Isolation, Maintenance and Expansion of Adult Hematopoietic Stem/Progenitor Cells and Leukemic Stem Cells
Abstract
Simple Summary
Abstract
1. The Adult HSC—A Rare, Self-Renewing Cell
| HSC Surface Markers | |||||
|---|---|---|---|---|---|
| Murine | Refs. | Human | Refs. | ||
| CD19, CD45R CD11b, Ly-6G CD3 | Lineage negative selection (Lin−) | [28,29,30] | CD45RA | CD45 isoform with specific molecular weight | [31] |
| CD117 | Type III transmembrane tyrosine kinase receptor (c-KIT) | [29,32,33,34] | CD38 | Cyclic ADP ribose hydrolase | [31,35] |
| SCA-1 | Lymphocyte activation protein-6A (Ly-6A/E) | [29,30,32,33] | CD49f | Integrin α-6 | [20,31] |
| CD48, CD150 | Signaling lymphocyte activation molecule (SLAM) family protein | [9,11,12,16] | CD90 | Thy1 | [30,31,36] |
| CD34 | Transmembrane phosphor-glycoprotein | [9,16,33,37] | CD34 | Transmembrane phosphor-glycoprotein | [29,36,38,39] |
| CD201 | Endothelial protein C receptor (EPCR) | [40,41] | CD201 | Endothelial protein C receptor (EPCR) | [41,42] |
| CD135 | Fms-like tyrosine kinase 3 receptor (FLT3-R); FLK2 | [9,16,33] | CD133 | AC133, Prominin-1 | [43,44] |
| GPI-80 | Glycosylphosphatidyl Inositol-Anchored Protein GPI-80 | [21,23] | |||
2. Leukemic Stem Cells (LSCs)—Villain to Its Heathy Counterpart
3. BM Niche of LT/ST-HSCs and LSCs
4. Essential Factors for HSC Quiescence and Self-Renewal—Cell Cycle Components as Mediators
5. Signaling and Metabolic Changes in LSCs
6. Major Challenges Culturing LT-HSCs
7. Maintaining Quiescent LT-HSCs
8. Expansion Techniques to Retain LT/ST-HSC Phenotype Ex Vivo
8.1. 2D Methods
8.1.1. Suspension Culture of Murine LT/ST-HSCs
8.1.2. Suspension Culture of Human LT/ST-HSCs
8.1.3. Suspension Culture of Human LSCs
8.2. 2.5D Methods
8.2.1. Co-Culturing LT/ST-HSCs
8.2.2. Co-Culturing LSCs
8.3. 3D Methods
8.3.1. 3D Culture of LT/ST-HSCs
8.3.2. 3D Culture of LSCs
9. Immortalized Hematopoietic Stem/Progenitor Cell Lines
9.1. Immortalization via HoxB8 and HoxB4
9.2. EML (Erythroid, Myeloid, and Lymphocytic) Cell Line
9.3. Immortalization via Lhx2—HPCLSK Cell Lines
9.4. Leukemic Stem Cell Lines Using the HPCLSK System
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Becker, A.J.; McCulloch, E.A.; Till, J.E. Cytological Demonstration of the Clonal Nature of Spleen Colonies Derived from Transplanted Mouse Marrow Cells. Nature 1963, 197, 452–454. [Google Scholar] [CrossRef]
- Cole, L.J.; Fishler, M.C.; Bond, V.P.S. Subcellular Fractionation of Mouse Spleen Radiation Protection Activity. Proc. Natl. Acad. Sci. USA 1953, 39, 759–772. [Google Scholar] [CrossRef]
- Till, J.E.; McCulloch, E.A. A Direct Measurement of the Radiation Sensitivity of Normal Mouse Bone Marrow Cells. Radiat. Res. 1961, 14, 213–222. [Google Scholar] [CrossRef]
- Morrison, S.J.; Weissman, I.L. The Long-Term Repopulating Subset of Hematopoietic Stem Cells Is Deterministic and Isolatable by Phenotype. Immunity 1994, 1, 661–673. [Google Scholar] [CrossRef]
- Bartelmez, S.H.; Andrews, R.G.; Bernstein, I.D. Uncovering the Heterogeneity of Hematopoietic Repopulating Cells. Exp. Hematol. 1991, 19, 861–862. [Google Scholar]
- Pietras, E.M.; Reynaud, D.; Kang, Y.-A.; Carlin, D.; Calero-Nieto, F.J.; Leavitt, A.D.; Stuart, J.M.; Göttgens, B.; Passegué, E. Functionally Distinct Subsets of Lineage-Biased Multipotent Progenitors Control Blood Production in Normal and Regenerative Conditions. Cell Stem Cell 2015, 17, 35–46. [Google Scholar] [CrossRef]
- Challen, G.A.; Pietras, E.M.; Wallscheid, N.C.; Signer, R.A.J. Simplified Murine Multipotent Progenitor Isolation Scheme: Establishing a Consensus Approach for Multipotent Progenitor Identification. Exp. Hematol. 2021, 104, 55–63. [Google Scholar] [CrossRef]
- Busch, K.; Klapproth, K.; Barile, M.; Flossdorf, M.; Holland-Letz, T.; Schlenner, S.M.; Reth, M.; Höfer, T.; Rodewald, H.-R. Fundamental Properties of Unperturbed Haematopoiesis from Stem Cells In Vivo. Nature 2015, 518, 542–546. [Google Scholar] [CrossRef]
- Cabezas-Wallscheid, N.; Klimmeck, D.; Hansson, J.; Lipka, D.B.; Reyes, A.; Wang, Q.; Weichenhan, D.; Lier, A.; von Paleske, L.; Renders, S.; et al. Identification of Regulatory Networks in HSCs and Their Immediate Progeny via Integrated Proteome, Transcriptome, and DNA Methylome Analysis. Cell Stem Cell 2014, 15, 507–522. [Google Scholar] [CrossRef]
- Kent, D.G.; Copley, M.R.; Benz, C.; Wöhrer, S.; Dykstra, B.J.; Ma, E.; Cheyne, J.; Zhao, Y.; Bowie, M.B.; Zhao, Y.; et al. Prospective Isolation and Molecular Characterization of Hematopoietic Stem Cells with Durable Self-Renewal Potential. Blood 2009, 113, 6342–6350. [Google Scholar] [CrossRef]
- Kiel, M.J.; Yilmaz, Ö.H.; Iwashita, T.; Yilmaz, O.H.; Terhorst, C.; Morrison, S.J. SLAM Family Receptors Distinguish Hematopoietic Stem and Progenitor Cells and Reveal Endothelial Niches for Stem Cells. Cell 2005, 121, 1109–1121. [Google Scholar] [CrossRef]
- Morita, Y.; Ema, H.; Nakauchi, H. Heterogeneity and Hierarchy within the Most Primitive Hematopoietic Stem Cell Compartment. J. Exp. Med. 2010, 207, 1173–1182. [Google Scholar] [CrossRef]
- Oguro, H.; Ding, L.; Morrison, S.J. SLAM Family Markers Resolve Functionally Distinct Subpopulations of Hematopoietic Stem Cells and Multipotent Progenitors. Cell Stem Cell 2013, 13, 102–116. [Google Scholar] [CrossRef]
- Rabe, J.L.; Hernandez, G.; Chavez, J.S.; Mills, T.S.; Nerlov, C.; Pietras, E.M. CD34 and EPCR Coordinately Enrich Functional Murine Hematopoietic Stem Cells under Normal and Inflammatory Conditions. Exp. Hematol. 2020, 81, 1–15.e6. [Google Scholar] [CrossRef]
- Sawai, C.M.; Babovic, S.; Upadhaya, S.; Knapp, D.J.H.F.; Lavin, Y.; Lau, C.M.; Goloborodko, A.; Feng, J.; Fujisaki, J.; Ding, L.; et al. Hematopoietic Stem Cells Are the Major Source of Multilineage Hematopoiesis in Adult Animals. Immunity 2016, 45, 597–609. [Google Scholar] [CrossRef]
- Wilson, A.; Laurenti, E.; Oser, G.; van der Wath, R.C.; Blanco-Bose, W.; Jaworski, M.; Offner, S.; Dunant, C.F.; Eshkind, L.; Bockamp, E.; et al. Hematopoietic Stem Cells Reversibly Switch from Dormancy to Self-Renewal during Homeostasis and Repair. Cell 2008, 135, 1118–1129. [Google Scholar] [CrossRef]
- Yamamoto, R.; Morita, Y.; Ooehara, J.; Hamanaka, S.; Onodera, M.; Rudolph, K.L.; Ema, H.; Nakauchi, H. Clonal Analysis Unveils Self-Renewing Lineage-Restricted Progenitors Generated Directly from Hematopoietic Stem Cells. Cell 2013, 154, 1112–1126. [Google Scholar] [CrossRef]
- Fares, I.; Chagraoui, J.; Lehnertz, B.; MacRae, T.; Mayotte, N.; Tomellini, E.; Aubert, L.; Roux, P.P.; Sauvageau, G. EPCR Expression Marks UM171-Expanded CD34+ Cord Blood Stem Cells. Blood 2017, 129, 3344–3351. [Google Scholar] [CrossRef]
- Gordon, P.R.; Leimig, T.; Babarin-Dorner, A.; Houston, J.; Holladay, M.; Mueller, I.; Geiger, T.; Handgretinger, R. Large-Scale Isolation of CD133+ Progenitor Cells from G-CSF Mobilized Peripheral Blood Stem Cells. Bone Marrow Transplant. 2003, 31, 17–22. [Google Scholar] [CrossRef][Green Version]
- Notta, F.; Doulatov, S.; Laurenti, E.; Poeppl, A.; Jurisica, I.; Dick, J.E. Isolation of Single Human Hematopoietic Stem Cells Capable of Long-Term Multilineage Engraftment. Science 2011, 333, 218–221. [Google Scholar] [CrossRef]
- Prashad, S.L.; Calvanese, V.; Yao, C.Y.; Kaiser, J.; Wang, Y.; Sasidharan, R.; Crooks, G.; Magnusson, M.; Mikkola, H.K.A. GPI-80 Defines Self-Renewal Ability in Hematopoietic Stem Cells during Human Development. Cell Stem Cell 2015, 16, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Radtke, S.; Pande, D.; Cui, M.; Perez, A.M.; Chan, Y.-Y.; Enstrom, M.; Schmuck, S.; Berger, A.; Eunson, T.; Adair, J.E.; et al. Purification of Human CD34+CD90+ HSCs Reduces Target Cell Population and Improves Lentiviral Transduction for Gene Therapy. Mol. Ther.-Methods Clin. Dev. 2020, 18, 679–691. [Google Scholar] [CrossRef] [PubMed]
- Sumide, K.; Matsuoka, Y.; Kawamura, H.; Nakatsuka, R.; Fujioka, T.; Asano, H.; Takihara, Y.; Sonoda, Y. A Revised Road Map for the Commitment of Human Cord Blood CD34-Negative Hematopoietic Stem Cells. Nat. Commun. 2018, 9, 2202. [Google Scholar] [CrossRef] [PubMed]
- Zonari, E.; Desantis, G.; Petrillo, C.; Boccalatte, F.E.; Lidonnici, M.R.; Kajaste-Rudnitski, A.; Aiuti, A.; Ferrari, G.; Naldini, L.; Gentner, B. Efficient Ex Vivo Engineering and Expansion of Highly Purified Human Hematopoietic Stem and Progenitor Cell Populations for Gene Therapy. Stem Cell Rep. 2017, 8, 977–990. [Google Scholar] [CrossRef]
- Dussiau, C.; Boussaroque, A.; Gaillard, M.; Bravetti, C.; Zaroili, L.; Knosp, C.; Friedrich, C.; Asquier, P.; Willems, L.; Quint, L.; et al. Hematopoietic Differentiation Is Characterized by a Transient Peak of Entropy at a Single-Cell Level. BMC Biol. 2022, 20, 60. [Google Scholar] [CrossRef]
- Laurenti, E.; Göttgens, B. From Haematopoietic Stem Cells to Complex Differentiation Landscapes. Nature 2018, 553, 418–426. [Google Scholar] [CrossRef]
- Velten, L.; Haas, S.F.; Raffel, S.; Blaszkiewicz, S.; Islam, S.; Hennig, B.P.; Hirche, C.; Lutz, C.; Buss, E.C.; Nowak, D.; et al. Human Haematopoietic Stem Cell Lineage Commitment Is a Continuous Process. Nat. Cell Biol. 2017, 19, 271–281. [Google Scholar] [CrossRef]
- Morrison, S.; Lagasse, E.; Weissman, I. Demonstration That Thy(Lo) Subsets of Mouse Bone Marrow That Express High Levels of Lineage Markers Are Not Significant Hematopoietic Progenitors. Blood 1994, 83, 3480–3490. [Google Scholar] [CrossRef]
- Osawa, M.; Hanada, K.; Hamada, H.; Nakauchi, H. Long-Term Lymphohematopoietic Reconstitution by a Single CD34-Low/Negative Hematopoietic Stem Cell. Science 1996, 273, 242–245. [Google Scholar] [CrossRef]
- Spangrude, G.J.; Heimfeld, S.; Weissman, I.L. Purification and Characterization of Mouse Hematopoietic Stem Cells. Science 1988, 241, 58–62. [Google Scholar] [CrossRef]
- Majeti, R.; Park, C.Y.; Weissman, I.L. Identification of a Hierarchy of Multipotent Hematopoietic Progenitors in Human Cord Blood. Cell Stem Cell 2007, 1, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Okada, S.; Nakauchi, H.; Nagayoshi, K.; Nishikawa, S.; Miura, Y.; Suda, T. In Vivo and in Vitro Stem Cell Function of C-Kit- and Sca-1-Positive Murine Hematopoietic Cells. Blood 1992, 80, 3044–3050. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Bryder, D.; Adolfsson, J.; Nygren, J.; Månsson, R.; Sigvardsson, M.; Jacobsen, S.E.W. Identification of Lin–Sca1+kit+CD34+Flt3– Short-Term Hematopoietic Stem Cells Capable of Rapidly Reconstituting and Rescuing Myeloablated Transplant Recipients. Blood 2005, 105, 2717–2723. [Google Scholar] [CrossRef] [PubMed]
- Kent, D.; Copley, M.; Benz, C.; Dykstra, B.; Bowie, M.; Eaves, C. Regulation of Hematopoietic Stem Cells by the Steel Factor/KIT Signaling Pathway. Clin. Cancer Res. 2008, 14, 1926–1930. [Google Scholar] [CrossRef]
- Deaglio, S.; Mehta, K.; Malavasi, F. Human CD38: A (r)Evolutionary Story of Enzymes and Receptors. Leuk. Res. 2001, 25, 1–12. [Google Scholar] [CrossRef]
- Baum, C.M.; Weissman, I.L.; Tsukamoto, A.S.; Buckle, A.M.; Peault, B. Isolation of a Candidate Human Hematopoietic Stem-Cell Population. Proc. Natl. Acad. Sci. USA 1992, 89, 2804–2808. [Google Scholar] [CrossRef]
- Matsubara, A.; Iwama, A.; Yamazaki, S.; Furuta, C.; Hirasawa, R.; Morita, Y.; Osawa, M.; Motohashi, T.; Eto, K.; Ema, H.; et al. Endomucin, a CD34-like Sialomucin, Marks Hematopoietic Stem Cells throughout Development. J. Exp. Med. 2005, 202, 1483–1492. [Google Scholar] [CrossRef]
- Civin, C.I.; Strauss, L.C.; Brovall, C.; Fackler, M.J.; Schwartz, J.F.; Shaper, J.H. Antigenic Analysis of Hematopoiesis. III. A Hematopoietic Progenitor Cell Surface Antigen Defined by a Monoclonal Antibody Raised against KG-1a Cells. J. Immunol. 1984, 133, 157–165. [Google Scholar]
- Tjrannfjord, G.E.; Steen, R.; Egeland, T. Characterization of CD34+ Peripheral Blood Cells from Healthy Adults Mobilized by Recombinant Human Granulocyte Colony-Stimulating Factor. Blood 1994, 84, 2795–2801. [Google Scholar] [CrossRef]
- Balazs, A.B.; Fabian, A.J.; Esmon, C.T.; Mulligan, R.C. Endothelial Protein C Receptor (CD201) Explicitly Identifies Hematopoietic Stem Cells in Murine Bone Marrow. Blood 2006, 107, 2317–2321. [Google Scholar] [CrossRef]
