Frequency and Significance of Body Weight Loss During Immunochemotherapy in Patients with Advanced Non-Small Cell Lung Cancer
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Outcome and Exposure
2.3. Statistical Analysis
3. Results
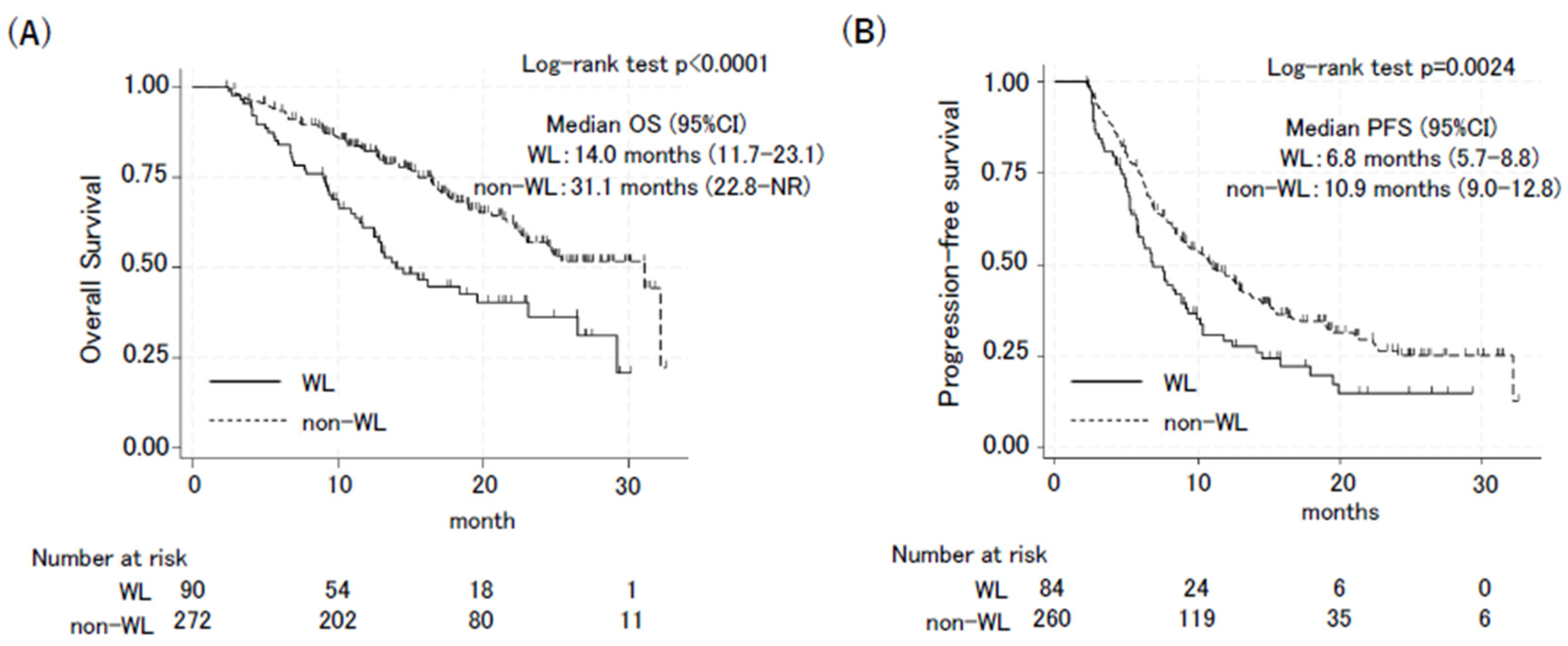
3.1. More Than One-Third of the Patients Significantly Lost Weight During Immunochemotherapy
3.2. Body Weight Loss During Treatment Negatively Affected Outcomes of ICI Plus Chemotherapy
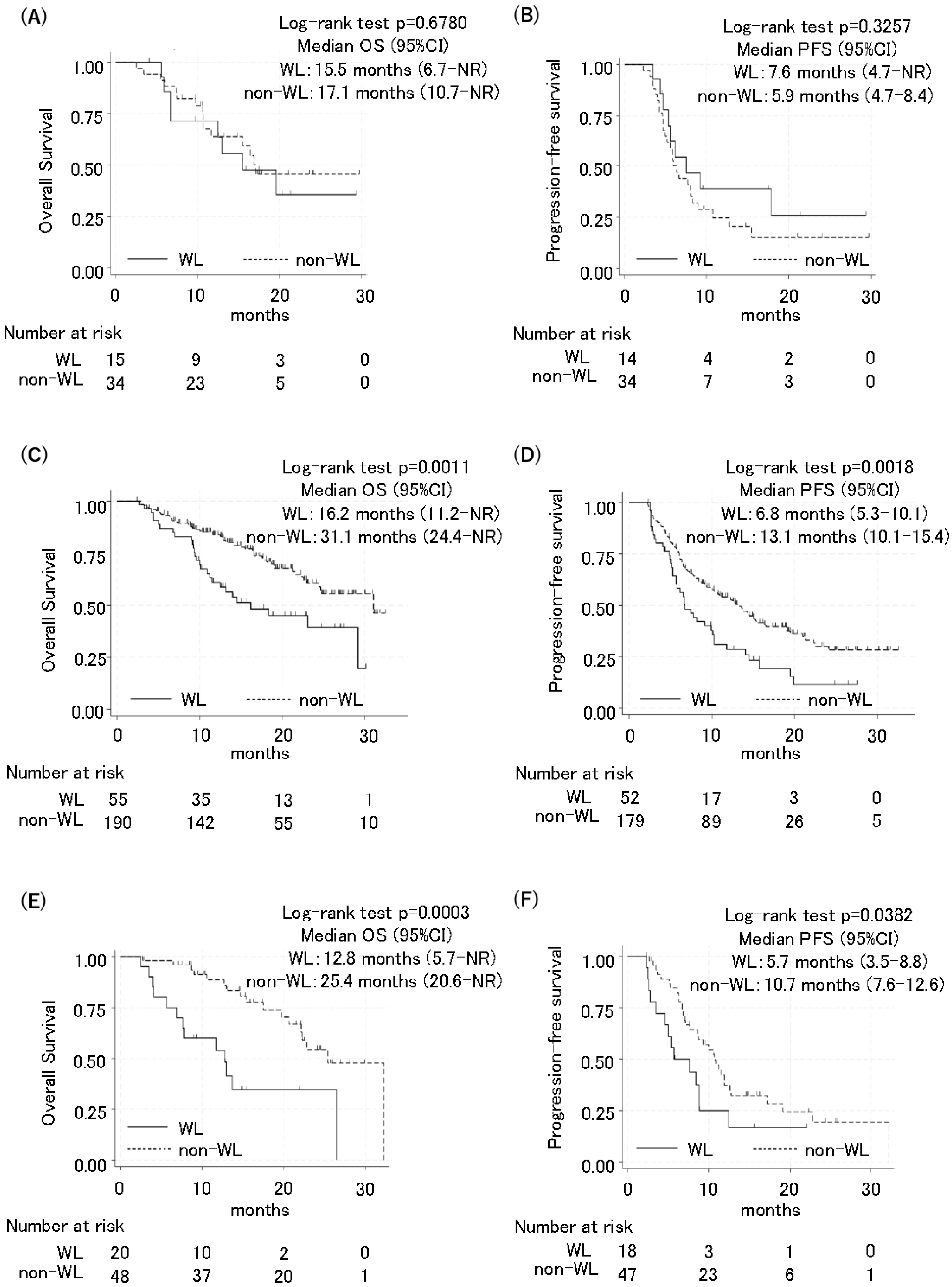
3.3. Poor Prognosis Due to Weight Loss Was Also Observed in Patients with Standard Weight and Obesity
3.4. Frequency of Most Immune-Related Adverse Events Was Not Different Between Groups
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gandhi, L.; Rodríguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab Plus Chemotherapy in Metastatic Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Luft, A.; Vicente, D.; Tafreshi, A.; Gümüş, M.; Mazières, J.; Hermes, B.; Çay Şenler, F.; Csőszi, T.; Fülöp, A.; et al. Pembrolizumab Plus Chemotherapy for Squamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2040–2051. [Google Scholar] [CrossRef]
- Socinski, M.A.; Jotte, R.M.; Cappuzzo, F.; Orlandi, F.; Stroyakovskiy, D.; Nogami, N.; Rodríguez-Abreu, D.; Moro-Sibilot, D.; Thomas, C.A.; Barlesi, F.; et al. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N. Engl. J. Med. 2018, 378, 2288–2301. [Google Scholar] [CrossRef]
- West, H.; McCleod, M.; Hussein, M.; Morabito, A.; Rittmeyer, A.; Conter, H.J.; Kopp, H.G.; Daniel, D.; McCune, S.; Mekhail, T.; et al. Atezolizumab in Combination with Carboplatin Plus Nab-Paclitaxel Chemotherapy Compared with Chemotherapy Alone as First-Line Treatment for Metastatic Non-Squamous Non-Small-Cell Lung Cancer (IMpower130): A Multicentre, Randomised, Open-Label, Phase 3 Trial. Lancet Oncol. 2019, 20, 924–937. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Ciuleanu, T.E.; Cobo, M.; Schenker, M.; Zurawski, B.; Menezes, J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O.; et al. First-Line Nivolumab Plus Ipilimumab Combined with Two Cycles of Chemotherapy in Patients with Non-Small-Cell Lung Cancer (CheckMate 9LA): An International, Randomised, Open-Label, Phase 3 Trial. Lancet Oncol. 2021, 22, 198–211. [Google Scholar] [CrossRef]
