Antitumor Activities of Interleukin-12 in Melanoma
Abstract
Simple Summary
Abstract
1. Introduction
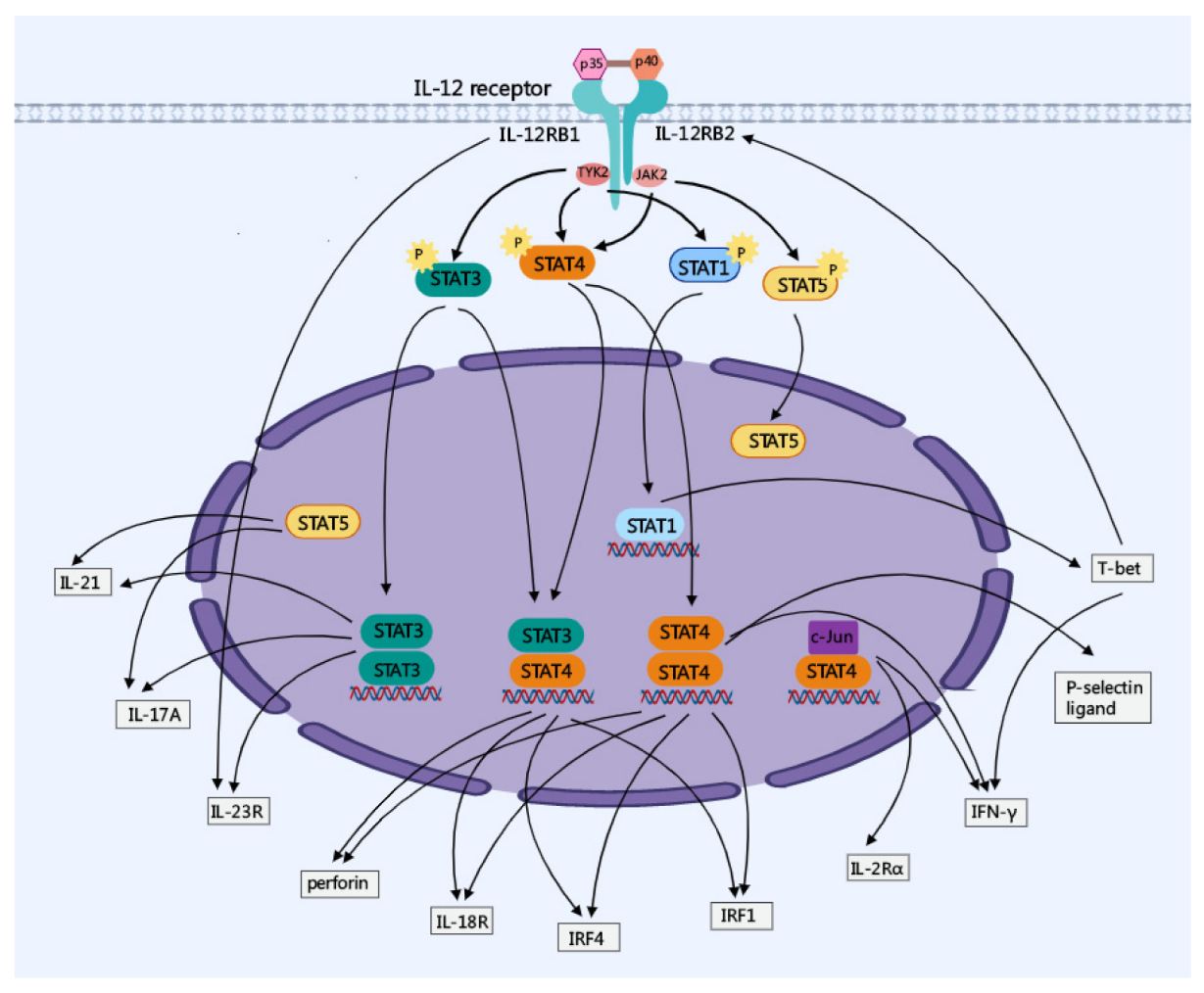
2. Major Molecular Regulation of IL-12 Signaling
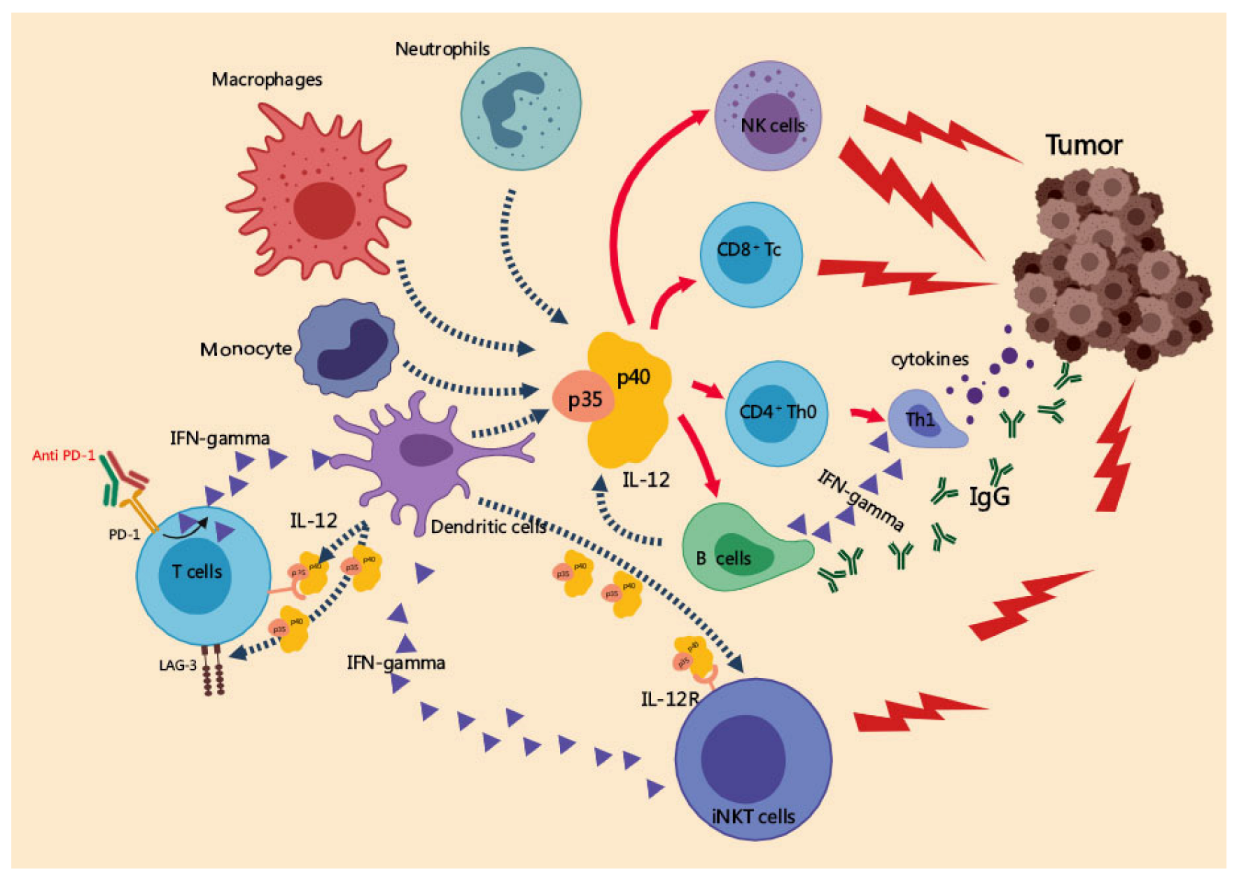
3. Anti-Tumor Effects of IL-12 in Melanoma
3.1. Clinical Administration of IL-12 in Patients with Melanoma
3.2. IL-12 Therapy in Combination with ICI
3.3. IL-12 Therapy in Combination with Other Cytokines
| Year | Reagents | Treatments | Objects | Results | Reference |
|---|---|---|---|---|---|
| 2015 | IL-18 | IL-12 with IL-18 | PBMC from patients with melanoma | NK cell activity↑ | [48] |
| 2015 | IL-18 | Linear DNA encoding IL-12/ IL-18 injection | Grey Horse | Total leukocyte and neutrophil counts↑, lymphocyte numbers↓ | [50] |
| 2016 | IL-2 | IL-12 with IL-2 | PBMC from patients with melanoma | IL-2Rα and IL-12R expression↑, NK cell activity↑ | [46] |
| 2017 | IL-2 | IL-12 with IL-2 | PBMC from patients with melanoma | CD4+CD25brightCD27+ Treg cells↓ | [25] |
| 2018 | TNF-α | IL-12 and TNF-α co-expression in situ vaccination | Murine B16F10 melanoma model | Tumor growth↓, local effectiveness from 80 to 100% | [55] |
| 2021 | GM-CSF | IL-12 and GM-CSF co-expression | Murine B16F10 melanoma model | Tumor growth↓, CD4+ and CD8+ T cell recruitment↑ | [52] |
| 2022 | CXCL9 | IL-12 plasmid with CXCL9 plasmid | Murine B16F10 melanoma model | Tumor growth↓, DC licensing↑, CD8+ T cell↑ | [54] |
| 2022 | IL-27 GM-CSF | LNP encapsulated with mRNAs encoding cytokines including IL-12, IL-27 and GM-CSF | Murine B16F10 melanoma model | Tumor growth↓, NK and CD8+ T cells↑ | [51] |
3.4. IL-12 in Combination with Other Therapeutic Reagents
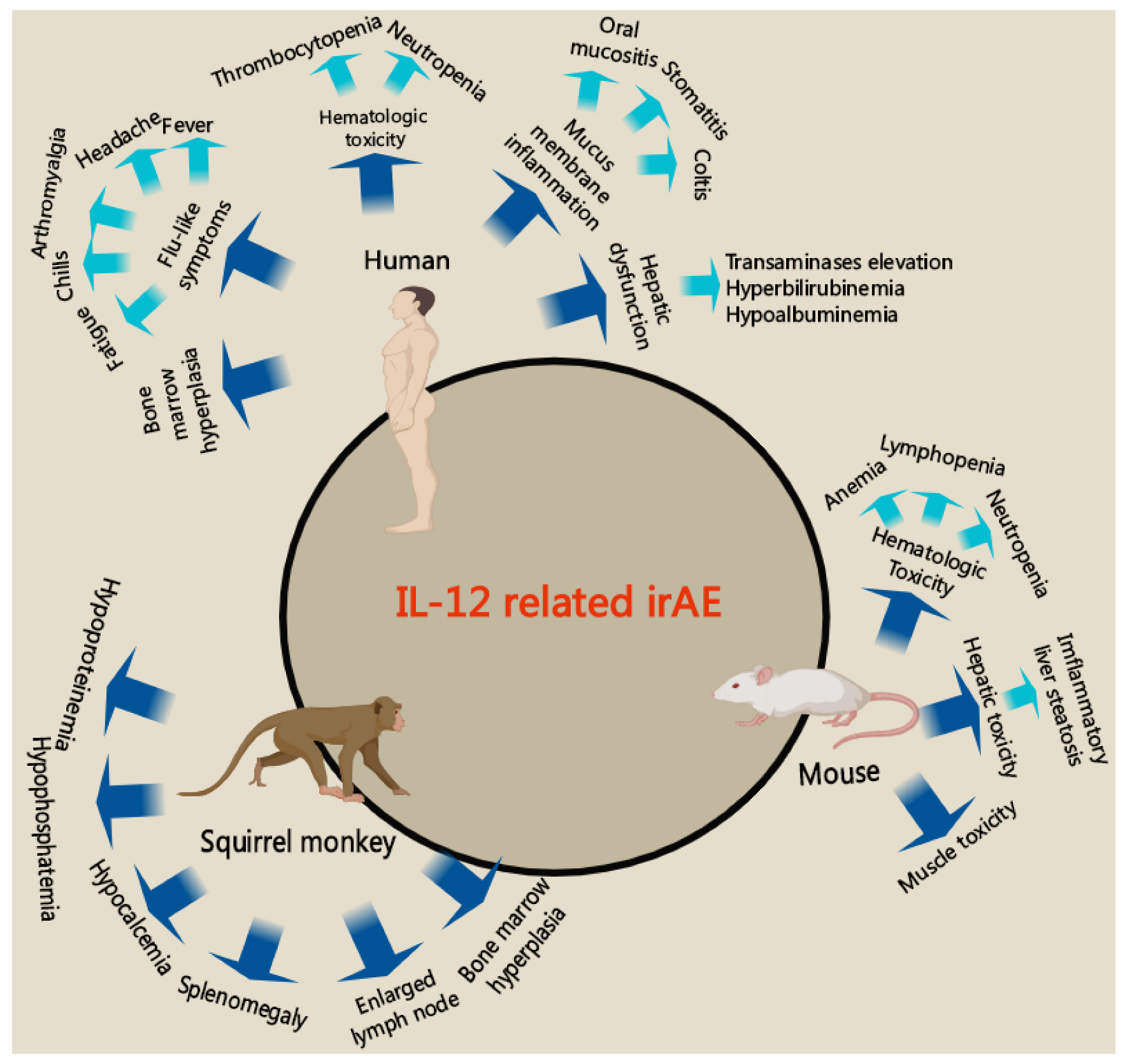
4. Strategies to Reduce the Immune-Related Side Effects of IL-12 Therapy
4.1. Immune-Related Side Effects of IL-12 Therapy
4.2. Strategies to Improve the Effectiveness and Safety
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Gray-Schopfer, V.; Wellbrock, C.; Marais, R. Melanoma biology and new targeted therapy. Nature 2007, 445, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Killock, D. Braf+meki and ici triplets show promise in melanoma. Nat. Rev. Clin. Oncol. 2019, 16, 525. [Google Scholar] [CrossRef]
- Tetu, P.; Baroudjian, B.; Lebbe, C. Targeting braf and mek inhibitors in melanoma in the metastatic, neoadjuvant and adjuvant setting. Curr. Opin. Oncol. 2020, 32, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Stachyra-Strawa, P.; Ciesielka, M.; Janiszewski, M.; Grzybowska-Szatkowska, L. The role of immunotherapy and moleculartargeted therapy in the treatment of melanoma (review). Oncol. Rep. 2021, 46, 158. [Google Scholar] [CrossRef] [PubMed]
- Gentzler, R.; Hall, R.; Kunk, P.R.; Gaughan, E.; Dillon, P.; Slingluff, C.L., Jr.; Rahma, O.E. Beyond melanoma: Inhibiting the pd-1/pd-l1 pathway in solid tumors. Immunotherapy 2016, 8, 583–600. [Google Scholar] [CrossRef]
- Bagchi, S.; Yuan, R.; Engleman, E.G. Immune checkpoint inhibitors for the treatment of cancer: Clinical impact and mechanisms of response and resistance. Annu. Rev. Pathol. 2021, 16, 223–249. [Google Scholar] [CrossRef]
- Han, D.; Pottin-Clemenceau, C.; Imro, M.A.; Scudeletti, M.; Doucet, C.; Puppo, F.; Brouty-Boye, D.; Vedrenne, J.; Sahraoui, Y.; Brailly, H.; et al. Il2 triggers a tumor progression process in a melanoma cell line melp derived from a patient whose metastasis increased in size during il2/infalpha biotherapy. Oncogene 1996, 12, 1015–1023. [Google Scholar]
