Emerging Canonical and Non-Canonical Roles of Granzyme B in Health and Disease
Abstract
Simple Summary
Abstract
1. Introduction
2. Canonical Roles of Granzyme B
2.1. Granzyme B Regulation in CTL and NK Cells
2.2. Regulating Self-Inflicted Damage
2.3. Transcriptional Regulation
2.4. Translational Regulation
3. Non-Lymphocytic Granzyme B Production
3.1. Mast Cells
3.2. Myeloid Derived Suppressor Cells
3.3. Signaling Mechanisms for Granzyme B Production in Non-Lymphocytic Cells
4. Non-Canonical Roles of Granzyme B
4.1. Role of Granzyme B in Inhibiting Infectious Diseases
4.2. Cytotoxicity towards Other Immune Cells
4.3. Cell Type Differentiation
4.4. Granzyme B Expression in Cancer
4.5. Extracellular Matrix Remodeling
5. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cullen, S.P.; Martin, S.J. Mechanisms of granule-dependent killing. Cell Death Differ. 2008, 15, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Casciola-Rosen, L.; Garcia-Calvo, M.; Bull, H.G.; Becker, J.W.; Hines, T.; Thornberry, N.A.; Rosen, A. Mouse and human granzyme B have distinct tetrapeptide specificities and abilities to recruit the bid pathway. J. Biol. Chem. 2007, 282, 4545–4552. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, N.J.; Sedelies, K.A.; Browne, K.A.; Wowk, M.E.; Newbold, A.; Sutton, V.R.; Clarke, C.J.P.; Oliaro, J.; Lindemann, R.K.; Bird, P.I.; et al. A central role for Bid in granzyme B-induced apoptosis. J. Biol. Chem. 2005, 280, 4476–4482. [Google Scholar] [CrossRef] [PubMed]
- Jans, D.A.; Jans, P.; Briggs, L.J.; Sutton, V.; Trapani, J.A. Nuclear transport of granzyme B (fragmentin-2). Dependence of perforin in vivo and cytosolic factors in vitro. J. Biol. Chem. 1996, 271, 30781–30789. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Beresford, P.J.; Greenberg, A.H.; Lieberman, J. Granzymes A and B directly cleave lamins and disrupt the nuclear lamina during granule-mediated cytolysis. Proc. Natl. Acad. Sci. USA 2001, 98, 5746–5751. [Google Scholar] [CrossRef] [PubMed]
- Chaitanya, G.V.; Babu, P.P. Differential PARP Cleavage: An Indication of Heterogeneous Forms of Cell Death and Involvement of Multiple Proteases in the Infarct of Focal Cerebral Ischemia in Rat. Cell. Mol. Neurobiol. 2009, 29, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; He, H.; Wang, K.; Shi, X.; Wang, Y.; Su, Y.; Wang, Y.; Li, D.; Liu, W.; Zhang, Y.; et al. Granzyme A from cytotoxic lymphocytes cleaves GSDMB to trigger pyroptosis in target cells. Science 2020, 368, eaaz7548. [Google Scholar] [CrossRef] [PubMed]
- Jans, D.A.; Briggs, L.J.; Jans, P.; Froelich, C.J.; Parasivam, G.; Kumar, S.; Sutton, V.R.; Trapani, J.A. Nuclear targeting of the serine protease granzyme A (fragmentin-1). J. Cell Sci. 1998, 111 Pt 17, 2645–2654. [Google Scholar] [CrossRef] [PubMed]
- Martinvalet, D.; Zhu, P.; Lieberman, J. Granzyme A induces caspase-independent mitochondrial damage, a required first step for apoptosis. Immunity 2005, 22, 355–370. [Google Scholar] [CrossRef] [PubMed]
- Beresford, P.J.; Jaju, M.; Friedman, R.S.; Yoon, M.J.; Lieberman, J. A role for heat shock protein 27 in CTL-mediated cell death. J. Immunol. 1998, 161, 161–167. [Google Scholar] [PubMed]
- Pardo, J.; Bosque, A.; Brehm, R.; Wallich, R.; Naval, J.; Müllbacher, A.; Anel, A.; Simon, M.M. Apoptotic pathways are selectively activated by granzyme A and/or granzyme B in CTL-mediated target cell lysis. J. Cell Biol. 2004, 167, 457–468. [Google Scholar] [CrossRef]
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. T cells and MHC proteins. In Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. Available online: https://www.ncbi.nlm.nih.gov/books/NBK26926/ (accessed on 30 July 2021).
