Short-Term Fasting Synergizes with Solid Cancer Therapy by Boosting Antitumor Immunity
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Fasting Causes Metabolic Reprogramming, Stimulates Autophagy, and Can Increase Immunogenic Cell Death
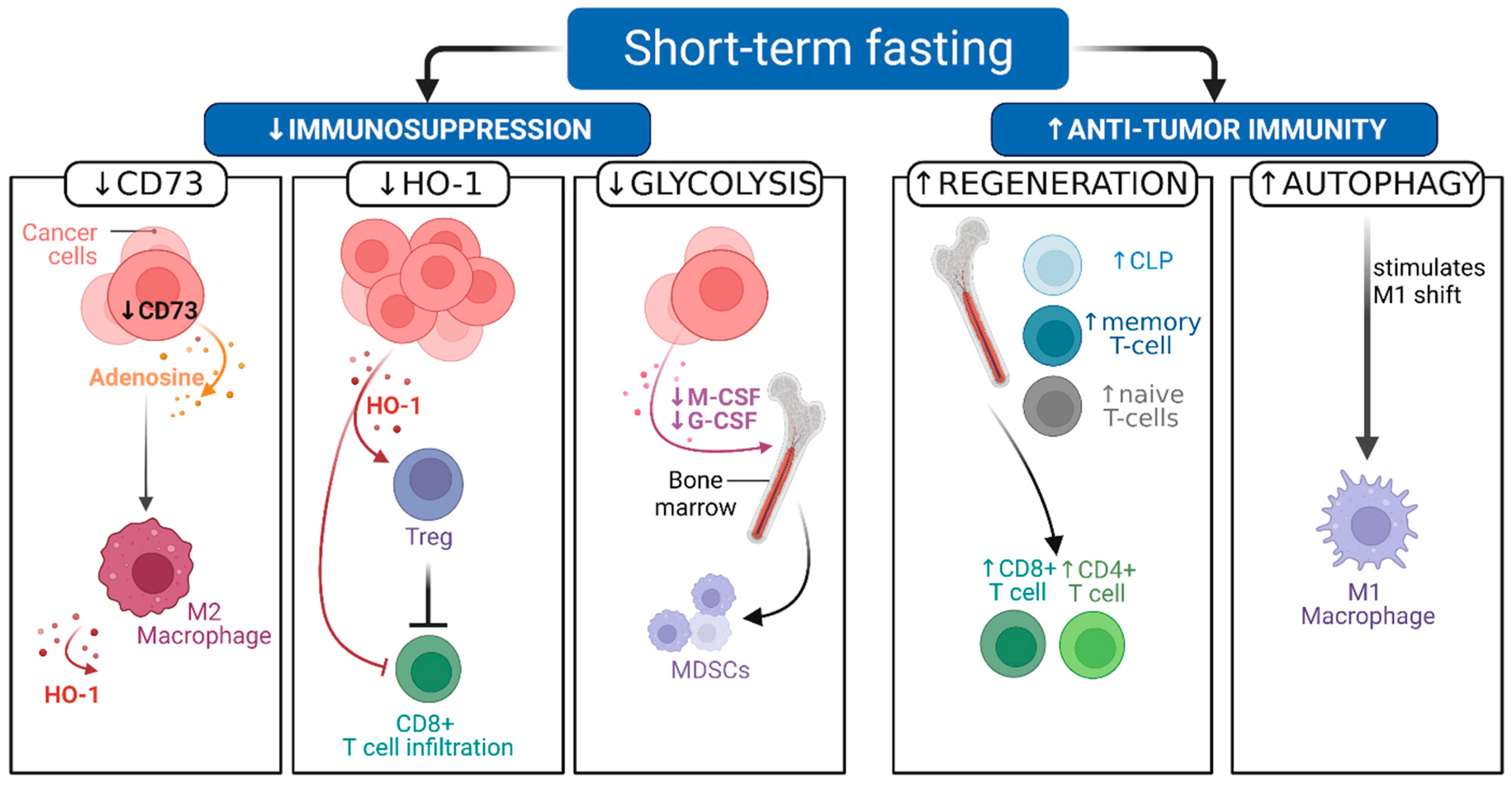
2.2. Pre-Clinical Evidence Shows That STF Can Decrease Immunosuppression and Boost Antitumor Immunity
2.3. Clinical Studies on Fasting Combined with Cancer Treatment
2.4. Fasting Can Synergize with Other Cancer Therapies, including Endocrine, Radiation, and Immunotherapy
3. Discussion
4. Conclusions
5. Key Messages
- Preclinical evidence shows that STF can reduce adverse events and improve the antitumor effects of chemotherapy.
