68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Recurrent Prostate Cancer: Diagnostic Performance and Association with Clinical and Histopathological Data
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. 68Ga-PSMA PET/MRI Acquisition Protocol
- an axial T2 weighted sequence with large field of view (FOV): FSE, TR = 10,235 ms; TE = 99.7 ms, FOV = 32 × 32 cm2; voxel size = 0.9 × 0.9 × 5 mm3,
- an axial T2 weighted sequence with small FOV: PROPELLER, TR = 9578 ms, TE = 151 ms, FOV = 18 × 18 cm2, voxel size = 0.6 × 0.6 × 3 mm3,
- a sagittal T2 weighted sequence with small FOV: PROPELLER, TR = 9578 ms, TE = 151 ms, FOV = 18 × 18 cm2, voxel size = 0.6 × 0.6 × 3 mm3,
- a diffusion weighted imaging (DWI) sequence with small FOV: TR = 6643 ms, TR = 79.5 ms, FOV = 18 × 18 cm2, voxel size = 1.8 × 1.8 × 3 mm3; b = 50, 800, 1400; 2000 s/mm2 and ADC maps,
- T1-Lava Flex sequence of the whole pelvic region pre-contrast and post-contrast: TR = 5 ms, TE = 1.7 ms, FOV: 44 × 35.2 cm2, voxel size = 1.3 × 1.2 × 2 mm3,
- a high temporal resolution T1 perfusion sequence after IV injection of 0.1 mmol/kg bolus of gadobutrol (Gadovist, Bayer Schering Pharma, Germany) at a flow rate of 3.5 mL/s: DISCO, TR = 5.1 ms, TE = 1.7 ms, FOV = 29 × 29 cm2, Voxel size = 1.9 × 2.2 × 3 mm 3, 88 dynamics.
2.3. 68Ga-DOTA-RM2 PET/MRI Acquisition Protocol
2.4. PET/MR Image Analysis
2.5. Lesion Validation
2.6. Statistical Analyses
3. Results
3.1. Patients
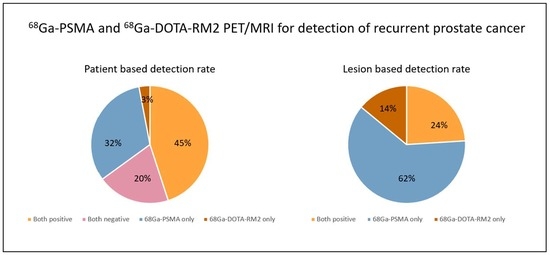
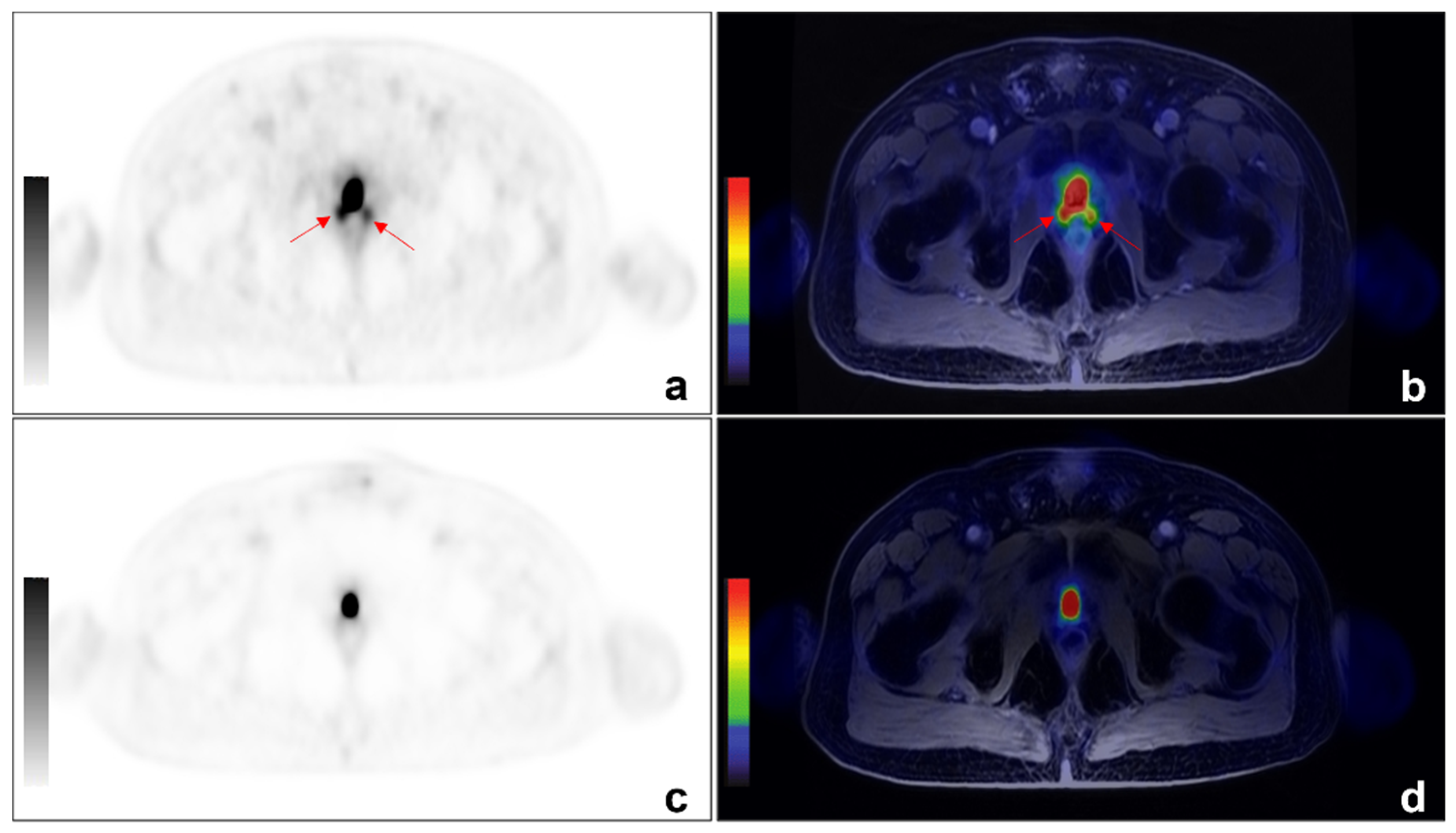
3.2. PET/MRI Findings and Comparison between 68Ga-PSMA and 68Ga-DOTA-RM2
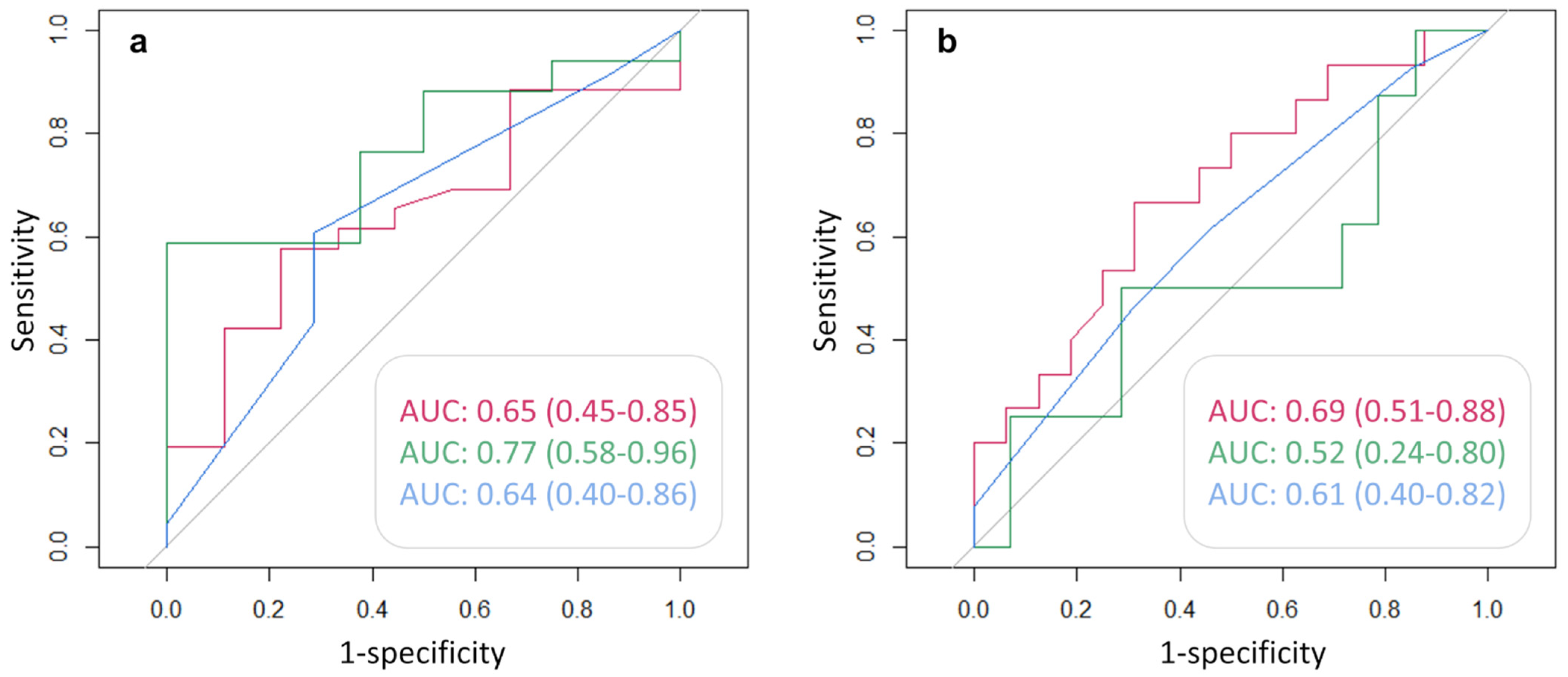
3.3. Associations between Semi-Quantitative Imaging Parameters and Clinical Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cornford, P.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer. Part II-2020 Update: Treatment of Relapsing and Metastatic Prostate Cancer. Eur. Urol. 2021, 79, 263–282. [Google Scholar] [CrossRef]