- Stinchcombe, J.C.; Bossi, G.; Booth, S.; Griffiths, G.M. The Immunological Synapse of CTL Contains a Secretory Domain and Membrane Bridges. Immunity 2001, 15, 751–761. [Google Scholar] [CrossRef]
- Smyth, M.J.; Thia, K.Y.T.; Cretney, E.; Kelly, J.M.; Snook, M.B.; Forbes, C.A.; Scalzo, A.A. Perforin Is a Major Contributor to NK Cell Control of Tumor Metastasis. J. Immunol. 1999, 162, 6658–6662. [Google Scholar]
- Smyth, M.J.; Hayakawa, Y.; Takeda, K.; Yagita, H. New aspects of natural-killer-cell surveillance and therapy of cancer. Nat. Rev. Cancer 2002, 2, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Yannelli, J.R.; Sullivan, J.A.; Mandell, G.L.; Engelhard, V.H. Reorientation and fusion of cytotoxic T lymphocyte granules after interaction with target cells as determined by high resolution cinemicrography. J. Immunol. 1986, 136, 377–382. [Google Scholar] [PubMed]
- Peters, P.J.; Geuze, H.J.; van der Donk, H.A.; Slot, J.W.; Griffith, J.M.; Stam, N.J.; Clevers, H.C.; Borst, J. Molecules relevant for T cell-target cell interaction are present in cytolytic granules of human T lymphocytes. Eur. J. Immunol. 1989, 19, 1469–1475. [Google Scholar] [CrossRef] [PubMed]
- Purbhoo, M.A.; Irvine, D.J.; Huppa, J.B.; Davis, M.M. T cell killing does not require the formation of a stable mature immunological synapse. Nat. Immunol. 2004, 5, 524–530. [Google Scholar] [CrossRef]
- Guan, J.; Miah, S.M.S.; Wilson, Z.S.; Erick, T.K.; Banh, C.; Brossay, L. Role of Type I Interferon Receptor Signaling on NK Cell Development and Functions. PLoS ONE 2004, 9, e111302. [Google Scholar] [CrossRef]
- Wong, J.L.; Muthuswamy, R.; Bartlett, D.L.; Kalinski, P. IL-18-based combinatorial adjuvants promote the intranodal production of CCL19 by NK cells and dendritic cells of cancer patients. Oncoimmunology 2013, 2, e26245. [Google Scholar] [CrossRef][Green Version]
- Farhadi, N.; Lambert, L.; Triulzi, C.; Openshaw, P.J.M.; Guerra, N.; Culley, F.J. Natural killer cell NKG2D and granzyme B are critical for allergic pulmonary inflammation. J. Allergy Clin. Immunol. 2014, 133, 827–835.e3. [Google Scholar] [CrossRef]
- Trapani, J.A.; Smyth, M.J. Functional significance of the perforin/granzyme cell death pathway. Nat. Rev. Immunol. 2002, 2, 735–747. [Google Scholar] [CrossRef]
- Shresta, S.; Pham, C.T.; Thomas, D.A.; Graubert, T.A.; Ley, T.J. How do cytotoxic lymphocytes kill their targets? Curr. Opin. Immunol. 1998, 10, 581–587. [Google Scholar] [CrossRef]
- Grossman, W.J.; Verbsky, J.W.; Tollefsen, B.L.; Kemper, C.; Atkinson, J.P.; Ley, T.J. Differential expression of granzymes A and B in human cytotoxic lymphocyte subsets and T regulatory cells. Blood 2004, 104, 2840–2848. [Google Scholar] [CrossRef] [PubMed]
- Kelso, A.; Costelloe, E.O.; Johnson, B.J.; Groves, P.; Buttigieg, K.; Fitzpatrick, D.R. The genes for perforin, granzymes A–C and IFN-γ are differentially expressed in single CD8+ T cells during primary activation. Int. Immunol. 2002, 14, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Dudimah, F.D.; Griffey, D.; Wang, X.; Whalen, M.M. Activation of p44/42 MAPK Plays a Role in the TBT-induced Loss of Human Natural Killer (NK) Cell Function. Cell Biol. Toxicol. 2010, 26, 435–444. [Google Scholar] [CrossRef][Green Version]
- Özdemir, Ö.; Savaşan, S. Combinational IL-2/IL-15 induction does not further enhance IL-15-induced lymphokine-activated killer cell cytotoxicity against human leukemia/lymphoma cells. Clin. Immunol. 2005, 115, 240–249. [Google Scholar] [CrossRef]
- Mishra, R.; Chen, A.T.; Welsh, R.M.; Szomolanyi-Tsuda, E. NK cells and gammadelta T cells mediate resistance to polyomavirus-induced tumors. PLoS Pathog. 2010, 6, e1000924. [Google Scholar] [CrossRef] [PubMed]