- Preclinical evidence demonstrates that STF can potentiate antitumor immunity, in part by enhancing immunogenicity and relieving tumor-induced immunosuppression.
- Clinical trials show that short-term fasting around chemotherapy is safe in a select group of fit patients and may increase the effectiveness of chemotherapy.
- (Pre-)clinical evidence suggests fasting regimens might alleviate immunosuppressive effects of chemotherapy.
- More studies are required to confirm that STF boosts the efficacy of cancer therapy, such as chemotherapy and immunotherapy, by enhancing tumor immunity.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AKT, Akt | protein kinase B |
| AMPK | adenosine monophosphate-activated protein kinase |
| ATP | adenosine 5′-triphosphate |
| cAMP-PKA | cyclic adenosine monophosphate protein kinase A |
| CLP | common lymphoid progenitor |
| CP | cyclophosphamide |
| CR | complete response |
| CT | chemotherapy |
| DC | dendritic cells |
| DSR | differential stress resistance |
| DXR | doxorubicin |
| ECM | extracellular matrix |
| EGR1 | early growth response 1 |
| FMD | fasting mimicking diet |
| G-CSF | granulocyte colony-stimulating factor |
| HER2 | human epidermal growth factor receptor 2 |
| HO-1 | heme oxygenase-1 |
| HR+ | hormone receptor positive |
| ICD | immunogenic cell death |
| IFNγ | interferon gamma |
| IGF-1 | insulin growth factor 1 |
| IGF1R | insulin growth factor 1 receptor |
| M-CSF | macrophage colony-stimulating factor |
| MDSC | myeloid derived suppressor cells |
| mTOR | mammalian target of rapamycin |
| MTX | mitoxantrone |
| NK | natural killer cell |
| OX | oxaliplatin |
| PBMC | peripheral blood mononuclear cell |
| PD1 | programmed cell death protein 1 |
| PD-L1 | programmed cell death protein 1 ligand |
| PI3K | phosphoinositide 3-kinase |
| PR | partial response |
| QoL | quality of life |
| ROS | reactive oxygen species |
| RT | radiation therapy |
| STF | short-term fasting |
| TAC | docetaxel/doxorubicin/cyclophosphamide |
| TAM | tumor associated macrophages |
| TIL | tumor infiltrating lymphocyte |
| Tregs | regulatory T cells |
References
- Zitvogel, L.; Apetoh, L.; Ghiringhelli, F.; Kroemer, G. Immunological aspects of cancer chemotherapy. Nat. Rev. Immunol. 2008, 8, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Andre, F.; Dieci, M.V.; Dubsky, P.; Sotiriou, C.; Curigliano, G.; Denkert, C.; Loi, S. Molecular pathways: Involvement of immune pathways in the therapeutic response and outcome in breast cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2013, 19, 28–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pietrocola, F.; Pol, J.; Vacchelli, E.; Rao, S.; Enot, D.P.; Baracco, E.E.; Levesque, S.; Castoldi, F.; Jacquelot, N.; Yamazaki, T.; et al. Caloric Restriction Mimetics Enhance Anticancer Immunosurveillance. Cancer Cell 2016, 30, 147–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Biase, S.; Lee, C.; Brandhorst, S.; Manes, B.; Buono, R.; Cheng, C.W.; Cacciottolo, M.; Martin-Montalvo, A.; de Cabo, R.; Wei, M.; et al. Fasting-Mimicking Diet Reduces HO-1 to Promote T Cell-Mediated Tumor Cytotoxicity. Cancer Cell 2016, 30, 136–146. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Raffaghello, L.; Brandhorst, S.; Safdie, F.M.; Bianchi, G.; Martin-Montalvo, A.; Pistoia, V.; Wei, M.; Hwang, S.; Merlino, A.; et al. Fasting cycles retard growth of tumors and sensitize a range of cancer cell types to chemotherapy. Sci. Transl. Med. 2012, 4, 124ra27. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Safdie, F.M.; Raffaghello, L.; Wei, M.; Madia, F.; Parrella, E.; Hwang, D.; Cohen, P.; Bianchi, G.; Longo, V.D.; et al. Reduced levels of IGF-I mediate differential protection of normal and cancer cells in response to fasting and improve chemotherapeutic index. Cancer Res. 2010, 70, 1564–1572. [Google Scholar] [CrossRef] [Green Version]
