Multidisciplinary Tumor Board in the Management of Patients with Colorectal Liver Metastases: A Single-Center Review of 847 Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Protocol
2.2. Definitions and Follow-Up Protocol
2.3. Multidisciplinary Tumor Board
2.4. Data Presentation and Study Endpoints
2.5. Statistical Analysis
3. Results
3.1. Patient Baseline Characteristics
3.2. Patient Allocation Based on the MDT Decision
3.3. Survival Analysis and Risk Factors for Overall Survival
3.4. Systemic versus Locoregional Therapy: Propensity-Score Matching
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Number of Patients | 250 | ||
|---|---|---|---|
| SEX = male (%) | 171 (68.4) | N stage of primary tumor (%) | |
| Age (median [IQR]) | 63.00 [54.00, 71.75] | 0 | 48 (19.2) |
| ECOG-PS (%) | 1 | 60 (24.0) | |
| 0 | 154 (61.6) | 2 | 60 (24.0) |
| 1 | 67 (26.8) | Unknown | 82 (32.8) |
| 2 | 26 (10.4) | KRAS (%) | |
| 3 | 2 (0.8) | Mut | 102 (40.8) |
| 4 | 1 (0.4) | WT | 89 (35.6) |
| Site of primary tumor (%) | unknown | 59 (23.6) | |
| ileum | 2 (0.8) | Number of metastases | |
| Cecum | 17 (6.8) | <=4, | 104 (41.6) |
| Ascending colon | 57 (22.8) | >4, | 129 (51.6) |
| Transversal colon | 17 (6.8) | Unknown | 17 (6.8) |
| Descending colon | 98 (39.2) | Synchronous vs. Metachronous | |
| Rectum | 59 (23.6) | Metachronous (%) | 78 (31.2) |
| G stage of primary tumor (%) | Localization | ||
| 1 | 3 (1.2) | Unilobar | 76 (30.4) |
| 2 | 82 (32.8) | Bilobar | 173 (69.2) |
| 3 | 52 (20.8) | Unknown | 1 (0.4) |
| 4 | 1 (0.4) | Size (median [IQR]) | 3.50 [2.20, 5.60] |
| unknown | 112 (44.8) | N mets (median [IQR]) | 5.00 [2.00, 10.00] |
| T stage of primary tumor (%) | CEA ng/mL (median [IQR]) | 19.00 [5.00, 69.50] | |
| 0 | 1 ( 0.4) | Time to treatment (median [IQR]) | 17.00 [7.00, 27.00] |
| 1 | 1 (0.4) | ||
| 2 | 15 (6.0) | ||
| 3 | 115 (46.0) | ||
| 4 | 36 (14.4) | ||
| unknown | 82 (32.8) |
References
- Siegel, R.L.; Miller, K.D.; Sauer, A.G.; Fedewa, S.A.; Butterly, L.F.; Anderson, J.C.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 145–164. [Google Scholar] [CrossRef]
- Ballantyne, G.H.; Quin, J. Surgical treatment of liver metastases in patients with colorectal cancer. Cancer 1993, 71, 4252–4266. [Google Scholar] [CrossRef]
- van der Pool, A.E.M.; Damhuis, R.A.; Ijzermans, J.N.M.; de Wilt, J.H.W.; Eggermont, A.M.M.; Kranse, R.; Verhoef, C. Trends in incidence, treatment and survival of patients with stage IV colorectal cancer: A population-based series. Color. Dis. 2010, 14, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Borner, M.M. Neoadjuvant chemotherapy for unresectable liver metastases of colorectal cancer-too good to be true? Ann. Oncol. 1999, 10, 623–626. [Google Scholar] [CrossRef]