- Meresse, B.; Korneychuk, N.; Malamut, G.; Cerf-Bensussan, N. Interleukin-15, a master piece in the immunological jigsaw of celiac disease. Dig. Dis. 2015, 33, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.C.; Rafii, S.; Granelli-Piperno, A.; Trapani, J.A.; Young, J.D. Perforin and serine esterase gene expression in stimulated human T cells. Kinetics, mitogen requirements, and effects of cyclosporin A. J. Exp. Med. 1989, 170, 2105–2118. [Google Scholar] [CrossRef]
- Waldmann, T.A.; Dubois, S.; Tagaya, Y. Contrasting Roles of IL-2 and IL-15 in the Life and Death of Lymphocytes: Implications for Immunotherapy. Immunity 2001, 14, 105–110. [Google Scholar] [CrossRef]
- Johnston, J.A.; Bacon, C.M.; Finbloom, D.S.; Rees, R.C.; Kaplan, D.; Shibuya, K.; Ortaldo, J.R.; Gupta, S.; Chen, Y.Q.; Giri, J.D. Tyrosine phosphorylation and activation of STAT5, STAT3, and Janus kinases by interleukins 2 and 15. Proc. Natl. Acad. Sci. USA 1995, 92, 8705–8709. [Google Scholar] [CrossRef] [PubMed]
- Moretta, A.; Bottino, C.; Vitale, M.; Pende, D.; Biassoni, R.; Mingari, M.C.; Moretta, L. Receptors for hla class-i molecules in human natural killer cells. Annu. Rev. Immunol. 1996, 14, 619–648. [Google Scholar] [CrossRef] [PubMed]
- Lanier, L.L. NK cell receptors. Annu. Rev. Immunol. 1998, 16, 359–393. [Google Scholar] [CrossRef] [PubMed]
- Karreci, E.S.; Eskandari, S.K.; Dotiwala, F.; Routray, S.K.; Kurdi, A.T.; Assaker, J.P.; Luckyanchykov, P.; Mihali, A.B.; Maarouf, O.; Borges, T.J.; et al. Human regulatory T cells undergo self-inflicted damage via granzyme pathways upon activation. JCI Insight 2017, 2, 91599. [Google Scholar] [CrossRef] [PubMed]
- Silverman, G.A.; Bird, P.I.; Carrell, R.W.; Church, F.C.; Coughlin, P.B.; Gettins, P.G.W.; Irving, J.A.; Lomas, D.A.; Luke, C.J.; Moyer, R.W.; et al. The Serpins Are an Expanding Superfamily of Structurally Similar but Functionally Diverse Proteins. J. Biol. Chem. 2001, 276, 33293–33296. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Park, S.-M.; Wang, Y.; Shah, R.; Liu, N.; Murmann, A.E.; Wang, C.-R.; Peter, M.E.; Ashton-Rickardt, P.G. Serine Protease Inhibitor 6 Protects Cytotoxic T Cells from Self-Inflicted Injury by Ensuring the Integrity of Cytotoxic Granules. Immunity 2006, 24, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Azzi, J.; Skartsis, N.; Mounayar, M.; Magee, C.N.; Batal, I.; Ting, C.; Moore, R.; Riella, L.V.; Ohori, S.; Abdoli, R.; et al. Serine protease inhibitor-6 plays a critical role in protecting murine Granzyme B-producing regulatory T-cells. J. Immunol. 2013, 191, 2319–2327. [Google Scholar] [CrossRef] [PubMed]
- Ansari, A.W.; Temblay, J.N.; Alyahya, S.H.; Ashton-Rickardt, P.G. Serine protease inhibitor 6 protects iNKT cells from self-inflicted damage. J. Immunol. 2010, 185, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Frégeau, C.J.; Bleackley, R.C. Transcription of two cytotoxic cell protease genes is under the control of different regulatory elements. Nucleic Acids Res. 1991, 19, 5583–5590. [Google Scholar] [CrossRef] [PubMed]
- Haddad, P.; Wargnier, A.; Bourge, J.F.; Sasportes, M.; Paul, P. A promoter element of the human serine esterase granzyme B gene controls specific transcription in activated T cells. Eur. J. Immunol. 1993, 23, 625–629. [Google Scholar] [CrossRef]
- Babichuk, C.K.; Duggan, B.L.; Bleackley, R.C. In Vivo regulation of murine granzyme B gene transcription in activated primary T cells. J. Biol. Chem. 1996, 271, 16485–16493. [Google Scholar] [CrossRef] [PubMed]