- De Groot, S.; Pijl, H.; van der Hoeven, J.J.M.; Kroep, J.R. Effects of short-term fasting on cancer treatment. J. Exp. Clin. Cancer Res. 2019, 38, 209. [Google Scholar] [CrossRef] [Green Version]
- De Groot, S.; Lugtenberg, R.T.; Cohen, D.; Welters, M.J.P.; Ehsan, I.; Vreeswijk, M.P.G.; Smit, V.T.; de Graaf, H.; Heijns, J.B.; Portielje, J.E.A.; et al. Fasting mimicking diet as an adjunct to neoadjuvant chemotherapy for breast cancer in the multicentre randomized phase 2 DIRECT trial. Nat. Commun. 2020, 11, 3083. [Google Scholar] [CrossRef]
- Safdie, F.M.; Dorf, F.T.; Quinn, D.; Fontana, L.; Wei, M.; Lee, C.; Cohen, P.; Longo, V.D. Fasting and cancer treatment in humans: A case series report. Aging 2009, 1, 988–1007. [Google Scholar] [CrossRef] [Green Version]
- Ajona, D.; Ortiz-Espinosa, S.; Lozano, T.; Exposito, F.; Calvo, A.; Valencia, K.; Redrado, M.; Remírez, A.; Lecanda, F.; Alignani, D.; et al. Short-term starvation reduces IGF-1 levels to sensitize lung tumors to PD-1 immune checkpoint blockade. Nat. Cancer 2020, 1, 75–85. [Google Scholar] [CrossRef] [Green Version]
- Vernieri, C.; Fuca, G.; Ligorio, F.; Huber, V.; Vingiani, A.; Iannelli, F.; Raimondi, A.; Rinchai, D.; Frige, G.; Belfiore, A.; et al. Fasting-mimicking diet is safe and reshapes metabolism and antitumor immunity in cancer patients. Cancer Discov. 2022, 12, 90–107. [Google Scholar] [CrossRef] [PubMed]
- Longo, V.D.; Mattson, M.P. Fasting: Molecular mechanisms and clinical applications. Cell Metab. 2014, 19, 181–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerndt, P.R.; Naughton, J.L.; Driscoll, C.E.; Loxterkamp, D.A. Fasting: The history, pathophysiology and complications. West J. Med. 1982, 137, 379–399. [Google Scholar]
- Finnell, J.S.; Saul, B.C.; Goldhamer, A.C.; Myers, T.R. Is fasting safe? A chart review of adverse events during medically supervised, water-only fasting. BMC Complement. Altern. Med. 2018, 18, 67. [Google Scholar] [CrossRef] [PubMed]
- Brandhorst, S.; Choi, I.Y.; Wei, M.; Cheng, C.W.; Sedrakyan, S.; Navarrete, G.; Dubeau, L.; Pen Yap, L.; Park, R.; Vinciguerra, M.; et al. A Periodic Diet that Mimics Fasting Promotes Multi-System Regeneration, Enhanced Cognitive Performance, and Healthspan. Cell Metab. 2015, 22, 86–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patterson, R.E.; Sears, D.D. Metabolic Effects of Intermittent Fasting. Annu. Rev. Nutr. 2017, 37, 371–393. [Google Scholar] [CrossRef] [Green Version]
- Madeo, F.; Pietrocola, F.; Eisenberg, T.; Kroemer, G. Caloric restriction mimetics: Towards a molecular definition. Nat. Rev. Drug Discov. 2014, 13, 727–740. [Google Scholar] [CrossRef]
- Turbitt, W.J.; Demark-Wahnefried, W.; Peterson, C.M.; Norian, L.A. Targeting Glucose Metabolism to Enhance Immunotherapy: Emerging Evidence on Intermittent Fasting and Calorie Restriction Mimetics. Front. Immunol. 2019, 10, 1402. [Google Scholar] [CrossRef]
- Raffaghello, L.; Lee, C.; Safdie, F.M.; Wei, M.; Madia, F.; Bianchi, G.; Longo, V.D. Starvation-dependent differential stress resistance protects normal but not cancer cells against high-dose chemotherapy. Proc. Natl. Acad. Sci. USA 2008, 105, 8215–8220. [Google Scholar] [CrossRef] [Green Version]
- Kroemer, G.; Galluzzi, L.; Kepp, O.; Zitvogel, L. Immunogenic cell death in cancer therapy. Annu. Rev. Immunol. 2013, 31, 51–72. [Google Scholar] [CrossRef]
