Lung Cancer Screening: New Perspective and Challenges in Europe
Abstract
Simple Summary
Abstract
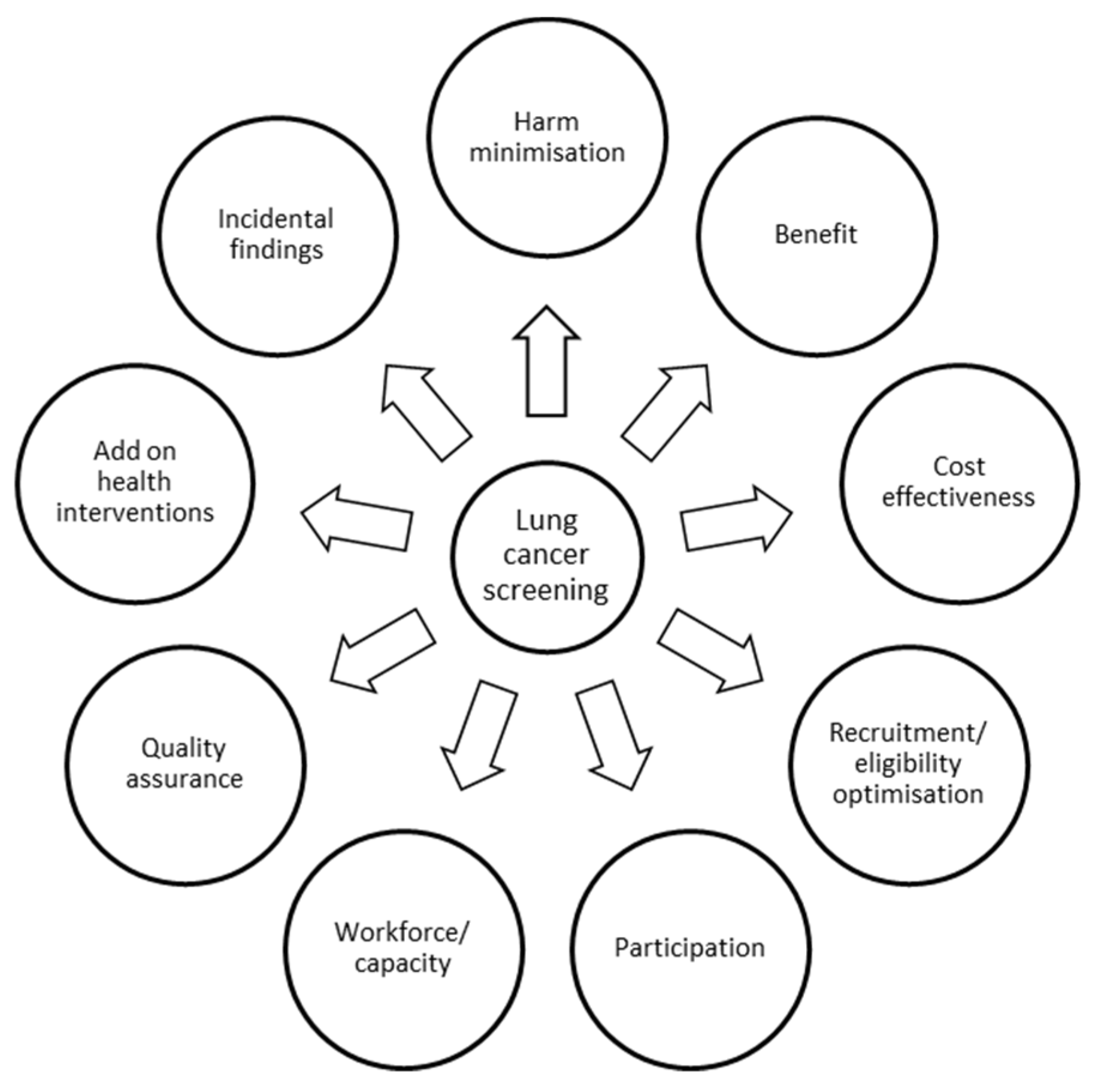
1. Introduction
2. Recruitment, Eligibility Optimisation and Participation
3. Harm Minimization
4. Cost Effectiveness and Add-On Health Interventions
5. Incidental Findings
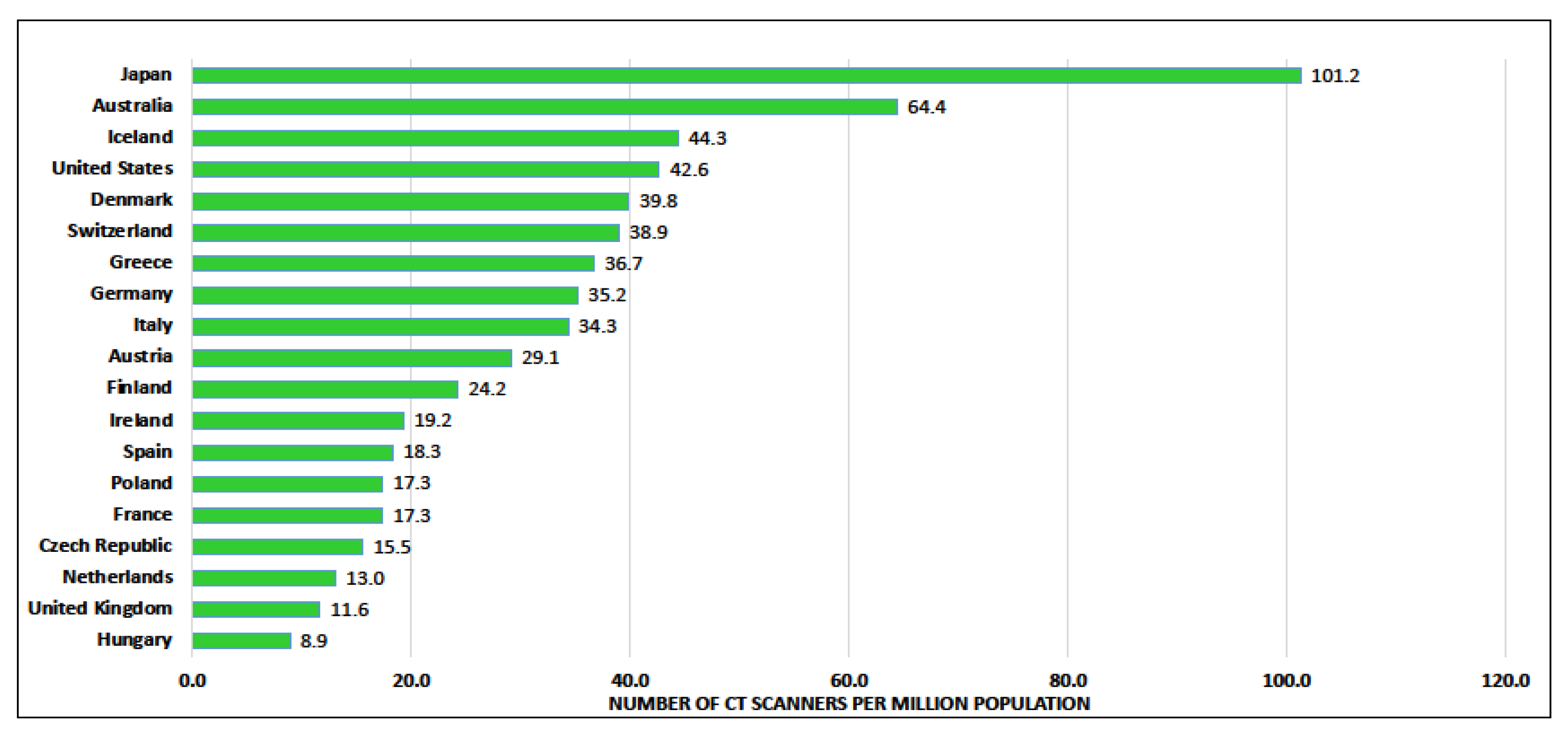
6. Workforce/Capacity
- Participation of thoracic surgeons in CT screening programmes;
- Training and clinical profile for surgeons participating in CT screening programmes;
- The use of minimally invasive thoracic surgery and other relevant surgical issues;
- Associated elements such as smoking cessation, nodule evaluation algorithms and pathology reports.