- Ploussard, G.; Almeras, C.; Briganti, A.; Giannarini, G.; Hennequin, C.; Ost, P.; Renard-Penna, R.; Salin, A.; Lebret, T.; Villers, A.; et al. Management of Node Only Recurrence after Primary Local Treatment for Prostate Cancer: A Systematic Review of the Literature. J. Urol. 2015, 194, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Strauss, D.S.; Sachpekidis, C.; Kopka, K.; Pan, L.; Haberkorn, U.; Dimitrakopoulou-Strauss, A. Pharmacokinetic studies of [68Ga]Ga-PSMA-11 in patients with biochemical recurrence of prostate cancer: Detection, differences in temporal distribution and kinetic modelling by tissue type. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 4472–4482. [Google Scholar] [CrossRef]
- Maurer, T.; Eiber, M.; Schwaiger, M.; Gschwend, J.E. Current use of PSMA-PET in prostate cancer management. Nat. Rev. Urol. 2016, 13, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Woo, S.; Kim, Y.J.; Suh, C.H. Impact of 68Ga-PSMA PET on the Management of Patients with Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. 2018, 74, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Fendler, W.P.; Ferdinandus, J.; Czernin, J.; Eiber, M.; Flavell, R.R.; Behr, S.C.; Wu, I.K.; Lawhn-Heath, C.; Pampaloni, M.H.; Reiter, R.E.; et al. Impact of 68Ga-PSMA-11 PET on the Management of Recurrent Prostate Cancer in a Prospective Single-Arm Clinical Trial. J. Nucl. Med. 2020, 61, 1793–1799. [Google Scholar] [CrossRef]
- Mansi, R.; Fleischmann, A.; Macke, H.R.; Reubi, J.C. Targeting GRPR in urological cancers—From basic research to clinical application. Nat. Rev. Urol. 2013, 10, 235–244. [Google Scholar] [CrossRef]