- Eriau, E.; Paillet, J.; Kroemer, G.; Pol, J.G. Metabolic Reprogramming by Reduced Calorie Intake or Pharmacological Caloric Restriction Mimetics for Improved Cancer Immunotherapy. Cancers 2021, 13, 1260. [Google Scholar] [CrossRef] [PubMed]
- Tesniere, A.; Schlemmer, F.; Boige, V.; Kepp, O.; Martins, I.; Ghiringhelli, F.; Aymeric, L.; Michaud, M.; Apetoh, L.; Barault, L.; et al. Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene 2010, 29, 482–491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Obeid, M.; Tesniere, A.; Ghiringhelli, F.; Fimia, G.M.; Apetoh, L.; Perfettini, J.-L.; Castedo, M.; Mignot, G.; Panaretakis, T.; Casares, N.; et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat. Med. 2007, 13, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Michaud, M.; Martins, I.; Sukkurwala, A.Q.; Adjemian, S.; Ma, Y.; Pellegatti, P.; Shen, S.; Kepp, O.; Scoazec, M.; Mignot, G.; et al. Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science 2011, 334, 1573–1577. [Google Scholar] [CrossRef]
- De Groot, S.; Röttgering, B.; Gelderblom, H.; Pijl, H.; Szuhai, K.; Kroep, J.R. Unraveling the Resistance of IGF-Pathway Inhibition in Ewing Sarcoma. Cancers 2020, 12, 3568. [Google Scholar] [CrossRef]
- Nencioni, A.; Caffa, I.; Cortellino, S.; Longo, V.D. Fasting and cancer: Molecular mechanisms and clinical application. Nat. Rev. Cancer 2018, 18, 707–719. [Google Scholar] [CrossRef]
- Chi, H. Regulation and function of mTOR signalling in T cell fate decisions. Nat. Rev. Immunol. 2012, 12, 325–338. [Google Scholar] [CrossRef]
- Warburg, O. The Metabolism of Carcinoma Cells. J. Cancer Res. 1925, 9, 148–163. [Google Scholar] [CrossRef] [Green Version]
- Liberti, M.V.; Locasale, J.W. The Warburg Effect: How Does it Benefit Cancer Cells? Trends Biochem. Sci. 2016, 41, 211–218. [Google Scholar] [CrossRef] [Green Version]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef] [Green Version]
- Bianchi, G.; Martella, R.; Ravera, S.; Marini, C.; Capitanio, S.; Orengo, A.; Emionite, L.; Lavarello, C.; Amaro, A.; Petretto, A.; et al. Fasting induces anti-Warburg effect that increases respiration but reduces ATP-synthesis to promote apoptosis in colon cancer models. Oncotarget 2015, 6, 11806–11819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadeghian, M.; Rahmani, S.; Khalesi, S.; Hejazi, E. A review of fasting effects on the response of cancer to chemotherapy. Clin. Nutr. 2020, 40, 1669–1681. [Google Scholar] [CrossRef] [PubMed]
- Bagherniya, M.; Butler, A.E.; Barreto, G.E.; Sahebkar, A. The effect of fasting or calorie restriction on autophagy induction: A review of the literature. Ageing Res. Rev. 2018, 47, 183–197. [Google Scholar] [CrossRef]
- Yamazaki, T.; Bravo-San Pedro, J.M.; Galluzzi, L.; Kroemer, G.; Pietrocola, F. Autophagy in the cancer-immunity dialogue. Adv. Drug Deliv. Rev. 2021, 169, 40–50. [Google Scholar] [CrossRef]
- Sharifi, M.N.; Mowers, E.E.; Drake, L.E.; Collier, C.; Chen, H.; Zamora, M.; Mui, S.; Macleod, K.F. Autophagy Promotes Focal Adhesion Disassembly and Cell Motility of Metastatic Tumor Cells through the Direct Interaction of Paxillin with LC3. Cell Rep. 2016, 15, 1660–1672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, P.; Wang, H.; He, Z.; Chen, X.; Wu, Q.; Chen, W.; Sun, Z.; Weng, M.; Zhu, M.; Ma, D.; et al. Fasting inhibits colorectal cancer growth by reducing M2 polarization of tumor-associated macrophages. Oncotarget 2017, 8, 74649–74660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takakuwa, T.; Nakashima, Y.; Koh, H.; Nakane, T.; Nakamae, H.; Hino, M. Short-Term Fasting Induces Cell Cycle Arrest in Immature Hematopoietic Cells and Increases the Number of Naïve T Cells in the Bone Marrow of Mice. Acta Haematol. 2019, 141, 189–198. [Google Scholar] [CrossRef] [Green Version]