- Wargnier, A.; Lafaurie, C.; Legros-Maïda, S.; Bourge, J.F.; Sigaux, F.; Sasportes, M.; Paul, P. Down-regulation of human granzyme B expression by glucocorticoids. Dexamethasone inhibits binding to the Ikaros and AP-1 regulatory elements of the granzyme B promoter. J. Biol. Chem. 1998, 273, 35326–35331. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Guilloty, F.; Pipkin, M.E.; Djuretic, I.M.; Levanon, D.; Lotem, J.; Lichtenheld, M.G.; Groner, Y.; Rao, A. Runx3 and T-box proteins cooperate to establish the transcriptional program of effector CTLs. J. Exp. Med. 2009, 206, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Babichuk, C.K.; Bleackley, R.C. Mutational analysis of the murine granzyme B gene promoter in primary T cells and a T cell clone. J. Biol. Chem. 1997, 272, 18564–18571. [Google Scholar] [CrossRef] [PubMed]
- de Bruijn, M.; Dzierzak, E. Runx transcription factors in the development and function of the definitive hematopoietic system. Blood 2017, 129, 2061–2069. [Google Scholar] [CrossRef] [PubMed]
- Boto, P.; Csuth, T.I.; Szatmari, I. RUNX3-Mediated Immune Cell Development and Maturation. Crit. Rev. Immunol. 2018, 38, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Fehniger, T.A.; Cai, S.F.; Cao, X.; Bredemeyer, A.J.; Presti, R.M.; French, A.R.; Ley, T.J. Acquisition of Murine NK Cell Cytotoxicity Requires the Translation of a Pre-existing Pool of Granzyme B and Perforin mRNAs. Immunity 2007, 26, 798–811. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.M.; Mahmoud, A.B.; Makrigiannis, A.P. Licensed and Unlicensed NK Cells: Differential Roles in Cancer and Viral Control. Front. Immunol. 2016, 7, 166. [Google Scholar] [CrossRef] [PubMed]
- Strik, M.C.M.; de Koning, P.J.A.; Kleijmeer, M.J.; Bladergroen, B.A.; Wolbink, A.M.; Griffith, J.M.; Wouters, D.; Fukuoka, Y.; Schwartz, L.B.; Hack, C.E.; et al. Human mast cells produce and release the cytotoxic lymphocyte associated protease granzyme B upon activation. Mol. Immunol. 2007, 44, 3462–3472. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, G. Granzymes A and B are targeted to the lytic granules of lymphocytes by the mannose-6-phosphate receptor. J. Cell Biol. 1993, 120, 885–896. [Google Scholar] [CrossRef] [PubMed]
- Prakash, M.D.; Bird, C.H.; Bird, P.I. Active and zymogen forms of granzyme B are constitutively released from cytotoxic lymphocytes in the absence of target cell engagement. Immunol. Cell Biol. 2009, 87, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.R.; McGuire, M.J.; Thiele, D.L. Dipeptidyl peptidase I is enriched in granules of In Vitro- and In Vivo-activated cytotoxic T lymphocytes. J. Immunol. 1993, 150, 4733–4742. [Google Scholar] [PubMed]
- Smyth, M.J.; McGuire, M.J.; Thia, K.Y. Expression of recombinant human granzyme B. A processing and activation role for dipeptidyl peptidase I. J. Immunol. 1995, 154, 6299–6305. [Google Scholar] [PubMed]
- Galli, S.J.; Nakae, S.; Tsai, M. Mast cells in the development of adaptive immune responses. Nat. Immunol. 2005, 6, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Pardo, J.; Wallich, R.; Ebnet, K.; Iden, S.; Zentgraf, H.; Martin, P.; Ekiciler, A.; Prins, A.; Müllbacher, A.; Huber, M.; et al. Granzyme B is expressed in mouse mast cells In Vivo and In Vitro and causes delayed cell death independent of perforin. Cell Death Differ. 2007, 14, 1768–1779. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.T.; Lim, D.; Granville, D.J. Granzyme B in skin inflammation and disease. Matrix Biol. 2019, 75–76, 126–140. [Google Scholar] [CrossRef] [PubMed]
- Wroblewski, M.; Bauer, R.; Córdova, M.C.; Udonta, F.; Ben-Batalla, I.; Legler, K.; Hauser, C.; Egberts, J.; Janning, M.; Velthaus, J.; et al. Mast cells decrease efficacy of anti-angiogenic therapy by secreting matrix-degrading granzyme B. Nat. Commun. 2017, 8, 269. [Google Scholar] [CrossRef] [PubMed]