- Van Cutsem, E.; Cervantes, A.; Adam, R.; Sobrero, A.; Van Krieken, J.H.; Aderka, D.; Arnold, D. Faculty Opinions recommendation of ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann. Oncol. 2018, 27, 1386–1422. [Google Scholar] [CrossRef]
- Benson, A.B.; Venook, A.P.; Al-Hawary, M.M.; Arain, M.A.; Chen, Y.-J.; Ciombor, K.K.; Gurski, L.A. Colon Cancer, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2021, 19, 329–359. [Google Scholar] [CrossRef]
- Ruers, T.; Van Coevorden, F.; Punt, C.J.A.; Pierie, J.-P.E.N.; Borel-Rinkes, I.; Ledermann, J.A.; Poston, G.; Bechstein, W.; Lentz, M.-A.; Mauer, M.; et al. Local Treatment of Unresectable Colorectal Liver Metastases: Results of a Randomized Phase II Trial. JNCI J. Natl. Cancer Inst. 2017, 109, djx015. [Google Scholar] [CrossRef]
- Nordlinger, B.; Sorbye, H.; Glimelius, B.; Poston, G.J.; Schlag, P.M.; Rougier, P.; Bechstein, W.O.; Primrose, J.N.; Walpole, E.T.; Finch-Jones, M.; et al. Perioperative FOLFOX4 chemotherapy and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC 40983): Long-term results of a randomised, controlled, phase 3 trial. Lancet Oncol. 2013, 14, 1208–1215. [Google Scholar] [CrossRef]
- Cucchetti, A.; Ferrero, A.; Cescon, M.; Donadon, M.; Russolillo, N.; Ercolani, G.; Stacchini, G.; Mazzotti, F.; Torzilli, G.; Pinna, A.D.; et al. Cure Model Survival Analysis After Hepatic Resection for Colorectal Liver Metastases. Ann. Surg. Oncol. 2014, 22, 1908–1914. [Google Scholar] [CrossRef]
- Torzilli, G.; Viganò, L.; Gatti, A.; Costa, G.; Cimino, M.; Procopio, F.; Donadon, M.; Del Fabbro, D. Twelve-year experience of “radical but conservative” liver surgery for colorectal metastases: Impact on surgical practice and oncologic efficacy. HPB 2017, 19, 775–784. [Google Scholar] [CrossRef]
- Sorbye, H. Recurrence Patterns After Resection of Liver Metastases from Colorectal Cancer. Recent Results Cancer Res. 2014, 203, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Kopetz, S.; Chang, G.J.; Overman, M.J.; Eng, C.; Sargent, D.; Larson, D.W.; Grothey, A.; Vauthey, J.-N.; Nagorney, D.M.; McWilliams, R.R. Improved Survival in Metastatic Colorectal Cancer Is Associated With Adoption of Hepatic Resection and Improved Chemotherapy. J. Clin. Oncol. 2009, 27, 3677–3683. [Google Scholar] [CrossRef] [PubMed]
- Hong, N.J.L.; Wright, F.C.; Gagliardi, A.R.; Paszat, L.F. Examining the potential relationship between multidisciplinary cancer care and patient survival: An international literature review. J. Surg. Oncol. 2010, 102, 125–134. [Google Scholar] [CrossRef]
