Antibiotic Therapy in Integrated Oncology and Palliative Cancer Care: An Observational Study
Abstract
:Simple Summary
Abstract
1. Introduction
- What are the differences and similarities in PROMs and clinical and paraclinical features at admission for palliative care cancer patients with and without infections?
- What are the differences and similarities in the development of symptoms and clinical and paraclinical features during the hospital stay for palliative cancer patients with and without infections?
- Are acutely admitted patients receiving integrated oncology services more often treated with intravenous antibiotics compared to patients receiving palliative cancer care only?
- What is the survival for patients with advanced cancer, receiving integrated oncology and palliative care services, and treated for infections necessitating acute hospital admission?
2. Materials and Methods
2.1. Study Design
2.2. Data Collection and Assessments
2.3. Statistical Analysis
2.4. Ethics
3. Results
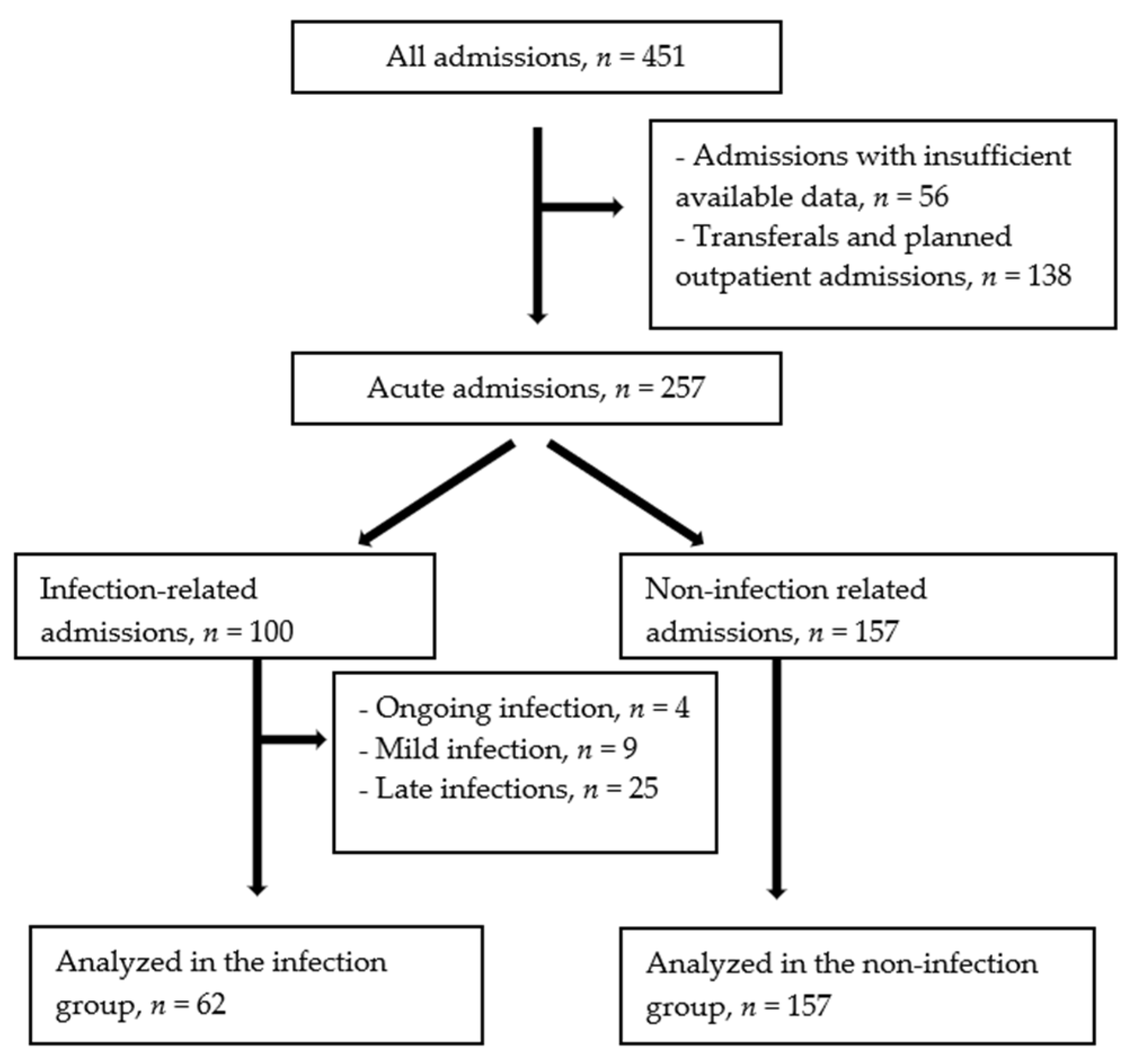
3.1. Inclusions and Exclusions
3.2. Patient Demographics
3.3. Infection Characteristics
3.4. Group Comparison at Admission
3.5. Dynamic Group Comparison during the Hospital Stay
3.6. Antibiotic Treatment and Survival in Patients with and without Integrated Oncology Care
4. Discussion
4.1. Statement of Principal Findings
4.2. Appraisal of Methods
4.3. Comparison with Previous Work
4.4. Implications and Further Work
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Admission Mean | Day 10. Mean | Difference | 95% Confidence Interval of the Difference | p-Value, * | ||
|---|---|---|---|---|---|---|
| Physiology | Systolic blood pressure, mmHg | 122 | 137 | −15.06 | −22.24–(−7.87) | <0.010 |
| Heart rate, bpm | 90 | 80 | 9.51 | 4.58–14.44 | <0.001 | |
| Temperature, °C | 37.3 | 36.5 | 0.72 | 0.39–10.58 | <0.001 | |
| Total NEWS score | 2.96 | 1.36 | 1.60 | 0.87–2.34 | <0.001 | |
| Paraclinical | C-reactive protein, mg/L | 147.8 | 81.4 | 66.41 | 41.70–91.19 | <0.001 |
| Leucocytes, ×109/L | 9.9 | 8.4 | 1.48 | 0.29–2.66 | 0.016 | |
