FXYD3 Expression Predicts Poor Prognosis in Renal Cell Carcinoma with Immunosuppressive Tumor Microenvironment
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Survival Analysis with FXYD3 mRNA Levels in the Public Database
2.2. Estimated TIL Fraction
2.3. Gene Co-Expression Network Analysis
2.4. Statistical Analysis
3. Results
3.1. Clinical Characteristics by FXYD3 Expression Level in KIRC Patients
3.2. Prognostic Impact of FXYD3 Gene Expression in KIRC
3.3. Infiltrating Immune Cells and FXYD3 mRNA Levels
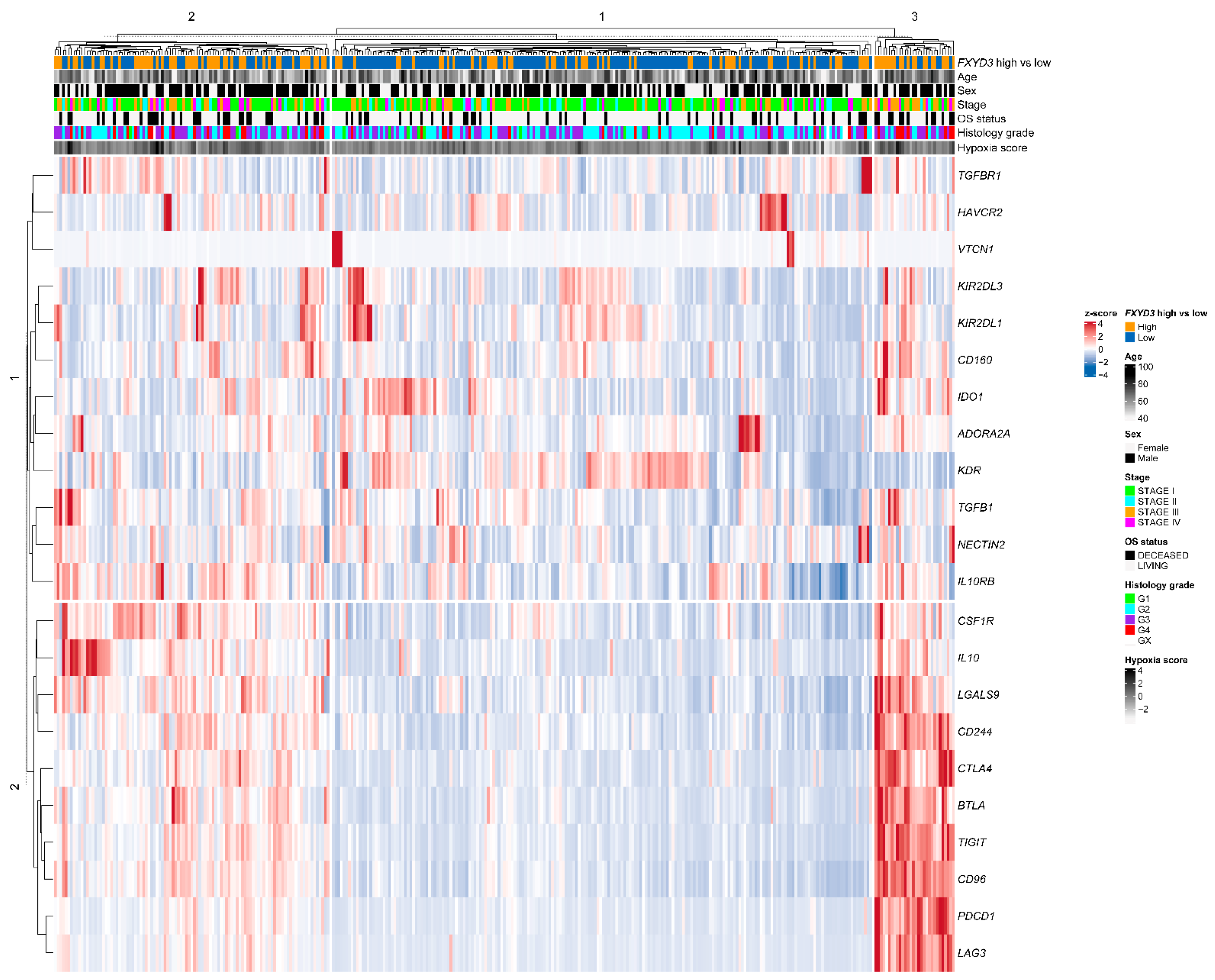
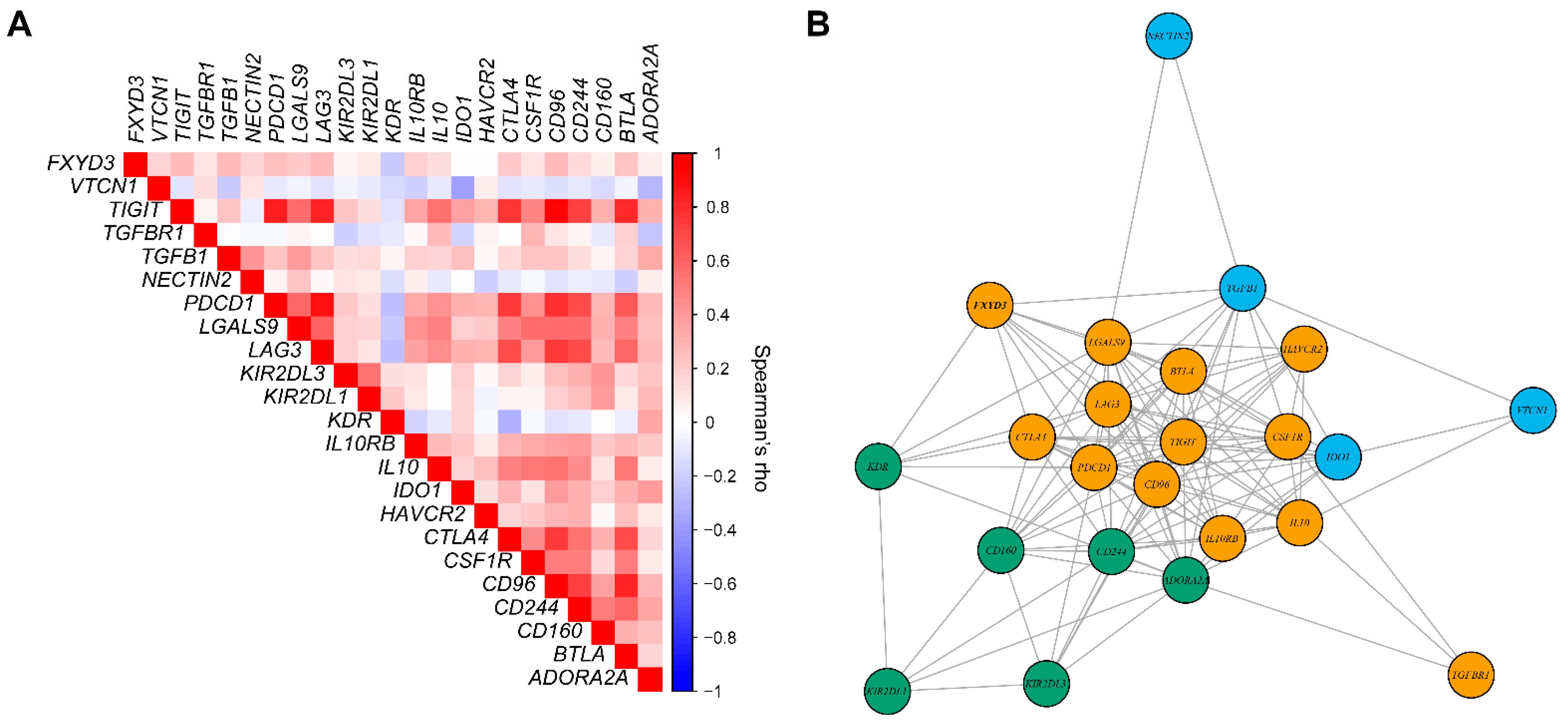
3.4. Expression of Immunoinhibitory Genes and FXYD3 mRNA Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hsieh, J.J.; Purdue, M.P.; Signoretti, S.; Swanton, C.; Albiges, L.; Schmidinger, M.; Heng, D.Y.; Larkin, J.; Ficarra, V. Renal cell carcinoma. Nat. Rev. Dis. Prim. 2017, 3, 17009. [Google Scholar] [CrossRef]
- Makhov, P.; Joshi, S.; Ghatalia, P.; Kutikov, A.; Uzzo, R.G.; Kolenko, V.M. Resistance to Systemic Therapies in Clear Cell Renal Cell Carcinoma: Mechanisms and Management Strategies. Mol. Cancer Ther. 2018, 17, 1355–1364. [Google Scholar] [CrossRef]
- Nukui, A.; Masuda, A.; Abe, H.; Arai, K.; Yoshida, K.-I.; Kamai, T. Increased serum level of soluble interleukin-2 receptor is associated with a worse response of metastatic clear cell renal cell carcinoma to interferon alpha and sequential VEGF-targeting therapy. BMC Cancer 2017, 17, 372. [Google Scholar] [CrossRef] [PubMed]
- Ueda, K.; Suekane, S.; Kurose, H.; Chikui, K.; Nakiri, M.; Nishihara, K.; Matsuo, M.; Kawahara, A.; Yano, H.; Igawa, T. Prognostic value of PD-1 and PD-L1 expression in patients with metastatic clear cell renal cell carcinoma. Urol. Oncol. 2018, 36, 499.e9–499.e16. [Google Scholar] [CrossRef] [PubMed]
