Sex Differences in Taxane Toxicities
Abstract
:Simple Summary
Abstract
1. Introduction
2. Paclitaxel and Taxane Background
3. Translational and Clinical Research
3.1. Pharmacokinetics
3.2. Adverse Drug Reactions
3.3. Neurotoxicity
3.3.1. Peripheral Neuropathy
3.3.2. Cognitive Dysfunction
4. Basic Research
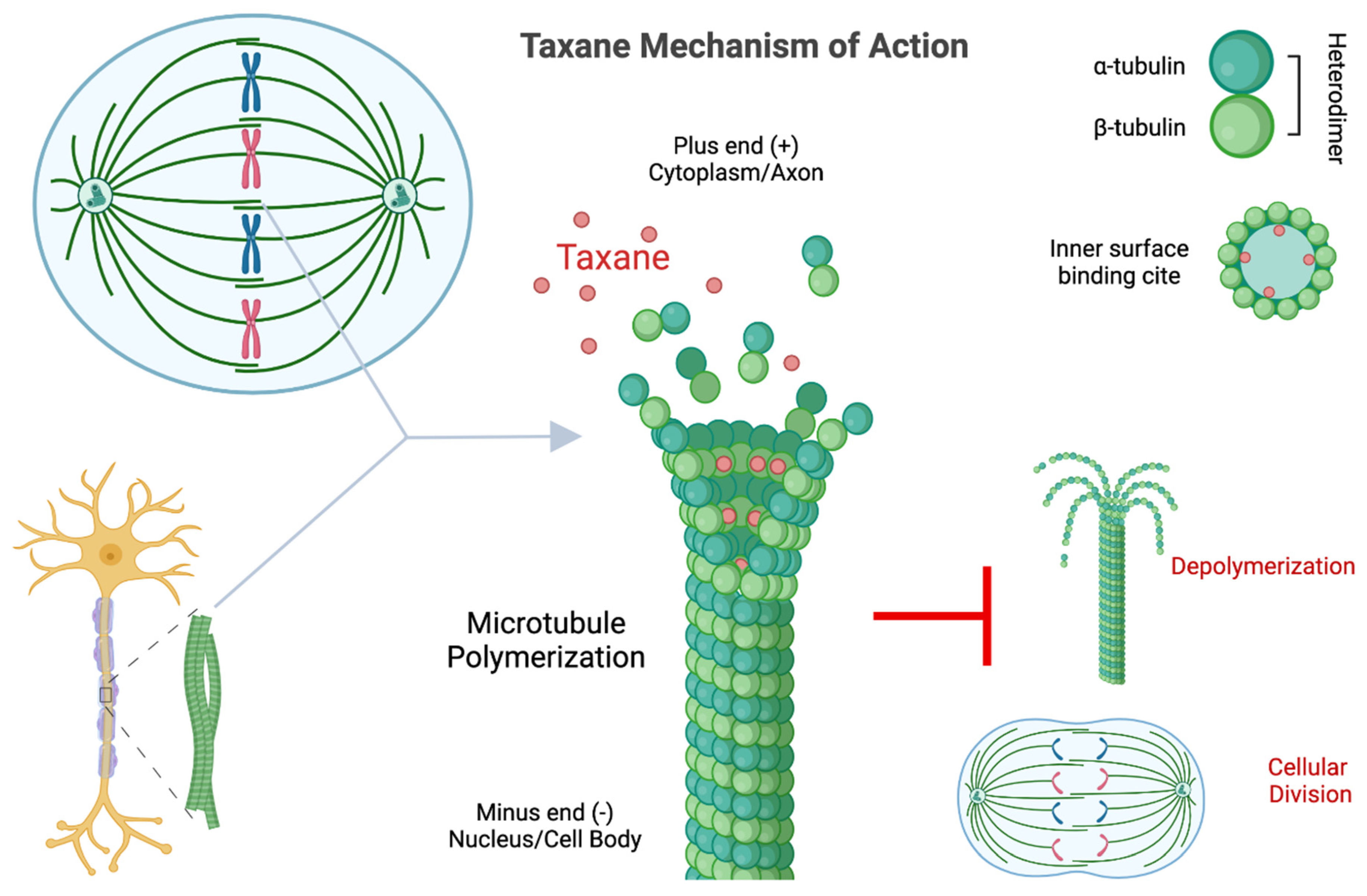
4.1. Mitotic Arrest
4.2. Microtubule Dynamic Dysfunction
4.2.1. Androgen Receptor Signaling Dysfunction
4.2.2. Estrogen-Mediated Microtubule Dynamics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tran, C.; Knowles, S.R.; Liu, B.A.; Shear, N.H. Gender Differences in Adverse Drug Reactions. J. Clin. Pharmacol. 1998, 38, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Rademaker, M. Do Women Have More Adverse Drug Reactions? Am. J. Clin. Dermatol. 2001, 2, 349–351. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.D. Sex and Racial Differences in Pharmacological Response: Where Is the Evidence? Pharmacogenetics, Pharmacokinetics, and Pharmacodynamics. J. Women’s Health 2005, 14, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Bren, L. FDA Consumer; Federal Drug Administration: Washington, DC, USA, 2005; Volume 39, pp. 10–15. [PubMed]
- Schmetzer, O.; Flörcken, A. Sex Differences in the Drug Therapy for Oncologic Diseases. Handb. Exp. Pharmacol. 2012, 214, 411–442. [Google Scholar] [CrossRef]
- Wang, J.; Huang, Y. Pharmacogenomics of Sex Difference in Chemotherapeutic Toxicity. Curr. Drug Discov. Technol. 2007, 4, 59–68. [Google Scholar] [CrossRef]
- Haupt, S.; Caramia, F.; Klein, S.L.; Rubin, J.B.; Haupt, Y. Sex Disparities Matter in Cancer Development and Therapy. Nat. Rev. Cancer 2021, 21, 393–407. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-I.; Lim, H.; Moon, A. Sex Differences in Cancer: Epidemiology, Genetics and Therapy. Biomol. Ther. 2018, 26, 335–342. [Google Scholar] [CrossRef]
- Cook, M.B.; McGlynn, K.A.; Devesa, S.S.; Freedman, N.D.; Anderson, W.F. Sex Disparities in Cancer Mortality and Survival. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1629–1637. [Google Scholar] [CrossRef] [Green Version]
- Islam, M.M.; Iqbal, U.; Walther, B.A.; Nguyen, P.A.; Li, Y.C.; Dubey, N.K.; Poly, T.N.; Masud, J.H.B.; Atique, S.; Syed-Abdul, S. Gender-Based Personalized Pharmacotherapy: A Systematic Review. Arch. Gynecol. Obstet. 2017, 295, 1305–1317. [Google Scholar] [CrossRef]
- Davidson, M.; Wagner, A.D.; Kouvelakis, K.; Nanji, H.; Starling, N.; Chau, I.; Watkins, D.; Rao, S.; Peckitt, C.; Cunningham, D. Influence of Sex on Chemotherapy Efficacy and Toxicity in Oesophagogastric Cancer: A Pooled Analysis of Four Randomised Trials. Eur. J. Cancer 2019, 121, 40–47. [Google Scholar] [CrossRef]
- Wagner, A.D.; Oertelt-Prigione, S.; Adjei, A.; Buclin, T.; Cristina, V.; Csajka, C.; Coukos, G.; Dafni, U.; Dotto, G.-P.; Ducreux, M.; et al. Gender Medicine and Oncology: Report and Consensus of an ESMO Workshop. Ann. Oncol. 2019, 30, 1914–1924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, A.D. Sex Differences in Cancer Chemotherapy Effects, and Why We Need to Reconsider BSA-Based Dosing of Chemotherapy. ESMO Open 2020, 5, 770. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends—An Update. Cancer Epidemiol. Biomark. Prev. 2016, 25, 16–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahles, T.A.; Saykin, A.J. Breast Cancer Chemotherapy-Related Cognitive Dysfunction. Clin. Breast Cancer 2002, 3, S84–S90. [Google Scholar] [CrossRef]
- Wefel, J.S.; Schagen, S.B. Chemotherapy-Related Cognitive Dysfunction. Curr. Neurol. Neurosci. Rep. 2012, 12, 267–275. [Google Scholar] [CrossRef]
- Koppelmans, V.; Breteler, M.M.B.; Boogerd, W.; Seynaeve, C.; Schagen, S.B. Late Effects of Adjuvant Chemotherapy for Adult Onset Non-CNS Cancer; Cognitive Impairment, Brain Structure and Risk of Dementia. Crit. Rev. Oncol. Hematol. 2013, 88, 87–101. [Google Scholar] [CrossRef]
- Jansen, C.; Miaskowski, C.; Dodd, M.; Dowling, G.; Kramer, J. Potential Mechanisms for Chemotherapy-Induced Impairments in Cognitive Function. Oncol. Nurs. Forum 2005, 32, 1151–1163. [Google Scholar] [CrossRef] [Green Version]
- Stivers, N.N. Sex Differences in Taxane Cancer Treatment Toxicities. Available online: https://escholarship.org/uc/item/58p5p3pp#author (accessed on 23 June 2022).