- Chae, Y.K.; Chang, S.; Ko, T.; Anker, J.; Agte, S.; Iams, W.; Choi, W.M.; Lee, K.; Cruz, M. Epithelial-mesenchymal transition (emt) signature is inversely associated with t-cell infiltration in non-small cell lung cancer (nsclc). Sci. Rep. 2018, 8, 2918. [Google Scholar] [CrossRef]
- Charlet, A.; Kappenstein, M.; Keye, P.; Klasener, K.; Endres, C.; Poggio, T.; Gorantla, S.P.; Kreutmair, S.; Sanger, J.; Illert, A.L.; et al. The il-3, il-5, and gm-csf common receptor beta chain mediates oncogenic activity of flt3-itd-positive aml. Leukemia 2022, 36, 701–711. [Google Scholar] [CrossRef]
- Briukhovetska, D.; Dorr, J.; Endres, S.; Libby, P.; Dinarello, C.A.; Kobold, S. Interleukins in cancer: From biology to therapy. Nat. Rev. Cancer 2021, 21, 481–499. [Google Scholar] [CrossRef]
- Parihar, R.; Dierksheide, J.; Hu, Y.; Carson, W.E. Il-12 enhances the natural killer cell cytokine response to ab-coated tumor cells. J. Clin. Investig. 2002, 110, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Schoenhaut, D.S.; Chua, A.O.; Wolitzky, A.G.; Quinn, P.M.; Dwyer, C.M.; McComas, W.; Familletti, P.C.; Gately, M.K.; Gubler, U. Cloning and expression of murine il-12. J. Immunol. 1992, 148, 3433–3440. [Google Scholar] [PubMed]
- Xu, H.; Zhang, Y.; Hua, Y.; Chen, T.; Wang, H.; Wu, W. Il-12 p35 silenced dendritic cells modulate immune responses by blocking il-12 signaling through jak-stat pathway in t lymphocytes. Biochem. Biophys. Res. Commun. 2007, 353, 812–816. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, Y.; Gu, W.; Sun, B. Th1/th2 cell differentiation and molecular signals. Adv. Exp. Med. Biol. 2014, 841, 15–44. [Google Scholar] [PubMed]
- Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat. Rev. Immunol. 2003, 3, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Afkarian, M.; Sedy, J.R.; Yang, J.; Jacobson, N.G.; Cereb, N.; Yang, S.Y.; Murphy, T.L.; Murphy, K.M. T-bet is a stat1-induced regulator of il-12r expression in naïve cd4+ t cells. Nat. Immunol. 2020, 3, 549–557. [Google Scholar] [CrossRef]
- Glassman, C.R.; Mathiharan, Y.K.; Jude, K.M.; Su, L.; Panova, O.; Lupardus, P.J.; Spangler, J.B.; Ely, L.K.; Thomas, C.; Skiniotis, G.; et al. Structural basis for il-12 and il-23 receptor sharing reveals a gateway for shaping actions on t versus nk cells. Cell 2021, 184, 983–999.e924. [Google Scholar] [CrossRef]
- Vignali, D.A.; Kuchroo, V.K. Il-12 family cytokines: Immunological playmakers. Nat. Immunol. 2012, 13, 722–728. [Google Scholar] [CrossRef]
- Sun, Y.; Yang, J.; Yang, T.; Li, Y.; Zhu, R.; Hou, Y.; Liu, Y. Co-delivery of il-12 cytokine gene and cisplatin prodrug by a polymetformin-conjugated nanosystem for lung cancer chemo-gene treatment through chemotherapy sensitization and tumor microenvironment modulation. Acta Biomater. 2021, 128, 447–461. [Google Scholar] [CrossRef]
- Liu, X.; Gao, X.; Zheng, S.; Wang, B.; Li, Y.; Zhao, C.; Muftuoglu, Y.; Chen, S.; Li, Y.; Yao, H.; et al. Modified nanoparticle mediated il-12 immunogene therapy for colon cancer. Nanomedicine 2017, 13, 1993–2004. [Google Scholar] [CrossRef]
- Huang, J.; Liu, Y.; Au, B.C.; Barber, D.L.; Arruda, A.; Schambach, A.; Rothe, M.; Minden, M.D.; Paige, C.J.; Medin, J.A. Preclinical validation: Lv/il-12 transduction of patient leukemia cells for immunotherapy of aml. Mol. Ther. Methods Clin. Dev. 2016, 3, 16074. [Google Scholar] [CrossRef] [PubMed]
- Mittal, D.; Vijayan, D.; Putz, E.M.; Aguilera, A.R.; Markey, K.A.; Straube, J.; Kazakoff, S.; Nutt, S.L.; Takeda, K.; Hill, G.R.; et al. Interleukin-12 from cd103(+) batf3-dependent dendritic cells required for nk-cell suppression of metastasis. Cancer Immunol. Res. 2017, 5, 1098–1108. [Google Scholar] [CrossRef] [PubMed]