- Collins, N.; Han, S.J.; Enamorado, M.; Link, V.M.; Huang, B.; Moseman, E.A.; Kishton, R.J.; Shannon, J.P.; Dixit, D.; Schwab, J.P.; et al. The Bone Marrow Protects and Optimizes Immunological Memory during Dietary Restriction. Cell 2019, 178, 1088–1101.e15. [Google Scholar] [CrossRef]
- Cheng, C.W.; Adams, G.B.; Perin, L.; Wei, M.; Zhou, X.; Lam, B.S.; Da Sacco, S.; Mirisola, M.; Quinn, D.I.; Dorff, T.B.; et al. Prolonged fasting reduces IGF-1/PKA to promote hematopoietic-stem-cell-based regeneration and reverse immunosuppression. Cell Stem Cell 2014, 14, 810–823. [Google Scholar] [CrossRef] [Green Version]
- Pomatto-Watson, L.C.D.; Bodogai, M.; Bosompra, O.; Kato, J.; Wong, S.; Carpenter, M.; Duregon, E.; Chowdhury, D.; Krishna, P.; Ng, S.; et al. Daily caloric restriction limits tumor growth more effectively than caloric cycling regardless of dietary composition. Nat. Commun. 2021, 12, 6201. [Google Scholar] [CrossRef]
- Speiser, D.E.; Ho, P.-C.; Verdeil, G. Regulatory circuits of T cell function in cancer. Nat. Rev. Immunol. 2016, 16, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-H.; Qiu, J.; O’Sullivan, D.; Buck, M.D.; Noguchi, T.; Curtis, J.D.; Chen, Q.; Gindin, M.; Gubin, M.M.; van der Windt, G.J.W.; et al. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell 2015, 162, 1229–1241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pearce, E.L.; Walsh, M.C.; Cejas, P.J.; Harms, G.M.; Shen, H.; Wang, L.S.; Jones, R.G.; Choi, Y. Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 2009, 460, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Blagih, J.; Coulombe, F.; Vincent, E.E.; Dupuy, F.; Galicia-Vázquez, G.; Yurchenko, E.; Raissi, T.C.; van der Windt, G.J.W.; Viollet, B.; Pearce, E.L.; et al. The energy sensor AMPK regulates T cell metabolic adaptation and effector responses in vivo. Immunity 2015, 42, 41–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michalek, R.D.; Gerriets, V.A.; Jacobs, S.R.; Macintyre, A.N.; MacIver, N.J.; Mason, E.F.; Sullican, S.A.; Nichols, A.G.; Rathmell, J.C. Cutting edge: Distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J. Immunol. 2011, 186, 3299–3303. [Google Scholar] [CrossRef] [Green Version]
- Kishton, R.J.; Sukumar, M.; Restifo, N.P. Metabolic Regulation of T Cell Longevity and Function in Tumor Immunotherapy. Cell Metab. 2017, 26, 94–109. [Google Scholar] [CrossRef] [Green Version]
- Sukumar, M.; Liu, J.; Ji, Y.; Subramanian, M.; Crompton, J.G.; Yu, Z.; Roychoudhuri, R.; Palmer, D.C.; Muranski, P.; Karoly, E.D.; et al. Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. J. Clin. Investig. 2013, 123, 4479–4488. [Google Scholar] [CrossRef]
- Vinay, D.S.; Ryan, E.P.; Pawelec, G.; Talib, W.H.; Stagg, J.; Elkord, E.; Lichtor, T.; Denker, W.K.; Whelan, R.L.; Kumara, H.M.C.S.; et al. Immune evasion in cancer: Mechanistic basis and therapeutic strategies. Semin. Cancer Biol. 2015, 35, S185–S198. [Google Scholar] [CrossRef]
- Hato, S.V.; Khong, A.; de Vries, I.J.M.; Lesterhuis, W.J. Molecular Pathways: The Immunogenic Effects of Platinum-Based Chemotherapeutics. Clin. Cancer Res. 2014, 20, 2831–2837. [Google Scholar] [CrossRef] [Green Version]
- Alaluf, E.; Vokaer, B.; Detavernier, A.; Azouz, A.; Splittgerber, M.; Carrette, A.; Boon, L.; Libert, F.; Soares, M.; Le Moine, A.; et al. Heme oxygenase-1 orchestrates the immunosuppressive program of tumor-associated macrophages. JCI Insight 2020, 5, e133929. [Google Scholar] [CrossRef]