- Walsh, V.; Goodman, J. From Taxol to Taxol®: The Changing Identities and Ownership of an Anti-Cancer Drug. Med. Anthropol. 2002, 21, 307–336. [Google Scholar] [CrossRef]
- Renneberg, R. Biotech History: Yew Trees, Paclitaxel Synthesis and Fungi. Biotechnol. J. 2007, 2, 1207–1209. [Google Scholar] [CrossRef]
- Wiernik, P.H.; Schwartz, E.L.; Strauman, J.J.; Dutcher, J.P.; Lipton, R.B.; Paietta, E. Phase I Clinical and Pharmacokinetic Study of Taxol. Cancer Res. 1987, 47, 2486–2493. [Google Scholar]
- Kampan, N.C.; Madondo, M.T.; McNally, O.M.; Quinn, M.; Plebanski, M. Paclitaxel and Its Evolving Role in the Management of Ovarian Cancer. BioMed Res. Int. 2015, 2015, 413076. [Google Scholar] [CrossRef] [PubMed]
- Weaver, B.A. How Taxol/Paclitaxel Kills Cancer Cells. Mol. Biol. Cell 2014, 25, 2677–2681. [Google Scholar] [CrossRef] [PubMed]
- Wani, M.C.; Taylor, H.L.; Wall, M.E.; Coggon, P.; Mcphail, A.T. Plant Antitumor Agents.VI. The Isolation and Structure of Taxol, a Novel Antileukemic and Antitumor Agent from Taxus brevifolia. J. Am. Chem. Soc. 1971, 93, 2325–2327. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, D.A.; Johnson, R.K. Cytologic Evidence That Taxol, an Antineoplastic Agent from Taxus brevifolia, Acts as a Mitotic Spindle Poison. Cancer Treat. Rep. 1978, 62, 1219–1222. [Google Scholar] [PubMed]
- Schiff, P.B.; Horwitz, S.B. Taxol Stabilizes Microtubules in Mouse Fibroblast Cells. Proc. Natl. Acad. Sci. USA 1980, 77, 1561–1565. [Google Scholar] [CrossRef] [Green Version]
- Schiff, P.B.; Fant, J.; Horwitz, S.B. Promotion of Microtubule Assembly in Vitro by Taxol. Nature 1979, 277, 665–667. [Google Scholar] [CrossRef]
- McGuire, W.P.; Rowinsky, E.K.; Rosenhein, N.B.; Grumbine, F.C.; Ettinger, D.S.; Armstrong, D.K.; Donehower, R.C. Taxol: A Unique Antineoplastic Agent with Significant Activity in Advanced Ovarian Epithelial Neoplasms. Ann. Intern. Med. 1989, 111, 273–279. [Google Scholar] [CrossRef]
- United States Congress; House Committee on Small Business; Subcommittee on Regulation; Business Opportunities, and Energy. Exclusive Agreements between Federal Agencies and Bristol-Myers Squibb Co for Drug Development; United States. Congress. House. Committee on Small Business. Subcommittee on Regulation, Business Opportunities, and Energy: Washington, DC, USA, 1992.