- Mollica, V.; Santoni, M.; Matrana, M.R.; Basso, U.; De Giorgi, U.; Rizzo, A.; Maruzzo, M.; Marchetti, A.; Rosellini, M.; Bleve, S.; et al. Concomitant Proton Pump Inhibitors and Outcome of Patients Treated with Nivolumab Alone or Plus Ipilimumab for Advanced Renal Cell Carcinoma. Target. Oncol. 2022, 17, 61–68. [Google Scholar] [CrossRef]
- Geering, K.; Béguin, P.; Garty, H.; Karlish, S.; Füzesi, M.; Horisberger, J.-D.; Crambert, G. FXYD Proteins: New Tissue- and Isoform-Specific Regulators of Na,K-ATPase. Ann. N. Y. Acad. Sci. 2003, 986, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Franzin, C.M.; Gong, X.M.; Teriete, P.; Marassi, F.M. Structures of the FXYD regulatory proteins in lipid micelles and membranes. J. Bioenerg. Biomembr. 2007, 39, 379–383. [Google Scholar] [CrossRef][Green Version]
- Li, Y.; Zhang, X.; Xu, S.; Ge, J.; Liu, J.; Li, L.; Fang, G.; Meng, Y.; Zhang, H.; Sun, X. Expression and clinical significance of FXYD3 in endometrial cancer. Oncol. Lett. 2014, 8, 517–522. [Google Scholar] [CrossRef]
- Gordon, G.J.; Richards, W.G.; Sugarbaker, D.J.; Jaklitsch, M.T.; Bueno, R. A prognostic test for adenocarcinoma of the lung from gene expression profiling data. Cancer Epidemiol. Biomark. Prev. 2003, 12, 905–910. [Google Scholar]
- Widegren, E.; Önnesjö, S.; Arbman, G.; Kayed, H.; Zentgraf, H.; Kleeff, J.; Zhang, H.; Sun, X.-F. Expression of FXYD3 Protein in Relation to Biological and Clinicopathological Variables in Colorectal Cancers. Chemotherapy 2009, 55, 407–413. [Google Scholar] [CrossRef]