- Fukudome, K.; Esmon, C.T. Molecular Cloning and Expression of Murine and Bovine Endothelial Cell Protein C/Activated Protein C Receptor (EPCR). J. Biol. Chem. 1995, 270, 5571–5577. [Google Scholar] [CrossRef]
- Martin, G.H.; Park, C.Y. EPCR: A Novel Marker of Cultured Cord Blood HSCs. Blood 2017, 129, 3279–3280. [Google Scholar] [CrossRef]
- Gallacher, L.; Murdoch, B.; Wu, D.M.; Karanu, F.N.; Keeney, M.; Bhatia, M. Isolation and Characterization of Human CD34−Lin− and CD34+Lin− Hematopoietic Stem Cells Using Cell Surface Markers AC133 and CD7. Blood 2000, 95, 2813–2820. [Google Scholar] [CrossRef]
- Shmelkov, S.V.; St.Clair, R.; Lyden, D.; Rafii, S. AC133/CD133/Prominin-1. Int. J. Biochem. Cell Biol. 2005, 37, 715–719. [Google Scholar] [CrossRef]
- Vetrie, D.; Helgason, G.V.; Copland, M. The Leukaemia Stem Cell: Similarities, Differences and Clinical Prospects in CML and AML. Nat. Rev. Cancer 2020, 20, 158–173. [Google Scholar] [CrossRef]
- Holyoake, T.; Jiang, X.; Eaves, C.; Eaves, A. Isolation of a Highly Quiescent Subpopulation of Primitive Leukemic Cells in Chronic Myeloid Leukemia. Blood 1999, 94, 2056–2064. [Google Scholar] [CrossRef]
- Lapidot, T.; Sirard, C.; Vormoor, J.; Murdoch, B.; Hoang, T.; Caceres-Cortes, J.; Minden, M.; Paterson, B.; Caligiuri, M.A.; Dick, J.E. A Cell Initiating Human Acute Myeloid Leukaemia after Transplantation into SCID Mice. Nature 1994, 367, 645–648. [Google Scholar] [CrossRef]
- Guan, Y.; Gerhard, B.; Hogge, D.E. Detection, Isolation, and Stimulation of Quiescent Primitive Leukemic Progenitor Cells from Patients with Acute Myeloid Leukemia (AML). Blood 2003, 101, 3142–3149. [Google Scholar] [CrossRef]
- Schwartz, D.M.; Farley, T.K.; Richoz, N.; Yao, C.; Shih, H.-Y.; Petermann, F.; Zhang, Y.; Sun, H.-W.; Hayes, E.; Mikami, Y.; et al. Retinoic Acid Receptor Alpha Represses a Th9 Transcriptional and Epigenomic Program to Reduce Allergic Pathology. Immunity 2019, 50, 106–120.e10. [Google Scholar] [CrossRef]
- Zhang, B.; Li, L.; Ho, Y.; Li, M.; Marcucci, G.; Tong, W.; Bhatia, R. Heterogeneity of Leukemia-Initiating Capacity of Chronic Myelogenous Leukemia Stem Cells. J. Clin. Investig. 2016, 126, 975–991. [Google Scholar] [CrossRef]
- Holyoake, T.L.; Vetrie, D. The Chronic Myeloid Leukemia Stem Cell: Stemming the Tide of Persistence. Blood 2017, 129, 1595–1606. [Google Scholar] [CrossRef] [PubMed]
- Pollyea, D.A.; Jordan, C.T. Therapeutic Targeting of Acute Myeloid Leukemia Stem Cells. Blood 2017, 129, 1627–1635. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.; Majeti, R. Biology and Relevance of Human Acute Myeloid Leukemia Stem Cells. Blood 2017, 129, 1577–1585. [Google Scholar] [CrossRef] [PubMed]
- Griessinger, E.; Vargaftig, J.; Horswell, S.; Taussig, D.C.; Gribben, J.; Bonnet, D. Acute Myeloid Leukemia Xenograft Success Prediction: Saving Time. Exp. Hematol. 2018, 59, 66–71.e4. [Google Scholar] [CrossRef]
- Pearce, D.J.; Taussig, D.; Zibara, K.; Smith, L.-L.; Ridler, C.M.; Preudhomme, C.; Young, B.D.; Rohatiner, A.Z.; Lister, T.A.; Bonnet, D. AML Engraftment in the NOD/SCID Assay Reflects the Outcome of AML: Implications for Our Understanding of the Heterogeneity of AML. Blood 2006, 107, 1166–1173. [Google Scholar] [CrossRef]
- Ran, D.; Schubert, M.; Pietsch, L.; Taubert, I.; Wuchter, P.; Eckstein, V.; Bruckner, T.; Zoeller, M.; Ho, A.D. Aldehyde Dehydrogenase Activity among Primary Leukemia Cells Is Associated with Stem Cell Features and Correlates with Adverse Clinical Outcomes. Exp. Hematol. 2009, 37, 1423–1434. [Google Scholar] [CrossRef]
- Gerber, J.M.; Qin, L.; Kowalski, J.; Smith, B.D.; Griffin, C.A.; Vala, M.S.; Collector, M.I.; Perkins, B.; Zahurak, M.; Matsui, W.; et al. Characterization of Chronic Myeloid Leukemia Stem Cells. Am. J. Hematol. 2011, 86, 31–37. [Google Scholar] [CrossRef]
- Hehlmann, R. How I Treat CML Blast Crisis. Blood 2012, 120, 737–747. [Google Scholar] [CrossRef]
- Tanizaki, R.; Nomura, Y.; Miyata, Y.; Minami, Y.; Abe, A.; Hanamura, A.; Sawa, M.; Murata, M.; Kiyoi, H.; Matsushita, T.; et al. Irrespective of CD34 Expression, Lineage-Committed Cell Fraction Reconstitutes and Re-Establishes Transformed Philadelphia Chromosome-Positive Leukemia in NOD/SCID/IL-2Rγc−/− Mice. Cancer Sci. 2010, 101, 631–638. [Google Scholar] [CrossRef]
- Bonnet, D.; Dick, J.E. Human Acute Myeloid Leukemia Is Organized as a Hierarchy That Originates from a Primitive Hematopoietic Cell. Nat. Med. 1997, 3, 730–737. [Google Scholar] [CrossRef]
- Hope, K.J.; Jin, L.; Dick, J.E. Acute Myeloid Leukemia Originates from a Hierarchy of Leukemic Stem Cell Classes That Differ in Self-Renewal Capacity. Nat. Immunol. 2004, 5, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Eppert, K.; Takenaka, K.; Lechman, E.R.; Waldron, L.; Nilsson, B.; van Galen, P.; Metzeler, K.H.; Poeppl, A.; Ling, V.; Beyene, J.; et al. Stem Cell Gene Expression Programs Influence Clinical Outcome in Human Leukemia. Nat. Med. 2011, 17, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Taussig, D.C.; Vargaftig, J.; Miraki-Moud, F.; Griessinger, E.; Sharrock, K.; Luke, T.; Lillington, D.; Oakervee, H.; Cavenagh, J.; Agrawal, S.G.; et al. Leukemia-Initiating Cells from Some Acute Myeloid Leukemia Patients with Mutated Nucleophosmin Reside in the CD34(−) Fraction. Blood 2010, 115, 1976–1984. [Google Scholar] [CrossRef] [PubMed]
- Goardon, N.; Marchi, E.; Atzberger, A.; Quek, L.; Schuh, A.; Soneji, S.; Woll, P.; Mead, A.; Alford, K.A.; Rout, R.; et al. Coexistence of LMPP-like and GMP-like Leukemia Stem Cells in Acute Myeloid Leukemia. Cancer Cell 2011, 19, 138–152. [Google Scholar] [CrossRef]
- Kirstetter, P.; Schuster, M.B.; Bereshchenko, O.; Moore, S.; Dvinge, H.; Kurz, E.; Theilgaard-Mönch, K.; Månsson, R.; Pedersen, T.Å.; Pabst, T.; et al. Modeling of C/EBPα Mutant Acute Myeloid Leukemia Reveals a Common Expression Signature of Committed Myeloid Leukemia-Initiating Cells. Cancer Cell 2008, 13, 299–310. [Google Scholar] [CrossRef]
- Somervaille, T.C.P.; Matheny, C.J.; Spencer, G.J.; Iwasaki, M.; Rinn, J.L.; Witten, D.M.; Chang, H.Y.; Shurtleff, S.A.; Downing, J.R.; Cleary, M.L. Hierarchical Maintenance of MLL Myeloid Leukemia Stem Cells Employs a Transcriptional Program Shared with Embryonic Rather Than Adult Stem Cells. Cell Stem Cell 2009, 4, 129–140. [Google Scholar] [CrossRef]
- Somervaille, T.C.P.; Cleary, M.L. Identification and Characterization of Leukemia Stem Cells in Murine MLL-AF9 Acute Myeloid Leukemia. Cancer Cell 2006, 10, 257–268. [Google Scholar] [CrossRef]
- Ishikawa, F.; Yoshida, S.; Saito, Y.; Hijikata, A.; Kitamura, H.; Tanaka, S.; Nakamura, R.; Tanaka, T.; Tomiyama, H.; Saito, N.; et al. Chemotherapy-Resistant Human AML Stem Cells Home to and Engraft within the Bone-Marrow Endosteal Region. Nat. Biotechnol. 2007, 25, 1315–1321. [Google Scholar] [CrossRef]
- Quek, L.; Otto, G.W.; Garnett, C.; Lhermitte, L.; Karamitros, D.; Stoilova, B.; Lau, I.-J.; Doondeea, J.; Usukhbayar, B.; Kennedy, A.; et al. Genetically Distinct Leukemic Stem Cells in Human CD34− Acute Myeloid Leukemia Are Arrested at a Hemopoietic Precursor-like Stage. J. Exp. Med. 2016, 213, 1513–1535. [Google Scholar] [CrossRef]
- Huntly, B.J.P.; Shigematsu, H.; Deguchi, K.; Lee, B.H.; Mizuno, S.; Duclos, N.; Rowan, R.; Amaral, S.; Curley, D.; Williams, I.R.; et al. MOZ-TIF2, but Not BCR-ABL, Confers Properties of Leukemic Stem Cells to Committed Murine Hematopoietic Progenitors. Cancer Cell 2004, 6, 587–596. [Google Scholar] [CrossRef]
- Sadovnik, I.; Hoelbl-Kovacic, A.; Herrmann, H.; Eisenwort, G.; Cerny-Reiterer, S.; Warsch, W.; Hoermann, G.; Greiner, G.; Blatt, K.; Peter, B.; et al. Identification of CD25 as STAT5-Dependent Growth Regulator of Leukemic Stem Cells in Ph+ CML. Clin. Cancer Res. 2016, 22, 2051–2061. [Google Scholar] [CrossRef] [PubMed]
- Sadovnik, I.; Herrmann, H.; Eisenwort, G.; Blatt, K.; Hoermann, G.; Mueller, N.; Sperr, W.R.; Valent, P. Expression of CD25 on Leukemic Stem Cells in BCR-ABL1 + CML: Potential Diagnostic Value and Functional Implications. Exp. Hematol. 2017, 51, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.; Kitamura, H.; Hijikata, A.; Tomizawa-Murasawa, M.; Tanaka, S.; Takagi, S.; Uchida, N.; Suzuki, N.; Sone, A.; Najima, Y.; et al. Identification of Therapeutic Targets for Quiescent, Chemotherapy-Resistant Human Leukemia Stem Cells. Sci. Transl. Med. 2010, 2, 17ra9. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, H.; Cerny-Reiterer, S.; Gleixner, K.V.; Blatt, K.; Herndlhofer, S.; Rabitsch, W.; Jager, E.; Mitterbauer-Hohendanner, G.; Streubel, B.; Selzer, E.; et al. CD34+/CD38- Stem Cells in Chronic Myeloid Leukemia Express Siglec-3 (CD33) and Are Responsive to the CD33-Targeting Drug Gemtuzumab/Ozogamicin. Haematologica 2012, 97, 219–226. [Google Scholar] [CrossRef]
- Herrmann, H.; Sadovnik, I.; Eisenwort, G.; Rülicke, T.; Blatt, K.; Herndlhofer, S.; Willmann, M.; Stefanzl, G.; Baumgartner, S.; Greiner, G.; et al. Delineation of Target Expression Profiles in CD34+/CD38− and CD34+/CD38+ Stem and Progenitor Cells in AML and CML. Blood Adv. 2020, 4, 5118–5132. [Google Scholar] [CrossRef]
- Jilani, I.; Estey, E.; Huh, Y.; Joe, Y.; Manshouri, T.; Yared, M.; Giles, F.; Kantarjian, H.; Cortes, J.; Thomas, D.; et al. Differences in CD33 Intensity Between Various Myeloid Neoplasms. Am. J. Clin. Pathol. 2002, 118, 560–566. [Google Scholar] [CrossRef]
- Kersten, B.; Valkering, M.; Wouters, R.; van Amerongen, R.; Hanekamp, D.; Kwidama, Z.; Valk, P.; Ossenkoppele, G.; Zeijlemaker, W.; Kaspers, G.; et al. CD45RA, a Specific Marker for Leukaemia Stem Cell Sub-Populations in Acute Myeloid Leukaemia. Br. J. Haematol. 2016, 173, 219–235. [Google Scholar] [CrossRef]
- Wisniewski, D.; Affer, M.; Willshire, J.; Clarkson, B. Further Phenotypic Characterization of the Primitive Lineage− CD34+CD38−CD90+CD45RA− Hematopoietic Stem Cell/Progenitor Cell Sub-Population Isolated from Cord Blood, Mobilized Peripheral Blood and Patients with Chronic Myelogenous Leukemia. Blood Cancer J. 2011, 1, e36. [Google Scholar] [CrossRef]
- Greenlee-Wacker, M.C.; Galvan, M.D.; Bohlson, S.S. CD93: Recent Advances and Implications in Disease. Curr. Drug Targets 2012, 13, 411–420. [Google Scholar] [CrossRef]
- Inoue, A.; Kobayashi, C.I.; Shinohara, H.; Miyamoto, K.; Yamauchi, N.; Yuda, J.; Akao, Y.; Minami, Y. Chronic Myeloid Leukemia Stem Cells and Molecular Target Therapies for Overcoming Resistance and Disease Persistence. Int. J. Hematol. 2018, 108, 365–370. [Google Scholar] [CrossRef]
- Kinstrie, R.; Horne, G.A.; Morrison, H.; Irvine, D.; Munje, C.; Castañeda, E.G.; Moka, H.A.; Dunn, K.; Cassels, J.E.; Parry, N.; et al. CD93 Is Expressed on Chronic Myeloid Leukemia Stem Cells and Identifies a Quiescent Population Which Persists after Tyrosine Kinase Inhibitor Therapy. Leukemia 2020, 34, 1613–1625. [Google Scholar] [CrossRef] [PubMed]
- Charrin, S.; Manié, S.; Billard, M.; Ashman, L.; Gerlier, D.; Boucheix, C.; Rubinstein, E. Multiple Levels of Interactions within the Tetraspanin Web. Biochem. Biophys. Res. Commun. 2003, 304, 107–112. [Google Scholar] [CrossRef]
- Touzet, L.; Dumezy, F.; Roumier, C.; Berthon, C.; Bories, C.; Quesnel, B.; Preudhomme, C.; Boyer, T. CD9 in Acute Myeloid Leukemia: Prognostic Role and Usefulness to Target Leukemic Stem Cells. Cancer Med. 2019, 8, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- van Rhenen, A.; van Dongen, G.A.M.S.; Kelder, A.; Rombouts, E.J.; Feller, N.; Moshaver, B.; Walsum, M.S.; Zweegman, S.; Ossenkoppele, G.J.; Jan Schuurhuis, G. The Novel AML Stem Cell–Associated Antigen CLL-1 Aids in Discrimination between Normal and Leukemic Stem Cells. Blood 2007, 110, 2659–2666. [Google Scholar] [CrossRef]
- Askmyr, M.; Ågerstam, H.; Hansen, N.; Gordon, S.; Arvanitakis, A.; Rissler, M.; Juliusson, G.; Richter, J.; Järås, M.; Fioretos, T. Selective Killing of Candidate AML Stem Cells by Antibody Targeting of IL1RAP. Blood 2013, 121, 3709–3713. [Google Scholar] [CrossRef]
- Jaras, M.; Johnels, P.; Hansen, N.; Agerstam, H.; Tsapogas, P.; Rissler, M.; Lassen, C.; Olofsson, T.; Bjerrum, O.W.; Richter, J.; et al. Isolation and Killing of Candidate Chronic Myeloid Leukemia Stem Cells by Antibody Targeting of IL-1 Receptor Accessory Protein. Proc. Natl. Acad. Sci. USA 2010, 107, 16280–16285. [Google Scholar] [CrossRef]