- Tuma, R.S. Taxol’s Journey from Discovery to Use. Oncol. Times 2003, 25, 52–57. [Google Scholar] [CrossRef]
- Yared, J.A.; Tkaczuk, K.H. Update on Taxane Development: New Analogs and New Formulations. Drug Des. Dev. Ther. 2012, 6, 371–384. [Google Scholar] [CrossRef] [Green Version]
- Eisenhauer, E. Docetaxel: Current Status and Future Prospects. J. Clin. Oncol. 1995, 13, 2865–2868. [Google Scholar] [CrossRef]
- Paller, C.J.; Antonarakis, E.S. Cabazitaxel: A Novel Second-Line Treatment for Metastatic Castration-Resistant Prostate Cancer. Drug Des. Dev. Ther. 2011, 5, 117–124. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, E. Gender-Related Differences in Pharmacokinetics and Their Clinical Significance. J. Clin. Pharm. Ther. 1999, 24, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, B.C.; Csajka, C.; Dotto, G.P.; Wagner, A.D. Sex Differences in Efficacy and Toxicity of Systemic Treatments: An Undervalued Issue in the Era of Precision Oncology. J. Clin. Oncol. 2018, 36, 2680–2683. [Google Scholar] [CrossRef]
- Joerger, M.; Huitema, A.D.R.; van den Bongard, D.H.J.G.; Schellens, J.H.M.; Beijnen, J.H. Quantitative Effect of Gender, Age, Liver Function, and Body Size on the Population Pharmacokinetics of Paclitaxel in Patients with Solid Tumors. Clin. Cancer Res. 2006, 12, 2150–2157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schrijvers, D. Role of Red Blood Cells in Pharmacokinetics of Chemotherapeutic Agents. Clin. Pharmacokinet. 2003, 42, 779–791. [Google Scholar] [CrossRef]
- Murphy, W.G. The Sex Difference in Haemoglobin Levels in Adults—Mechanisms, Causes, and Consequences. Blood Rev. 2014, 28, 41–47. [Google Scholar] [CrossRef]
- Maloney, S.M.; Hoover, C.A.; Morejon-Lasso, L.V.; Prosperi, J.R. Mechanisms of Taxane Resistance. Cancers 2020, 12, 3323. [Google Scholar] [CrossRef]
- Thornton, L.M.; Carson, W.E.; Shapiro, C.L.; Farrar, W.B.; Andersen, B.L. Delayed Emotional Recovery after Taxane-Based Chemotherapy. Cancer 2008, 113, 638–647. [Google Scholar] [CrossRef] [Green Version]
- Wheatley-Price, P.; Le Maître, A.; Ding, K.; Leighl, N.; Hirsh, V.; Seymour, L.; Bezjak, A.; Shepherd, F.A. The Influence of Sex on Efficacy, Adverse Events, Quality of Life, and Delivery of Treatment in National Cancer Institute of Canada Clinical Trials Group Non-Small Cell Lung Cancer Chemotherapy Trials. J. Thorac. Oncol. 2010, 5, 640–648. [Google Scholar] [CrossRef] [Green Version]
- Cristina, V.; Mahachie, J.; Mauer, M.; Buclin, T.; van Cutsem, E.; Roth, A.; Wagner, A.D. Association of Patient Sex with Chemotherapy-Related Toxic Effects: A Retrospective Analysis of the PETACC-3 Trial Conducted by the EORTC Gastrointestinal Group. JAMA Oncol. 2018, 4, 1003–1006. [Google Scholar] [CrossRef]
- Miller, K.D.; Nogueira, L.; Mariotto, A.B.; Rowland, J.H.; Yabroff, K.R.; Alfano, C.M.; Jemal, A.; Kramer, J.L.; Siegel, R.L. Cancer Treatment and Survivorship Statistics, 2019. CA Cancer J. Clin. 2019, 69, 363–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [Green Version]
- Klein, S.L.; Flanagan, K.L. Sex Differences in Immune Responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Márquez, E.J.; Chung, C.; Marches, R.; Rossi, R.J.; Nehar-Belaid, D.; Eroglu, A.; Mellert, D.J.; Kuchel, G.A.; Banchereau, J.; Ucar, D. Sexual-Dimorphism in Human Immune System Aging. Nat. Commun. 2020, 11, 751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roved, J.; Westerdahl, H.; Hasselquist, D. Sex Differences in Immune Responses: Hormonal Effects, Antagonistic Selection, and Evolutionary Consequences. Horm. Behav. 2017, 88, 95–105. [Google Scholar] [CrossRef]
- Weiss, R.B.; Donehower, R.C.; Wiernik, P.H.; Ohnuma, T.; Gralla, R.J.; Trump, D.L.; Baker, R., Jr.; van Echo, D.A.; von Hoff, D.D.; Leyland-Jones, B. Hypersensitivity Reactions from Taxol. J. Clin. Oncol. 2016, 8, 1263–1268. [Google Scholar] [CrossRef]
- Castells, M.C.; Tennant, N.M.; Sloane, D.E.; Hsu, F.I.; Barrett, N.A.; Hong, D.I.; Laidlaw, T.M.; Legere, H.J.; Nallamshetty, S.N.; Palis, R.I.; et al. Hypersensitivity Reactions to Chemotherapy: Outcomes and Safety of Rapid Desensitization in 413 Cases. J. Allergy Clin. Immunol. 2008, 122, 574–580. [Google Scholar] [CrossRef]
- Limsuwan, T.; Castells, M. Outcomes and Safety of Rapid Desensitization for Chemotherapy Hypersensitivity. Expert Opin. Drug Saf. 2010, 9, 39–53. [Google Scholar] [CrossRef]
- Picard, M.; Castells, M.C. Re-Visiting Hypersensitivity Reactions to Taxanes: A Comprehensive Review. Clin. Rev. Allergy Immunol. 2014, 49, 177–191. [Google Scholar] [CrossRef]
- Lansinger, O.M.; Biedermann, S.; He, Z.; Colevas, A.D. Do Steroids Matter? A Retrospective Review of Premedication for Taxane Chemotherapy and Hypersensitivity Reactions. J. Clin. Oncol. 2021, 39, 3583–3590. [Google Scholar] [CrossRef]
- Kemper, E.M.; van Zandbergen, A.E.; Cleypool, C.; Mos, H.A.; Boogerd, W.; Beijnen, J.H.; van Tellingen, O. Increased Penetration of Paclitaxel into the Brain by Inhibition of P-Glycoprotein. Clin. Cancer Res. 2003, 9, 2849–2855. [Google Scholar]
- Balayssac, D.; Cayre, A.; Authier, N.; Bourdu, S.; Penault-Llorca, F.; Gillet, J.P.; Maublant, J.; Eschalier, A.; Coudore, F. Patterns of P-Glycoprotein Activity in the Nervous System during Vincristine-Induced Neuropathy in Rats. J. Peripher. Nerv. Syst. 2005, 10, 301–310. [Google Scholar] [CrossRef]
- Cisternino, S.; Bourasset, F.; Archimbaud, Y.; Sémiond, D.; Sanderink, G.; Scherrmann, J.-M. Nonlinear Accumulation in the Brain of the New Taxoid TXD258 Following Saturation of P-Glycoprotein at the Blood–Brain Barrier in Mice and Rats. Br. J. Pharmacol. 2003, 138, 1367–1375. [Google Scholar] [CrossRef] [Green Version]
- Huehnchen, P.; Boehmerle, W.; Springer, A.; Freyer, D.; Endres, M. A Novel Preventive Therapy for Paclitaxel-Induced Cognitive Deficits: Preclinical Evidence from C57BL/6 Mice. Transl. Psychiatry 2017, 7, e1185. [Google Scholar] [CrossRef] [Green Version]
- Montague, K.; Malcangio, M. The Therapeutic Potential of Monocyte/Macrophage Manipulation in the Treatment of Chemotherapy-Induced Painful Neuropathy. Front. Mol. Neurosci. 2017, 10, 397. [Google Scholar] [CrossRef]
- da Costa, R.; Passos, G.F.; Quintão, N.L.M.; Fernandes, E.S.; Maia, J.R.L.C.B.; Campos, M.M.; Calixto, J.B. Taxane-Induced Neurotoxicity: Pathophysiology and Therapeutic Perspectives. Br. J. Pharmacol. 2020, 177, 3127–3146. [Google Scholar] [CrossRef] [PubMed]