- Jin, M.; Zhang, H.; Yang, J.; Zheng, Z.; Liu, K. Expression mode and prognostic value of FXYD family members in colon cancer. Aging 2021, 13, 18404–18422. [Google Scholar] [CrossRef] [PubMed]
- Loftås, P.; Önnesjö, S.; Widegren, E.; Adell, G.; Kayed, H.; Kleeff, J.; Zentgraf, H.; Sun, X.-F. Expression of FXYD-3 is an Independent Prognostic Factor in Rectal Cancer Patients With Preoperative Radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.J.; Li, Q.J.; Le, Y.; Ouyang, H.Y.; He, M.K.; Yu, Z.S.; Zhang, Y.F.; Shi, M. Prognostic significance of sodium-potassium ATPase regulator, FXYD3, in human hepatocellular carcinoma. Oncol. Lett. 2018, 15, 3024–3030. [Google Scholar] [CrossRef] [PubMed]
- Finotello, F.; Mayer, C.; Plattner, C.; Laschober, G.; Rieder, D.; Hackl, H.; Krogsdam, A.; Loncova, Z.; Posch, W.; Wilflingseder, D.; et al. Molecular and pharmacological modulators of the tumor immune contexture revealed by deconvolution of RNA-seq data. Genome Med. 2019, 11, 34. [Google Scholar] [CrossRef]
- Charoentong, P.; Finotello, F.; Angelova, M.; Mayer, C.; Efremova, M.; Rieder, D.; Hackl, H.; Trajanoski, Z. Pan-cancer Immunogenomic Analyses Reveal Genotype-Immunophenotype Relationships and Predictors of Response to Checkpoint Blockade. Cell Rep. 2017, 18, 248–262. [Google Scholar] [CrossRef] [PubMed]
- Edeline, J.; Mottier, S.; Vigneau, C.; Jouan, F.; Perrin, C.; Zerrouki, S.; Fergelot, P.; Patard, J.-J.; Rioux-Leclercq, N. Description of 2 angiogenic phenotypes in clear cell renal cell carcinoma. Hum. Pathol. 2012, 43, 1982–1990. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio cancer genomics portal: An open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.E.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative Analysis of Complex Cancer Genomics and Clinical Profiles Using the cBioPortal. Sci. Signal. 2013, 6, l1. [Google Scholar] [CrossRef]