- Huang, S.; Liu, Y.; Chen, Y.; Yeh, Y.; Huang, H. CD69 Partially Inhibits Apoptosis and Erythroid Differentiation via CD24, and Their Knockdown Increase Imatinib Sensitivity in BCR-ABL-positive Cells. J. Cell. Physiol. 2018, 233, 7467–7479. [Google Scholar] [CrossRef]
- Landberg, N.; von Palffy, S.; Askmyr, M.; Lilljebjörn, H.; Sandén, C.; Rissler, M.; Mustjoki, S.; Hjorth-Hansen, H.; Richter, J.; Ågerstam, H.; et al. CD36 Defines Primitive Chronic Myeloid Leukemia Cells Less Responsive to Imatinib but Vulnerable to Antibody-Based Therapeutic Targeting. Haematologica 2018, 103, 447–455. [Google Scholar] [CrossRef]
- Sachs, K.; Sarver, A.L.; Noble-Orcutt, K.E.; LaRue, R.S.; Antony, M.L.; Chang, D.; Lee, Y.; Navis, C.M.; Hillesheim, A.L.; Nykaza, I.R.; et al. Single-Cell Gene Expression Analyses Reveal Distinct Self-Renewing and Proliferating Subsets in the Leukemia Stem Cell Compartment in Acute Myeloid Leukemia. Cancer Res. 2020, 80, 458–470. [Google Scholar] [CrossRef]
- O’Reilly, E.; Zeinabad, H.A.; Szegezdi, E. Hematopoietic versus Leukemic Stem Cell Quiescence: Challenges and Therapeutic Opportunities. Blood Rev. 2021, 50, 100850. [Google Scholar] [CrossRef]
- Zhao, M.; Li, L. Dissecting the Bone Marrow HSC Niches. Cell Res. 2016, 26, 975–976. [Google Scholar] [CrossRef] [PubMed]
- Crane, G.M.; Jeffery, E.; Morrison, S.J. Adult Haematopoietic Stem Cell Niches. Nat. Rev. Immunol. 2017, 17, 573–590. [Google Scholar] [CrossRef]
- Kunisaki, Y.; Bruns, I.; Scheiermann, C.; Ahmed, J.; Pinho, S.; Zhang, D.; Mizoguchi, T.; Wei, Q.; Lucas, D.; Ito, K.; et al. Arteriolar Niches Maintain Haematopoietic Stem Cell Quiescence. Nature 2013, 502, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Acar, M.; Kocherlakota, K.S.; Murphy, M.M.; Peyer, J.G.; Oguro, H.; Inra, C.N.; Jaiyeola, C.; Zhao, Z.; Luby-Phelps, K.; Morrison, S.J. Deep Imaging of Bone Marrow Shows Non-Dividing Stem Cells Are Mainly Perisinusoidal. Nature 2015, 526, 126–130. [Google Scholar] [CrossRef]
- Poulos, M.G.; Guo, P.; Kofler, N.M.; Pinho, S.; Gutkin, M.C.; Tikhonova, A.; Aifantis, I.; Frenette, P.S.; Kitajewski, J.; Rafii, S.; et al. Endothelial Jagged-1 Is Necessary for Homeostatic and Regenerative Hematopoiesis. Cell Rep. 2013, 4, 1022–1034. [Google Scholar] [CrossRef] [PubMed]
- Winkler, I.G.; Barbier, V.; Nowlan, B.; Jacobsen, R.N.; Forristal, C.E.; Patton, J.T.; Magnani, J.L.; Lévesque, J.-P. Vascular Niche E-Selectin Regulates Hematopoietic Stem Cell Dormancy, Self Renewal and Chemoresistance. Nat. Med. 2012, 18, 1651–1657. [Google Scholar] [CrossRef]
- Galán-Díez, M.; Kousteni, S. The Osteoblastic Niche in Hematopoiesis and Hematological Myeloid Malignancies. Curr. Mol. Biol. Rep. 2017, 3, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Itkin, T.; Ludin, A.; Gradus, B.; Gur-Cohen, S.; Kalinkovich, A.; Schajnovitz, A.; Ovadya, Y.; Kollet, O.; Canaani, J.; Shezen, E.; et al. FGF-2 Expands Murine Hematopoietic Stem and Progenitor Cells via Proliferation of Stromal Cells, c-Kit Activation, and CXCL12 down-Regulation. Blood 2012, 120, 1843–1855. [Google Scholar] [CrossRef]
- Takam Kamga, P.; Bazzoni, R.; Dal Collo, G.; Cassaro, A.; Tanasi, I.; Russignan, A.; Tecchio, C.; Krampera, M. The Role of Notch and Wnt Signaling in MSC Communication in Normal and Leukemic Bone Marrow Niche. Front. Cell Dev. Biol. 2021, 8, 599276. [Google Scholar] [CrossRef]
- Yamazaki, S.; Ema, H.; Karlsson, G.; Yamaguchi, T.; Miyoshi, H.; Shioda, S.; Taketo, M.M.; Karlsson, S.; Iwama, A.; Nakauchi, H. Nonmyelinating Schwann Cells Maintain Hematopoietic Stem Cell Hibernation in the Bone Marrow Niche. Cell 2011, 147, 1146–1158. [Google Scholar] [CrossRef]
- Arai, F.; Hosokawa, K.; Toyama, H.; Matsumoto, Y.; Suda, T. Role of N-Cadherin in the Regulation of Hematopoietic Stem Cells in the Bone Marrow Niche: Function of N-Cadherin in the Regulation of HSPCs. Ann. N. Y. Acad. Sci. 2012, 1266, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Zhou, H.; Nemes, K.; Yen, D.; Zhao, W.; Bramlett, C.; Wang, B.; Lu, R.; Shen, K. Membrane-Bound SCF and VCAM-1 Synergistically Regulate the Morphology of Hematopoietic Stem Cells. J. Cell Biol. 2021, 220, e202010118. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, M.A.; DiPersio, J.F. Mobilization of Hematopoietic Stem and Leukemia Cells. J. Leukoc. Biol. 2012, 91, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Coulombel, L.; Auffray, I.; Gaugler, M.-H.; Rosemblatt, M. Expression and Function of Integrins on Hematopoietic Progenitor Cells. Acta Haematol. 1997, 97, 13–21. [Google Scholar] [CrossRef]
- Lee-Thedieck, C.; Spatz, J.P. Biophysical Regulation of Hematopoietic Stem Cells. Biomater. Sci. 2014, 2, 1548–1561. [Google Scholar] [CrossRef]
- Martino, F.; Perestrelo, A.R.; Vinarský, V.; Pagliari, S.; Forte, G. Cellular Mechanotransduction: From Tension to Function. Front. Physiol. 2018, 9, 824. [Google Scholar] [CrossRef]
- Alonso, J.L.; Goldmann, W.H. Cellular mechanotransduction. AIMS Biophys. 2016, 3, 50–62. [Google Scholar] [CrossRef]
- Riether, C.; Schürch, C.M.; Ochsenbein, A.F. Regulation of Hematopoietic and Leukemic Stem Cells by the Immune System. Cell Death Differ. 2015, 22, 187–198. [Google Scholar] [CrossRef]
- Schepers, K.; Pietras, E.M.; Reynaud, D.; Flach, J.; Binnewies, M.; Garg, T.; Wagers, A.J.; Hsiao, E.C.; Passegué, E. Myeloproliferative Neoplasia Remodels the Endosteal Bone Marrow Niche into a Self-Reinforcing Leukemic Niche. Cell Stem Cell 2013, 13, 285–299. [Google Scholar] [CrossRef]
- Sipkins, D.A.; Wei, X.; Wu, J.W.; Runnels, J.M.; Côté, D.; Means, T.K.; Luster, A.D.; Scadden, D.T.; Lin, C.P. In Vivo Imaging of Specialized Bone Marrow Endothelial Microdomains for Tumour Engraftment. Nature 2005, 435, 969–973. [Google Scholar] [CrossRef]
- Brenner, A.K.; Nepstad, I.; Bruserud, Ø. Mesenchymal Stem Cells Support Survival and Proliferation of Primary Human Acute Myeloid Leukemia Cells through Heterogeneous Molecular Mechanisms. Front. Immunol. 2017, 8, 106. [Google Scholar] [CrossRef] [PubMed]
- Jacamo, R.; Chen, Y.; Wang, Z.; Ma, W.; Zhang, M.; Spaeth, E.L.; Wang, Y.; Battula, V.L.; Mak, P.Y.; Schallmoser, K.; et al. Reciprocal Leukemia-Stroma VCAM-1/VLA-4-Dependent Activation of NF-KB Mediates Chemoresistance. Blood 2014, 123, 2691–2702. [Google Scholar] [CrossRef] [PubMed]
- García-García, A.; Klein, T.; Born, G.; Hilpert, M.; Scherberich, A.; Lengerke, C.; Skoda, R.C.; Bourgine, P.E.; Martin, I. Culturing Patient-Derived Malignant Hematopoietic Stem Cells in Engineered and Fully Humanized 3D Niches. Proc. Natl. Acad. Sci. USA 2021, 118, e2114227118. [Google Scholar] [CrossRef]
- Ito, S.; Barrett, A.J.; Dutra, A.; Pak, E.; Miner, S.; Keyvanfar, K.; Hensel, N.F.; Rezvani, K.; Muranski, P.; Liu, P.; et al. Long Term Maintenance of Myeloid Leukemic Stem Cells Cultured with Unrelated Human Mesenchymal Stromal Cells. Stem Cell Res. 2015, 14, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ema, H. Mechanisms of Self-Renewal in Hematopoietic Stem Cells. Int. J. Hematol. 2016, 103, 498–509. [Google Scholar] [CrossRef]
- Gothot, A.; van der Loo, J.C.M.; Clapp, D.W.; Srour, E.F. Cell Cycle-Related Changes in Repopulating Capacity of Human Mobilized Peripheral Blood CD34+ Cells in Non-Obese Diabetic/Severe Combined Immune-Deficient Mice. Blood 1998, 92, 2641–2649. [Google Scholar] [CrossRef] [PubMed]
- Florian, M.C.; Dörr, K.; Niebel, A.; Daria, D.; Schrezenmeier, H.; Rojewski, M.; Filippi, M.-D.; Hasenberg, A.; Gunzer, M.; Scharffetter-Kochanek, K.; et al. Cdc42 Activity Regulates Hematopoietic Stem Cell Aging and Rejuvenation. Cell Stem Cell 2012, 10, 520–530. [Google Scholar] [CrossRef]
- Ito, K.; Turcotte, R.; Cui, J.; Zimmerman, S.E.; Pinho, S.; Mizoguchi, T.; Arai, F.; Runnels, J.M.; Alt, C.; Teruya-Feldstein, J.; et al. Self-Renewal of a Purified Tie2+ Hematopoietic Stem Cell Population Relies on Mitochondrial Clearance. Science 2016, 354, 1156–1160. [Google Scholar] [CrossRef]
- Loeffler, D.; Wehling, A.; Schneiter, F.; Zhang, Y.; Müller-Bötticher, N.; Hoppe, P.S.; Hilsenbeck, O.; Kokkaliaris, K.D.; Endele, M.; Schroeder, T. Asymmetric Lysosome Inheritance Predicts Activation of Haematopoietic Stem Cells. Nature 2019, 573, 426–429. [Google Scholar] [CrossRef]
- Ting, S.B.; Deneault, E.; Hope, K.; Cellot, S.; Chagraoui, J.; Mayotte, N.; Dorn, J.F.; Laverdure, J.-P.; Harvey, M.; Hawkins, E.D.; et al. Asymmetric Segregation and Self-Renewal of Hematopoietic Stem and Progenitor Cells with Endocytic Ap2a2. Blood 2012, 119, 2510–2522. [Google Scholar] [CrossRef]
- Nebenfuehr, S.; Kollmann, K.; Sexl, V. The Role of CDK6 in Cancer. Int. J. Cancer 2020, 147, 2988–2995. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Rollins, B.J. Cyclin C/Cdk3 Promotes Rb-Dependent G0 Exit. Cell 2004, 117, 239–251. [Google Scholar] [CrossRef]
- Besson, A.; Dowdy, S.F.; Roberts, J.M. CDK Inhibitors: Cell Cycle Regulators and Beyond. Dev. Cell 2008, 14, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.; Yoshihara, H.; Hosokawa, K.; Tai, I.; Shinmyozu, K.; Tsukahara, F.; Maru, Y.; Nakayama, K.; Nakayama, K.I.; Suda, T. P57Kip2 and P27Kip1 Cooperate to Maintain Hematopoietic Stem Cell Quiescence through Interactions with Hsc70. Cell Stem Cell 2011, 9, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Dao, M.A.; Hwa, J.; Nolta, J.A. Molecular Mechanism of Transforming Growth Factor Beta-mediated Cell-cycle Modulation in Primary Human CD34(+) Progenitors. Blood 2002, 99, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Yang, P.; Shen, H.; Yu, H.; Song, X.; Zhang, L.; Zhang, P.; Cheng, H.; Xie, Z.; Hao, S.; et al. Small-Molecule Inhibitors Targeting INK4 Protein P18INK4C Enhance Ex Vivo Expansion of Haematopoietic Stem Cells. Nat. Commun. 2015, 6, 6328. [Google Scholar] [CrossRef]
- Yuan, Y.; Shen, H.; Franklin, D.S.; Scadden, D.T.; Cheng, T. In Vivo Self-Renewing Divisions of Haematopoietic Stem Cells Are Increased in the Absence of the Early G1-Phase Inhibitor, P18INK4C. Nat. Cell Biol. 2004, 6, 436–442. [Google Scholar] [CrossRef]
- Hilpert, M.; Legrand, C.; Bluteau, D.; Balayn, N.; Betems, A.; Bluteau, O.; Villeval, J.-L.; Louache, F.; Gonin, P.; Debili, N.; et al. P19INK4d Controls Hematopoietic Stem Cells in a Cell-Autonomous Manner during Genotoxic Stress and through the Microenvironment during Aging. Stem Cell Rep. 2014, 3, 1085–1102. [Google Scholar] [CrossRef]
- Cheung, T.H.; Rando, T.A. Molecular Regulation of Stem Cell Quiescence. Nat. Rev. Mol. Cell Biol. 2013, 14, 329–340. [Google Scholar] [CrossRef]
- Hao, S.; Chen, C.; Cheng, T. Cell Cycle Regulation of Hematopoietic Stem or Progenitor Cells. Int. J. Hematol. 2016, 103, 487–497. [Google Scholar] [CrossRef]
- Bowie, M.B.; Kent, D.G.; Copley, M.R.; Eaves, C.J. Steel Factor Responsiveness Regulates the High Self-Renewal Phenotype of Fetal Hematopoietic Stem Cells. Blood 2007, 109, 5043–5048. [Google Scholar] [CrossRef]
- Fox, N.; Priestley, G.; Papayannopoulou, T.; Kaushansky, K. Thrombopoietin Expands Hematopoietic Stem Cells after Transplantation. J. Clin. Investig. 2002, 110, 389–394. [Google Scholar] [CrossRef]
- Kunisato, A.; Chiba, S.; Nakagami-Yamaguchi, E.; Kumano, K.; Saito, T.; Masuda, S.; Yamaguchi, T.; Osawa, M.; Kageyama, R.; Nakauchi, H.; et al. HES-1 Preserves Purified Hematopoietic Stem Cells Ex Vivo and Accumulates Side Population Cells in Vivo. Blood 2003, 101, 1777–1783. [Google Scholar] [CrossRef]
- Stier, S.; Cheng, T.; Dombkowski, D.; Carlesso, N.; Scadden, D.T. Notch1 Activation Increases Hematopoietic Stem Cell Self-Renewal in Vivo and Favors Lymphoid over Myeloid Lineage Outcome. Blood 2002, 99, 2369–2378. [Google Scholar] [CrossRef]
- Arai, F.; Hirao, A.; Ohmura, M.; Sato, H.; Matsuoka, S.; Takubo, K.; Ito, K.; Koh, G.Y.; Suda, T. Tie2/Angiopoietin-1 Signaling Regulates Hematopoietic Stem Cell Quiescence in the Bone Marrow Niche. Cell 2004, 118, 149–161. [Google Scholar] [CrossRef]
- Nie, Y.; Han, Y.-C.; Zou, Y.-R. CXCR4 Is Required for the Quiescence of Primitive Hematopoietic Cells. J. Exp. Med. 2008, 205, 777–783. [Google Scholar] [CrossRef]
- Yamazaki, S.; Iwama, A.; Takayanagi, S.; Eto, K.; Ema, H.; Nakauchi, H. TGF-β as a Candidate Bone Marrow Niche Signal to Induce Hematopoietic Stem Cell Hibernation. Blood 2009, 113, 1250–1256. [Google Scholar] [CrossRef]