- Scripture, C.D.; Figg, W.D.; Sparreboom, A. Peripheral Neuropathy Induced by Paclitaxel: Recent Insights and Future Perspectives. Curr. Neuropharmacol. 2006, 4, 165–172. [Google Scholar] [CrossRef] [Green Version]
- Tofthagen, C.; McAllister, R.D.; Visovsky, C. Peripheral Neuropathy Caused by Paclitaxel and Docetaxel: An Evaluation and Comparison of Symptoms. J. Adv. Pract. Oncol. 2013, 4, 204–215. [Google Scholar] [CrossRef]
- Augusto, C.; Pietro, M.; Cinzia, M.; Sergio, C.; Sara, C.; Luca, G.; Scaioli, V. Peripheral Neuropathy Due to Paclitaxel: Study of the Temporal Relationships between the Therapeutic Schedule and the Clinical Quantitative Score (QST) and Comparison with Neurophysiological Findings. J. Neuro-Oncol. 2008, 86, 89–99. [Google Scholar] [CrossRef]
- Sarosy, G.; Kohn, E.; Stone, D.A.; Rothenberg, M.; Jacob, J.; Adamo, D.O.; Ognibene, F.P.; Cunnion, R.E.; Reed, E. Phase I Study of Taxol and Granulocyte Colony-Stimulating Factor in Patients with Refractory Ovarian Cancer. J. Clin. Oncol. 1992, 10, 1165–1170. [Google Scholar] [CrossRef]
- Seretny, M.; Currie, G.L.; Sena, E.S.; Ramnarine, S.; Grant, R.; Macleod, M.R.; Colvin, L.A.; Fallon, M. Incidence, Prevalence, and Predictors of Chemotherapy-Induced Peripheral Neuropathy: A Systematic Review and Meta-Analysis. Pain® 2014, 155, 2461–2470. [Google Scholar] [CrossRef] [Green Version]
- Tanabe, Y.; Hashimoto, K.; Shimizu, C.; Hirakawa, A.; Harano, K.; Yunokawa, M.; Yonemori, K.; Katsumata, N.; Tamura, K.; Ando, M.; et al. Paclitaxel-Induced Peripheral Neuropathy in Patients Receiving Adjuvant Chemotherapy for Breast Cancer. Int. J. Clin. Oncol. 2011, 18, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Nakai, Y.; Tanaka, N.; Ichikawa, K.; Miyake, M.; Anai, S.; Fujimoto, K. Appropriate Number of Docetaxel Cycles in Castration-Resistant Prostate Cancer Patients Considering Peripheral Neuropathy and Oncological Control. Chemotherapy 2020, 65, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Winters-Stone, K.M.; Horak, F.; Jacobs, P.G.; Trubowitz, P.; Dieckmann, N.F.; Stoyles, S.; Faithfull, S. Falls, Functioning, and Disability Among Women with Persistent Symptoms of Chemotherapy-Induced Peripheral Neuropathy. J. Clin. Oncol. 2017, 35, 2604–2612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kober, K.M.; Mazor, M.; Abrams, G.; Olshen, A.; Conley, Y.P.; Hammer, M.; Schumacher, M.; Chesney, M.; Smoot, B.; Mastick, J.; et al. Phenotypic Characterization of Paclitaxel-Induced Peripheral Neuropathy in Cancer Survivors. J. Pain Symptom Manag. 2018, 56, 908–919.e3. [Google Scholar] [CrossRef]
- Windebank, A.J.; Grisold, W. Chemotherapy-Induced Neuropathy. J. Peripher. Nerv. Syst. 2008, 13, 27–46. [Google Scholar] [CrossRef]
- Weimer, L.H. Medication-Induced Peripheral Neuropathy. Curr. Neurol. Neurosci. Rep. 2003, 3, 86–92. [Google Scholar] [CrossRef]
- Mogil, J.S. Sex Differences in Pain and Pain Inhibition: Multiple Explanations of a Controversial Phenomenon. Nat. Rev. Neurosci. 2012, 13, 859–866. [Google Scholar] [CrossRef]
- Vallejo, R.; Tilley, D.M.; Vogel, L.; Benyamin, R. The Role of Glia and the Immune System in the Development and Maintenance of Neuropathic Pain. Pain Pract. 2010, 10, 167–184. [Google Scholar] [CrossRef]
- Boyette-Davis, J.; Xin, W.; Zhang, H.; Dougherty, P.M. Intraepidermal Nerve Fiber Loss Corresponds to the Development of Taxol-Induced Hyperalgesia and Can Be Prevented by Treatment with Minocycline. Pain® 2011, 152, 308–313. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, H.; Zhang, H.; Kosturakis, A.K.; Jawad, A.B.; Dougherty, P.M. Toll-Like Receptor 4 Signaling Contributes to Paclitaxel-Induced Peripheral Neuropathy. J. Pain 2014, 15, 712–725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosen, S.; Ham, B.; Mogil, J.S. Sex Differences in Neuroimmunity and Pain. J. Neurosci. Res. 2017, 95, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Gregus, A.M.; Levine, I.S.; Eddinger, K.A.; Yaksh, T.L.; Buczynski, M.W. Sex Differences in Neuroimmune and Glial Mechanisms of Pain. Pain® 2021, 162, 2186–2200. [Google Scholar] [CrossRef]
- Sorge, R.E.; Mapplebeck, J.C.S.; Rosen, S.; Beggs, S.; Taves, S.; Alexander, J.K.; Martin, L.J.; Austin, J.-S.; Sotocinal, S.G.; Chen, D.; et al. Different Immune Cells Mediate Mechanical Pain Hypersensitivity in Male and Female Mice. Nat. Neurosci. 2015, 18, 1081–1083. [Google Scholar] [CrossRef] [Green Version]
- Luo, X.; Huh, Y.; Bang, S.; He, Q.; Zhang, L.; Matsuda, M.; Ji, R.-R. Macrophage Toll-like Receptor 9 Contributes to Chemotherapy-Induced Neuropathic Pain in Male Mice. J. Neurosci. 2019, 39, 6848–6864. [Google Scholar] [CrossRef]
- Wang, Y.; Li, N.; Zhao, Y.; Zhang, L.J. Effects of Female Sex Hormones on Chemotherapeutic Paclitaxel-Induced Neuropathic Pain and Involvement of Inflammatory Signal. J. Biol. Regul. Homeost. Agents 2018, 32, 1157–1163. [Google Scholar]
- Bale, T.L.; Epperson, C.N. Sex Differences and Stress across the Lifespan. Nat. Neurosci. 2015, 18, 1413–1420. [Google Scholar] [CrossRef]
- Ferrari, L.F.; Araldi, D.; Green, P.; Levine, J.D. Marked Sexual Dimorphism in Neuroendocrine Mechanisms for the Exacerbation of Paclitaxel-Induced Painful Peripheral Neuropathy by Stress. Pain 2020, 161, 865–874. [Google Scholar] [CrossRef]
- Luo, X.; Gu, Y.; Tao, X.; Serhan, C.N.; Ji, R.-R. Resolvin D5 Inhibits Neuropathic and Inflammatory Pain in Male but Not Female Mice: Distinct Actions of D-Series Resolvins in Chemotherapy-Induced Peripheral Neuropathy. Front. Pharmacol. 2019, 10, 745. [Google Scholar] [CrossRef]
- Lopes, D.M.; Malek, N.; Edye, M.; Jager, S.B.; McMurray, S.; McMahon, S.B.; Denk, F. Sex Differences in Peripheral Not Central Immune Responses to Pain-Inducing Injury. Sci. Rep. 2017, 7, 16460. [Google Scholar] [CrossRef] [Green Version]
- Parvathy, S.S.; Masocha, W. Coadministration of Indomethacin and Minocycline Attenuates Established Paclitaxel-Induced Neuropathic Thermal Hyperalgesia: Involvement of Cannabinoid CB1 Receptors. Sci. Rep. 2015, 5, 10541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Natri, H.; Garcia, A.R.; Buetow, K.H.; Trumble, B.C.; Wilson, M.A. The Pregnancy Pickle: Evolved Immune Compensation Due to Pregnancy Underlies Sex Differences in Human Diseases. Trends Genet. 2019, 35, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, J.; Kaiser, J. Cancer, Chemotherapy and Cognitive Dysfunction. US Neurol. 2016, 12, 43–45. [Google Scholar] [CrossRef] [Green Version]