- Yoshihara, K.; Shahmoradgoli, M.; Martínez, E.; Vegesna, R.; Kim, H.; Torres-Garcia, W.; Trevino, V.; Shen, H.; Laird, P.W.; Levine, D.A.; et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat. Commun. 2013, 4, 2612. [Google Scholar] [CrossRef]
- Ru, B.; Wong, C.N.; Tong, Y.; Zhong, J.Y.; Zhong, S.S.W.; Wu, W.C.; Chu, K.C.; Wong, C.Y.; Lau, C.Y.; Chen, I.; et al. TISIDB: An integrated repository portal for tumor–immune system interactions. Bioinformatics 2019, 35, 4200–4202. [Google Scholar] [CrossRef]
- Li, T.; Fan, J.; Wang, B.; Traugh, N.; Chen, Q.; Liu, J.S.; Li, B.; Liu, X.S. TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res. 2017, 77, e108–e110. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Fu, J.; Zeng, Z.; Cohen, D.; Li, J.; Chen, Q.; Li, B.; Liu, X.S. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020, 48, W509–W514. [Google Scholar] [CrossRef] [PubMed]
- Buffa, F.M.; Harris, A.L.; West, C.M.; Miller, C.J. Large meta-analysis of multiple cancers reveals a common, compact and highly prognostic hypoxia metagene. Br. J. Cancer 2010, 102, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, E.; Long, J.; Hu, Z.; Peng, J.; Liu, L.; Tang, F.; Li, L.; Ouyang, Y.; Zeng, Z. Immune infiltration in renal cell carcinoma. Cancer Sci. 2019, 110, 1564–1572. [Google Scholar] [CrossRef]
- Zhu, Z.L.; Yan, B.-Y.; Zhang, Y.; Yang, Y.-H.; Wang, M.-W.; Zentgraf, H.; Zhang, X.-H.; Sun, X.-F. Overexpression of FXYD-3 Is Involved in the Tumorigenesis and Development of Esophageal Squamous Cell Carcinoma. Dis. Markers 2013, 35, 195–202. [Google Scholar] [CrossRef]