| Neutrophils, ×109/L | 8.36 | 6.77 | 1.59 | 0.43–2.74 | 0.008 | |
| Symptom, NRS | Average pain | 4.22 | 3.17 | 1.04 | 0.28–1.81 | 0.008 |
| Worst pain | 5.45 | 3.48 | 1.98 | 0.95–3.00 | <0.001 | |
| Tiredness | 5.66 | 4.40 | 1.26 | 0.57–1.94 | <0.001 | |
| Drowsiness | 5.81 | 4.45 | 1.36 | 0.63–2.09 | <0.001 | |
| Nausea | 2.09 | 1.51 | 0.57 | −0.13–1.28 | 0.109 | |
| Appetite | 4.58 | 3.76 | 0.82 | −0.21–1.85 | 0.114 | |
| Shortness of breath | 3.57 | 2.72 | 0.85 | 0.27–1.43 | 0.005 | |
| Depression | 3.36 | 2.87 | 0.49 | −0.08–1.05 | 0.087 | |
| Anxiety | 2.71 | 2.47 | 0.24 | −0.44–0.93 | 0.475 | |
| Well-being | 4.51 | 4.02 | 0.49 | −0.22–1.20 | 0.122 | |
| Sleep | 5.15 | 4.07 | 1.09 | 0.23–1.90 | 0.010 | |
| Constipation | 3.07 | 1.73 | 1.34 | 0.35–2.33 | 0.009 |
| Admission Mean | Day 10. Mean | Difference | 95% Confidence Interval of the Difference | p-Value * | ||
|---|---|---|---|---|---|---|
| Physiology | Systolic blood pressure, mmHg | 126 | 129 | −2.56 | −6.38–1.25 | 0.186 |
| Heart rate, bpm | 84 | 84 | 0.43 | −2.99–3.86 | 0.803 | |
| Temperature, °C | 36.7 | 36.7 | 0.04 | −0.11–0.20 | 0.593 | |
| Total NEWS score | 1.73 | 1.77 | −0.45 | −0.51–0.42 | 0.848 | |
| Paraclinical | C-reactive protein, mg/L | 56.0 | 56.6 | −0.69 | −9.67–8.29 | 0.879 |
| Leucocytes, ×109/L | 10.7 | 10.6 | 0.08 | −0.74–0.90 | 0.846 | |
| Neutrophils, ×109/L | 8.73 | 8.41 | 0.32 | −0.43–1.06 | 0.402 | |
| Symptom, NRS | Average pain | 4.28 | 2.88 | 1.40 | 0.88–1.92 | <0.001 |
| Worst pain | 5.48 | 4.08 | 1.40 | 0.79–2.01 | <0.001 | |
| Tiredness | 6.07 | 4.62 | 1.45 | 0.92–1.97 | <0.001 | |
| Drowsiness | 5.55 | 4.38 | 1.12 | 0.65–1.69 | <0.001 | |
| Nausea | 2.77 | 1.85 | 0.92 | 0.46–1.38 | <0.001 | |
| Appetite | 4.83 | 3.47 | 1.37 | 0.81–1.92 | <0.001 | |
| Shortness of breath | 3.48 | 2.41 | 1.06 | 0.56–1.57 | <0.001 | |
| Depression | 3.71 | 3.12 | 0.59 | 0.11–1.08 | 0.017 | |
| Anxiety | 3.08 | 2.36 | 0.71 | 0.28–1.15 | 0.002 | |
| Well-being | 5.09 | 4.05 | 1.05 | 0.49–1.61 | <0.001 | |
| Sleep | 3.74 | 3.12 | 0.63 | 0.02–1.23 | 0.042 | |