- Gogishvili, M.; Melkadze, T.; Makharadze, T.; Giorgadze, D.; Dvorkin, M.; Penkov, K.; Laktionov, K.; Nemsadze, G.; Nechaeva, M.; Rozhkova, I.; et al. Cemiplimab Plus Chemotherapy versus Chemotherapy Alone in Non-Small Cell Lung Cancer: A Randomized, Controlled, Double-Blind Phase 3 Trial. Nat. Med. 2022, 28, 2374–2380. [Google Scholar] [CrossRef]
- Turner, D.C.; Kondic, A.G.; Anderson, K.M.; Robinson, A.G.; Garon, E.B.; Riess, J.W.; Jain, L.; Mayawala, K.; Kang, J.; Ebbinghaus, S.W.; et al. Pembrolizumab Exposure-Response Assessments Challenged by Association of Cancer Cachexia and Catabolic Clearance. Clin. Cancer Res. 2018, 24, 5841–5849. [Google Scholar] [CrossRef]
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.; Bruera, E.; Fainsinger, R.L.; Jatoi, A.; Loprinzi, C.; MacDonald, N.; Mantovani, G.; et al. Definition and Classification of Cancer Cachexia: An International Consensus. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Scott, H.R.; McMillan, D.C.; Forrest, L.M.; Brown, D.J.F.; McArdle, C.S.; Milroy, R. The Systemic Inflammatory Response, Weight Loss, Performance Status and Survival in Patients with Inoperable Non-Small Cell Lung Cancer. Br. J. Cancer 2002, 87, 264–267. [Google Scholar] [CrossRef]
- Shepshelovich, D.; Xu, W.; Lu, L.; Fares, A.; Yang, P.; Christiani, D.; Zhang, J.; Shiraishi, K.; Ryan, B.M.; Chen, C.; et al. Body Mass Index (BMI), BMI Change, and Overall Survival in Patients with SCLC and NSCLC: A Pooled Analysis of the International Lung Cancer Consortium. J. Thorac. Oncol. 2019, 14, 1594–1607. [Google Scholar] [CrossRef]
- Degens, J.H.R.J.; Dingemans, A.M.C.; Willemsen, A.C.H.; Gietema, H.A.; Hurkmans, D.P.; Aerts, J.G.; Hendriks, L.E.L.; Schols, A.M.W.J. The Prognostic Value of Weight and Body Composition Changes in Patients with Non-Small-Cell Lung Cancer Treated with Nivolumab. J. Cachexia Sarcopenia Muscle 2021, 12, 657–664. [Google Scholar] [CrossRef]
- Blauwhoff-Buskermolen, S.; Versteeg, K.S.; De Van Der Schueren, M.A.E.; Den Braver, N.R.; Berkhof, J.; Langius, J.A.E.; Verheul, H.M.W. Loss of Muscle Mass During Chemotherapy Is Predictive for Poor Survival of Patients with Metastatic Colorectal Cancer. J. Clin. Oncol. 2016, 34, 1339–1344. [Google Scholar] [CrossRef]