- Mena, E.; Lindenberg, L.M.; Choyke, P.L. New Targets for PET Molecular Imaging of Prostate Cancer. Semin. Nucl. Med. 2019, 49, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Wieser, G.; Popp, I.; Christian Rischke, H.; Drendel, V.; Grosu, A.L.; Bartholoma, M.; Weber, W.A.; Mansi, R.; Wetterauer, U.; Schultze-Seemann, W.; et al. Diagnosis of recurrent prostate cancer with PET/CT imaging using the gastrin-releasing peptide receptor antagonist 68Ga-RM2: Preliminary results in patients with negative or inconclusive [(18)F]Fluoroethylcholine-PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1463–1472. [Google Scholar] [CrossRef]
- Baratto, L.; Song, H.; Duan, H.; Hatami, N.; Bagshaw, H.; Buyyounouski, M.; Hancock, S.; Shah, S.A.; Srinivas, S.; Swift, P.; et al. PSMA- and GRPR-targeted PET: Results from 50 Patients with Biochemically Recurrent Prostate Cancer. J. Nucl. Med. 2021, 62, 1545–1549. [Google Scholar] [CrossRef]
- Iagaru, A. Will GRPR Compete with PSMA as a Target in Prostate Cancer? J. Nucl. Med. 2017, 58, 1883–1884. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mapelli, P.; Ghezzo, S.; Samanes Gajate, A.M.; Preza, E.; Brembilla, G.; Cucchiara, V.; Ahmed, N.; Bezzi, C.; Presotto, L.; Bettinardi, V.; et al. Preliminary Results of an Ongoing Prospective Clinical Trial on the Use of 68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Staging of High-Risk Prostate Cancer Patients. Diagnostics 2021, 11, 2068. [Google Scholar] [CrossRef] [PubMed]
- Fassbender, T.F.; Schiller, F.; Zamboglou, C.; Drendel, V.; Kiefer, S.; Jilg, C.A.; Grosu, A.L.; Mix, M. Voxel-based comparison of [68Ga]Ga-RM2-PET/CT and [68Ga]Ga-PSMA-11-PET/CT with histopathology for diagnosis of primary prostate cancer. EJNMMI Res. 2020, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Minamimoto, R.; Hancock, S.; Schneider, B.; Chin, F.T.; Jamali, M.; Loening, A.; Vasanawala, S.; Gambhir, S.S.; Iagaru, A. Pilot Comparison of (6)(8)Ga-RM2 PET and (6)(8)Ga-PSMA-11 PET