7. Quality Assurance
8. The European Union (EU) and Lung Cancer Screening
- Implement new cancer screening tests in routine healthcare only after they have been evaluated in randomised controlled trials;
- Run trials, in addition to those on screening-specific parameters and mortality, on subsequent treatment procedures, clinical outcomes, side effects, morbidity and quality of life;
- Assess the level of evidence concerning the effects of new methods by pooling trial results from representative settings;
- Consider the introduction into routine healthcare of potentially promising new screening tests, which are currently being evaluated in randomised controlled trials, once the evidence is conclusive and other relevant aspects, such as cost effectiveness in the different healthcare systems, have been taken into account.
- By 2024 the European Commission should develop and publish new guidelines on lung cancer screening for high-risk groups;
- By 2026 at least five EU Member States should have incorporated these guidelines into their national cancer plans;
- By 2027, all EU Member States should have put in place a strategy for the early detection of lung cancer in the high-risk population.
9. Implementation of Lung Cancer Screening in Europe
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- National Lung Screening Trial Research Team; Aberle, D.R.; Adams, A.M.; Berg, C.D.; Black, W.C.; Clapp, J.D.; Fagerstrom, F.M.; Gareen, I.F.; Gatsonis, C.; Marcus, P.M.; et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N. Engl. J. Med. 2011, 365, 395–409. [Google Scholar] [PubMed]
- de Koning, H.J.; van der Aalst, C.M.; de Jong, P.A.; Scholten, E.T.; Nackaerts, K.; Heuvelmans, M.A.; Lammers, J.J.; Weenink, C.; Yousaf-Khan, U.; Horeweg, N.; et al. Reduced Lung-Cancer Mortality with Volume CT Screening in a Randomized Trial. N. Engl. J. Med. 2020, 382, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Becker, N.; Motsch, E.; Trotter, A.; Heussel, C.P.; Dienemann, H.; Schnabel, P.A.; Kauczor, H.U.; Maldonado, S.G.; Miller, A.B.; Kaaks, R.; et al. Lung cancer mortality reduction by LDCT screening-Results from the randomized German LUSI trial. Int. J. Cancer 2020, 146, 1503–1513. [Google Scholar] [CrossRef] [PubMed]
- Pastorino, U.; Silva, M.; Sestini, S.; Sabia, F.; Boeri, M.; Cantarutti, A.; Sverzellati, N.; Sozzi, G.; Corrao, G.; Marchiano, A. Prolonged lung cancer screening reduced 10-year mortality in the MILD trial: New confirmation of lung cancer screening efficacy. Ann. Oncol. 2019, 30, 1162–1169. [Google Scholar] [CrossRef]
- Field, J.K.; Vulkan, D.; Davies, M.P.A.; Baldwin, D.R.; Brain, K.E.; Devaraj, A.; Eisen, T.; Gosney, J.; Green, B.A.; Holemans, J.A.; et al. Lung cancer mortality reduction by LDCT screening: UKLS randomised trial results and international meta-analysis. Lancet Reg. Health Eur. 2021, 10, 100179. [Google Scholar] [CrossRef]
- Duma, N.; Santana-Davila, R.; Molina, J.R. Non-Small Cell Lung Cancer: Epidemiology, Screening, Diagnosis, and Treatment. Mayo Clin. Proc. 2019, 94, 1623–1640. [Google Scholar] [CrossRef]
- Tammemagi, M.C.; Schmidt, H.; Martel, S.; McWilliams, A.; Goffin, J.R.; Johnston, M.R.; Tremblay, A.; Bhatia, R.; Liu, G.; Soghrati, K.; et al. Participant selection for lung cancer screening by risk modelling (the Pan-Canadian Early Detection of Lung Cancer [PanCan] study): A single-arm, prospective study. Lancet Oncol. 2017, 18, 1523–1531. [Google Scholar] [CrossRef]