| Constipation | 3.61 | 2.36 | 1.25 | 0.61–1.89 | <0.001 |
| Integrated Cancer Care n = 81. | Palliative Cancer Care n = 136. | ||
|---|---|---|---|
| Receiving antibiotics, n (%) | 24 (30) | 37 (27) | |
| Radiology, n (%) | X-ray | 26 (32) | 52 (38) |
| Computer tomography | 34 (42) | 47 (35) | |
| Ultrasound | 7 (9) | 15 (11) | |
| Magnetic resonance imaging | 21 (26) | 10 (7) | |
| Interventions, n (%) | Radiological intervention 1 | 7 (9) | 11 (8) |
| Rehydration | 38 (47) | 68 (50) | |
| Nutrition | 10 (12) | 15 (11) | |
| Transfusion | 10 (12) | 23 (17) | |
| Surgery | 1 (5) | 0 | |
| In-hospital mortality, n (%) | 4 (5) | 26 (19) |
References
- Kaasa, S.; Loge, J.H.; Aapro, M.; Albreht, T.; Anderson, R.; Bruera, E.; Brunelli, C.; Caraceni, A.; Cervantes, A.; Currow, D.C.; et al. Integration of oncology and palliative care: A Lancet Oncology Commission. Lancet Oncol. 2018, 19, e588–e653. [Google Scholar] [CrossRef] [Green Version]
- Advancing Cancer Therapy. Nat. Cancer 2021, 2, 245–246. [CrossRef] [PubMed]
- World Health Organization. National Cancer Control Programs: Policies and Managerial Guidelines, 2nd ed.; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Macedo, F.; Nunes, C.; Ladeira, K.; Pinho, F.; Saraiva, N.; Bonito, N.; Pinto, L.; Gonçalves, F. Antimicrobial therapy in palliative care: An overview. Support Care Cancer 2018, 26, 1361–1367. [Google Scholar] [CrossRef]
- Making precision oncology the standard of care. Lancet Oncol. 2017, 18, 835. [CrossRef]
- Cardona-Morrell, M.; Kim, J.; Turner, R.M.; Anstey, M.; Mitchell, I.A.; Hillman, K. Non-beneficial treatments in hospital at the end of life: A systematic review on extent of the problem. Int. J. Qual. Health Care 2016, 28, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Hodgkinson, S.; Ruegger, J.; Field-Smith, A.; Latchem, S.; Latchem, S.H. Care of Dying Adults in the Last Days of Life. Clin. Med. 2015, 16, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Health TNDo. National Guidelines for Palliative Cancer Care; The Norwegian Directorate of Health: Oslo, Norway, 2015.