- Vitali, M.; Ripamonti, C.I.; Roila, F.; Proto, C.; Signorelli, D.; Imbimbo, M.; Corrao, G.; Brissa, A.; Rosaria, G.; de Braud, F.; et al. Cognitive Impairment and Chemotherapy: A Brief Overview. Crit. Rev. Oncol. Hematol. 2017, 118, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Myers, J.S. Chemotherapy-Related Cognitive Impairment: The Breast Cancer Experience. Oncol. Nurs. Forum 2012, 39, E31–E40. [Google Scholar] [CrossRef] [Green Version]
- Loh, K.P.; Janelsins, M.C.; Mohile, S.G.; Holmes, H.M.; Hsu, T.; Inouye, S.K.; Karuturi, M.S.; Kimmick, G.G.; Lichtman, S.M.; Magnuson, A.; et al. Chemotherapy-Related Cognitive Impairment in Older Patients with Cancer. J. Geriatr. Oncol. 2016, 7, 270–280. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Caeyenberghs, K. Longitudinal Assessment of Chemotherapy-Induced Changes in Brain and Cognitive Functioning: A Systematic Review. Neurosci. Biobehav. Rev. 2018, 92, 304–317. [Google Scholar] [CrossRef]
- Argyriou, A.A.; Assimakopoulos, K.; Iconomou, G.; Giannakopoulou, F.; Kalofonos, H.P. Either Called “Chemobrain” or “Chemofog,” the Long-Term Chemotherapy-Induced Cognitive Decline in Cancer Survivors Is Real. J. Pain Symptom Manag. 2011, 41, 126–139. [Google Scholar] [CrossRef]
- Janelsins, M.C.; Heckler, C.E.; Peppone, L.J.; Kamen, C.; Mustian, K.M.; Mohile, S.G.; Magnuson, A.; Kleckner, I.R.; Guido, J.J.; Young, K.L.; et al. Cognitive Complaints in Survivors of Breast Cancer after Chemotherapy Compared with Age-Matched Controls: An Analysis from a Nationwide, Multicenter, Prospective Longitudinal Study. J. Clin. Oncol. Am. Soc. Clin. Oncol. 2017, 35, 506–514. [Google Scholar] [CrossRef]
- Liang, L.; Wei, J.; Tian, L.; Nagendra, B.V.P.; Gao, F.; Zhang, J.; Xu, L.; Wang, H.; Huo, F.Q. Paclitaxel Induces Sex-Biased Behavioral Deficits and Changes in Gene Expression in Mouse Prefrontal Cortex. Neuroscience 2020, 426, 168–178. [Google Scholar] [CrossRef]
- Chmielewski-Stivers, N.; Petit, B.; Ollivier, J.; Monceau, V.; Tsoutsou, P.; Pousa, A.Q.; Lin, X.; Limoli, C.; Vozenin, M.-C. Sex-Specific Differences in Toxicity Following Systemic Paclitaxel Treatment and Localized Cardiac Radiotherapy. Cancers 2021, 13, 3973. [Google Scholar] [CrossRef] [PubMed]
- Hausmann, M. Why Sex Hormones Matter for Neuroscience: A Very Short Review on Sex, Sex Hormones, and Functional Brain Asymmetries. J. Neurosci. Res. 2017, 95, 40–49. [Google Scholar] [CrossRef] [Green Version]
- Gaignard, P.; Liere, P.; Thérond, P.; Schumacher, M.; Slama, A.; Guennoun, R. Role of Sex Hormones on Brain Mitochondrial Function, with Special Reference to Aging and Neurodegenerative Diseases. Front. Aging Neurosci. 2017, 9, 406. [Google Scholar] [CrossRef]
- Médale-Giamarchi, C.; Lajoie-Mazenc, I.; Malissein, E.; Meunier, E.; Couderc, B.; Bergé, Y.; Filleron, T.; Keller, L.; Marty, C.; Lacroix-Triki, M.; et al. RhoB Modifies Estrogen Responses in Breast Cancer Cells by Influencing Expression of the Estrogen Receptor. Breast Cancer Res. 2013, 15, R6. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Lian, C.L.; Zheng, H.; Lin, L.E.; Yu, Y.F.; Lin, Q.; Wu, S.G. Cognitive Dysfunction in Patients with Nasopharyngeal Carcinoma after Induction Chemotherapy. Oral Oncol. 2020, 111, 104921. [Google Scholar] [CrossRef] [PubMed]
- Cerulla, N.; Arcusa, À.; Navarro, J.-B.; Garolera, M.; Enero, C.; Chico, G.; Fernández-Morales, L. Role of Taxanes in Chemotherapy-Related Cognitive Impairment: A Prospective Longitudinal Study. Breast Cancer Res. Treat. 2017, 164, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Wefel, J.S.; Saleeba, A.K.; Buzdar, A.U.; Meyers, C.A. Acute and Late Onset Cognitive Dysfunction Associated with Chemotherapy in Women with Breast Cancer. Cancer 2010, 116, 3348–3356. [Google Scholar] [CrossRef] [Green Version]
- Brown, T.; McElroy, T.; Simmons, P.; Walters, H.; Ntagwabira, F.; Wang, J.; Byrum, S.D.; Allen, A.R. Cognitive Impairment Resulting from Treatment with Docetaxel, Doxorubicin, and Cyclophosphamide. Brain Res. 2021, 1760, 147397. [Google Scholar] [CrossRef]
- Alibhai, S.M.H.; Breunis, H.; Timilshina, N.; Hansen, A.R.; Joshua, A.M.; Warde, P.R.; Gregg, R.W.; Fleshner, N.E.; Tomlinson, G.; Hotte, S.J.; et al. The Effect of Docetaxel, Enzalutamide, Abiraterone, and Radium-223 on Cognitive Function in Older Men with Metastatic Castrate-Resistant Prostate Cancer (MCRPC). J. Clin. Oncol. 2020, 38, 73. [Google Scholar] [CrossRef]
- Lange, M.; Heutte, N.; Rigal, O.; Noal, S.; Kurtz, J.-E.; Lévy, C.; Allouache, D.; Rieux, C.; Lefel, J.; Clarisse, B.; et al. Decline in Cognitive Function in Older Adults with Early-Stage Breast Cancer After Adjuvant Treatment. Oncologist 2016, 21, 1337–1348. [Google Scholar] [CrossRef] [Green Version]
- Fardell, J.E.; Zhang, J.; de Souza, R.; Vardy, J.; Johnston, I.; Allen, C.; Henderson, J.; Piquette-Miller, M. The Impact of Sustained and Intermittent Docetaxel Chemotherapy Regimens on Cognition and Neural Morphology in Healthy Mice. Psychopharmacology 2013, 231, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Seigers, R.; Loos, M.; van Tellingen, O.; Boogerd, W.; Smit, A.B.; Schagen, S.B. Cognitive Impact of Cytotoxic Agents in Mice. Psychopharmacology 2014, 232, 17–37. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, C.K.; O’Mara, S.M. Long-Term Cognitive Dysfunction in the Rat Following Docetaxel Treatment Is Ameliorated by the Phosphodiesterase-4 Inhibitor, Rolipram. Behav. Brain Res. 2015, 290, 84–89. [Google Scholar] [CrossRef]
- Panoz-Brown, D.; Carey, L.M.; Smith, A.E.; Gentry, M.; Sluka, C.M.; Corbin, H.E.; Wu, J.E.; Hohmann, A.G.; Crystal, J.D. The Chemotherapeutic Agent Paclitaxel Selectively Impairs Reversal Learning While Sparing Prior Learning, New Learning and Episodic Memory. Neurobiol. Learn. Mem. 2017, 144, 259–270. [Google Scholar] [CrossRef] [Green Version]
- Office of Science Policy. Significant Research Advances Enabled by HeLa Cells. Available online: https://osp.od.nih.gov/scientific-sharing/hela-cells-timeline/ (accessed on 16 August 2021).