- Hwang, M.P.; Fecek, R.J.; Qin, T.; Storkus, W.J.; Wang, Y. Single injection of il-12 coacervate as an effective therapy against b16-f10 melanoma in mice. J. Control Release Off. J. Control Release Soc. 2020, 318, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Rayman, P.; Pavicic, P.G., Jr.; Tannenbaum, C.; Hamilton, T.; Montero, A.; Ko, J.; Gastman, B.; Finke, J.; Ernstoff, M.; et al. Ex vivo conditioning with il-12 protects tumor-infiltrating cd8(+) t cells from negative regulation by local ifn-gamma. Cancer Immunol. Immunother. 2019, 68, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Mirjacic Martinovic, K.M.; Vuletic, A.M.; Lj Babovic, N.; Dzodic, R.R.; Konjevic, G.M.; Jurisic, V.B. Attenuated in vitro effects of ifn-alpha, il-2 and il-12 on functional and receptor characteristics of peripheral blood lymphocytes in metastatic melanoma patients. Cytokine 2017, 96, 30–40. [Google Scholar] [CrossRef]
- Hinrichs, C.S.; Rosenberg, S.A. Exploiting the curative potential of adoptive t-cell therapy for cancer. Immunol. Rev. 2014, 257, 56–71. [Google Scholar] [CrossRef]
- Tucker, C.G.; Mitchell, J.S.; Martinov, T.; Burbach, B.J.; Beura, L.K.; Wilson, J.C.; Dwyer, A.J.; Singh, L.M.; Mescher, M.F.; Fife, B.T. Adoptive t cell therapy with il-12-preconditioned low-avidity t cells prevents exhaustion and results in enhanced t cell activation, enhanced tumor clearance, and decreased risk for autoimmunity. J. Immunol. 2020, 205, 1449–1460. [Google Scholar] [CrossRef]
- Shenoy, G.N.; Greene, C.J.; Bhatta, M.; Baroja, M.L.; Loyall, J.L.; Balu-Iyer, S.V.; Kelleher, R.J., Jr.; Carreno, B.M.; Linette, G.P.; Shultz, L.D.; et al. Preclinical evaluation of cancer immune therapy using patient-derived tumor antigen-specific t cells in a novel xenograft platform. Clin. Transl. Immunol. 2021, 10, e1246. [Google Scholar] [CrossRef]
- Chalmers, Z.R.; Connelly, C.F.; Fabrizio, D.; Gay, L.; Ali, S.M.; Ennis, R.; Schrock, A.; Campbell, B.; Shlien, A.; Chmielecki, J.; et al. Analysis of 100,000 human cancer genomes reveals the landscape of tumor mutational burden. Genome Med. 2017, 9, 34. [Google Scholar] [CrossRef]
- Tugues, S.; Burkhard, S.H.; Ohs, I.; Vrohlings, M.; Nussbaum, K.; Vom Berg, J.; Kulig, P.; Becher, B. New insights into il-12-mediated tumor suppression. Cell Death Differ. 2015, 22, 237–246. [Google Scholar] [CrossRef]
- Carreno, B.M.; Becker-Hapak, M.; Huang, A.; Chan, M.; Alyasiry, A.; Lie, W.R.; Aft, R.L.; Cornelius, L.A.; Trinkaus, K.M.; Linette, G.P. Il-12p70-producing patient dc vaccine elicits tc1-polarized immunity. J. Clin. Investig. 2013, 123, 3383–3394. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Morgan, R.A.; Beane, J.D.; Zheng, Z.; Dudley, M.E.; Kassim, S.H.; Nahvi, A.V.; Ngo, L.T.; Sherry, R.M.; Phan, G.Q.; et al. Tumor-infiltrating lymphocytes genetically engineered with an inducible gene encoding interleukin-12 for the immunotherapy of metastatic melanoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2015, 21, 2278–2288. [Google Scholar] [CrossRef] [PubMed]
- Cha, E.; Daud, A. Plasmid il-12 electroporation in melanoma. Hum. Vaccines Immunother. 2012, 8, 1734–1738. [Google Scholar] [CrossRef] [PubMed]
- Algazi, A.; Bhatia, S.; Agarwala, S.; Molina, M.; Lewis, K.; Faries, M.; Fong, L.; Levine, L.P.; Franco, M.; Oglesby, A.; et al. Intratumoral delivery of tavokinogene telseplasmid yields systemic immune responses in metastatic melanoma patients. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2020, 31, 532–540. [Google Scholar] [CrossRef]