- Ten Haaf, K.; Jeon, J.; Tammemagi, M.C.; Han, S.S.; Kong, C.Y.; Plevritis, S.K.; Feuer, E.J.; de Koning, H.J.; Steyerberg, E.W.; Meza, R. Risk prediction models for selection of lung cancer screening candidates: A retrospective validation study. PLoS Med. 2017, 14, e1002277. [Google Scholar] [CrossRef]
- Tammemagi, M.C.; Katki, H.A.; Hocking, W.G.; Church, T.R.; Caporaso, N.; Kvale, P.A.; Chaturvedi, A.K.; Silvestri, G.A.; Riley, T.L.; Commins, J.; et al. Selection criteria for lung-cancer screening. N. Engl. J. Med. 2013, 368, 728–736. [Google Scholar] [CrossRef]
- Raji, O.Y.; Duffy, S.W.; Agbaje, O.F.; Baker, S.G.; Christiani, D.C.; Cassidy, A.; Field, J.K. Predictive accuracy of the Liverpool Lung Project risk model for stratifying patients for computed tomography screening for lung cancer: A case-control and cohort validation study. Ann. Intern. Med. 2012, 157, 242–250. [Google Scholar] [CrossRef]
- Katki, H.A.; Kovalchik, S.A.; Petito, L.C.; Cheung, L.C.; Jacobs, E.; Jemal, A.; Berg, C.D.; Chaturvedi, A.K. Implications of Nine Risk Prediction Models for Selecting Ever-Smokers for Computed Tomography Lung Cancer Screening. Ann. Intern. Med. 2018, 169, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Ten Haaf, K.; Bastani, M.; Cao, P.; Jeon, J.; Toumazis, I.; Han, S.S.; Plevritis, S.K.; Blom, E.F.; Kong, C.Y.; Tammemagi, M.C.; et al. A Comparative Modeling Analysis of Risk-Based Lung Cancer Screening Strategies. J. Natl. Cancer Inst. 2020, 112, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Myles, J.P.; van Tongeren, M.; Page, R.D.; Liloglou, T.; Duffy, S.W.; Filed, J.K. The LLP risk model: An individual risk prediction model for lung cancer. Br. J. Cancer 2008, 98, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Lifford, K.J.; Carter, B.; McRonald, F.; Yadegarfar, G.; Baldwin, D.R.; Weller, D.; Hansell, D.M.; Duffy, S.W.; Field, J.K.; et al. Barriers to uptake among high-risk individuals declining participation in lung cancer screening: A mixed methods analysis of the UK Lung Cancer Screening (UKLS) trial. BMJ Open 2015, 5, e008254. [Google Scholar] [CrossRef]
- Dickson, J.L.; Quaife, S.L.; Horst, C.; Tisi, S.; Hall, H.; Verghese, P.; Mullin, A.; Sarpong, R.; Teague, J.; Farrelly, R.; et al. P18 The SUMMIT study: Uptake from re-invitation. Thorax 2021, 76, A94–A95. [Google Scholar]
- Crosbie, P.A.; Gabe, R.; Simmonds, I.; Kennedy, M.; Rogerson, S.; Ahmed, N.; Baldwin, D.R.; Booton, R.; Cochrane, A.; Darby, M.; et al. Yorkshire Lung Screening Trial (YLST): Protocol for a randomised controlled trial to evaluate invitation to community-based low-dose CT screening for lung cancer versus usual care in a targeted population at risk. BMJ Open 2020, 10, e037075. [Google Scholar] [CrossRef]
- Quaife, S.L.; Ruparel, M.; Dickson, J.L.; Beeken, R.J.; McEwen, A.; Baldwin, D.R.; Bhowmik, A.; Navani, N.; Sennett, K.; Duffy, S.W.; et al. Lung Screen Uptake Trial (LSUT): Randomized Controlled Clinical Trial Testing Targeted Invitation Materials. Am. J. Respir. Crit. Care Med. 2020, 201, 965–975. [Google Scholar] [CrossRef]
- Pham, D.; Bhandari, S.; Pinkston, C.; Oechsli, M.; Kloecker, G. Lung Cancer Screening Registry Reveals Low-dose CT Screening Remains Heavily Underutilized. Clin. Lung Cancer 2020, 21, e206–e211. [Google Scholar] [CrossRef]
