Risk of Gynecological Cancers in Cholecystectomized Women: A Large Nationwide Cohort Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Datasets
2.2. Follow-Up
2.3. Statistical Analyses
3. Results
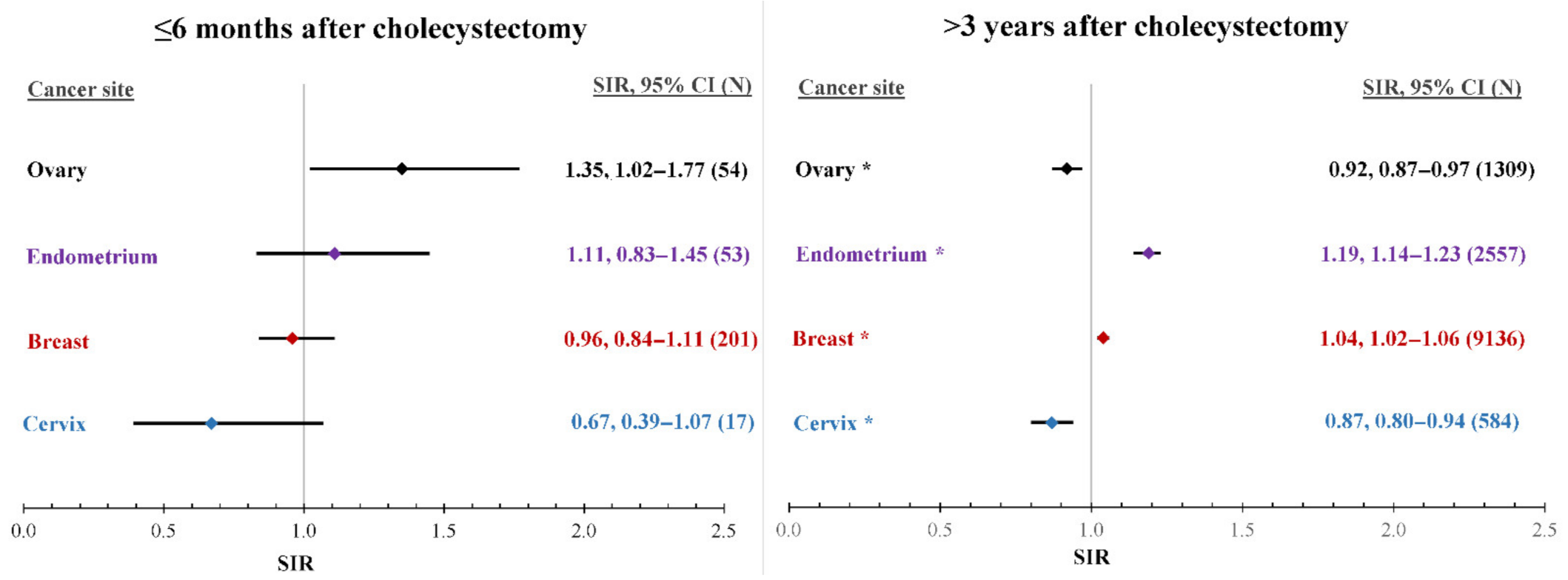
3.1. SIRs by Time after Cholecystectomy
3.2. SIRs by Age at Cholecystectomy
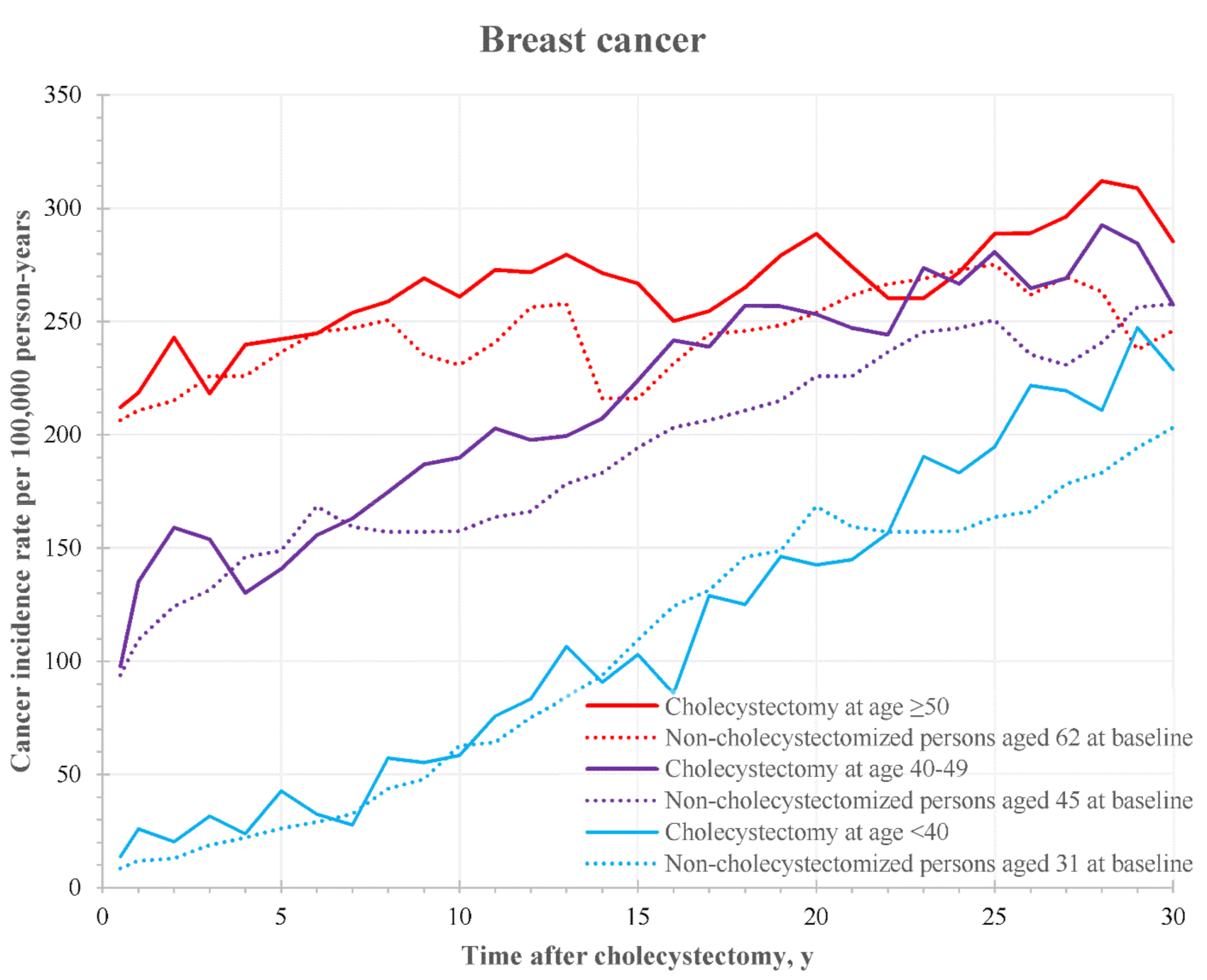
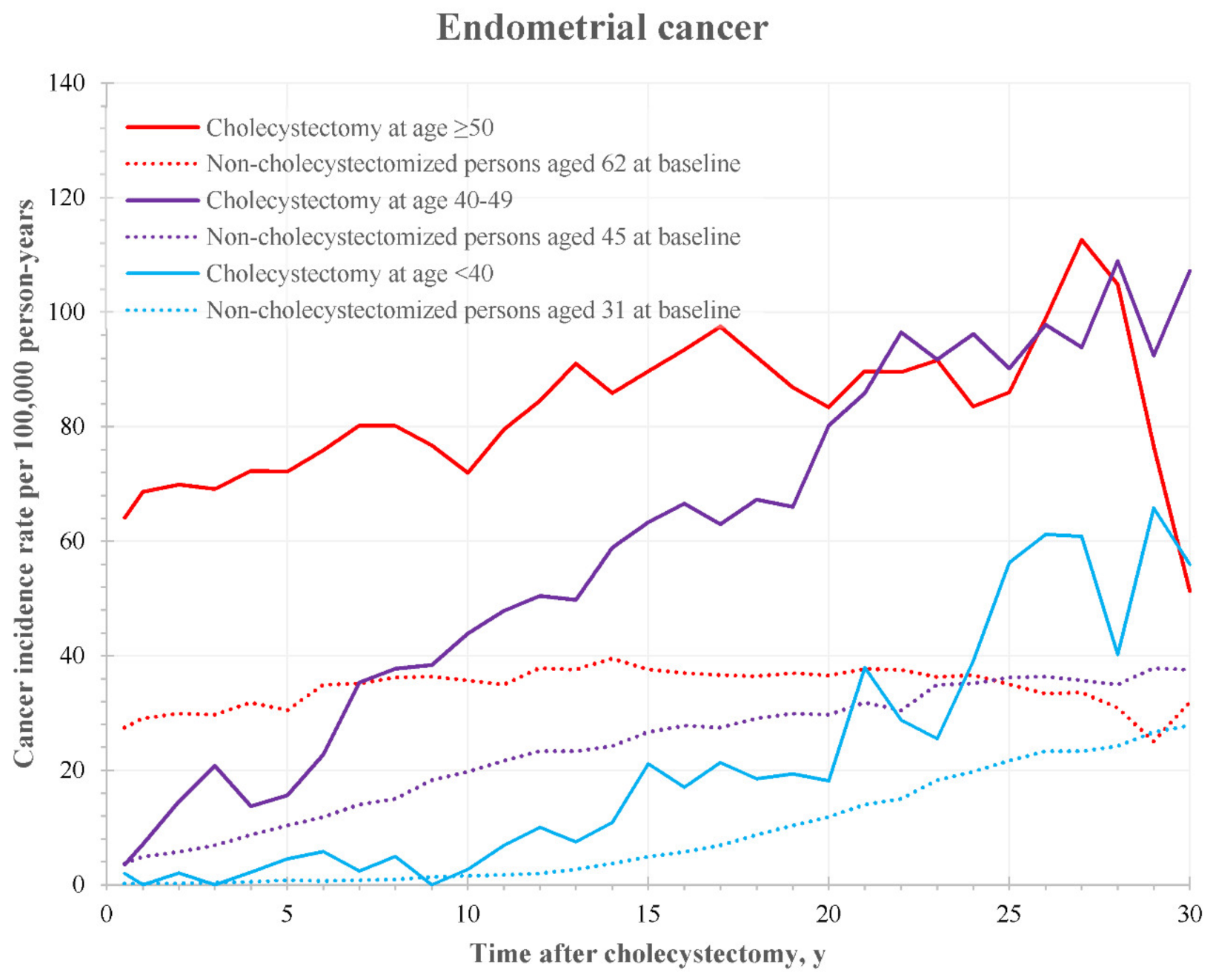
3.3. Age-Specific Incidence Curves
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Townsend, C.; Evers, B.; Mattox, K.; Beauchamp, R. Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice; Wb Saunders: Philadelphia, PA, USA, 2016. [Google Scholar]
- Schirmer, B.D.; Winters, K.L.; Edlich, R.F. Cholelithiasis and Cholecystitis. J. Long-Term Eff. Med. Implant. 2005, 15, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Völzke, H.; Baumeister, S.E.; Alte, D.; Hoffmann, W.; Schwahn, C.; Simon, P.; John, U.; Lerch, M.M. Independent Risk Factors for Gallstone Formation in a Region with High Cholelithiasis Prevalence. Digestion 2005, 71, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, T. Prevalence of gallstones in a Danish population. Am. J. Epidemiol. 1987, 126, 912–921. [Google Scholar] [CrossRef]
- Valdivieso, V.; Covarrubias, C.; Siegel, F.; Cruz, F. Pregnancy and cholelithiasis: Pathogenesis and natural course of gallstones diagnosed in early puerperium. Hepatology 1993, 17, 1–4. [Google Scholar] [PubMed]
- Scragg, R.K.; McMichael, A.J.; Seamark, R.F. Oral contraceptives, pregnancy, and endogenous oestrogen in gall stone disease—A case-control study. Br. Med. J. 1984, 288, 1795–1799. [Google Scholar] [CrossRef] [PubMed]
- Uhler, M.L.; Marks, J.W.; Judd, H.L. Estrogen replacement therapy and gallbladder disease in postmenopausal women. Menopause 2000, 7, 162–167. [Google Scholar] [CrossRef]