in Patients with Biochemically Recurrent Prostate Cancer. J. Nucl. Med. 2016, 57, 557–562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panebianco, V.; Barchetti, F.; Sciarra, A.; Musio, D.; Forte, V.; Gentile, V.; Tombolini, V.; Catalano, C. Prostate cancer recurrence after radical prostatectomy: The role of 3-T diffusion imaging in multi-parametric magnetic resonance imaging. Eur. Radiol. 2013, 23, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Park, J.J.; Kim, C.K.; Park, S.Y.; Park, B.K.; Lee, H.M.; Cho, S.W. Prostate cancer: Role of pretreatment multiparametric 3-T MRI in predicting biochemical recurrence after radical prostatectomy. AJR Am. J. Roentgenol. 2014, 202, W459–W465. [Google Scholar] [CrossRef] [PubMed]
- Roach, M., 3rd; Hanks, G.; Thames, H., Jr.; Schellhammer, P.; Shipley, W.U.; Sokol, G.H.; Sandler, H. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: Recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 965–974. [Google Scholar] [CrossRef]
- Baratto, L.; Duan, H.; Laudicella, R.; Toriihara, A.; Hatami, N.; Ferri, V.; Iagaru, A. Physiological 68Ga-RM2 uptake in patients with biochemically recurrent prostate cancer: An atlas of semi-quantitative measurements. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 115–122. [Google Scholar] [CrossRef]
- Demirci, E.; Sahin, O.E.; Ocak, M.; Akovali, B.; Nematyazar, J.; Kabasakal, L. Normal distribution pattern and physiological variants of 68Ga-PSMA-11 PET/CT imaging. Nucl. Med. Commun. 2016, 37, 1169–1179. [Google Scholar] [CrossRef]
- Draulans, C.; De Roover, R.; van der Heide, U.A.; Kerkmeijer, L.; Smeenk, R.J.; Pos, F.; Vogel, W.V.; Nagarajah, J.; Janssen, M.; Isebaert, S.; et al. Optimal 68Ga-PSMA and (18)F-PSMA PET window levelling for gross tumour volume delineation in primary prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 30 November 2021).