- Greaney, S.K.; Algazi, A.P.; Tsai, K.K.; Takamura, K.T.; Chen, L.; Twitty, C.G.; Zhang, L.; Paciorek, A.; Pierce, R.H.; Le, M.H.; et al. Intratumoral plasmid il12 electroporation therapy in patients with advanced melanoma induces systemic and intratumoral t-cell responses. Cancer Immunol. Res. 2020, 8, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Algazi, A.P.; Twitty, C.G.; Tsai, K.K.; Le, M.; Pierce, R.; Browning, E.; Hermiz, R.; Canton, D.A.; Bannavong, D.; Oglesby, A.; et al. Phase ii trial of il-12 plasmid transfection and pd-1 blockade in immunologically quiescent melanoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2020, 26, 2827–2837. [Google Scholar] [CrossRef]
- Mansurov, A.; Ishihara, J.; Hosseinchi, P.; Potin, L.; Marchell, T.M.; Ishihara, A.; Williford, J.M.; Alpar, A.T.; Raczy, M.M.; Gray, L.T.; et al. Collagen-binding il-12 enhances tumour inflammation and drives the complete remission of established immunologically cold mouse tumours. Nat. Biomed. Eng. 2020, 4, 531–543. [Google Scholar] [CrossRef]
- Abusarah, J.; Khodayarian, F.; El-Hachem, N.; Salame, N.; Olivier, M.; Balood, M.; Roversi, K.; Talbot, S.; Bikorimana, J.P.; Chen, J.; et al. Engineering immunoproteasome-expressing mesenchymal stromal cells: A potent cellular vaccine for lymphoma and melanoma in mice. Cell Rep. Med. 2021, 2, 100455. [Google Scholar] [CrossRef]
- Kos, S.; Lopes, A.; Preat, V.; Cemazar, M.; Lampreht Tratar, U.; Ucakar, B.; Vanvarenberg, K.; Sersa, G.; Vandermeulen, G. Intradermal DNA vaccination combined with dual ctla-4 and pd-1 blockade provides robust tumor immunity in murine melanoma. PLoS ONE 2019, 14, e0217762. [Google Scholar] [CrossRef]
- Hong, H.; Wang, X.; Song, X.; Fawal, G.E.; Wang, K.; Jiang, D.; Pei, Y.; Wang, Z.; Wang, H. Transdermal delivery of interleukin-12 gene targeting dendritic cells enhances the anti-tumour effect of programmed cell death protein 1 monoclonal antibody. Biomater. Transl. 2021, 2, 151–164. [Google Scholar]
- Hewitt, S.L.; Bailey, D.; Zielinski, J.; Apte, A.; Musenge, F.; Karp, R.; Burke, S.; Garcon, F.; Mishra, A.; Gurumurthy, S.; et al. Intratumoral il12 mrna therapy promotes th1 transformation of the tumor microenvironment. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2020, 26, 6284–6298. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, A.; Naka, T.; Kubo, M. Socs proteins, cytokine signalling and immune regulation. Nat. Rev. Immunol. 2007, 7, 454–465. [Google Scholar] [CrossRef]
- Weiss, J.M.; Subleski, J.J.; Wigginton, J.M.; Wiltrout, R.H. Immunotherapy of cancer by il-12-based cytokine combinations. Expert Opin. Biol. Ther. 2007, 7, 1705–1721. [Google Scholar] [CrossRef]
- Mullard, A. Restoring il-2 to its cancer immunotherapy glory. Nat. Rev. Drug Discov. 2021, 20, 163–165. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A. Il-2: The first effective immunotherapy for human cancer. J. Immunol. 2014, 192, 5451–5458. [Google Scholar] [CrossRef] [PubMed]
- Mirjacic Martinovic, K.M.; Babovic, N.L.; Dzodic, R.R.; Jurisic, V.B.; Ninkovic, A.Z.; Konjevic, G.M. Beneficial in-vitro effects of interleukin-2, interleukin-12, and their combination on functional and receptor characteristics of natural killer cells in metastatic melanoma patients with normal serum lactate dehydrogenase levels. Melanoma Res. 2016, 26, 551–564. [Google Scholar] [CrossRef]