- Podkalicka, P.; Mucha, O.; Józkowicz, A.; Dulak, J.; Łoboda, A. Heme oxygenase inhibition in cancers: Possible tools and targets. Contemp. Oncol. 2018, 22, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.H.; Sørensen, R.B.; Brimnes, M.K.; Svane, I.M.; Becker, J.C.; thor Straten, P. Identification of heme oxygenase-1-specific regulatory CD8+ T cells in cancer patients. J. Clin. Investig. 2009, 119, 2245–2256. [Google Scholar] [CrossRef] [PubMed]
- Verbist, K.C.; Guy, C.S.; Milasta, S.; Liedmann, S.; Kamiński, M.M.; Wang, R.; Green, D.R. Metabolic maintenance of cell asymmetry following division in activated T lymphocytes. Nature 2016, 532, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.; Lu, Y.; Zhang, Y.; Yu, M.; Ma, S.; Lyu, S. Intermittent fasting suppressed splenic CD205+ G-MDSC accumulation in a murine breast cancer model by attenuating cell trafficking and inducing apoptosis. Food Sci. Nutr. 2021, 9, 5517–5526. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tanikawa, T.; Kryczek, I.; Xia, H.; Li, G.; Wu, K.; Wei, S.; Zhao, L.; Vatan, L.; Wen, B.; et al. Aerobic Glycolysis Controls Myeloid-Derived Suppressor Cells and Tumor Immunity via a Specific CEBPB Isoform in Triple-Negative Breast Cancer. Cell Metab. 2018, 28, 87–103.e6. [Google Scholar] [CrossRef] [Green Version]
- Movahedi, K.; Laoui, D.; Gysemans, C.; Baeten, M.; Stangé, G.; Van den Bossche, J.; Mack, M.; Pipeleers, D.; In’t Veld, P.; De Baetselier, P.; et al. Different Tumor Microenvironments Contain Functionally Distinct Subsets of Macrophages Derived from Ly6C(high) Monocytes. Cancer Res. 2010, 70, 5728–5739. [Google Scholar] [CrossRef] [Green Version]
- Vitale, I.; Manic, G.; Coussens, L.M.; Kroemer, G.; Galluzzi, L. Macrophages and Metabolism in the Tumor Microenvironment. Cell Metab. 2019, 30, 36–50. [Google Scholar] [CrossRef]
- Esteban-Martínez, L.; Sierra-Filardi, E.; McGreal, R.S.; Salazar-Roa, M.; Mariño, G.; Seco, E.; Durand, S.; Enot, D.; Graña, O.; Malumbres, M.; et al. Programmed mitophagy is essential for the glycolytic switch during cell differentiation. EMBO J. 2017, 36, 1688–1706. [Google Scholar] [CrossRef]
- Kaneda, M.M.; Messer, K.S.; Ralainirina, N.; Li, H.; Leem, C.J.; Gorjestani, S.; Woo, G.; Nguyen, A.V.; Figueiredo, C.C.; Foubert, P.; et al. PI3Kγ is a molecular switch that controls immune suppression. Nature 2016, 539, 437–442. [Google Scholar] [CrossRef] [Green Version]
- Fridman, W.H.; Zitvogel, L.; Sautès-Fridman, C.; Kroemer, G. The immune contexture in cancer prognosis and treatment. Nat. Rev. Clin. Oncol. 2017, 14, 717–734. [Google Scholar] [CrossRef]
- Arnold, J.N.; Magiera, L.; Kraman, M.; Fearon, D.T. Tumoral immune suppression by macrophages expressing fibroblast activation protein-α and heme oxygenase-1. Cancer Immunol. Res. 2014, 2, 121–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muliaditan, T.; Opzoomer, J.W.; Caron, J.; Okesola, M.; Kosti, P.; Lall, S.; Van Hemelrijck, M.; Dazzi, F.; Tutt, A.; Grigoriadis, A.; et al. Repurposing Tin Mesoporphyrin as an Immune Checkpoint Inhibitor Shows Therapeutic Efficacy in Preclinical Models of Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 1617–1628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luu Hoang, K.N.; Anstee, J.E.; Arnold, J.N. The Diverse Roles of Heme Oxygenase-1 in Tumor Progression. Front. Immunol. 2021, 12, 658315. [Google Scholar] [CrossRef]
- Badar, T.; Ismail, A.; AlShanqeeti, A. Safety and Feasability of Muslim Fasting While Receiving Chemotherapy. IOSR J. Pharm. 2014, 4, 15–20. [Google Scholar] [CrossRef]