- Zahnd, W.E.; Eberth, J.M. Lung Cancer Screening Utilization: A Behavioral Risk Factor Surveillance System Analysis. Am. J. Prev. Med. 2019, 57, 250–255. [Google Scholar] [CrossRef]
- Ruparel, M.; Quaife, S.L.; Dickson, J.L.; Horst, C.; Tisi, S.; Hall, H.; Taylor, M.; Ahmed, A.; Shaw, P.; Burke, S.; et al. Lung Screen Uptake Trial: Results from a single lung cancer screening round. Thorax 2020, 75, 908–912. [Google Scholar] [CrossRef]
- Callister, M.E.; Baldwin, D.R.; Akram, A.R.; Barnard, S.; Cane, P.; Draffan, J.; Franks, K.; Gleeson, F.; Graham, R.; Malhotra, P.; et al. British Thoracic Society guidelines for the investigation and management of pulmonary nodules. Thorax 2015, 70, ii1–ii54. [Google Scholar] [CrossRef] [PubMed]
- MacMahon, H.; Naidich, D.P.; Goo, J.M.; Lee, K.S.; Leung, A.N.C.; Mayo, J.R.; Mehta, A.C.; Ohno, Y.; Powell, C.A.; Prokop, M.; et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology 2017, 284, 228–243. [Google Scholar] [CrossRef] [PubMed]
- Devaraj, A.; van Ginneken, B.; Nair, A.; Baldwin, D. Use of Volumetry for Lung Nodule Management: Theory and Practice. Radiology 2017, 284, 630–644. [Google Scholar] [CrossRef] [PubMed]
- Jeon, K.N.; Goo, J.M.; Lee, C.H.; Lee, Y.; Choo, J.Y.; Lee, N.K.; Shim, M.S.; Lee, I.S.; Kim, K.G.; Gierada, D.S.; et al. Computer-aided nodule detection and volumetry to reduce variability between radiologists in the interpretation of lung nodules at low-dose screening computed tomography. Investig. Radiol. 2012, 47, 457–461. [Google Scholar] [CrossRef] [PubMed]
- National Lung Screening Trial Research Team. Lung Cancer Incidence and Mortality with Extended Follow-up in the National Lung Screening Trial. J. Thorac Oncol. 2019, 14, 1732–1742. [Google Scholar] [CrossRef]
- Field, J.K.; Hansell, D.M.; Duffy, S.W.; Baldwin, D.R. CT screening for lung cancer: Countdown to implementation. Lancet Oncol. 2013, 14, e591–e600. [Google Scholar] [CrossRef]
- Balata, H.; Ruparel, M.; O’Dowd, E.; Ledson, M.; Field, J.K.; Duffy, S.W.; Quaife, S.L.; Sharman, A.; Janes, S.; Baldwin, D.; et al. Analysis of the baseline performance of five UK lung cancer screening programmes. Lung Cancer 2021, 161, 136–140. [Google Scholar] [CrossRef]
- Rampinelli, C.; De Marco, P.; Origgi, D.; Maisonneuve, P.; Casiraghi, M.; Veronesi, G.; Spaggiari, L.; Bellomi, M. Exposure to low dose computed tomography for lung cancer screening and risk of cancer: Secondary analysis of trial data and risk-benefit analysis. BMJ 2017, 356, j347. [Google Scholar] [CrossRef]
- Toumazis, I.; de Nijs, K.; Cao, P.; Bastani, M.; Munshi, V.; Ten Haaf, K.; Jeon, J.; Gazelle, G.S.; Feuer, E.J.; de Koning, H.J.; et al. Cost-effectiveness Evaluation of the 2021 US Preventive Services Task Force Recommendation for Lung Cancer Screening. JAMA Oncol. 2021, 7, 1833–1842. [Google Scholar] [CrossRef]
- Black, W.C.; Gareen, I.F.; Soneji, S.S.; Sicks, J.D.; Keeler, E.B.; Aberle, D.R.; Naeim, A.; Church, T.R.; Silvestri, G.A.; Gorelick, J.; et al. Cost-effectiveness of CT screening in the National Lung Screening Trial. N. Engl. J. Med. 2014, 371, 1793–1802. [Google Scholar] [CrossRef]