- Hulley, S.; Grady, D.; Bush, T.; Furberg, C.; Herrington, D.; Riggs, B.; Vittinghoff, E.; Heart and Estrogen/progestin Replacement Study (HERS) Research Group. Randomized Trial of Estrogen Plus Progestin for Secondary Prevention of Coronary Heart Disease in Postmenopausal Women. JAMA J. Am. Med. Assoc. 1998, 280, 605–613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cirillo, D.; Wallace, R.B.; Rodabough, R.J.; Greenland, P.; Lacroix, A.Z.; Limacher, M.C.; Larson, J.C. Effect of Estrogen Therapy on Gallbladder Disease. JAMA J. Am. Med. Assoc. 2005, 293, 330–339. [Google Scholar] [CrossRef]
- Stampfer, M.J.; Maclure, K.M.; Colditz, G.; Manson, J.E.; Willett, W.C. Risk of symptomatic gallstones in women with severe obesity. Am. J. Clin. Nutr. 1992, 55, 652–658. [Google Scholar] [CrossRef] [Green Version]
- Attili, A.F.; Capocaccia, R.; Carulli, N.; Festi, D.; Roda, E.; Barbara, L.; Capocaccia, L.; Menotti, A.; Okolicsanyi, L.; Ricci, G.; et al. Factors associated with gallstone disease in the MICOL experience. Multicenter Italian Study on Epidemiology of Cholelithiasis. Hepatology 1997, 26, 809–818. [Google Scholar] [CrossRef]
- Shiffman, M.L.; Sugerman, H.J.; Kellum, J.M.; Brewer, W.H.; Moore, E.W. Gallstone formation after rapid weight loss: A prospective study in patients undergoing gastric bypass surgery for treatment of morbid obesity. Am. J. Gastroenterol. 1991, 86, 1000–1005. [Google Scholar]
- Brown, S.B.; Hankinson, S.E. Endogenous estrogens and the risk of breast, endometrial, and ovarian cancers. Steroids 2015, 99, 8–10. [Google Scholar] [CrossRef]
- Pfeiffer, R.M.; Park, Y.; Kreimer, A.R.; Lacey, J.V., Jr.; Pee, D.; Greenlee, R.T.; Buys, S.S.; Hollenbeck, A.; Rosner, B.; Gail, M.H.; et al. Risk Prediction for Breast, Endometrial, and Ovarian Cancer in White Women Aged 50 y or Older: Derivation and Validation from Population-Based Cohort Studies. PLoS Med. 2013, 10, e1001492. [Google Scholar] [CrossRef]
- Hannaford, P.C.; Selvaraj, S.; Elliott, A.M.; Angus, V.; Iversen, L.; Lee, A.J. Cancer risk among users of oral contraceptives: Cohort data from the Royal College of General Practitioner’s oral contraception study. BMJ 2007, 335, 651. [Google Scholar] [CrossRef] [Green Version]