- Zhu, Z.L.; Zhao, Z.-R.; Zhang, Y.; Yang, Y.-H.; Wang, Z.-M.; Cui, D.-S.; Wang, M.-W.; Kleeff, J.; Kayed, H.; Yan, B.-Y.; et al. Expression and significance of FXYD-3 protein in gastric adenocarcinoma. Dis. Markers 2010, 28, 63–69. [Google Scholar] [CrossRef]
- Kayed, H.; Kleeff, J.; Kolb, A.; Ketterer, K.; Keleg, S.; Felix, K.; Giese, T.; Penzel, R.; Zentgraf, H.; Büchler, M.W.; et al. FXYD3 is overexpressed in pancreatic ductal adenocarcinoma and influences pancreatic cancer cell growth. Int. J. Cancer 2006, 118, 43–54. [Google Scholar] [CrossRef]
- Yamamoto, H.; Okumura, K.; Toshima, S.; Mukaisho, K.-I.; Sugihara, H.; Hattori, T.; Kato, M.; Asano, S. FXYD3 Protein Involved in Tumor Cell Proliferation Is Overproduced in Human Breast Cancer Tissues. Biol. Pharm. Bull. 2009, 32, 1148–1154. [Google Scholar] [CrossRef][Green Version]
- Herrmann, P.; Aronica, S.M. Estrogen and tamoxifen up-regulate FXYD3 on breast cancer cells: Assessing the differential roles of ER α and ZEB1. SpringerPlus 2015, 4, 245. [Google Scholar] [CrossRef]
- Liu, C.C.; Teh, R.; Mozar, C.A.; Baxter, R.C.; Rasmussen, H.H. Silencing overexpression of FXYD3 protein in breast cancer cells amplifies effects of doxorubicin and γ-radiation on Na+/K+-ATPase and cell survival. Breast Cancer Res. Treat. 2016, 155, 203–213. [Google Scholar] [CrossRef]
