Inhibition of MACC1-Induced Metastasis in Esophageal and Gastric Adenocarcinomas
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Tissue Samples
2.3. Cell Lines and Medium
2.4. Cloning and Transduction
2.5. MACC1 Expression Analysis
2.6. Proliferation
2.7. Migration
2.8. In Vitro Drug Treatment
2.9. Cell-Line-Derived Xenograft Model
2.10. Statistics
3. Results
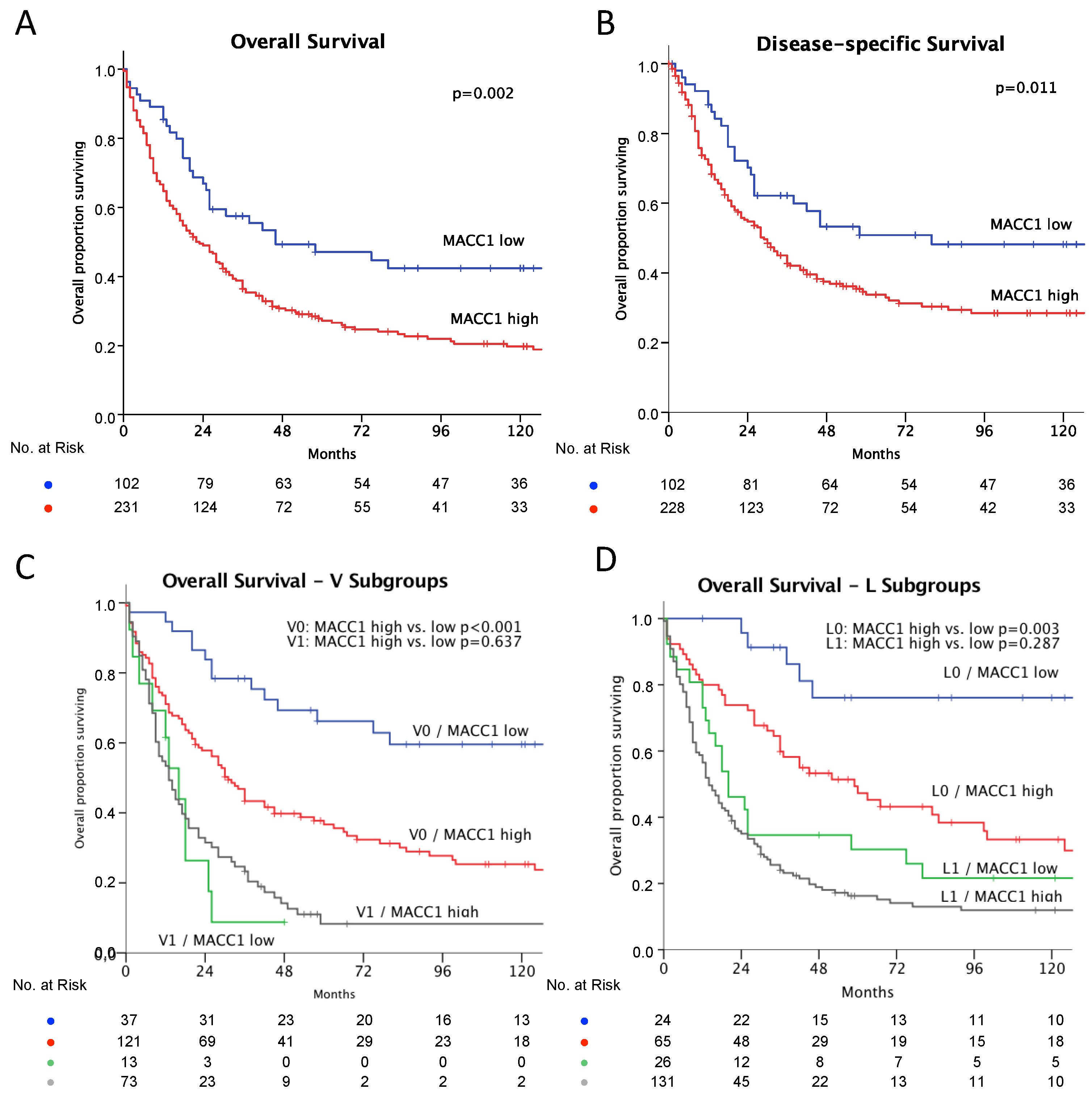
3.1. Patients
3.2. MACC1 Expression
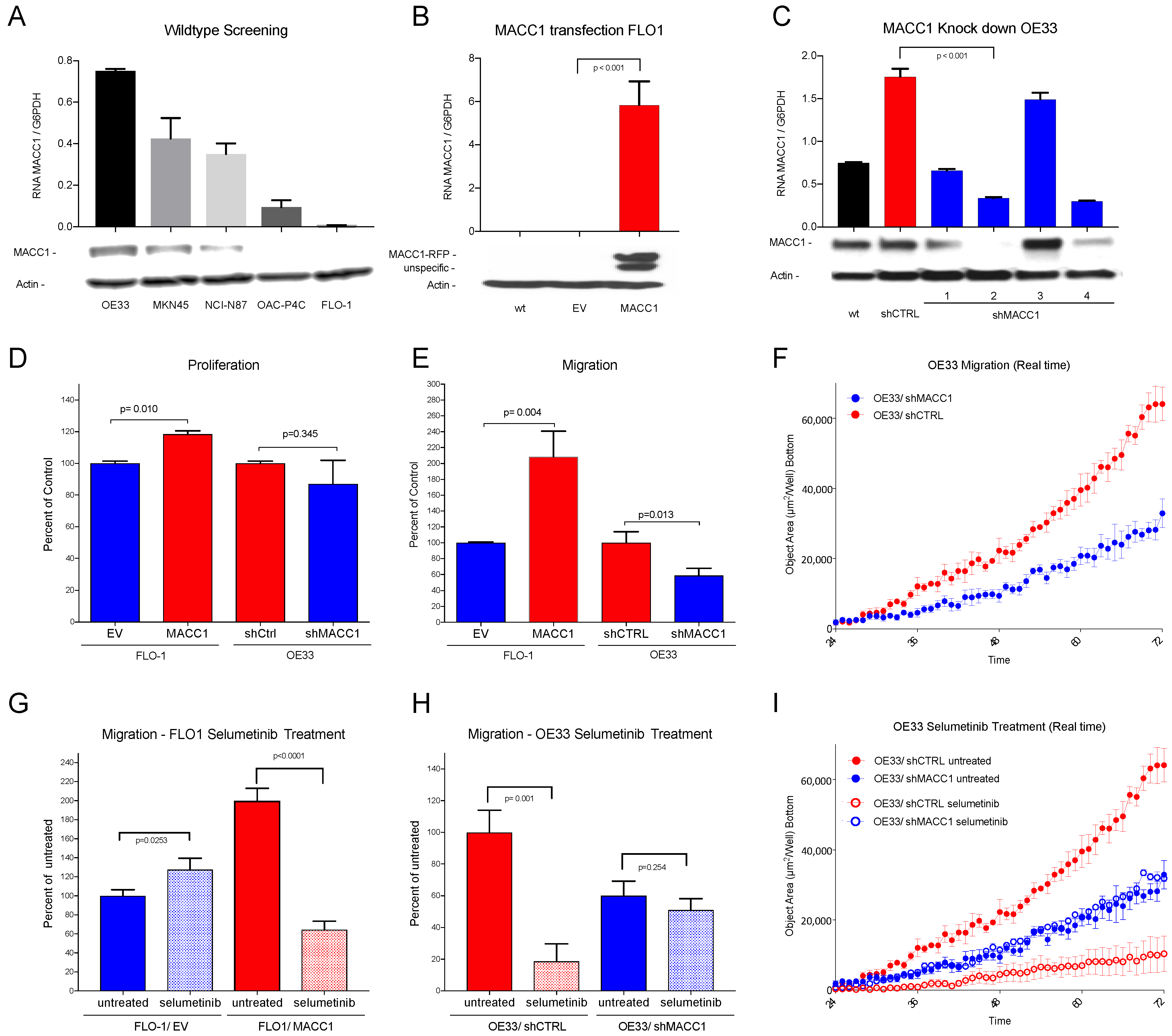
3.3. In Vitro Analysis
3.4. In Vivo Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Son, T. Multi-institutional validation of the 8th AJCC TNM staging system for gastric cancer: Analysis of survival data from high-volume Eastern centers and the SEER database. J. Surg. Oncol. 2019, 120, 676–684. [Google Scholar] [CrossRef]
- Riihimäki, M.; Hemminki, A.; Sundquist, K.; Sundquist, J.; Hemminki, K. Metastatic spread in patients with gastric cancer. Oncotarget 2016, 7, 52307–52316. [Google Scholar] [CrossRef] [Green Version]
- He, X.; Wu, W.; Lin, Z.; Ding, Y.; Si, J.; Sun, L. Validation of the American Joint Committee on Cancer (AJCC) 8th edition stage system for gastric cancer patients: A population-based analysis American Joint Committee on Cancer. Gastric Cancer 2018, 21, 391–400. [Google Scholar] [CrossRef]
- Stein, U.; Walther, W.; Arlt, F.; Schwabe, H.; Smith, J.; Fichtner, I.; Birchmeier, W.; Schlag, P.M. MACC1, a newly identified key regulator of HGF-MET signaling, predicts colon cancer metastasis. Nat. Med. 2009, 15, 59–67. [Google Scholar] [CrossRef]
- Schmid, F.; Wang, Q.; Huska, M.R.; Andrade-Navarro, M.A.; Lemm, M.; Fichtner, I.; Dahlmann, M.; Kobelt, D.; Walther, W.; Smith, J.; et al. SPON2, a newly identified target gene of MACC1, drives colorectal cancer metastasis in mice and is prognostic for colorectal cancer patient survival. Oncogene 2016, 35, 5942–5952. [Google Scholar] [CrossRef]
- Lemos, C.; Hardt, M.S.; Juneja, M.; Voss, C.; Forster, S.; Jerchow, B.; Haider, W.; Blaker, H.; Stein, U. MACC1 induces tumor progression in transgenic mice and colorectal cancer patients via increased pluripotency Markers Nanog and oct4. Clin. Cancer Res. 2016, 22, 2812–2824. [Google Scholar] [CrossRef] [Green Version]