- Meijerink, M.R.; Puijk, R.S.; Van Tilborg, A.A.J.M.; Henningsen, K.H.; Fernandez, L.G.; Neyt, M.; Heymans, J.; Frankema, J.S.; De Jong, K.P.; Richel, D.J.; et al. Radiofrequency and Microwave Ablation Compared to Systemic Chemotherapy and to Partial Hepatectomy in the Treatment of Colorectal Liver Metastases: A Systematic Review and Meta-Analysis. Cardiovasc. Interv. Radiol. 2018, 41, 1189–1204. [Google Scholar] [CrossRef] [PubMed]
- Fiorentini, G.; Aliberti, C.; Tilli, M.; Coschiera, P.; Mambrini, A.; Giordani, P.; Alessandroni, P.; Benea, G. Hepatic arterial embolization adopting polyvinyl alcohol microspheres preloaded with irinotecan versus systemic chemotherapy for hepatic metastases from colorectal cancer: A phase III clinical research trial of activity and quality of life. J. Clin. Oncol. 2012, 30, 587. [Google Scholar] [CrossRef]
- Kurilova, I.; Bendet, A.; Petre, E.N.; Boas, F.E.; Kaye, E.; Gonen, M.; Covey, A.; Brody, L.A.; Brown, K.T.; Kemeny, N.E.; et al. Factors Associated With Local Tumor Control and Complications After Thermal Ablation of Colorectal Cancer Liver Metastases: A 15-year Retrospective Cohort Study. Clin. Color. Cancer 2020, 20, e82–e95. [Google Scholar] [CrossRef] [PubMed]
- Shady, W.; Petre, E.N.; Gonen, M.; Erinjeri, J.P.; Brown, K.T.; Covey, A.M.; Alago, W.; Durack, J.; Maybody, M.; Brody, L.A.; et al. Percutaneous Radiofrequency Ablation of Colorectal Cancer Liver Metastases: Factors Affecting Outcomes—A 10-year Experience at a Single Center. Radiology 2016, 278, 601–611. [Google Scholar] [CrossRef]
- Nieuwenhuizen, S.; Dijkstra, M.; Puijk, R.S.; Timmer, F.E.F.; Nota, I.M.; Opperman, J.; Bemd, B.V.D.; Geboers, B.; Ruarus, A.H.; Schouten, E.A.C.; et al. Thermal Ablation versus Stereotactic Ablative Body Radiotherapy to Treat Unresectable Colorectal Liver Metastases: A Comparative Analysis from the Prospective Amsterdam CORE Registry. Cancers 2021, 13, 4303. [Google Scholar] [CrossRef]
- Dijkstra, M.; Nieuwenhuizen, S.; Puijk, R.; Timmer, F.; Geboers, B.; Schouten, E.; Opperman, J.; Scheffer, H.; Vries, J.; Swijnenburg, R.-J.; et al. Thermal Ablation Compared to Partial Hepatectomy for Recurrent Colorectal Liver Metastases: An Amsterdam Colorectal Liver Met Registry (AmCORE) Based Study. Cancers 2021, 13, 2769. [Google Scholar] [CrossRef]
- Tanis, E.; Nordlinger, B.; Mauer, M.; Sorbye, H.; van Coevorden, F.; Gruenberger, T.; Schlag, P.; Punt, C.; Ledermann, J.; Ruers, T. Local recurrence rates after radiofrequency ablation or resection of colorectal liver metastases. Analysis of the European Organisation for Research and Treatment of Cancer #40004 and #40983. Eur. J. Cancer 2014, 50, 912–919. [Google Scholar] [CrossRef]