- Hendel, A.; Hsu, I.; Granville, D.J. Granzyme B releases vascular endothelial growth factor from extracellular matrix and induces vascular permeability. Lab. Investig. 2014, 94, 716–725. [Google Scholar] [CrossRef] [PubMed]
- Ueshima, C.; Kataoka, T.R.; Hirata, M.; Furuhata, A.; Suzuki, E.; Toi, M.; Tsuruyama, T.; Okayama, Y.; Haga, H. The Killer Cell Ig-like Receptor 2DL4 Expression in Human Mast Cells and Its Potential Role in Breast Cancer Invasion. Cancer Immunol. Res. 2015, 3, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Bronte, V.; Brandau, S.; Chen, S.-H.; Colombo, M.P.; Frey, A.B.; Greten, T.F.; Mandruzzato, S.; Murray, P.J.; Ochoa, A.; Ostrand-Rosenberg, S.; et al. Recommendations for myeloid-derived suppressor cell nomenclature and characterization standards. Nat. Commun. 2016, 7, 12150. [Google Scholar] [CrossRef]
- Condamine, T.; Gabrilovich, D.I. Molecular mechanisms regulating myeloid-derived suppressor cell differentiation and function. Trends Immunol. 2011, 32, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Eksioglu, E.A.; Zhou, J.; Zhang, L.; Djeu, J.; Fortenbery, N.; Epling-Burnette, P.; van Bijnen, S.; Dolstra, H.; Cannon, J.; et al. Induction of myelodysplasia by myeloid-derived suppressor cells. J. Clin. Investig. 2013, 123, 4595–4611. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.-J.; Kim, H.; Suk, K.; Lee, W.-H. Macrophages express granzyme B in the lesion areas of atherosclerosis and rheumatoid arthritis. Immunol. Lett. 2007, 111, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Dufait, I.; Pardo, J.; Escors, D.; de Vlaeminck, Y.; Jiang, H.; Keyaerts, M.; de Ridder, M.; Breckpot, K. Perforin and Granzyme B Expressed by Murine Myeloid-Derived Suppressor Cells: A Study on Their Role in Outgrowth of Cancer Cells. Cancers 2019, 11, 808. [Google Scholar] [CrossRef] [PubMed]
- Hagn, M.; Belz, G.T.; Kallies, A.; Sutton, V.R.; Thia, K.Y.; Tarlinton, D.M.; Hawkins, E.D.; Trapani, J.A. Activated Mouse B Cells Lack Expression of Granzyme B. J. Immunol. 2012, 188, 3886–3892. [Google Scholar] [CrossRef] [PubMed]
- Jahrsdörfer, B.; Vollmer, A.; Blackwell, S.E.; Maier, J.; Sontheimer, K.; Beyer, T.; Mandel, B.; Lunov, O.; Tron, K.; Nienhaus, G.U.; et al. Granzyme B produced by human plasmacytoid dendritic cells suppresses T-cell expansion. Blood 2010, 115, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Riley, J.K.; Takeda, K.; Akira, S.; Schreiber, R.D. Interleukin-10 Receptor Signaling through the JAK-STAT Pathway Requirement for Two Distinct Receptor-Derived Signals for Anti-Inflammatory Action. J. Biol. Chem. 1999, 274, 16513–16521. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.Y.; Clark, E.A. The role of CD40 and CD40L in Dendritic Cells. Semin. Immunol. 2009, 21, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Walsh, M.C.; Choi, Y. The role of TRAF6 in signal transduction and the immune response. Microbes Infect. 2004, 6, 1333–1338. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, Y.; Onoé, K. Distinct regulation of CD40-mediated interleukin-6 and interleukin-12 productions via mitogen-activated protein kinase and nuclear factor κB-inducing kinase in mature dendritic cells. Immunology 2006, 117, 526–535. [Google Scholar] [CrossRef]
- Kerkmann, M.; Rothenfusser, S.; Hornung, V.; Towarowski, A.; Wagner, M.; Sarris, A.; Giese, T.; Endres, S.; Hartmann, G. Activation with CpG-A and CpG-B Oligonucleotides Reveals Two Distinct Regulatory Pathways of Type I IFN Synthesis in Human Plasmacytoid Dendritic Cells. J. Immunol. 2003, 170, 4465–4474. [Google Scholar] [CrossRef] [PubMed]