- Ando, C.; Ichihara, E.; Yokoyama, T.; Inoue, K.; Tamura, T.; Fujiwara, K.; Oda, N.; Kano, H.; Kishino, D.; Watanabe, K.; et al. More than One-Third of Advanced Non-Small-Cell Lung Cancer Patients Do Not Receive Immunochemotherapy Due to Intolerance. J. Cancer Res. Clin. Oncol. 2023, 149, 4933–4938. [Google Scholar] [CrossRef]
- Nishimura, T.; Ichihara, E.; Yokoyama, T.; Inoue, K.; Tamura, T.; Sato, K.; Oda, N.; Kano, H.; Kishino, D.; Kawai, H.; et al. The Effect of Pleural Effusion on Prognosis in Patients with Non-Small Cell Lung Cancer Undergoing Immunochemotherapy: A Retrospective Observational Study. Cancers 2022, 14, 6184. [Google Scholar] [CrossRef]
- Tokunaga, T.; Matsuzawa, Y.; Kotani, K.; Keno, Y.; Kobatake, T.; Fujioka, S.; Tarui, S. Ideal Body Weight Estimated from the Body Mass Index with the Lowest Morbidity. Int. J. Obes. 1991, 15, 2010254. [Google Scholar]
- Shiroyama, T.; Nagatomo, I.; Koyama, S.; Hirata, H.; Nishida, S.; Miyake, K.; Fukushima, K.; Shirai, Y.; Mitsui, Y.; Takata, S.; et al. Impact of Sarcopenia in Patients with Advanced Non-Small Cell Lung Cancer Treated with PD-1 Inhibitors: A Preliminary Retrospective Study. Sci. Rep. 2019, 9, 2447. [Google Scholar] [CrossRef]
- Yang, R.; Cheung, M.C.; Pedroso, F.E.; Byrne, M.M.; Koniaris, L.G.; Zimmers, T.A. Obesity and Weight Loss at Presentation of Lung Cancer Are Associated with Opposite Effects on Survival. J. Surg. Res. 2011, 170, e75–e83. [Google Scholar] [CrossRef]
- Martin, L.; Senesse, P.; Gioulbasanis, I.; Antoun, S.; Bozzetti, F.; Deans, C.; Strasser, F.; Thoresen, L.; Jagoe, R.T.; Chasen, M.; et al. Diagnostic Criteria for the Classification of Cancer-Associated Weight Loss. J. Clin. Oncol. 2015, 33, 90–99. [Google Scholar] [CrossRef]
- Rounis, K.; Makrakis, D.; Tsigkas, A.P.; Georgiou, A.; Galanakis, N.; Papadaki, C.; Monastirioti, A.; Vamvakas, L.; Kalbakis, K.; Vardakis, N.; et al. Cancer Cachexia Syndrome and Clinical Outcome in Patients with Metastatic Non-Small Cell Lung Cancer Treated with PD-1/PD-L1 Inhibitors: Results from a Prospective, Observational Study. Transl. Lung Cancer Res. 2021, 10, 3538–3549. [Google Scholar] [CrossRef]