- Buza-Vidas, N.; Antonchuk, J.; Qian, H.; Månsson, R.; Luc, S.; Zandi, S.; Anderson, K.; Takaki, S.; Nygren, J.M.; Jensen, C.T.; et al. Cytokines Regulate Postnatal Hematopoietic Stem Cell Expansion: Opposing Roles of Thrombopoietin and LNK. Genes Dev. 2006, 20, 2018–2023. [Google Scholar] [CrossRef]
- Geddis, A.E.; Fox, N.E.; Kaushansky, K. Phosphatidylinositol 3-Kinase Is Necessary but Not Sufficient for Thrombopoietin-Induced Proliferation in Engineered Mpl-Bearing Cell Lines as Well as in Primary Megakaryocytic Progenitors. J. Biol. Chem. 2001, 276, 34473–34479. [Google Scholar] [CrossRef]
- Kimura, S.; Roberts, A.W.; Metcalf, D.; Alexander, W.S. Hematopoietic Stem Cell Deficiencies in Mice Lacking C-Mpl, the Receptor for Thrombopoietin. Proc. Natl. Acad. Sci. USA 1998, 95, 1195–1200. [Google Scholar] [CrossRef]
- Kollmann, S.; Grausenburger, R.; Klampfl, T.; Prchal-Murphy, M.; Bastl, K.; Pisa, H.; Knab, V.M.; Brandstoetter, T.; Doma, E.; Sperr, W.R.; et al. A STAT5B–CD9 Axis Determines Self-Renewal in Hematopoietic and Leukemic Stem Cells. Blood 2021, 138, 2347–2359. [Google Scholar] [CrossRef] [PubMed]
- Rojnuckarin, P.; Drachman, J.G.; Kaushansky, K. Thrombopoietin-Induced Activation of the Mitogen-Activated Protein Kinase (MAPK) Pathway in Normal Megakaryocytes: Role in Endomitosis. Blood 1999, 94, 1273–1282. [Google Scholar] [CrossRef] [PubMed]
- Seita, J.; Weissman, I.L. Hematopoietic Stem Cell: Self-Renewal versus Differentiation: Hematopoietic Stem Cell. Wiley Interdiscip. Rev. Syst. Biol. Med. 2010, 2, 640–653. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, H.; Arai, F.; Hosokawa, K.; Hagiwara, T.; Takubo, K.; Nakamura, Y.; Gomei, Y.; Iwasaki, H.; Matsuoka, S.; Miyamoto, K.; et al. Thrombopoietin/MPL Signaling Regulates Hematopoietic Stem Cell Quiescence and Interaction with the Osteoblastic Niche. Cell Stem Cell 2007, 1, 685–697. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.C.; Lodish, H.F. Cytokines Regulating Hematopoietic Stem Cell Function. Curr. Opin. Hematol. 2008, 15, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Antonchuk, J.; Sauvageau, G.; Humphries, R.K. HOXB4 Overexpression Mediates Very Rapid Stem Cell Regeneration and Competitive Hematopoietic Repopulation. Exp. Hematol. 2001, 29, 1125–1134. [Google Scholar] [CrossRef]
- Qian, H.; Buza-Vidas, N.; Hyland, C.D.; Jensen, C.T.; Antonchuk, J.; Månsson, R.; Thoren, L.A.; Ekblom, M.; Alexander, W.S.; Jacobsen, S.E.W. Critical Role of Thrombopoietin in Maintaining Adult Quiescent Hematopoietic Stem Cells. Cell Stem Cell 2007, 1, 671–684. [Google Scholar] [CrossRef]
- Maurer, B.; Kollmann, S.; Pickem, J.; Hoelbl-Kovacic, A.; Sexl, V. STAT5A and STAT5B—Twins with Different Personalities in Hematopoiesis and Leukemia. Cancers 2019, 11, 1726. [Google Scholar] [CrossRef]
- Wang, Z.; Li, G.; Tse, W.; Bunting, K.D. Conditional Deletion of STAT5 in Adult Mouse Hematopoietic Stem Cells Causes Loss of Quiescence and Permits Efficient Nonablative Stem Cell Replacement. Blood 2009, 113, 4856–4865. [Google Scholar] [CrossRef]
- Fatrai, S.; Wierenga, A.T.J.; Daenen, S.M.G.J.; Vellenga, E.; Schuringa, J.J. Identification of HIF2α as an Important STAT5 Target Gene in Human Hematopoietic Stem Cells. Blood 2011, 117, 3320–3330. [Google Scholar] [CrossRef]
- Lampreia, F.P.; Carmelo, J.G.; Anjos-Afonso, F. Notch Signaling in the Regulation of Hematopoietic Stem Cell. Curr. Stem Cell Rep. 2017, 3, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Giovannini, C.; Gramantieri, L.; Minguzzi, M.; Fornari, F.; Chieco, P.; Grazi, G.L.; Bolondi, L. CDKN1C/P57 Is Regulated by the Notch Target Gene Hes1 and Induces Senescence in Human Hepatocellular Carcinoma. Am. J. Pathol. 2012, 181, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Guiu, J.; Shimizu, R.; D’Altri, T.; Fraser, S.T.; Hatakeyama, J.; Bresnick, E.H.; Kageyama, R.; Dzierzak, E.; Yamamoto, M.; Espinosa, L.; et al. Hes Repressors Are Essential Regulators of Hematopoietic Stem Cell Development Downstream of Notch Signaling. J. Exp. Med. 2013, 210, 71–84. [Google Scholar] [CrossRef]
- Palomero, T.; Lim, W.K.; Odom, D.T.; Sulis, M.L.; Real, P.J.; Margolin, A.; Barnes, K.C.; O’Neil, J.; Neuberg, D.; Weng, A.P.; et al. NOTCH1 Directly Regulates C-MYC and Activates a Feed-Forward-Loop Transcriptional Network Promoting Leukemic Cell Growth. Proc. Natl. Acad. Sci. USA 2006, 103, 18261–18266. [Google Scholar] [CrossRef]
- de Pooter, R.F.; Schmitt, T.M.; de la Pompa, J.L.; Fujiwara, Y.; Orkin, S.H.; Zúñiga-Pflücker, J.C. Notch Signaling Requires GATA-2 to Inhibit Myelopoiesis from Embryonic Stem Cells and Primary Hemopoietic Progenitors. J. Immunol. 2006, 176, 5267–5275. [Google Scholar] [CrossRef]
- Perez-Roger, I. Cyclins D1 and D2 Mediate Myc-Induced Proliferation via Sequestration of P27Kip1 and P21Cip1. EMBO J. 1999, 18, 5310–5320. [Google Scholar] [CrossRef]
- Zhang, S.; Li, Y.; Wu, Y.; Shi, K.; Bing, L.; Hao, J. Wnt/β-Catenin Signaling Pathway Upregulates c-Myc Expression to Promote Cell Proliferation of P19 Teratocarcinoma Cells. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2012, 295, 2104–2113. [Google Scholar] [CrossRef]
- Fleming, H.E.; Janzen, V.; Lo Celso, C.; Guo, J.; Leahy, K.M.; Kronenberg, H.M.; Scadden, D.T. Wnt Signaling in the Niche Enforces Hematopoietic Stem Cell Quiescence and Is Necessary to Preserve Self-Renewal In Vivo. Cell Stem Cell 2008, 2, 274–283. [Google Scholar] [CrossRef]
- Luis, T.C.; Naber, B.A.E.; Roozen, P.P.C.; Brugman, M.H.; de Haas, E.F.E.; Ghazvini, M.; Fibbe, W.E.; van Dongen, J.J.M.; Fodde, R.; Staal, F.J.T. Canonical Wnt Signaling Regulates Hematopoiesis in a Dosage-Dependent Fashion. Cell Stem Cell 2011, 9, 345–356. [Google Scholar] [CrossRef]
- Zhang, Y.; Dépond, M.; He, L.; Foudi, A.; Kwarteng, E.O.; Lauret, E.; Plo, I.; Desterke, C.; Dessen, P.; Fujii, N.; et al. CXCR4/CXCL12 Axis Counteracts Hematopoietic Stem Cell Exhaustion through Selective Protection against Oxidative Stress. Sci. Rep. 2016, 6, 37827. [Google Scholar] [CrossRef]
- Blank, U.; Karlsson, S. TGF-b Signaling in the Control of Hematopoietic Stem Cells. Blood 2015, 125, 3542–3550. [Google Scholar] [CrossRef] [PubMed]
- Dao, M.A.; Taylor, N.; Nolta, J.A. Reduction in Levels of the Cyclin-Dependent Kinase Inhibitor P27kip-1 Coupled with Transforming Growth Factor Neutralization Induces Cell-Cycle Entry and Increases Retroviral Transduction of Primitive Human Hematopoietic Cells. Proc. Natl. Acad. Sci. USA 1998, 95, 13006–13011. [Google Scholar] [CrossRef] [PubMed]
- Ducos, K.; Panterne, B.; Fortunel, N.; Hatzfeld, A.; Monier, M.-N.; Hatzfeld, J. p21(cip1) mRNA is Controlled by Endogenous Transforming Growth Factor-Beta1 in Quiescent Human Hematopoietic Stem/Progenitor Cells. J. Cell. Physiol. 2000, 184, 80–85. [Google Scholar] [CrossRef]
- Vaidya, A.; Kale, V. Hematopoietic Stem Cells, Their Niche, and the Concept of Co-Culture Systems: A Critical Review. J. Stem Cells 2015, 19, 13–31. [Google Scholar]
- Wang, X.; Dong, F.; Zhang, S.; Yang, W.; Yu, W.; Wang, Z.; Zhang, S.; Wang, J.; Ma, S.; Wu, P.; et al. TGF-Β1 Negatively Regulates the Number and Function of Hematopoietic Stem Cells. Stem Cell Rep. 2018, 11, 274–287. [Google Scholar] [CrossRef]
- Asai, T.; Liu, Y.; Di Giandomenico, S.; Bae, N.; Ndiaye-Lobry, D.; Deblasio, A.; Menendez, S.; Antipin, Y.; Reva, B.; Wevrick, R.; et al. Necdin, a P53 Target Gene, Regulates the Quiescence and Response to Genotoxic Stress of Hematopoietic Stem/Progenitor Cells. Blood 2012, 120, 1601–1612. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Elf, S.E.; Miyata, Y.; Sashida, G.; Liu, Y.; Huang, G.; Di Giandomenico, S.; Lee, J.M.; Deblasio, A.; Menendez, S.; et al. P53 Regulates Hematopoietic Stem Cell Quiescence. Cell Stem Cell 2009, 4, 37–48. [Google Scholar] [CrossRef]
- Iavarone, A.; Garg, P.; Lasorella, A.; Hsu, J.; Israel, M.A. The Helix-Loop-Helix Rotein Id-2 Enhances Cell ProliferPion and Binds to the Retinobl-Asto a Protein. Genes Dev. 1994, 16, 1270–1284. [Google Scholar] [CrossRef]
- Mori, S. Lactation Defect in Mice Lacking the Helix-Loop-Helix Inhibitor Id2. EMBO J. 2000, 19, 5772–5781. [Google Scholar] [CrossRef]
- Taniura, H.; Matsumoto, K.; Yoshikawa, K. Physical and Functional Interactions of Neuronal Growth Suppressor Necdin with P53. J. Biol. Chem. 1999, 274, 16242–16248. [Google Scholar] [CrossRef]
- Lacorazza, H.D.; Yamada, T.; Liu, Y.; Miyata, Y.; Sivina, M.; Nunes, J.; Nimer, S.D. The Transcription Factor MEF/ELF4 Regulates the Quiescence of Primitive Hematopoietic Cells. Cancer Cell 2006, 9, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Ficara, F.; Murphy, M.J.; Lin, M.; Cleary, M.L. Pbx1 Regulates Self-Renewal of Long-Term Hematopoietic Stem Cells by Maintaining Their Quiescence. Cell Stem Cell 2008, 2, 484–496. [Google Scholar] [CrossRef] [PubMed]
- Goyama, S.; Nitta, E.; Yoshino, T.; Kako, S.; Watanabe-Okochi, N.; Shimabe, M.; Imai, Y.; Takahashi, K.; Kurokawa, M. EVI-1 Interacts with Histone Methyltransferases SUV39H1 and G9a for Transcriptional Repression and Bone Marrow Immortalization. Leukemia 2010, 24, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Laurenti, E.; Frelin, C.; Xie, S.; Ferrari, R.; Dunant, C.F.; Zandi, S.; Neumann, A.; Plumb, I.; Doulatov, S.; Chen, J.; et al. CDK6 Levels Regulate Quiescence Exit in Human Hematopoietic Stem Cells. Cell Stem Cell 2015, 16, 302–313. [Google Scholar] [CrossRef]
- Scheicher, R.; Hoelbl-Kovacic, A.; Bellutti, F.; Tigan, A.-S.; Prchal-Murphy, M.; Heller, G.; Schneckenleithner, C.; Salazar-Roa, M.; Zöchbauer-Müller, S.; Zuber, J.; et al. CDK6 as a Key Regulator of Hematopoietic and Leukemic Stem Cell Activation. Blood 2015, 125, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.; Laurenti, E.; Trumpp, A. Balancing Dormant and Self-Renewing Hematopoietic Stem Cells. Curr. Opin. Genet. Dev. 2009, 19, 461–468. [Google Scholar] [CrossRef]
- Takizawa, H.; Regoes, R.R.; Boddupalli, C.S.; Bonhoeffer, S.; Manz, M.G. Dynamic Variation in Cycling of Hematopoietic Stem Cells in Steady State and Inflammation. J. Exp. Med. 2011, 208, 273–284. [Google Scholar] [CrossRef]
- Trumpp, A.; Essers, M.; Wilson, A. Awakening Dormant Haematopoietic Stem Cells. Nat. Rev. Immunol. 2010, 10, 201–209. [Google Scholar] [CrossRef]
- Cortes, J. Natural History and Staging of Chronic Myelogenous Leukemia. Hematol. Oncol. Clin. N. Am. 2004, 18, 569–584. [Google Scholar] [CrossRef]
- Corces, M.R.; Buenrostro, J.D.; Wu, B.; Greenside, P.G.; Chan, S.M.; Koenig, J.L.; Snyder, M.P.; Pritchard, J.K.; Kundaje, A.; Greenleaf, W.J.; et al. Lineage-Specific and Single-Cell Chromatin Accessibility Charts Human Hematopoiesis and Leukemia Evolution. Nat. Genet. 2016, 48, 1193–1203. [Google Scholar] [CrossRef]
- Jan, M.; Snyder, T.M.; Corces-Zimmerman, M.R.; Vyas, P.; Weissman, I.L.; Quake, S.R.; Majeti, R. Clonal Evolution of Preleukemic Hematopoietic Stem Cells Precedes Human Acute Myeloid Leukemia. Sci. Transl. Med. 2012, 4, 149ra118. [Google Scholar] [CrossRef]
- Lu, C.; Ward, P.S.; Kapoor, G.S.; Rohle, D.; Turcan, S.; Abdel-Wahab, O.; Edwards, C.R.; Khanin, R.; Figueroa, M.E.; Melnick, A.; et al. IDH Mutation Impairs Histone Demethylation and Results in a Block to Cell Differentiation. Nature 2012, 483, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Gilliland, D.G.; Griffin, J.D. The Roles of FLT3 in Hematopoiesis and Leukemia. Blood 2002, 100, 1532–1542. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Small, D. Mutant FLT3 Signaling Contributes to a Block in Myeloid Differentiation. Leuk. Lymphoma 2005, 46, 1679–1687. [Google Scholar] [CrossRef] [PubMed]
- Papaemmanuil, E.; Gerstung, M.; Bullinger, L.; Gaidzik, V.I.; Paschka, P.; Roberts, N.D.; Potter, N.E.; Heuser, M.; Thol, F.; Bolli, N.; et al. Genomic Classification and Prognosis in Acute Myeloid Leukemia. N. Engl. J. Med. 2016, 374, 2209–2221. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.N.; Kalleda, N.; Stavropoulou, V.; Schwaller, J. The Impact of the Cellular Origin in Acute Myeloid Leukemia: Learning from Mouse Models. HemaSphere 2019, 3, e152. [Google Scholar] [CrossRef]
- Ng, S.W.K.; Mitchell, A.; Kennedy, J.A.; Chen, W.C.; McLeod, J.; Ibrahimova, N.; Arruda, A.; Popescu, A.; Gupta, V.; Schimmer, A.D.; et al. A 17-Gene Stemness Score for Rapid Determination of Risk in Acute Leukaemia. Nature 2016, 540, 433–437. [Google Scholar] [CrossRef]