- Komel, T.; Bosnjak, M.; Kranjc Brezar, S.; De Robertis, M.; Mastrodonato, M.; Scillitani, G.; Pesole, G.; Signori, E.; Sersa, G.; Cemazar, M. Gene electrotransfer of il-2 and il-12 plasmids effectively eradicated murine b16.F10 melanoma. Bioelectrochemistry 2021, 141, 107843. [Google Scholar] [CrossRef]
- Mirjacic Martinovic, K.; Babovic, N.; Dzodic, R.; Jurisic, V.; Matkovic, S.; Konjevic, G. Favorable in vitro effects of combined il-12 and il-18 treatment on nk cell cytotoxicity and cd25 receptor expression in metastatic melanoma patients. J. Transl. Med. 2015, 13, 120. [Google Scholar] [CrossRef]
- Schnabel, C.L.; Steinig, P.; Koy, M.; Schuberth, H.J.; Juhls, C.; Oswald, D.; Wittig, B.; Willenbrock, S.; Murua Escobar, H.; Pfarrer, C.; et al. Immune response of healthy horses to DNA constructs formulated with a cationic lipid transfection reagent. BMC Vet. Res. 2015, 11, 140. [Google Scholar] [CrossRef]
- Mahlmann, K.; Feige, K.; Juhls, C.; Endmann, A.; Schuberth, H.J.; Oswald, D.; Hellige, M.; Doherr, M.; Cavalleri, J.M. Local and systemic effect of transfection-reagent formulated DNA vectors on equine melanoma. BMC Vet. Res. 2015, 11, 132. [Google Scholar] [CrossRef]
- Liu, J.-Q.; Zhang, C.; Zhang, X.; Yan, J.; Zeng, C.; Talebian, F.; Lynch, K.; Zhao, W.; Hou, X.; Du, S.; et al. Intratumoral delivery of il-12 and il-27 mrna using lipid nanoparticles for cancer immunotherapy. J. Control Release 2022, 345, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Moon, D.; Kong, S.J.; Lee, Y.S.; Yoo, Y.; Kim, S.; Kim, C.; Chon, H.J.; Kim, J.H.; Choi, K.J. Antitumor effects of il-12 and gm-csf co-expressed in an engineered oncolytic hsv-1. Gene Ther. 2021, 28, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Ji, Y.; Gattinoni, L.; Zhang, L.; Yu, Z.; Restifo, N.P.; Rosenberg, S.A.; Morgan, R.A. Modulating the differentiation status of ex vivo-cultured anti-tumor t cells using cytokine cocktails. Cancer Immunol. Immunother. 2013, 62, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Nguyen, B.; Mukhopadhyay, A.; Han, M.; Zhang, J.; Gujar, R.; Salazar, J.; Hermiz, R.; Svenson, L.; Browning, E.; et al. Amplification of the cxcr3/cxcl9 axis via intratumoral electroporation of plasmid cxcl9 synergizes with plasmid il-12 therapy to elicit robust anti-tumor immunity. Mol. Ther. Oncolytics 2022, 25, 174–188. [Google Scholar] [CrossRef]
- Kamensek, U.; Cemazar, M.; Lampreht Tratar, U.; Ursic, K.; Sersa, G. Antitumor in situ vaccination effect of tnfalpha and il-12 plasmid DNA electrotransfer in a murine melanoma model. Cancer Immunol. Immunother. 2018, 67, 785–795. [Google Scholar] [CrossRef]
- Dorr, R.T.; Meyers, R.; Snead, K.; Liddil, J.D. Analytical and biological inequivalence of two commercial formulations of the antitumor agent bleomycin. Cancer Chemother. Pharmacol. 1998, 42, 149–154. [Google Scholar] [CrossRef]
- Locke, F.; Clark, J.I.; Gajewski, T.F. A phase ii study of oxaliplatin, docetaxel, and gm-csf in patients with previously treated advanced melanoma. Cancer Chemother. Pharmacol. 2010, 65, 509–514. [Google Scholar] [CrossRef]
- Hoffmann, R.; Muller, I.; Neuber, K.; Lassmann, S.; Buer, J.; Probst, M.; Oevermann, K.; Franzke, A.; Kirchner, H.; Ganser, A.; et al. Risk and outcome in metastatic malignant melanoma patients receiving dtic, cisplatin, bcnu and tamoxifen followed by immunotherapy with interleukin 2 and interferon alpha2a. Br. J. Cancer 1998, 78, 1076–1080. [Google Scholar] [CrossRef][Green Version]