- Wang, M.W.; Gu, P.; Zhang, Z.-Y.; Zhu, Z.-L.; Geng, Y.; Kayed, H.; Zentgraf, H.; Sun, X.-F. FXYD3 Expression in Gliomas and its Clinicopathological Significance. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2009, 18, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Okudela, K.; Yazawa, T.; Ishii, J.; Woo, T.; Mitsui, H.; Bunai, T.; Sakaeda, M.; Shimoyamada, H.; Sato, H.; Tajiri, M.; et al. Down-Regulation of FXYD3 Expression in Human Lung Cancers: Its Mechanism and Potential Role in Carcinogenesis. Am. J. Pathol. 2009, 175, 2646–2656. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Arimochi, J.; Ohashi-Kobayashi, A.; Maeda, M. Interaction of Mat-8 (FXYD-3) with Na+/K+-ATPase in Colorectal Cancer Cells. Biol. Pharm. Bull. 2007, 30, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Loftas, P.; Arbman, G.; Sun, X.-F.; Edler, D.; Syk, E.; Hallböök, O. FXYD-3 expression in relation to local recurrence of rectal cancer. Radiat. Oncol. J. 2016, 34, 52–58. [Google Scholar] [CrossRef]
- Meding, S.; Balluff, B.; Elsner, M.; Schöne, C.; Rauser, S.; Nitsche, U.; Maak, M.; Schäfer, A.; Hauck, S.; Ueffing, M.; et al. Tissue-based proteomics reveals FXYD3, S100A11 and GSTM3 as novel markers for regional lymph node metastasis in colon cancer. J. Pathol. 2012, 228, 459–470. [Google Scholar] [CrossRef]
- Schödel, J.; Grampp, S.; Maher, E.R.; Moch, H.; Ratcliffe, P.J.; Russo, P.; Mole, D.R. Hypoxia, Hypoxia-inducible Transcription Factors, and Renal Cancer. Eur. Urol. 2016, 69, 646–657. [Google Scholar] [CrossRef]
- Drake, C.G.; Stein, M.N. The Immunobiology of Kidney Cancer. J. Clin. Oncol. 2018, 36, 3547–3552. [Google Scholar] [CrossRef]
- Kim, M.C.; Jin, Z.; Kolb, R.; Borcherding, N.; Chatzkel, J.A.; Falzarano, S.M.; Zhang, W. Updates on Immunotherapy and Immune Landscape in Renal Clear Cell Carcinoma. Cancers 2021, 13, 5856. [Google Scholar] [CrossRef]