- Pichorner, A.; Sack, U.; Kobelt, D.; Kelch, I.; Arlt, F.; Smith, J.; Walther, W.; Schlag, P.M.; Stein, U. In vivo imaging of colorectal cancer growth and metastasis by targeting MACC1 with shRNA in xenografted mice. Clin. Exp. Metastasis 2012, 29, 573–583. [Google Scholar] [CrossRef]
- Budczies, J.; Kluck, K.; Walther, W.; Stein, U. Decoding and targeting the molecular basis of MACC1-driven metastatic spread: Lessons from big data mining and clinical-experimental approaches. Semin. Cancer Biol. 2020, 60, 365–379. [Google Scholar] [CrossRef]
- Radhakrishnan, H.; Walther, W.; Zincke, F.; Kobelt, D.; Imbastari, F.; Erdem, M.; Kortüm, B.; Dahlmann, M.; Stein, U. MACC1—The first decade of a key metastasis molecule from gene discovery to clinical translation. Cancer Metastasis Rev. 2018, 37, 805–820. [Google Scholar] [CrossRef]
- Guo, T.; Yang, J.; Yao, J.; Zhang, Y.; Da, M.; Duan, Y. Expression of MACC1 and c-Met in human gastric cancer and its clinical significance. Cancer Cell Int. 2013, 13, 121. [Google Scholar] [CrossRef] [Green Version]
- Ma, J.; Ma, J.; Meng, Q.; Zhao, Z.S.; Xu, W.J. Prognostic value and clinical pathology of MACC-1 and c-MET expression in gastric carcinoma. Pathol. Oncol. Res. 2013, 19, 821–832. [Google Scholar] [CrossRef]
- Wang, L.; Wu, Y.; Lin, L.; Liu, P.; Huang, H.; Liao, W.; Zheng, D.; Zuo, Q.; Sun, L.; Huang, N.; et al. Metastasis-associated in colon cancer-1 upregulation predicts a poor prognosis of gastric cancer, and promotes tumor cell proliferation and invasion. Int. J. Cancer 2013, 133, 1419–1430. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Duan, J.; Jiang, Y.; Wang, L.; Huang, N.; Lin, L.; Liao, Y.; Liao, W. Metastasis-associated in colon cancer-1 upregulates vascular endothelial growth factor-C/D to promote lymphangiogenesis in human gastric cancer. Cancer Lett. 2015, 357, 242–253. [Google Scholar] [CrossRef]
- Koh, Y.W.; Hur, H.; Lee, D. Increased MACC1 expression indicates a poor prognosis independent of MET expression in gastric adenocarcinoma. Pathol. Res. Pract. 2016, 212, 93–100. [Google Scholar] [CrossRef]