- de Matos-Neto, E.M.; Lima, J.D.C.C.; de Pereira, W.O.; Figuerêdo, R.G.; Riccardi, D.M.D.R.; Radloff, K.; das Neves, R.X.; Camargo, R.G.; Maximiano, L.F.; Tokeshi, F.; et al. Systemic Inflammation in Cachexia—Is Tumor Cytokine Expression Profile the Culprit? Front. Immunol. 2015, 6, 629. [Google Scholar] [CrossRef]
- Fukahori, M.; Shibata, M.; Hamauchi, S.; Kasamatsu, E.; Machii, K. A Retrospective Cohort Study to Investigate the Incidence of Cancer-Related Weight Loss During Chemotherapy in Gastric Cancer Patients. Support. Care Cancer 2021, 29, 341–348. [Google Scholar] [CrossRef]
- Miyawaki, T.; Naito, T.; Yabe, M.; Kodama, H.; Nishioka, N.; Miyawaki, E.; Mamesaya, N.; Kobayashi, H.; Omori, S.; Wakuda, K.; et al. Impact of Weight Loss on Treatment with PD-1/PD-L1 Inhibitors Plus Chemotherapy in Advanced Non-Small-Cell Lung Cancer. Support. Care Cancer 2022, 30, 1633–1641. [Google Scholar] [CrossRef]
- Bonomi, P.D.; Crawford, J.; Dunne, R.F.; Roeland, E.J.; Smoyer, K.E.; Siddiqui, M.K.; McRae, T.D.; Rossulek, M.I.; Revkin, J.H.; Tarasenko, L.C. Mortality Burden of Pre-Treatment Weight Loss in Patients with Non-Small-Cell Lung Cancer: A Systematic Literature Review and Meta-Analysis. J. Cachexia Sarcopenia Muscle 2024, 15, 1226–1239. [Google Scholar] [CrossRef]
- Cortellini, A.; Bersanelli, M.; Buti, S.; Cannita, K.; Santini, D.; Perrone, F.; Giusti, R.; Tiseo, M.; Michiara, M.; Di Marino, P.; et al. A Multicenter Study of Body Mass Index in Cancer Patients Treated with Anti-PD-1/PD-L1 Immune Checkpoint Inhibitors: When Overweight Becomes Favorable. J. Immunother. Cancer 2019, 7, 57. [Google Scholar] [CrossRef]
- McQuade, J.L.; Daniel, C.R.; Hess, K.R.; Mak, C.; Wang, D.Y.; Rai, R.R.; Park, J.J.; Haydu, L.E.; Spencer, C.; Wongchenko, M.; et al. Association of Body-Mass Index and Outcomes in Patients with Metastatic Melanoma Treated with Targeted Therapy, Immunotherapy, or Chemotherapy: A Retrospective, Multicohort Analysis. Lancet Oncol. 2018, 19, 310–322. [Google Scholar] [CrossRef]
- Wang, Z.; Aguilar, E.G.; Luna, J.I.; Dunai, C.; Khuat, L.T.; Le, C.T.; Mirsoian, A.; Minnar, C.M.; Stoffel, K.M.; Sturgill, I.R.; et al. Paradoxical Effects of Obesity on T Cell Function During Tumor Progression and PD-1 Checkpoint Blockade. Nat. Med. 2019, 25, 141–151. [Google Scholar] [CrossRef]