- Mui, A.L.; Wakao, H.; O’Farrell, A.M.; Harada, N.; Miyajima, A. Interleukin-3, granulocyte-macrophage colony stimulating factor and interleukin-5 transduce signals through two STAT5 homologs. EMBO J. 1995, 14, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Gerada, C.; Steain, M.; Campbell, T.M.; McSharry, B.; Slobedman, B.; Abendroth, A. Granzyme B Cleaves Multiple Herpes Simplex Virus 1 and Varicella-Zoster Virus (VZV) Gene Products, and VZV ORF4 Inhibits Natural Killer Cell Cytotoxicity. J. Virol. 2019, 93, e01140-19. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Cheng, A.-C.; Wang, M.-S.; Jia, R.-Y.; Sun, K.-F.; Yang, Q.; Wu, Y.; Zhu, D.; Chen, S.; Liu, M.-F.; et al. The suppression of apoptosis by α-herpesvirus. Cell Death Dis. 2017, 8, e2749. [Google Scholar] [CrossRef] [PubMed]
- Marcet-Palacios, M.; Duggan, B.L.; Shostak, I.; Barry, M.; Geskes, T.; Wilkins, J.A.; Yanagiya, A.; Sonenberg, N.; Bleackley, R.C. Granzyme B Inhibits Vaccinia Virus Production through Proteolytic Cleavage of Eukaryotic Initiation Factor 4 Gamma 3. PLoS Pathog. 2011, 7, e1002447. [Google Scholar] [CrossRef] [PubMed]
- Rajani, D.K.; Walch, M.; Martinvalet, D.; Thomas, M.P.; Lieberman, J. Alterations in RNA processing during immune-mediated programmed cell death. Proc. Natl. Acad. Sci. USA 2012, 109, 8688–8693. [Google Scholar] [CrossRef] [PubMed]
- Dotiwala, F.; Mulik, S.; Polidoro, R.B.; Ansara, J.A.; Burleigh, B.A.; Walch, M.; Gazzinelli, R.T.; Lieberman, J. Killer lymphocytes use granulysin, perforin and granzymes to kill intracellular parasites. Nat. Med. 2016, 22, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Walch, M.; Dotiwala, F.; Mulik, S.; Thiery, J.; Kirchhausen, T.; Clayberger, C.; Krensky, A.M.; Martinvalet, D.; Lieberman, J. Cytotoxic cells kill intracellular bacteria through Granulysin-mediated delivery of Granzymes. Cell 2014, 157, 1309–1323. [Google Scholar] [CrossRef] [PubMed]
- Dotiwala, F.; Santara, S.S.; Binker-Cosen, A.A.; Li, B.; Chandrasekaran, S.; Lieberman, J. Granzyme B Disrupts Central Metabolism and Protein Synthesis in Bacteria to Promote an Immune Cell Death Program. Cell 2017, 171, 1125–1137.e11. [Google Scholar] [CrossRef] [PubMed]
- León, D.L.; Matthey, P.; Fellay, I.; Blanchard, M.; Martinvalet, D.; Mantel, P.-Y.; Filgueira, L.; Walch, M. Granzyme B Attenuates Bacterial Virulence by Targeting Secreted Factors. iScience 2020, 23, 100932. [Google Scholar] [CrossRef]
- Zhao, D.-M.; Thornton, A.M.; DiPaolo, R.J.; Shevach, E.M. Activated CD4+CD25+ T cells selectively kill B lymphocytes. Blood 2006, 107, 3925–3932. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, C.A.; Christiansson, L.H.; Thörn, I.; Mangsbo, S.; Paul-Wetterberg, G.; Sundström, C.; Tötterman, T.H.; Simonsson, B.; Enblad, G.; Frisk, P.; et al. Both CD4+ FoxP3+ and CD4+ FoxP3- T cells from patients with B-cell malignancy express cytolytic markers and kill autologous leukaemic B cells In Vitro. Immunology 2011, 133, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Grossman, W.J.; Verbsky, J.W.; Barchet, W.; Colonna, M.; Atkinson, J.P.; Ley, T.J. Human T Regulatory Cells Can Use the Perforin Pathway to Cause Autologous Target Cell Death. Immunity 2004, 21, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Pfisterer, K.; Forster, F.; Paster, W.; Supper, V.; Ohradanova-Repic, A.; Eckerstorfer, P.; Zwirzitz, A.; Donner, C.; Boulegue, C.; Schiller, H.B.; et al. The Late Endosomal Transporter CD222 Directs the Spatial Distribution and Activity of Lck. J. Immunol. 2014, 193, 2718–2732. [Google Scholar] [CrossRef] [PubMed]