- Lu, G.; Zhou, L.; Zhang, X.; Zhu, B.; Wu, S.; Song, W.; Gong, X.; Wang, D.; Tao, Y. The expression of metastasis-associated in colon cancer-1 and KAI1 in gastric adenocarcinoma and their clinical significance. World J. Surg. Oncol. 2016, 14, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Xie, Q.; Xiang, C.; Wang, G.; Lei, K.; Wang, Y. MACC1 upregulation promotes gastric cancer tumor cell metastasis and predicts a poor prognosis. J. Zhejiang Univ. Sci. B 2016, 17, 361–366. [Google Scholar] [CrossRef]
- Dong, G.; Wang, M.; Gu, G.; Li, S.; Sun, X.; Li, Z.; Cai, H.; Zhu, Z. MACC1 and HGF are associated with survivalin patients with gastric cancer. Oncol. Lett. 2018, 15, 3207–3213. [Google Scholar] [CrossRef] [Green Version]
- Kobelt, D.; Perez-Hernandez, D.; Fleuter, C.; Dahlmann, M.; Zincke, F.; Smith, J.; Migotti, R.; Popp, O.; Burock, S.; Walther, W.; et al. The newly identified MEK1 tyrosine phosphorylation target MACC1 is druggable by approved MEK1 inhibitors to restrict colorectal cancer metastasis. Oncogene 2021, 40, 5286–5301. [Google Scholar] [CrossRef]
- Becker, M.; Nitsche, A.; Neumann, C.; Aumann, J.; Junghahn, I.; Fichtner, I. Sensitive PCR method for the detection and real-time quantification of human cells in xenotransplantation systems. Br. J. Cancer 2002, 87, 1328–1335. [Google Scholar] [CrossRef] [Green Version]
- Jin, Y.; Zhou, K.; Zhao, W.; Han, R.; Huo, X.; Yang, F.; Chen, J. Clinicopathological and prognostic significance of metastasis-associated in colon cancer-1 in gastric cancer: A meta-analysis. Int. J. Biol. Markers 2019, 34, 27–32. [Google Scholar] [CrossRef]
- Burock, S.; Herrmann, P.; Wendler, I.; Niederstrasser, M.; Wernecke, K.; Stein, U. Circulating metastasis associated in colon cancer 1 transcripts in gastric cancer patient plasma as diagnostic and prognostic biomarker. World J. Gastroenterol. 2015, 21, 333–341. [Google Scholar] [CrossRef]
- Lin, S.J.; Gagnon-Bartsch, J.A.; Tan, I.B.; Earle, S.; Ruff, L.; Pettinger, K.; Ylstra, B.; Van Grieken, N.; Rha, S.Y.; Chung, H.C.; et al. Signatures of tumour immunity distinguish Asian and non-Asian gastric adenocarcinomas. Gut 2015, 64, 1721–1731. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Y.; Bertagnolli, M.M. Comparison of Gastric Cancer Survival Between Caucasian and Asian Patients Treated in the United States: Results from the Surveillance Epidemiology and End Results (SEER) Database. Ann. Surg. Oncol. 2015, 22, 2965–2971. [Google Scholar] [CrossRef]