- Field, J.K.; Duffy, S.W.; Baldwin, D.R.; Whynes, D.K.; Devaraj, A.; Brain, K.E.; Eisen, T.; Gosney, J.; Green, B.A.; Holemans, J.A.; et al. UK Lung Cancer RCT Pilot Screening Trial: Baseline findings from the screening arm provide evidence for the potential implementation of lung cancer screening. Thorax 2016, 71, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Hinde, S.; Crilly, T.; Balata, H.; Bartlett, R.; Crilly, J.; Barber, P.; Threlfall, A.; Tonge, J.; Booton, R.; Crosbie, P.A. The cost-effectiveness of the Manchester ‘lung health checks’, a community-based lung cancer low-dose CT screening pilot. Lung Cancer 2018, 126, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Cressman, S.; Peacock, S.J.; Tammemagi, M.C.; Evans, W.K.; Leighl, N.B.; Goffin, J.R.; Tremblay, A.; Liu, G.; Manos, D.; MacEachern, P.; et al. The Cost-Effectiveness of High-Risk Lung Cancer Screening and Drivers of Program Efficiency. J. Thorac Oncol. 2017, 12, 1210–1222. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Sidorenkov, G.; Heuvelmans, M.A.; Groen, H.J.M.; Vermeulen, K.M.; Greuter, M.J.W.; de Bock, G.H. Cost-effectiveness of lung cancer screening with low-dose computed tomography in heavy smokers: A microsimulation modelling study. Eur. J. Cancer 2020, 135, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Hofer, F.; Kauczor, H.U.; Stargardt, T. Cost-utility analysis of a potential lung cancer screening program for a high-risk population in Germany: A modelling approach. Lung Cancer 2018, 124, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Sun, Y.; Wang, K.; Wang, Z.; Li, D.; Fan, M.; Bu, X.; Chen, M.; Ren, H. Cost Effectiveness of Lung Cancer Screening with Low-Dose CT in Heavy Smokers in China. Cancer Prev. Res. 2022, 15, 37–44. [Google Scholar] [CrossRef]
- Murray, R.L.; Brain, K.; Britton, J.; Quinn-Scoggins, H.D.; Lewis, S.; McCutchan, G.M.; Quaife, S.L.; Wu, Q.; Ashurst, A.; Baldwin, D.; et al. Yorkshire Enhanced Stop Smoking (YESS) study: A protocol for a randomised controlled trial to evaluate the effect of adding a personalised smoking cessation intervention to a lung cancer screening programme. BMJ Open 2020, 10, e037086. [Google Scholar] [CrossRef]
- Van der Aalst, C.O.M.; Oudkerk, M.; Ten Haaf, K.; Baldwin, D.; Murray, R.; O’Dowd, E.; Kaaks, R.; Katzke, V.; Becker, N.; Espinas, J.; et al. Towards personalized lung cancer CT screening in Europe. Eur. Respir. J. 2020, 56 (Suppl. S64), 4171. [Google Scholar]
- van de Wiel, J.C.; Wang, Y.; Xu, D.M.; van der Zaag-Loonen, H.J.; van der Jagt, E.J.; van Klaveren, R.J.; Oudkerk, M.; NELSON Study Group. Neglectable benefit of searching for incidental findings in the Dutch-Belgian lung cancer screening trial (NELSON) using low-dose multidetector CT. Eur. Radiol. 2007, 17, 1474–1482. [Google Scholar] [CrossRef]
- Bartlett, E.C.; Belsey, J.; Derbyshire, J.; Morris, K.; Chen, M.; Addis, J.; Martins, M.; Ridge, C.A.; Desai, S.R.; Mirsadraee, S.; et al. Implications of incidental findings from lung screening for primary care: Data from a UK pilot. NPJ Prim. Care Respir. Med. 2021, 31, 36. [Google Scholar] [CrossRef]
- Horst, C.D.J.; Dickson, J.; Tisi, S.; Hall, H.; Verghese, P.; Mullin, A.; Farrelli, L.; Levermore, C.; Gyertson, K.; Clarke, C.; et al. P41.04 The SUMMIT Study: Pulmonary Nodule and Incidental Findings in the First 10,000 Participants of a Population-Based Low-Dose CT Screening Study. J. Thorac. Oncol. 2021, 16, S473–S474. [Google Scholar] [CrossRef]
- Silvestrin, C. Europe’s Looming Radiology Capacity Challenge: A Comparative Study. 2016. Available online: https://www.telemedicineclinic.com (accessed on 26 April 2022).