- Zarour, H.M. Reversing T-cell Dysfunction and Exhaustion in Cancer. Clin. Cancer Res. 2016, 22, 1856–1864. [Google Scholar] [CrossRef]
- Okoye, I.; Xu, L.; Motamedi, M.; Parashar, P.; Walker, J.W.; Elahi, S. Galectin-9 expression defines exhausted T cells and impaired cytotoxic NK cells in patients with virus-associated solid tumors. J. Immunother. Cancer 2020, 8, e001849. [Google Scholar] [CrossRef]
- Remark, R.; Alifano, M.; Cremer, I.; Lupo, A.; Dieu-Nosjean, M.-C.; Riquet, M.; Crozet, L.; Ouakrim, H.; Goc, J.; Cazes, A.; et al. Characteristics and Clinical Impacts of the Immune Environments in Colorectal and Renal Cell Carcinoma Lung Metastases: Influence of Tumor Origin. Clin. Cancer Res. 2013, 19, 4079–4091. [Google Scholar] [CrossRef]
- Donskov, F.; von der Maase, H. Impact of Immune Parameters on Long-Term Survival in Metastatic Renal Cell Carcinoma. J. Clin. Oncol. 2006, 24, 1997–2005. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Lou, L.; Ye, J.; Zhang, S. Prognostic role of the neutrophil-lymphocyte ratio in renal cell carcinoma: A meta-analysis. BMJ Open 2015, 5, e006404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Huang, Y.; Lindstrom, A.R.; Lin, T.-Y.; Lam, K.S.; Li, Y. Peptide-based materials for cancer immunotherapy. Theranostics 2019, 9, 7807–7825. [Google Scholar] [CrossRef] [PubMed]
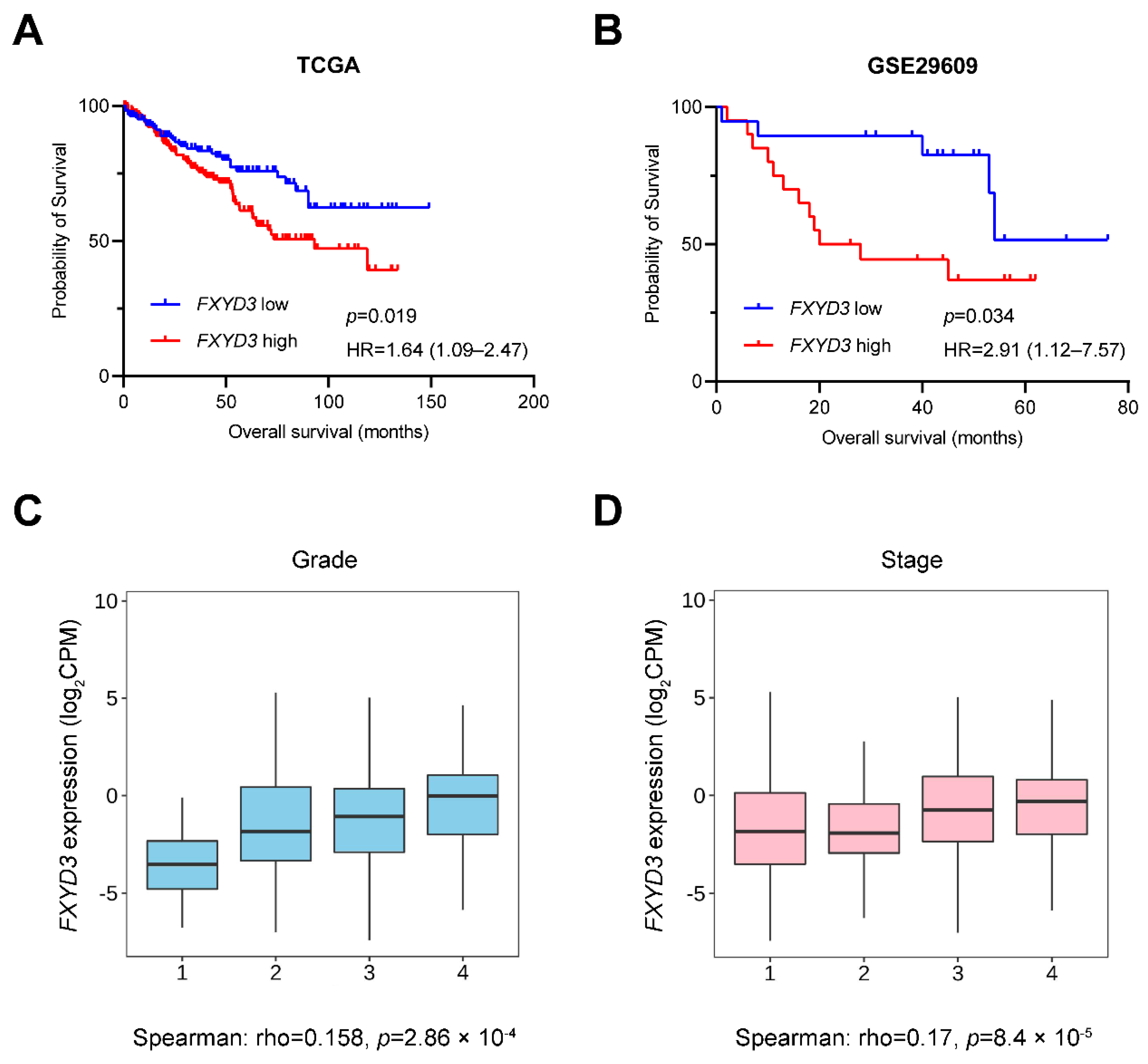
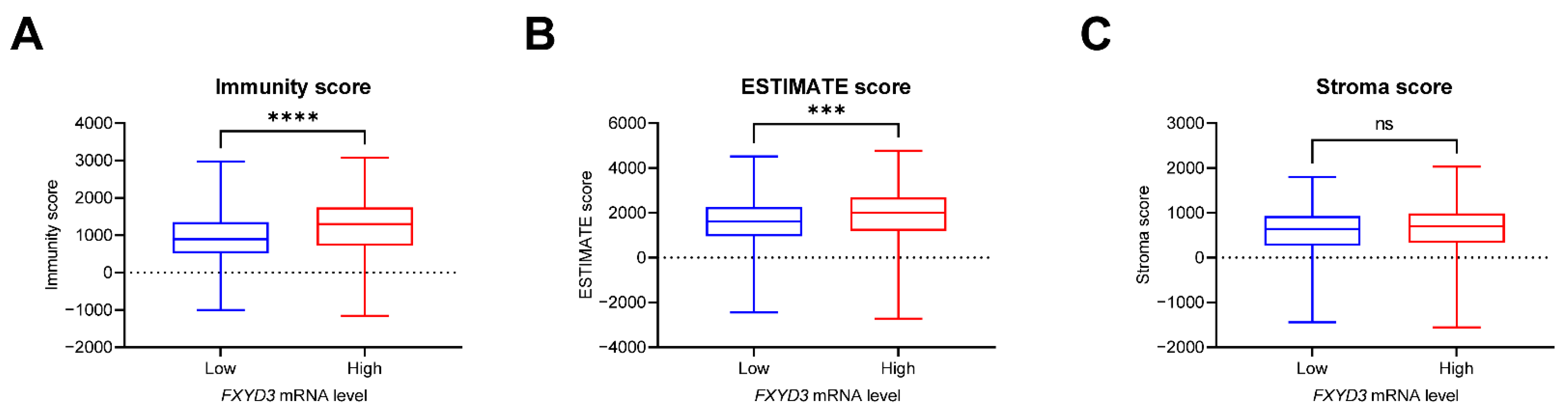
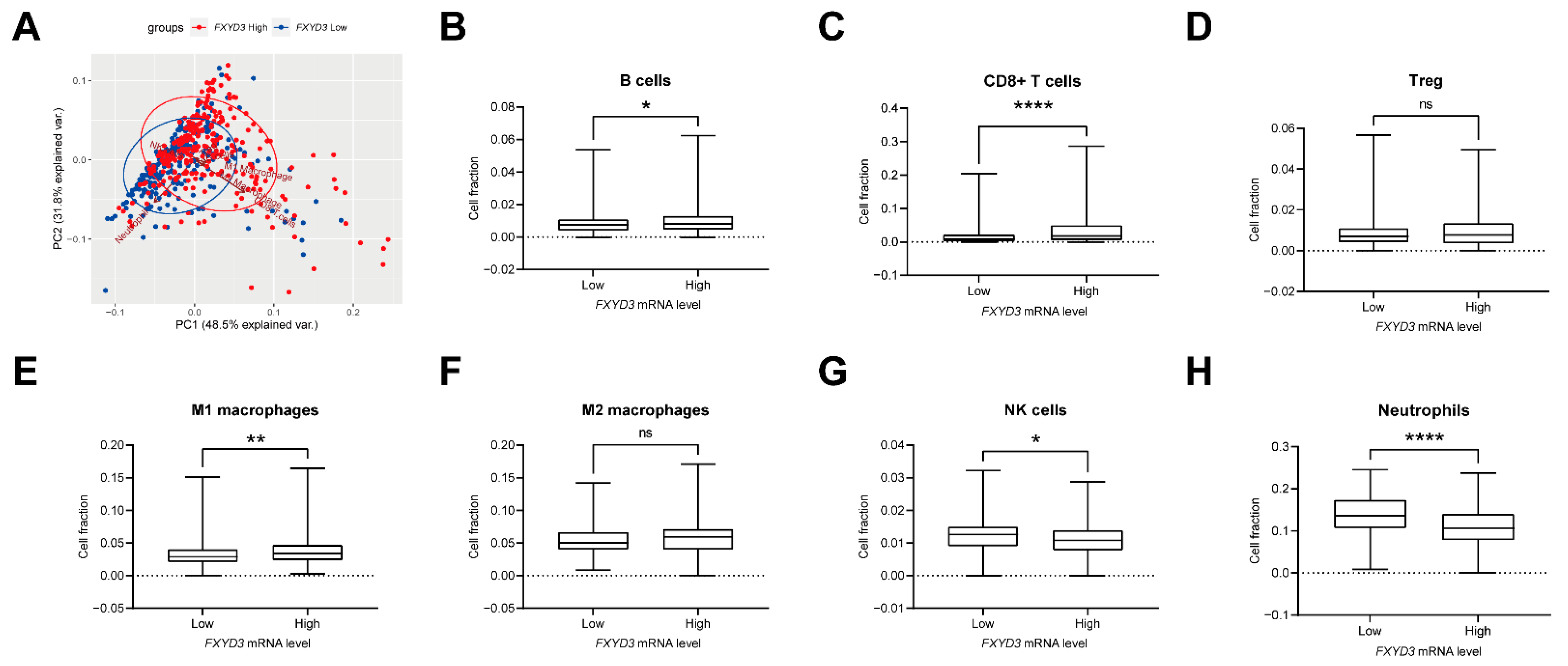
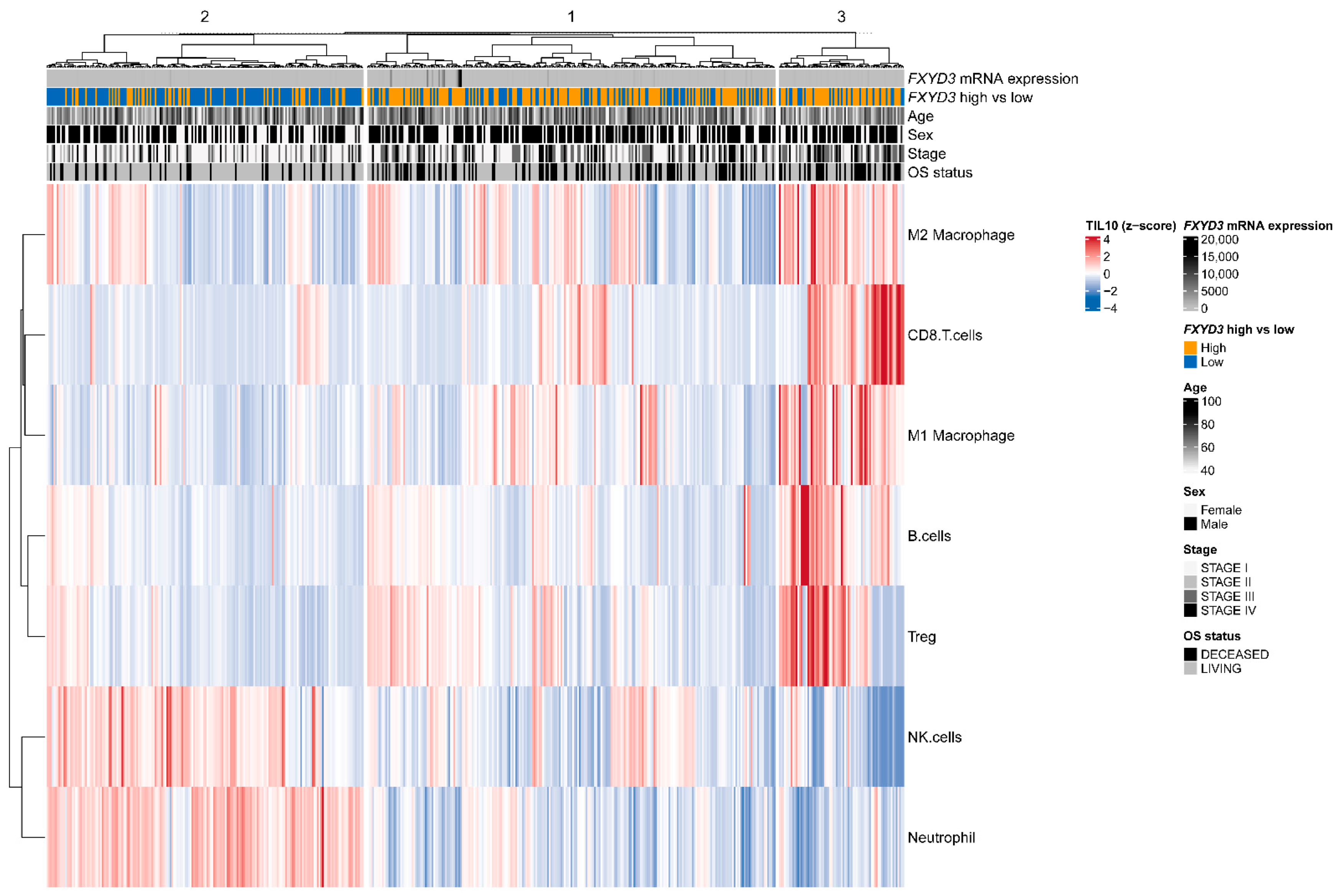
| Characteristics | FXYD3 Low n = 255 | FXYD3 High n = 255 | p † |
|---|---|---|---|
| Age (median [range]) | 60.0 (29–90) | 61.0 (32–90) | 0.047 |
| Sex | |||
| Male | 150 | 175 | 0.021 |
| Female | 105 | 80 | |