- Lordan, J.; Karanjia, N.; Quiney, N.; Fawcett, W.; Worthington, T. A 10-year study of outcome following hepatic resection for colorectal liver metastases—The effect of evaluation in a multidisciplinary team setting. Eur. J. Surg. Oncol. EJSO 2009, 35, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Pillay, B.; Wootten, A.C.; Crowe, H.; Corcoran, N.; Tran, B.; Bowden, P.; Crowe, J.; Costello, A.J. The impact of multidisciplinary team meetings on patient assessment, management and outcomes in oncology settings: A systematic review of the literature. Cancer Treat. Rev. 2016, 42, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Basso, M.; Corallo, S.; Calegari, M.A.; Zurlo, I.V.; Ardito, F.; Vellone, M.; Marchesani, S.; Orlandi, A.; Dadduzio, V.; Fucà, G.; et al. The impact of multidisciplinary team management on outcome of hepatic resection in liver-limited colorectal metastases. Sci. Rep. 2020, 10, 10871. [Google Scholar] [CrossRef] [PubMed]
- Viganò, L.; Langella, S.; Ferrero, A.; Russolillo, N.; Sperti, E.; Capussotti, L. Colorectal Cancer with Synchronous Resectable Liver Metastases: Monocentric Management in a Hepatobiliary Referral Center Improves Survival Outcomes. Ann. Surg. Oncol. 2012, 20, 938–945. [Google Scholar] [CrossRef]
- Fleissig, A.; Jenkins, V.; Catt, S.; Fallowfield, L. Multidisciplinary teams in cancer care: Are they effective in the UK? Lancet Oncol. 2006, 7, 935–943. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–656. [Google Scholar] [CrossRef]
- Neeman, E.; Gresham, G.; Ovasapians, N.; Hendifar, A.; Tuli, R.; Figlin, R.; Shinde, A. Comparing Physician and Nurse Eastern Cooperative Oncology Group Performance Status (ECOG-PS) Ratings as Predictors of Clinical Outcomes in Patients with Cancer. Oncologist 2019, 24, e1460–e1466. [Google Scholar] [CrossRef]
- Strasberg, S.; Belghiti, J.; Clavien, P.-A.; Gadzijev, E.; Garden, J.; Lau, W.-Y.; Makuuchi, M.; Strong, R. The Brisbane 2000 Terminology of Liver Anatomy and Resections. HPB 2000, 2, 333–339. [Google Scholar] [CrossRef]
- Clavien, P.A.; Barkun, J.; de Oliveira, M.L.; Vauthey, J.N.; Dindo, D.; Schulick, R.D.; de Santibañes, E.; Pekolj, J.; Slankamenac, K.; Bassi, C.; et al. The Clavien-Dindo Classification of Surgical Complications: Five-year experience. Ann. Surg. 2009, 250, 187–196. [Google Scholar] [CrossRef]
- Schwartz, L.H.; Litière, S.; de Vries, E.; Ford, R.; Gwyther, S.; Mandrekar, S.; Seymour, L.; Bogaerts, J.; Chen, A.; Dancey, J. RECIST 1.1-Update and clarification: From the RECIST committee. Eur. J. Cancer 2016, 62, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Weiser, M.R. AJCC 8th Edition: Colorectal Cancer. Ann. Surg. Oncol. 2018, 25, 1454–1455. [Google Scholar] [CrossRef] [PubMed]