- Horwitz, S.B.; Goldman, I.D. A Conversation with Susan Band Horwitz. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Brabander, M.; Geuens, G.; Nuydens, R.; Willebrords, R.; de Mey, J. Taxol Induces the Assembly of Free Microtubules in Living Cells and Blocks the Organizing Capcity of the Centrosomes and Kinetochores. Proc. Natl. Acad. Sci. USA 1981, 78, 5608–5612. [Google Scholar] [CrossRef] [Green Version]
- Shi, J.; Orth, J.D.; Mitchison, T. Cell Type Variation in Responses to Antimitotic Drugs That Target Microtubules and Kinesin-5. Cancer Res. 2008, 68, 3269–3276. [Google Scholar] [CrossRef] [Green Version]
- Milas, L.; Hunter, N.R.; Kurdoglu, B.; Mason, K.A.; Meyn, R.E.; Stephens, L.C.; Peters, L.J. Kinetics of Mitotic Arrest and Apoptosis in Murine Mammary and Ovarian Tumors Treated with Taxol. Cancer Chemother. Pharmacol. 1995, 35, 297–303. [Google Scholar] [CrossRef]
- Waters, J.C.; Chen, R.H.; Murray, A.W.; Salmon, E.D. Localization of Mad2 to Kinetochores Depends on Microtubule Attachment, Not Tension. J. Cell Biol. 1998, 141, 1181–1191. [Google Scholar] [CrossRef] [Green Version]
- Sudo, T.; Nitta, M.; Saya, H.; Ueno, N.T. Dependence of Paclitaxel Sensitivity on a Functional Spindle Assembly Checkpoint. Cancer Res. 2004, 64, 2502–2508. [Google Scholar] [CrossRef] [Green Version]
- Komlodi-Pasztor, E.; Sackett, D.; Wilkerson, J.; Fojo, T. Mitosis Is Not a Key Target of Microtubule Agents in Patient Tumors. Nat. Rev. Clin. Oncol. 2011, 8, 244–250. [Google Scholar] [CrossRef]
- Mitchison, T.J. The Proliferation Rate Paradox in Antimitotic Chemotherapy. Mol. Biol. Cell 2012, 23, 1–6. [Google Scholar] [CrossRef]
- Milross, C.G.; Mason, K.A.; Hunter, N.R.; Chung, W.K.; Peters, L.J.; Milas, L. Relationship of Mitotic Arrest and Apoptosis to Antitumor Effect of Paclitaxel. J. Natl. Cancer Inst. 1996, 88, 1308–1314. [Google Scholar] [CrossRef]
- Chakravarty, A.; Shinde, V.; Tabernero, J.; Cervantes, A.; Cohen, R.B.; Dees, E.C.; Burris, H.; Infante, J.R.; Macarulla, T.; Elez, E.; et al. Phase I Assessment of New Mechanism-Based Pharmacodynamic Biomarkers for MLN8054, a Small-Molecule Inhibitor of Aurora A Kinase. Cancer Res. 2011, 71, 675–685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Florian, S.; Mitchison, T.J. Anti-Microtubule Drugs. In The Mitotic Spindle. Methods in Molecular Biology; Chang, P., Ohi, R., Eds.; Humana Press Inc.: New York, NY, USA, 2016. [Google Scholar] [CrossRef]
- Gundersen, G.G.; Cook, T.A. Microtubules and Signal Transduction. Curr. Opin. Cell Biol. 1999, 11, 81–94. [Google Scholar] [CrossRef]
- Etienne-Manneville, S. From Signaling Pathways to Microtubule Dynamics: The Key Players. Curr. Opin. Cell Biol. 2010, 22, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Zasadil, L.M.; Andersen, K.A.; Yeum, D.; Rocque, G.B.; Wilke, L.G.; Tevaarwerk, A.J.; Raines, R.T.; Burkard, M.E.; Weaver, B.A. Cytotoxicity of Paclitaxel in Breast Cancer Is Due to Chromosome Missegregation on Multipolar Spindles. Sci. Transl. Med. 2014, 6, 229ra43. [Google Scholar] [CrossRef] [Green Version]
- Thadani-Mulero, M.; Nanus, D.M.; Giannakakou, P. Androgen Receptor on the Move: Boarding the Microtubule Expressway to the Nucleus. Cancer Res. 2012, 72, 4611–4615. [Google Scholar] [CrossRef] [Green Version]
- Carbonaro, M.; Escuin, D.; O’Brate, A.; Thadani-Mulero, M.; Giannakakou, P. Microtubules Regulate Hypoxia-Inducible Factor-1α Protein Trafficking and Activity: Implications for Taxane Therapy. J. Biol. Chem. 2012, 287, 11859–11869. [Google Scholar] [CrossRef] [Green Version]
- Darshan, M.S.; Loftus, M.S.; Thadani-Mulero, M.; Levy, B.P.; Escuin, D.; Zhou, X.K.; Gjyrezi, A.; Chanel-Vos, C.; Shen, R.; Tagawa, S.T.; et al. Taxane-Induced Blockade to Nuclear Accumulation of the Androgen Receptor Predicts Clinical Responses in Metastatic Prostate Cancer. Cancer Res. 2011, 71, 6019–6029. [Google Scholar] [CrossRef] [Green Version]
- Jordan, M.A.; Wendell, K.; Gardiner, S.; Brent Derry, W.; Copp, H.; Wilson, L. Mitotic Block Induced in HeLa Cells by Low Concentrations of Paclitaxel (Taxol) Results in Abnormal Mitotic Exit and Apoptotic Cell Death. Cancer Res. 1996, 56, 816–825. [Google Scholar] [PubMed]
- Torres, K.; Horwitz, S.B. Mechanisms of Taxol-Induced Cell Death Are Concentration Dependent. Cancer Res. 1998, 58, 3620–3626. [Google Scholar] [PubMed]
- Jordan, M.A.; Toso, R.J.; Thrower, D.; Wilson, L. Mechanism of Mitotic Block and Inhibition of Cell Proliferation by Taxol at Low Concentrations. Proc. Natl. Acad. Sci. USA 1993, 90, 9552–9556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ikui, A.E.; Chia-Ping, H.Y.; Matsumoto, T.; Horwitz, S.B. Low Concentrations of Taxol Cause Mitotic Delay Followed by Premature Dissociation of P55CDC from Mad2 and BubR1 and Abrogation of the Spindle Checkpoint, Leading to Aneuploidy. Cell Cycle 2005, 4, 1385–1388. [Google Scholar] [CrossRef] [Green Version]
- Zhu, M.-L.; Horbinski, C.M.; Garzotto, M.; Qian, D.Z.; Beer, T.M.; Kyprianou, N. Tubulin-Targeting Chemotherapy Impairs Androgen Receptor Activity in Prostate Cancer. Cancer Res. 2010, 70, 7992–8002. [Google Scholar] [CrossRef] [Green Version]