- Vallée, J.; Cadot, E.; Grillo, F.; Parizot, I.; Chauvin, P. The combined effects of activity space and neighbourhood of residence on participation in preventive health-care activities: The case of cervical screening in the Paris metropolitan area (France). Health Place 2010, 16, 838–852. [Google Scholar] [CrossRef] [Green Version]
- Grillo, F.; Vallée, J.; Chauvin, P. Inequalities in cervical cancer screening for women with or without a regular consulting in primary care for gynaecological health, in Paris, France. Prev. Med. 2012, 54, 259–265. [Google Scholar] [CrossRef]
- Mali, N.R.; Mali, R. Effect of Structured Education on Knowledge Regarding Prevention of Cervical Cancer among A.N.M. Students. Original research. IJSR Int. J. Sci. Res. 2014, 3, 17. [Google Scholar]
- Bosch, F.X.; Muñoz, N.; José, F.X.B.; Izarzugaza, I.; Gili, M.; Viladiu, P.; Tormo, M.J.; Moreo, P.; Ascunce, N.; Gonzalez, L.C.; et al. Risk factors for cervical cancer in Colombia and Spain. Int. J. Cancer 1992, 52, 750–758. [Google Scholar] [CrossRef]
- Barlow, L.; Westergren, K.; Holmberg, L.; Talbäck, M. The completeness of the Swedish Cancer Register—A sample survey for year 1998. Acta Oncol. 2009, 48, 27–33. [Google Scholar] [CrossRef]
- Drasar, B.S.; Irving, D. Environmental Factors and Cancer of the Colon and Breast. Br. J. Cancer 1973, 27, 167–172. [Google Scholar] [CrossRef] [Green Version]
- MacMahon, B.; Cole, P.; Brown, J. Etiology of Human Breast Cancer: A Review. JNCI J. Natl. Cancer Inst. 1973, 50, 21–42. [Google Scholar] [CrossRef]
- SShabanzadeh, D.M.; Sørensen, L.T.; Jørgensen, T. Association Between Screen-Detected Gallstone Disease and Cancer in a Cohort Study. Gastroenterology 2017, 152, 1965–1974.e1. [Google Scholar] [CrossRef]
- Wang, H.H.; Liu, M.; Clegg, D.J.; Portincasa, P.; Wang, D.Q.-H. New insights into the molecular mechanisms underlying effects of estrogen on cholesterol gallstone formation. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2009, 1791, 1037–1047. [Google Scholar] [CrossRef] [Green Version]