- Saijo, N. The role of pharmacoethnicity in the development of cytotoxic and molecular targeted drugs in oncology. Yonsei Med. J. 2013, 54, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Lui, F.H.; Tuan, B.; Swenson, S.L.; Wong, R.J. Ethnic disparities in gastric cancer incidence and survival in the USA: An updated analysis of 1992-2009 SEER data. Dig. Dis. Sci. 2014, 59, 3027–3034. [Google Scholar] [CrossRef]
- Ohtsu, A.; Shah, M.A.; Van Cutsem, E.; Rha, S.Y.; Sawaki, A.; Park, S.R.; Lim, H.Y.; Yamada, Y.; Wu, J.; Langer, B.; et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: A randomized, double-blind, placebo-controlled phase III study. J. Clin. Oncol. 2011, 29, 3968–3976. [Google Scholar] [CrossRef]
- Ge, S.-H.; Wu, X.; Wang, X.; Xing, X.-F.; Zhang, L.-H.; Zhu, Y.-B.; Du, H.; Dong, B.; Hu, Y.; Ji, J.-F. Over-expression of Metastasis-associated in Colon Cancer-1 (MACC1) Associates with Better Prognosis of Gastric Cancer Patients. Chin. J. Cancer Res. 2011, 23, 153–159. [Google Scholar] [CrossRef] [Green Version]
- Zheng, G.; You, C.; Qiu, Z.; Zheng, X. Expression of MACC1 protein in gastric cancer and its effect on proliferation and invasion of gastric cancer cells. Cell. Mol. Biol. 2020, 66, 111. [Google Scholar] [CrossRef]
- Janjigian, Y.Y.; Shitara, K.; Moehler, M.; Garrido, M.; Salman, P.; Shen, L.; Wyrwicz, L.; Yamaguchi, K.; Skoczylas, T.; Campos Bragagnoli, A.; et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): A randomised, open-label, phase 3 trial. Lancet 2021, 398, 27–40. [Google Scholar] [CrossRef]
- Bang, Y.-J.; Van Cutsem, E.; Feyereislova, A.; Chung, H.C.; Shen, L.; Sawaki, A.; Lordick, F.; Ohtsu, A.; Omuro, Y.; Satoh, T.; et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet 2010, 376, 687–697. [Google Scholar] [CrossRef]
- Abrahao-machado, L.F.; Scapulatempo-neto, C.; Abrahao-machado, L.F.; Scapulatempo-, C. 2016 Gastric Cancer: Global view HER2 testing in gastric cancer: An update. World J. Gastroenterol. 2016, 22, 4619–4625. [Google Scholar] [CrossRef]