- Feldman, B.J.; Feldman, D. The Development of Androgen-Independent Prostate Cancer. Nat. Rev. Cancer 2001, 1, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Watson, P.A.; Arora, V.K.; Sawyers, C.L. Emerging Mechanisms of Resistance to Androgen Receptor Inhibitors in Prostate Cancer. Nat. Rev. Cancer 2015, 15, 701–711. [Google Scholar] [CrossRef] [Green Version]
- Attard, G.; Richards, J.; de Bono, J.S. New Strategies in Metastatic Prostate Cancer: Targeting the Androgen Receptor Signaling Pathway. Clin. Cancer Res. 2011, 17, 1649–1657. [Google Scholar] [CrossRef] [Green Version]
- Antonarakis, E.S.; Tagawa, S.T.; Galletti, G.; Worroll, D.; Ballman, K.; Vanhuyse, M.; Sonpavde, G.; North, S.; Albany, C.; Tsao, C.-K.; et al. Randomized, Noncomparative, Phase II Trial of Early Switch from Docetaxel to Cabazitaxel or Vice Versa, with Integrated Biomarker Analysis, in Men with Chemotherapy-Naïve, Metastatic, Castration-Resistant Prostate Cancer. J. Clin. Oncol. 2017, 35, 3181–3188. [Google Scholar] [CrossRef] [Green Version]
- de Bono, J.S.; Oudard, S.; Ozguroglu, M.; Hansen, S.; Machiels, J.-P.; Kocak, I.; Gravis, G.; Bodrogi, I.; Mackenzie, M.J.; Shen, L.; et al. Prednisone plus Cabazitaxel or Mitoxantrone for Metastatic Castration-Resistant Prostate Cancer Progressing after Docetaxel Treatment: A Randomised Open-Label Trial. Lancet 2010, 376, 1147–1154. [Google Scholar] [CrossRef]
- Petrylak, D.P.; Tangen, C.M.; Hussain, M.H.A.; Lara, P.N.; Jones, J.A.; Taplin, M.E.; Burch, P.A.; Berry, D.; Moinpour, C.; Kohli, M.; et al. Docetaxel and Estramustine Compared with Mitoxantrone and Prednisone for Advanced Refractory Prostate Cancer. N. Engl. J. Med. 2004, 351, 1513–1520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tannock, I.F.; de Wit, R.; Berry, W.R.; Horti, J.; Pluzanska, A.; Chi, K.N.; Oudard, S.; Théodore, C.; James, N.D.; Turesson, I.; et al. Docetaxel plus Prednisone or Mitoxantrone plus Prednisone for Advanced Prostate Cancer. N. Engl. J. Med. 2004, 351, 1502–1512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuroda, K.; Liu, H.; Kim, S.; Guo, M.; Navarro, V.; Bander, N.H. Docetaxel Down-Regulates the Expression of Androgen Receptor and Prostate-Specific Antigen but Not Prostate-Specific Membrane Antigen in Prostate Cancer Cell Lines: Implications for PSA Surrogacy. Prostate 2009, 69, 1579–1585. [Google Scholar] [CrossRef]
- Gan, L.; Chen, S.; Wang, Y.; Watahiki, A.; Bohrer, L.; Sun, Z.; Wang, Y.; Huang, H. Inhibition of the Androgen Receptor as a Novel Mechanism of Taxol Chemotherapy in Prostate Cancer. Cancer Res. 2009, 69, 8386–8394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kampa, M.; Kogia, C.; Theodoropoulos, P.A.; Anezinis, P.; Charalampopoulos, I.; Papakonstanti, E.A.; Stathopoulos, E.N.; Hatzoglou, A.; Stournaras, C.; Gravanis, A.; et al. Activation of Membrane Androgen Receptors Potentiates the Antiproliferative Effects of Paclitaxel on Human Prostate Cancer Cells. Mol. Cancer Ther. 2006, 5, 1342–1351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lai, J.-J.; Lai, K.-P.; Zeng, W.; Chuang, K.-H.; Altuwaijri, S.; Chang, C. Androgen Receptor Influences on Body Defense System via Modulation of Innate and Adaptive Immune Systems: Lessons from Conditional AR Knockout Mice. Am. J. Pathol. 2012, 181, 1504–1512. [Google Scholar] [CrossRef] [Green Version]
- Yeh, S.; Tsai, M.-Y.; Xu, Q.; Mu, X.-M.; Lardy, H.; Huang, K.-E.; Lin, H.; Yeh, S.-D.; Altuwaijri, S.; Zhou, X.; et al. Generation and Characterization of Androgen Receptor Knockout (ARKO) Mice: An in Vivo Model for the Study of Androgen Functions in Selective Tissues. Proc. Natl. Acad. Sci. USA 2002, 99, 13498–13503. [Google Scholar] [CrossRef] [Green Version]
- Kerkhofs, S.; Denayer, S.; Haelens, A.; Claessens, F. Androgen Receptor Knockout and Knock-in Mouse Models. J. Mol. Endocrinol. 2009, 42, 11–17. [Google Scholar] [CrossRef] [Green Version]
- Venken, K.; de Gendt, K.; Boonen, S.; Ophoff, J.; Bouillon, R.; Swinnen, J.V.; Verhoeven, G.; Vanderschueren, D. Relative Impact of Androgen and Estrogen Receptor Activation in the Effects of Androgens on Trabecular and Cortical Bone in Growing Male Mice: A Study in the Androgen Receptor Knockout Mouse Model. J. Bone Miner. Res. 2006, 21, 576–585. [Google Scholar] [CrossRef]
- Komura, K.; Jeong, S.H.; Hinohara, K.; Qu, F.; Wang, X.; Hiraki, M.; Azuma, H.; Lee, G.S.M.; Kantoff, P.W.; Sweeney, C.J. Resistance to Docetaxel in Prostate Cancer Is Associated with Androgen Receptor Activation and Loss of KDM5D Expression. Proc. Natl. Acad. Sci. USA 2016, 113, 6259–6264. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Chi, P.; Rockowitz, S.; Iaquinta, P.J.; Shamu, T.; Shukla, S.; Gao, D.; Sirota, I.; Carver, B.S.; Wongvipat, J.; et al. ETS Factors Reprogram the Androgen Receptor Cistrome and Prime Prostate Tumorigenesis in Response to PTEN Loss. Nat. Med. 2013, 19, 1023–1029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, J.; Yu, J.; Mani, R.-S.; Cao, Q.; Brenner, C.J.; Cao, X.; Wang, X.; Wu, L.; Li, J.; Hu, M.; et al. An Integrated Network of Androgen Receptor, Polycomb, and TMPRSS2-ERG Gene Fusions in Prostate Cancer Progression. Cancer Cell 2010, 17, 443–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galletti, G.; Matov, A.; Beltran, H.; Fontugne, J.; Miguel Mosquera, J.; Cheung, C.; MacDonald, T.Y.; Sung, M.; O’Toole, S.; Kench, J.G.; et al. ERG Induces Taxane Resistance in Castration-Resistant Prostate Cancer. Nat. Commun. 2014, 5, 5548. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Jamalruddin, M.A.B.; Au, C.C.; Mukhtar, E.; Portella, L.; Berger, A.; Worroll, D.; Rickman, D.S.; Nanus, D.M.; Giannakakou, P. AR-V7 Exhibits Non-Canonical Mechanisms of Nuclear Import and Chromatin Engagement in Castrate-Resistant Prostate Cancer. bioRxiv 2021. [Google Scholar] [CrossRef]