- Ichihara, E.; Harada, D.; Inoue, K.; Sato, K.; Hosokawa, S.; Kishino, D.; Watanabe, K.; Ochi, N.; Oda, N.; Hara, N.; et al. The Impact of Body Mass Index on the Efficacy of Anti-PD-1/PD-L1 Antibodies in Patients with Non-Small Cell Lung Cancer. Lung Cancer 2020, 139, 140–145. [Google Scholar] [CrossRef]
- Antoun, S.; Lanoy, E.; Ammari, S.; Farhane, S.; Martin, L.; Robert, C.; Planchard, D.; Routier, E.; Voisin, A.L.; Messayke, S.; et al. Protective Effect of Obesity on Survival in Cancers Treated with Immunotherapy Vanishes When Controlling for Type of Cancer, Weight Loss and Reduced Skeletal Muscle. Eur. J. Cancer 2023, 178, 49–59. [Google Scholar] [CrossRef]
- Baguley, B.J.; Edbrooke, L.; Denehy, L.; Prado, C.M.; Kiss, N. A Rapid Review of Nutrition and Exercise Approaches to Managing Unintentional Weight Loss, Muscle Loss, and Malnutrition in Cancer. Oncologist 2024, oyae261. [Google Scholar] [CrossRef]
- Langius, J.A.E.; Zandbergen, M.C.; Eerenstein, S.E.J.; van Tulder, M.W.; Leemans, C.R.; Kramer, M.H.H.; Weijs, P.J.M. Effect of Nutritional Interventions on Nutritional Status, Quality of Life and Mortality in Patients with Head and Neck Cancer Receiving (Chemo)Radiotherapy: A Systematic Review. Clin. Nutr. 2013, 32, 671–678. [Google Scholar] [CrossRef]
- Baldwin, C.; Spiro, A.; McGough, C.; Norman, A.R.; Gillbanks, A.; Thomas, K.; Cunningham, D.; O’Brien, M.; Andreyev, H.J.N. Simple Nutritional Intervention in Patients with Advanced Cancers of the Gastrointestinal Tract, Non-Small Cell Lung Cancers or Mesothelioma and Weight Loss Receiving Chemotherapy: A Randomised Controlled Trial. J. Hum. Nutr. Diet. 2011, 24, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Muscaritoli, M.; Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN Practical Guideline: Clinical Nutrition in Cancer. Clin. Nutr. 2021, 40, 2898–2913. [Google Scholar] [CrossRef] [PubMed]
- Bargetzi, L.; Brack, C.; Herrmann, J.; Bargetzi, A.; Hersberger, L.; Bargetzi, M.; Kaegi-Braun, N.; Tribolet, P.; Gomes, F.; Hoess, C.; et al. Nutritional Support During the Hospital Stay Reduces Mortality in Patients with Different Types of Cancers: Secondary Analysis of a Prospective Randomized Trial. Ann. Oncol. 2021, 32, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