- Motyka, B.; Korbutt, G.; Pinkoski, M.J.; Heibein, J.A.; Caputo, A.; Hobman, M.; Barry, M.; Shostak, I.; Sawchuk, T.; Holmes, C.F.B.; et al. Mannose 6-Phosphate/Insulin-like Growth Factor II Receptor Is a Death Receptor for Granzyme B during Cytotoxic T Cell–Induced Apoptosis. Cell 2000, 103, 491–500. [Google Scholar] [CrossRef]
- Ahmed, K.A.; Wang, L.; Griebel, P.; Mousseau, D.D.; Xiang, J. Differential expression of mannose-6-phosphate receptor regulates T cell contraction. J. Leukoc. Biol. 2015, 98, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Hlongwane, P.; Mungra, N.; Madheswaran, S.; Akinrinmade, O.A.; Chetty, S.; Barth, S. Human Granzyme B Based Targeted Cytolytic Fusion Proteins. Biomedicines 2018, 6, 72. [Google Scholar] [CrossRef] [PubMed]
- Kurschus, F.C.; Jenne, D.E. Delivery and therapeutic potential of human granzyme B. Immunol. Rev. 2010, 235, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Liang, S.; Wang, T.; Yu, S.; Yang, R.; Hou, T.; Liu, Y.; He, C.; Zhang, N. Engineering Thermo-pH Dual Responsive Hydrogel for Enhanced Tumor Accumulation, Penetration, and Chemo-Protein Combination Therapy. Int. J. Nanomed. 2020, 15, 4739–4752. [Google Scholar] [CrossRef] [PubMed]
- Paydarnia, N.; Nikkhoi, S.K.; Fakhravar, A.; Mehdiabdol, M.; Heydarzadeh, H.; Ranjbar, S. Synergistic effect of granzyme B-azurin fusion protein on breast cancer cells. Mol. Biol. Rep. 2019, 46, 3129–3140. [Google Scholar] [CrossRef]
- Sharma, V.; Delgado, M.; Ganea, D. Granzyme B, a New Player in Activation-Induced Cell Death, Is Down-Regulated by Vasoactive Intestinal Peptide in Th2 but Not Th1 Effectors. J. Immunol. 2006, 176, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Hoek, K.L.; Greer, M.J.; McClanahan, K.G.; Nazmi, A.; Piazuelo, M.B.; Singh, K.; Wilson, K.T.; Olivares-Villagómez, D. Granzyme B prevents aberrant IL-17 production and intestinal pathogenicity in CD4+ T cells. Mucosal Immunol. 2021, 14, 1088–1099. [Google Scholar] [CrossRef] [PubMed]
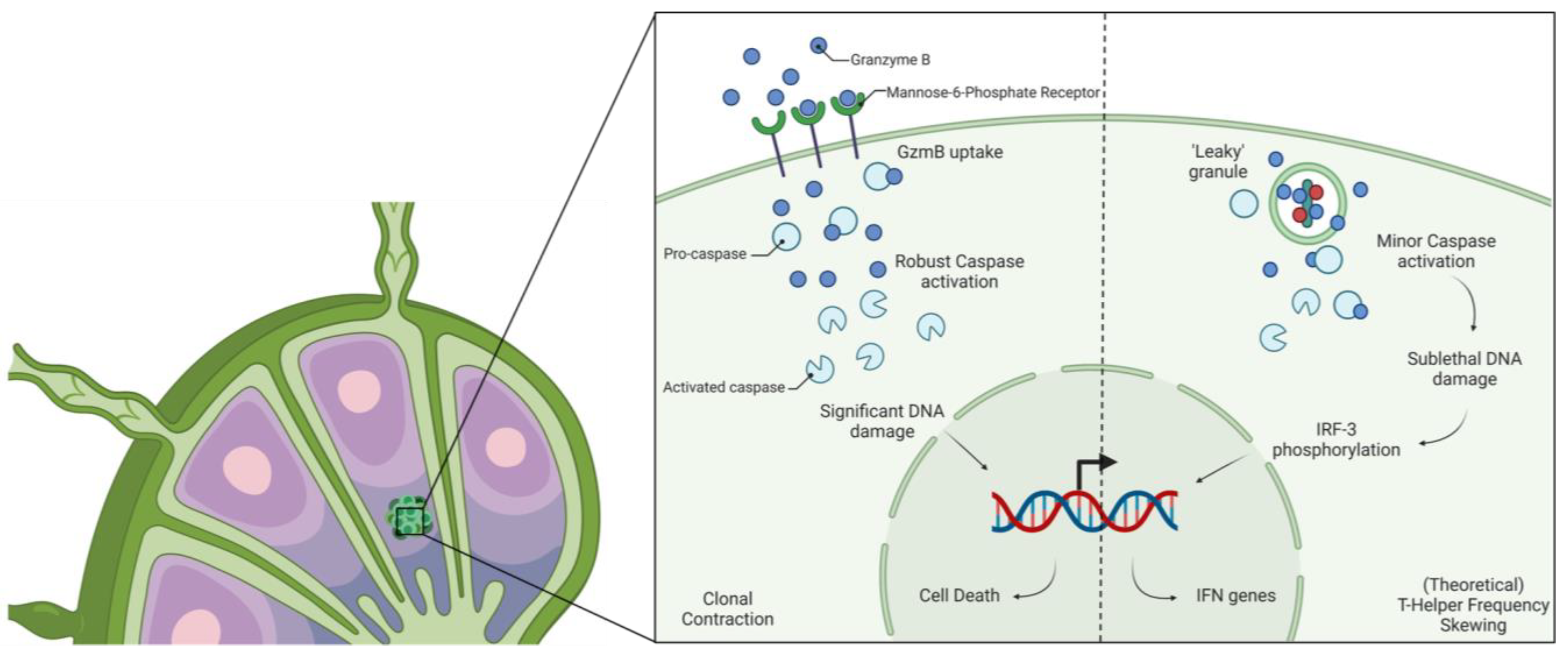
- Gapud, E.J.; Trejo-Zambrano, M.I.; Gomez-Banuelos, E.; Tiniakou, E.; Antiochos, B.; Granville, D.J.; Andrade, F.; Casciola-Rosen, L.; Rosen, A. Granzyme B Induces IRF-3 Phosphorylation through a Perforin-Independent Proteolysis-Dependent Signaling Cascade without Inducing Cell Death. J. Immunol. 2021, 206, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Mossu, A.; Daoui, A.; Bonnefoy, F.; Aubergeon, L.; Saas, P.; Perruche, S. Plasmacytoid Dendritic Cells Die by the CD8 T Cell–Dependent Perforin Pathway during Acute Nonviral Inflammation. J. Immunol. 2016, 197, 1672–1682. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Couturier, J.; Yu, X.; Medina, M.A.; Kozinetz, C.A.; Lewis, D.E. Granzyme B secretion by human memory CD4 T cells is less strictly regulated compared to memory CD8 T cells. BMC Immunol. 2014, 15, 36. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Sharma, S.C.; Das, S.N. Dynamics of regulatory T cells (Tregs) in patients with oral squamous cell carcinoma. J. Surg. Oncol. 2017, 116, 1103–1113. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Li, C.; Zheng, Y.; Liu, X. Identification of pyroptosis-related signature for cervical cancer predicting prognosis. Aging 2021, 13, 24795–24814. [Google Scholar] [CrossRef] [PubMed]
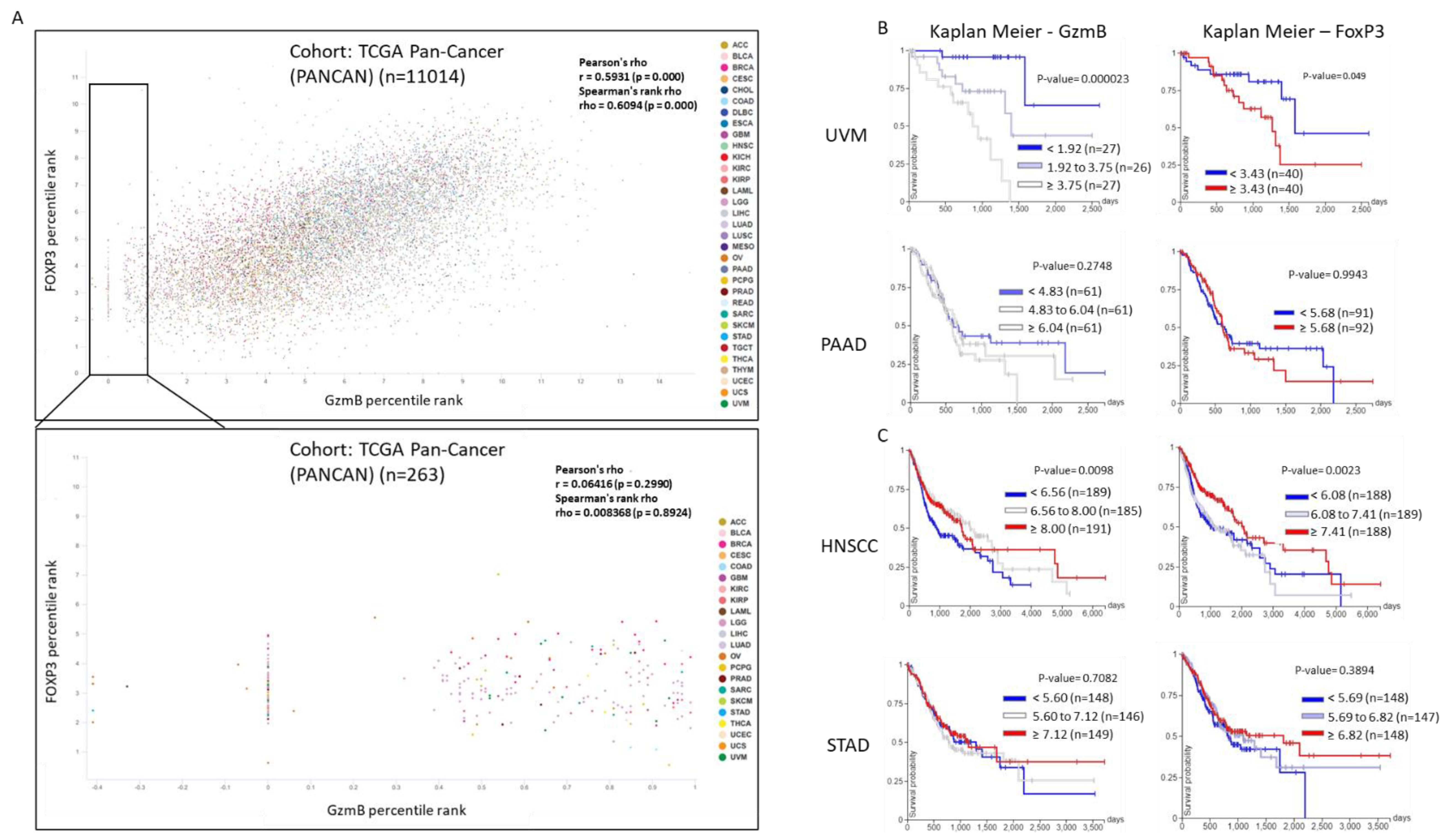
- Goldman, M.J.; Craft, B.; Hastie, M.; Repečka, K.; McDade, F.; Kamath, A.; Banerjee, A.; Luo, Y.; Rogers, D.; Brooks, A.N.; et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. 2020, 38. [Google Scholar] [CrossRef] [PubMed]