- Hu, R.; Dunn, T.A.; Wei, S.; Isharwal, S.; Veltri, R.W.; Humphreys, E.; Han, M.; Partin, A.W.; Vessella, R.L.; Isaacs, W.B.; et al. Ligand-Independent Androgen Receptor Variants Derived from Splicing of Cryptic Exons Signify Hormone-Refractory Prostate Cancer. Cancer Res. 2009, 69, 16–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hörnberg, E.; Ylitalo, E.B.; Crnalic, S.; Antti, H.; Stattin, P.; Widmark, A.; Bergh, A.; Wikström, P. Expression of Androgen Receptor Splice Variants in Prostate Cancer Bone Metastases Is Associated with Castration-Resistance and Short Survival. PLoS ONE 2011, 6, e19059. [Google Scholar] [CrossRef] [Green Version]
- Maughan, B.L.; Antonarakis, E.S. Clinical Relevance of Androgen Receptor Splice Variants in Castration-Resistant Prostate Cancer. Curr. Treat. Options Oncol. 2015, 16, 57. [Google Scholar] [CrossRef]
- Tagawa, S.T.; Antonarakis, E.S.; Gjyrezi, A.; Galletti, G.; Kim, S.; Worroll, D.; Stewart, J.; Zaher, A.; Szatrowski, T.P.; Ballman, K.V.; et al. Expression of AR-V7 and ARv567es in Circulating Tumor Cells Correlates with Outcomes to Taxane Therapy in Men with Metastatic Prostate Cancer Treated in TAXYNERGY. Clin. Cancer Res. 2019, 25, 1880–1888. [Google Scholar] [CrossRef] [Green Version]
- Rizzo, A.; Mollica, V.; Rosellini, M.; Marchetti, A.; Ricci, A.D.; Fiorentino, M.; Battelli, N.; Santoni, M.; Massari, F. Exploring the Association between Metastatic Sites and Androgen Receptor Splice Variant 7 (AR-V7) in Castration-Resistant Prostate Cancer Patients: A Meta-Analysis of Prospective Clinical Trials. Pathol. Res. Pract. 2021, 222, 153440. [Google Scholar] [CrossRef]
- Antonarakis, E.S.; Lu, C.; Wang, H.; Luber, B.; Nakazawa, M.; Roeser, J.C.; Chen, Y.; Mohammad, T.A.; Chen, Y.; Fedor, H.L.; et al. AR-V7 and Resistance to Enzalutamide and Abiraterone in Prostate Cancer. N. Engl. J. Med. 2014, 371, 1028–1038. [Google Scholar] [CrossRef] [Green Version]
- Kipp, J.L.; Ramirez, V.D. Estradiol and Testosterone Have Opposite Effects on Microtubule Polymerization. Neuroendocrinology 2003, 77, 258–272. [Google Scholar] [CrossRef] [PubMed]
- Butler, R.; Leigh, P.N.; Gallo, J.-M. Androgen-Induced up-Regulation of Tubulin Isoforms in Neuroblastoma Cells. J. Neurochem. 2001, 78, 854–861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lo, Y.C.; Cormier, O.; Liu, T.; Nettles, K.W.; Katzenellenbogen, J.A.; Stearns, T.; Altman, R.B. Pocket Similarity Identifies Selective Estrogen Receptor Modulators as Microtubule Modulators at the Taxane Site. Nat. Commun. 2019, 10, 1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dougherty, M.K.; Schumaker, L.M.; Jordan, V.C.; Welshons, W.V.; Curran, E.M.; Ellis, M.J.; El-Ashry, D. Estrogen Receptor Expression and Sensitivity to Paclitaxel in Breast Cancer. Cancer Biol. Ther. 2004, 3, 460–467. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, T.; Iwasaki, A. Sex Differences in Immune Responses. Science 2021, 371, 347–348. [Google Scholar] [CrossRef]
- Takahashi, T.; Ellingson, M.K.; Wong, P.; Israelow, B.; Lucas, C.; Klein, J.; Silva, J.; Mao, T.; Oh, J.E.; Tokuyama, M.; et al. Sex Differences in Immune Responses That Underlie COVID-19 Disease Outcomes. Nature 2020, 588, 315–320. [Google Scholar] [CrossRef]
- Scully, E.P.; Haverfield, J.; Ursin, R.L.; Tannenbaum, C.; Klein, S.L. Considering How Biological Sex Impacts Immune Responses and COVID-19 Outcomes. Nat. Rev. Immunol. 2020, 20, 442–447. [Google Scholar] [CrossRef]
- Brodin, P. Immune Determinants of COVID-19 Disease Presentation and Severity. Nat. Med. 2021, 27, 28–33. [Google Scholar] [CrossRef]
- Jin, J.-M.; Bai, P.; He, W.; Wu, F.; Liu, X.-F.; Han, D.-M.; Liu, S.; Yang, J.-K. Gender Differences in Patients with COVID-19: Focus on Severity and Mortality. Front. Public Health 2020, 8, 152. [Google Scholar] [CrossRef]
- Peckham, H.; de Gruijter, N.M.; Raine, C.; Radziszewska, A.; Ciurtin, C.; Wedderburn, L.R.; Rosser, E.C.; Webb, K.; Deakin, C.T. Male Sex Identified by Global COVID-19 Meta-Analysis as a Risk Factor for Death and ITU Admission. Nat. Commun. 2020, 11, 6317. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chmielewski, N.N.; Limoli, C.L. Sex Differences in Taxane Toxicities. Cancers 2022, 14, 3325. https://doi.org/10.3390/cancers14143325
Chmielewski NN, Limoli CL. Sex Differences in Taxane Toxicities. Cancers. 2022; 14(14):3325. https://doi.org/10.3390/cancers14143325
Chicago/Turabian StyleChmielewski, Nicole N., and Charles L. Limoli. 2022. "Sex Differences in Taxane Toxicities" Cancers 14, no. 14: 3325. https://doi.org/10.3390/cancers14143325