- Matsuo, K.; Chen, L.; Matsuzaki, S.; Mandelbaum, R.S.; Ciesielski, K.M.; Silva, J.P.; Klar, M.; Roman, L.D.; Accordino, M.K.; Melamed, A.; et al. Opportunistic Salpingectomy at the Time of Laparoscopic Cholecystectomy for Ovarian Cancer Prevention. Ann. Surg. 2022, in press. [Google Scholar] [CrossRef]
- Dutta, U.; Bush, N.; Kalsi, D.; Popli, P.; Kapoor, V.K. Epidemiology of gallbladder cancer in India. Chin. Clin. Oncol. 2019, 8, 33. [Google Scholar] [CrossRef]
- Rawla, P.; Sunkara, T.; Thandra, K.C.; Barsouk, A. Epidemiology of gallbladder cancer. Clin. Exp. Hepatol. 2019, 5, 93–102. [Google Scholar] [CrossRef]
- Giovannucci, E.; Colditz, G.; Stampfer, M.J. A meta-analysis of cholecystectomy and risk of colorectal cancer. Gastroenterology 1993, 105, 130–141. [Google Scholar] [CrossRef]
- Ren, X.; Xu, J.; Zhang, Y.; Chen, G.; Zhang, Y.; Huang, Q.; Liu, Y. Bacterial Alterations in Post-Cholecystectomy Patients Are Associated With Colorectal Cancer. Front. Oncol. 2020, 10, 1418. [Google Scholar] [CrossRef]
- Wang, W.; Wang, J.; Li, J.; Yan, P.; Jin, Y.; Zhang, R.; Yue, W.; Guo, Q.; Geng, J. Cholecystectomy Damages Aging-Associated Intestinal Microbiota Construction. Front. Microbiol. 2018, 9, 1402. [Google Scholar] [CrossRef] [PubMed]
- Chiba, T.; Marusawa, H.; Ushijima, T. Inflammation-Associated Cancer Development in Digestive Organs: Mechanisms and Roles for Genetic and Epigenetic Modulation. Gastroenterology 2012, 143, 550–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nogueira, L.; Freedman, N.D.; Engels, E.A.; Warren, J.L.; Castro, F.; Koshiol, J. Gallstones, Cholecystectomy, and Risk of Digestive System Cancers. Am. J. Epidemiol. 2014, 179, 731–739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lagergren, J.; Mattsson, F.; El-Serag, H.; Nordenstedt, H. Increased risk of hepatocellular carcinoma after cholecystectomy. Br. J. Cancer 2011, 105, 154–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, T.; Yang, Y.-X. Cholecystectomy and the Risk of Colorectal Cancer. Am. J. Gastroenterol. 2005, 100, 1813–1820. [Google Scholar] [CrossRef]