- Ursic, K.; Kos, S.; Kamensek, U.; Cemazar, M.; Miceska, S.; Markelc, B.; Bucek, S.; Staresinic, B.; Prevodnik, V.K.; Hellerg, R.; et al. Potentiation of electrochemotherapy effectiveness by immunostimulation with il-12 gene electrotransfer in mice is dependent on tumor immune status. J. Control Release 2021, 332, 623–635. [Google Scholar] [CrossRef]
- Milevoj, N.; Tratar, U.L.; Nemec, A.; Brozic, A.; Znidar, K.; Sersa, G.; Cemazar, M.; Tozon, N. A combination of electrochemotherapy, gene electrotransfer of plasmid encoding canine il-12 and cytoreductive surgery in the treatment of canine oral malignant melanoma. Res. Vet. Sci. 2019, 122, 40–49. [Google Scholar] [CrossRef]
- Ott, P.A.; Hodi, F.S.; Buchbinder, E.I. Inhibition of immune checkpoints and vascular endothelial growth factor as combination therapy for metastatic melanoma: An overview of rationale, preclinical evidence, and initial clinical data. Front. Oncol. 2015, 5, 202. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.M.; Hong, J.; Yun, C.O. Oncolytic adenovirus coexpressing interleukin-12 and shvegf restores antitumor immune function and enhances antitumor efficacy. Oncotarget 2016, 7, 84965–84980. [Google Scholar] [CrossRef]
- Chinnasamy, D.; Yu, Z.; Kerkar, S.P.; Zhang, L.; Morgan, R.A.; Restifo, N.P.; Rosenberg, S.A. Local delivery of interleukin-12 using t cells targeting vegf receptor-2 eradicates multiple vascularized tumors in mice. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 1672–1683. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Kang, S.J.; Jeong, H.Y.; Kim, M.W.; Park, S.I.; Lee, Y.K.; Kim, H.S.; Kim, K.S.; Park, Y.S. Anti-egfr immunonanoparticles containing il12 and salmosin genes for targeted cancer gene therapy. Int. J. Oncol. 2016, 49, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Nguyen, B.; Lee, J.Y.; Browning, E.; Zhang, J.; Mukhopadhyay, A.; Gujar, R.; Salazar, J.; Hermiz, R.; Svenson, L.; et al. Intratumoral electroporation of plasmid encoded il12 and membrane-anchored anti-cd3 increases systemic tumor immunity. Mol. Cancer Res. 2022, 20, 983–995. [Google Scholar] [CrossRef]
- Jiang, J.; Zhang, Y.; Peng, K.; Wang, Q.; Hong, X.; Li, H.; Fan, G.; Zhang, Z.; Gong, T.; Sun, X. Combined delivery of a tgf-β inhibitor and an adenoviral vector expressing interleukin-12 potentiates cancer immunotherapy. Acta Biomater. 2017, 1, 114–123. [Google Scholar] [CrossRef]
- Leleux, J.A.; Albershardt, T.C.; Reeves, R.; James, R.; Krull, J.; Parsons, A.J.; Ter Meulen, J.; Berglund, P. Intratumoral expression of il-12 from lentiviral or rna vectors acts synergistically with tlr4 agonist (gla) to generate anti-tumor immunological memory. PLoS ONE 2021, 16, e0259301. [Google Scholar] [CrossRef]
- Remic, T.; Sersa, G.; Ursic, K.; Cemazar, M.; Kamensek, U. Development of tumor cell-based vaccine with il-12 gene electrotransfer as adjuvant. Vaccines 2020, 8, 111. [Google Scholar] [CrossRef]
- Atkins, M.B.; Robertson, M.J.; Gordon, M.; Lotze, M.T.; DeCoste, M.; DuBois, J.S.; Ritz, J.; Sandler, A.B.; Edington, H.D.; Garzone, P.D.; et al. Phase i evaluation of intravenous recombinant human interleukin 12 in patients with advanced malignancies. Clin. Cancer Res. 1997, 3, 409–417. [Google Scholar]
- Halin, C.; Rondini, S.; Nilsson, F.; Berndt, A.; Kosmehl, H.; Zardi, L.; Neri, D. Enhancement of the antitumor activity of interleukin-12 by targeted delivery to neovasculature. Nat. Biotechnol. 2002, 20, 264–269. [Google Scholar] [CrossRef]