- Duployez, N.; Marceau-Renaut, A.; Villenet, C.; Petit, A.; Rousseau, A.; Ng, S.W.K.; Paquet, A.; Gonzales, F.; Barthélémy, A.; Leprêtre, F.; et al. The Stem Cell-Associated Gene Expression Signature Allows Risk Stratification in Pediatric Acute Myeloid Leukemia. Leukemia 2019, 33, 348–357. [Google Scholar] [CrossRef]
- Li, S.; Garrett-Bakelman, F.E.; Chung, S.S.; Sanders, M.A.; Hricik, T.; Rapaport, F.; Patel, J.; Dillon, R.; Vijay, P.; Brown, A.L.; et al. Distinct Evolution and Dynamics of Epigenetic and Genetic Heterogeneity in Acute Myeloid Leukemia. Nat. Med. 2016, 22, 792–799. [Google Scholar] [CrossRef]
- Simsek, T.; Kocabas, F.; Zheng, J.; DeBerardinis, R.J.; Mahmoud, A.I.; Olson, E.N.; Schneider, J.W.; Zhang, C.C.; Sadek, H.A. The Distinct Metabolic Profile of Hematopoietic Stem Cells Reflects Their Location in a Hypoxic Niche. Cell Stem Cell 2010, 7, 380–390. [Google Scholar] [CrossRef]
- Kuntz, E.M.; Baquero, P.; Michie, A.M.; Dunn, K.; Tardito, S.; Holyoake, T.L.; Helgason, G.V.; Gottlieb, E. Targeting Mitochondrial Oxidative Phosphorylation Eradicates Therapy-Resistant Chronic Myeloid Leukemia Stem Cells. Nat. Med. 2017, 23, 1234–1240. [Google Scholar] [CrossRef] [PubMed]
- Nieborowska-Skorska, M.; Kopinski, P.K.; Ray, R.; Hoser, G.; Ngaba, D.; Flis, S.; Cramer, K.; Reddy, M.M.; Koptyra, M.; Penserga, T.; et al. Rac2-MRC-CIII–Generated ROS Cause Genomic Instability in Chronic Myeloid Leukemia Stem Cells and Primitive Progenitors. Blood 2012, 119, 4253–4263. [Google Scholar] [CrossRef] [PubMed]
- Sallmyr, A.; Fan, J.; Datta, K.; Kim, K.-T.; Grosu, D.; Shapiro, P.; Small, D.; Rassool, F. Internal Tandem Duplication of FLT3 (FLT3/ITD) Induces Increased ROS Production, DNA Damage, and Misrepair: Implications for Poor Prognosis in AML. Blood 2008, 111, 3173–3182. [Google Scholar] [CrossRef] [PubMed]
- Adane, B.; Ye, H.; Khan, N.; Pei, S.; Minhajuddin, M.; Stevens, B.M.; Jones, C.L.; D’Alessandro, A.; Reisz, J.A.; Zaberezhnyy, V.; et al. The Hematopoietic Oxidase NOX2 Regulates Self-Renewal of Leukemic Stem Cells. Cell Rep. 2019, 27, 238–254.e6. [Google Scholar] [CrossRef]
- Grossmann, V.; Kohlmann, A.; Zenger, M.; Schindela, S.; Eder, C.; Weissmann, S.; Schnittger, S.; Kern, W.; Müller, M.C.; Hochhaus, A.; et al. A Deep-Sequencing Study of Chronic Myeloid Leukemia Patients in Blast Crisis (BC-CML) Detects Mutations in 76.9% of Cases. Leukemia 2011, 25, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Tubbs, A.; Nussenzweig, A. Endogenous DNA Damage as a Source of Genomic Instability in Cancer. Cell 2017, 168, 644–656. [Google Scholar] [CrossRef]
- Rebechi, M.T.; Pratz, K.W. Genomic Instability Is a Principle Pathologic Feature of FLT3 ITD Kinase Activity in Acute Myeloid Leukemia Leading to Clonal Evolution and Disease Progression. Leuk. Lymphoma 2017, 58, 2040–2050. [Google Scholar] [CrossRef]
- Lagadinou, E.D.; Sach, A.; Callahan, K.; Rossi, R.M.; Neering, S.J.; Minhajuddin, M.; Ashton, J.M.; Pei, S.; Grose, V.; O’Dwyer, K.M.; et al. BCL-2 Inhibition Targets Oxidative Phosphorylation and Selectively Eradicates Quiescent Human Leukemia Stem Cells. Cell Stem Cell 2013, 12, 329–341. [Google Scholar] [CrossRef]
- Hao, X.; Gu, H.; Chen, C.; Huang, D.; Zhao, Y.; Xie, L.; Zou, Y.; Shu, H.S.; Zhang, Y.; He, X.; et al. Metabolic Imaging Reveals a Unique Preference of Symmetric Cell Division and Homing of Leukemia-Initiating Cells in an Endosteal Niche. Cell Metab. 2019, 29, 950–965.e6. [Google Scholar] [CrossRef]
- Pei, S.; Minhajuddin, M.; Adane, B.; Khan, N.; Stevens, B.M.; Mack, S.C.; Lai, S.; Rich, J.N.; Inguva, A.; Shannon, K.M.; et al. AMPK/FIS1-Mediated Mitophagy Is Required for Self-Renewal of Human AML Stem Cells. Cell Stem Cell 2018, 23, 86–100.e6. [Google Scholar] [CrossRef]
- Hattori, A.; Tsunoda, M.; Konuma, T.; Kobayashi, M.; Nagy, T.; Glushka, J.; Tayyari, F.; McSkimming, D.; Kannan, N.; Tojo, A.; et al. Cancer Progression by Reprogrammed BCAA Metabolism in Myeloid Leukaemia. Nature 2017, 545, 500–504. [Google Scholar] [CrossRef]
- Bazinet, A.; Popradi, G. A General Practitioner’s Guide to Hematopoietic Stem-Cell Transplantation. Curr. Oncol. 2019, 26, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Jenq, R.R.; van den Brink, M.R.M. Allogeneic Haematopoietic Stem Cell Transplantation: Individualized Stem Cell and Immune Therapy of Cancer. Nat. Rev. Cancer 2010, 10, 213–221. [Google Scholar] [CrossRef] [PubMed]
- van Galen, P.; Kreso, A.; Mbong, N.; Kent, D.G.; Fitzmaurice, T.; Chambers, J.E.; Xie, S.; Laurenti, E.; Hermans, K.; Eppert, K.; et al. The Unfolded Protein Response Governs Integrity of the Haematopoietic Stem-Cell Pool during Stress. Nature 2014, 510, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Karigane, D.; Kobayashi, H.; Morikawa, T.; Ootomo, Y.; Sakai, M.; Nagamatsu, G.; Kubota, Y.; Goda, N.; Matsumoto, M.; Nishimura, E.K.; et al. P38α Activates Purine Metabolism to Initiate Hematopoietic Stem/Progenitor Cell Cycling in Response to Stress. Cell Stem Cell 2016, 19, 192–204. [Google Scholar] [CrossRef]
- Koide, S.; Sigurdsson, V.; Radulovic, V.; Saito, K.; Zheng, Z.; Lang, S.; Soneji, S.; Iwama, A.; Miharada, K. CD244 Expression Represents Functional Decline of Murine Hematopoietic Stem Cells after in Vitro Culture. iScience 2022, 25, 103603. [Google Scholar] [CrossRef]
- Kruta, M.; Sunshine, M.J.; Chua, B.A.; Fu, Y.; Chawla, A.; Dillingham, C.H.; Hidalgo San Jose, L.; De Jong, B.; Zhou, F.J.; Signer, R.A.J. Hsf1 Promotes Hematopoietic Stem Cell Fitness and Proteostasis in Response to Ex Vivo Culture Stress and Aging. Cell Stem Cell 2021, 28, 1950–1965.e6. [Google Scholar] [CrossRef]
- Ludin, A.; Itkin, T.; Gur-Cohen, S.; Mildner, A.; Shezen, E.; Golan, K.; Kollet, O.; Kalinkovich, A.; Porat, Z.; D’Uva, G.; et al. Monocytes-Macrophages That Express α-Smooth Muscle Actin Preserve Primitive Hematopoietic Cells in the Bone Marrow. Nat. Immunol. 2012, 13, 1072–1082. [Google Scholar] [CrossRef]
- Miharada, K.; Sigurdsson, V.; Karlsson, S. Dppa5 Improves Hematopoietic Stem Cell Activity by Reducing Endoplasmic Reticulum Stress. Cell Rep. 2014, 7, 1381–1392. [Google Scholar] [CrossRef]
- Walter, D.; Lier, A.; Geiselhart, A.; Thalheimer, F.B.; Huntscha, S.; Sobotta, M.C.; Moehrle, B.; Brocks, D.; Bayindir, I.; Kaschutnig, P.; et al. Exit from Dormancy Provokes DNA-Damage-Induced Attrition in Haematopoietic Stem Cells. Nature 2015, 520, 549–552. [Google Scholar] [CrossRef]
- Xiao, N.; Jani, K.; Morgan, K.; Okabe, R.; Cullen, D.E.; Jesneck, J.L.; Raffel, G.D. Hematopoietic Stem Cells Lacking Ott1 Display Aspects Associated with Aging and Are Unable to Maintain Quiescence during Proliferative Stress. Blood 2012, 119, 4898–4907. [Google Scholar] [CrossRef] [PubMed]
- Christodoulou, C.; Spencer, J.A.; Yeh, S.-C.A.; Turcotte, R.; Kokkaliaris, K.D.; Panero, R.; Ramos, A.; Guo, G.; Seyedhassantehrani, N.; Esipova, T.V.; et al. Live-Animal Imaging of Native Haematopoietic Stem and Progenitor Cells. Nature 2020, 578, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Kubota, Y.; Takubo, K.; Suda, T. Bone Marrow Long Label-Retaining Cells Reside in the Sinusoidal Hypoxic Niche. Biochem. Biophys. Res. Commun. 2008, 366, 335–339. [Google Scholar] [CrossRef]
- Parmar, K.; Mauch, P.; Vergilio, J.-A.; Sackstein, R.; Down, J.D. Distribution of Hematopoietic Stem Cells in the Bone Marrow According to Regional Hypoxia. Proc. Natl. Acad. Sci. USA 2007, 104, 5431–5436. [Google Scholar] [CrossRef]
- Winkler, I.G.; Barbier, V.; Wadley, R.; Zannettino, A.C.W.; Williams, S.; Lévesque, J.-P. Positioning of Bone Marrow Hematopoietic and Stromal Cells Relative to Blood Flow in Vivo: Serially Reconstituting Hematopoietic Stem Cells Reside in Distinct Nonperfused Niches. Blood 2010, 116, 375–385. [Google Scholar] [CrossRef]
- Broxmeyer, H.E.; O’Leary, H.A.; Huang, X.; Mantel, C. The Importance of Hypoxia and Extra Physiologic Oxygen Shock/Stress for Collection and Processing of Stem and Progenitor Cells to Understand True Physiology/Pathology of These Cells Ex Vivo. Curr. Opin. Hematol. 2015, 22, 273–278. [Google Scholar] [CrossRef]
- Cipolleschi, M.; Dello Sbarba, P.; Olivotto, M. The Role of Hypoxia in the Maintenance of Hematopoietic Stem Cells. Blood 1993, 82, 2031–2037. [Google Scholar] [CrossRef] [PubMed]
- Danet, G.H.; Pan, Y.; Luongo, J.L.; Bonnet, D.A.; Simon, M.C. Expansion of Human SCID-Repopulating Cells under Hypoxic Conditions. J. Clin. Investig. 2003, 112, 126–135. [Google Scholar] [CrossRef]
- Ivanović, Z.; Sbarba, P.D.; Trimoreau, F.; Faucher, J.-L.; Praloran, V. Primitive Human HPCs Are Better Maintained and Expanded in Vitro at 1 Percent Oxygen than at 20 Percent. Transfusion 2000, 40, 1482–1488. [Google Scholar] [CrossRef]
- Kobayashi, H.; Morikawa, T.; Okinaga, A.; Hamano, F.; Hashidate-Yoshida, T.; Watanuki, S.; Hishikawa, D.; Shindou, H.; Arai, F.; Kabe, Y.; et al. Environmental Optimization Enables Maintenance of Quiescent Hematopoietic Stem Cells Ex Vivo. Cell Rep. 2019, 28, 145–158.e9. [Google Scholar] [CrossRef]
- Pernes, G.; Flynn, M.C.; Lancaster, G.I.; Murphy, A.J. Fat for Fuel: Lipid Metabolism in Haematopoiesis. Clin. Transl. Immunol. 2019, 8, e1098. [Google Scholar] [CrossRef]
- Purton, L.E. Roles of Retinoids and Retinoic Acid Receptors in the Regulation of Hematopoietic Stem Cell Self-Renewal and Differentiation. PPAR Res. 2007, 2007, 087934. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.-H.; Gudas, L.J. Retinoids, Retinoic Acid Receptors, and Cancer. Annu. Rev. Pathol. Mech. Dis. 2011, 6, 345–364. [Google Scholar] [CrossRef]
- Cabezas-Wallscheid, N.; Buettner, F.; Sommerkamp, P.; Klimmeck, D.; Ladel, L.; Thalheimer, F.B.; Pastor-Flores, D.; Roma, L.P.; Renders, S.; Zeisberger, P.; et al. Vitamin A-Retinoic Acid Signaling Regulates Hematopoietic Stem Cell Dormancy. Cell 2017, 169, 807–823.e19. [Google Scholar] [CrossRef] [PubMed]
- Oedekoven, C.A.; Belmonte, M.; Bode, D.; Hamey, F.K.; Shepherd, M.S.; Che, J.L.C.; Boyd, G.; McDonald, C.; Belluschi, S.; Diamanti, E.; et al. Hematopoietic Stem Cells Retain Functional Potential and Molecular Identity in Hibernation Cultures. Stem Cell Rep. 2021, 16, 1614–1628. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, A.C.; Ishida, R.; Kikuchi, M.; Sudo, K.; Morita, M.; Crisostomo, R.V.; Yamamoto, R.; Loh, K.M.; Nakamura, Y.; Watanabe, M.; et al. Long-Term Ex Vivo Haematopoietic-Stem-Cell Expansion Allows Nonconditioned Transplantation. Nature 2019, 571, 117–121. [Google Scholar] [CrossRef]
- Wilkinson, A.C.; Ishida, R.; Nakauchi, H.; Yamazaki, S. Long-Term Ex Vivo Expansion of Mouse Hematopoietic Stem Cells. Nat. Protoc. 2020, 15, 628–648. [Google Scholar] [CrossRef] [PubMed]
- Yonemura, Y.; Ku, H.; Lyman, S.D.; Ogawa, M. In Vitro Expansion of Hematopoietic Progenitors and Maintenance of Stem Cells: Comparison Between FLT3/FLK-2 Ligand and KIT Ligand. Blood 1997, 89, 1915–1921. [Google Scholar] [CrossRef]
- Zhang, S.; Morita, M.; Wang, Z.; Ooehara, J.; Zhang, S.; Xie, M.; Bai, H.; Yu, W.; Wang, X.; Dong, F.; et al. Interleukin-12 Supports in Vitro Self-Renewal of Long-Term Hematopoietic Stem Cells. Blood Sci. 2019, 1, 92–101. [Google Scholar] [CrossRef]
- Huynh, H.; Iizuka, S.; Kaba, M.; Kirak, O.; Zheng, J.; Lodish, H.F.; Zhang, C.C. Insulin-Like Growth Factor-Binding Protein 2 Secreted by a Tumorigenic Cell Line Supports Ex Vivo Expansion of Mouse Hematopoietic Stem Cells. Stem Cells 2008, 26, 1628–1635. [Google Scholar] [CrossRef]
- Ieyasu, A.; Ishida, R.; Kimura, T.; Morita, M.; Wilkinson, A.C.; Sudo, K.; Nishimura, T.; Ohehara, J.; Tajima, Y.; Lai, C.-Y.; et al. An All-Recombinant Protein-Based Culture System Specifically Identifies Hematopoietic Stem Cell Maintenance Factors. Stem Cell Rep. 2017, 8, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.L.; Eaves, C.J. Expansion in Vitro of Adult Murine Hematopoietic Stem Cells with Transplantable Lympho-Myeloid Reconstituting Ability. Proc. Natl. Acad. Sci. USA 1997, 94, 13648–13653. [Google Scholar] [CrossRef] [PubMed]
- Yonemura, Y.; Ku, H.; Hirayama, F.; Souza, L.M.; Ogawa, M. Interleukin 3 or Interleukin 1 Abrogates the Reconstituting Ability of Hematopoietic Stem Cells. Proc. Natl. Acad. Sci. USA 1996, 93, 4040–4044. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, M.; Yonemura, Y.; Ku, H. In Vitro Expansion of Hematopoietic Stem Cells. Stem Cells 1997, 15, 7–12. [Google Scholar] [CrossRef]
