localized Prostate Cancer. Clin. Genitourin. Cancer 2024, 22, 102113. [Google Scholar] [CrossRef] [PubMed]
- Madersbacher, S.; Kratzik, C.; Szabo, N.; Susani, M.; Vingers, L.; Marberger, M. Tissue ablation in benign prostatic hyperplasia with high-intensity focused ultrasound. Eur. Urol. 1993, 23 (Suppl. S1), 39–43. [Google Scholar] [CrossRef] [PubMed]
- Mearini, L.; Nunzi, E.; Giovannozzi, S.; Lepri, L.; Lolli, C.; Giannantoni, A. Urodynamic evaluation after high-intensity focused ultrasound for patients with prostate cancer. Prostate Cancer 2014, 2014, 462153. [Google Scholar] [CrossRef]
- Altieri, V.M.; Di Bello, F.; Saldutto, P.; Rocca, R.L.; Romani, M.L.; Vena, W.; Mauro, E.D.; Verratti, V.; Sangiorgi, G.; Lacono, F.; et al. Outcomes and safety of trans perineal laser ablation of the prostate: A systematic review. World J. Urol. 2025, 43, 385. [Google Scholar] [CrossRef]
- Duwe, G.; Boehm, K.; Haack, M.; Sparwasser, P.; Brandt, M.P.; Mager, R.; Tsaur, I.; Haferkamp, A.; Hofner, T. Single-center, prospective phase 2 trial of high-intensity focused ultrasound (HIFU) in patients with unilateral localized prostate cancer: Good functional results but oncologically not as safe as expected. World J. Urol. 2023, 41, 1293–1299. [Google Scholar] [CrossRef]
- Ziglioli, F.; Baciarello, M.; Maspero, G.; Bellini, V.; Bocchialini, T.; Cavalieri, D.; Bignami, E.G.; Maestroni, U. Oncologic outcome, side effects and comorbidity of high-intensity focused ultrasound (HIFU) for localized prostate cancer. A review. Ann. Med. Surg. 2020, 56, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Grivas, N.; Eberli, D.; van der Poel, H. How Sound Are High-intensity Focused Ultrasound Data? Eur. Urol. 2025, 87, 534–535. [Google Scholar] [CrossRef] [PubMed]
- Giganti, F.; Dickinson, L.; Orczyk, C.; Haider, A.; Freeman, A.; Emberton, M.; Allen, C.; Moore, C.M. Prostate Imaging after Focal Ablation (PI-FAB): A Proposal for a Scoring System for Multiparametric MRI of the Prostate After Focal Therapy. Eur. Urol. Oncol. 2023, 6, 629–634. [Google Scholar] [CrossRef] [PubMed]
| Included Patients (n = 51) | |
|---|---|
| Patient excluded | 1 |
| Age (years), median (IQR) | 66.9 (60.5–73.5) |
| PSA (ng/mL), median (IQR) | 7.55 (5.4–9.4) |
| Prostate volume, mL, median (IQR) | 39.5 (29–50) |
| ISUP | |
| 1 | 20 (38%) |
| 2 | 25 (49%) |
| 3 | 6 (12%) |
| mpMRI lesion (PI-RADS score) | |
| 3 | 6 (12%) |
| 4 | 28 (55%) |
| 5 | 16 (31%) |
| Clinical Stage | |
| T1c | 37 (73%) |
| T2 | 8 (16%) |
| Medical history | |
| 5ARI | 5 (10%) |
| Alpha-blocker | 11 (22%) |
| Anticoagulation | 18 (35%) |
| Prior prostate surgery (TUR/P) | 1 (2%) |
| Charlson Comorbidity Index, mean (IQR) | 4.2 (3–8) |
| Smoker (actively) | 12 (24%) |
| Diabetes | 9 (18%) |
| Positive family history of prostate cancer | 5 (10%) |
| All (n = 51) | |
|---|---|
| Target volume, mL—median (IQR) | 12.2 (9.9–13.0) |
| Procedure time, min—median (IQR) | 84.6 (70.5–96.5) |
| Intraoperative CEUS application | 18 (35%) |
| Foley catheter duration, days—median (IQR) | 5 (3–5) |
| All (n = 51) | |
|---|---|
| Any kind of AE | 14 (27%) |
| Hospital admission ≤ 30 days post-HIFU | 1 (2%) |
| Transient acute urinary retention | 11 (22%) |
| With transurethral catheter placement | 5 (10%) |
| With suprapubic catheter placement 1 | 6 (12%) |
| With post-HIFU TURP for deobstruction | 2 (4%) |
| Urinary Tract Infection | 3 (6%) |
| Epididymitis | 2 (4%) |
| Bladder Cancer | 1 (2%) |
| Incontinence | 1 (2%) |
| Death | 2 (4%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosta, G.; Turba, S.; Mun, D.-H.; Shehab, A.; Saciri, L.; Engelhardt, P.F.; Weisz, P.; Riedl, C.; Ameli, G.; Doblhammer, S.; et al. Focal Therapy Using High-Intensity Focused Ultrasound for Low- and Intermediate-Risk Prostate Cancer: Results from a Prospective, Multicenter Feasibility Trial. Cancers 2025, 17, 3429. https://doi.org/10.3390/cancers17213429
Rosta G, Turba S, Mun D-H, Shehab A, Saciri L, Engelhardt PF, Weisz P, Riedl C, Ameli G, Doblhammer S, et al. Focal Therapy Using High-Intensity Focused Ultrasound for Low- and Intermediate-Risk Prostate Cancer: Results from a Prospective, Multicenter Feasibility Trial. Cancers. 2025; 17(21):3429. https://doi.org/10.3390/cancers17213429
Chicago/Turabian StyleRosta, Gabor, Simon Turba, Dong-Ho Mun, Azad Shehab, Leon Saciri, Paul F. Engelhardt, Patricia Weisz, Claus Riedl, Ghazal Ameli, Stephan Doblhammer, and et al. 2025. "Focal Therapy Using High-Intensity Focused Ultrasound for Low- and Intermediate-Risk Prostate Cancer: Results from a Prospective, Multicenter Feasibility Trial" Cancers 17, no. 21: 3429. https://doi.org/10.3390/cancers17213429
APA StyleRosta, G., Turba, S., Mun, D.-H., Shehab, A., Saciri, L., Engelhardt, P. F., Weisz, P., Riedl, C., Ameli, G., Doblhammer, S., & Fajkovic, H. (2025). Focal Therapy Using High-Intensity Focused Ultrasound for Low- and Intermediate-Risk Prostate Cancer: Results from a Prospective, Multicenter Feasibility Trial. Cancers, 17(21), 3429. https://doi.org/10.3390/cancers17213429