- Schoemig-Markiefka, B.; Eschbach, J.; Scheel, A.H.; Pamuk, A.; Rueschoff, J.; Zander, T.; Buettner, R.; Schroeder, W.; Bruns, C.J.; Loeser, H.; et al. Optimized PD-L1 scoring of gastric cancer. Gastric Cancer 2021, 24, 1115–1122. [Google Scholar] [CrossRef]
- Ahn, S.; Brant, R.; Sharpe, A.; Dry, J.R.; Hodgson, D.R.; Kilgour, E.; Kim, K.; Kim, S.T.; Park, S.H.; Kang, W.K.; et al. Correlation between MEK signature and Ras gene alteration in advanced gastric cancer. Oncotarget 2017, 8, 107492–107499. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Kim, S.T.; Kozarewa, I.; Kim, H.K.; Kim, K.; Mortimer, P.G.; Hollingsworth, S.J.; Lee, J.; Park, M.; Park, S.H.; et al. Selumetinib plus docetaxel as second-line chemotherapy in KRAS mutant, KRAS amplified or MEK signatured gastric cancer patients: First arm of the umbrella trial in GC though the molecular screening, VIKTORY trial. J. Clin. Oncol. 2018, 36, 4061. [Google Scholar] [CrossRef]


| Total | MACC1 Low | Macc1 High | Exluced | p | |||||
|---|---|---|---|---|---|---|---|---|---|
| n | (%) | n | (%) | n | (%) | n | (%) | ||
| Gender | |||||||||
| Female | 131 | (36.4) | 14 | (10.7) | 82 | (62.6) | 35 | (26.7) | 0.083 |
| Male | 229 | (63.6) | 41 | (17.9) | 129 | (56.3) | 59 | (25.8) | |
| Age Group | |||||||||
| <65 years | 159 | (44.2) | 37 | (23.9) | 111 | (69.8) | 53 | (33.3) | 0.051 |
| ≥65 years | 201 | (55.8) | 18 | (9.0) | 100 | (49.8) | 41 | (20.4) | |
| Localization | |||||||||
| AEG | 60 | (83.3) | 5 | (8.3) | 41 | (68.3) | 14 | (23.3) | 0.161 |
| Stomach | 300 | (16.7) | 50 | (16.7) | 170 | (56.7) | 80 | (26.7) | |
| Tumor Stage | |||||||||
| T1 | 47 | (13.1) | 15 | (31.9) | 22 | (46.8) | 10 | (21.3) | 0.001 |
| T2 | 140 | (38.9) | 25 | (17.9) | 81 | (57.9) | 34 | (24.3) | |
| T3 | 122 | (33.9) | 10 | (8.2) | 86 | (70.5) | 26 | (21.3) | |
| T4 | 32 | (8.9) | 5 | (15.6) | 22 | (68.8) | 5 | (15.6) | |
| Tx | 19 | (5.3) | 0 | (0.0) | 0 | (0.0) | 19 | (100.0) | |
| Node Stage | |||||||||
| N0 | 105 | (29.29 | 19 | (18.1) | 51 | (48.6) | 35 | (33.3) | 0.293 |