| WL Group n = 141 | Non-WL Group n = 229 | ||
|---|---|---|---|
| Median pre-treatment BMI (IQR) | 22.8 (20.0–25.0) | 22.1 (19.9–23.9) | p = 0.168 |
| Median age, years | 71 (40–83) | 69 (34–84) | p = 0.014 |
| Performance status | p = 0.086 | ||
| 0–1 | 124 (87.9%) | 214 (93.4%) | |
| 2–4 | 17 (22.1%) | 15 (6.6%) | |
| Smoking history | p = 0.294 | ||
| Yes | 120 (85.1%) | 186 (81.2%) | |
| No | 19 (13.5%) | 42 (18.3%) | |
| Unknown | 2 (1.4%) | 1 (0.5%) | |
| Sex | p = 0.691 | ||
| Males | 114 (80.9%) | 180 (78.6%) | |
| Females | 27 (19.1%) | 49 (21.4%) | |
| Stage (rec/advanced/unknown) | p = 0.342 | ||
| Advanced | 114 (80.9%) | 170 (74.2%) | |
| Postoperative recurrence | 26 (18.4%) | 57 (24.9%) | |
| Other | 1 (0.7%) | 2 (0.9%) | |
| Histology (non-sq/sq) | p = 0.455 | ||
| Non-Sq | 110 (78.0%) | 170 (74.2%) | |
| Sq | 31 (22.0%) | 59 (25.8%) | |
| PD-L1 expression | p = 0.163 | ||
| <50% | 84 (59.6%) | 149 (65.1%) | |
| ≥50% | 31 (22.0%) | 54 (23.6%) | |
| unknown | 26 18.4%) | 26 (11.3%) | |
| EGFR/ALK mutation | p = 0.853 | ||
| Yes | 12 (8.5%) | 22 (9.6%) | |
| No or undetermined | 129 (91.5%) | 207 (90.4%) | |
| Combination regimen | p = 0.887 | ||
| Pembrolizumab-combined | 118 (83.7%) | 189 (82.5%) | |
| Atezolizumab-combined | 23 (16.3%) | 40 (17.5%) |
| WL Group n = 141 | Non-WL Group n = 229 | p-Value | |
|---|---|---|---|
| Best response rate | |||
| CR | 1 (0.7%) | 4 (1.7%) | |
| PR | 82 (58.2%) | 125 (54.6%) | |
| SD | 45 (31.9%) | 79 (34.5%) | |
| PD | 11 (7.8%) | 14 (6.1%) | |
| Unknown | 2 | 7 | |
| Disease control rate | 128 (90.7%) | 208 (90.8%) | p = 1.000 |
| Objective response rate | 83 (58.7%) | 129 (56.3%) | p = 0.666 |
| PFS HR (95% CI) | p-Value | OS HR (95% CI) | p-Value | |
|---|---|---|---|---|
| Body weight loss | 0.002 | <0.001 | ||
| Yes | 1.69 (1.20–2.36) | 2.24 (1.51–3.36) | ||
| No | 1.0 (reference) | 1.0 (reference) |
| WL Group n = 141 | Non-WL Group n = 229 | Fisher’s Exact Test | |
|---|---|---|---|
| Any irAEs | |||
| Any grade | 59 (41.8%) | 88 (38.4%) | p = 0.514 |
| Grade 3 or more | 24 (17.0%) | 25 (10.9%) | p = 0.114 |
| Hypothyroidism | |||
| Any grade | 6 (4.3%) | 6 (2.6%) | p = 0.389 |