- Bauersfeld, S.P.; Kessler, C.S.; Wischnewsky, M.; Jaensch, A.; Steckhan, N.; Stange, R.; Kunz, B.; Brückner, B.; Sehouli, J.; Michalsen, A. The effects of short-term fasting on quality of life and tolerance to chemotherapy in patients with breast and ovarian cancer: A randomized cross-over pilot study. BMC Cancer 2018, 18, 476. [Google Scholar] [CrossRef] [PubMed]
- Dorff, T.B.; Groshen, S.; Garcia, A.; Shah, M.; Tsao-Wei, D.; Pham, H.; Cheng, C.W.; Brandhorst, S.; Cohen, P.; Wei, M.; et al. Safety and feasibility of fasting in combination with platinum-based chemotherapy. BMC Cancer 2016, 16, 360. [Google Scholar] [CrossRef] [Green Version]
- de Groot, S.; Vreeswijk, M.P.; Welters, M.J.; Gravesteijn, G.; Boei, J.J.; Jochems, A.; Houtsma, D.; Putter, H.; Van der Hoeven, J.J.; Nortier, J.W.; et al. The effects of short-term fasting on tolerance to (neo) adjuvant chemotherapy in HER2-negative breast cancer patients: A randomized pilot study. BMC Cancer 2015, 15, 652. [Google Scholar] [CrossRef] [Green Version]
- Lugtenberg, R.T.; de Groot, S.; Kaptein, A.A.; Fischer, M.J.; Kranenbarg, E.M.-K.; Carpentier, M.D.; Cohen, D.; de Graaf, H.; Heijns, J.B.; Portielje, J.E.A.; et al. Quality of life and illness perceptions in patients with breast cancer using a fasting mimicking diet as an adjunct to neoadjuvant chemotherapy in the phase 2 DIRECT (BOOG 2013-14) trial. Breast Cancer Res. Treat. 2021, 185, 741–758. [Google Scholar] [CrossRef]
- Caffa, I.; Spagnolo, V.; Vernieri, C.; Valdemarin, F.; Becherini, P.; Wei, M.; Brandhorst, S.; Zucal, C.; Driehuis, E.; Ferrando, L.; et al. Fasting-mimicking diet and hormone therapy induce breast cancer regression. Nature 2020, 583, 620–624. [Google Scholar] [CrossRef]
- Manukian, G.; Kivolowitz, C.; DeAngelis, T.; Shastri, A.A.; Savage, J.E.; Camphausen, K.; Rodeck, U.; Zarif, J.C.; Simone, N.L. Caloric Restriction Impairs Regulatory T cells Within the Tumor Microenvironment After Radiation and Primes Effector T cells. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 1341–1349. [Google Scholar] [CrossRef]
- Lévesque, S.; Le Naour, J.; Pietrocola, F.; Paillet, J.; Kremer, M.; Castoldi, F.; Baracco, E.E.; Wang, Y.; Vacchelli, E.; Stoll, G.; et al. A synergistic triad of chemotherapy.; immune checkpoint inhibitors.; and caloric restriction mimetics eradicates tumors in mice. Oncoimmunology 2019, 8, e1657375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nikolich-Zugich, J.; Messaoudi, I. Mice and flies and monkeys too: Caloric restriction rejuvenates the aging immune system of non-human primates. Exp. Gerontol. 2005, 40, 884–893. [Google Scholar] [CrossRef] [PubMed]

| Author | Mouse Model | Treatment | Outcome, Findings |
|---|---|---|---|
| Pietrocola et al., 2016 [3] | Autophagy competent TC-1 non-small cell lung cancer | Hydroxycitrate + MTX or OX | Improved tumor growth control mediated by Treg depletion; autophagy required |
| Colorectal cancer | Hydroxycitrate + MTX or OX | Improved tumor growth control mediated by Treg depletion; autophagy required | |
| MCA205 fibrosarcoma | Hydroxycitrate + MTX or OX Hydroxycitrate + cisplatin 48-h STF | Improved tumor growth control mediated by Treg depletion; autophagy required Tumor growth control not improved; autophagy induction similar to hydroxycitrate | |
| Di Biase et al., 2016 [4] | 4T1 breast cancer, B16 melanoma | 2–3 cycles, 4-day FMD every 2 weeks + DXR or CP | Delayed tumor progression; intratumoral Treg reduction, HO-1 dependent; increased CD8+ TILs |
| Increased CLP in bone marrow, increased circulating naïve T cells | |||
| Takakuwa et al., 2019 [37] | C57Bl/6 mice | 48-h STF | Increased naïve CD4 and CD8 T cells in bone marrow |