- Yagi, M.; Ritchie, K.A.; Sitnicka, E.; Storey, C.; Roth, G.J.; Bartelmez, S. Sustained Ex Vivo Expansion of Hematopoietic Stem Cells Mediated by Thrombopoietin. Proc. Natl. Acad. Sci. USA 1999, 96, 8126–8131. [Google Scholar] [CrossRef]
- Du, J.; He, H.; Li, Z.; He, J.; Bai, Z.; Liu, B.; Lan, Y. Integrative Transcriptomic Analysis of Developing Hematopoietic Stem Cells in Human and Mouse at Single-Cell Resolution. Biochem. Biophys. Res. Commun. 2021, 558, 161–167. [Google Scholar] [CrossRef]
- Eaves, C.J. Hematopoietic Stem Cells: Concepts, Definitions, and the New Reality. Blood 2015, 125, 2605–2613. [Google Scholar] [CrossRef]
- Göttgens, B. Regulatory Network Control of Blood Stem Cells. Blood 2015, 125, 2614–2620. [Google Scholar] [CrossRef]
- Radtke, F.; Wilson, A.; Stark, G.; Bauer, M.; van Meerwijk, J.; MacDonald, H.R.; Aguet, M. Deficient T Cell Fate Specification in Mice with an Induced Inactivation of Notch1. Immunity 1999, 10, 547–558. [Google Scholar] [CrossRef]
- Dallas, M.H.; Varnum-Finney, B.; Delaney, C.; Kato, K.; Bernstein, I.D. Density of the Notch Ligand Delta1 Determines Generation of B and T Cell Precursors from Hematopoietic Stem Cells. J. Exp. Med. 2005, 201, 1361–1366. [Google Scholar] [CrossRef]
- Varnum-Finney, B.; Brashem-Stein, C.; Bernstein, I.D. Combined Effects of Notch Signaling and Cytokines Induce a Multiple Log Increase in Precursors with Lymphoid and Myeloid Reconstituting Ability. Blood 2003, 101, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Dallas, M.H.; Varnum-Finney, B.; Martin, P.J.; Bernstein, I.D. Enhanced T-Cell Reconstitution by Hematopoietic Progenitors Expanded Ex Vivo Using the Notch Ligand Delta. Blood 2007, 109, 3579–3587. [Google Scholar] [CrossRef] [PubMed]
- Tolosano, E.; Altruda, F. Hemopexin: Structure, Function, and Regulation. DNA Cell Biol. 2002, 21, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Farahbakhshian, E.; Verstegen, M.M.; Visser, T.P.; Kheradmandkia, S.; Geerts, D.; Arshad, S.; Riaz, N.; Grosveld, F.; van Til, N.P.; Meijerink, J.P.P. Angiopoietin-Like Protein 3 Promotes Preservation of Stemness during Ex Vivo Expansion of Murine Hematopoietic Stem Cells. PLoS ONE 2014, 9, e105642. [Google Scholar] [CrossRef]
- Stein, E.M.; DiNardo, C.D.; Pollyea, D.A.; Fathi, A.T.; Roboz, G.J.; Altman, J.K.; Stone, R.M.; DeAngelo, D.J.; Levine, R.L.; Flinn, I.W.; et al. Enasidenib in Mutant IDH2 Relapsed or Refractory Acute Myeloid Leukemia. Blood 2017, 130, 722–731. [Google Scholar] [CrossRef]
- Kragh-Hansen, U. Molecular Aspects of Ligand Binding to Serum Albumin. Pharmacol. Rev. 1981, 33, 17–53. [Google Scholar]
- Nishimura, T.; Hsu, I.; Martinez-Krams, D.C.; Nakauchi, Y.; Majeti, R.; Yamazaki, S.; Nakauchi, H.; Wilkinson, A.C. Use of Polyvinyl Alcohol for Chimeric Antigen Receptor T-Cell Expansion. Exp. Hematol. 2019, 80, 16–20. [Google Scholar] [CrossRef]
- Zhang, C.C.; Lodish, H.F. Insulin-like Growth Factor 2 Expressed in a Novel Fetal Liver Cell Population Is a Growth Factor for Hematopoietic Stem Cells. Blood 2004, 103, 2513–2521. [Google Scholar] [CrossRef]
- Fan, X.; Gay, F.P.; Lim, F.W.; Ang, J.M.; Chu, P.P.; Bari, S.; Hwang, W.Y. Low-Dose Insulin-like Growth Factor Binding Proteins 1 and 2 and Angiopoietin-like Protein 3 Coordinately Stimulate Ex Vivo Expansion of Human Umbilical Cord Blood Hematopoietic Stem Cells as Assayed in NOD/SCID Gamma Null Mice. Stem Cell Res. Ther. 2014, 5, 71. [Google Scholar] [CrossRef]
- Ribeiro-Filho, A.C.; Levy, D.; Ruiz, J.L.M.; da Cunha Mantovani, M.; Bydlowski, S.P. Traditional and Advanced Cell Cultures in Hematopoietic Stem Cell Studies. Cells 2019, 8, 1628. [Google Scholar] [CrossRef]
- Zhang, C.C.; Kaba, M.; Iizuka, S.; Huynh, H.; Lodish, H.F. Angiopoietin-like 5 and IGFBP2 Stimulate Ex Vivo Expansion of Human Cord Blood Hematopoietic Stem Cells as Assayed by NOD/SCID Transplantation. Blood 2008, 111, 3415–3423. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri-Fard, S.; Niazi, V.; Taheri, M.; Basiri, A. Effect of Small Molecule on Ex Vivo Expansion of Cord Blood Hematopoietic Stem Cells: A Concise Review. Front. Cell Dev. Biol. 2021, 9, 649115. [Google Scholar] [CrossRef] [PubMed]
- Boitano, A.E.; Wang, J.; Romeo, R.; Bouchez, L.C.; Parker, A.E.; Sutton, S.E.; Walker, J.R.; Flaveny, C.A.; Perdew, G.H.; Denison, M.S.; et al. Aryl Hydrocarbon Receptor Antagonists Promote the Expansion of Human Hematopoietic Stem Cells. Science 2010, 329, 1345–1348. [Google Scholar] [CrossRef] [PubMed]
- Shpall, E.J.; Rezvani, K. Cord Blood Expansion Has Arrived. Blood 2021, 138, 1381–1382. [Google Scholar] [CrossRef]
- Thordardottir, S.; Hangalapura, B.N.; Hutten, T.; Cossu, M.; Spanholtz, J.; Schaap, N.; Radstake, T.R.D.J.; van der Voort, R.; Dolstra, H. The Aryl Hydrocarbon Receptor Antagonist StemRegenin 1 Promotes Human Plasmacytoid and Myeloid Dendritic Cell Development from CD34+ Hematopoietic Progenitor Cells. Stem Cells Dev. 2014, 23, 955–967. [Google Scholar] [CrossRef]
- Singh, K.P.; Casado, F.L.; Opanashuk, L.A.; Gasiewicz, T.A. The Aryl Hydrocarbon Receptor Has a Normal Function in the Regulation of Hematopoietic and Other Stem/Progenitor Cell Populations. Biochem. Pharmacol. 2009, 77, 577–587. [Google Scholar] [CrossRef]
- Liu, H.; Ramachandran, I.; Gabrilovich, D.I. Regulation of Plasmacytoid Dendritic Cell Development in Mice by Aryl Hydrocarbon Receptor. Immunol. Cell Biol. 2014, 92, 200–203. [Google Scholar] [CrossRef]
- Wagner, J.E.; Brunstein, C.G.; Boitano, A.E.; DeFor, T.E.; McKenna, D.; Sumstad, D.; Blazar, B.R.; Tolar, J.; Le, C.; Jones, J.; et al. Phase I/II Trial of StemRegenin-1 Expanded Umbilical Cord Blood Hematopoietic Stem Cells Supports Testing as a Stand-Alone Graft. Cell Stem Cell 2016, 18, 144–155. [Google Scholar] [CrossRef]
- Casper, R.F.; Quesne, M.; Rogers, I.M.; Shirota, T.; Jolivet, A.; Milgrom, E.; Savouret, J.-F. Resveratrol Has Antagonist Activity on the Aryl Hydrocarbon Receptor: Implications for Prevention of Dioxin Toxicity. Mol. Pharmacol. 1999, 56, 784–790. [Google Scholar]
- Lin, H.-Y.; Lansing, L.; Merillon, J.-M.; Davis, F.B.; Tang, H.-Y.; Shih, A.; Vitrac, X.; Krisa, S.; Keating, T.; Cao, H.J.; et al. Integrin AVβ3 Contains a Receptor Site for Resveratrol. FASEB J. 2006, 20, 1742–1744. [Google Scholar] [CrossRef]
- Chagraoui, J.; Girard, S.; Spinella, J.-F.; Simon, L.; Bonneil, E.; Mayotte, N.; MacRae, T.; Coulombe-Huntington, J.; Bertomeu, T.; Moison, C.; et al. UM171 Preserves Epigenetic Marks That Are Reduced in Ex Vivo Culture of Human HSCs via Potentiation of the CLR3-KBTBD4 Complex. Cell Stem Cell 2021, 28, 48–62.e6. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Roy, J.; Lachance, S.; Delisle, J.-S.; Marinier, A.; Busque, L.; Roy, D.-C.; Barabé, F.; Ahmad, I.; Bambace, N.; et al. Hematopoietic Stem Cell Transplantation Using Single UM171-Expanded Cord Blood: A Single-Arm, Phase 1–2 Safety and Feasibility Study. Lancet Haematol. 2020, 7, e134–e145. [Google Scholar] [CrossRef]
- Fares, I.; Chagraoui, J.; Gareau, Y.; Gingras, S.; Ruel, R.; Mayotte, N.; Csaszar, E.; Knapp, D.J.H.F.; Miller, P.; Ngom, M.; et al. Pyrimidoindole Derivatives Are Agonists of Human Hematopoietic Stem Cell Self-Renewal. Science 2014, 345, 1509–1512. [Google Scholar] [CrossRef] [PubMed]
- Peled, T.; Shoham, H.; Aschengrau, D.; Yackoubov, D.; Frei, G.; Rosenheimer, G.N.; Lerrer, B.; Cohen, H.Y.; Nagler, A.; Fibach, E.; et al. Nicotinamide, a SIRT1 Inhibitor, Inhibits Differentiation and Facilitates Expansion of Hematopoietic Progenitor Cells with Enhanced Bone Marrow Homing and Engraftment. Exp. Hematol. 2012, 40, 342–355.e1. [Google Scholar] [CrossRef]
- Peck, B.; Chen, C.-Y.; Ho, K.-K.; Di Fruscia, P.; Myatt, S.S.; Coombes, R.C.; Fuchter, M.J.; Hsiao, C.-D.; Lam, E.W.-F. SIRT Inhibitors Induce Cell Death and P53 Acetylation through Targeting Both SIRT1 and SIRT2. Mol. Cancer Ther. 2010, 9, 844–855. [Google Scholar] [CrossRef]
- Horwitz, M.E.; Chao, N.J.; Rizzieri, D.A.; Long, G.D.; Sullivan, K.M.; Gasparetto, C.; Chute, J.P.; Morris, A.; McDonald, C.; Waters-Pick, B.; et al. Umbilical Cord Blood Expansion with Nicotinamide Provides Long-Term Multilineage Engraftment. J. Clin. Investig. 2014, 124, 3121–3128. [Google Scholar] [CrossRef]
- Papa, L.; Zimran, E.; Djedaini, M.; Ge, Y.; Ozbek, U.; Sebra, R.; Sealfon, S.C.; Hoffman, R. Ex Vivo Human HSC Expansion Requires Coordination of Cellular Reprogramming with Mitochondrial Remodeling and P53 Activation. Blood Adv. 2018, 2, 2766–2779. [Google Scholar] [CrossRef]
- Zimran, E.; Papa, L.; Djedaini, M.; Patel, A.; Iancu-Rubin, C.; Hoffman, R. Expansion and Preservation of the Functional Activity of Adult Hematopoietic Stem Cells Cultured Ex Vivo with a Histone Deacetylase Inhibitor. Stem Cells Transl. Med. 2020, 9, 531–542. [Google Scholar] [CrossRef]
- Rocha, V.; Gluckman, E. Clinical Use of Umbilical Cord Blood Hematopoietic Stem Cells. Biol. Blood Marrow Transplant. 2006, 12, 34–41. [Google Scholar] [CrossRef]
- Delaney, C.; Heimfeld, S.; Brashem-Stein, C.; Voorhies, H.; Manger, R.L.; Bernstein, I.D. Notch-Mediated Expansion of Human Cord Blood Progenitor Cells Capable of Rapid Myeloid Reconstitution. Nat. Med. 2010, 16, 232–236. [Google Scholar] [CrossRef]
- Papa, L.; Djedaini, M.; Hoffman, R. Ex Vivo HSC Expansion Challenges the Paradigm of Unidirectional Human Hematopoiesis. Ann. N. Y. Acad. Sci. 2020, 1466, 39–50. [Google Scholar] [CrossRef] [PubMed]
- MacMillan, M.L.; Weisdorf, D.J.; Brunstein, C.G.; Cao, Q.; DeFor, T.E.; Verneris, M.R.; Blazar, B.R.; Wagner, J.E. Acute Graft-versus-Host Disease after Unrelated Donor Umbilical Cord Blood Transplantation: Analysis of Risk Factors. Blood 2009, 113, 2410–2415. [Google Scholar] [CrossRef] [PubMed]
- Kasai, F.; Hirayama, N.; Ozawa, M.; Iemura, M.; Kohara, A. Changes of Heterogeneous Cell Populations in the Ishikawa Cell Line during Long-Term Culture: Proposal for an in Vitro Clonal Evolution Model of Tumor Cells. Genomics 2016, 107, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Dexter, T.M.; Moore, M.A.; Sheridan, A.P. Maintenance of Hemopoietic Stem Cells and Production of Differentiated Progeny in Allogeneic and Semiallogeneic Bone Marrow Chimeras in Vitro. J. Exp. Med. 1977, 145, 1612–1616. [Google Scholar] [CrossRef]
- Klco, J.M.; Spencer, D.H.; Miller, C.A.; Griffith, M.; Lamprecht, T.L.; O’Laughlin, M.; Fronick, C.; Magrini, V.; Demeter, R.T.; Fulton, R.S.; et al. Functional Heterogeneity of Genetically Defined Subclones in Acute Myeloid Leukemia. Cancer Cell 2014, 25, 379–392. [Google Scholar] [CrossRef]
- Miles, L.A.; Bowman, R.L.; Merlinsky, T.R.; Csete, I.S.; Ooi, A.T.; Durruthy-Durruthy, R.; Bowman, M.; Famulare, C.; Patel, M.A.; Mendez, P.; et al. Single-Cell Mutation Analysis of Clonal Evolution in Myeloid Malignancies. Nature 2020, 587, 477–482. [Google Scholar] [CrossRef]
- Zeijlemaker, W.; Gratama, J.W.; Schuurhuis, G.J. Tumor Heterogeneity Makes AML a “Moving Target” for Detection of Residual Disease: Phenotype Instability and MRD in AML. Cytom. B Clin. Cytom. 2014, 86, 3–14. [Google Scholar] [CrossRef]
- Brenner, A.K.; Aasebø, E.; Hernandez-Valladares, M.; Selheim, F.; Berven, F.; Grønningsæter, I.-S.; Bartaula-Brevik, S.; Bruserud, Ø. The Capacity of Long-Term in Vitro Proliferation of Acute Myeloid Leukemia Cells Supported Only by Exogenous Cytokines Is Associated with a Patient Subset with Adverse Outcome. Cancers 2019, 11, 73. [Google Scholar] [CrossRef]
- Lin, H.; Damen, J.E.; Walasek, M.A.; Szilvassy, S.J.; Turhan, A.G.; Louis, S.A.; Eaves, A.C.; Wognum, A.W. Feeder-Free and Serum-Free in Vitro Assay for Measuring the Effect of Drugs on Acute and Chronic Myeloid Leukemia Stem/Progenitor Cells. Exp. Hematol. 2020, 90, 52–64.e11. [Google Scholar] [CrossRef]
- Pabst, C.; Krosl, J.; Fares, I.; Boucher, G.; Ruel, R.; Marinier, A.; Lemieux, S.; Hébert, J.; Sauvageau, G. Identification of Small Molecules That Support Human Leukemia Stem Cell Activity Ex Vivo. Nat. Methods 2014, 11, 436–442. [Google Scholar] [CrossRef]
- Huang, J.; Nguyen-McCarty, M.; Hexner, E.O.; Danet-Desnoyers, G.; Klein, P.S. Maintenance of Hematopoietic Stem Cells through Regulation of Wnt and MTOR Pathways. Nat. Med. 2012, 18, 1778–1785. [Google Scholar] [CrossRef]