- Lagergren, J.; Ye, W.; Ekbom, A. Intestinal cancer after cholecystectomy: Is bile involved in carcinogenesis? Gastroenterology 2001, 121, 542–547. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, H.; Li, L.; Ai, M.; Gong, Z.; He, Y.; Dong, Y.; Xu, S.; Wang, J.; Jin, B.; et al. Cholecystectomy can increase the risk of colorectal cancer: A meta-analysis of 10 cohort studies. PLoS ONE 2017, 12, e0181852. [Google Scholar] [CrossRef] [Green Version]
- Torp, N.M.U.; Kristensen, S.B.; Mortensen, F.V.; Kirkegård, J. Cholecystitis and risk of pancreatic, liver, and biliary tract cancer in patients undergoing cholecystectomy. HPB 2020, 22, 1258–1264. [Google Scholar] [CrossRef]
- Bernstein, H.; Bernstein, C.; Payne, C.M.; Dvorakova, K.; Garewal, H. Bile acids as carcinogens in human gastrointestinal cancers. Mutat. Res. Mutat. Res. 2005, 589, 47–65. [Google Scholar] [CrossRef]
- Michels, K.B.; Hausen, H.Z. HPV vaccine for all. Lancet 2009, 374, 268–270. [Google Scholar] [CrossRef]
| Time after Cholecystectomy | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1–6 Months | 7–12 Months | 13–36 Months | >3 Years | All | |||||||||||||||||
| Cancer | N | SIR | 95% CI | N | SIR | 95% CI | N | SIR | 95% CI | N | SIR | 95% CI | N | SIR | 95% CI | ICD-7 | |||||
| Breast | 201 | 0.96 | 0.84 | 1.11 | 217 | 1.05 | 0.92 | 1.20 | 877 | 1.08 | 1.01 | 1.15 | 9136 | 1.04 * | 1.02 | 1.06 | 10,431 | 1.04 * | 1.02 | 1.06 | 170 |
| Cervix | 17 | 0.67 | 0.39 | 1.07 | 16 | 0.65 | 0.37 | 1.05 | 88 | 0.94 | 0.75 | 1.15 | 584 | 0.87 * | 0.80 | 0.94 | 705 | 0.86 * | 0.80 | 0.93 | 171 |
| Endometrium | 53 | 1.11 | 0.83 | 1.45 | 55 | 1.16 | 0.88 | 1.51 | 223 | 1.19 * | 1.04 | 1.36 | 2557 | 1.19 * | 1.14 | 1.23 | 2888 | 1.18 * | 1.14 | 1.23 | 172 |