| Stage | |||
| I | 146 | 108 | 0.0026 |
| II | 34 | 32 | |
| III | 71 | 108 | |
| IV | 4 | 7 | |
| Histologic grade | |||
| G1 | 12 | 1 | < 0.0001 |
| G2 | 121 | 93 | |
| G3 | 95 | 105 | |
| G4 | 21 | 54 | |
| GX | 4 | 1 | |
| Buffa hypoxia score (median [range]) | −1 (−33–35) | 5 (−19–43) | < 0.001 |
| Mutation count (median [range]) | 49 (8–426) | 55 (10–591) | 0.272 |
| Variable | HR | 95% CI | p-Value |
|---|---|---|---|
| Age at diagnosis | 1.033 | 1.018–1.048 | <0.0001 |
| Male (ref: Female) | 0.903 | 0.656–1.244 | 0.532 |
| Stage at initial diagnosis (ref: Stage 0/I) | |||
| II | 1.311 | 0.704–2.440 | 0.394 |
| III | 2.519 | 1.675–3.788 | <0.0001 |
| IV | 7.081 | 4.818–10.41 | <0.0001 |
| FXYD3 mRNA level | 1.117 | 1.013–1.232 | 0.027 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yonekura, S.; Ueda, K. FXYD3 Expression Predicts Poor Prognosis in Renal Cell Carcinoma with Immunosuppressive Tumor Microenvironment. Cancers 2022, 14, 3596. https://doi.org/10.3390/cancers14153596
Yonekura S, Ueda K. FXYD3 Expression Predicts Poor Prognosis in Renal Cell Carcinoma with Immunosuppressive Tumor Microenvironment. Cancers. 2022; 14(15):3596. https://doi.org/10.3390/cancers14153596
Chicago/Turabian StyleYonekura, Satoru, and Kosuke Ueda. 2022. "FXYD3 Expression Predicts Poor Prognosis in Renal Cell Carcinoma with Immunosuppressive Tumor Microenvironment" Cancers 14, no. 15: 3596. https://doi.org/10.3390/cancers14153596
APA StyleYonekura, S., & Ueda, K. (2022). FXYD3 Expression Predicts Poor Prognosis in Renal Cell Carcinoma with Immunosuppressive Tumor Microenvironment. Cancers, 14(15), 3596. https://doi.org/10.3390/cancers14153596