- Marra, A.R.; Puig-Asensio, M.; Balkenende, E.; Livorsi, D.J.; Goto, M.; Perencevich, E.N. Antibiotic use during end-of-life care: A systematic literature review and meta-analysis. Infect. Control Hosp. Epidemiol. 2021, 42, 523–529. [Google Scholar] [CrossRef]
- Chen, L.-K.; Chou, Y.-C.; Hsu, P.-S.; Tsai, S.-T.; Hwang, S.-J.; Wu, B.-Y.; Lin, M.-H.; Chen, T.-W. Antibiotic prescription for fever episodes in hospice patients. Support Care Cancer 2002, 10, 538–541. [Google Scholar] [CrossRef]
- Vitetta, L.; Kenner, D.; Sali, A. Bacterial infections in terminally ill hospice patients. J. Pain Symptom Manag. 2000, 20, 326–334. [Google Scholar] [CrossRef]
- Reinbolt, R.E.; Shenk, A.M.; White, P.H.; Navari, R.M. Symptomatic treatment of infections in patients with advanced cancer receiving hospice care. J. Pain Symptom Manag. 2005, 30, 175–182. [Google Scholar] [CrossRef]
- Mirhosseini, M.; Oneschuk, D.; Hunter, B.; Hanson, J.; Quan, H.; Amigo, P. The role of antibiotics in the management of infection-related symptoms in advanced cancer patients. J. Palliat. Care 2006, 22, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, J.H.; Albrecht, J.S.; Fromme, E.K.; Noble, B.N.; McGregor, J.C.; Comer, A.C.; Furuno, J.P. Antimicrobial use for symptom management in patients receiving hospice and palliative care: A systematic review. J. Palliat. Med. 2013, 16, 1658–1674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seow, H.; Barbera, L.; Sutradhar, R.; Howell, D.; Dudgeon, D.; Atzema, C.; Liu, Y.; Husain, A.; Sussman, J.; Earle, C. Trajectory of performance status and symptom scores for patients with cancer during the last six months of life. J. Clin. Oncol. 2011, 29, 1151–1158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamma, P.D.; Avdic, E.; Li, D.X.; Dzintars, K.; Cosgrove, S.E. Association of Adverse Events With Antibiotic Use in Hospitalized Patients. JAMA Intern. Med. 2017, 177, 1308–1315. [Google Scholar] [CrossRef] [Green Version]
- Davey, P.; Marwick, C.A.; Scott, C.L.; Charani, E.; McNeil, K.; Brown, E.; Ramsay, C.R.; Wiffen, P.J.; Wilcox, M. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst. Rev. 2017, 2. [Google Scholar] [CrossRef] [Green Version]
- Berwick, D.M. Avoiding overuse-the next quality frontier. Lancet 2017, 390, 102–104. [Google Scholar] [CrossRef]
- Thronæs, M.; Løhre, E.T.; Kvikstad, A.; Brenne, E.; Norvaag, R.; Aalberg, K.O.; Moen, M.K.; Jakobsen, G.; Klepstad, P.; Solberg, A. Interventions a3nd symptom relief in hospital palliative cancer care: Results from a prospective longitudinal study. Support Care Cancer 2021, 29, 6595–6603. [Google Scholar] [CrossRef]
- Hui, D.; Bruera, E. The Edmonton Symptom Assessment System 25 Years Later: Past, Present, and Future Developments. J. Pain Symptom Manag. 2017, 53, 630–643. [Google Scholar] [CrossRef] [Green Version]
- Hannon, B.; Dyck, M.; Pope, A.; Swami, N.; Banerjee, S.; Mak, E.; Bryson, J.; Rodin, G.; Ridley, J.; Lo, C. Modified Edmonton Symptom Assessment System including constipation and sleep: Validation in outpatients with cancer. J. Pain Symptom Manag. 2015, 49, 945–952. [Google Scholar] [CrossRef]