- Fendler, W.P.; Calais, J.; Eiber, M.; Flavell, R.R.; Mishoe, A.; Feng, F.Y.; Nguyen, H.G.; Reiter, R.E.; Rettig, M.B.; Okamoto, S.; et al. Assessment of 68Ga-PSMA-11 PET Accuracy in Localizing Recurrent Prostate Cancer: A Prospective Single-Arm Clinical Trial. JAMA Oncol. 2019, 5, 856–863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoffmann, M.A.; Buchholz, H.G.; Wieler, H.J.; Miederer, M.; Rosar, F.; Fischer, N.; Muller-Hubenthal, J.; Trampert, L.; Pektor, S.; Schreckenberger, M. PSA and PSA Kinetics Thresholds for the Presence of 68Ga-PSMA-11 PET/CT-Detectable Lesions in Patients With Biochemical Recurrent Prostate Cancer. Cancers 2020, 12, 389. [Google Scholar] [CrossRef] [Green Version]
- Afshar-Oromieh, A.; da Cunha, M.L.; Wagner, J.; Haberkorn, U.; Debus, N.; Weber, W.; Eiber, M.; Holland-Letz, T.; Rauscher, I. Performance of [68Ga]Ga-PSMA-11 PET/CT in patients with recurrent prostate cancer after prostatectomy-a multi-centre evaluation of 2533 patients. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2925–2934. [Google Scholar] [CrossRef]
- Giesel, F.L.; Knorr, K.; Spohn, F.; Will, L.; Maurer, T.; Flechsig, P.; Neels, O.; Schiller, K.; Amaral, H.; Weber, W.A.; et al. Detection Efficacy of (18)F-PSMA-1007 PET/CT in 251 Patients with Biochemical Recurrence of Prostate Cancer After Radical Prostatectomy. J. Nucl. Med. 2019, 60, 362–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kranzbuhler, B.; Nagel, H.; Becker, A.S.; Muller, J.; Huellner, M.; Stolzmann, P.; Muehlematter, U.; Guckenberger, M.; Kaufmann, P.A.; Eberli, D.; et al. Clinical performance of 68Ga-PSMA-11 PET/MRI for the detection of recurrent prostate cancer following radical prostatectomy. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.A.; Buchholz, H.G.; Wieler, H.J.; Hofner, T.; Muller-Hubenthal, J.; Trampert, L.; Schreckenberger, M. The positivity rate of 68Gallium-PSMA-11 ligand PET/CT depends on the serum PSA-value in patients with biochemical recurrence of prostate cancer. Oncotarget 2019, 10, 6124–6137. [Google Scholar] [CrossRef]
- von Eyben, F.E.; Picchio, M.; von Eyben, R.; Rhee, H.; Bauman, G. 68Ga-Labeled Prostate-specific Membrane Antigen Ligand Positron Emission Tomography/Computed Tomography for Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. Focus 2018, 4, 686–693. [Google Scholar] [CrossRef] [Green Version]
- Ceci, F.; Uprimny, C.; Nilica, B.; Geraldo, L.; Kendler, D.; Kroiss, A.; Bektic, J.; Horninger, W.; Lukas, P.; Decristoforo, C.; et al. 68Ga-PSMA PET/CT for restaging recurrent prostate cancer: Which factors are associated with PET/CT detection rate? Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1284–1294. [Google Scholar] [CrossRef] [Green Version]
- Pereira Mestre, R.; Treglia, G.; Ferrari, M.; Pascale, M.; Mazzara, C.; Azinwi, N.C.; Llado, A.; Stathis, A.; Giovanella, L.; Roggero, E. Correlation between PSA kinetics and PSMA-PET in prostate cancer restaging: A meta-analysis. Eur. J. Clin. Investig. 2019, 49, e13063. [Google Scholar] [CrossRef] [PubMed]





| Pt | Age (years) | GS | PSA (ng/mL) | Treatment | Adjuvant Therapy |
|---|---|---|---|---|---|
| 1 | 64 | 4 + 4 | 9.86 | RP | None |
| 2 | 66 | 4 + 3 | 0.53 | RP | None |
| 3 | 75 | 3 + 3 | 0.54 | RP | None |