- Haukoos, J.S.; Lewis, R.J. The Propensity Score. JAMA J. Am. Med Assoc. 2015, 314, 1637–1638. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.-T.; Jiang, J.-K.; Chang, S.-C.; Yang, S.-H.; Lin, C.-C.; Lin, H.-H.; Wang, H.-S.; Chen, W.-S.; Lin, T.-C.; Lin, J.-K. Improved outcomes of colorectal cancer patients with liver metastases in the era of the multidisciplinary teams. Int. J. Color. Dis. 2015, 31, 403–411. [Google Scholar] [CrossRef]
- Pawlik, T.M.; Laheru, D.; Hruban, R.H.; Coleman, J.; Wolfgang, C.L.; Campbell, K.; Ali, S.; Fishman, E.K.; Schulick, R.D.; Herman, J.M.; et al. Evaluating the Impact of a Single-Day Multidisciplinary Clinic on the Management of Pancreatic Cancer. Ann. Surg. Oncol. 2008, 15, 2081–2088. [Google Scholar] [CrossRef]
- Munro, A.; Brown, M.; Niblock, P.G.; Steele, R.; Carey, A.F. Do Multidisciplinary Team (MDT) processes influence survival in patients with colorectal cancer? A population-based experience. BMC Cancer 2015, 15, 686. [Google Scholar] [CrossRef]
- Yopp, A.C.; Mansour, J.C.; Beg, M.S.; Arenas, J.; Trimmer, C.; Reddick, M.; Pedrosa, I.; Khatri, G.; Yakoo, T.; Meyer, J.J.; et al. Establishment of a Multidisciplinary Hepatocellular Carcinoma Clinic is Associated with Improved Clinical Outcome. Ann. Surg. Oncol. 2013, 21, 1287–1295. [Google Scholar] [CrossRef]
- Adam, R.; De Gramont, A.; Figueras, J.; Guthrie, A.; Kokudo, N.; Kunstlinger, F.; Loyer, E.; Poston, G.; Rougier, P.; Rubbia-Brandt, L.; et al. The Oncosurgery Approach to Managing Liver Metastases from Colorectal Cancer: A Multidisciplinary International Consensus. Oncologist 2012, 17, 1225–1239. [Google Scholar] [CrossRef]
- Torzilli, G.; Adam, R.; Viganò, L.; Imai, K.; Goransky, J.; Fontana, A.P.; Toso, C.; Majno, P.; De Santibañes, E. Surgery of Colorectal Liver Metastases: Pushing the Limits. Liver Cancer 2016, 6, 80–89. [Google Scholar] [CrossRef]
- Prades, J.; Remue, E.; Van Hoof, E.; Borras, J.M. Is it worth reorganising cancer services on the basis of multidisciplinary teams (MDTs)? A systematic review of the objectives and organisation of MDTs and their impact on patient outcomes. Health Policy 2015, 119, 464–474. [Google Scholar] [CrossRef]
- Ottevanger, N.; Hilbink, M.; Weenk, M.; Janssen, R.; Vrijmoeth, T.; De Vries, A.; Hermens, R. Oncologic multidisciplinary team meetings: Evaluation of quality criteria. J. Eval. Clin. Pract. 2013, 19, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Keating, N.L.; Landrum, M.B.; Lamont, E.B.; Bozeman, S.R.; Shulman, L.N.; McNeil, B.J. Tumor Boards and the Quality of Cancer Care. JNCI J. Natl. Cancer Inst. 2012, 105, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Ke, K.M.; Blazeby, J.M.; Strong, S.; Carroll, E.F.; Ness, A.R.; Hollingworth, W. Are multidisciplinary teams in secondary care cost-effective? A systematic review of the literature. Cost Eff. Resour. Alloc. 2013, 11, 7–13. [Google Scholar] [CrossRef]