- Lohre, E.T.; Thronaes, M.; Brunelli, C.; Kaasa, S.; Klepstad, P. An in-hospital clinical care pathway with integrated decision support for cancer pain management reduced pain intensity and needs for hospital stay. Support Care Cancer 2019, 28, 671–682. [Google Scholar] [CrossRef]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P.M. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Physicians, R.C. National Early Warning Score (NEWS): Standardising the Assessment of Acute-Illness Severity in the NHS; Report of a Working Party; RCP: London, UK, 2012. [Google Scholar]
- Cheng, H.G.; Phillips, M.R. Secondary analysis of existing data: Opportunities and implementation. Shanghai Arch. Psychiatry 2014, 26, 371–375. [Google Scholar] [PubMed]
- Talari, K.; Goyal, M. Retrospective studies-utility and caveats. J. R. Coll. Physicians Edinb. 2020, 50, 398–402. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Moldawer, L.L. Parallels between cancer and infectious disease. N. Engl. J. Med. 2014, 371, 380–383. [Google Scholar] [CrossRef] [Green Version]
- Temel, J.S.; Greer, J.A.; Muzikansky, A.; Gallagher, E.R.; Admane, S.; Jackson, V.A. Early palliative care for patients with metastatic non-small-cell lung cancer. N. Engl. J. Med. 2010, 363, 733–742. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Cancer Pain Relief and Palliative Care-Report of a WHO Expert Committee; World Health Organization: Geneva, Switzerland, 1990. [Google Scholar]
- Dagli, O.; Tasdemir, E.; Ulutasdemir, N. Palliative care infections and antibiotic cost: A vicious circle. Aging Male 2020, 23, 98–105. [Google Scholar] [CrossRef]
- Diakos, C.I.; Charles, K.A.; McMillan, D.C.; Clarke, S.J. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014, 15, e493–e503. [Google Scholar] [CrossRef]
- Evans, S.S.; Repasky, E.A.; Fisher, D.T. Fever and the thermal regulation of immunity: The immune system feels the heat. Nat. Rev. Immunol. 2015, 15, 335–349. [Google Scholar] [CrossRef]
- Cecconi, M.; Evans, L.; Levy, M.; Rhodes, A. Sepsis and septic shock. Lancet 2018, 392, 75–87. [Google Scholar] [CrossRef]
- Thompson, A.J.; Silveira, M.J.; Vitale, C.A.; Malani, P.N. Antimicrobial use at the end of life among hospitalized patients with advanced cancer. Am. J. Hosp. Palliat. Care 2012, 29, 599–603. [Google Scholar] [CrossRef]
- Vento, S.; Cainelli, F. Infections in patients with cancer undergoing chemotherapy: Aetiology, prevention, and treatment. Lancet Oncol. 2003, 4, 595–604. [Google Scholar] [CrossRef]
- Hjelholt, T.J.; Rasmussen, T.B.; Seesaghur, A.; Hernandez, R.K.; Marongiu, A.; Obel, N.; Sørensen, H.T.; Pedersen, A.B. Risk of infections and mortality in Danish patients with cancer diagnosed with bone metastases: A population-based cohort study. BMJ Open 2021, 11, 049831. [Google Scholar] [CrossRef]
- Good, P.D.; Cavenagh, J.; Ravenscroft, P.J. Survival after enrollment in an Australian palliative care program. J. Pain Symptom Manag. 2004, 27, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Thatcher, N.; Girling, D.J.; Hopwood, P.; Sambrook, R.J.; Qian, W.; Stephens, R.J. Improving survival without reducing quality of life in small-cell lung cancer patients by increasing the dose-intensity of chemotherapy with granulocyte colony-stimulating factor support: Results of a British Medical Research Council Multicenter Randomized Trial. Medical Research Council Lung Cancer Working Party. J. Clin. Oncol. 2000, 18, 395–404. [Google Scholar] [PubMed]