| N1 | 108 | (30.0) | 18 | (16.7) | 65 | (60.2) | 25 | (23.1) | |
| N2 | 71 | (19.7) | 10 | (14.1) | 45 | (63.4) | 16 | (22.5) | |
| N3 | 75 | (20.8) | 8 | (10.7) | 50 | (66.7) | 17 | (22.7) | |
| Nx | 1 | (0.3) | - | - | - | - | - | - | |
| Distant Metastasis | |||||||||
| M0 | 245 | (68.1) | 48 | (19.6) | 149 | (60.8) | 48 | (19.6) | 0.012 |
| M1 | 115 | (31.9) | 7 | (6.1) | 62 | (53.9) | 46 | (40.0) | |
| Lymphatic Vessel Invasion | |||||||||
| L0 | 115 | (31.9) | 24 | (20.9) | 66 | (57.4) | 25 | (21.7) | 0.07 |
| L1 | 206 | (57.2) | 26 | (12.6) | 131 | (63.6) | 49 | (23.8) | |
| Unknown | 39 | (10.8) | - | - | - | - | - | - | |
| Vein Invasion | |||||||||
| V0 | 201 | (55.8) | 37 | (18.4) | 122 | (60.7) | 42 | (20.9) | 0.139 |
| V1 | 114 | (31.7) | 13 | (11.4) | 73 | (64.0) | 28 | (24.6) | |
| Unknown | 45 | (12.5) | - | - | - | - | - | - | |
| Grading | |||||||||
| G1 | 1 | (0.3) | 1 | (100.0) | 0 | (0.0) | 0 | (0.0) | 0.001 |
| G2 | 90 | (25.0) | 23 | (25.6) | 48 | (53.3) | 19 | (21.1) | |
| G3 | 266 | (73.9) | 29 | (10.9) | 71 | (26.7) | 166 | (62.4) | |
| Unknown | 3 | (0.8) | - | - | - | - | - | - | |
| Lauren Classification | |||||||||
| Intestinal | 122 | (33.9) | 26 | (21.3) | 69 | (56.6) | 27 | (22.1) | 0.048 |
| Diffuse | 190 | (52.8) | 19 | (10.0) | 114 | (60.0) | 57 | (30.0) | |
| Mixed | 45 | (12.5) | 8 | (17.8) | 27 | (60.0) | 10 | (22.2) | |
| Unknown | 3 | (0.8) | - | - | - | - | - | - | |
| Missmatch Repair System | |||||||||
| proficient | 278 | (77.2) | 49 | (17.6) | 183 | (65.8) | 46 | (16.5) | <0.0001 |
| deficient | 39 | (10.8) | 5 | (12.8) | 35 | (89.7) | 9 | (23.1) | |
| unkown | 43 | (11.9) | - | - | - | - | - | - | |
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| Median Survival Months (95% CI) | p | Odds Ratio | Low | High | p | |
| Tumor Stage | ||||||
| T1 | 181.19 (164.7–197.7) | <0.0001 | 1.798 | 1.467 | 2.203 | <0.0001 |
| T2 | 79.72 (67.2–92.2) | |||||
| T3 | 32.76 (23.5–42.1) | |||||
| T4 | 14.13 (9.3–19.0) | |||||
| Node Stage | ||||||
| N0 | 135.79 (121.3–150.3) | <0.0001 | 1.595 | 1.353 | 1.879 | <0.0001 |
| N1 | 64.32 (51.6–77.0) | |||||
| N2 | 24.06 (16.6–31.6) | |||||
| N3 | 17.92 (10.8–25.0) | |||||