- Bhavanasi, D.; Wen, K.W.; Liu, X.; Vergez, F.; Danet-Desnoyers, G.; Carroll, M.; Huang, J.; Klein, P.S. Signaling Mechanisms That Regulate Ex Vivo Survival of Human Acute Myeloid Leukemia Initiating Cells. Blood Cancer J. 2017, 7, 636. [Google Scholar] [CrossRef][Green Version]
- Wilkinson, A.C.; Igarashi, K.J.; Nakauchi, H. Haematopoietic Stem Cell Self-Renewal in Vivo and Ex Vivo. Nat. Rev. Genet. 2020, 21, 541–554. [Google Scholar] [CrossRef]
- Breems, D.A.; Blokland, E.A.W.; Siebel, K.E.; Mayen, A.E.M.; Engels, L.J.A.; Ploemacher, R.E. Stroma-Contact Prevents Loss of Hematopoietic Stem Cell Quality During Ex Vivo Expansion of CD34+ Mobilized Peripheral Blood Stem Cells. Blood 1998, 91, 111–117. [Google Scholar] [CrossRef]
- Robinson, S.N.; Ng, J.; Niu, T.; Yang, H.; McMannis, J.D.; Karandish, S.; Kaur, I.; Fu, P.; Del Angel, M.; Messinger, R.; et al. Superior Ex Vivo Cord Blood Expansion Following Co-Culture with Bone Marrow-Derived Mesenchymal Stem Cells. Bone Marrow Transplant. 2006, 37, 359–366. [Google Scholar] [CrossRef]
- de Lima, M.; McNiece, I.; Robinson, S.N.; Munsell, M.; Eapen, M.; Horowitz, M.; Alousi, A.; Saliba, R.; McMannis, J.D.; Kaur, I.; et al. Cord-Blood Engraftment with Ex Vivo Mesenchymal-Cell Coculture. N. Engl. J. Med. 2012, 367, 2305–2315. [Google Scholar] [CrossRef]
- Méndez-Ferrer, S.; Michurina, T.V.; Ferraro, F.; Mazloom, A.R.; MacArthur, B.D.; Lira, S.A.; Scadden, D.T.; Ma’Ayan, A.; Enikolopov, G.N.; Frenette, P.S. Mesenchymal and Haematopoietic Stem Cells Form a Unique Bone Marrow Niche. Nature 2010, 466, 829–834. [Google Scholar] [CrossRef]
- Nakahara, F.; Borger, D.K.; Wei, Q.; Pinho, S.; Maryanovich, M.; Zahalka, A.H.; Suzuki, M.; Cruz, C.D.; Wang, Z.; Xu, C.; et al. Engineering a Haematopoietic Stem Cell Niche by Revitalizing Mesenchymal Stromal Cells. Nat. Cell Biol. 2019, 21, 560–567. [Google Scholar] [CrossRef]
- Butler, J.M.; Nolan, D.J.; Vertes, E.L.; Varnum-Finney, B.; Kobayashi, H.; Hooper, A.T.; Seandel, M.; Shido, K.; White, I.A.; Kobayashi, M.; et al. Endothelial Cells Are Essential for the Self-Renewal and Repopulation of Notch-Dependent Hematopoietic Stem Cells. Cell Stem Cell 2010, 6, 251–264. [Google Scholar] [CrossRef]
- Seandel, M.; Butler, J.M.; Kobayashi, H.; Hooper, A.T.; White, I.A.; Zhang, F.; Vertes, E.L.; Kobayashi, M.; Zhang, Y.; Shmelkov, S.V.; et al. Generation of a Functional and Durable Vascular Niche by the Adenoviral E4ORF1 Gene. Proc. Natl. Acad. Sci. USA 2008, 105, 19288–19293. [Google Scholar] [CrossRef]
- Li, H.; Pei, H.; Xie, X.; Wang, S.; Jia, Y.; Zhang, B.; Fan, Z.; Liu, Y.; Bai, Y.; Han, Y.; et al. Liver Sinusoidal Endothelial Cells Promote the Expansion of Human Cord Blood Hematopoietic Stem and Progenitor Cells. Int. J. Mol. Sci. 2019, 20, 1985. [Google Scholar] [CrossRef]
- Azadniv, M.; Myers, J.R.; McMurray, H.R.; Guo, N.; Rock, P.; Coppage, M.L.; Ashton, J.; Becker, M.W.; Calvi, L.M.; Liesveld, J.L. Bone Marrow Mesenchymal Stromal Cells from Acute Myelogenous Leukemia Patients Demonstrate Adipogenic Differentiation Propensity with Implications for Leukemia Cell Support. Leukemia 2020, 34, 391–403. [Google Scholar] [CrossRef]
- Bruserud, Ø.; Gjertsen, B.T.; von Volkman, H.L. In Vitro Culture of Human Acute Myelogenous Leukemia (AML) Cells in Serum-Free Media: Studies of Native AML Blasts and AML Cell Lines. J. Hematother. Stem Cell Res. 2000, 9, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Garrido, S.M.; Appelbaum, F.R.; Willman, C.L.; Banker, D.E. Acute Myeloid Leukemia Cells Are Protected from Spontaneous and Drug-Induced Apoptosis by Direct Contact with a Human Bone Marrow Stromal Cell Line (HS-5). Exp. Hematol. 2001, 29, 448–457. [Google Scholar] [CrossRef]
- Mayani, H.; Flores-Figueroa, E.; Chávez-González, A. In Vitro Biology of Human Myeloid Leukemia. Leuk. Res. 2009, 33, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Cucchi, D.G.J.; Groen, R.W.J.; Janssen, J.J.W.M.; Cloos, J. Ex Vivo Cultures and Drug Testing of Primary Acute Myeloid Leukemia Samples: Current Techniques and Implications for Experimental Design and Outcome. Drug Resist. Updat. 2020, 53, 100730. [Google Scholar] [CrossRef]
- van Gosliga, D.; Schepers, H.; Rizo, A. Establishing Long-Term Cultures with Self-Renewing Acute Myeloid Leukemia Stem/Progenitor Cells. Exp. Hematol. 2007, 35, 1538–1549. [Google Scholar] [CrossRef]
- Griessinger, E.; Anjos-Afonso, F.; Pizzitola, I.; Rouault-Pierre, K.; Vargaftig, J.; Taussig, D.; Gribben, J.; Lassailly, F.; Bonnet, D. A Niche-Like Culture System Allowing the Maintenance of Primary Human Acute Myeloid Leukemia-Initiating Cells: A New Tool to Decipher Their Chemoresistance and Self-Renewal Mechanisms. Stem Cells Transl. Med. 2014, 3, 520–529. [Google Scholar] [CrossRef]
- Rombouts, W.; Broyl, A.; Martens, A.; Slater, R.; Ploemacher, R. Human Acute Myeloid Leukemia Cells with Internal Tandem Duplications in the Flt3 Gene Show Reduced Proliferative Ability in Stroma Supported Long-Term Cultures. Leukemia 1999, 13, 1071–1078. [Google Scholar] [CrossRef]
- Zhou, B.; Tsaknakis, G.; Coldwell, K.E.; Khoo, C.P.; Roubelakis, M.G.; Chang, C.-H.; Pepperell, E.; Watt, S.M. A Novel Function for the Haemopoietic Supportive Murine Bone Marrow MS-5 Mesenchymal Stromal Cell Line in Promoting Human Vasculogenesis and Angiogenesis. Br. J. Haematol. 2012, 157, 299–311. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.; Malek, S.N.; Zheng, P.; Liu, Y. Targeting HIF1α Eliminates Cancer Stem Cells in Hematological Malignancies. Cell Stem Cell 2011, 8, 399–411. [Google Scholar] [CrossRef]
- Chandran, P.; Le, Y.; Li, Y.; Sabloff, M.; Mehic, J.; Rosu-Myles, M.; Allan, D.S. Mesenchymal Stromal Cells from Patients with Acute Myeloid Leukemia Have Altered Capacity to Expand Differentiated Hematopoietic Progenitors. Leuk. Res. 2015, 39, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Geyh, S.; Rodríguez-Paredes, M.; Jäger, P.; Khandanpour, C.; Cadeddu, R.-P.; Gutekunst, J.; Wilk, C.M.; Fenk, R.; Zilkens, C.; Hermsen, D.; et al. Functional Inhibition of Mesenchymal Stromal Cells in Acute Myeloid Leukemia. Leukemia 2016, 30, 683–691. [Google Scholar] [CrossRef]
- Battula, V.L.; Le, P.M.; Sun, J.C.; Nguyen, K.; Yuan, B.; Zhou, X.; Sonnylal, S.; McQueen, T.; Ruvolo, V.; Michel, K.A.; et al. AML-Induced Osteogenic Differentiation in Mesenchymal Stromal Cells Supports Leukemia Growth. JCI Insight 2017, 2, e90036. [Google Scholar] [CrossRef] [PubMed]
- Binato, R.; de Almeida Oliveira, N.C.; Du Rocher, B.; Abdelhay, E. The Molecular Signature of AML Mesenchymal Stromal Cells Reveals Candidate Genes Related to the Leukemogenic Process. Cancer Lett. 2015, 369, 134–143. [Google Scholar] [CrossRef]
- Desbourdes, L.; Javary, J.; Charbonnier, T.; Ishac, N.; Bourgeais, J.; Iltis, A.; Chomel, J.-C.; Turhan, A.; Guilloton, F.; Tarte, K.; et al. Alteration Analysis of Bone Marrow Mesenchymal Stromal Cells from De Novo Acute Myeloid Leukemia Patients at Diagnosis. Stem Cells Dev. 2017, 26, 709–722. [Google Scholar] [CrossRef]
- Le, Y.; Fraineau, S.; Chandran, P.; Sabloff, M.; Brand, M.; Lavoie, J.R.; Gagne, R.; Rosu-Myles, M.; Yauk, C.L.; Richardson, R.B.; et al. Adipogenic Mesenchymal Stromal Cells from Bone Marrow and Their Hematopoietic Supportive Role: Towards Understanding the Permissive Marrow Microenvironment in Acute Myeloid Leukemia. Stem Cell Rev. Rep. 2016, 12, 235–244. [Google Scholar] [CrossRef]
- Chang-Liu, C.-M.; Woloschak, G.E. Effect of Passagenumber on Cellular Response to DNA-Damaging Agents: Cell Survival and Gene Expression. Cancer Lett. 1995, 10, 77–86. [Google Scholar] [CrossRef]
- Raic, A.; Naolou, T.; Mohra, A.; Chatterjee, C.; Lee-Thedieck, C. 3D Models of the Bone Marrow in Health and Disease: Yesterday, Today, and Tomorrow. MRS Commun. 2019, 9, 37–52. [Google Scholar] [CrossRef]
- Salamanna, F.; Contartese, D.; Maglio, M.; Fini, M. A Systematic Review on in Vitro 3D Bone Metastases Models: A New Horizon to Recapitulate the Native Clinical Scenario? Oncotarget 2016, 7, 44803–44820. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, C.; Hellwarth, P.B.; Bao, X. Biomaterials for stem cell engineering and biomanufacturing. Bioact. Mater. 2019, 4, 366–379. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.M.; Futrega, K.; Osiecki, M.; Kabiri, M.; Kul, B.; Rice, A.; Atkinson, K.; Brooke, G.; Doran, M. Micromarrows—Three-Dimensional Coculture of Hematopoietic Stem Cells and Mesenchymal Stromal Cells. Tissue Eng. Part C Methods 2012, 18, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Edmondson, R.; Broglie, J.J.; Adcock, A.F.; Yang, L. Three-Dimensional Cell Culture Systems and Their Applications in Drug Discovery and Cell-Based Biosensors. Assay Drug Dev. Technol. 2014, 12, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Białkowska, K.; Komorowski, P.; Bryszewska, M.; Miłowska, K. Spheroids as a Type of Three-Dimensional Cell Cultures-Examples of Methods of Preparation and the Most Important Application. Int. J. Mol. Sci. 2020, 21, 6225. [Google Scholar] [CrossRef]
- Ham, S.L.; Joshi, R.; Thakuri, P.S.; Tavana, H. Liquid-Based Three-Dimensional Tumor Models for Cancer Research and Drug Discovery. Exp. Biol. Med. 2016, 241, 939–954. [Google Scholar] [CrossRef]
- Lewis, E.E.L.; Wheadon, H.; Lewis, N.; Yang, J.; Mullin, M.; Hursthouse, A.; Stirling, D.; Dalby, M.J.; Berry, C.C. A Quiescent, Regeneration-Responsive Tissue Engineered Mesenchymal Stem Cell Bone Marrow Niche Model via Magnetic Levitation. ACS Nano 2016, 10, 8346–8354. [Google Scholar] [CrossRef]
- Costa, E.C.; de Melo-Diogo, D.; Moreira, A.F.; Carvalho, M.P.; Correia, I.J. Spheroids Formation on Non-Adhesive Surfaces by Liquid Overlay Technique: Considerations and Practical Approaches. Biotechnol. J. 2018, 13, 1700417. [Google Scholar] [CrossRef]
- Lee-Thedieck, C.; Spatz, J.P. Artificial Niches: Biomimetic Materials for Hematopoietic Stem Cell Culture. Macromol. Rapid Commun. 2012, 33, 1432–1438. [Google Scholar] [CrossRef]
- Liu, S.; Tang, J.; Ji, F.; Lin, W.; Chen, S. Recent Advances in Zwitterionic Hydrogels: Preparation, Property, and Biomedical Application. Gels 2022, 8, 46. [Google Scholar] [CrossRef]
- Bai, T.; Li, J.; Sinclair, A.; Imren, S.; Merriam, F.; Sun, F.; O’Kelly, M.B.; Nourigat, C.; Jain, P.; Delrow, J.J.; et al. Expansion of Primitive Human Hematopoietic Stem Cells by Culture in a Zwitterionic Hydrogel. Nat. Med. 2019, 25, 1566–1575. [Google Scholar] [CrossRef]
- Jiang, S.; Cao, Z. Ultralow-Fouling, Functionalizable, and Hydrolyzable Zwitterionic Materials and Their Derivatives for Biological Applications. Adv. Mater. 2010, 22, 920–932. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, L.J.; Rogers, Z.J.; Colombani, T.; Memic, A.; Bencherif, S.A. Injectable Cryogels for Biomedical Applications. Trends Biotechnol. 2020, 38, 418–431. [Google Scholar] [CrossRef] [PubMed]
- Raic, A.; Rödling, L.; Kalbacher, H.; Lee-Thedieck, C. Biomimetic Macroporous PEG Hydrogels as 3D Scaffolds for the Multiplication of Human Hematopoietic Stem and Progenitor Cells. Biomaterials 2014, 35, 929–940. [Google Scholar] [CrossRef] [PubMed]
- Ventura Ferreira, M.S.; Jahnen-Dechent, W.; Labude, N.; Bovi, M.; Hieronymus, T.; Zenke, M.; Schneider, R.K.; Neurs, S. Cord Blood-Hematopoietic Stem Cell Expansion in 3D Fibrin Scaffolds with Stromal Support. Biomaterials 2012, 33, 6987–6997. [Google Scholar] [CrossRef] [PubMed]
- Franc, S.; Rousseau, J.C.; Garrone, R.; van der Rest, M.; Moradi-Améli, M. Microfibrillar Composition of Umbilical Cord Matrix: Characterization of Fibrillin, Collagen VI and Intact Collagen, V. Placenta 1998, 19, 95–104. [Google Scholar] [CrossRef]
- Jadalannagari, S.; Converse, G.; McFall, C.; Buse, E.; Filla, M.; Villar, M.T.; Artigues, A.; Mellot, A.J.; Wang, J.; Detamore, M.S.; et al. Decellularized Wharton’s Jelly from Human Umbilical Cord as a Novel 3D Scaffolding Material for Tissue Engineering Applications. PLoS ONE 2017, 12, e0172098. [Google Scholar] [CrossRef]
- Rödling, L.; Schwedhelm, I.; Kraus, S.; Bieback, K.; Hansmann, J.; Lee-Thedieck, C. 3D Models of the Hematopoietic Stem Cell Niche under Steady-State and Active Conditions. Sci. Rep. 2017, 7, 4625. [Google Scholar] [CrossRef]