| Grade 3 or more | 0 (0%) | 0 (0%) | |
| Pneumonitis | |||
| Any grade | 21 (14.9%) | 33 (14.4%) | p = 0.881 |
| Grade 3 or more | 11 (7.8%) | 12 (5.2%) | p = 0.377 |
| Infusion reaction | |||
| Any grade | 0 (0%) | 1 (0.4%) | |
| Grade 3 or more | 0 (0%) | 0 (0%) | |
| Colitis | |||
| Any grade | 5 (3.5%) | 4 (1.7%) | p = 0.275 |
| Grade 3 or more | 2 (1.4%) | 3 (1.3%) | p = 0.930 |
| Hepatitis | |||
| Any grade | 1 (0.7%) | 7 (3.1%) | p = 0.132 |
| Grade 3 or more | 1 (0.7%) | 3 (1.3%) | p = 0.587 |
| Skin reaction | |||
| Any grade | 8 (5.7%) | 27 (11.8%) | p = 0.051 |
| Grade 3 or more | 1 (0.7%) | 0 (0%) | |
| Hypophysitis | |||
| Any grade | 3 (2.1%) | 2 (0.9%) | p = 0.310 |
| Grade 3 or more | 3 (2.1%) | 2 (0.9%) | p = 0.310 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taoka, M.; Ichihara, E.; Yokoyama, T.; Inoue, K.; Tamura, T.; Sato, A.; Oda, N.; Kano, H.; Nakamura, K.; Kawai, H.; et al. Frequency and Significance of Body Weight Loss During Immunochemotherapy in Patients with Advanced Non-Small Cell Lung Cancer. Cancers 2024, 16, 4089. https://doi.org/10.3390/cancers16234089
Taoka M, Ichihara E, Yokoyama T, Inoue K, Tamura T, Sato A, Oda N, Kano H, Nakamura K, Kawai H, et al. Frequency and Significance of Body Weight Loss During Immunochemotherapy in Patients with Advanced Non-Small Cell Lung Cancer. Cancers. 2024; 16(23):4089. https://doi.org/10.3390/cancers16234089
Chicago/Turabian StyleTaoka, Masataka, Eiki Ichihara, Toshihide Yokoyama, Koji Inoue, Tomoki Tamura, Akiko Sato, Naohiro Oda, Hirohisa Kano, Kayo Nakamura, Haruyuki Kawai, and et al. 2024. "Frequency and Significance of Body Weight Loss During Immunochemotherapy in Patients with Advanced Non-Small Cell Lung Cancer" Cancers 16, no. 23: 4089. https://doi.org/10.3390/cancers16234089
APA StyleTaoka, M., Ichihara, E., Yokoyama, T., Inoue, K., Tamura, T., Sato, A., Oda, N., Kano, H., Nakamura, K., Kawai, H., Inoue, M., Ochi, N., Fujimoto, N., Ichikawa, H., Ando, C., Oze, I., Kiura, K., Maeda, Y., & Hotta, K. (2024). Frequency and Significance of Body Weight Loss During Immunochemotherapy in Patients with Advanced Non-Small Cell Lung Cancer. Cancers, 16(23), 4089. https://doi.org/10.3390/cancers16234089