- Diagnostic Radiography Workforce UK Census 2020. The Society and College of Radiographers 2020. Available online: https://www.sor.org/learning-advice/professional-body-guidance-and-publications/documents-and-publications/reports-and-surveys/or-diagnostic-radiography-workforce-uk-census-2020 (accessed on 26 April 2022).
- Rodin, D.; Grover, S.; Xu, M.J.; Hanna, T.P.; Olson, R.; Schreiner, L.J.; Munshi, A.; Mornex, F.; Palma, D.; Gaspar, L.E.; et al. Radiotherapeutic Management of Non-Small Cell Lung Cancer in the Minimal Resource Setting. J. Thorac Oncol. 2016, 11, 21–29. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Woodward, R. Organisation for Economic Co-Operation and Development (OECD); Routledge: London, UK, 2022. [Google Scholar]
- Pedersen, J.H.; Rzyman, W.; Veronesi, G.; D’Amico, T.A.; Van Schil, P.; Molins, L.; Massard, G.; Rocco, G. Recommendations from the European Society of Thoracic Surgeons (ESTS) regarding computed tomography screening for lung cancer in Europe. Eur. J. Cardiothorac. Surg. 2017, 51, 411–420. [Google Scholar] [CrossRef] [PubMed]
- NHS England. Quality Assurance Standards prepared for the Targeted Lung Health Checks Programme. In National Cancer Programme Targeted Screening for Lung Cancer with Low Radiation Dose Computed Tomography; National Health Service (NHS): UK, 2019. [Google Scholar]
- NHS England. Standard Protocol prepared for the Targeted Lung Health Checks Programme. In National Cancer Programme Targeted Screening for Lung Cancer with Low Radiation Dose Computed Tomography; National Health Service (NHS): UK, 2019. [Google Scholar]
- Greer, S.L.F.N.; Rozenblum, S.; Jarman, H.; Palm, W.; Elliott, H.A. (Eds.) European Observatory on Health Systems and Policies. In Everything You Always Wanted to Know about European Union Health Policies but Were Afraid to Ask, 2nd ed.; European Observatory on Health Systems and Policies: Copenhagen, Denmark, 2019. [Google Scholar]
- Basu, P.; Ponti, A.; Anttila, A.; Ronco, G.; Senore, C.; Vale, D.B.; Sagnan, N.; Tomatis, M.; Soerjomataram, I.; Zakelj, M.P.; et al. Status of implementation and organization of cancer screening in The European Union Member States-Summary results from the second European screening report. Int. J. Cancer 2018, 142, 44–56. [Google Scholar] [CrossRef]
- Rzyman, W.; Szurowska, E.; Adamek, M. Implementation of lung cancer screening at the national level: Polish example. Transl. Lung Cancer Res. 2019, 8, S95–S105. [Google Scholar] [CrossRef]
| Question 1: Is there as of 1 January 2022 an ONGOING population-based nationwide lung cancer screening (LCS) programme with low dose CT scan in your country? |
| Yes: 1/23 (4.3%) No: 22/23 (95.7%) → If NO, will there be one starting in 2022? Yes: 3/22 (13.6%) No: 19/22 (86.4%) → If NO: does your country require a formal EU-recommendation before embarking on lung cancer screening? Yes: 3/22 (13.6%) No: 19/22 (86.4%) → If NO: is the policy and regulatory process governing the decision to implement nation-wide screening programmes well defined in your country? Yes: 12/21 (57.1%) No: 9/21 (42.9%) |
| Question 2: Are institutions in your country currently participating in feasibility projects, piloting the implementation of LCS with low dose CT scan in your country? |