- Murad, M.H.; Asi, N.; Alsawas, M.; Alahdab, F. New evidence pyramid. Evid. Based Med. 2016, 21, 125–127. [Google Scholar] [CrossRef] [Green Version]
| Characteristic | Infection Group (n = 62) | Non-Infection Group (n =157) | |
|---|---|---|---|
| Age, mean years (range) | 65.8 (29–94) | 70.2 (30–98) | |
| Gender, % male | 69 | 56 | |
| Cancer type, n (%) | Gastrointestinal (GI) | 27 (44) | 86 (55) |
| Urological | 23 (37) | 26 (17) | |
| Breast | 3 (5) | 17 (11) | |
| Lung | 0 | 1 (1) | |
| Head-neck | 2 (3) | 13 (8) | |
| Other | 7 (11) | 14 (8) | |
| Metastasis, n (%) | Yes | 55 (89) | 113 (72) |
| No | 7 (11) | 14 (9) | |
| Comorbidities, n (%) | Cardiovascular disease | 21 (34) | 22 (14) |
| Diabetes | 6 (10) | 20 (13) | |
| Kidney disease | 0 | 4 (3) | |
| Musculoskeletal disease | 5 (8) | 17 (11) | |
| Psychiatric disorder | 3 (5) | 22 (14) | |
| COPD 1 | 4 (7) | 7 (5) | |
| Liver disease | 1 (2) | 0 | |
| Other | 14 (22) | 31 (19) | |
| ECOG 2 performance status score, n (%) | 0 | 0 | 2 (1) |
| 1–2 | 26 (42) | 63 (40) | |
| 3–4 | 35 (57) | 91 (58) | |
| Missing | 1 (2) | 1 (1) | |
| Trajectory, n (%) | Palliative cancer care | 37 (60) | 99 (63) |
| Integrated cancer care | 24 (39) | 57(36) | |
| Information not available | 1 (2) | 1 (1) | |
| Radiology, n (%) | X-ray | 37 (60) | 41 (26) |
| Computer tomography | 28 (45) | 55 (35) | |
| Ultrasound | 7 (11) | 15 (10) | |
| Magnetic resonance imaging | 4 (7) | 27 (17) | |
| No | 10 (16) | 57 (36) | |
| Interventions n (%) | Radiological intervention 3 | 5 (8) | 13 (8) |
| Rehydration | 43 (69) | 65 (41) | |
| Nutrition (iv or nasogastric feeding tube) | 10 (16) | 15 (10) | |
| Transfusion | 20 (32) | 15 (10) | |
| Surgery | 1(2) | 3 (2) | |
| Use of multidisiplinary team | 24 (39) | 52 (33) | |
| Mean hospital length of stay, mean days (range) | 8.2 (2–29) | 6.8 (0–39) | |
| Survival after admission, median days (95% CI 4) | 58.5 (41–86) | 36 (28–44) | |
| In-hospital mortality n (%) | 8 (12.9) | 22 (14.0) | |
| Characteristic | Infection Group, n = 62 | |
|---|---|---|
| Source of infection, n (%) | Lungs | 32 (52) |
| Urinary | 10 (16) | |
| GI | 6 (10) | |
| Skin or soft tissue | 1 (2) | |
| Other (se under) | 1 (2) | |
| Unknown | 20 (32) | |
| More than one focus | 8 (13) | |
| Blood culture, n (%) | Positive | 6 (10) |
| Gram-negative | 4 (7) | |
| Gram-positive | 1 (2) | |
| Poly-microbial or fungal BSI | 1 (2) | |
| Negative | 48 (77) | |
| Not taken | 8 (13) | |
| Urine culture, n (%) | Positive | 12 (20) |
| Negative | 32 (52) | |
| Not taken | 18 (29) | |
| Other cultures, n (%) | Positive | 2 (3) |
| Negative | 1 (1) | |
| Not taken | 59 (95) | |
| Antibiotics |
| 13 (21) |
| 13 (21) | |
| 13 (21) | |
| 9 (15) | |
| 3 (5) | |
| 9 (15) | |
| 1 (2) | |
| 1 (2) | |
| Antibiotic change, n (%) | No | 41 (66) |
| Yes | 21 (34) | |
| Due to resistance | 6 (10) | |
| Therapy failure | 7 (11) | |
| De-escalation | 1 (2) | |
| Uncertain | 6 (10) | |
| Antibiotic appropriateness after resistance, n (%) | Yes | 14 (22) |
| No | 4 (7) | |
| Negative blood culture | 38 (61) | |