- Kaneda, M.; Kashiwamura, S.; Ueda, H.; Sawada, K.; Sugihara, A.; Terada, N.; Kimura-Shimmyo, A.; Fukuda, Y.; Shimoyama, T.; Okamura, H. Inflammatory liver steatosis caused by il-12 and il-18. J. Interf. Cytokine Res. 2003, 23, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Lasek, W.; Zagozdzon, R.; Jakobisiak, M. Interleukin 12: Still a promising candidate for tumor immunotherapy? Cancer Immunol. Immunother. 2014, 63, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Kamensek, U.; Tesic, N.; Sersa, G.; Cemazar, M. Clinically usable interleukin 12 plasmid without an antibiotic resistance gene: Functionality and toxicity study in murine melanoma model. Cancers 2018, 10, 60. [Google Scholar] [CrossRef] [PubMed]
- Mansurov, A.; Hosseinchi, P.; Chang, K.; Lauterbach, A.L.; Gray, L.T.; Alpar, A.T.; Budina, E.; Slezak, A.J.; Kang, S.; Cao, S.; et al. Masking the immunotoxicity of interleukin-12 by fusing it with a domain of its receptor via a tumour-protease-cleavable linker. Nat. Biomed. Eng. 2022, 6, 819–829. [Google Scholar] [CrossRef]
- Burkart, C.; Mukhopadhyay, A.; Shirley, S.A.; Connolly, R.J.; Wright, J.H.; Bahrami, A.; Campbell, J.S.; Pierce, R.H.; Canton, D.A. Improving therapeutic efficacy of il-12 intratumoral gene electrotransfer through novel plasmid design and modified parameters. Gene Ther. 2018, 25, 93–103. [Google Scholar] [CrossRef]
- Galvan, D.L.; O’Neil, R.T.; Foster, A.E.; Huye, L.; Bear, A.; Rooney, C.M.; Wilson, M.H. Anti-tumor effects after adoptive transfer of il-12 transposon-modified murine splenocytes in the ot-i-melanoma mouse model. PLoS ONE 2015, 10, e0140744. [Google Scholar] [CrossRef]
- Zhang, L.; Davies, J.S.; Serna, C.; Yu, Z.; Restifo, N.P.; Rosenberg, S.A.; Morgan, R.A.; Hinrichs, C.S. Enhanced efficacy and limited systemic cytokine exposure with membrane-anchored interleukin-12 t-cell therapy in murine tumor models. J. Immunother. Cancer 2020, 8, e000210. [Google Scholar] [CrossRef]
- Hu, J.; Yang, Q.; Zhang, W.; Du, H.; Chen, Y.; Zhao, Q.; Dao, L.; Xia, X.; Natalie Wall, F.; Zhang, Z.; et al. Cell membrane-anchored and tumor-targeted il-12 (attil12)-t cell therapy for eliminating large and heterogeneous solid tumors. J. Immunother. Cancer 2022, 10, e003633. [Google Scholar] [CrossRef]
- Viano, M.E.; Baez, N.S.; Reynolds, D.; Matellon, M.; Young, H.A.; Rodriguez-Galan, M.C. Safety levels of systemic il-12 induced by cdna expression as a cancer therapeutic. Immunotherapy 2022, 14, 115–133. [Google Scholar]
- Qian, Y.; Qiao, S.; Dai, Y.; Xu, G.; Dai, B.; Lu, L.; Yu, X.; Luo, Q.; Zhang, Z. Molecular-targeted immunotherapeutic strategy for melanoma via dual-targeting nanoparticles delivering small interfering rna to tumor-associated macrophages. ACS Nano 2017, 11, 9536–9549. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Q.; Wu, C.; Wang, J.; Su, M.; Deng, J. Hypoxia-responsive nanogel as il-12 carrier for anti-cancer therapy. Nanotechnology 2021, 32, 095107. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Zhu, X.; Wu, L.; Qiu, L. Cationic polyphosphazene vesicles for cancer immunotherapy by efficient in vivo cytokine il-12 plasmid delivery. Biomacromolecules 2016, 17, 2199–2209. [Google Scholar] [CrossRef] [PubMed]
- Kulach, N.; Pilny, E.; Cichon, T.; Czapla, J.; Jarosz-Biej, M.; Rusin, M.; Drzyzga, A.; Matuszczak, S.; Szala, S.; Smolarczyk, R. Mesenchymal stromal cells as carriers of il-12 reduce primary and metastatic tumors of murine melanoma. Sci. Rep. 2021, 11, 18335. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Li, Y.; Zeng, L.; Zhang, X.; Zhou, Z.; Zheng, M.; Wan, H. Intratumoral injection of dendritic cells overexpressing interleukin12 inhibits melanoma growth. Oncol. Rep. 2019, 42, 370–376. [Google Scholar]