| Yes: 11/23 (47.8%) → If YES, is there a national screening protocol or task force? Yes: 7/11 (63.6%) No: 4/11 (36.4%) No: 12/23 (52,2%) → If NO: will there be one starting in 2022? Yes: 2/12 No: 8/12 No answer/unclear: 2/12 |
| Question 3: Has any cost-effectiveness study been published regarding LCS in your country? |
| Yes: 5/23 (21.7%) No: 18/23 (78.3%) → If NO: is a cost-effectiveness study required for lung cancer screening to be implemented? Yes: 9/18 (50%) No: 9/18 (50%) |
| Question 4: Who will cover the costs of a national population based lung cancer screening programme in your country? |
| Health authority and/or National health service: 16/23 (69.6%) Insurance companies: 4/23 (17.4%) Other: 3/23 (13.0%) |
| Question 5: Who decides on the implementation of a population based LCS in your country? |
| National Health minister or Health board: 19/23 (82.7%) Board of directors of health insurance companies: 3/23 (13.0%) Regional or local health authority: 2/23 (8.7%) Other: 3/23 (13.0%) |
| Question 6: Will the implementation of lung cancer screening be conditional of a structured smoking cessation intervention? |
| Yes: 9/23 (39.1%) Very likely: 8/23 (34.8%) Likely: 4/23 (17.4%) Unlikely: 2/23 (8.7%) No: 0/23 |
| Question 7: What are according to you the main hurdles for the implementation of a population-based national lung cancer screening programme in your country? Ranking of cost–public opinion on stigma of lung cancer–capacity of CT-scans and radiologists–willingness of GP’s/radiologists-others |
| Most important hurdle: Cost 10/23 (43.5%) Public opinion on stigma of lung cancer 3/23 (13.0%) Capacity of CT-scans and radiologists 5/23 (21.7%) Willingness of general practitioners’s/radiologists 2/23 (8.7%) Other 3/23 (13.0%) Second most important hurdle: Cost 9/23 (39.1%) Public opinion on stigma of lung cancer 4/23 (17.4%) Capacity of CT-scans and radiologists 6/23 (26.1%) Willingness of general practitioners’s/radiologists 3/23 (13.0%) Other 1/23 (4.3%) Third most important hurdle: Cost 4/23 (17.4%) Public opinion on stigma of lung cancer 0/23 Capacity of CT-scans and radiologists 5/23 (21.7%) Willingness of general practitioners’s/radiologists 7/23 (30.4%) Other 7/23 (30.4%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Meerbeeck, J.P.; O’Dowd, E.; Ward, B.; Van Schil, P.; Snoeckx, A. Lung Cancer Screening: New Perspective and Challenges in Europe. Cancers 2022, 14, 2343. https://doi.org/10.3390/cancers14092343
Van Meerbeeck JP, O’Dowd E, Ward B, Van Schil P, Snoeckx A. Lung Cancer Screening: New Perspective and Challenges in Europe. Cancers. 2022; 14(9):2343. https://doi.org/10.3390/cancers14092343
Chicago/Turabian StyleVan Meerbeeck, Jan P., Emma O’Dowd, Brian Ward, Paul Van Schil, and Annemiek Snoeckx. 2022. "Lung Cancer Screening: New Perspective and Challenges in Europe" Cancers 14, no. 9: 2343. https://doi.org/10.3390/cancers14092343
APA StyleVan Meerbeeck, J. P., O’Dowd, E., Ward, B., Van Schil, P., & Snoeckx, A. (2022). Lung Cancer Screening: New Perspective and Challenges in Europe. Cancers, 14(9), 2343. https://doi.org/10.3390/cancers14092343