- Bessy, T.; Itkin, T.; Passaro, D. Bioengineering the Bone Marrow Vascular Niche. Front. Cell Dev. Biol. 2021, 9, 645496. [Google Scholar] [CrossRef]
- Kotha, S.S.; Hayes, B.J.; Phong, K.T.; Redd, M.A.; Bomsztyk, K.; Ramakrishnan, A.; Torok-Storb, B.; Zheng, Y. Engineering a Multicellular Vascular Niche to Model Hematopoietic Cell Trafficking. Stem Cell Res. Ther. 2018, 9, 77. [Google Scholar] [CrossRef]
- Reinisch, A.; Hernandez, D.C.; Schallmoser, K.; Majeti, R. Generation and Use of a Humanized Bone-Marrow-Ossicle Niche for Hematopoietic Xenotransplantation into Mice. Nat. Protoc. 2017, 12, 2169–2188. [Google Scholar] [CrossRef]
- Torisawa, Y.; Spina, C.S.; Mammoto, T.; Mammoto, A.; Weaver, J.C.; Tat, T.; Collins, J.J.; Ingber, D.E. Bone Marrow–on–a–Chip Replicates Hematopoietic Niche Physiology in Vitro. Nat. Methods 2014, 11, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Aljitawi, O.S.; Li, D.; Xiao, Y.; Zhang, D.; Ramachandran, K.; Stehno-Bittel, L.; Van Veldhuizen, P.; Lin, T.L.; Kambhampati, S.; Garimella, R. A Novel Three-Dimensional Stromal-Based Model for in Vitro Chemotherapy Sensitivity Testing of Leukemia Cells. Leuk. Lymphoma 2014, 55, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Blanco, T.M.; Mantalaris, A.; Bismarck, A.; Panoskaltsis, N. The Development of a Three-Dimensional Scaffold for Ex Vivo Biomimicry of Human Acute Myeloid Leukaemia. Biomaterials 2010, 31, 2243–2251. [Google Scholar] [CrossRef]
- Bray, L.J.; Binner, M.; Körner, Y.; von Bonin, M.; Bornhäuser, M.; Werner, C. A Three-Dimensional Ex Vivo Tri-Culture Model Mimics Cell-Cell Interactions between Acute Myeloid Leukemia and the Vascular Niche. Haematologica 2017, 102, 1215–1226. [Google Scholar] [CrossRef] [PubMed]
- Leisten, I.; Kramann, R.; Ventura Ferreira, M.S.; Bovi, M.; Neuss, S.; Ziegler, P.; Wagner, W.; Knüchel, R.; Schneider, R.K. 3D Co-Culture of Hematopoietic Stem and Progenitor Cells and Mesenchymal Stem Cells in Collagen Scaffolds as a Model of the Hematopoietic Niche. Biomaterials 2012, 33, 1736–1747. [Google Scholar] [CrossRef]
- Tun, T.; Miyoshi, H.; Aung, T.; Takahashi, S.; Shimizu, R.; Kuroha, T.; Yamamoto, M.; Ohshima, N. Effect of Growth Factors on Ex Vivo Bone Marrow Cell Expansion Using Three-Dimensional Matrix Support. Artif. Organs 2002, 26, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Velliou, E.G.; Dos Santos, S.B.; Papathanasiou, M.M.; Fuentes-Gari, M.; Misener, R.; Panoskaltsis, N.; Pistikopoulos, E.N.; Mantalaris, A. Towards Unravelling the Kinetics of an Acute Myeloid Leukaemia Model System under Oxidative and Starvation Stress: A Comparison between Two- and Three-Dimensional Cultures. Bioprocess Biosyst. Eng. 2015, 38, 1589–1600. [Google Scholar] [CrossRef]
- Argiropoulos, B.; Humphries, R.K. Hox Genes in Hematopoiesis and Leukemogenesis. Oncogene 2007, 26, 6766–6776. [Google Scholar] [CrossRef]
- Doma, E.; Mayer, I.M.; Brandstoetter, T.; Maurer, B.; Grausenburger, R.; Menzl, I.; Zojer, M.; Hoelbl-Kovacic, A.; Carlsson, L.; Heller, G.; et al. A Robust Approach for the Generation of Functional Hematopoietic Progenitor Cell Lines to Model Leukemic Transformation. Blood Adv. 2021, 5, 39–53. [Google Scholar] [CrossRef]
- Geoffroy, M.-C.; Esnault, C.; de Thé, H. Retinoids in Hematology: A Timely Revival? Blood 2021, 137, 2429–2437. [Google Scholar] [CrossRef]
- Sauvageau, G. Overexpression of HOXB4 in Hematopoietic Cells Causes the Selective Expansion of more Primitive Populations in Vitro and Vivo. Genes Dev. 1995, 14, 1753–1765. [Google Scholar] [CrossRef] [PubMed]
- Redecke, V.; Wu, R.; Zhou, J.; Finkelstein, D.; Chaturvedi, V.; High, A.A.; Häcker, H. Hematopoietic Progenitor Cell Lines with Myeloid and Lymphoid Potential. Nat. Methods 2013, 10, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.; Sitnicka, E.; Collins, S. Lymphohematopoietic Progenitors Immortalized by a Retroviral Vector Harboring a Dominant-Negative Retinoic Acid Receptor Can Recapitulate Lymphoid, Myelold, and Erythrold Development. Genes Dev. 1994, 12, 2831–2841. [Google Scholar] [CrossRef]
- Lappin, T.R.; Grier, D.G.; Thompson, A.; Halliday, H.L. Hox Genes: Seductive Science, Mysterious Mechanisms. Ulst. Med. J. 2006, 9, 23–31. [Google Scholar]
- Abuhantash, M.; Collins, E.M.; Thompson, A. Role of the HOXA Cluster in HSC Emergence and Blood Cancer. Biochem. Soc. Trans. 2021, 49, 1817–1827. [Google Scholar] [CrossRef]
- Yu, H.; Neale, G.; Zhang, H.; Lee, H.M.; Ma, Z.; Zhou, S.; Forget, B.G.; Sorrentino, B.P. Downregulation of Prdm16 MRNA Is a Specific Antileukemic Mechanism during HOXB4-Mediated HSC Expansion in Vivo. Blood 2014, 124, 1737–1747. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Morita, K.; Hill, M.C.; Jiang, Y.; Kitano, A.; Saito, Y.; Wang, F.; Mao, X.; Hoegenauer, K.A.; Morishita, K.; et al. PRDM16s Transforms Megakaryocyte-Erythroid Progenitors into Myeloid Leukemia–Initiating Cells. Blood 2019, 134, 614–625. [Google Scholar] [CrossRef]
- Cheng, H.; Zheng, Z.; Cheng, T. New Paradigms on Hematopoietic Stem Cell Differentiation. Protein Cell 2020, 11, 34–44. [Google Scholar] [CrossRef]
- Antonchuk, J.; Sauvageau, G.; Humphries, R.K. HOXB4-Induced Expansion of Adult Hematopoietic Stem Cells Ex Vivo. Cell 2002, 109, 39–45. [Google Scholar] [CrossRef]
- Krosl, J.; Sauvageau, G. AP-1 Complex Is Effector of Hox-Induced Cellular Proliferation and Transformation. Oncogene 2000, 8, 5134–5141. [Google Scholar] [CrossRef]
- Ohta, H.; Sekulovic, S.; Bakovic, S.; Eaves, C.J.; Pineault, N.; Gasparetto, M.; Smith, C.; Sauvageau, G.; Humphries, R.K. Near-Maximal Expansions of Hematopoietic Stem Cells in Culture Using NUP98-HOX Fusions. Exp. Hematol. 2007, 35, 817–830. [Google Scholar] [CrossRef]
- Beslu, N.; Krosl, J.; Laurin, M.; Mayotte, N.; Humphries, K.R.; Sauvageau, G. Molecular Interactions Involved in HOXB4-Induced Activation of HSC Self-Renewal. Blood 2004, 104, 2307–2314. [Google Scholar] [CrossRef] [PubMed]
- Cellot, S.; Krosl, J.; Chagraoui, J.; Meloche, S.; Humphries, R.K.; Sauvageau, G. Sustained in Vitro Trigger of Self-Renewal Divisions in Hoxb4hiPbx1lo Hematopoietic Stem Cells. Exp. Hematol. 2007, 35, 802.e1–802.e19. [Google Scholar] [CrossRef]
- Pineault, N.; Abramovich, C.; Ohta, H.; Humphries, R.K. Differential and Common Leukemogenic Potentials of Multiple NUP98-Hox Fusion Proteins Alone or with Meis1. Mol. Cell. Biol. 2004, 24, 1907–1917. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.; Broxmeyer, H.E.; Lee, M.R. Generating Autologous Hematopoietic Cells from Human-Induced Pluripotent Stem Cells through Ectopic Expression of Transcription Factors. Curr. Opin. Hematol. 2017, 24, 283–288. [Google Scholar] [CrossRef]
- Izawa, K.; Yamazaki, S.; Becker, H.J.; Bhadury, J.; Kakegawa, T.; Sakaguchi, M.; Tojo, A. Activated HoxB4-Induced Hematopoietic Stem Cells from Murine Pluripotent Stem Cells via Long-Term Programming. Exp. Hematol. 2020, 89, 68–79.e7. [Google Scholar] [CrossRef] [PubMed]
- Weston, W.; Zayas, J.; Perez, R.; George, J.; Jurecic, R. Dynamic Equilibrium of Heterogeneous and Interconvertible Multipotent Hematopoietic Cell Subsets. Sci. Rep. 2015, 4, 5199. [Google Scholar] [CrossRef]
- Dai, B.; Wang, P. In Vitro Differentiation of Adult Bone Marrow Progenitors into Antigen-Specific CD4 Helper T Cells Using Engineered Stromal Cells Expressing a Notch Ligand and a Major Histocompatibility Complex Class II Protein. Stem Cells Dev. 2009, 18, 235–245. [Google Scholar] [CrossRef]
- Kutlesa, S.; Zayas, J.; Valle, A.; Levy, R.B.; Jurecic, R. T-Cell Differentiation of Multipotent Hematopoietic Cell Line EML in the OP9-DL1 Coculture System. Exp. Hematol. 2009, 37, 909–923. [Google Scholar] [CrossRef][Green Version]
- Pina, C.; Fugazza, C.; Tipping, A.J.; Brown, J.; Soneji, S.; Teles, J.; Peterson, C.; Enver, T. Inferring Rules of Lineage Commitment in Haematopoiesis. Nat. Cell Biol. 2012, 14, 287–294. [Google Scholar] [CrossRef]
- Ye, Z.-J.; Kluger, Y.; Lian, Z.; Weissman, S.M. Two Types of Precursor Cells in a Multipotential Hematopoietic Cell Line. Proc. Natl. Acad. Sci. USA 2005, 102, 18461–18466. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.M.; Zhang, H.; Schulz, V.; Tuck, D.P.; Forget, B.G. Downstream Targets of HOXB4 in a Cell Line Model of Primitive Hematopoietic Progenitor Cells. Blood 2010, 116, 720–730. [Google Scholar] [CrossRef] [PubMed]
- Pinto do O, P.; Kolterud, Å.; Carlsson, L. Expression of the LIM-Homeobox Gene LH2 Generates Immortalized Steel Factor-Dependent Multipotent Hematopoietic Precursors. EMBO J. 1998, 17, 5744–5756. [Google Scholar] [CrossRef] [PubMed]
- do Pinto, Ó.P.; Richter, K.; Carlsson, L. Hematopoietic Progenitor/Stem Cells Immortalized ByLhx2 Generate Functional Hematopoietic Cells in Vivo. Blood 2002, 99, 3939–3946. [Google Scholar] [CrossRef] [PubMed]
- Hobert, O.; Westphal, H. Functions of LIM-Homeobox Genes. Trends Genet. 2000, 16, 75–83. [Google Scholar] [CrossRef]
- Porter, F.D.; Drago, J.; Xu, Y.; Cheema, S.S.; Wassif, C.; Huang, S.-P.; Lee, E.; Grinberg, A.; Massalas, J.S.; Bodine, D.; et al. Lhx2, a LIM Homeobox Gene, Is Required for Eye, Forebrain, and Definitive Erythrocyte Development. Development 1997, 124, 2935–2944. [Google Scholar] [CrossRef]
- Kitajima, K.; Kawaguchi, M.; Iacovino, M.; Kyba, M.; Hara, T. Molecular Functions of the LIM-Homeobox Transcription Factor Lhx2 in Hematopoietic Progenitor Cells Derived from Mouse Embryonic Stem Cells. Stem Cells 2013, 31, 2680–2689. [Google Scholar] [CrossRef]
- Adnan-Awad, S.; Kim, D.; Hohtari, H.; Javarappa, K.K.; Brandstoetter, T.; Mayer, I.; Potdar, S.; Heckman, C.A.; Kytölä, S.; Porkka, K.; et al. Characterization of P190-Bcr-Abl Chronic Myeloid Leukemia Reveals Specific Signaling Pathways and Therapeutic Targets. Leukemia 2021, 35, 1964–1975. [Google Scholar] [CrossRef]
- Schmalzbauer, B.S.; Thondanpallil, T.; Heller, G.; Schirripa, A.; Sperl, C.M.; Mayer, I.M.; Knab, V.M.; Nebenfuehr, S.; Zojer, M.; Mueller, A.C.; et al. CDK6 Degradation Is Counteracted by p16INK4A and p18INK4C in AML. Cancers 2022, 14, 1554. [Google Scholar] [CrossRef]
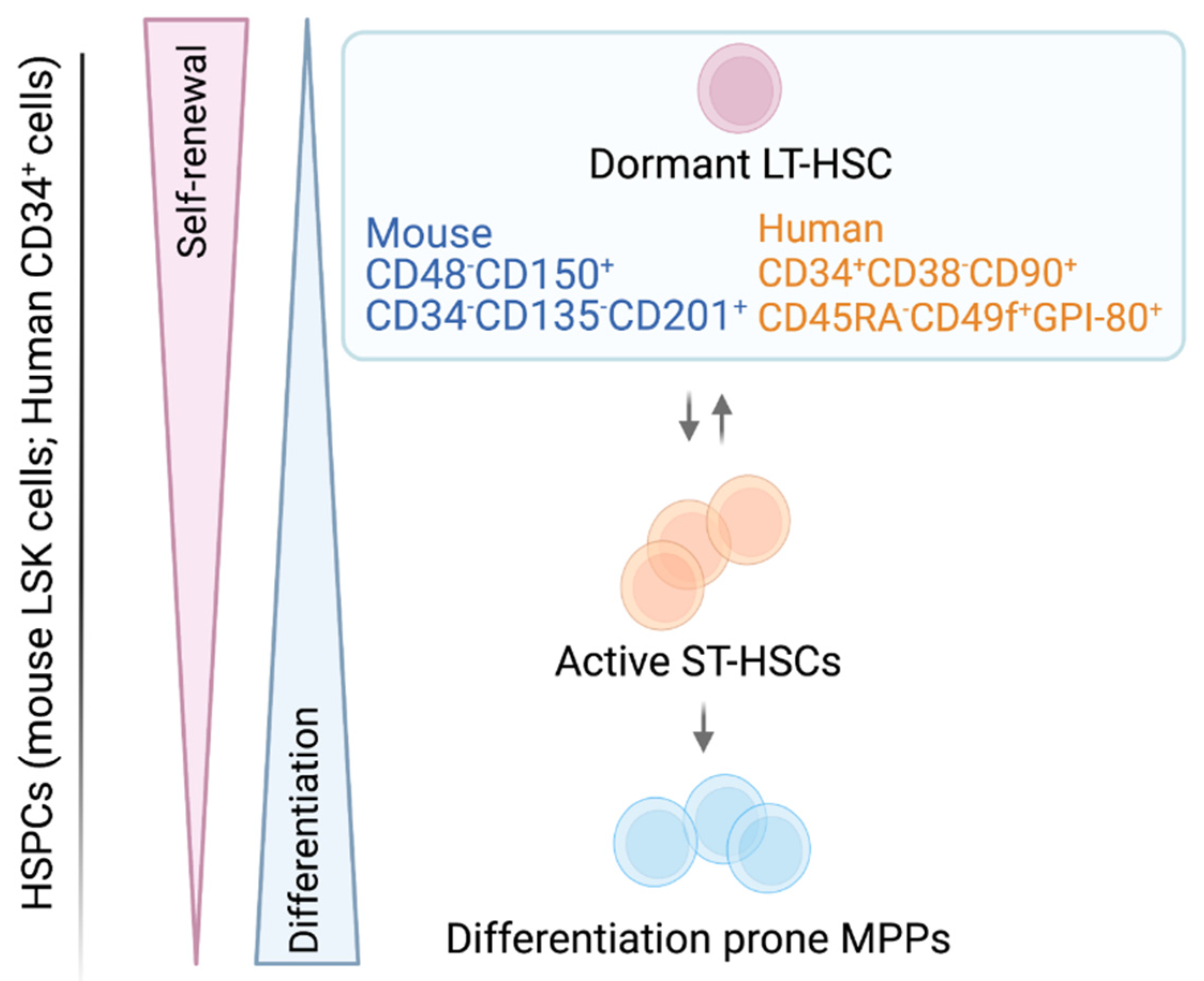
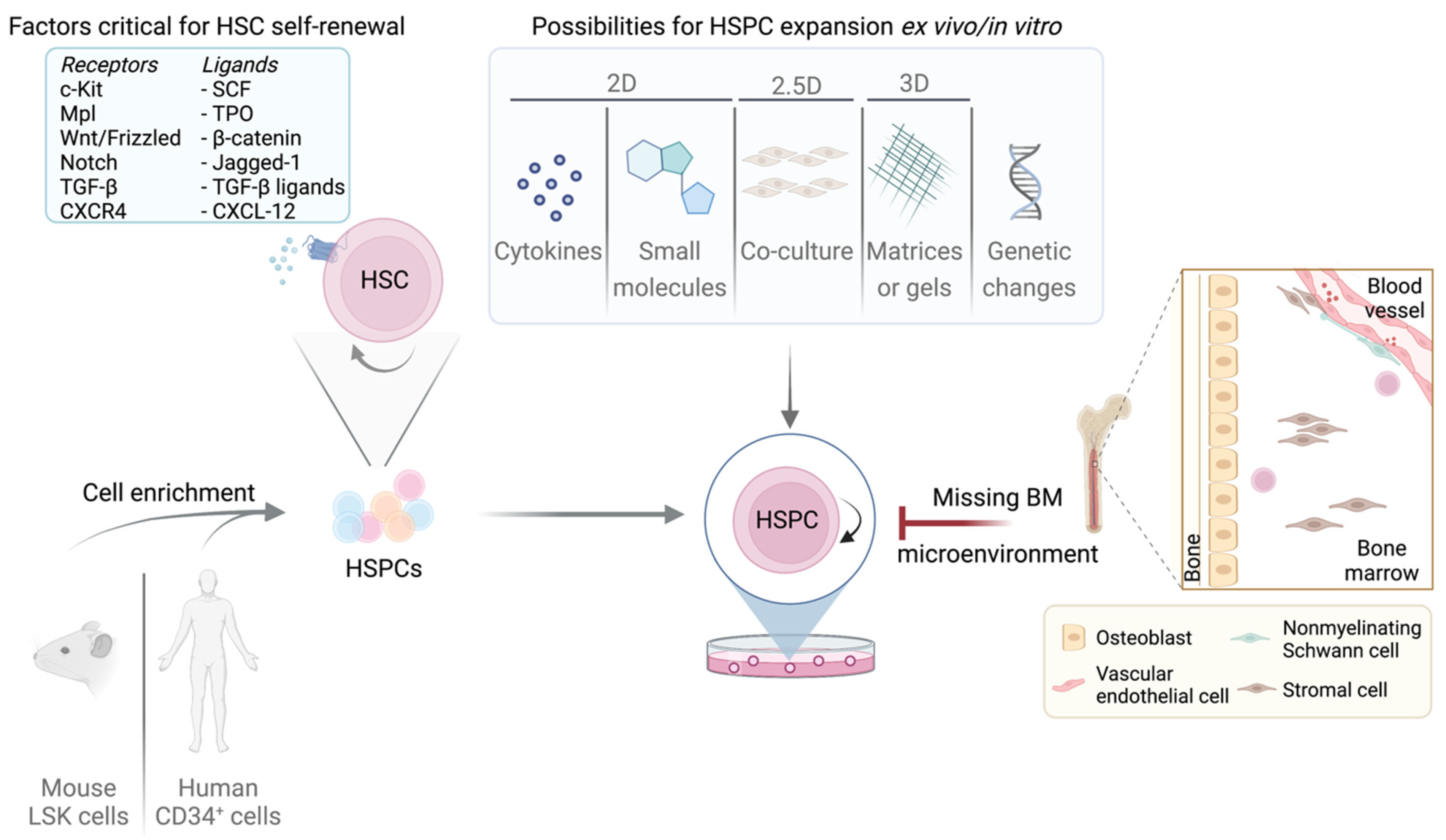
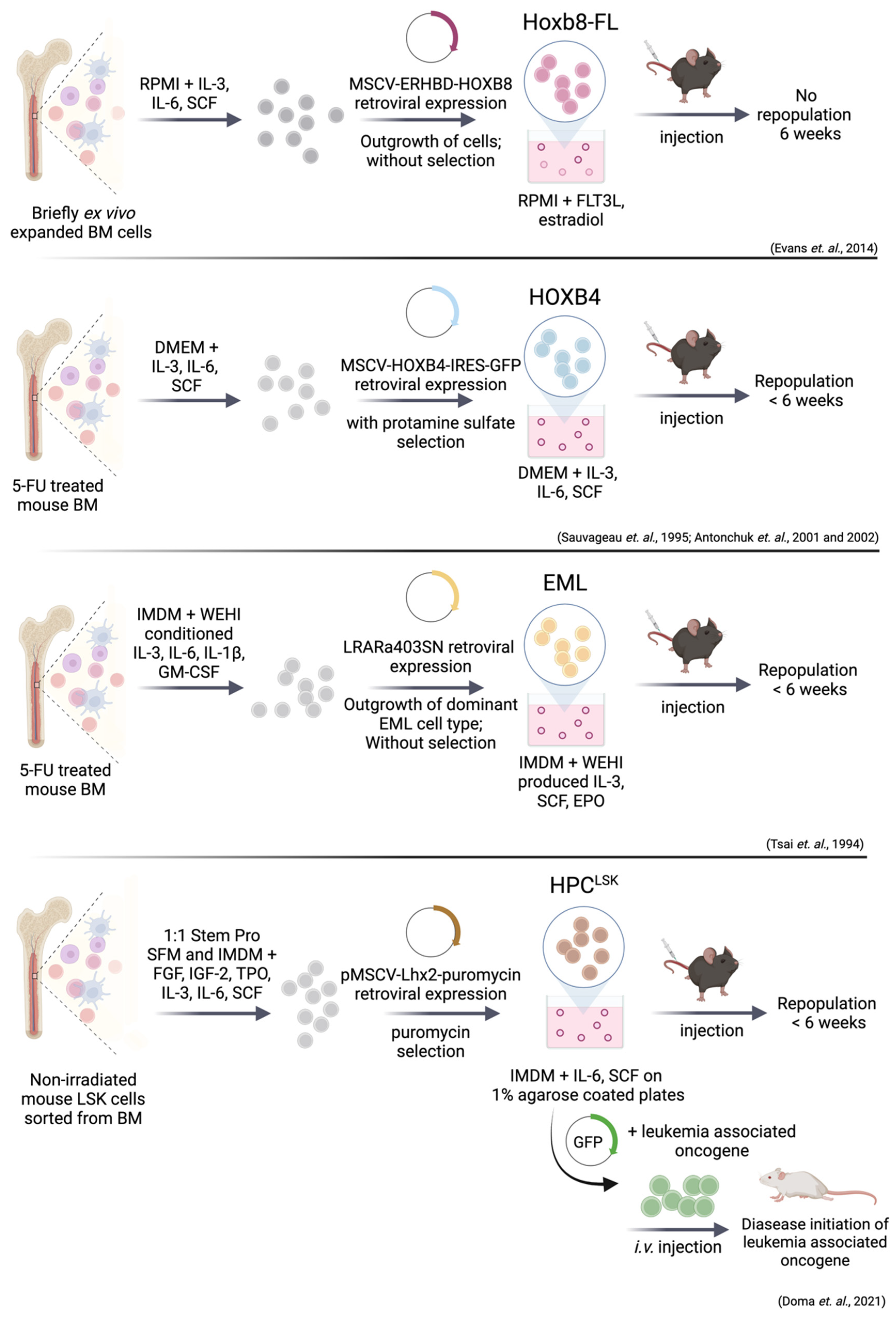
| Human LSC Surface Markers | |||||
|---|---|---|---|---|---|
| AML and CML Markers | Alternative Name | Refs. | AML and CML Markers | Alternative Name | Refs. |
| CD25 | IL-2Rα chain | [71,72,73] | CD33 | Siglec-3 | [72,74,75,76] |
| CD45RA | CD45, also known as protein tyrosine phosphatase, receptor (PTPRC) | [77,78] | CD93 | C-type lectin-like domain (CTLD) containing glycoprotein | [79,80,81] |
| CD9 | Motility Related Protein-1 (MRP-1) Tetraspanin-29 (TSPAN29) | [72,82,83] | CD123 | Interleukin 3 receptor subunit α (IL-3R α) | [72] |
| CD371 | C-type lectin domain family 12 member A (CLEC12A); CLL-1 antigen | [72,84] | IL1RAP | Interleukin 1 receptor accessory protein | [85,86] |
| CD69 | C-Type Lectin Domain Family 2, Member C (CLEC2C) Activation Inducer Molecule (AIM) | [87,88,89] | CD36 | Thrombospondin Receptor Platelet Collagen Receptor Glycoprotein IIIb (GP3B) | [88,89] |
| CD43 | Leukosialin | [75] | CD44 | Pgp-1, multidrug resistance protein 1 (MDR1) | [75] |
| CD45 | Leukocyte common antigen (LCA) | [75] | CD157 | Bone marrow stromal cell antigen 1 (BST1) | [75] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayer, I.M.; Hoelbl-Kovacic, A.; Sexl, V.; Doma, E. Isolation, Maintenance and Expansion of Adult Hematopoietic Stem/Progenitor Cells and Leukemic Stem Cells. Cancers 2022, 14, 1723. https://doi.org/10.3390/cancers14071723
Mayer IM, Hoelbl-Kovacic A, Sexl V, Doma E. Isolation, Maintenance and Expansion of Adult Hematopoietic Stem/Progenitor Cells and Leukemic Stem Cells. Cancers. 2022; 14(7):1723. https://doi.org/10.3390/cancers14071723
Chicago/Turabian StyleMayer, Isabella Maria, Andrea Hoelbl-Kovacic, Veronika Sexl, and Eszter Doma. 2022. "Isolation, Maintenance and Expansion of Adult Hematopoietic Stem/Progenitor Cells and Leukemic Stem Cells" Cancers 14, no. 7: 1723. https://doi.org/10.3390/cancers14071723
APA StyleMayer, I. M., Hoelbl-Kovacic, A., Sexl, V., & Doma, E. (2022). Isolation, Maintenance and Expansion of Adult Hematopoietic Stem/Progenitor Cells and Leukemic Stem Cells. Cancers, 14(7), 1723. https://doi.org/10.3390/cancers14071723