| Year | NCT Number/ Study Phase | Treatment | Patients Number | Outcomes | Reference |
|---|---|---|---|---|---|
| 2013 | NCT00683670 Phase I | Vaccination with CD40L/IFN-γ–matured, IL-12p70–producing DCs | 7 | One CR, two PR, one SD in melanoma patients | [31] |
| 2015 | NCI 1-C-011 Phase I | Autologous TIL transduced with a gene encoding a single chain IL-12 driven by a nuclear factor of activated T cells promoter (NFAT.IL12) | 33 | One CR, nine PR in melanoma patients | [32] |
| 2020 | NCT 01502293 Phase II | Intratumorally with plasmid encoding IL-12 (tavokinogene telseplasmid; tavo), 0.5 mg/mL followed by electroporation (six pulses, 1500 V/cm) | 30 | Five CR, seven PD in melanoma patients | [34] |
| 2020 | NCT02493361 Phase II | Tavo was administered intratumorally days 1, 5, and 8 every 6 weeks while pembrolizumab (200 mg, i.v.) was administered every 3 weeks. | 23 | Nine CR, two PR, three SD, nine PD in melanoma patients | [36] |
| Year | Reagents | Treatments | Objects | Results | Reference |
|---|---|---|---|---|---|
| 2019 | Anti- PD-1 anti-CTLA-4 | IL-12 plasmid intradermal injection with anti- PD-1 or anti-CTLA-4 | Murine B16F10 melanoma model | Tumor growth↓ | [39] |
| 2020 | Pembrolizumab | Tavo encoding IL-12 with pembrolizumab | Patients with melanoma | Immune infiltration↑, ORR 48% (11/23) | [36] |
| 2020 | Anti-PD-L1 | Modified CD8+ T cells with mIL-12 mRNA with anti-PD-L1 | Murine B16F10 melanoma model | Anti-tumor immunity↑, tumor growth↓, un-injected distal lesions↓ | [41] |
| 2020 | Anti-PD-1 anti-CTLA-4 | A collagen-binding domain fused to IL-12 with anti- PD-1 or anti-CTLA-4 | Murine B16F10 melanoma model | Tumor growth↓, antigen-specific immunological memory↑ | [37] |
| Year | Reagents | Treatments | Objects | Results | Reference |
|---|---|---|---|---|---|
| 1998 | Cisplatin | IL-12 plasmid with cisplatin | Murine B16F10 melanoma | No significant improvement | [58] |
| 2012 | VEGFR-2 | T cells cotransduced with an anti-VEGFR- 2 CAR and a constitutively expressed single-chain murine IL-12 or an inducible IL-12 gene after host lymphodepletion | Murine B16F10 melanoma model | Tumor growth↓ CD11b+Gr1+ cells↓ | [63] |
| 2016 | shVEGF | Oncolytic adenovirus co-expressing IL-12 and VEGF-shRNA | Murine B16F10 melanoma model | Tumor growth↓ CD4+, CD8+ T cells↑, NK cells and DCs↑ | [62] |
| 2017 | TGF-β inhibitor | IL-12 plasmid and SB-505124 | Murine B16F10 melanoma model | Tumor growth↓ CD4+, CD8+ T and NK cells↑ | [66] |
| 2021 | GLA formulated in a stable emulsion | IL-12 plasmid and GLA-SE | Murine B16F10 melanoma model | Tumor growth↓, CD8+ T cells↑ | [67] |
| 2022 | Membrane- anchored anti-CD3 | Electroporation of IL-12 and membrane-anchored anti-CD3 plasmids | Patients with unresectable, stage III/IV melanomas | Restored the function of TIL isolated from a patient with melanoma actively progressing on PD-1 blockage | [65] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, W.; Pan, J.; Pan, J. Antitumor Activities of Interleukin-12 in Melanoma. Cancers 2022, 14, 5592. https://doi.org/10.3390/cancers14225592
Gao W, Pan J, Pan J. Antitumor Activities of Interleukin-12 in Melanoma. Cancers. 2022; 14(22):5592. https://doi.org/10.3390/cancers14225592
Chicago/Turabian StyleGao, Wei, Jun Pan, and Jianping Pan. 2022. "Antitumor Activities of Interleukin-12 in Melanoma" Cancers 14, no. 22: 5592. https://doi.org/10.3390/cancers14225592
APA StyleGao, W., Pan, J., & Pan, J. (2022). Antitumor Activities of Interleukin-12 in Melanoma. Cancers, 14(22), 5592. https://doi.org/10.3390/cancers14225592