- Rogers, M.; Matheson, L.; Garrard, B.; Maher, B.; Cowdery, S.; Luo, W.; Reed, M.; Riches, S.; Pitson, G.; Ashley, D. Comparison of outcomes for cancer patients discussed and not discussed at a multidisciplinary meeting. Public Health 2017, 149, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Meagher, A.P. Colorectal cancer: Are multidisciplinary team meetings a waste of time? ANZ J. Surg. 2013, 83, 101–103. [Google Scholar] [CrossRef]
- Eigenmann, F. Multidisciplinary team meetings encourage overtreatment. BMJ 2015, 351, h4630. [Google Scholar] [CrossRef]
- Chinai, N.; Bintcliffe, F.; Armstrong, E.; Teape, J.; Jones, B.; Hosie, K. Does every patient need to be discussed at a multidisciplinary team meeting? Clin. Radiol. 2013, 68, 780–784. [Google Scholar] [CrossRef]
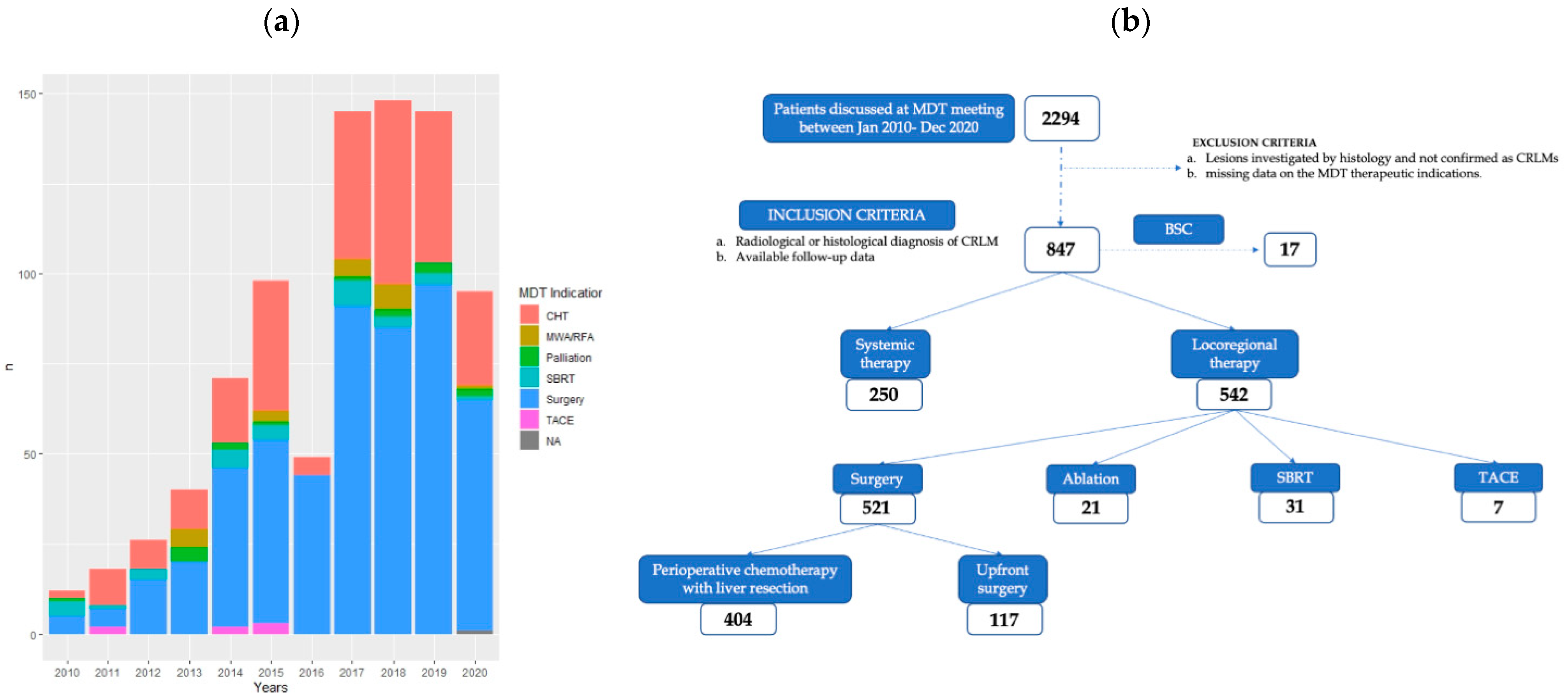
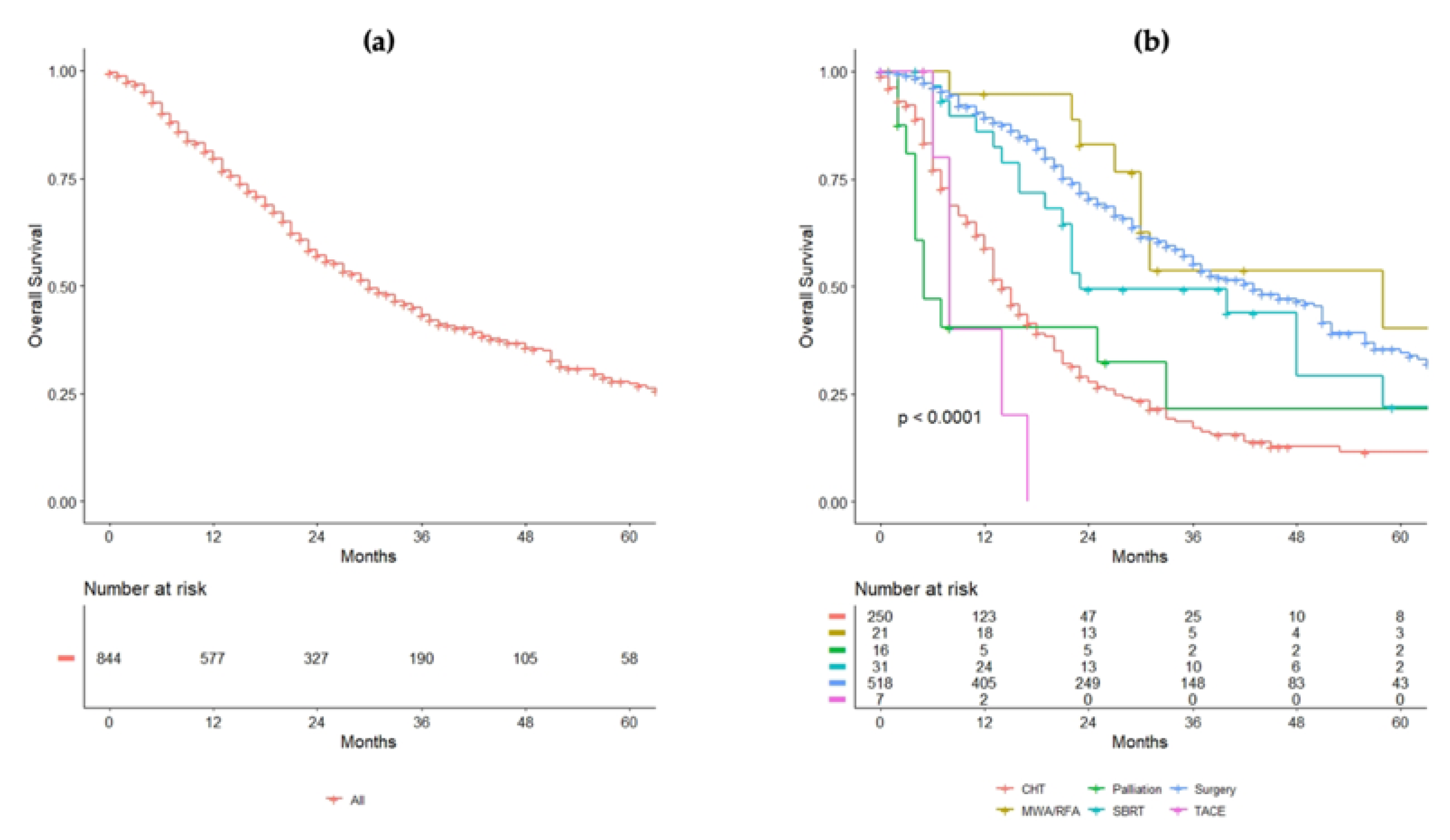
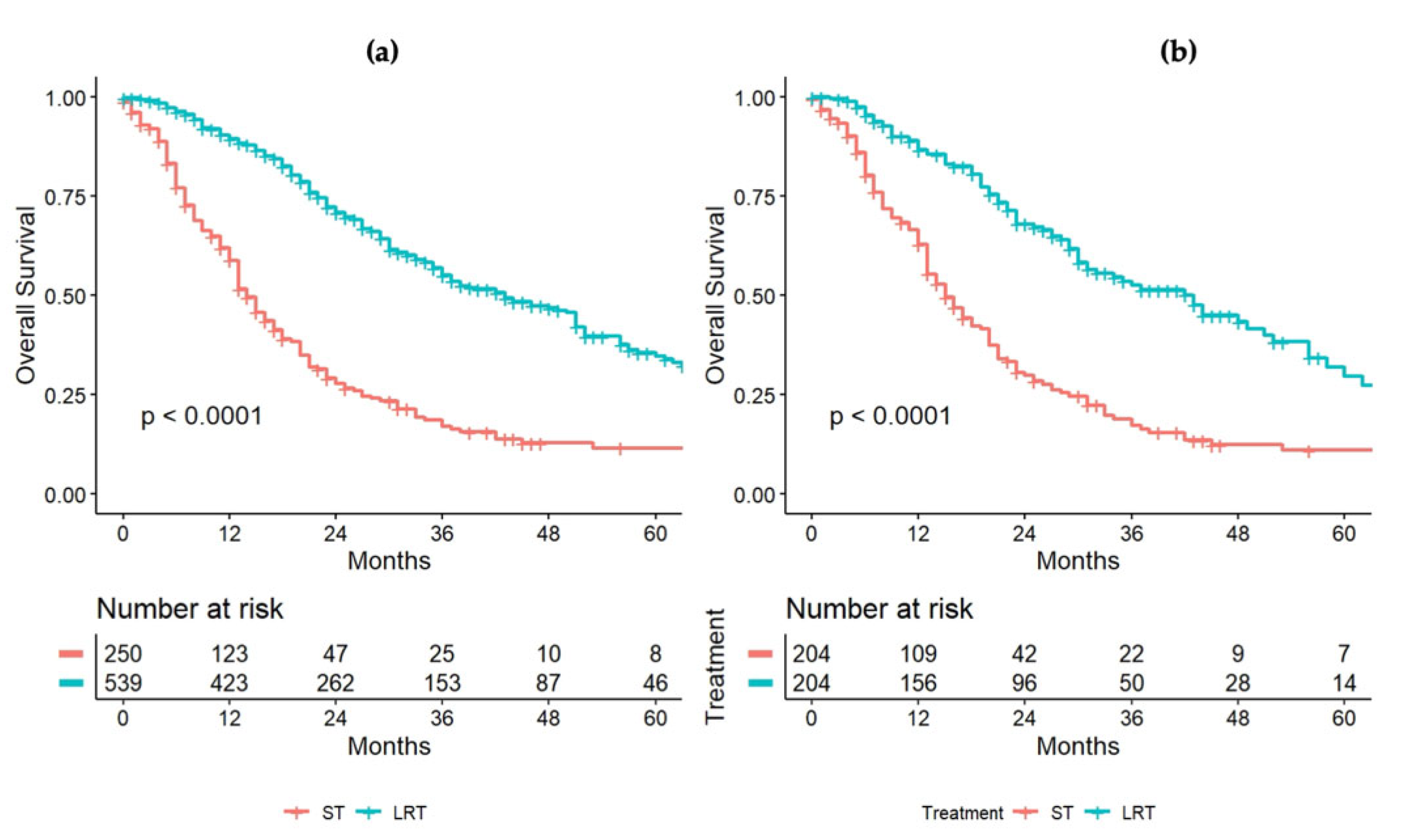
| Variable | Overall Series |
|---|---|
| Number | 847 |
| Male sex (%) | 549 (64.8) |
| Age (median; IQR) | 63.00; 55.00, 71.00 |
| ECOG PS (%) | |
| 0 | 566 (66.8) |
| 1 | 227 (26.8) |
| 2 | 49 (5.8) |
| 3 | 5 (0.6) |
| Site of primary tumor (%) | |
| ileum | 2 (0.2) |
| cecum | 59 (7.0) |
| ascending colon | 168 (19.8) |
| transversal colon | 51 (6.0) |
| descending colon | 340 (40.1) |
| rectum | 227 (26.8) |
| T stage (%) | |
| T 1-2 | 110 (12.9) |
| T 3-4 | 610 (72.1) |
| NA | 127 (15.0) |
| N status (%) | |
| 0 | 220 (26.0) |
| 1 | 286 (33.8) |
| 2 | 209 (24.7) |
| NA | 132 (15.6) |
| KRAS mutational status (%) | |
| Mutated | 325 (38.4) |
| Wild type | 352 (41.6) |
| NA | 170 (20.1) |
| NRAS mutational status (%) | |
| Mutated | 42 (5.0) |
| Wild type | 462 (54.5) |
| NA | 343 (40.5) |
| BRAF mutational status (%) | |
| Mutated | 22 (2.6) |
| Wild type | 441 (52.1) |
| NA | 384 (45.3) |
| Disease staging | |
| Computed tomography (%) | 842 (99.4) |
| Magnetic resonance imaging (%) | 733 (86.5) |
| Positron emission tomography (%) | 703 (83.0) |
| Metachronous disease (%) | 318 (37.5) |
| Liver bilobar disease | 496 (58.6) |
| Size of the largest metastasis (cm) (median; IQR) | 3.00; 1.90, 5.00 |
| Number of metastases (median; IQR) | 4.00; 2.00, 8.00 |
| Extrahepatic disease (%) | 245 (28.9) |
| CEA (ng/mL) (median; IQR) | 7.00; 3.00, 30.00 |
| MDT response (%) | |
| Surgery | 521 (61.5) |
| Systemic therapy | 250 (29.5) |
| SBRT | 31 (3.7) |
| MWA/RFA | 21 (2.5) |
| Palliation | 17 (2) |
| TACE | 7 (0.8) |
| Perioperative chemotherapy | 452 (53.3) |
| N of perioperative cycles (median; IQR) | 5.00; 0.00, 10.00 |