| No blood culture | 6 (10) | |
| Antibiotics after change |
| 2 (3) |
| 1 (2) | |
| 4 (7) | |
| 3(5) | |
| 2 (3) | |
| 2 (3) | |
| 4 (7) | |
| 1 (2) | |
| 2 (3) | |
| Days of intravenous treatment, mean (range) | 7.15 (1–20) |
| Infection Group, n = 62. Mean | Non-Infection Group, n = 157. Mean | p-Value 1 | ||
|---|---|---|---|---|
| Systolic blood pressure, mmHg | 120 | 126 | 0.070 | |
| Physiology | Heart rate, bpm 2 | 92 | 84 | 0.004 |
| Temperature, °C | 37.3 | 36.7 | <0.001 | |
| Total NEWS score | 3.47 | 1.79 | <0.001 | |
| Paraclinical | C-reactive protein, mg/L | 144.1 | 50.5 | <0.001 |
| Leucocytes, ×109/L | 10.2 | 10.6 | 0.688 | |
| Neutrophils ×109/L | 8.67 | 8.48 | 0.839 | |
| Symptoms at admission, NRS | Average pain | 4.12 | 4.09 | 0.959 |
| Worst pain | 5.40 | 5.40 | 0.997 | |
| Tiredness | 5.60 | 5.80 | 0.626 | |
| Drowsiness | 5.65 | 5.32 | 0.430 | |
| Nausea | 1.98 | 2.71 | 0.115 | |
| Appetite | 4.62 | 4.98 | 0.515 | |
| Shortness of breath | 3.69 | 3.41 | 0.585 | |
| Depression | 3.38 | 3.64 | 0.611 | |
| Anxiety | 2.80 | 2.97 | 0.719 | |
| Well-being | 4.61 | 4.81 | 0.601 | |
| Sleep | 5.02 | 3.91 | 0.017 | |
| Constipation | 2.96 | 3.40 | 0.455 | |
| Difference 1 | Infection Group, n = 62. Mean | Non-Infection Group, n = 157. Mean | p-Value 2 | |
|---|---|---|---|---|
| Physiology | Diff systolic blood pressure | −15.06 | −2.56 | 0.001 |
| Diff heart rate | 9.51 | 0.43 | 0.005 | |
| Diff temperature | 0.72 | 0.04 | <0.001 | |
| Diff total NEWS score | 1.60 | −0.05 | <0.001 | |
| Diff C-reactive protein | 66.4 | −0.7 | <0.001 | |
| Paraclinical | Diff leucocytes | 1.48 | 0.08 | 0.051 |
| Diff neutrophils | 1.59 | 0.32 | 0.057 | |
| Symptoms | Diff average pain. | 1.04 | 1.40 | 0.440 |
| Diff worst pain. | 1.98 | 1.40 | 0.302 | |
| Diff tiredness | 1.26 | 1.45 | 0.667 | |
| Diff drowsiness | 1.36 | 1.17 | 0.670 | |
| Diff nausea | 0.57 | 0.92 | 0.409 | |
| Diff appetite | 0.82 | 1.37 | 0.309 | |
| Diff shortness of breath | 0.85 | 1.06 | 0.605 | |
| Diff depression | 0.49 | 0.59 | 0.793 | |
| Diff anxiety | 0.24 | 0.71 | 0.236 | |
| Diff well-being | 0.49 | 1.05 | 0.232 | |
| Diff sleep | 1.09 | 0.63 | 0.378 | |
| Diff constipation | 1.34 | 1.25 | 0.870 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moen, M.K.; Løhre, E.T.; Jakobsen, G.; Thronæs, M.; Klepstad, P. Antibiotic Therapy in Integrated Oncology and Palliative Cancer Care: An Observational Study. Cancers 2022, 14, 1602. https://doi.org/10.3390/cancers14071602
Moen MK, Løhre ET, Jakobsen G, Thronæs M, Klepstad P. Antibiotic Therapy in Integrated Oncology and Palliative Cancer Care: An Observational Study. Cancers. 2022; 14(7):1602. https://doi.org/10.3390/cancers14071602
Chicago/Turabian StyleMoen, Martine Kjølberg, Erik Torbjørn Løhre, Gunnhild Jakobsen, Morten Thronæs, and Pål Klepstad. 2022. "Antibiotic Therapy in Integrated Oncology and Palliative Cancer Care: An Observational Study" Cancers 14, no. 7: 1602. https://doi.org/10.3390/cancers14071602
APA StyleMoen, M. K., Løhre, E. T., Jakobsen, G., Thronæs, M., & Klepstad, P. (2022). Antibiotic Therapy in Integrated Oncology and Palliative Cancer Care: An Observational Study. Cancers, 14(7), 1602. https://doi.org/10.3390/cancers14071602