| 4 | 78 | 4 + 5 | 0.212 | RP | RT |
| 5 | 73 | 4 + 3 | 4.75 | RP | RT |
| 6 | 77 | 4 + 4 | 0.237 | RP | None |
| 7 | 58 | 4 + 3 | 1.3 | RP | None |
| 8 | 64 | 4 + 3 | 2.5 | RP | RT |
| 9 | 79 | 4 + 5 | 2 | RP | RT; ADT |
| 10 | 49 | 4 + 5 | 0.82 | RP | None |
| 11 | 84 | NA | 3.24 | RT | None |
| 12 | 79 | 5 + 3 | 1.53 | RP | RT; ADT |
| 13 | 75 | 3 + 4 | 0.39 | RP | None |
| 14 | 78 | 4 + 5 | 0.5 | RP | RT |
| 15 | 76 | 4 + 5 | 0.41 | RP | RT |
| 16 | 67 | 4 + 4 | 2 | RP | None |
| 17 | 76 | 3 + 4 | 0.41 | RP | None |
| 18 | 81 | NA | 3.53 | RT | None |
| 19 | 71 | 4 + 3 | 4.3 | RP | RT |
| 20 | 72 | 3 + 4 | 0.22 | RP | RT |
| 21 | 77 | 2 + 4 | 1.21 | RP | None |
| 22 | 77 | 4 + 5 | 4.63 | RP | RT |
| 23 | 50 | 5 + 5 | 14.4 | RP | None |
| 24 | 69 | 4 + 5 | 0.2 | RP | None |
| 25 | 59 | 4 + 4 | 0.26 | RP | None |
| 26 | 53 | NA | 0.25 | RP | None |
| 27 | 75 | 4 + 3 | 0.27 | RP | None |
| 28 | 62 | 3 + 3 | 0.68 | RP | None |
| 29 | 70 | 5 + 4 | 2.16 | RP | None |
| 30 | 73 | 4 + 3 | 0.4 | RP | None |
| 31 | 64 | 4 + 5 | 0.23 | RP | None |
| 32 | 69 | NA | 0.37 | RP | RT |
| 33 | 74 | 5 + 4 | 0.74 | RP | RT |
| 34 | 62 | 5 + 4 | 0.34 | RP | RT |
| 35 | 60 | 4 + 3 | 0.48 | RP | None |
| Pt | 68Ga-PSMA Findings | 68Ga-DOTA-RM2 Findings | PET Findings Validation |
|---|---|---|---|
| 1 | Left perirectal lesion | Left perirectal lesion | Confirmation on conventional imaging at baseline |
| 2 | Right obturator LN; right laterocervical LN | Right obturator LN | Right obturator LN confirmed on conventional imaging at baseline |
| 3 | Negative | Negative | No evidence of disease on conventional imaging either at baseline or follow-up |
| 4 | Left humerus | NA | Decrease in PSA level greater than 50% after RT on the site of pathological 68Ga-PSMA uptake |
| 5 | Left supraclavicular LN; 2 left paraortic LNs; left iliac bone; left sacral ala | Left synchondrosis; 8 left paraortic LNs; interaortocaval LN; left retroclavicular LN; 2 right retrocrural LNs; 2 left common iliac LNs | Left iliac bone and left sacral ala confirmed on conventional imaging at baseline |
| 6 | Negative | NA | No evidence of disease on conventional imaging either at baseline or follow-up and stable level of PSA during follow-up |
| 7 | Negative | Negative | No evidence of disease on conventional imaging or 11C-choline and 68Ga-PSMA PET either at baseline or follow-up |
| 8 | Bilateral prostatic fossa (2) | Negative | Progression on follow-up 68Ga-PSMA PET studies associated with an increase in PSA level |
| 9 | Left lateral rectal wall; left common iliac LN; left paramedian presacral LN | left lateral rectal wall; left common iliac LN; left paramedian presacral LN | Left lateral rectal wall confirmed on conventional imaging at baseline |
| 10 | Right iliac ala | NA | Decrease in PSA level greater than 50% after RT on the site of pathological 68Ga-PSMA uptake |
| 11 | Right prostate lobe; right internal iliac LN; left iliac bone | Right prostate lobe | Disappearance of 68Ga-PSMA uptake on follow-up PET scans after systemic treatment associated with a decrease in PSA level greater than 50% |
| 12 | Right vesical-urethral anastomosis; 2 left laterocervical LNs; left retroclavicular LN; left dorsal LN | Right vesical-urethral anastomosis | Right vesical-urethral anastomosis confirmed on conventional imaging at baseline |