| RECIST (%) * | |
| CR | 1 (0.2) |
| PR | 287 (63.5) |
| SD | 69 (15.3) |
| PD | 88 (19.5) |
| NA | 7 (1.5) |
| Time to treatment (months) (median; IQR) | 27.00; 13.00, 51.00 |
| VARIABLE | HR Univariable | HR Multivariable | |
|---|---|---|---|
| Age | Mean (SD) | 1.00 (0.99–1.01, p = 0.806) | - |
| Site of primary tumor | cecum | 1.0 | - |
| ileum | 0.59 (0.08–4.35, p = 0.602) | - | |
| ascending colon | 1.13 (0.72–1.78, p = 0.582) | - | |
| transversal colon | 1.04 (0.58–1.84, p = 0.903) | ||
| descending colon | 0.76 (0.49–1.17, p = 0.208) | - | |
| rectum | 1.05 (0.68–1.63, p = 0.823) | - | |
| T stage | 1 | 1.0 | - |
| 2 | 1.12 (0.12–10.75, p = 0.923) | - | |
| 3 | 2.72 (0.37–19.75, p = 0.322) | - | |
| 4 | 2.41 (0.34–17.21, p = 0.380) | - | |
| N status | 0 | 1.0 | - |
| 1 | 1.21 (0.93–1.56, p = 0.152) | 1.11 (0.79–1.55, p = 0.554) | |
| 2 | 1.66 (1.27–2.18, p < 0.001) | 1.71 (1.20–2.43, p = 0.003) | |
| KRAS | Wild type | 1.0 | - |
| Mutated | 0.77 (0.62–0.95, p = 0.016) | 0.72 (0.54–0.96, p = 0.024) | |
| Systemic therapy versus loco-regional therapy | Systemic therapy | 1.0 | - |
| Locoregional treatment | 0.32 (0.26–0.39, p < 0.001) | 0.21 (0.14–0.29, p < 0.001) | |
| Bilobar disease | No | 1.0 | - |
| Yes | 1.79 (1.47–2.18, p < 0.001) | 1.22 (0.86–1.72, p = 0.265) | |
| Synchronous vs. Metachronous | synchronous | 1.0 | - |
| metachronous | 0.84 (0.69–1.02, p = 0.084) | - | |
| Size of metastases | Mean (SD) | 1.04 (1.03–1.06, p < 0.001) | 1.11 (1.06–1.17, p < 0.001) |
| Number of metastases | Mean (SD) | 1.02 (1.01–1.03, p < 0.001) | 1.00 (0.98–1.02, p = 0.842) |
| CEA (ng/mL) | Mean (SD) | 1.00 (1.00–1.00, p < 0.001) | 1.00 (1.00–1.00, p = 0.050) |
| Time to treatment | Mean (SD) | 1.00 (1.00–1.00, p = 0.076) | - |
| Order of resection | Simultaneous resection | 1.0 | - |
| Liver first | 0.94 (0.58–1.53, p = 0.817) | - | |
| Bowel first | 1.12 (0.78–1.61, p = 0.542) | - |
| PRE-Propensity SCORE | POST-Propensity SCORE | |||||
|---|---|---|---|---|---|---|
| ST | LRT | p | ST | LRT | p | |
| n | 250 | 542 | 209 | 209 | ||
| SEX = male (%) | 171 (68.4) | 340 (62.7) | 0.15 | 144 (68.9) | 131 (62.7) | 0.216 |
| Age (median [IQR]) | 63.00 [54.00, 71.75] | 63.00 [55.00, 70.00] | 0.754 | 63.00 [55.00, 72.00] | 61.00 [53.00, 70.00] | 0.048 |
| T stage (%) | <0.001 | 0.245 | ||||
| 0 | 1 (0.4) | 4 (0.7) | 7 (3.3) | 2 (1.0) | ||
| 1 | 1 (0.4) | 14 (2.6) | 12 (5.7) | 7 (3.3) | ||
| 2 | 15 (6.0) | 68 (12.6) | 24 (11.5) | 24 (11.5) | ||
| 3 | 115 (46.0) | 324 (59.8) | 108 (51.7) | 105 (50.2) | ||
| 4 | 36 (14.4) | 94 (17.3) | 58 (27.8) | 71 (34.0) | ||
| NA | 82 (32.8) | 38 (7) | ||||
| N stage (%) | <0.001 | 0.747 | ||||