| Ovary | 54 | 1.35 | 1.02 | 1.77 | 45 | 1.14 | 0.84 | 1.53 | 169 | 1.10 | 0.94 | 1.28 | 1309 | 0.92 * | 0.87 | 0.97 | 1577 | 0.95 | 0.91 | 1.00 | 175 |
| Age at Cholecystectomy (Excluding Cancers Diagnosed in the First 6 Months after Cholecystectomy) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| <40 Years | 40–49 Years | ≥50 Years | All | |||||||||||||
| Cancer | N | SIR | 95% CI | N | SIR | 95% CI | N | SIR | 95% CI | N | SIR | 95% CI | ||||
| Breast | 2583 | 1.00 | 0.96 | 1.04 | 2184 | 1.05 | 1.01 | 1.09 | 5463 | 1.07 * | 1.04 | 1.10 | 10,230 | 1.05 * | 1.03 | 1.07 |
| Cervix | 294 | 1.00 | 0.89 | 1.12 | 125 | 0.82 | 0.68 | 0.98 | 269 | 0.78 * | 0.69 | 0.87 | 688 | 0.87 * | 0.80 | 0.93 |
| Endometrium | 527 | 1.18 * | 1.08 | 1.28 | 577 | 1.15 * | 1.06 | 1.25 | 1731 | 1.20 * | 1.14 | 1.26 | 2835 | 1.19 * | 1.14 | 1.23 |
| Ovary | 362 | 0.99 | 0.89 | 1.10 | 306 | 0.89 | 0.79 | 1.00 | 855 | 0.94 | 0.88 | 1.01 | 1523 | 0.94 * | 0.90 | 0.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kharazmi, E.; Sundquist, K.; Sundquist, J.; Fallah, M.; Bermejo, J.L. Risk of Gynecological Cancers in Cholecystectomized Women: A Large Nationwide Cohort Study. Cancers 2022, 14, 1484. https://doi.org/10.3390/cancers14061484
Kharazmi E, Sundquist K, Sundquist J, Fallah M, Bermejo JL. Risk of Gynecological Cancers in Cholecystectomized Women: A Large Nationwide Cohort Study. Cancers. 2022; 14(6):1484. https://doi.org/10.3390/cancers14061484
Chicago/Turabian StyleKharazmi, Elham, Kristina Sundquist, Jan Sundquist, Mahdi Fallah, and Justo Lorenzo Bermejo. 2022. "Risk of Gynecological Cancers in Cholecystectomized Women: A Large Nationwide Cohort Study" Cancers 14, no. 6: 1484. https://doi.org/10.3390/cancers14061484
APA StyleKharazmi, E., Sundquist, K., Sundquist, J., Fallah, M., & Bermejo, J. L. (2022). Risk of Gynecological Cancers in Cholecystectomized Women: A Large Nationwide Cohort Study. Cancers, 14(6), 1484. https://doi.org/10.3390/cancers14061484