| 13 | Right vesical-urethral anastomosis; left common iliac LN | Left common iliac LN | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline |
| 14 | Negative | Negative | Evidence of disease on conventional imaging at baseline (vesical-urethral anastomosis and right iliac bone) and increase in PSA level during follow-up |
| 15 | Right vesical-urethral anastomosis | Negative | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline |
| 16 | Left pubis; right V rib | Negative | Left pubis confirmed by conventional imaging at baseline |
| 17 | Negative | NA | Evidence of disease on conventional imaging at baseline (vesical-urethral anastomosis) |
| 18 | Negative | Negative | No evidence of disease on conventional imaging either at baseline or follow-up |
| 19 | Left pulmonary hilum; left acetabulum | Left pulmonary hilum | Disappearance of 68Ga-PSMA uptake on follow-up PET scans after systemic treatment associated with a decrease in PSA level greater than 50% |
| 20 | Left retrolateral vesical-urethral anastomosis | Negative | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline |
| 21 | Left obturator LN, left III rib | Left obturator LN; left III rib | Confirmed on conventional imaging at baseline |
| 22 | Bilateral iliac LNs (2); left rectus abdominis muscle; bilateral pleura (2) | Negative | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline. Pleura confirmed on histological analysis of surgically resected specimens. |
| 23 | Multiple LNs (16); multiple skeletal lesions (27) | Left retroclavicular LN; left paraortic LN; multiple hips (6) | Multiple LN and skeletal lesions confirmed on conventional imaging at baseline |
| 24 | Right perirectal LN; right pubic bone | Negative | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline |
| 25 | Small trochanter | Negative | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline and decrease in PSA level greater than 50% after RT on the site of pathological 68Ga-PSMA uptake |
| 26 | Negative | Negative | Evidence of disease on conventional imaging at baseline (paraortic and aortocaval LN) |
| 27 | Right VIII rib | Negative | No evidence of disease on conventional imaging either at baseline or follow-up |
| 28 | D7 right hemisome | Negative | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline |
| 29 | D10 right hemisome, right X rib, D8 | D10 right hemisome | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline |
| 30 | Negative | Negative | No evidence of disease on conventional imaging either at baseline or follow-up |
| 31 | Negative | Right thigh-bone | Confirmation of 68Ga-DOTA-RM2 PET findings on conventional imaging at baseline |
| 32 | Paracaval LN | Negative | Confirmation of 68Ga-PSMA PET findings on conventional imaging at baseline |
| 33 | Right iliac LN; 2 right obturator LNs | Right iliac LN; 2 right obturator LNs | Confirmed on conventional imaging at baseline and disappearance of 68Ga-PSMA uptake on follow-up PET scans after systemic treatment associated with a decrease in PSA level greater than 50% |
| 34 | Left IX rib | Left IX rib | Confirmed on conventional imaging at baseline |
| 35 | Right obturator LN | Right obturator LN | Confirmed on conventional imaging at baseline |