- Oztas, E.; Ozler, S.; Tokmak, A.; Yilmaz, N.; Celik, H.T.; Kazancı, F.H.; Danisman, N.; Ergin, M.; Yakut, H.I. Increased levels of serum granzyme-B is associated with insulin resistance and increased cardiovascular risk in adolescent polycystic ovary syndrome patients. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 198, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Qiao, J.; Zhou, M.; Li, Z.; Ren, J.; Gao, G.; Zhen, J.; Cao, G.; Ding, L. Elevated serum granzyme B levels are associated with disease activity and joint damage in patients with rheumatoid arthritis. J. Int. Med. Res. 2020, 48, 300060520962954. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Nakamura, T.; Adachi, H.; Yagita, H.; Okumura, K. Antigen-independent T cell activation mediated by a very late activation antigen-like extracellular matrix receptor. Eur. J. Immunol. 1991, 21, 1559–1562. [Google Scholar] [CrossRef] [PubMed]
- Prakash, M.D.; Munoz, M.A.; Jain, R.; Tong, P.L.; Koskinen, A.; Regner, M.; Kleifeld, O.; Ho, B.; Olson, M.; Turner, S.J.; et al. Granzyme B Promotes Cytotoxic Lymphocyte Transmigration via Basement Membrane Remodeling. Immunity 2014, 41, 960–972. [Google Scholar] [CrossRef] [PubMed]
- Backes, C.; Kuentzer, J.; Lenhof, H.-P.; Comtesse, N.; Meese, E. GraBCas: A bioinformatics tool for score-based prediction of Caspase- and Granzyme B-cleavage sites in protein sequences. Nucleic Acids Res. 2005, 33, W208–W213. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Li, F.; Leier, A.; Marquez-Lago, T.T.; Akutsu, T.; Haffari, G.; Chou, K.-C.; Webb, G.I.; Pike, R.N. PROSPERous: High-throughput prediction of substrate cleavage sites for 90 proteases with improved accuracy. Bioinformatics 2018, 34, 684–687. [Google Scholar] [CrossRef] [PubMed]
- Varnum-Finney, B.; Purton, L.E.; Yu, M.; Brashem-Stein, C.; Flowers, D.; Staats, S.; Moore, K.A.; le Roux, I.; Mann, R.; Gray, G.; et al. The Notch ligand, Jagged-1, influences the development of primitive hematopoietic precursor cells. Blood 1998, 91, 4084–4091. [Google Scholar] [CrossRef] [PubMed]
- Olivier, A.; Lauret, E.; Gonin, P.; Galy, A. The Notch ligand delta-1 is a hematopoietic development cofactor for plasmacytoid dendritic cells. Blood 2006, 107, 2694–2701. [Google Scholar] [CrossRef] [PubMed]
- van de Walle, I.; de Smet, G.; Gärtner, M.; de Smedt, M.; Waegemans, E.; Vandekerckhove, B.; Leclercq, G.; Plum, J.; Aster, J.C.; Bernstein, I.D.; et al. Jagged2 acts as a Delta-like Notch ligand during early hematopoietic cell fate decisions. Blood 2011, 117, 4449–4459. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Charng, W.-L.; Bellen, H.J. Chapter five—Endocytosis and intracellular trafficking of notch and its ligands. In Current Topics in Developmental Biology; Kopan, R., Ed.; Academic Press: Cambridge, MA, USA, 2010; Volume 92, pp. 165–200. [Google Scholar] [CrossRef]
- van Tetering, G.; Bovenschen, N.; Meeldijk, J.; van Diest, P.J.; Vooijs, M. Cleavage of Notch1 by granzyme B disables its transcriptional activity. Biochem. J. 2011, 437, 313–322. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tibbs, E.; Cao, X. Emerging Canonical and Non-Canonical Roles of Granzyme B in Health and Disease. Cancers 2022, 14, 1436. https://doi.org/10.3390/cancers14061436
Tibbs E, Cao X. Emerging Canonical and Non-Canonical Roles of Granzyme B in Health and Disease. Cancers. 2022; 14(6):1436. https://doi.org/10.3390/cancers14061436
Chicago/Turabian StyleTibbs, Ellis, and Xuefang Cao. 2022. "Emerging Canonical and Non-Canonical Roles of Granzyme B in Health and Disease" Cancers 14, no. 6: 1436. https://doi.org/10.3390/cancers14061436
APA StyleTibbs, E., & Cao, X. (2022). Emerging Canonical and Non-Canonical Roles of Granzyme B in Health and Disease. Cancers, 14(6), 1436. https://doi.org/10.3390/cancers14061436