| Nx | 13.00 (13.0–13.0) | |||||
| Lymphatic Vessel Invasion | ||||||
| L0 | 119.68 (103.3–136.0) | <0.0001 | 1.004 | 0.774 | 1.302 | 0.976 |
| L1 | 43.27 (34.9–51.6) | |||||
| Lx | 32.7 (15.9–49.1) | |||||
| Vein Invasion | ||||||
| V0 | 99.00 (86.2–111.8) | <0.0001 | 1.003 | 0.778 | 1.292 | 0.984 |
| V1 | 34.56 (24.7–44.4) | |||||
| Vx | 32.22 (16.6–47.8) | |||||
| Grading | ||||||
| 1 | - | 0.038 | 0.828 | 0.551 | 1.243 | 0.791 |
| 2 | 88.71 (72.7–104.7) | |||||
| 3 | 76.4 (65.8–87.0) | |||||
| Lauren Classification | ||||||
| intestinal | 95.97 (82.0–109.9) | 0.002 | 1.038 | 0.786 | 1.373 | 0.791 |
| diffuse | 69.83 (56.8–82.9) | |||||
| mixed | 69.40 (51.4–87.4) | |||||
| Mismatch Repair System | ||||||
| proficient | 47.22 (28.1–66.3) | 0.025 | 0.759 | 0.494 | 1.165 | 0.207 |
| deficient | 79.77 (70.2–89.4) | |||||
| MACC1 | ||||||
| low | 87.06 (66.5–107.6) | 0.002 | 1.513 | 1.013 | 2.258 | 0.043 |
| high | 54.20 (45.0–63.4) | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Treese, C.; Werchan, J.; von Winterfeld, M.; Berg, E.; Hummel, M.; Timm, L.; Rau, B.; Daberkow, O.; Walther, W.; Daum, S.; et al. Inhibition of MACC1-Induced Metastasis in Esophageal and Gastric Adenocarcinomas. Cancers 2022, 14, 1773. https://doi.org/10.3390/cancers14071773
Treese C, Werchan J, von Winterfeld M, Berg E, Hummel M, Timm L, Rau B, Daberkow O, Walther W, Daum S, et al. Inhibition of MACC1-Induced Metastasis in Esophageal and Gastric Adenocarcinomas. Cancers. 2022; 14(7):1773. https://doi.org/10.3390/cancers14071773
Chicago/Turabian StyleTreese, Christoph, Jessica Werchan, Moritz von Winterfeld, Erika Berg, Michael Hummel, Lena Timm, Beate Rau, Ole Daberkow, Wolfgang Walther, Severin Daum, and et al. 2022. "Inhibition of MACC1-Induced Metastasis in Esophageal and Gastric Adenocarcinomas" Cancers 14, no. 7: 1773. https://doi.org/10.3390/cancers14071773
APA StyleTreese, C., Werchan, J., von Winterfeld, M., Berg, E., Hummel, M., Timm, L., Rau, B., Daberkow, O., Walther, W., Daum, S., Kobelt, D., & Stein, U. (2022). Inhibition of MACC1-Induced Metastasis in Esophageal and Gastric Adenocarcinomas. Cancers, 14(7), 1773. https://doi.org/10.3390/cancers14071773