| Imaging Modality | Stratification | No. of Patients | Positive Results, No. (%) | p Value | Adjusted p Value |
|---|---|---|---|---|---|
| 68Ga-PSMA PET/MRI | PSA | ||||
| <0.5 | 15 | 10 (67) | 0.339 | 0.509 | |
| 0.5–2 | 9 | 6 (67) | |||
| ≥2 | 11 | 10 (91) | |||
| PSA DT | |||||
| <6 | 9 | 9 (100) | 0.022 | 0.065 | |
| ≥6 | 16 | 8 (50) | |||
| Not available | 10 | 9 (90) | |||
| GS | |||||
| ≤3 + 4 | 6 | 4 (67) | 0.603 | 0.603 | |
| ≥4 + 3 | 24 | 19 (79) | |||
| Not available | 5 | 3 (60) | |||
| 68Ga-DOTA-RM2 PET/MRI | PSA | ||||
| <0.5 | 12 | 4 (33) | 0.390 | 0.993 | |
| 0.5–2 | 8 | 4 (50) | |||
| ≥2 | 11 | 7 (64) | |||
| PSA DT | |||||
| <6 | 9 | 4 (44) | 0.662 | 0.993 | |
| ≥6 | 13 | 4 (31) | |||
| Not available | 9 | 7 (78) | |||
| GS | |||||
| ≤3 + 4 | 5 | 2 (40) | 1 | 1 | |
| ≥4 + 3 | 21 | 11 (52) | |||
| Not available | 5 | 2 (40) |
| Imaging Modality | Stratification | Positive Lesions, No. (Average) |
|---|---|---|
| 68Ga-PSMA PET/MRI | PSA | |
| <0.5 | 12 (0.8) | |
| 0.5–2 | 14 (1.56) | |
| ≥2 | 69 (6.27) | |
| PSA DT | ||
| <6 | 15 (1.67) | |
| ≥6 | 17 (1.06) | |
| Not available | 63 (6.3) | |
| GS | ||
| ≤3 + 4 | 6 (1) | |
| ≥4 + 3 | 84 (3.5) | |
| Not available | 5 (1) | |
| 68Ga-DOTA-RM2 PET/MRI | PSA | |
| <0.5 | 4 (0.33) | |
| 0.5–2 | 7 (0.88) | |
| ≥2 | 30 (2.73) | |
| PSA DT | ||
| <6 | 4 (0.44) | |
| ≥6 | 4 (0.31) | |
| Not available | 33 (3.66) | |
| GS | ||
| ≤3 + 4 | 3 (0.6) | |
| ≥4 + 3 | 36 (1.71) | |
| Not available | 2 (0.4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mapelli, P.; Ghezzo, S.; Samanes Gajate, A.M.; Preza, E.; Palmisano, A.; Cucchiara, V.; Brembilla, G.; Bezzi, C.; Rigamonti, R.; Magnani, P.; et al. 68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Recurrent Prostate Cancer: Diagnostic Performance and Association with Clinical and Histopathological Data. Cancers 2022, 14, 334. https://doi.org/10.3390/cancers14020334
Mapelli P, Ghezzo S, Samanes Gajate AM, Preza E, Palmisano A, Cucchiara V, Brembilla G, Bezzi C, Rigamonti R, Magnani P, et al. 68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Recurrent Prostate Cancer: Diagnostic Performance and Association with Clinical and Histopathological Data. Cancers. 2022; 14(2):334. https://doi.org/10.3390/cancers14020334
Chicago/Turabian StyleMapelli, Paola, Samuele Ghezzo, Ana Maria Samanes Gajate, Erik Preza, Anna Palmisano, Vito Cucchiara, Giorgio Brembilla, Carolina Bezzi, Riccardo Rigamonti, Patrizia Magnani, and et al. 2022. "68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Recurrent Prostate Cancer: Diagnostic Performance and Association with Clinical and Histopathological Data" Cancers 14, no. 2: 334. https://doi.org/10.3390/cancers14020334
APA StyleMapelli, P., Ghezzo, S., Samanes Gajate, A. M., Preza, E., Palmisano, A., Cucchiara, V., Brembilla, G., Bezzi, C., Rigamonti, R., Magnani, P., Toninelli, E., Bettinardi, V., Suardi, N., Gianolli, L., Scifo, P., Briganti, A., De Cobelli, F., Esposito, A., & Picchio, M. (2022). 68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Recurrent Prostate Cancer: Diagnostic Performance and Association with Clinical and Histopathological Data. Cancers, 14(2), 334. https://doi.org/10.3390/cancers14020334