| Collins et al., 2019 [38] | C57Bl/6 mice (several types), with B16 melanoma cell line | Calorie reduction 50% for 6 weeks | Central accumulation memory T cells associated with ↑tumor immunity and survival benefit; transient peripheral ↓CD4 & CD8 T cells (1 w); central increase CD4 and CD8 T cells (at 3 w) |
| Cheng et al., 2015 [39] | C57BL/6 J mice | 48–120 h STF around CP administration for 6 cycles in 12–14 days | Hematopoietic stem cell regeneration, IGF-1/PKA dependent; reduced chemotherapy induced mortality; reduced DNA damage in bone marrow cells |
| Pomatto-Watson et al., 2021 [40] | 4T1 breast cancer | 4-day FMD, 2 cycles in 28 days | Reduced MDSCs frequency in spleen; intratumoral MDSC frequency; increased T cell to MDSC ratio in PBMC |
| Sun et al., 2017 [36] | Colorectal cancer, in vivo | Alternate day fasting for 2 weeks | Tumor growth inhibition; less M2 polarization of macrophages |
| CT26 and RAW264.7 cells, in vitro | Mechanism in vitro: CD73 reduction, blunted adenosine release into ECM |
| Author, Location | Human Participant | Treatment | Outcome |
|---|---|---|---|
| Safdie et al., 2009, USC, USA [9] | n = 10, with different malignancies | Variable, water-only fasting 48–140 h prior to and/or 5–56 h after | Fewer side effects (self-reported) |
| Dorff et al., 2016, USC, USA (NCT00936364) [66] | n = 20, with different malignancies | Platinum-based chemotherapy with 24, 48, or 72 h of water-only fasting | Reduced DNA damage in leukocytes; trend towards less grade 3 or 4 neutropenia |
| Bauersfeld et al., 2018, Charite University, Germany (NCT01954836) [65] | n = 34, women with gynecological cancer | Water-only fasting 60 h around chemotherapy administration | Higher QoL score association |
| de Groot et al. LUMC, the Netherlands (NCT01304251) [67] | n = 13, women with breast cancer (HER2-negative) | TAC CT and 48 h STF | Reduced DNA-damage in PBMCs |
| de Groot et al., 2020, LUMC, the Netherlands (NCT02126449) [8] | n = 131, women with breast cancer (HER2-negative) | Randomized 4-day FMD or regular diet around neoadjuvant CT, maximum of 8 cycles | Increased rate of CR or PR in ITT; increased rate of pathological response per protocol; reduced DNA-damage in PBMCs; QoL non-significant improved in FMD arm; similar grade 3–4 toxicity between arms |
| Vernieri et al., 2022, University of Milan, Italy (NCT03340935, NCT03454282) [11] | n = 101, with different malignancies | 5-day FMD every 3–4 weeks | Reduction peripheral MDSCs (n = 38); boost CD4 and CD8 T cell peripheral; increased NK cytotoxic activity; intratumoral: ↑CD8 TIL, ↑DCs, ↑NKs; systemic increased IFNγ |
| Breast cancer subgroup (n = 18) | Intratumoral: ↑M1 macrophage |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Gruil, N.; Pijl, H.; van der Burg, S.H.; Kroep, J.R. Short-Term Fasting Synergizes with Solid Cancer Therapy by Boosting Antitumor Immunity. Cancers 2022, 14, 1390. https://doi.org/10.3390/cancers14061390
de Gruil N, Pijl H, van der Burg SH, Kroep JR. Short-Term Fasting Synergizes with Solid Cancer Therapy by Boosting Antitumor Immunity. Cancers. 2022; 14(6):1390. https://doi.org/10.3390/cancers14061390
Chicago/Turabian Stylede Gruil, Nadia, Hanno Pijl, Sjoerd H. van der Burg, and Judith R. Kroep. 2022. "Short-Term Fasting Synergizes with Solid Cancer Therapy by Boosting Antitumor Immunity" Cancers 14, no. 6: 1390. https://doi.org/10.3390/cancers14061390
APA Stylede Gruil, N., Pijl, H., van der Burg, S. H., & Kroep, J. R. (2022). Short-Term Fasting Synergizes with Solid Cancer Therapy by Boosting Antitumor Immunity. Cancers, 14(6), 1390. https://doi.org/10.3390/cancers14061390