| 0 | 48 (19.2) | 152 (28.1) | 95 (45.5) | 91 (43.5) | ||
| 1 | 60 (24.0) | 208 (38.4) | 59 (28.2) | 56 (26.8) | ||
| 2 | 60 (24.0) | 140 (25.8) | 55 (26.3) | 62 (29.7) | ||
| NA | 82 (32.8) | 42 (7.7) | ||||
| KRAS WT (%) | 89 (46.6%) | 246 (54.3%) | 0.089 | 128 (61.2%) | 119 (56.9%) | 0.426 |
| Metachronous disease (%) | 78 (31.2) | 214 (39.5) | 0.029 | 72 (34.4) | 70 (33.5) | 0.918 |
| Bilobar disease (%) | 173 (69.2) | 297 (54.8) | <0.001 | 133 (63.6) | 137 (65.6) | 0.759 |
| Extrahepatic disease (%) | 93 (37.2) | 138 (25.5) | 0.001 | 69 (33.0) | 74 (35.4) | 0.68 |
| Size of the largest metastasis (cm) | ||||||
| Median [IQR] | 3.50 [2.20, 5.60] | 3.00 [1.80, 4.50] | <0.001 | 3.20 [2.00, 5.20] | 3.30 [1.80, 5.40] | 0.967 |
| N of metastases | ||||||
| Median [IQR] | 5.00 [2.00, 10.00] | 4.00 [2.00, 7.00] | <0.001 | 5.00 [2.00, 10.00] | 5.00 [2.00, 10.00] | 0.671 |
| CEA (ng/ml) | ||||||
| Median [IQR] | 19.00 [5.00, 69.50] | 5.60 [3.00, 18.00] | <0.001 | 8.60 [4.00, 16.00] | 7.20 [3.00, 18.00] | 0.718 |
| Time to treatment (months) | ||||||
| Median [IQR] | 17.00 [7.00, 27.00] | 30.00 [15.00, 55.00] | <0.001 | 8.00 [6.00, 13.00] | 20.00 [8.00, 48.00] | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milana, F.; Famularo, S.; Luberto, A.; Rimassa, L.; Scorsetti, M.; Comito, T.; Pressiani, T.; Franzese, C.; Poretti, D.; Di Tommaso, L.; et al. Multidisciplinary Tumor Board in the Management of Patients with Colorectal Liver Metastases: A Single-Center Review of 847 Patients. Cancers 2022, 14, 3952. https://doi.org/10.3390/cancers14163952
Milana F, Famularo S, Luberto A, Rimassa L, Scorsetti M, Comito T, Pressiani T, Franzese C, Poretti D, Di Tommaso L, et al. Multidisciplinary Tumor Board in the Management of Patients with Colorectal Liver Metastases: A Single-Center Review of 847 Patients. Cancers. 2022; 14(16):3952. https://doi.org/10.3390/cancers14163952
Chicago/Turabian StyleMilana, Flavio, Simone Famularo, Antonio Luberto, Lorenza Rimassa, Marta Scorsetti, Tiziana Comito, Tiziana Pressiani, Ciro Franzese, Dario Poretti, Luca Di Tommaso, and et al. 2022. "Multidisciplinary Tumor Board in the Management of Patients with Colorectal Liver Metastases: A Single-Center Review of 847 Patients" Cancers 14, no. 16: 3952. https://doi.org/10.3390/cancers14163952
APA StyleMilana, F., Famularo, S., Luberto, A., Rimassa, L., Scorsetti, M., Comito, T., Pressiani, T., Franzese, C., Poretti, D., Di Tommaso, L., Personeni, N., Rodari, M., Pedicini, V., Donadon, M., & Torzilli, G. (2022). Multidisciplinary Tumor Board in the Management of Patients with Colorectal Liver Metastases: A Single-Center Review of 847 Patients. Cancers, 14(16), 3952. https://doi.org/10.3390/cancers14163952










